Current State of the Neurotrophin-Based Pharmaceutics in the Treatment of Neurodegenerative Diseases and Neuroinflammation
Abstract
1. Introduction
2. Classification of Nerve Growth Factors and Their Receptors
- (1)
- NGF, BDNF, NT-3, NT-4;
- (2)
- Glial cell line-derived neurotrophic factor GDNF;
- (3)
- Ciliary neurotrophic factor CNTF
- (1)
- Neurotrophins (NTs);
- (2)
- GDNFs and GDNF family ligands (GFLs);
- (3)
- Neuropoietic cytokines, also known as the interleukin-6 family (IL-6).
- (1)
- NGF, BDNF, NT-3, and NT-4;
- (2)
- Neurturin (NRTN), artemin (ARTN), and persephin (PSPN);
- (3)
- Cytokines including IL-6, IL-11, IL-27, leukemia inhibitor factor (LIF), ciliary neurotrophic factor (CNTF), cardiotrophin 1 (CT-1), neuropoietin, cardiotrophin-like cytokine (CLC), also known as novel neurotrophin 1 (NNT1), and meteorin;
- (4)
- CDNF/MANF.
- -
- High-affinity nerve growth factor receptor TrkA (NTRK1) for NGF;
- -
- TrkB (NTRK2) for BDNF and NT-4;
- -
- TrkC (NTRK3) for NT-3;
- -
- P75NTR (NGFR, LNGFR) for NGF, BDNF, NT-3, and NT-4/5;
- -
- CNTFRα, IL-6Rα (CD126), LIFR, p-GP130 for CNTF [13];
- -
- GFRα-RET complex for GDNF: GFRα1 for GDNF, GFRα2 for NRTN, GFRα3 for ARTN and GFRα4 for PSPN;
- -
- GRP78-NBD (nucleotide-binding domain of GRP78) for both CDNF and MANF
- -
- IRE1α (inositol-requiring enzyme 1) for both CDNF and MANF [14].
3. NGF
3.1. NFG Agonists
- Promote tumor progression (e.g., NGF upregulates p75NTR in triple-negative breast cancer);
- Induce chemotherapy resistance in triple-negative breast cancers by inhibiting apoptosis;
- Potentially trigger carcinogenesis [33].
3.2. NGF Antagonists
4. BDNF
4.1. BDNF Agonists
4.2. BDNF Antagonists
5. NT-3
5.1. NT-3 Agonists
5.2. NT-3 Antagonists
6. GDNF
6.1. GDNF Agonists
6.2. GDNF Antagonists
7. CNTF
7.1. CNTF Agonists
7.2. CNTF Antagonists
8. Limitations and Future Prospects for Neurotrophin-Based Pharmaceutics
8.1. Limitations
8.1.1. Selectivity
8.1.2. Resistance
P-Glycoprotein–Mediated Resistance
Immune-Mediated Resistance
Resistance Mutations
8.1.3. Delivery
8.1.4. Disease-Stage-Related Limitations
8.2. Future Prospects
- High selectivity. It is advisable to prioritize the development of highly selective agonists and antagonists of neurotrophins capable of binding exclusively to a specific receptor subtype.
- Advanced delivery systems. To enhance the blood–brain barrier penetration, the development of novel delivery complexes, such as mesenchymal stem cell-derived extracellular vesicles [205], is recommended. For small molecules, the focus should be on creating new drug formulations such as nanocarriers or liposomes.
- Optimized administration and technique. Optimizing and standardizing the administration routes, surgical techniques, and injection procedures are recommended, integrating imaging guidance to simplify and reduce the cost of these interventions. For gene therapy, the use of new vectors with consideration for AAV serotype tropism is advised [206,207].
- Optimal dosing. The dosage of neurotrophins is a highly debated topic. Regarding BDNF, the data are controversial: some studies indicate that overdosing can cause pro-epileptogenic effects in animals and increases neuronal excitability [208], while others show that the overexpression of BDNF leads to the protection of brain cells from epileptic seizures [209,210].
- Personalized pharmacogenomics. Prior to clinical trials, creating the personalized pharmacogenomic reports for enrolled patients is recommended. This involves assessing the polymorphisms in neurotrophin and receptor genes, as well as in the detoxification system (e.g., P-gp, cytochrome P450 isoforms, GST isoforms), for instance, the Val66Met polymorphism in BDNF can negate synaptic plasticity and treatment outcomes [211].
- Capability to distinguish pro- and mature forms of NTs. Meticulous distinction between the pro- and mature forms of neurotrophins, such as pro-BDNF and mature BDNF, and the evaluation of their respective levels, are recommended [212].
- Assessing the disease status with respect to neuroinflammation. It is recommended to account for the extent of neuroinflammation using methods such as TSPO PET imaging, measuring sTREM2/GFAP (soluble triggering receptors expressed on myeloid cells-2/glial fibrillary acid protein) in CSF [213], plasma/CSF neurofilament light chain (NfL) level, and cytokine profiling. Monitoring the dynamics of these pharmacodynamic markers is crucial for assessing the direct therapeutic effect.
- Transition to plasma biomarkers. A shift from reliance on CSF markers to incorporating the validated plasma biomarkers is needed. Systemic inflammation markers, such as C-reactive protein, should consistently be included in the analysis.
- Account for comorbidities. Considering comorbid conditions, such as insulin resistance, is essential. It is also important that no other disease-modifying therapies be administered for at least two months prior to the start of the study period.
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Samaddar, S.; Redhwan, M.A.M.; Eraiah, M.M.; Koneri, R. Neurotrophins in Peripheral Neuropathy: Exploring Pathophysiological Mechanisms and Emerging Therapeutic Opportunities. CNS Neurol. Disord. Drug Targets 2025, 24, 91–101. [Google Scholar] [CrossRef]
- Lei, M.; Liu, Q.; Nie, J.; Huang, R.; Mei, Y.; Pan, D.; Chen, Y.; Liu, W. Impact and Mechanisms of Action of BDNF on Neurological Disorders, Cancer, and Cardiovascular Diseases. CNS Neurosci. Ther. 2024, 30, e70138. [Google Scholar] [CrossRef] [PubMed]
- Numakawa, T.; Kajihara, R. The Role of Brain-Derived Neurotrophic Factor as an Essential Mediator in Neuronal Functions and the Therapeutic Potential of Its Mimetics for Neuroprotection in Neurologic and Psychiatric Disorders. Molecules 2025, 30, 848. [Google Scholar] [CrossRef]
- Alqahtani, S.M.; Al-Kuraishy, H.M.; Al-Gareeb, A.I.; Albuhadily, A.K.; Alexiou, A.; Papadakis, M.; Hemeda, L.R.; Faheem, S.A.; Batiha, G.E.-S. Unlocking Alzheimer’s Disease: The Role of BDNF Signaling in Neuropathology and Treatment. Neuromol. Med. 2025, 27, 36. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.L.; Zhang, L.; Zhu, Y.; Bao, T.Y.; Zhu, Y.T.; Chen, Y.T.; Pang, H.Q. Exploring the Cellular and Molecular Basis of Nerve Growth Factor in Cerebral Ischemia Recovery. Neuroscience 2025, 566, 190–197. [Google Scholar] [CrossRef]
- Zhang, T.; Si, H.; Liao, J.; Ma, R. Association of Plasma BDNF and MMP-9 Levels with Mild Cognitive Impairment: A Matched Case-Control Study. Sci. Rep. 2024, 14, 30911. [Google Scholar] [CrossRef]
- Gluck, L.; Gerstein, B.; Kaunzner, U.W. Repair Mechanisms of the Central Nervous System: From Axon Sprouting to Remyelination. Neurotherapeutics 2025, 22, e00583. [Google Scholar] [CrossRef] [PubMed]
- Aloe, L. Rita Levi-Montalcini and the Discovery of NGF, the First Nerve Cell Growth Factor. Arch. Ital. Biol. 2011, 149, 175–181. [Google Scholar] [CrossRef]
- Götz, R.; Köster, R.; Winkler, C.; Raulf, F.; Lottspeich, F.; Schartl, M.; Thoenen, H. Neurotrophin-6 Is a New Member of the Nerve Growth Factor Family. Nature 1994, 372, 266–269. [Google Scholar] [CrossRef]
- Castilla-Cortázar, I.; Iturrieta, I.; García-Magariño, M.; Puche, J.E.; Martín-Estal, I.; Aguirre, G.A.; Femat-Roldan, G.; Cantu-Martinez, L.; Muñoz, Ú. Neurotrophic Factors and Their Receptors Are Altered by the Mere Partial IGF-1 Deficiency. Neuroscience 2019, 404, 445–458. [Google Scholar] [CrossRef]
- Sobańska, M.; Sobański, D.; Staszkiewicz, R.; Gogol, P.; Strojny, D.; Pawłaszek, T.; Dammerman, W.; Grabarek, B.O. Modulation of Neurturin Expression by Lumbosacral Spinal Stenosis, Lifestyle Factors, and Glycemic Dysregulation. Biomedicines 2025, 13, 1102. [Google Scholar] [CrossRef] [PubMed]
- Sims, S.K.; Wilken-Resman, B.; Smith, C.J.; Mitchell, A.; McGonegal, L.; Sims-Robinson, C. Brain-Derived Neurotrophic Factor and Nerve Growth Factor Therapeutics for Brain Injury: The Current Translational Challenges in Preclinical and Clinical Research. Neural Plast. 2022, 2022, 3889300. [Google Scholar] [CrossRef]
- Yong, J.; Groeger, S.; von Bremen, J.; Ruf, S. Ciliary Neurotrophic Factor (CNTF) and Its Receptors Signal Regulate Cementoblasts Apoptosis through a Mechanism of ERK1/2 and Caspases Signaling. Int. J. Mol. Sci. 2022, 23, 8335. [Google Scholar] [CrossRef]
- Graewert, M.A.; Volkova, M.; Jonasson, K.; Määttä, J.A.E.; Gräwert, T.; Mamidi, S.; Kulesskaya, N.; Evenäs, J.; Johnsson, R.E.; Svergun, D.; et al. Structural Basis of CDNF Interaction with the UPR Regulator GRP78. Nat. Commun. 2024, 15, 8175. [Google Scholar] [CrossRef]
- Chaldakov, G.N.; Aloe, L.; Yanev, S.G.; Fiore, M.; Tonchev, A.B.; Vinciguerra, M.; Evtimov, N.T.; Ghenev, P.; Dikranian, K. Trackins (Trk-Targeting Drugs): A Novel Therapy for Different Diseases. Pharmaceuticals 2024, 17, 961. [Google Scholar] [CrossRef]
- Koya, P.C.; Kolla, S.C.; Madala, V.; Sayana, S.B. Potential of Nerve Growth Factor (NGF)- and Brain-Derived Neurotrophic Factor (BDNF)-Targeted Gene Therapy for Alzheimer’s Disease: A Narrative Review. Cureus 2025, 17, e85814. [Google Scholar] [CrossRef]
- Zeliadt, N. Rita Levi-Montalcini: NGF, the Prototypical Growth Factor. Proc. Natl. Acad. Sci. USA 2013, 110, 4873–4876. [Google Scholar] [CrossRef] [PubMed]
- Bruno, F.; Abondio, P.; Montesanto, A.; Luiselli, D.; Bruni, A.C.; Maletta, R. The Nerve Growth Factor Receptor (NGFR/p75NTR): A Major Player in Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 3200. [Google Scholar] [CrossRef]
- Hirose, M.; Kuroda, Y.; Murata, E. NGF/TrkA Signaling as a Therapeutic Target for Pain. Pain Pract. 2016, 16, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Powell, R.; Woolf, C.J. Mechanism-Based Nonopioid Analgesic Targets. J. Clin. Investig. 2025, 135, e191346. [Google Scholar] [CrossRef]
- Gavioli, E.; Mantelli, F.; Cesta, M.C.; Sacchetti, M.; Allegretti, M. The History of Nerve Growth Factor: From Molecule to Drug. Biomolecules 2024, 14, 635. [Google Scholar] [CrossRef]
- Conroy, J.N.; Coulson, E.J. High-Affinity TrkA and p75 Neurotrophin Receptor Complexes: A Twisted Affair. J. Biol. Chem. 2022, 298, 101568. [Google Scholar] [CrossRef]
- El Baassiri, M.G.; Dosh, L.; Haidar, H.; Gerges, A.; Baassiri, S.; Leone, A.; Rappa, F.; Jurjus, A. Nerve Growth Factor and Burn Wound Healing: Update of Molecular Interactions with Skin Cells. Burns 2023, 49, 989–1002. [Google Scholar] [CrossRef]
- Lazarovici, P.; Gazit, A.; Staniszewska, I.; Marcinkiewicz, C.; Lelkes, P.I. Nerve Growth Factor (NGF) Promotes Angiogenesis in the Quail Chorioallantoic Membrane. Endothelium 2006, 13, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Rocco, M.L.; Soligo, M.; Manni, L.; Aloe, L. Nerve Growth Factor: Early Studies and Recent Clinical Trials. Curr. Neuropharmacol. 2018, 16, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000064300-NGFR/tissue (accessed on 15 December 2025).
- Adams, B.S.; Patel, A.R. Cenegermin. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Cattaneo, A.; Capsoni, S. Painless Nerve Growth Factor: A TrkA Biased Agonist Mediating a Broad Neuroprotection via Its Actions on Microglia Cells. Pharmacol. Res. 2019, 139, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Napoli, D.; Orsini, N.; Salamone, G.; Calvello, M.A.; Capsoni, S.; Cattaneo, A.; Strettoi, E. Human NGF “Painless” Ocular Delivery for Retinitis Pigmentosa: An In Vivo Study. eNeuro 2024, 11, 9. [Google Scholar] [CrossRef]
- Vacher, M.; Porter, T.; Villemagne, V.L.; Milicic, L.; Peretti, M.; Fowler, C.; Martins, R.; Rainey-Smith, S.; Ames, D.; Masters, C.L.; et al. Validation of a Priori Candidate Alzheimer’s Disease SNPs with Brain Amyloid-Beta Deposition. Sci. Rep. 2019, 9, 17069. [Google Scholar] [CrossRef]
- Kuo, S.C.; Lin, C.L.; Tsou, C.C.; Yeh, Y.W.; Yang, B.Z.; Chen, C.Y.; Huang, C.Y.; Huang, S.Y. Nerve Growth Factor Gene Polymorphisms May Be Associated with Heroin Dependence in Women but Do Not Mediate Specific Personality Traits. Eur. Arch. Psychiatry Clin. Neurosci. 2025, 275, 487–498. [Google Scholar] [CrossRef]
- Stepanyan, A.; Zakharyan, R.; Simonyan, A.; Tsakanova, G.; Arakelyan, A. Involvement of Polymorphisms of the Nerve Growth Factor and Its Receptor Encoding Genes in the Etiopathogenesis of Ischemic Stroke. BMC Med. Genet. 2018, 19, 33. [Google Scholar] [CrossRef]
- Chakravarthy, R.; Mnich, K.; Gorman, A.M. Nerve Growth Factor (NGF)-Mediated Regulation of p75(NTR) Expression Contributes to Chemotherapeutic Resistance in Triple Negative Breast Cancer Cells. Biochem. Biophys. Res. Commun. 2016, 478, 1541–1547. [Google Scholar] [CrossRef]
- Trentini, F.; Agnetti, V.; Manini, M.; Giovannetti, E.; Garajová, I. NGF-Mediated Crosstalk: Unraveling the Influence of Metabolic Deregulation on the Interplay between Neural and Pancreatic Cancer Cells and Its Impact on Patient Outcomes. Front. Pharmacol. 2024, 15, 1499414. [Google Scholar] [CrossRef]
- Wang, H.; Zheng, Q.; Lu, Z.; Lu, Z.; Wang, L.; Ding, L.; Xia, L.; Zhang, H.; Wang, M.; Chen, Y.; et al. Role of the Nervous System in Cancers: A Review. Cell Death Discov. 2021, 7, 76. [Google Scholar] [CrossRef]
- Schmitd, L.B.; Perez-Pacheco, C.; D’Silva, N.J. Neural Influence on Cancer Invasion and Metastasis. In Cancer Neuroscience; Amit, M., Scheff, N.N., Eds.; Springer: Cham, Switzerland, 2023; pp. 63–84. [Google Scholar] [CrossRef]
- Dwivedi, S.; Krishnan, A. Neural Invasion: A Scenic Trail for the Nervous Tumor and Hidden Therapeutic Opportunity. Am. J. Cancer Res. 2020, 10, 2258–2270. [Google Scholar]
- Capossela, L.; Gatto, A.; Ferretti, S.; Di Sarno, L.; Graglia, B.; Massese, M.; Soligo, M.; Chiaretti, A. Multifaceted Roles of Nerve Growth Factor: A Comprehensive Review with a Special Insight into Pediatric Perspectives. Biology 2024, 13, 546. [Google Scholar] [CrossRef]
- Barker, P.A.; Mantyh, P.; Arendt-Nielsen, L.; Viktrup, L.; Tive, L. Nerve Growth Factor Signaling and Its Contribution to Pain. J. Pain Res. 2020, 13, 1223–1241. [Google Scholar] [CrossRef] [PubMed]
- Sheffield, K.S.; Kennedy, A.E.; Scott, J.A.; Ross, G.M. Characterizing Nerve Growth Factor-p75(NTR) Interactions and Small Molecule Inhibition Using Surface Plasmon Resonance Spectroscopy. Anal. Biochem. 2016, 493, 21–26. [Google Scholar] [CrossRef]
- Lin, J.; Jia, S.; Zhang, W.; Nian, M.; Liu, P.; Yang, L.; Zuo, J.; Li, W.; Zeng, H.; Zhang, X. Recent Advances in Small Molecule Inhibitors for the Treatment of Osteoarthritis. J. Clin. Med. 2023, 12, 1986. [Google Scholar] [CrossRef] [PubMed]
- DiMartino, S.J.; Gao, H.; Eng, S.; Valenzuela, G.; Fuerst, T.; Emeremni, C.; Ho, T.; Hassan, H.E.; Turner, K.C.; Davis, J.D.; et al. Efficacy and Safety of Fasinumab in an NSAID-Controlled Study in Patients with Pain Due to Osteoarthritis of the Knee or Hip. BMC Musculoskelet. Disord. 2025, 26, 192. [Google Scholar] [CrossRef]
- Gao, Y.; Hu, Z.; Huang, Y.; Liu, W.; Ren, C. Efficacy and Safety of Anti-Nerve Growth Factor Antibody Therapy for Hip and Knee Osteoarthritis: A Meta-Analysis. Orthop. J. Sports Med. 2022, 10, 23259671221088590. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Zhou, J.; Long, Z.; Li, Y.; Sun, J.; Luo, Y.; Wang, W. Targeting Nerve Growth Factor, a New Option for Treatment of Osteoarthritis: A Network Meta-Analysis of Comparative Efficacy and Safety with Traditional Drugs. Aging 2020, 13, 1051–1070. [Google Scholar] [CrossRef]
- Jin, D.; Yang, H.; Chen, Z.; Hong, Y.; Ma, H.; Xu, Z.; Cao, B.; Fei, F.; Zhang, Y.; Wu, W.; et al. Effect of the Novel Anti-NGF Monoclonal Antibody DS002 on the Metabolomics of Pain Mediators, Cartilage and Bone. Front. Pharmacol. 2024, 15, 1396790. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Cao, B.; Huang, L.; Yang, Y.; Geng, Y.; Xie, P.; Zhao, Y.; Lin, H.; Wang, K.; Wang, C.; et al. First-in-Human Study to Assess the Safety, Tolerability, Pharmacokinetics and Immunogenicity of DS002, an Anti-Nerve Growth Factor Monoclonal Antibody. Front. Pharmacol. 2022, 13, 1075309. [Google Scholar] [CrossRef]
- Conaghan, P.G.; Guermazi, A.; Katz, N.; Bihlet, A.R.; Rom, D.; Perkins, M.C.; Hughes, B.; Herholdt, C.; Bombelka, I.; Westbrook, S. A Randomised Controlled Phase II Trial of LEVI-04, a Novel Neurotrophin-3 Inhibitor, in Knee Osteoarthritis Demonstrates Substantially Improved Pain and Function without Deleterious Effects on Joint Structure. Rheumatology 2025, 64, keaf142.004. [Google Scholar] [CrossRef]
- Malik, S.C.; Sozmen, E.G.; Baeza-Raja, B.; Le Moan, N.; Akassoglou, K.; Schachtrup, C. In Vivo Functions of p75NTR: Challenges and Opportunities for an Emerging Therapeutic Target. Trends Pharmacol. Sci. 2021, 42, 772–788. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://clinicaltrials.gov/study/NCT05618782 (accessed on 25 December 2025).
- Drilon, A.; Laetsch, T.W.; Kummar, S.; DuBois, S.G.; Lassen, U.N.; Demetri, G.D.; Nathenson, M.; Doebele, R.C.; Farago, A.F.; Pappo, A.S.; et al. Efficacy of Larotrectinib in TRK Fusion-Positive Cancers in Adults and Children. N. Engl. J. Med. 2018, 378, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Romero Garavito, A.; Díaz Martínez, V.; Juárez Cortés, E.; Negrete Díaz, J.V.; Montilla Rodríguez, L.M. Impact of Physical Exercise on the Regulation of Brain-Derived Neurotrophic Factor in People with Neurodegenerative Diseases. Front. Neurol. 2025, 15, 1505879. [Google Scholar] [CrossRef]
- Russo, L.; Giacomelli, C.; Fortino, M.; Marzo, T.; Ferri, G.; Calvello, M.; Viegi, A.; Magrì, A.; Pratesi, A.; Pietropaolo, A.; et al. Neurotrophic Activity and Its Modulation by Zinc Ion of a Dimeric Peptide Mimicking the Brain-Derived Neurotrophic Factor N-Terminal Region. ACS Chem. Neurosci. 2022, 13, 3453–3463. [Google Scholar] [CrossRef]
- Fan, X.; Wang, H.; Lv, X.; Wang, Q.; Yu, B.; Li, X.; Li, L.; Zhang, Y.; Ma, N.; Lu, Q.; et al. The pCREB/BDNF Pathway in the Hippocampus Is Involved in the Therapeutic Effect of Selective 5-HT Reuptake Inhibitors in Adult Male Rats Exposed to Blast Traumatic Brain Injury. Brain Sci. 2025, 15, 236. [Google Scholar] [CrossRef]
- Schulte-Herbrüggen, O.; Nassenstein, C.; Lommatzsch, M.; Quarcoo, D.; Renz, H.; Braun, A. Tumor Necrosis Factor-Alpha and Interleukin-6 Regulate Secretion of Brain-Derived Neurotrophic Factor in Human Monocytes. J. Neuroimmunol. 2005, 160, 204–209. [Google Scholar] [CrossRef]
- Lima Giacobbo, B.; Doorduin, J.; Klein, H.C.; Dierckx, R.A.J.O.; Bromberg, E.; de Vries, E.F.J. Brain-Derived Neurotrophic Factor in Brain Disorders: Focus on Neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312. [Google Scholar] [CrossRef]
- McGonigal, A.; Becker, C.; Fath, J.; Hammam, K.; Baumstarck, K.; Fernandes, S.; Giusiano, B.; Dufau, S.; Rheims, S.; Maillard, L.; et al. BDNF as Potential Biomarker of Epilepsy Severity and Psychiatric Comorbidity: Pitfalls in the Clinical Population. Epilepsy Res. 2023, 195, 107200. [Google Scholar] [CrossRef] [PubMed]
- Jhaveri, D.J.; McGonigal, A.; Becker, C.; Benoliel, J.J.; Nandam, L.S.; Soncin, L.; Kotwas, I.; Bernard, C.; Bartolomei, F. Stress and Epilepsy: Towards Understanding of Neurobiological Mechanisms for Better Management. eNeuro 2023, 10, ENEURO.0200-23.2023. [Google Scholar] [CrossRef]
- Bhavsar, K.; Tripathi, M.; Banerjee, J.; Srivastava, A.; Nidhi; Pandey, S.; Vohora, D. Brain-Derived Neurotrophic Factor but Not Beta-Secretase 1, Vascular Endothelial Growth Factor, Glial Fibrillary Acidic Protein and Interleukin-1β Correlate with Cognitive Impairment in Adult Persons with Epilepsy: A Cross-Sectional Single-Center Study from India. Front. Neurol. 2025, 16, 1540915. [Google Scholar] [CrossRef]
- Sun, S.R.; Zhao, J.N.; Bi, P.W.; Zhang, H.Y.; Li, G.X.; Yan, J.Z.; Li, Y.F.; Yin, Y.Y.; Cheng, H. Pharmacologically Activating BDNF/TrkB Signaling Exerted Rapid-Acting Antidepressant-Like Effects through Improving Synaptic Plasticity and Neuroinflammation. Metab. Brain Dis. 2025, 40, 158. [Google Scholar] [CrossRef]
- Antonijevic, M.; Charou, D.; Davis, A.; Curel, T.; Valcarcel, M.; Ramos, I.; Villacé, P.; Claeysen, S.; Dallemagne, P.; Gravanis, A.; et al. Development of Pleiotropic TrkB and 5-HT4 Receptor Ligands as Neuroprotective Agents. Molecules 2024, 29, 515. [Google Scholar] [CrossRef]
- Jang, S.; Liu, X.; Yepes, M.; Shepherd, K.R.; Miller, G.W.; Liu, Y.; Wilson, W.D.; Xiao, G.; Blanchi, B.; Sun, Y.E.; et al. A Selective TrkB Agonist with Potent Neurotrophic Activities by 7,8-Dihydroxyflavone. Proc. Natl. Acad. Sci. USA 2010, 107, 2687–2692. [Google Scholar] [CrossRef]
- Emili, M.; Guidi, S.; Uguagliati, B.; Giacomini, A.; Bartesaghi, R.; Stagni, F. Treatment with the Flavonoid 7,8-Dihydroxyflavone: A Promising Strategy for a Constellation of Body and Brain Disorders. Crit. Rev. Food Sci. Nutr. 2022, 62, 13–50. [Google Scholar] [CrossRef]
- Xu, S.; Ma, X.; Xiao, Y.; Zhang, T.; Ma, C.; Ma, Z. 7,8-DHF Modulates Aggressive Behavior in Sebastes schlegelii: Phenotype-Dependent Responses in Aggression-Dimorphic Individuals. Animals 2025, 15, 1463. [Google Scholar] [CrossRef]
- Fletcher, J.L.; Dill, L.K.; Wood, R.J.; Wang, S.; Robertson, K.; Murray, S.S.; Zamani, A.; Semple, B.D. Acute Treatment with TrkB Agonist LM22A-4 Confers Neuroprotection and Preserves Myelin Integrity in a Mouse Model of Pediatric Traumatic Brain Injury. Exp. Neurol. 2021, 339, 113652. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Bellot-Saez, A.; Phillips, M.L.; Yang, T.; Longo, F.M.; Pozzo-Miller, L. A Small-Molecule TrkB Ligand Restores Hippocampal Synaptic Plasticity and Object Location Memory in Rett Syndrome Mice. Dis. Model. Mech. 2017, 10, 837–845. [Google Scholar] [CrossRef]
- Totoson, P.; Pedard, M.; Marie, C.; Demougeot, C. Activation of Endothelial TrkB Receptors Induces Relaxation of Resistance Arteries. Vasc. Pharmacol. 2018, 106, 46–53. [Google Scholar] [CrossRef]
- Yu, G.; Wang, W. Protective Effects of LM22A-4 on Injured Spinal Cord Nerves. Int. J. Clin. Exp. Pathol. 2015, 8, 6526–6532. [Google Scholar]
- Lee, J.H.; Lee, K.H.; Ryu, J.H.; Kim, M.J.; Kim, E.; Lee, S.Y.; Han, S.C.; Choi, B.T.; Shin, Y.I.; Shin, H.K. LM22A-4-Loaded Smart Mesoporous Balls Enhance Neuroprotection and Functional Recovery after Ischemic Stroke. Biomed. Pharmacother. 2025, 183, 117863. [Google Scholar] [CrossRef]
- Parrado Fernandez, C.; Juric, S.; Backlund, M.; Dahlström, M.; Madjid, N.; Lidell, V.; Rasti, A.; Sandin, J.; Nordvall, G.; Forsell, P. Neuroprotective and Disease-Modifying Effects of the Triazinetrione ACD856, a Positive Allosteric Modulator of Trk-Receptors for the Treatment of Cognitive Dysfunction in Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 11159. [Google Scholar] [CrossRef]
- Önnestam, K.; Nilsson, B.; Rother, M.; Rein-Hedin, E.; Bylund, J.; Anderer, P.; Kemethofer, M.; Halldin, M.M.; Sandin, J.; Segerdahl, M. Safety, Tolerability, Pharmacokinetics and Quantitative Electroencephalography Assessment of ACD856, a Novel Positive Allosteric Modulator of Trk-Receptors Following Multiple Doses in Healthy Subjects. J. Prev. Alzheimers Dis. 2023, 10, 778–789. [Google Scholar] [CrossRef]
- Nilsson, B.; Bylund, J.; Halldin, M.M.; Rother, M.; Rein-Hedin, E.; Önnestam, K.; Segerdahl, M. ACD856, a Novel Positive Allosteric Modulator of Trk Receptors, Single Ascending Doses in Healthy Subjects: Safety and Pharmacokinetics. Eur. J. Clin. Pharmacol. 2024, 80, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Fedotcheva, T.A.; Shimanovsky, N.L. Neurosteroids Progesterone and Dehydroepiandrosterone: Molecular Mechanisms of Action in Neuroprotection and Neuroinflammation. Pharmaceuticals 2025, 18, 945. [Google Scholar] [CrossRef]
- Pardossi, S.; Fagiolini, A.; Cuomo, A. Variations in BDNF and Their Role in the Neurotrophic Antidepressant Mechanisms of Ketamine and Esketamine: A Review. Int. J. Mol. Sci. 2024, 25, 13098. [Google Scholar] [CrossRef]
- Tang, S.; Luo, W.; Wu, S.; Yuan, M.; Wen, J.; Zhong, G.; Shen, L.; Jiang, W.; Cheng, C.; Wu, X.; et al. Hippocampus-Targeted BDNF Gene Therapy to Rescue Cognitive Impairments of Alzheimer’s Disease in Multiple Mouse Models. Genes Dis. 2025, 13, 101649. [Google Scholar] [CrossRef]
- Karsuntseva, E.K.; Fursa, G.A.; Sosnovtseva, A.O.; Voronova, A.D.; Chadin, A.V.; Semkina, A.S.; Stepanova, O.V.; Chekhonin, V.P. Application of a New Gene-Cell Construct Based on the Olfactory Mucosa Escheating Cells Transduced with an Adenoviral Vector Encoding Mature BDNF in the Therapy of Spinal Cord Cysts. Bull. Exp. Biol. Med. 2022, 172, 617–621. [Google Scholar] [CrossRef]
- Stepanova, O.V.; Fursa, G.A.; Andretsova, S.S.; Karsuntseva, E.K.; Shishkina, V.S.; Chadin, A.V.; Voronova, A.D.; Semkina, A.S.; Reshetov, I.V.; Chekhonin, V.P. Transduced Olfactory Mucosa Cells Expressing Nerve Growth Factor for the Therapy of Experimental Spinal Cord Cysts. Bull. Exp. Biol. Med. 2024, 177, 552–558. [Google Scholar] [CrossRef]
- Jahani, N.; Dehnou, V.V.; Eslami, R.; Gahreman, D. Effects of Aerobic Exercise on Memory Functions and Serum Levels of BDNF and TrkB in Young Adult and Older Adult Male Football Players. J. Exerc. Sci. Fit. 2025, 23, 197–202. [Google Scholar] [CrossRef]
- Ishaq, S.; Shah, I.A.; Lee, S.D.; Wu, B.T. Effects of Exercise Training on Nigrostriatal Neuroprotection in Parkinson’s Disease: A Systematic Review. Front. Neurosci. 2025, 18, 1464168. [Google Scholar] [CrossRef]
- Vulturar, R.; Chiş, A.; Hambrich, M.; Kelemen, B.; Ungureanu, L.; Miu, A.C. Allelic Distribution of BDNF Val66Met Polymorphism in Healthy Romanian Volunteers. Transl. Neurosci. 2016, 7, 31–34. [Google Scholar] [CrossRef]
- Cadwallader, C.J.; VandeBunte, A.M.; Fischer, D.L.; Chen, C.; Diaz, V.E.; Lee, S.Y.; Chan, B.; Lario-Lago, A.; Rojas, J.C.; Ramos, E.M.; et al. BDNF Val66Met Polymorphism Moderates Associations between Physical Activity and Neurocognitive Outcomes in Older Adults. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2025, 11, e70106. [Google Scholar] [CrossRef]
- Khan, N.; Smith, M.T. Neurotrophins and Neuropathic Pain: Role in Pathobiology. Molecules 2015, 20, 10657–10688. [Google Scholar] [CrossRef]
- Hernández-Echeagaray, E. Neurotrophin-3 Modulates Synaptic Transmission. Vitam. Horm. 2020, 113, 89–113. [Google Scholar] [CrossRef]
- Wan, T.; Zhang, F.S.; Qin, M.Y.; Jiang, H.R.; Zhang, M.; Qu, Y.; Wang, Y.L.; Zhang, P.X. Growth Factors: Bioactive Macromolecular Drugs for Peripheral Nerve Injury Treatment—Molecular Mechanisms and Delivery Platforms. Biomed. Pharmacother. 2024, 170, 116024. [Google Scholar] [CrossRef]
- El Ouaamari, Y.; Van den Bos, J.; Willekens, B.; Cools, N.; Wens, I. Neurotrophic Factors as Regenerative Therapy for Neurodegenerative Diseases: Current Status, Challenges and Future Perspectives. Int. J. Mol. Sci. 2023, 24, 3866. [Google Scholar] [CrossRef]
- Bové, M.; Monto, F.; Guillem-Llobat, P.; Ivorra, M.D.; Noguera, M.A.; Zambrano, A.; Sirerol-Piquer, M.S.; Requena, A.C.; García-Alonso, M.; Tejerina, T.; et al. NT3/TrkC Pathway Modulates the Expression of UCP-1 and Adipocyte Size in Human and Rodent Adipose Tissue. Front. Endocrinol. 2021, 12, 630097. [Google Scholar] [CrossRef]
- Xu, X.; Song, L.; Li, Y.; Guo, J.; Huang, S.; Du, S.; Li, W.; Cao, R.; Cui, S. Neurotrophin-3 Promotes Peripheral Nerve Regeneration by Maintaining a Repair State of Schwann Cells after Chronic Denervation via the TrkC/ERK/c-Jun Pathway. J. Transl. Med. 2023, 21, 733. [Google Scholar] [CrossRef]
- Sekimoto, M.; Tsuji, T.; Matsuzaki, J.; Chamoto, K.; Koda, T.; Nemoto, K.; Degawa, M.; Nishimura, S.-I.; Nishimura, T. Functional Expression of the TrkC Gene, Encoding a High Affinity Receptor for NT-3, in Antigen-Specific T Helper Type 2 (Th2) Cells. Immunol. Lett. 2003, 88, 221–226. [Google Scholar] [CrossRef]
- Tong, L.; Ozes, B.; Moss, K.; Myers, M.; Attia, Z.; Vetter, T.A.; Trapp, B.D.; Sahenk, Z. AAV1.NT3 Gene Therapy Mitigates the Severity of Autoimmune Encephalomyelitis in the Mouse Model for Multiple Sclerosis. Gene Ther. 2025, 32, 410–420. [Google Scholar] [CrossRef]
- Meissner, S.; Lopez, S.; Rees, S.; O’Carroll, S.; Barker, D.; Harland, B.; Raos, B.; Svirskis, D. Safe Subdural Administration and Retention of a Neurotrophin-3-Delivering Hydrogel in a Rat Model of Spinal Cord Injury. Sci. Rep. 2024, 14, 25424. [Google Scholar] [CrossRef]
- de Miranda, A.S.; de Barros, J.L.V.M.; Teixeira, A.L. Is Neurotrophin-3 (NT-3): A Potential Therapeutic Target for Depression and Anxiety? Expert Opin. Ther. Targets 2020, 24, 1225–1238. [Google Scholar] [CrossRef]
- Brahimi, F.; Nassour, H.; Galan, A.; Guruswamy, R.; Ortiz, C.; Nejatie, A.; Nedev, H.; Trempe, J.F.; Saragovi, H.U. Selective Inhibitors of the TrkC.T1 Receptor Reduce Retinal Inflammation and Delay Neuronal Death in a Model of Retinitis Pigmentosa. PNAS Nexus 2025, 4, pgaf020. [Google Scholar] [CrossRef]
- Dravid, A.; O’Carroll, S.J.; Svirskis, D. Neurotrophins and Their Role in Axonal Outgrowth Following Spinal Cord Injury. In Cellular, Molecular, Physiological, and Behavioral Aspects of Spinal Cord Injury; Elsevier: Amsterdam, The Netherlands, 2022; pp. 215–227. [Google Scholar] [CrossRef]
- Proenca, C.C.; Song, M.; Lee, F.S. Differential Effects of BDNF and Neurotrophin 4 (NT4) on Endocytic Sorting of TrkB Receptors. J. Neurochem. 2016, 138, 397–406. [Google Scholar] [CrossRef]
- NT-3 Levels and Function in Individuals with CMT. Available online: https://clinicaltrials.gov/study/NCT05011006 (accessed on 25 December 2025).
- Yalvac, M.E.; Arnold, W.D.; Braganza, C.; Chen, L.; Mendell, J.R.; Sahenk, Z. AAV1.NT-3 Gene Therapy Attenuates Spontaneous Autoimmune Peripheral Polyneuropathy. Gene Ther. 2016, 23, 95–102. [Google Scholar] [CrossRef]
- Tonyan, S.; Pospelova, M.; Krasnikova, V.; Fionik, O.; Alekseeva, T.; Samochernykh, K.; Ivanova, N.; Vavilova, T.; Vasilieva, E.; Makhanova, A.; et al. Neurotrophin-3 (NT-3) as a Potential Biomarker of the Peripheral Nervous System Damage Following Breast Cancer Treatment. Pathophysiology 2023, 30, 110–122. [Google Scholar] [CrossRef]
- Kolik, L.G.; Konstantinipolsky, M.A.; Nikolaev, S.V.; Logvinov, I.O.; Antipova, T.A.; Gudasheva, T.A. Low-Molecular Neurotrophin-3 Mimetics with Different Patterns of Postreceptor Signaling Activation Attenuate Differentially Morphine Withdrawal in Rats. Biochem. Moscow 2024, 89, 1961–1969. [Google Scholar] [CrossRef]
- Magrì, A.; Tomasello, B.; Naletova, I.; Tabbì, G.; Cairns, W.R.L.; Greco, V.; Sciuto, S.; La Mendola, D.; Rizzarelli, E. New BDNF and NT-3 Cyclic Mimetics Concur with Copper to Activate Trophic Signaling Pathways as Potential Molecular Entities to Protect Old Brains from Neurodegeneration. Biomolecules 2024, 14, 1104. [Google Scholar] [CrossRef] [PubMed]
- Kaya, Z.B.; Santiago-Padilla, V.; Lim, M.; Boschen, S.L.; Atilla, P.; McLean, P.J. Optimizing SH-SY5Y Cell Culture: Exploring the Beneficial Effects of an Alternative Media Supplement on Cell Proliferation and Viability. Sci. Rep. 2024, 14, 4775. [Google Scholar] [CrossRef]
- Albaugh, P.; Fan, Y.; Mi, Y.; Sun, F.; Adrian, F.; Li, N.; Jia, Y.; Sarkisova, Y.; Kreusch, A.; Hood, T.; et al. Discovery of GNF-5837, a Selective TRK Inhibitor with Efficacy in Rodent Cancer Tumor Models. ACS Med. Chem. Lett. 2012, 3, 140–145. [Google Scholar] [CrossRef]
- Wang, L.-H.; Besirli, C.G.; Johnson, E.M., Jr. Mixed-Lineage Kinases: A Target for the Prevention of Neurodegenera-tion. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 451–474. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, E.; Marconi, A.; Truzzi, F.; Dallaglio, K.; Petrachi, T.; Humbert, P.; Schnebert, S.; Perrier, E.; Dumas, M.; Pincelli, C. Role of Neurotrophins on Dermal Fibroblast Survival and Differentiation. J. Cell. Physiol. 2012, 227, 1017–1025. [Google Scholar] [CrossRef]
- Raychaudhuri, S.K.; Raychaudhuri, S.P. NGF and Its Receptor System: A New Dimension in the Pathogenesis of Psoriasis and Psoriatic Arthritis. Ann. N. Y. Acad. Sci. 2009, 1173, 470–477. [Google Scholar] [CrossRef]
- DrugBank Online. Available online: https://go.drugbank.com/drugs/DB14723 (accessed on 5 September 2025).
- Barker, R.A.; Björklund, A.; Gash, D.M.; Whone, A.; Van Laar, A.; Kordower, J.H.; Bankiewicz, K.; Kieburtz, K.; Saarma, M.; Booms, S.; et al. GDNF and Parkinson’s Disease: Where Next? A Summary from a Recent Workshop. J. Park. Dis. 2020, 10, 875–891. [Google Scholar] [CrossRef]
- Mendes-Oliveira, J.; Campos, F.L.; Ferreira, S.A.; Tomé, D.; Fonseca, C.P.; Baltazar, G. Endogenous GDNF Is Unable to Halt Dopaminergic Injury Triggered by Microglial Activation. Cells 2024, 13, 74. [Google Scholar] [CrossRef]
- Mahato, A.K.; Saarma, M. Biology of RET Receptor and Its Ligands: Focus on the Nervous System. Endocr. Relat. Cancer 2025, 32, e240174. [Google Scholar] [CrossRef] [PubMed]
- Shchelchkova, N.A.; Kokaya, A.A.; Bezhenar’, V.F.; Rozhdestvenskaya, O.V.; Mamedova, M.A.; Mishchenko, T.A.; Mitroshina, E.V.; Vedunova, M.V. The Role of Brain-Derived Neurotrophic Factor and Glial Cell Line-Derived Neurotrophic Factor in Chronic Fetal Oxygen Deprivation. Sovrem. Tehnol. Med. 2020, 12, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Grodzki, L.M.; Bartsch, U. Survival and Axonal Regeneration of Retinal Ganglion Cells in a Mouse Optic Nerve Crush Model After a Cell-Based Intravitreal Co-Administration of Ciliary Neurotrophic Factor and Glial Cell Line-Derived Neurotrophic Factor at Different Post-Lesion Time Points. Cells 2025, 14, 643. [Google Scholar] [CrossRef]
- UniProt Consortium. GDNF Protein. Available online: https://www.uniprot.org/uniprotkb/P26441/entry (accessed on 12 December 2025).
- Mätlik, K.; Garton, D.R.; Montaño-Rodríguez, A.R.; Olfat, S.; Eren, F.; Casserly, L.; Damdimopoulos, A.; Panhelainen, A.; Porokuokka, L.L.; Kopra, J.J.; et al. Elevated Endogenous GDNF Induces Altered Dopamine Signalling in Mice and Correlates with Clinical Severity in Schizophrenia. Mol. Psychiatry 2022, 27, 3247–3261. [Google Scholar] [CrossRef]
- Marshall, P. Finding an Optimal Level of GDNF Overexpression: Insights from Dopamine Cycling. Cell. Mol. Neurobiol. 2023, 43, 3179–3189. [Google Scholar] [CrossRef]
- Heiss, J.D.; Ray-Chaudhury, A.; Kleiner, D.E.; Ehrlich, D.J.; Scott, G.; Edwards, N.A.; Goldstein, D.S.; Hammoud, D.A.; Hadaczek, P.; Van Laar, V.S.; et al. Persistent GDNF Expression 45 Months after Putaminal Infusion of AAV2-GDNF in a Patient with Parkinson’s Disease. Mov. Disord. 2024, 39, 1412–1417. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. A Study of NBIb-1817 (VY-AADC) in Participants with Parkinson’s Disease. Available online: https://clinicaltrials.gov/study/NCT06285643 (accessed on 12 December 2025).
- Van Laar, A.D.; Christine, C.W.; Phielipp, N.; Larson, P.S.; Elder, J.B.; Merola, A.; San Sebastian, W.; Fiandaca, M.S.; Kells, A.P.; Wisniewski, M.E.; et al. Intraputaminal Delivery of Adeno-Associated Virus Serotype 2-Glial Cell Line-Derived Neurotrophic Factor in Mild or Moderate Parkinson’s Disease. Mov. Disord. 2025, 40, 1297–1306. [Google Scholar] [CrossRef]
- Baloh, R.H.; Johnson, J.P.; Avalos, P.; Allred, P.; Svendsen, S.; Gowing, G.; Roxas, K.; Wu, A.; Donahue, B.; Osborne, S.; et al. Transplantation of human neural progenitor cells secreting GDNF into the spinal cord of patients with ALS: A phase 1/2a trial. Nat. Med. 2022, 28, 1813–1822. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chu, Y.; Bartus, R.T.; Manfredsson, F.P.; Olanow, C.W.; Kordower, J.H. Long-Term Post-Mortem Studies Following Neurturin Gene Therapy in Patients with Advanced Parkinson’s Disease. Brain 2020, 143, 960–975. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Kordower, J.H. Post-Mortem Studies of Neurturin Gene Therapy for Parkinson’s Disease: Two Subjects with 10 Years CERE120 Delivery. Mov. Disord. 2023, 38, 1728–1736. [Google Scholar] [CrossRef] [PubMed]
- Kells, A.; Van Laar, A.D.; Bankiewicz, K. GDNF Gene Therapy for Parkinson’s Disease. In Translational Neuroscience; Tuszynski, M.H., Ed.; Springer: Cham, Switzerland, 2025. [Google Scholar] [CrossRef]
- Gupta, A.; Mishra, S.K.; Lascelles, B.D.X. Emerging Evidence of Artemin/GFRα3 Signaling in Musculoskeletal Pain. Osteoarthr. Cartil. 2025, 33, 196–206. [Google Scholar] [CrossRef]
- Rolan, P.E.; O’Neill, G.; Versage, E.; Rana, J.; Tang, Y.; Galluppi, G.; Aycardi, E. First-In-Human, Double-Blind, Placebo-Controlled, Randomized, Dose-Escalation Study of BG00010, a Glial Cell Line-Derived Neurotrophic Factor Family Member, in Subjects with Unilateral Sciatica. PLoS ONE 2015, 10, e0125034. [Google Scholar] [CrossRef]
- Okun, M.S.; Foote, K.D. Parkinson’s Disease DBS: What, When, Who and Why? The Time Has Come to Tailor DBS Targets. Expert Rev. Neurother. 2010, 10, 1847–1857. [Google Scholar] [CrossRef]
- Huttunen, H.J.; Booms, S.; Sjögren, M.; Kerstens, V.; Johansson, J.; Holmnäs, R.; Koskinen, J.; Kulesskaya, N.; Fazio, P.; Woolley, M.; et al. Intraputamenal Cerebral Dopamine Neurotrophic Factor in Parkinson’s Disease: A Randomized, Double-Blind, Multicenter Phase 1 Trial. Mov. Disord. 2023, 38, 1209–1222. [Google Scholar] [CrossRef]
- Porcari, C.; Cattaneo, S.; Crippa, L.; Simonato, M.; Bettegazzi, B. Exploring the Diversity of Biological Processes Regulated by Glial Cell Line-Derived Neurotrophic Factor, a Pleiotropic Molecule with Therapeutic Potential. Front. Physiol. 2025, 16, 1618330. [Google Scholar] [CrossRef] [PubMed]
- Sidorova, Y.A.; Bespalov, M.M.; Wong, A.W.; Kambur, O.; Jokinen, V.; Lilius, T.O.; Suleymanova, I.; Karelson, G.; Rauhala, P.V.; Karelson, M.; et al. A Novel Small Molecule GDNF Receptor RET Agonist, BT13, Promotes Neurite Growth from Sensory Neurons in Vitro and Attenuates Experimental Neuropathy in the Rat. Front. Pharmacol. 2017, 8, 365. [Google Scholar] [CrossRef]
- Mahato, A.K.; Kopra, J.; Renko, J.M.; Visnapuu, T.; Korhonen, I.; Pulkkinen, N.; Bespalov, M.M.; Domanskyi, A.; Ronken, E.; Piepponen, T.P.; et al. Glial Cell Line-Derived Neurotrophic Factor Receptor Rearranged during Transfection Agonist Supports Dopamine Neurons in Vitro and Enhances Dopamine Release In Vivo. Mov. Disord. 2020, 35, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Haider, M.S.; Mahato, A.K.; Kotliarova, A.; Forster, S.; Böttcher, B.; Stahlhut, P.; Sidorova, Y.; Luxenhofer, R. Biolog-ical Activity In Vitro, Absorption, BBB Penetration, and Tolerability of Nanoformulation of BT44: RET Agonist with Disease-Modifying Potential for the Treatment of Neurodegeneration. Biomacromolecules 2023, 24, 4348–4365. [Google Scholar] [CrossRef]
- Sun, W.; Ye, B.; Chen, S.; Yang, H.; Zhou, Y.; Ge, L.; Liu, Y. Neuro–bone tissue engineering: Emerging mechanisms, potential strategies, and current challenges. Bone Res. 2023, 11, 65. [Google Scholar] [CrossRef]
- Saarma, M.; Karelson, M.; Bespalov, M.; Pilv, M. Methods of Facilitating Neural Cell Survival Using GDNF Family Ligand (GFL) Mimetics or RET Signaling Pathway Activators. U.S. Patent 8,901,129, 2 December 2014. [Google Scholar]
- Airaksinen, M.; Saarma, M. The GDNF Family: Signalling, Biological Functions and Therapeutic Value. Nat. Rev. Neurosci. 2002, 3, 383–394. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.J.; Gyorkos, A.M.; Spitsbergen, J.M. Short-Term Exercise Increases GDNF Protein Levels in the Spinal Cord of Young and Old Rats. Neuroscience 2013, 240, 258–268. [Google Scholar] [CrossRef]
- Subbiah, V.; Shen, T.; Terzyan, S.S.; Liu, X.; Hu, X.; Patel, K.P.; Hu, M.; Cabanillas, M.; Behrang, A.; Meric-Bernstam, F.; et al. Structural Basis of Acquired Resistance to Selpercatinib and Pralsetinib Mediated by Non-Gatekeeper RET Mutations. Ann. Oncol. 2021, 32, 261–268. [Google Scholar] [CrossRef]
- Mashanov, V.; Alwan, A.; Kim, M.W.; Lai, D.; Poerio, A.; Ju, Y.M.; Kim, J.H.; Yoo, J.J. Synergistic Effect of CNTF and GDNF on Directed Neurite Growth in Chick Embryo Dorsal Root Ganglia. PLoS ONE 2020, 15, e0240235. [Google Scholar] [CrossRef]
- Fantone, S.; Tossetta, G.; Montironi, R.; Senzacqua, M.; Marzioni, D.; Mazzucchelli, R. Ciliary Neurotrophic Factor (CNTF) and Its Receptor (CNTFRα) Signal through MAPK/ERK Pathway in Human Prostate Tissues: A Morphological and Biomolecular Study. Eur. J. Histochem. 2020, 64, 3147. [Google Scholar] [CrossRef]
- Ghasemi, M.; Alizadeh, E.; Saei Arezoumand, K.; Fallahi Motlagh, B.; Zarghami, N. Ciliary Neurotrophic Factor (CNTF) Delivery to Retina: An Overview of Current Research Advancements. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1694–1707. [Google Scholar] [CrossRef]
- Hu, Z.; Deng, N.; Liu, K.; Zhou, N.; Sun, Y.; Zeng, W. CNTF-STAT3-IL-6 Axis Mediates Neuroinflammatory Cascade across Schwann Cell-Neuron-Microglia. Cell Rep. 2020, 31, 107657. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Han, L.; Ni, H.; Ke, S.; Pan, T. Association of Cerebrospinal Fluid Ciliary Neurotrophic Factor Levels with Cognitive Decline and Disease Progression. J. Alzheimers Dis. 2025, 105, 573–581. [Google Scholar] [CrossRef]
- Do Rhee, K.; Wang, Y.; ten Hoeve, J.; Stiles, L.; Nguyen, T.T.T.; Zhang, X.; Vergnes, L.; Reue, K.; Shirihai, O.; Bok, D.; et al. Ciliary Neurotrophic Factor-Mediated Neuroprotection Involves Enhanced Glycolysis and Anabolism in Degenerating Mouse Retinas. Nat. Commun. 2022, 13, 7037. [Google Scholar] [CrossRef] [PubMed]
- Kauper, K.; Nystuen, A.; Orecchio, L.; Gonzalez-Lopez, E.; Lee, A.; Duncan, J.L.; Stewart, J.M.; Aaberg, T., Jr. Long-Term Durability of Ciliary Neurotrophic Factor-Releasing Revakinagene Taroretcel-lwey in Individuals With Retinal Degenerative Disorders. Invest. Ophthalmol. Vis. Sci. 2025, 66, 3. [Google Scholar] [CrossRef]
- Rafii, P.; Cruz, P.R.; Ettich, J.; Seibel, C.; Padrini, G.; Wittich, C.; Lang, A.; Petzsch, P.; Köhrer, K.; Moll, J.M.; et al. Engineered Interleukin-6-Derived Cytokines Recruit Artificial Receptor Complexes and Disclose CNTF Signaling via the OSMR. J. Biol. Chem. 2024, 300, 107251. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Hopkins, J.J.; Heier, J.S.; Birch, D.G.; Halperin, L.S.; Albini, T.A.; Brown, D.M.; Jaffe, G.J.; Tao, W.; Williams, G.A. Ciliary Neurotrophic Factor Delivered by Encapsulated Cell Intraocular Implants for Treatment of Geographic Atrophy in Age-Related Macular Degeneration. Proc. Natl. Acad. Sci. USA 2011, 108, 6241–6245. [Google Scholar] [CrossRef]
- Girmens, J.-F.; Sahel, J.-A.; Marazova, K. Dry Age-Related Macular Degeneration: A Currently Unmet Clinical Need. Intractable Rare Dis. Res. 2012, 1, 103–114. [Google Scholar] [CrossRef]
- Hoy, S.M. Revakinagene Taroretcel: First Approval. Mol. Diagn. Ther. 2025, 29, 553–561. [Google Scholar] [CrossRef]
- DrugBank Online. Ciliary Neurotrophic Factor. Available online: https://go.drugbank.com/drugs/DB05344 (accessed on 12 December 2025).
- Viswanadhapalli, S.; Dileep, K.V.; Zhang, K.Y.J.; Nair, H.B.; Vadlamudi, R.K. Targeting LIF/LIFR Signaling in Cancer. Genes Dis. 2021, 9, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Ferraguti, G.; Terracina, S.; Tarani, L.; Fanfarillo, F.; Allushi, S.; Caronti, B.; Tirassa, P.; Polimeni, A.; Lucarelli, M.; Cavalcanti, L.; et al. Nerve Growth Factor and the Role of Inflammation in Tumor Development. Curr. Issues Mol. Biol. 2024, 46, 965–989. [Google Scholar] [CrossRef]
- Cuéllar-Pérez, R.; Jauregui-Huerta, F.; Ruvalcaba-Delgadillo, Y.; Montero, S.; Lemus, M.; Roces de Álvarez-Buylla, E.; García-Estrada, J.; Luquín, S. K252a Prevents Microglial Activation Induced by Anoxic Stimulation of Carotid Bodies in Rats. Toxics 2023, 11, 871. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Pineda, V.G.; Nieto-Mendoza, E.; Torres-Cruz, F.M.; Hernández-Echeagaray, E. Neurotrophin-3 Rescues Striatal Synaptic Plasticity in Model of Neurodegeneration by PLC Signaling Activation. CNS Neurol. Disord. Drug Targets 2024, 23, 1488–1498. [Google Scholar] [CrossRef]
- Kasakura, N.; Murata, Y.; Suzuki, K.; Segi-Nishida, E. Role of Endogenous NT-3 in Neuronal Activity and Neurogenesis in the Hippocampal Dentate Gyrus. Neurosci. Res. 2025, 218, 104923. [Google Scholar] [CrossRef]
- Zwingelberg, S.B.; Bachmann, B.O.; Cursiefen, C. Real Life Data on Efficacy and Safety of Topical NGF Eye Drops (Cenegermin). Klin. Monatsblatter Augenheilkd 2020, 237, 1455–1461. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Xiao, L. Progress in AAV-Mediated In Vivo Gene Therapy and Its Applications in Central Nervous System Diseases. Int. J. Mol. Sci. 2025, 26, 2213. [Google Scholar] [CrossRef]
- Rafii, M.S.; Tuszynski, M.H.; Thomas, R.G.; Barba, D.; Brewer, J.B.; Rissman, R.A.; Siffert, J.; Aisen, P.S.; AAV2-NGF Study Team. Adeno-Associated Viral Vector (Serotype 2)-Nerve Growth Factor for Patients with Alzheimer Disease: A Randomized Clinical Trial. JAMA Neurol. 2018, 75, 834–841. [Google Scholar] [CrossRef]
- Liang, Z.-J.; Tan, J.; Tang, L.; Xie, Z.-B.; Chen, G.-J.; Liu, G.-J.; Yuan, L.; Wang, K.-X.; Ding, H.-P.; Qiu, H.; et al. NGF monoclonal antibody DS002 alleviates chemotherapy-induced peripheral neuropathy in rats. Acta Pharmacol. Sin. 2022, 43, 2841–2847. [Google Scholar] [CrossRef] [PubMed]
- Shanks, H.R.C.; Chen, K.; Reiman, E.M.; Blennow, K.; Cummings, J.L.; Massa, S.M.; Longo, F.M.; Börjesson-Hanson, A.; Windisch, M.; Schmitz, T.W. P75 Neurotrophin Receptor Modulation in Mild to Moderate Alzheimer Disease: A Randomized, Placebo-Controlled Phase 2a Trial. Nat. Med. 2024, 30, 1761–1770. [Google Scholar] [CrossRef] [PubMed]
- Sheffield, K.S.; Vohra, R.; Scott, J.A.; Ross, G.M. Using Surface Plasmon Resonance Spectroscopy to Characterize the Inhibition of NGF-p75(NTR) and proNGF-p75(NTR) Interactions by Small Molecule Inhibitors. Pharmacol. Res. 2016, 103, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Awad-Igbaria, Y.; Edelman, D.; Ianshin, E.; Abu-Ata, S.; Shamir, A.; Bornstein, J.; Palzur, E. Inflammation-Induced Mast Cell-Derived Nerve Growth Factor: A Key Player in Chronic Vulvar Pain? Brain 2025, 148, 331–346. [Google Scholar] [CrossRef]
- Kigami, D.; Butt, M.T.; Brown, D.L.; Matsumoto, M.; Ito, H. Neurotoxicity Studies with a Tropomyosin-Related Kinase A Inhibitor, ASP7962, on the Sympathetic and Sensory Nervous Systems in Rats. Toxicol. Lett. 2021, 344, 34–45. [Google Scholar] [CrossRef]
- Walsh, D.A.; Neogi, T. A Tale of Two TrkA Inhibitor Trials: Same Target, Divergent Results. Osteoarthr. Cartil. 2019, 27, 1575–1577. [Google Scholar] [CrossRef]
- Kang, Z.; Zheng, Z.; Guo, W. Efficacy of Agomelatine on Sleep Disorders and Lateral Habenula Neuronal Activity in Chronic Restraint Stress Depression Model Mice. Psychopharmacology 2025, 242, 353–360. [Google Scholar] [CrossRef]
- Wellington, N.J.; Boųcas, A.P.; Lagopoulos, J.; Quigley, B.L.; Kuballa, A.V. Molecular Pathways of Ketamine: A Systematic Review of Immediate and Sustained Effects on PTSD. Psychopharmacology 2025, 242, 1197–1243. [Google Scholar] [CrossRef]
- Sallie, F.N.; Pienaar, L.; Lubbe, A.; Xhakaza, S.; Manne, S.R.; de la Torre, B.G.; Albericio, F.; Mu Daniels, W.; Me Millen, A.; Baijnath, S. Neurobehavioral and Molecular Changes in a Rodent Model of ACTH-Induced HPA Axis Dysfunction. Brain Res. 2024, 1834, 148913. [Google Scholar] [CrossRef]
- Cai, J.; Chen, X.; Jin, Z.; Chi, Z.; Xiong, J. Effects of Adjunctive Esketamine on Depression in Elderly Patients Undergoing Hip Fracture Surgery: A Randomized Controlled Trial. BMC Anesthesiol. 2024, 24, 340. [Google Scholar] [CrossRef]
- Rengarajan, S.; Venkatachalam, S.; Pattabi, S.; Jones, S.; Prabhu, K.; Krishna, V.; Prasanth, K. Neuroprotection by Cerebrolysin and Citicoline Through the Upregulation of Brain-Derived Neurotrophic Factor (BDNF) Expression in the Affected Neural Cells: A Preliminary Clue Obtained Through an In Vitro Study. Cureus 2024, 16, e54665. [Google Scholar] [CrossRef]
- Eremenko, E.; Mittal, K.; Berner, O.; Kamenetsky, N.; Nemirovsky, A.; Elyahu, Y.; Monsonego, A. BDNF-Producing, Amy-loid β-Specific CD4 T Cells as Targeted Drug-Delivery Vehicles in Alzheimer’s Disease. EBioMedicine 2019, 43, 424–434. [Google Scholar] [CrossRef]
- Raychaudhuri, S.P.; Sanyal, M.; Weltman, H.; Kundu-Raychaudhuri, S. K252a, a High-Affinity Nerve Growth Factor Receptor Blocker, Improves Psoriasis: An In Vivo Study Using the Severe Combined Immunodeficient Mouse-Human Skin Model. J. Investig. Dermatol. 2004, 122, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Kuiper-Makris, C.; Fahle, L.; Zeitouny, C.; Vohlen, C.; Klymenko, O.; Stephan, S.; Mižik, I.; Bae-Gartz, I.; Selle, J.; Hirani, D.; et al. BDNF-TrkB Signaling Maintains AT2 Survival and Is Blocked in Hyperoxia-Induced Neonatal Lung Injury. Am. J. Respir. Cell Mol. Biol. 2025, 73, 517–529. [Google Scholar] [CrossRef]
- Gupta, R.; Dittmeier, M.; Wohlleben, G.; Nickl, V.; Bischler, T.; Luzak, V.; Wegat, V.; Doll, D.; Sodmann, A.; Bady, E.; et al. Atypical Cellular Responses Mediated by Intracellular Constitutive Active TrkB (NTRK2) Kinase Domains and a Solely Intracellular NTRK2-Fusion Oncogene. Cancer Gene Ther. 2024, 31, 1357–1379. [Google Scholar] [CrossRef] [PubMed]
- Peach, C.J.; Tonello, R.; Damo, E.; Gomez, K.; Calderon-Rivera, A.; Bruni, R.; Bansia, H.; Maile, L.; Manu, A.M.; Hahn, H.; et al. Neuropilin-1 Inhibition Suppresses Nerve Growth Factor Signaling and Nociception in Pain Models. J. Clin. Investig. 2024, 135, e183873. [Google Scholar] [CrossRef]
- Ding, X.; Zuo, Y.; Liu, Z.; Sun, Y.; Wang, L.; Xie, Y.; Liu, G.; Liu, C. Recombinant Neurotrophin-3 with the Ability to Penetrate the Blood-Brain Barrier: A New Strategy against Alzheimer’s Disease. Int. J. Biol. Macromol. 2025, 293, 139359. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Massa, S.M.; Tran, K.C.; Simmons, D.A.; Rajadas, J.; Zeng, A.Y.; Jang, T.; Carsanaro, S.; Longo, F.M. A Small Molecule TrkB/TrkC Neurotrophin Receptor Co-Activator with Distinctive Effects on Neuronal Survival and Process Outgrowth. Neuropharmacology 2016, 110, 343–361. [Google Scholar] [CrossRef]
- Nikiforov, D.M.; Povarnina, P.Y.; Gudasheva, T.A.; Nadorova, A.V.; Kolik, L.G.; Valdman, E.A.; Vakhitova, Y.V.; Seredenin, S.B. Study of the Pharmacological Activity Spectrum of the New Original NT-3 Mimetic Dipeptide GTS-302. Dokl. Biochem. Biophys. 2025, 520, 74–82. [Google Scholar] [CrossRef]
- Huttunen, H.J.; Saarma, M. CDNF Protein Therapy in Parkinson’s Disease. Cell Transplant. 2019, 28, 349–366. [Google Scholar] [CrossRef]
- Sidorova, Y.A.; Saarma, M. Small Molecules and Peptides Targeting Glial Cell Line-Derived Neurotrophic Factor Receptors for the Treatment of Neurodegeneration. Int. J. Mol. Sci. 2020, 21, 6575. [Google Scholar] [CrossRef]
- Goldberg, J.L.; Beykin, G.; Satterfield, K.R.; Nunez, M.; Lam, B.L.; Albini, T.A. Phase I NT-501 Ciliary Neurotrophic Factor Implant Trial for Primary Open-Angle Glaucoma: Safety, Neuroprotection, and Neuroenhancement. Oph-thalmol. Sci. 2023, 3, 100298. [Google Scholar] [CrossRef]
- Nishikubo, H.; Kawabata, K.; Kanei, S.; Aoyama, R.; Ma, D.; Sano, T.; Imanishi, D.; Sakuma, T.; Maruo, K.; Fan, C.; et al. Multi-Cancer Genome Profiling for Neurotrophic Tropomyosin Receptor Kinase (NTRK) Fusion Genes: Analysis of Profiling Database of 88,688 Tumors. Cancers 2025, 17, 2250. [Google Scholar] [CrossRef]
- Repasky, E.; Mohammadpour, H. Targeting Nerve Growth Factor: An Achilles’ Heel for Tumors? J. Immunother. Cancer 2025, 13, e011609. [Google Scholar] [CrossRef] [PubMed]
- Okuno, K.; Ikemura, K.; Okamoto, R.; Oki, K.; Watanabe, A.; Kuroda, Y.; Kidachi, M.; Fujino, S.; Nie, Y.; Higuchi, T.; et al. CAF-associated genes putatively representing distinct prognosis by in silico landscape of stromal components of colon cancer. PLoS ONE 2024, 19, e0299827. [Google Scholar] [CrossRef]
- Dudas, J.; Glueckert, R.; Greier, M.d.C.; Hofauer, B.G. The Relationships Among Perineural Invasion, Tumor–Nerve Interaction and Immunosuppression in Cancer. Onco 2025, 5, 25. [Google Scholar] [CrossRef]
- Miyaue, N.; Yamanishi, Y.; Ito, Y.; Ando, R.; Nagai, M. CSF Neopterin Levels Are Elevated in Various Neurological Diseases and Aging. J. Clin. Med. 2024, 13, 4542. [Google Scholar] [CrossRef] [PubMed]
- Terracina, S.; Ferraguti, G.; Tarani, L.; Fanfarillo, F.; Tirassa, P.; Ralli, M.; Iannella, G.; Polimeni, A.; Lucarelli, M.; Greco, A.; et al. Nerve Growth Factor and Autoimmune Diseases. Curr. Issues Mol. Biol. 2023, 45, 8950–8973. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Ao, X.; Xiang, X.; Zhang, J.; Cai, J.; Fu, J.; Zhang, W.; Zheng, Z.; Chu, J.; Huang, M.; et al. Mast cell activation by NGF drives the formation of trauma-induced heterotopic ossification. JCI Insight 2024, 10, e179759. [Google Scholar] [CrossRef]
- Lin, T.-H.; Chang, K.-H.; Chiu, Y.-J.; Weng, Z.-K.; Sun, Y.-C.; Lin, W.; Lee-Chen, G.-J.; Chen, C.-M. Neuroprotective Action of Coumarin Derivatives through Activation of TRKB-CREB-BDNF Pathway and Reduction of Caspase Activity in Neuronal Cells Expressing Pro-Aggregated Tau Protein. Int. J. Mol. Sci. 2022, 23, 12734. [Google Scholar] [CrossRef]
- Mitrovic, M.; Selakovic, D.; Jovicic, N.; Ljujic, B.; Rosic, G. BDNF/proBDNF Interplay in the Mediation of Neuronal Apoptotic Mechanisms in Neurodegenerative Diseases. Int. J. Mol. Sci. 2025, 26, 4926. [Google Scholar] [CrossRef]
- Farina, L.; Minnone, G.; Alivernini, S.; Caiello, I.; MacDonald, L.; Soligo, M.; Manni, L.; Tolusso, B.; Coppola, S.; Zara, E.; et al. Pro Nerve Growth Factor and Its Receptor p75NTR Activate Inflammatory Responses in Synovial Fibroblasts: A Novel Targetable Mechanism in Arthritis. Front. Immunol. 2022, 13, 818630. [Google Scholar] [CrossRef]
- Chen, R.; Chen, W.; Li, P.; Zhao, Y.; Zeng, Q.; Chen, W.; Cao, D. Function and application of brain derived neurotrophic factor precursors (Review). Int. J. Mol. Med. 2025, 56, 105. [Google Scholar] [CrossRef] [PubMed]
- Fedotcheva, T.A.; Shimanovsky, N.L. Pharmacological Strategies for Overcoming Multidrug Resistance to Chemotherapy. Pharm. Chem. J. 2023, 56, 1307–1313. [Google Scholar] [CrossRef]
- Chai, A.B.; Callaghan, R.; Gelissen, I.C. Regulation of P-Glycoprotein in the Brain. Int. J. Mol. Sci. 2022, 23, 14667. [Google Scholar] [CrossRef]
- Samario-Román, J.; Larqué, C.; Pánico, P.; Ortiz-Huidobro, R.I.; Velasco, M.; Escalona, R.; Hiriart, M. NGF and Its Role in Immunoendocrine Communication during Metabolic Syndrome. Int. J. Mol. Sci. 2023, 24, 1957. [Google Scholar] [CrossRef]
- Benzaquen, D.; Lawrence, Y.R.; Taussky, D.; Zwahlen, D.; Oehler, C.; Champion, A. The Crosstalk between Nerves and Cancer—A Poorly Understood Phenomenon and New Possibilities. Cancers 2024, 16, 1875. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Di Benedetto, S.; Müller, V. From Homeostasis to Neuroinflammation: Insights into Cellular and Molecular Interactions and Network Dynamics. Cells 2025, 14, 54. [Google Scholar] [CrossRef] [PubMed]
- Palasz, E.; Wilkaniec, A.; Stanaszek, L.; Andrzejewska, A.; Adamczyk, A. Glia-Neurotrophic Factor Relationships: Possible Role in Pathobiology of Neuroinflammation-Related Brain Disorders. Int. J. Mol. Sci. 2023, 24, 6321. [Google Scholar] [CrossRef]
- Policastro, G.; Brunelli, M.; Tinazzi, M.; Chiamulera, C.; Emerich, D.F.; Paolone, G. Cytokine-, Neurotrophin-, and Motor Rehabilitation-Induced Plasticity in Parkinson’s Disease. Neural Plast. 2020, 2020, 8814028. [Google Scholar] [CrossRef] [PubMed]
- Jaffal, S.; Khalil, R. Targeting nerve growth factor for pain relief: Pros and cons. Korean J. Pain 2024, 37, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Semenova, A.A.; Kharitonova, E.V.; Khilazheva, E.D.; Salmina, A.B. Valproic Acid Increases the Level of Expression of BDNF, GDNF and THEIR Receptors in the Embryonic Brain of Mice and Inducts the Stereotype Behavior. Cell Tissue Biol. 2021, 15, 563–567. [Google Scholar] [CrossRef]
- Sartor, G.C.; Malvezzi, A.M.; Kumar, A.; Andrade, N.S.; Wiedner, H.J.; Vilca, S.J.; Janczura, K.J.; Bagheri, A.; Al-Ali, H.; Powell, S.K.; et al. Enhancement of BDNF Expression and Memory by HDAC Inhibition Requires BET Bromodomain Reader Proteins. J. Neurosci. 2019, 39, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Wang, T.Y.; Lu, R.B.; Wang, L.J.; Chang, C.H.; Chiang, Y.C.; Tsai, K.W. Peripheral BDNF correlated with miRNA in BD-II patients. J. Psychiatr. Res. 2021, 136, 184–189. [Google Scholar] [CrossRef]
- Fedotcheva, T.A.; Shimanovsky, N.L. Pharmacological Strategies to Overcome Tumor Resistance to Anticancer Hormonal, Targeted, and Immunotherapy. Pharm. Chem. J. 2023, 57, 171–179. [Google Scholar] [CrossRef]
- Joshi, R.; Salton, S.R.J. Neurotrophin Crosstalk in the Etiology and Treatment of Neuropsychiatric and Neurodegenerative Disease. Front. Mol. Neurosci. 2022, 15, 932497. [Google Scholar] [CrossRef]
- Luz, M.; Mohr, E.; Fibiger, H.C. GDNF-induced cerebellar toxicity: A brief review. Neurotoxicology 2016, 52, 46–56. [Google Scholar] [CrossRef]
- Pardridge, W.M. Blood-Brain Barrier Drug Targeting Enables Neuroprotection in Brain Ischemia Following Delayed Intravenous Administration of Neurotrophins. In Madame Curie Bioscience Database; Landes Bioscience: Austin, TX, USA, 2000–2013. Available online: https://www.ncbi.nlm.nih.gov/books/NBK6007/ (accessed on 14 December 2025).
- Esteban-Ortega, G.M.; Torres-Campos, E.; Díaz-Guerra, M. Retrograde transport of neurotrophin receptor TrkB-FL induced by excitotoxicity regulates Golgi stability and is a target for stroke neuroprotection. Cell Death Dis. 2025, 16, 659. [Google Scholar] [CrossRef]
- Weihrauch, T.; Limberg, M.M.; Gray, N.; Schmelz, M.; Raap, U. Neurotrophins: Neuroimmune Interactions in Human Atopic Diseases. Int. J. Mol. Sci. 2023, 24, 6105. [Google Scholar] [CrossRef] [PubMed]
- Minnone, G.; De Benedetti, F.; Bracci-Laudiero, L. NGF and Its Receptors in the Regulation of Inflammatory Response. Int. J. Mol. Sci. 2017, 18, 1028. [Google Scholar] [CrossRef]
- Salazar-Formoso, V.; Morales-Sánchez, M.A. Efficacy of Superoxide Dismutase in the Treatment of Vitiligo: A Systematic Review. Cureus 2025, 17, e88335. [Google Scholar] [CrossRef]
- Kim, J.E.; Ji, Y.E.; Hwang, H.J.; Go, G.E.; Lim, H.J.; Yoo, J.; Kim, J.; Park, D.; Kim, E.H.; Kim, D.; et al. Engineered MSC-EVs loaded with BDNF-enhancing neuropeptides via a non-disruptive method enhance post-stroke neuroregeneration via intranasal delivery. J. Nanobiotechnol. 2025, 23, 594. [Google Scholar] [CrossRef]
- Moldavskii, D.; Gilazieva, Z.; Fattakhova, A.; Solovyeva, V.; Issa, S.; Sufianov, A.; Sufianova, G.; Rizvanov, A. AAV-Based Gene Therapy: Opportunities, Risks, and Scale-Up Strategies. Int. J. Mol. Sci. 2025, 26, 8282. [Google Scholar] [CrossRef]
- Byrne, B.J.; Flanigan, K.M.; Matesanz, S.E.; Finkel, R.S.; Waldrop, M.A.; D’Ambrosio, E.S.; Johnson, N.E.; Smith, B.K.; Bönnemann, C.; Carrig, S.; et al. Current clinical applications of AAV-mediated gene therapy. Mol. Ther. 2025, 33, 2479–2516. [Google Scholar] [CrossRef]
- Suen, P.C.; Wu, K.; Levine, E.S.; Mount, H.T.; Xu, J.L.; Lin, S.Y.; Black, I.B. Brain-derived neurotrophic factor rapidly enhances phosphorylation of the postsynaptic N-methyl-D-aspartate receptor subunit 1. Proc. Natl. Acad. Sci. USA 1997, 94, 8191–8195. [Google Scholar] [CrossRef]
- Turovskaya, M.V.; Gavrish, M.S.; Tarabykin, V.S.; Babaev, A.A. Overexpression of BDNF Suppresses the Epileptiform Activity in Cortical Neurons of Heterozygous Mice with a Transcription Factor Sip1 Deletion. Int. J. Mol. Sci. 2024, 25, 10537. [Google Scholar] [CrossRef] [PubMed]
- Kustova, A.O.; Gavrish, M.S.; Sergeeva, M.A.; Avlasenko, D.A.; Kiseleva, A.O.; Epifanova, E.A.; Babaev, A.A.; Mishchenko, T.A.; Vedunova, M.V. The Influence of Neurotrophic Factors BDNF and GDNF Overexpression on the Functional State of Mice and Their Adaptation to Audiogenic Seizures. Brain Sci. 2022, 12, 1039. [Google Scholar] [CrossRef]
- Toader, C.; Serban, M.; Munteanu, O.; Covache-Busuioc, R.-A.; Enyedi, M.; Ciurea, A.V.; Tataru, C.P. From Synaptic Plasticity to Neurodegeneration: BDNF as a Transformative Target in Medicine. Int. J. Mol. Sci. 2025, 26, 4271. [Google Scholar] [CrossRef] [PubMed]
- Panvino, F.; Paparella, R.; Tarani, F.; Lombardi, C.; Ferraguti, G.; Pisani, F.; Fiore, M.; Pancheva, R.; Ardizzone, I.; Tarani, L. Neurotrophins in Neurodevelopmental Disorders: A Narrative Review of the Literature. Int. J. Mol. Sci. 2025, 26, 8335. [Google Scholar] [CrossRef] [PubMed]
- Azzolini, F.; Gilio, L.; Pavone, L.; Iezzi, E.; Dolcetti, E.; Bruno, A.; Buttari, F.; Musella, A.; Mandolesi, G.; Guadalupi, L.; et al. Neuroinflammation Is Associated with GFAP and sTREM2 Levels in Multiple Sclerosis. Biomolecules 2022, 12, 222. [Google Scholar] [CrossRef]
- Fedotchev, A.I.; Zemlyanaya, A.A. Brain State-Dependent Non-Invasive Neurostimulation with EEG Feedback: Achievements and Prospects. Sovrem. Tehnol. Med. 2023, 15, 33. [Google Scholar] [CrossRef] [PubMed]
- de Paiva, C.S.; St. Leger, A.J.; Caspi, R.R. Mucosal immunology of the ocular surface. Mucosal Immunol. 2022, 15, 1143–1157. [Google Scholar] [CrossRef] [PubMed]
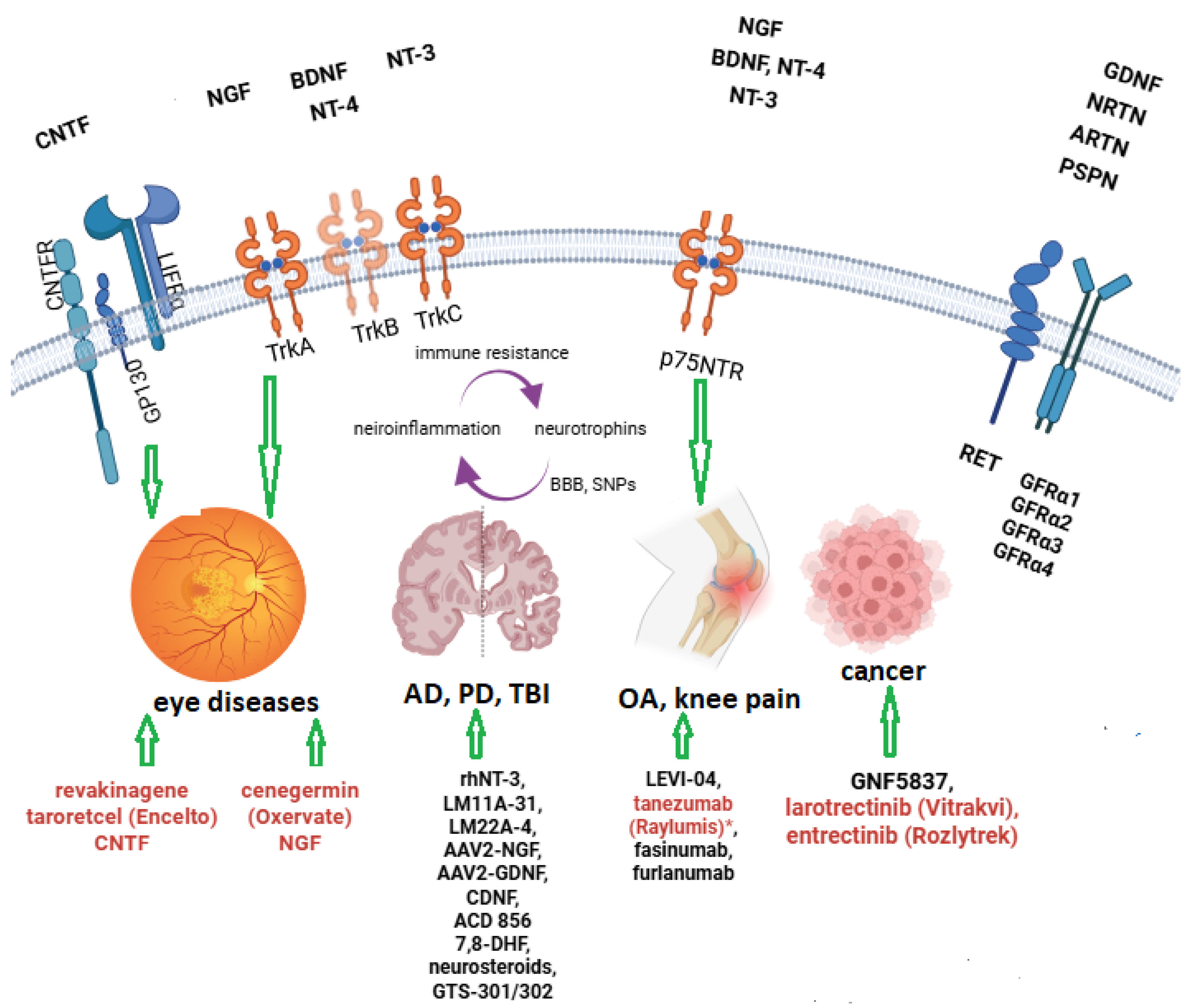
| NT | NGF | BDNF | NT-3 | GDNF | CNTF |
|---|---|---|---|---|---|
| Structure | Belongs to the family of neurotrophins | Belongs to the family of neurotrophins | Belongs to the same family as NGF and BDNF | Distantly related to TGF-β family | IL-6 family member |
| Receptor | High-affinity receptor TRkA and low-affinity receptor LNGFR/p75NTR | TRkB and LNGFR/p75NTR | TrkC | GFRα1 and RET | gp130, LIFR, CNTFR |
| Main function | Inhibits neuronal apoptosis, promotes axon regeneration and angiogenesis through its interaction with TrkA, a high-affinity receptor; induces pain through its interaction with p75NTR [5,146] | Regulates synaptic plasticity [12], prevents microglial activation after hypoxic stimulation [147] | Maintains striatal synaptic plasticity [148] and neuronal differentiation [149] | Improves development and survival of dopaminergic neurons [106] | Activates CNTFR and the two signaling β-receptors glycoprotein 130 (gp130) and leukemia inhibitory factor receptor (LIFR). Neuroprotective cytokine [150] |
| Drug | Mode of Action | Disease | Delivery Route | Key Results | Development Stage |
|---|---|---|---|---|---|
| Agonists/mimetics of NGF | |||||
| Cenegermin (Oxervate™) [27,150] | The recombinant human NGF | Neurotrophic keratitis | Eye drops | Restores corneal integrity | Approved |
| hNGFp [28] | The recombinant mutated form of human hNGF with reduced binding affinity to p75NTR | Retinitis pigmentosa | Eye drops | Reduces microglia-mediated inflammation | Investigational |
| AAV2-NGF [151,152] | The recombinant human NGF | AD | Intracerebral injections | No significant effect | Phase II clinical trial |
| Antagonists of NFG | |||||
| Anti-NGF antibody Tanezumab, fasinumab, furlanumab [43,44,45] | Anti-NGF antibody | OA | Intravenous injection, subcutaneous injection | Relieves pain but destroys the joint due to overload | Clinical trials stopped |
| Anti-NGF antibody DS002 [153] | Blocks NGF binding to TrkA | OA | Subcutaneous injection | Alleviates chemotherapy-induced peripheral neuropathy in rats | Investigational |
| LEVI-04 [47] | p75NTR fusion protein designed to bind excess NGF | OA | Intravenous injection | Clinically meaningful improvement in pain, function, and other outcomes | Phase II clinical trial |
| LM11A-31 [154] | BBB—penetrating small molecule, p75NTR antagonist | AD | Oral | Slows progression of pathophysiological features of AD | Phase II clinical trial |
| PD90780 [155] | NGF antagonist, prevents it from binding to p75NTR | Potential anti-cancer drug | In vitro, i.p. | Experimental chemical compound for in vitro studies | Investigational |
| Ro08-2750 [156] | Reversible NGF inhibitor | potential anti-cancer drug | i.p. | Inhibition of tumor growth (myeloid leukemia) | Investigational |
| ASP7962 [157] | TrkA inhibitor | OA | Oral | Analgesic efficacy | Phase II clinical trial |
| GZ 8998A [158] | TrkA inhibitor | OA | Intra-articular | Analgesic efficacy | Phase II clinical trial stopped |
| Agonists/mimetics of BDNF | |||||
| ACD856 [70,71] | Allosteric activator | AD | Intravenous and oral administration | Phase 1 clinical trial | |
| Agomelatine [159] ketamine [160] ACTH-analogues [161] esketamine [162] neurosteroids DHEA, progesterone, Allo [72] cerebrolysin [163] | Indirect stimulation of BDNF synthesis via enhancement of TrkB phosphorylation | Different indications | Different routes | Neuroprotective action | Approved, but not for neurodegenerative diseases |
| Cell and gene technologies [164] | Amyloid beta-protein (Aβ)-specific CD4 T cells, genetically engineered to express BDNF | AD | Intracerebroventricularly | Reduced levels of beta-secretase 1 (BACE1)-a protease essential in the cleavage process of the amyloid precursor protein-and ameliorated amyloid pathology and inflammation within the brain parenchyma | Investigational |
| AAV-BDNF [75] | Gene–cell construct with an adenoviral vector encoding mature BDNF | AD | Intrathecally | Improved motor activity of the hind limbs and reduced the size of cysts in rats | Investigational |
| Antagonists of BDNF | |||||
| K252a [165] | Non-selective Trk inhibitor | TBI, tumors | Intracerebroventricular infusion | Prevents brain damage, reduces tumor growth | Investigational |
| Ana12 [166] | Selective TrkB antagonist | TBI | intraperitoneal injection | Reduces pain behaviors and promotes locomotor function recovery | Investigational |
| Larotrectinib (LOXO-101) and Entrectinhib [167] | Trk kinase inhibitors | NTRK fusion-positive cancers | oral | Inhibits tumor cell growth and survival | Approved |
| EG00229 [168] | Inhibitor of co-receptor NRP1 (neurolipin-1) | neuropathic pain | In vitro | Suppresses NGF-stimulated excitation of human and mouse nociceptors neurons in vitro; causes a concentration-dependent inhibition of NGF-induced sensitization of transient receptor potential vanilloid-1 (TRPV1) on nociceptors | Investigational |
| Agonists/mimetics of NT-3 | |||||
| rhNT-3 or TAT [169] | Human HIV-produced transactivator of transcription)-fused recombinant neurotrophin-3 T-NT-3 to enhance NT-3 delivery | AD | Intraperitoneal injection | Inhibits oxidative stress, apoptosis, and inflammatory responses in neural cells by activating TrkC receptors and suppressing M1 microglial activation. In vivo, T-NT-3 improves cognitive and memory impairments in mice | Investigational |
| LM22B-10 [170] | TrkB/TrkC-activating compound | TBI, AD | Eye drops | Improves the healing speed of the corneal epithelium, corneal sensitivity, and corneal nerve density in regular and diabetic mice with corneal wounding | Investigational |
| GTS-302 [171] | TrkB/TrkC activating dipeptide | - | Intraperitoneal | Exhibits antidepressant-like activity anxiolytic and memory-enhancing activity; does not affect pain sensitivity in mice | Investigational |
| Agonists/mimetics of GDNF | |||||
| AAV2-GDNF [115] | Gene therapy | PD | Bilateral intraputaminal delivery | Well tolerated and associated with numerical stability (mild cohort) and improvement (moderate cohort) in clinical assessments at 18 months posttreatment | Phase Ib clinical trial |
| CNS10-NPC-GDNF [116] | Human neural progenitor cells secreting GDNF | ALS | Unilateral, motor cortex | One administration of engineered neural progenitors can provide new support cells and GDNF delivery to the ALS patient spinal cord for up to 42 months post-transplantation | Phase I/IIa clinical trial |
| Neublastin (artemin, BG00010) [117,118,119,120,121] | Selective ligand for the GDNF family receptor alpha-3 (GFRα3) co-receptor | Neuropathic pain, sciatica | Intravenous or subcutaneous | Anti-hyperalgesic effects | Investigational |
| CDNF [123,172] | Recombinant human CDNF | PD | Intraputamenally | No significant changes in motor symptom assessment between placebo and CDNF treatment groups | Phase I clinical trial |
| BT13 [125], BT18 [173] | RET receptor agonists | Glaucoma, neuropathy | Subcutaneous injections | Reduces mechanical hypersensitivity and restores the normal expression levels of sensory neuron markers in dorsal root ganglia | Investigational |
| Agonists/mimetics of CNTF | |||||
| Dapiclermin (Axokine) [140] | CNTF derivative | ALS, obesity and insulin resistance | Intravenous injection | Average weight loss of 6 pounds compared to 2 pounds in patients given a placebo; 70 percent of patients developed blocking antibodies that limited its continued effectiveness | Approve |
| NT-501, Revakinagene taroretcel (ENCELTO™) [142,174] | Recombinant CNTF | Idiopathic MacTel type 2 | Single-dose intravitreal implant | Induces a cascade of signaling events that promote photoreceptor survival | Approved |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Fedotcheva, T.A.; Shimanovsky, N.L. Current State of the Neurotrophin-Based Pharmaceutics in the Treatment of Neurodegenerative Diseases and Neuroinflammation. Med. Sci. 2026, 14, 15. https://doi.org/10.3390/medsci14010015
Fedotcheva TA, Shimanovsky NL. Current State of the Neurotrophin-Based Pharmaceutics in the Treatment of Neurodegenerative Diseases and Neuroinflammation. Medical Sciences. 2026; 14(1):15. https://doi.org/10.3390/medsci14010015
Chicago/Turabian StyleFedotcheva, Tatiana A., and Nikolay L. Shimanovsky. 2026. "Current State of the Neurotrophin-Based Pharmaceutics in the Treatment of Neurodegenerative Diseases and Neuroinflammation" Medical Sciences 14, no. 1: 15. https://doi.org/10.3390/medsci14010015
APA StyleFedotcheva, T. A., & Shimanovsky, N. L. (2026). Current State of the Neurotrophin-Based Pharmaceutics in the Treatment of Neurodegenerative Diseases and Neuroinflammation. Medical Sciences, 14(1), 15. https://doi.org/10.3390/medsci14010015





