Soil Erosion Induced by the Introduction of New Pasture Species in a Faxinal Farm of Southern Brazil
Abstract
1. Introduction
2. Materials and Methods
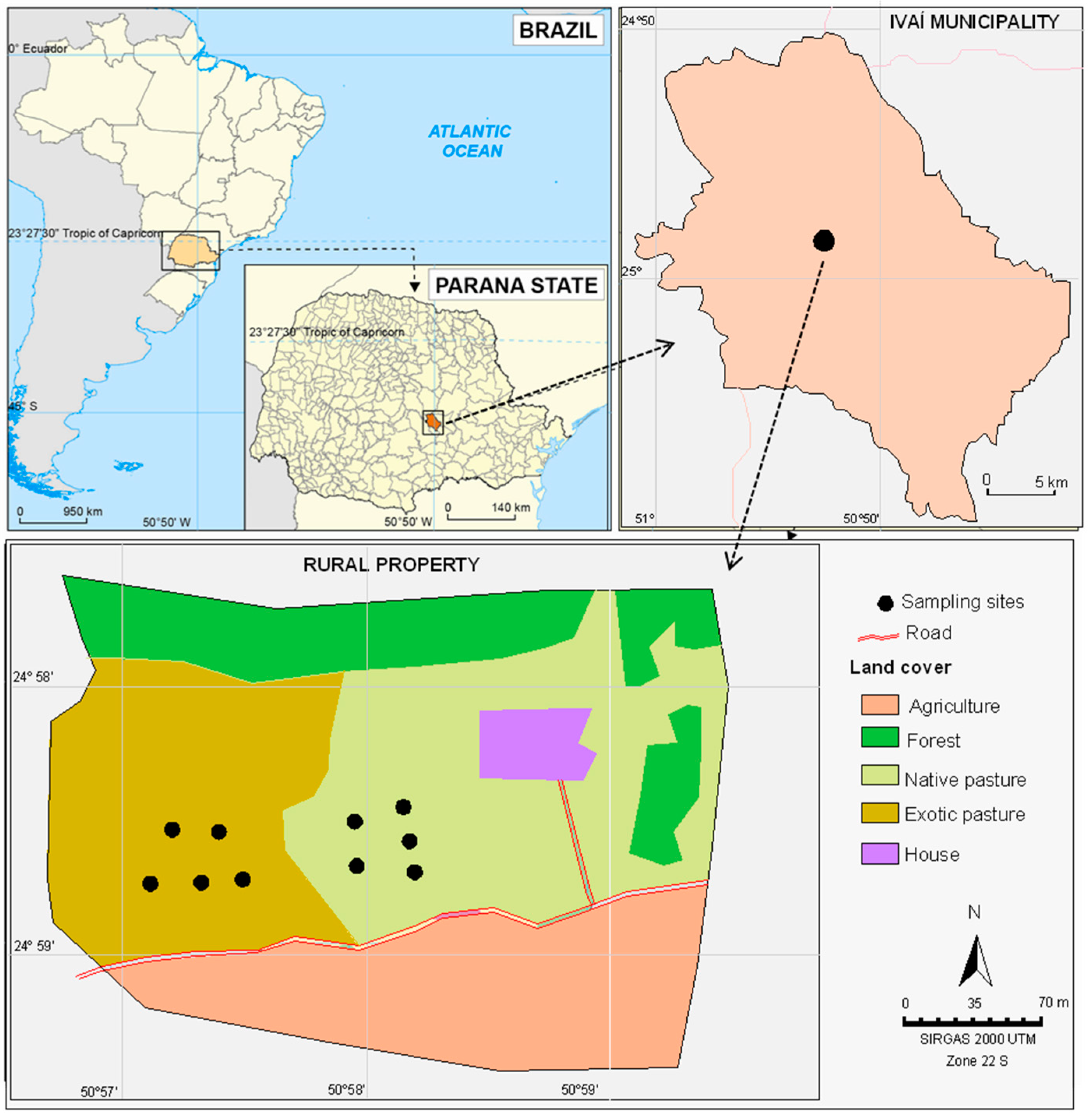
2.1. Study Area
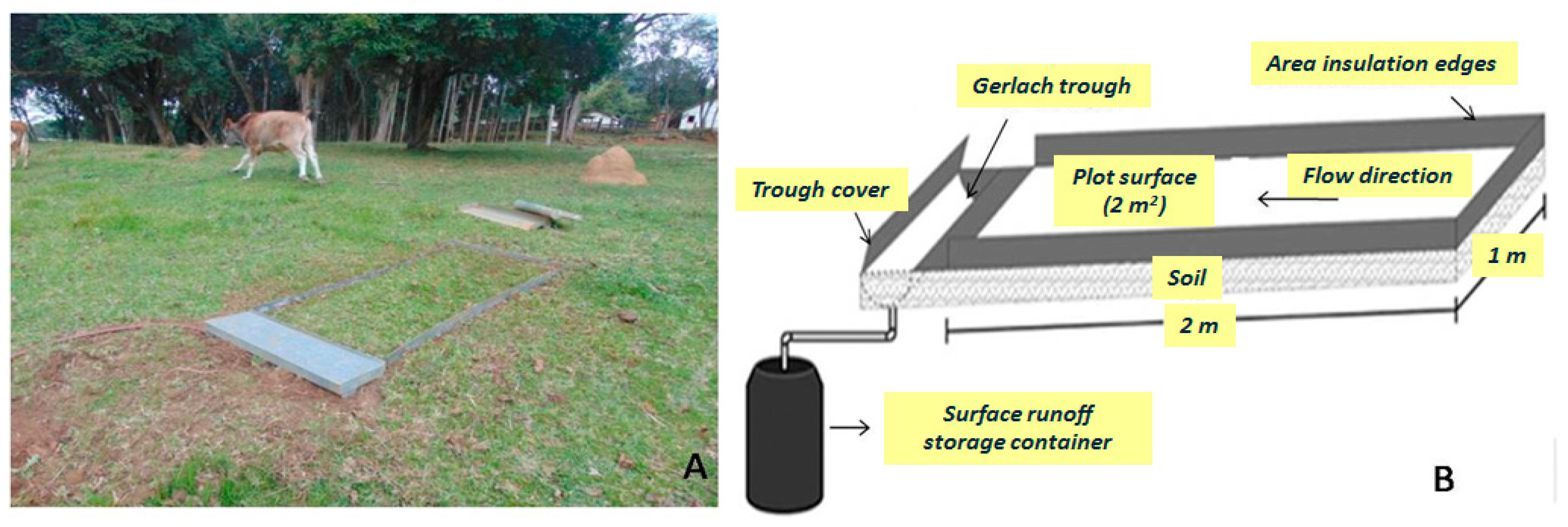
2.2. Sampling Design and Analysis
2.3. Data Analysis
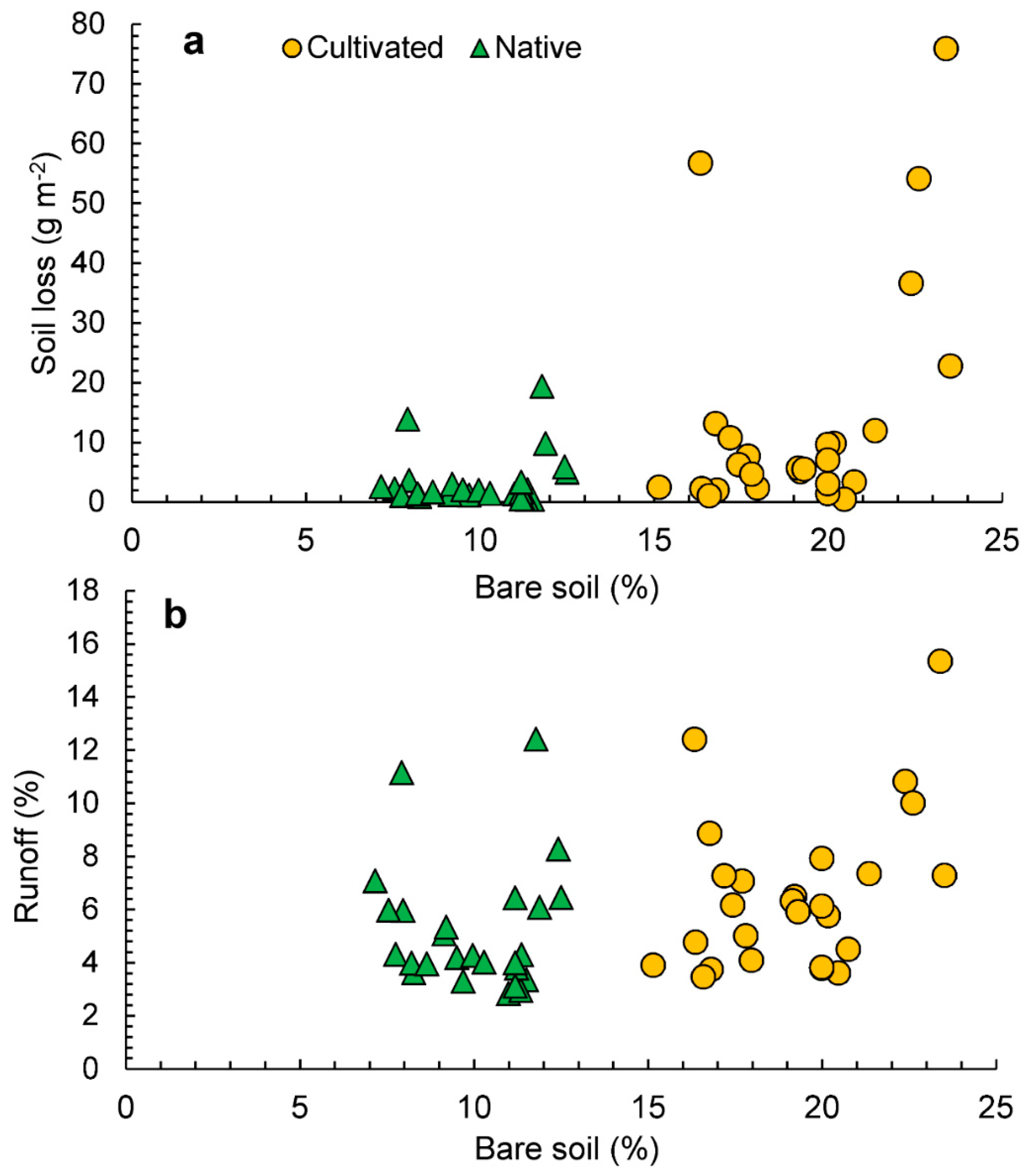
3. Results
3.1. Soil Properties and Pasture Production
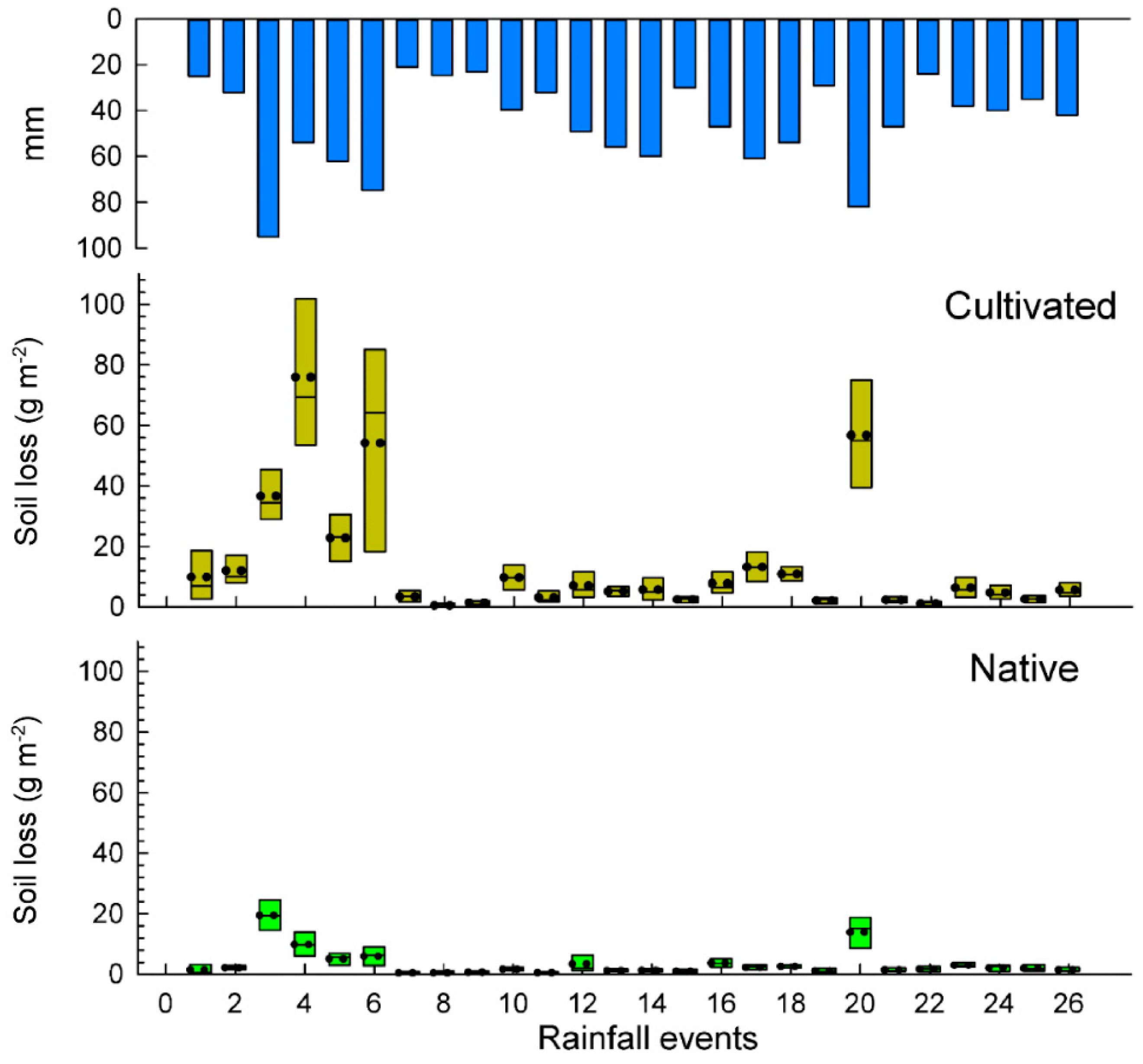
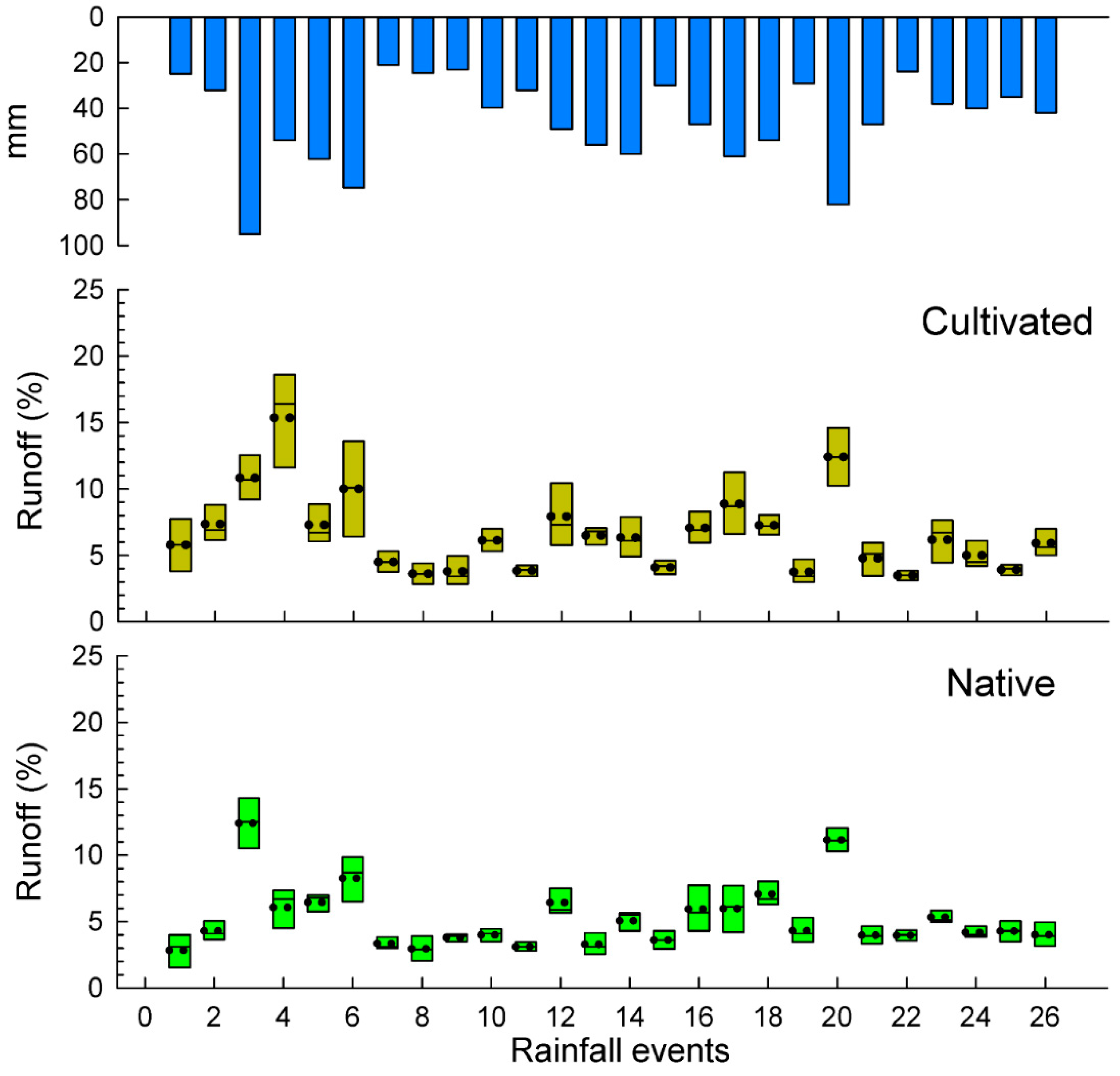
3.2. Soil and Water Losses
3.3. Bare Soil and Penetration Resistance
3.4. Relationships between Variables
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Campigoto, J.A.; Bona, A.N. A Hermenêutica e a origem dos faxinais. Revista de História Regional 2010, 14, 127–153. [Google Scholar] [CrossRef]
- Gonçalves Cunha, L.A. Desenvolvimento Rural e Desenvolvimento Territorial: O Caso do Paraná Tradicional. Ph.D. Thesis, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil, 2003. [Google Scholar]
- Watzlawick, L.F.; de Albuquerque, J.M.; Redin, C.G.; Longhi, R.V.; Longhi, S.J. Estrutura, diversidade e distribuição espacial da vegetação arbórea na Floresta Ombrófila Mista em Sistema Faxinal, Rebouças (PR). Ambiência 2011, 7, 415–427. [Google Scholar] [CrossRef][Green Version]
- Turner, B.L.; Tidwell, V.; Fernald, A.; Rivera, J.A.; Rodriguez, S.; Guldan, S.; Ochoa, C.; Hurd, B.; Boykin, K.; Cibils, A. Modeling acequia irrigation systems using system dynamics: Model development, evaluation, and sensitivity analyses to investigate effects of socio-economic and biophysical feedbacks. Sustainability 2016, 8, 1019. [Google Scholar] [CrossRef]
- Gunda, T.; Turner, B.; Tidwell, V.C. The influential role of sociocultural feedbacks on vommunity-managed irrigation system behaviors during times of water stress. Water Resour. Res. 2018, 54, 1–18. [Google Scholar]
- Chang, M.Y. Sistema Faxinal: Uma Forma de Organização Camponesa em Desagregação no Centro-sul do Paraná; Boletim Técnico IAPAR nº22: Londrina, Brazil1, 1988; p. 124. [Google Scholar]
- Thomaz, E.L.; Antoneli, V. Rain interception in a secondary fragment of araucaria forest with Faxinal, Guarapuava-PR. CERNE 2015, 21, 363–369. [Google Scholar] [CrossRef]
- Galvão Leite, D.M.; Cherumbim, A.A. Caracterização da criação animal em Sistema Faxinal. Rev. Bras. Agroecol. 2009, 4, 3959–3962. [Google Scholar]
- Martinkoski, L.; Vogel, G.F.; Jadoski, S.O.; Watzlawick, L.F. Soil physical quality under silvopastoral management and secondary forest. Floresta Ambiente 2017, 24, e20160282. [Google Scholar]
- Loch, D. Brachiaria decumbens (Signal grass)—A review with particular reference to Australia. Trop. Grassl. 1977, 11, 141–157. [Google Scholar]
- Wilson, J.R.; Hill, K.; Cameron, D.; Shelton, H.M. The growth of Paspalum notatum under the shade of a Eucalyptus grandis plantation canopy or in full sun. Trop. Grassl. 1990, 24, 24–28. [Google Scholar]
- Martha Júnior, G.B.; Corsi, M. Pastagens no Brasil: Situação atual e perspectivas. Preços Agrícolas 2001, 15, 3–6. [Google Scholar]
- Zhou, Z.; Gan, Z.; Shangguan, Z.; Dong, Z. Effects of grazing on soil physical properties and soil erodibility in semiarid grassland of the Northern Loess Plateau (China). CATENA 2010, 82, 87–91. [Google Scholar] [CrossRef]
- Pulido, M.; Schnabel, S.; Lavado Contador, J.F.; Lozano-Parra, J.; González, F. The impact of heavy grazing on soil quality and pasture production in rangelands of SW Spain. Land Degrad. Dev. 2018, 29, 219–230. [Google Scholar] [CrossRef]
- Vadas, P.A.; Busch, D.L.; Powell, J.M.; Brink, G.E. Monitoring runoff from cattle-grazed pastures for a phosphorus loss quantification tool. Agric. Ecosyst. Environ. 2015, 199, 124–131. [Google Scholar] [CrossRef]
- Pulido-Fernández, M.; Schnabel, S.; Lavado-Contador, J.F.; Miralles Mellado, I.; Ortega Pérez, R. Soil organic matter of Iberian open woodland rangelands as influenced by vegetation cover and land management. CATENA 2013, 109, 13–24. [Google Scholar] [CrossRef]
- Teague, W.; Dowhower, S.; Baker, S.; Ansley, R.; Kreuter, U.; Conover, D.; Waggoner, J. Soil and herbaceous plant responses to summer patch burns under continuous and rotational grazing. Agric. Ecosyst. Environ. 2010, 137, 113–123. [Google Scholar] [CrossRef]
- Van Oudenhoven, A.P.; Veerkamp, C.J.; Alkemade, R.; Leemans, R. Effects of different management regimes on soil erosion and surface runoff in semi-arid to sub-humid rangelands. J. Arid Environ. 2015, 121, 100–111. [Google Scholar] [CrossRef]
- Da Rocha Junior, P.R.; Andrade, F.V.; de Sá Mendonça, E.; Donagemma, G.K.; Fernandes, R.B.A.; Bhattharai, R.; Kalita, P.K. Soil, water, and nutrient losses from management alternatives for degraded pasture in Brazilian Atlantic Rainforest biome. Sci. Total Environ. 2017, 583, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.L.; Fuhrer, J.; Wuellner, M.; Menendez, H.M.; Dunn, B.H.; Gates, R. Scientific case studies in land-use driven soil erosion in the central United States: Why soil potential and risk concepts should be included in the principles of soil health. Int. Soil Water Conserv. Res. 2018, 6, 63–78. [Google Scholar] [CrossRef]
- Antoneli, V.; Thomaz, E.L. Comparação de infiltração de água no solo mensurada em período seco e úmido, em diferentes usos da terra na bacia do arroio Boa Vista, Guamiranga, Paraná. Ambiência 2009, 5, 301–318. [Google Scholar]
- Gerlach, T. Hillslope troughs for measuring sediment movement. Révue Géomorphologie Dynamique 1967, 4, 173. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Guidelines for Soil Description, 4th ed.; FAO: Rome, Italy, 2006; p. 97. [Google Scholar]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015; p. 203. [Google Scholar]
- Bouyoucos, G.J. Hydrometer method improved for making particle size analyses of soils. Agron. J. 1962, 54, 464–465. [Google Scholar] [CrossRef]
- Walkley, A.; Black, L.A. An examination of Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Statsoft. STATISTICA (Data Analysis Software System), Version 6. 2001. Available online: www.statsoft.com (accessed on 7 May 2018).
- Neill, C.; Davidson, E. Soil carbon accumulation or loss following deforestation for pasture in the Brazilian Amazon. In Global Climate Change and Tropical Ecosystems; Lal, R., Kimble, J.M., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 2000; pp. 197–211. [Google Scholar]
- Ye, C.; Guo, Z.; Li, Z.; Cai, C. The effect of Bahiagrass roots on soil erosion resistance of Aquults in subtropical China. Geomorphology 2017, 285, 82–93. [Google Scholar] [CrossRef]
- Agharkar, M.; Lomba, P.; Altpeter, F.; Zhang, H.; Kenworthy, K.; Lange, T. Stable expression of AtGA2ox1 in a low-input turfgrass (Paspalum notatum Flugge) reduces bioactive gibberellin levels and improves turf quality under field conditions. Plant Biotechnol. J. 2007, 5, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Panagos, P.; Borrelli, P.; Meusburger, K.; Alewell, C.; Lugato, E.; Montanarella, L. Estimating the soil erosion cover-management factor at the European scale. Land Use Policy 2015, 48, 38–50. [Google Scholar] [CrossRef]
- Stavi, I.; Ungar, E.D.; Lavee, H.; Sarah, P. Grazing-induced spatial variability of soil bulk density and content of moisture, organic carbon and calcium carbonate in a semi-arid rangeland. CATENA 2008, 75, 288–296. [Google Scholar] [CrossRef]
- da Silva, A.P.; Imhoff, S.; Corsi, M. Evaluation of soil compaction in an irrigated short-duration grazing system. Soil Tillage Res. 2003, 70, 83–90. [Google Scholar] [CrossRef]
- Bartley, R.; Roth, C.H.; Ludwig, J.; McJanett, D.; Liedloff, A.; Corfield, J.; Hawdon, A.; Abbott, B. Runoff and erosion from Australia’s tropical semi-arid rangelands: Influence of ground cover for differing space and time scales. Hydrol. Process. 2006, 20, 3317–3333. [Google Scholar] [CrossRef]
- Mulholland, B.; Fullen, M.A. Cattle trampling and soil compaction on loamy sands. Soil Use Manag. 1991, 7, 189–193. [Google Scholar] [CrossRef]
- Evans, R. Soil erosion in the UK initiated by grazing animals. Geography 1997, 17, 127–141. [Google Scholar] [CrossRef]
- Bilotta, G.S.; Brazier, R.E.; Haygarth, P.M. The impacts of grazing animals on the quality of soils, vegetation, and surface waters in intensively managed grasslands. Adv. Agron. 2007, 94, 237–280. [Google Scholar]
- Pulido, M.; Schnabel, S.; Contador, J.F.L.; Lozano-Parra, J.; Gómez-Gutiérrez, Á.; Brevik, E.C.; Cerdà, A. Reduction of the frequency of herbaceous roots as an effect of soil compaction induced by heavy grazing in rangelands of SW Spain. CATENA 2017, 158, 381–389. [Google Scholar] [CrossRef]
- Pulido, M.; Schnabel, S.; Contador, J.F.L.; Lozano-Parra, J.; Gómez-Gutiérrez, Á. Selecting indicators for assessing soil quality and degradation in rangelands of Extremadura (SW Spain). Ecol. Indic. 2017, 74, 49–61. [Google Scholar] [CrossRef]
| N | Rainfall Event | Rainfall Amount (mm) | N | Rainfall Event | Rainfall Amount (mm) |
|---|---|---|---|---|---|
| 1 | 24/07/2015 | 25.0 | 14 | 27/12/2015 | 60.0 |
| 2 | 13/08/2015 | 32.0 | 15 | 04/01/2016 | 30.0 |
| 3 | 04/09/2015 | 95.0 | 16 | 10/01/2016 | 47.0 |
| 4 | 09/09/2015 | 54.0 | 17 | 11/02/2016 | 61.0 |
| 5 | 27/09/2015 | 62.1 | 18 | 27/02/2016 | 54.0 |
| 6 | 22/10/2015 | 74.8 | 19 | 21/03/2016 | 29.0 |
| 7 | 01/11/2015 | 21.0 | 20 | 02/04/2016 | 82.0 |
| 8 | 05/11/2015 | 24.5 | 21 | 14/04/2016 | 47.0 |
| 9 | 14/11/2015 | 23.0 | 22 | 25/04/2016 | 24.0 |
| 10 | 16/11/2015 | 39.7 | 23 | 17/05/2016 | 38.0 |
| 11 | 17/11/2015 | 32.0 | 24 | 21/05/2016 | 40.0 |
| 12 | 19/11/2015 | 49.0 | 25 | 10/06/2016 | 35.0 |
| 13 | 19/12/2015 | 56.0 | 26 | 26/06/2016 | 42.0 |
| Variable | Native Pastures | Exotic Pastures |
|---|---|---|
| Slope (%) | 18 | 20 |
| Soil type [24] | Cambisol | Cambisol |
| Sand (%) | 36 | 34 |
| Silt (%) | 30 | 28 |
| Clay (%) | 34 | 38 |
| SOM (g kg−1) | 6.3 | 4.8 |
| Bulk density (g cm−3) | 1.29 ± 0.21a | 1.34 ± 0.19a |
| Porosity (%) | 51 ± 2.16a | 48 ± 1.97a |
| Pasture production (kg DM ha−1) | 204 ± 98b | 987 ± 201a |
| Land Use | Parameters | Runoff (L m−2) | Rc (%) | SC (g L−1) | Soil loss (g m−2) |
|---|---|---|---|---|---|
| Native pastures | Mean | 72.1 | 5.2 | 1.2 | 90.7 |
| SD | 2.6 | 2.4 | 0.6 | 4.5 | |
| Max | 11.8 | 12.4 | 3.0 | 19.5 | |
| Min | 0.7 | 2.8 | 0.4 | 0.5 | |
| CV | 7.0 | 5.7 | 0.3 | 19.8 | |
| Exotic pastures | Mean | 88.6 | 6.6 | 3.2 | 364.7 |
| SD | 2.8 | 2.9 | 2.1 | 19.6 | |
| Max | 10.3 | 15.4 | 9.2 | 76.0 | |
| Min | 0.8 | 3.5 | 0.7 | 0.6 | |
| CV | 7.8 | 8.7 | 4.5 | 385.8 | |
| Significant differences (Tukey test) | p < 0.036 | p < 0.001 | p < 0.001 | ||
| Land Use | Parameters | Bare Soil (%) | SPR (MPa) |
|---|---|---|---|
| Native pastures | Mean | 9.99 | 2.73 |
| SD | 1.66 | 0.32 | |
| Max | 12.50 | 3.50 | |
| Min | 7.16 | 2.40 | |
| CV | 16.63 | 0.10 | |
| Exotic pastures | Mean | 19.16 | 3.25 |
| SD | 2.31 | 0.25 | |
| Max | 23.50 | 3.70 | |
| Min | 15.14 | 2.90 | |
| CV | 12.08 | 0.06 |
| Native Pastures | |||||
| Rainfall | Bare soil | Soil loss | Water loss | Penetration resistance | |
| Rainfall | 1.000 | ||||
| Bare soil | 0.011 | 1.000 | |||
| Soil loss | 0.795 *** | 0.169 | 1.000 | ||
| Water loss | 0.884 *** | −0.001 | 0.909 *** | 1.000 | |
| Penetration resistance | 0.267 | 0.450 * | 0.532 ** | 0.303 | 1.000 |
| Exotic Pastures | |||||
| Rainfall | Bare soil | Soil loss | Water loss | Penetration resistance | |
| Rainfall | 1.000 | ||||
| Bare soil | 0.209 | 1.000 | |||
| Soil loss | 0.664 *** | 0.452 * | 1.000 | ||
| Water loss | 0.756 *** | 0.397 * | 0.919 *** | 1.000 | |
| Penetration resistance | 0.097 | 0.879 *** | 0.357 | 0.271 | 1.000 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antoneli, V.; Rebinski, E.A.; Bednarz, J.A.; Rodrigo-Comino, J.; Keesstra, S.D.; Cerdà, A.; Pulido Fernández, M. Soil Erosion Induced by the Introduction of New Pasture Species in a Faxinal Farm of Southern Brazil. Geosciences 2018, 8, 166. https://doi.org/10.3390/geosciences8050166
Antoneli V, Rebinski EA, Bednarz JA, Rodrigo-Comino J, Keesstra SD, Cerdà A, Pulido Fernández M. Soil Erosion Induced by the Introduction of New Pasture Species in a Faxinal Farm of Southern Brazil. Geosciences. 2018; 8(5):166. https://doi.org/10.3390/geosciences8050166
Chicago/Turabian StyleAntoneli, Valdemir, Everson A. Rebinski, João Anesio Bednarz, Jesús Rodrigo-Comino, Saskia Deborah Keesstra, Artemi Cerdà, and Manuel Pulido Fernández. 2018. "Soil Erosion Induced by the Introduction of New Pasture Species in a Faxinal Farm of Southern Brazil" Geosciences 8, no. 5: 166. https://doi.org/10.3390/geosciences8050166
APA StyleAntoneli, V., Rebinski, E. A., Bednarz, J. A., Rodrigo-Comino, J., Keesstra, S. D., Cerdà, A., & Pulido Fernández, M. (2018). Soil Erosion Induced by the Introduction of New Pasture Species in a Faxinal Farm of Southern Brazil. Geosciences, 8(5), 166. https://doi.org/10.3390/geosciences8050166










