Obsessive-Compulsive Disorders and Functional Urinary Disorders: A Fortuitous Association?
Abstract
1. Introduction
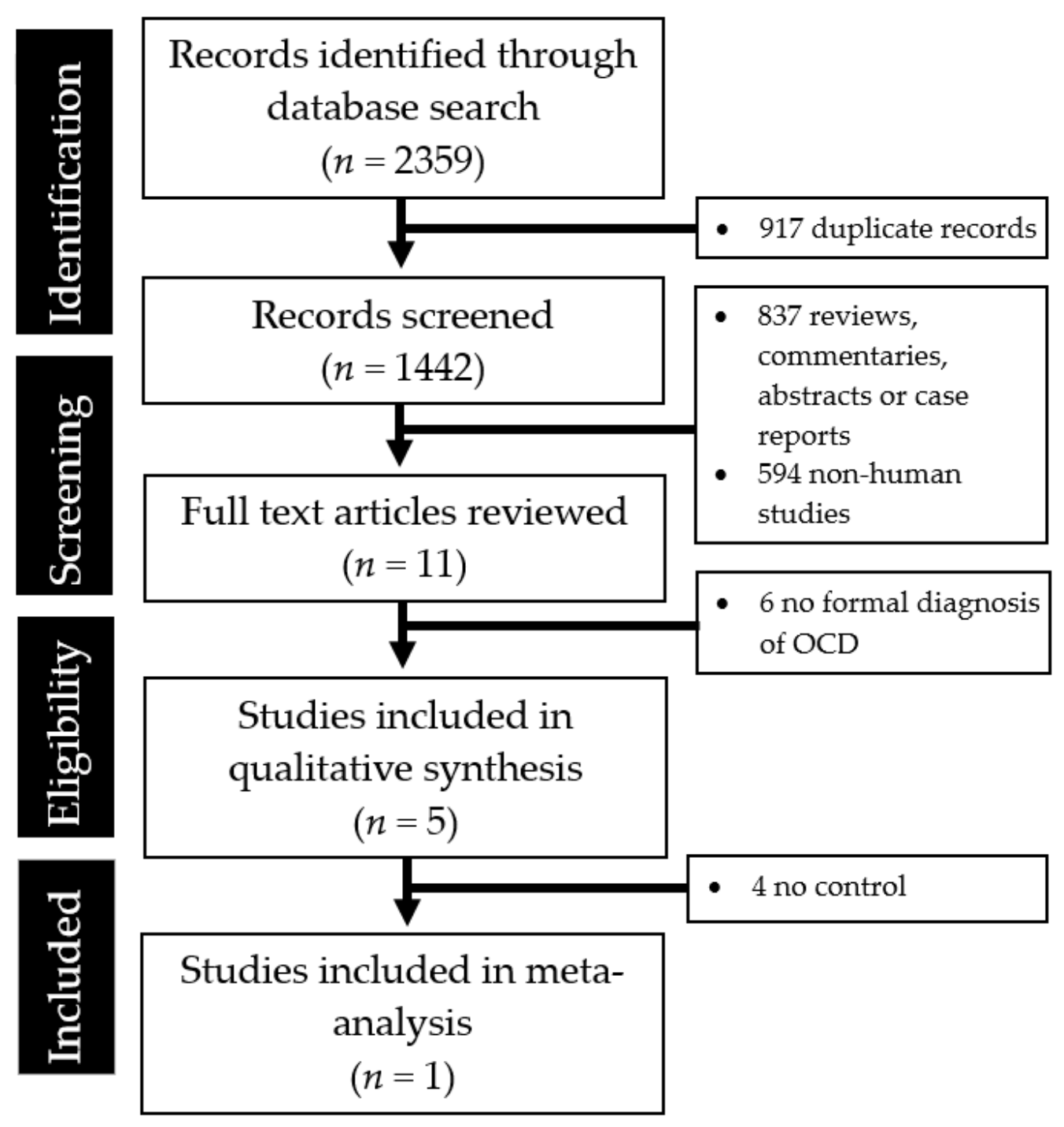
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Elenskaia, K.; Haidvogel, K.; Heidinger, C.; Doerfler, D.; Umek, W.; Hanzal, E. The greatest taboo: Urinary incontinence as a source of shame and embarrassment. Wien. Klin. Wochenschr. 2011, 123, 607–610. [Google Scholar] [CrossRef]
- Carvalhais, A.; Araújo, J.; Jorge, R.N.; Bø, K. Urinary incontinence and disordered eating in female elite athletes. J. Sci. Med. Sport 2019, 22, 140–144. [Google Scholar] [CrossRef]
- Vasudev, K.; Gupta, A.K. Incontinence and mood disorder: Is there an association? Case Rep. 2010, 2010, bcr0720092118. [Google Scholar] [CrossRef]
- Leue, C.; Kruimel, J.; Vrijens, D.; Masclee, A.; Van Os, J.; Van Koeveringe, G. Functional urological disorders: A sensitized defence response in the bladder–gut–brain axis. Nat. Rev. Urol. 2017, 14, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Von Gontard, A.; Hollmann, E. Comorbidity of functional urinary incontinence and encopresis: Somatic and behavioral associations. J. Urol. 2004, 171, 2644–2647. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.K.; Huang, K.H.; Chen, S.C.; Chang, H.C.; Yang, H.J.; Guo, Y.J. Correlation between clinical manifestations of nocturnal enuresis and attentional performance in children with attention deficit hyperactivity disorder (ADHD). J. Formos. Med. Assoc. 2013, 112, 41–47. [Google Scholar] [CrossRef]
- Gulisano, M.; Domini, C.; Capelli, M.; Pellico, A.; Rizzo, R. Importance of neuropsychiatric evaluation in children with primary monosymptomatic enuresis. J. Pediatric Urol. 2017, 13, 36.e1–36.e6. [Google Scholar] [CrossRef]
- Jiwanmall, S.A.; Kattula, D. Obsessive-Compulsive Disorder Presenting with Compulsions to Urinate Frequently. Indian J. Psychol. Med. 2016, 38, 364–365. [Google Scholar] [CrossRef]
- Rahmat, R. Urinary frequency: Going beyond the tract. Malays Fam. Physician 2020, 15, 58–60. [Google Scholar] [PubMed]
- Mowrer, O.H.; Mowrer, W.M. Enuresis—A method for its study and treatment. Am. J. Orthopsychiatry 1938, 8, 436–459. [Google Scholar] [CrossRef]
- Seki, M.; Zha, X.M.; Inamura, S.; Taga, M.; Matsuta, Y.; Aoki, Y.; Ito, H.; Yokoyama, O. Role of corticotropin-releasing factor on bladder function in rats with psychological stress. Sci. Rep. 2019, 9, 9828. [Google Scholar] [CrossRef]
- Ikeda, H.; Oyake, C.; Oonuki, Y.; Fuyama, M.; Watanabe, T.; Kyoda, T.; Tamura, S. Complete resolution of urinary incontinence with treatment improved the health-related quality of life of children with functional daytime urinary incontinence: A prospective study. Health Qual. Life Outcomes 2020, 18, 14. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Higgins, J.P. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Ahn, K.S.; Hong, H.P.; Kweon, H.J.; Ahn, A.L.; Oh, E.J.; Choi, J.K.; Cho, D.Y. Correlation between overactive bladder syndrome and obsessive compulsive disorder in women. Korean J. Fam. Med. 2016, 37, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Arlen, A.M.; Dewhurst, L.L.; Kirsch, S.S.; Dingle, A.D.; Scherz, H.C.; Kirsch, A.J. Phantom urinary incontinence in children with bladder-bowel dysfunction. Urology 2014, 84, 685–688. [Google Scholar] [CrossRef]
- Bernstein, G.A.; Victor, A.M.; Pipal, A.J.; Williams, K.A. Comparison of clinical characteristics of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections and childhood obsessive-compulsive disorder. J. Child Adolesc. Psychopharmacol. 2010, 20, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Jaspers-Fayer, F.; Han SH, J.; Chan, E.; McKenney, K.; Simpson, A.; Boyle, A.; Stewart, S.E. Prevalence of acute-onset subtypes in pediatric obsessive-compulsive disorder. J. Child Adolesc. Psychopharmacol. 2017, 27, 332–341. [Google Scholar] [CrossRef]
- Murphy, M.L.; Pichichero, M.E. Prospective identification and treatment of children with pediatric autoimmune neuropsychiatric disorder associated with group A streptococcal infection (PANDAS). Arch. Pediatrics Adolesc. Med. 2002, 156, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Holroyd-Leduc, J.M.; Tannenbaum, C.; Thorpe, K.E.; Straus, S.E. What type of urinary incontinence does this woman have? JAMA 2008, 299, 1446–1456. [Google Scholar] [CrossRef] [PubMed]
- Oades, R.D.; Daniels, R. Subclinical polydipsia and polyuria in young patients with schizophrenia or obsessive-compulsive disorder vs normal controls. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 1999, 23, 1329–1344. [Google Scholar] [CrossRef]
- Epstein, S.; Jenike, M.A. Disabling urinary obsessions: An uncommon variant of obsessive-compulsive disorder. Psychosomatics 1990, 31, 450–452. [Google Scholar] [CrossRef]
- Del Casale, A.; Sorice, S.; Padovano, A.; Simmaco, M.; Ferracuti, S.; Lamis, D.A.; Rapinesi, C.; Sani, G.; Girardi, P.; Kotzalidis, G.D.; et al. Psychopharmacological treatment of obsessive-compulsive disorder (OCD). Curr. Neuropharmacol. 2019, 17, 710–736. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, F.T.; Gilbert, A.R. Sertraline-Induced enuresis in a prepubertal child resolves after switching to fluoxetine. J. Child Adolesc. Psychopharmacol. 2010, 20, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Barnes, T.R.; Drake, M.J.; Paton, C. Nocturnal enuresis with antipsychotic medication. Br. J. Psychiatry 2012, 200, 7–9. [Google Scholar] [CrossRef]
- Bockaert, J.; Claeysen, S.; Compan, V.; Dumuis, A. 5-HT4 receptors: History, molecular pharmacology and brain functions. Neuropharmacology 2008, 55, 922–931. [Google Scholar] [CrossRef]
- Messori, E.; Rizzi, C.A.; Candura, S.M.; Lucchelli, A.; Balestra, B.; Tonini, M. 5-Hydroxytryptamine receptors that facilitate excitatory neuromuscular transmission in the guinea-pig isolated detrusor muscle. Br. J. Pharmacol. 1995, 115, 677–683. [Google Scholar] [CrossRef]
- Movig, K.L.L.; Leufkens, H.G.M.; Belitser, S.V.; Lenderink, A.W.; Egberts, A.C.G. Selective serotonin reuptake inhibitor-induced urinary incontinence. Pharmacoepidemiol. Drug Saf. 2002, 11, 271–279. [Google Scholar] [CrossRef]
- Torre, D.L.; Isgrò, S.; Muscatello, M.R.A.; Magno, C.; Melloni, D.; Meduri, M. Urinary incontinence in schizophrenic patients treated with atypical antipsychotics: Urodynamic findings and therapeutic perspectives. Int. J. Psychiatry Clin. Pract. 2005, 9, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Hermesh, H.; Aizenberg, D.; Weizman, A.; Lapidot, M.; Munitz, H.; Rivera, G.C.; Dion, P. Clomipramine-induced urinary dysfunction in an obsessive-compulsive adolescent. Drug Intell. Clin. Pharm. 1987, 21, 877–879. [Google Scholar] [CrossRef]
- Verhamme, K.M.; Sturkenboom, M.C.; Stricker, B.H.C.; Bosch, R. Drug-induced urinary retention. Drug Saf. 2008, 31, 373–388. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Country of Origin | Study Design and Study Population | Key Findings |
|---|---|---|---|
| Ahn et al., 2016 [15] | Korea | Case-control; n = 114 (57 women with OAB and 57 healthy female controls); Average age 50.65 ± 14.72 years |
|
| Arlen et al., 2014 [16] | United States | Retrospective chart review; n = 20 (2 boys and 18 girls); Average age 6.9 (range: 4 to 12 years) |
|
| Bernstein et al., 2010 [17] | United States | Case-control; n = 39 (24 boys and 15 girls); Average age 10.66 years |
|
| Jaspers-Fayer et al., 2017 [18] | Canada | Prospective cohort; n = 136 (54% males); Age range 6 to 19 years |
|
| Murphy & Pichichero, 2002 [19] | United States | Prospective case identification and followup; n = 12 (7 boys and 5 girls); Average age 5 years (range: 5 years 4 months to 10 years 11 months) |
|
| Study | Confounding | Selection | Measurement of Intervention | Missing Data | Measurement of Outcomes | Reported Result | Overall |
|---|---|---|---|---|---|---|---|
| Ahn et al., 2016 [15] | Serious | Serious | N/A | ? | Moderate | Moderate | Serious |
| Arlen et al., 2014 [16] | Serious | Serious | N/A | ? | Serious | Serious | Serious |
| Bernstein et al., 2010 [17] | Serious | Serious | N/A | ? | Moderate | Moderate | Serious |
| Jaspers-Fayer et al., 2017 [18] | Moderate | Serious | N/A | ? | Moderate | Moderate | Serious |
| Murphy & Pichichero, 2002 [19] | Serious | Serious | N/A | Moderate | Serious | Moderate | Serious |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ng, Q.X.; Lim, Y.L.; Loke, W.; Yeo, W.S.; Chee, K.T. Obsessive-Compulsive Disorders and Functional Urinary Disorders: A Fortuitous Association? Behav. Sci. 2021, 11, 89. https://doi.org/10.3390/bs11060089
Ng QX, Lim YL, Loke W, Yeo WS, Chee KT. Obsessive-Compulsive Disorders and Functional Urinary Disorders: A Fortuitous Association? Behavioral Sciences. 2021; 11(6):89. https://doi.org/10.3390/bs11060089
Chicago/Turabian StyleNg, Qin Xiang, Yu Liang Lim, Wayren Loke, Wee Song Yeo, and Kuan Tsee Chee. 2021. "Obsessive-Compulsive Disorders and Functional Urinary Disorders: A Fortuitous Association?" Behavioral Sciences 11, no. 6: 89. https://doi.org/10.3390/bs11060089
APA StyleNg, Q. X., Lim, Y. L., Loke, W., Yeo, W. S., & Chee, K. T. (2021). Obsessive-Compulsive Disorders and Functional Urinary Disorders: A Fortuitous Association? Behavioral Sciences, 11(6), 89. https://doi.org/10.3390/bs11060089








