Phase Evolution and Textural Changes during the Direct Conversion and Storage of CO2 to Produce Calcium Carbonate from Calcium Hydroxide
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. USAXS/SAXS/WAXS Measurements
3. Results
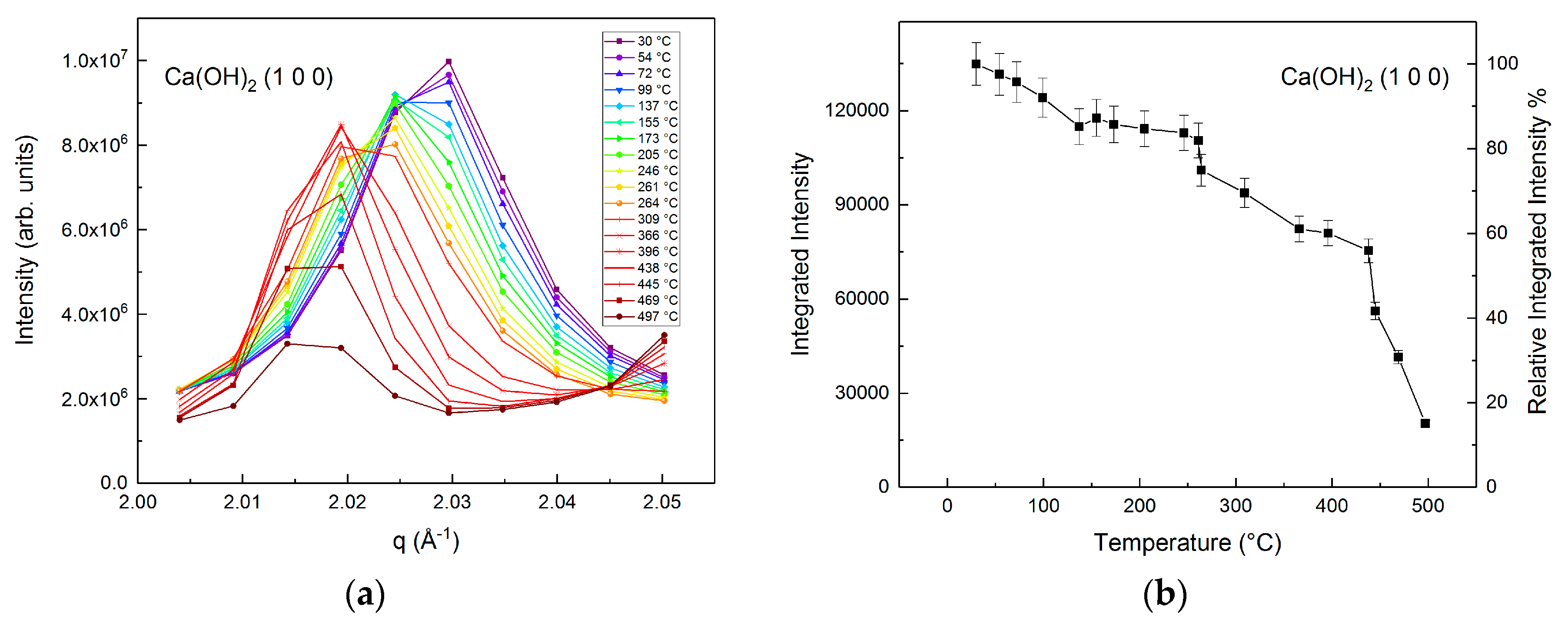
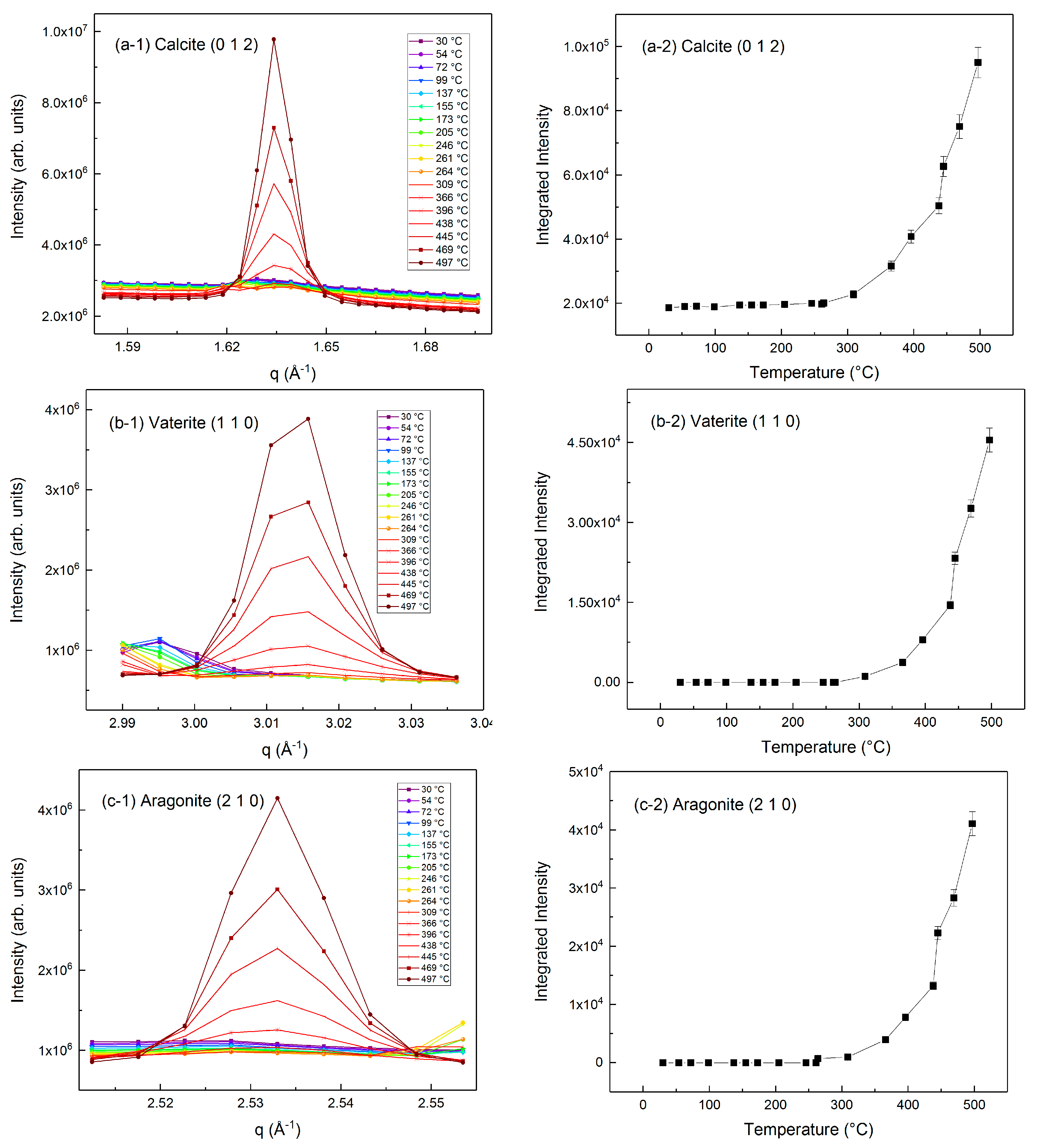
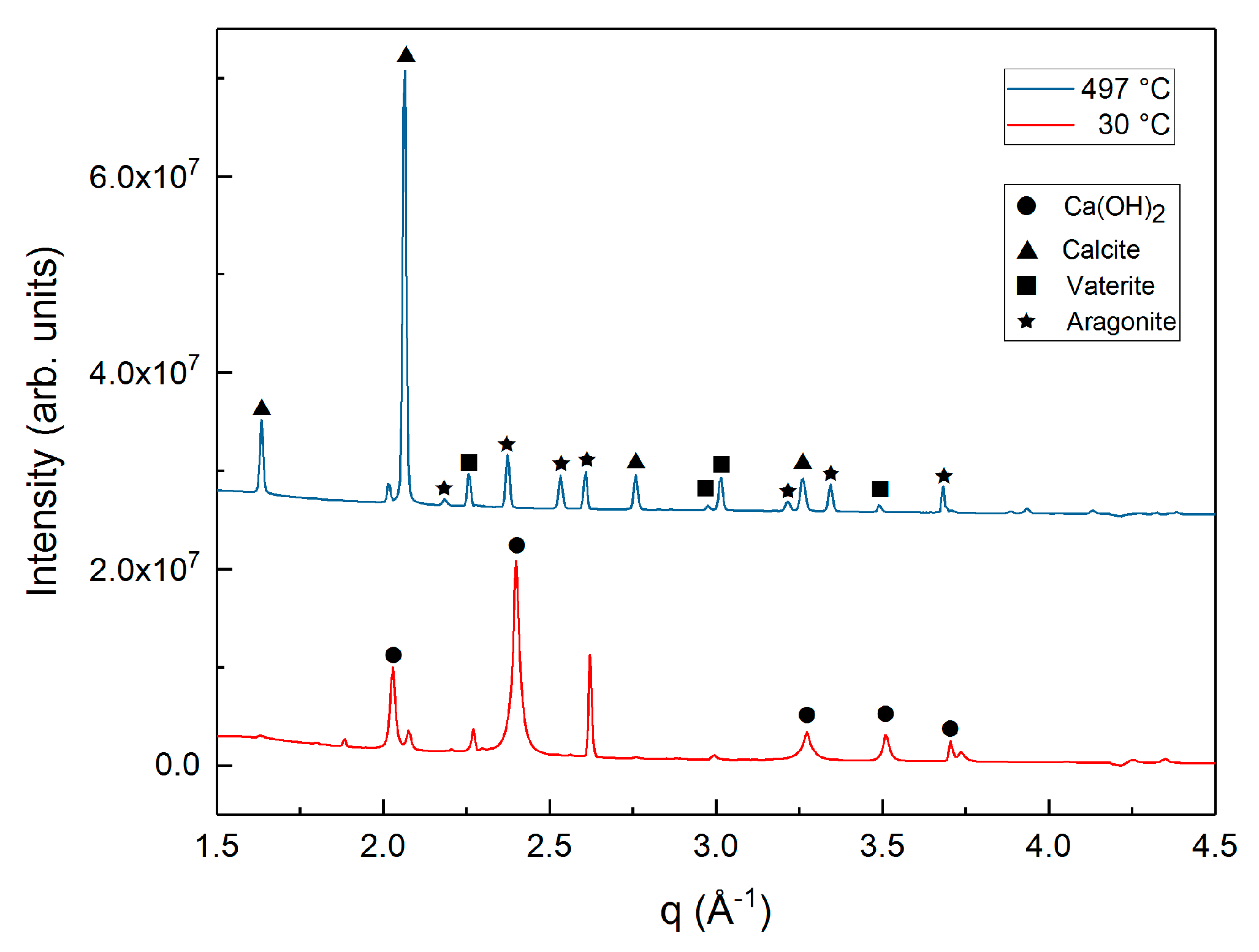
3.1. Phase Transformations during the Carbon Mineralization of Calcium Hydroxide
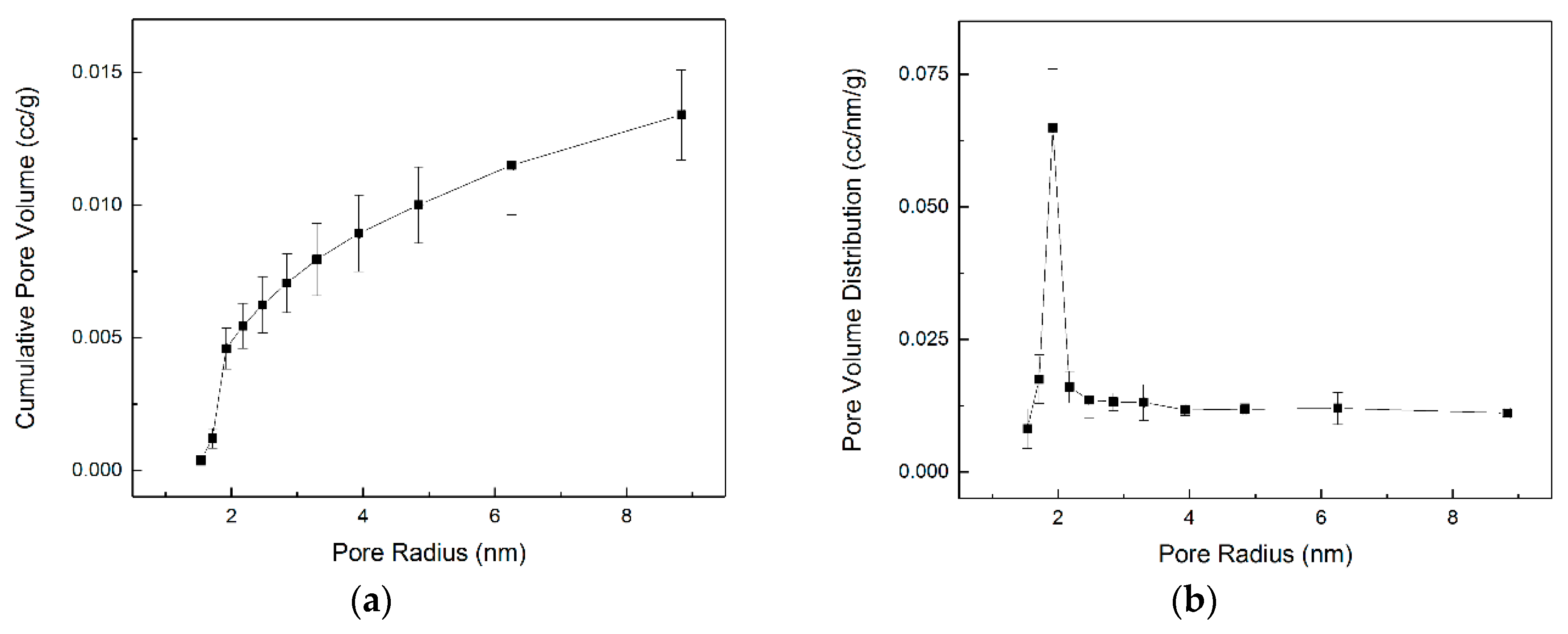
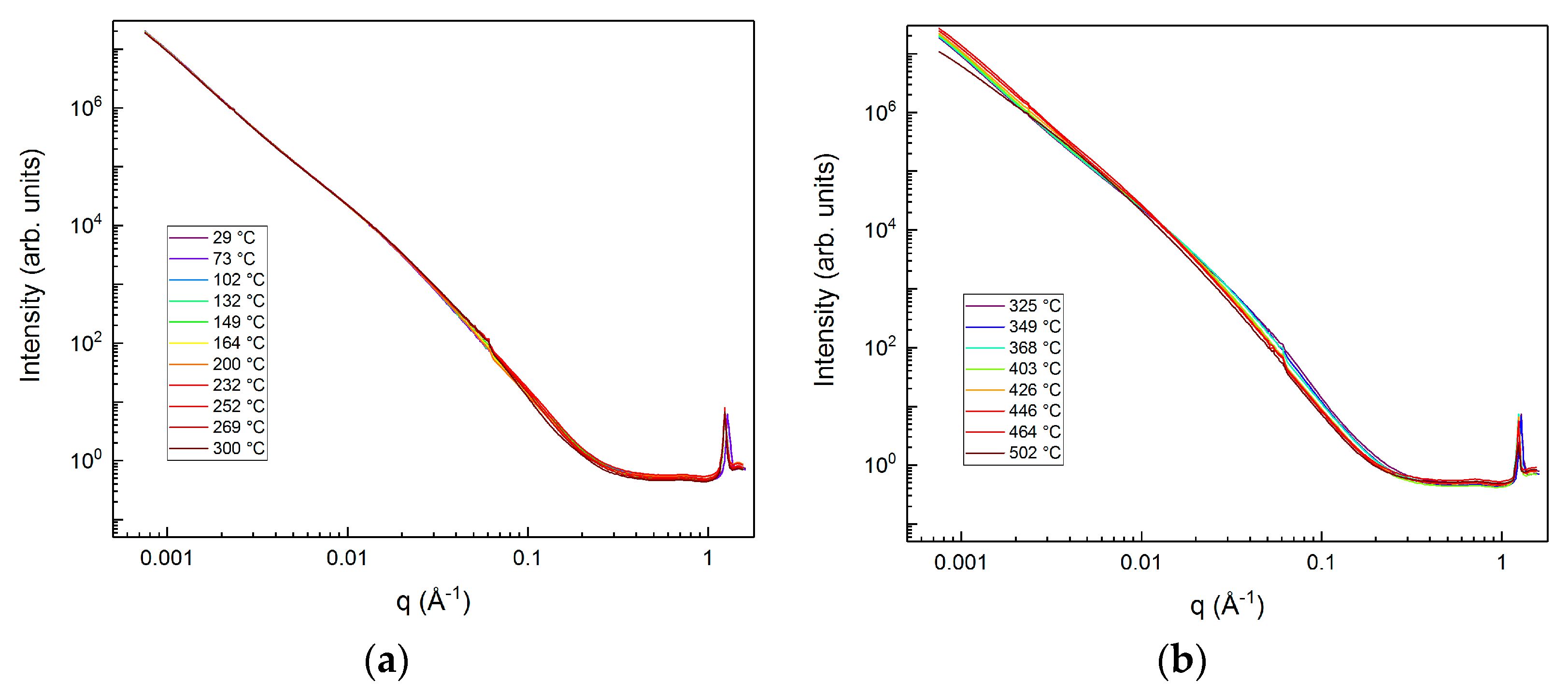
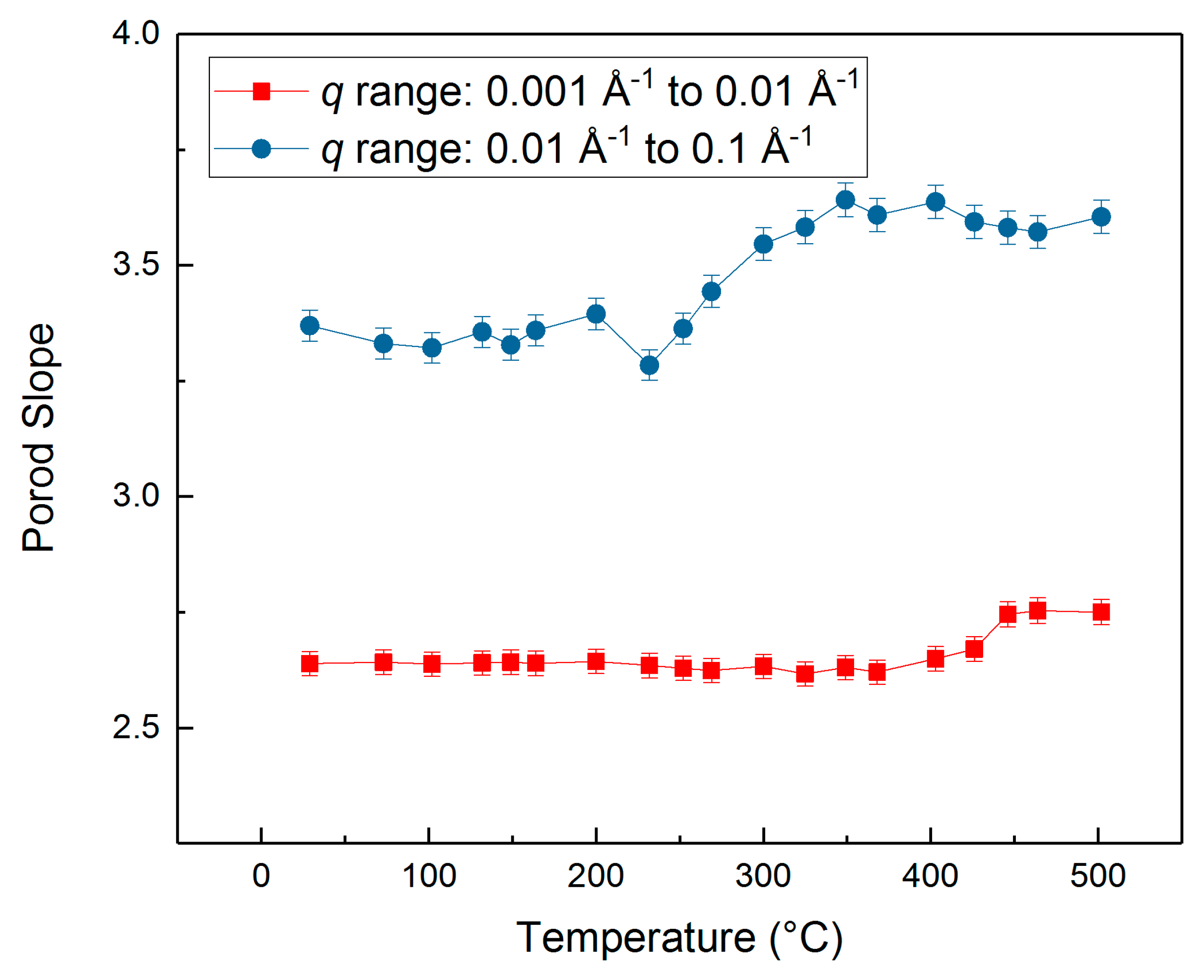
3.2. Evolution of the Pore–solid Interface during the Carbon Mineralization of Calcium Hydroxide
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fauth, D.J.; Goldberg, P.M.; Knoer, J.P.; Soong, Y.; O’Connor, W.K.; Dahlin, D.C.; Nilsen, D.N.; Walters, R.P.; Lackner, K.S.; Ziock, H.J.; et al. Carbon dioxide storage as mineral carbonates. ACS Div. Fuel Chem. Prepr. 2000, 45, 708–711. [Google Scholar]
- Lackner, K.S. Carbonate chemistry for sequestering fossil carbon. Annu. Rev. Energy Environ. 2002, 27, 193–232. [Google Scholar] [CrossRef]
- Seifritz, W. CO2 disposal by means of silicates. Nature 1990, 345, 486. [Google Scholar] [CrossRef]
- Metz, B. IPCC Special Report on Carbon Dioxide Capture and Storage; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Mission Innovation Carbon Capture, Utilization, and Storage (CCUS) Expert’s Workshop. Accelerating Breakthrough Innovation in Carbon Capture, Utilization, and Storage. Available online: https://www.energy.gov/fe/downloads/accelerating-breakthrough-innovation-carbon-capture-utilization-and-storage (accessed on 19 October 2018).
- Gadikota, G.; Matter, J.; Kelemen, P.; Park, A.-H.A. Chemical and morphological changes during olivine carbonation for CO2 storage in the presence of NaCl and NaHCO3. Phys. Chem. Chem. Phys. 2014, 16, 4679–4693. [Google Scholar] [CrossRef] [PubMed]
- Gadikota, G.; Swanson, E.; Zhao, H.; Park, A.-H.A. Experimental Design and Data Analysis for Accurate Estimation of Reaction Kinetics and Conversion for Carbon Mineralization. Ind. Eng. Chem. Res. 2014, 53, 6664–6676. [Google Scholar] [CrossRef]
- Gadikota, G.; Park, A.-H.A. Accelerated Carbonation of Ca- and Mg-Bearing Minerals and Industrial Wastes Using CO2. In Carbon Dioxide Utilisation: Closing the Carbon Cycle; Elsevier: Amsterdam, The Netherlands, 2015; pp. 115–137. [Google Scholar]
- Gadikota, G.; Fricker, K.J.; Jang, S.-H.; Park, A.-H.A. Carbonated silicate minerals and industrial wastes as construction materials. In Advances in CO2 Capture, Sequestration, and Conversion; American Chemical Society: Washington, DC, USA, 2015; pp. 295–322. [Google Scholar]
- Liu, M.; Gadikota, G. Chemo-morphological coupling during serpentine heat treatment for carbon mineralization. Fuel 2018, 227, 379–385. [Google Scholar] [CrossRef]
- Liu, M.; Gadikota, G. Integrated CO2 capture, conversion and storage to produce calcium carbonate using an amine looping strategy. Energy Fuels 2018. [Google Scholar] [CrossRef]
- Power, I.M.; Harrison, A.L.; Dipple, G.M.; Wilson, S.A.; Kelemen, P.B.; Hitch, M.; Southam, G. Carbon mineralization: From natural analogues to engineered systems. Rev. Mineral. Geochem. 2013, 77, 305–360. [Google Scholar] [CrossRef]
- Sanna, A.; Hall, M.R.; Maroto-Valer, M. Post-processing pathways in carbon capture and storage by mineral carbonation (CCSM) towards the introduction of carbon neutral materials. Energy Environ. Sci. 2012, 5, 7781–7796. [Google Scholar] [CrossRef]
- Gerdemann, S.J.; O’Connor, W.K.; Dahlin, D.C.; Penner, L.R.; Rush, H. Ex situ aqueous mineral carbonation. Environ. Sci. Technol. 2007, 41, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- McKelvy, M.J.; Chizmeshya, A.V.G.; Diefenbacher, J.; Béarat, H.; Wolf, G. Exploration of the role of heat activation in enhancing serpentine carbon sequestration reactions. Environ. Sci. Technol. 2004, 38, 6897–6903. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Yang, J.; Xiao, B. Review on treatment and utilization of bauxite residues in China. Int. J. Miner. Process. 2009, 93, 220–231. [Google Scholar] [CrossRef]
- Li, Y.; Sun, R.; Liu, C.; Liu, H.; Lu, C. CO2 capture by carbide slag from chlor-alkali plant in calcination/carbonation cycles. Int. J. Greenh. Gas Control 2012, 9, 117–123. [Google Scholar] [CrossRef]
- Cheng, J.; Zhou, J.; Liu, J.; Cao, X.; Cen, K. Physicochemical characterizations and desulfurization properties in coal combustion of three calcium and sodium industrial wastes. Energy Fuels 2009, 23, 2506–2516. [Google Scholar] [CrossRef]
- Cao, J.; Liu, F.; Lin, Q.; Zhang, Y. Hydrothermal synthesis of xonotlite from carbide slag. Prog. Nat. Sci. 2008, 18, 1147–1154. [Google Scholar] [CrossRef]
- Bobicki, E.R.; Liu, Q.; Xu, Z.; Zeng, H. Carbon capture and storage using alkaline industrial wastes. Prog. Energy Combust. Sci. 2012, 38, 302–320. [Google Scholar] [CrossRef]
- Li, B.; Duan, Y.; Luebke, D.; Morreale, B. Advances in CO2 capture technology: A patent review. Appl. Energy 2013, 102, 1439–1447. [Google Scholar] [CrossRef]
- Blamey, J.; Anthony, E.J.; Wang, J.; Fennell, P.S. The calcium looping cycle for large-scale CO2 capture. Prog. Energy Combust. Sci. 2010, 36, 260–279. [Google Scholar] [CrossRef]
- Liu, W.; An, H.; Qin, C.; Yin, J.; Wang, G.; Feng, B.; Xu, M. Performance enhancement of calcium oxide sorbents for cyclic CO2 capture—A review. Energy Fuels 2012, 26, 2751–2767. [Google Scholar] [CrossRef]
- Valverde, J.M. Ca-based synthetic materials with enhanced CO2 capture efficiency. J. Mater. Chem. A 2013, 1, 447–468. [Google Scholar] [CrossRef]
- Manovic, V.; Anthony, E.J. Lime-based sorbents for high-temperature CO2 capture-a review of sorbent modification methods. Int. J. Environ. Res. Public Health 2010, 7, 3129–3140. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Su, M.; Xie, X.; Wu, S.; Liu, C. CO2 capture performance of synthetic sorbent prepared from carbide slag and aluminum nitrate hydrate by combustion synthesis. Appl. Energy 2015, 145, 60–68. [Google Scholar] [CrossRef]
- Sun, R.; Li, Y.; Zhao, J.; Liu, C.; Lu, C. CO2 capture using carbide slag modified by propionic acid in calcium looping process for hydrogen production. Int. J. Hydrog. Energy 2013, 38, 13655–13663. [Google Scholar] [CrossRef]
- Gadikota, G.; Zhang, F.; Allen, A.J. Towards understanding the microstructural and structural changes in natural hierarchical materials for energy recovery: In-operando multiscale X-ray scattering characterization of Na- and Ca-montmorillonite on heating to 1150 °C. Fuel 2017, 196, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Gadikota, G.; Zhang, F.; Allen, A. In Situ angstrom-to-micrometer characterization of the structural and microstructural changes in kaolinite on heating using ultra small-angle, small-angle, and wide-angle X-ray scattering (USAXS/SAXS/WAXS). Ind. Eng. Chem. Res. 2017, 56, 11791–11801. [Google Scholar] [CrossRef]
- Gadikota, G.; Allen, A.J. Microstructural and structural characterization of materials for CO2 storage using multiscale X-ray scattering methods. In Materials and Processes for CO2 Capture, Conversion, and Sequestration; John Wiley & Sons: Hoboken, NJ, USA, 2017; Volume 27, pp. 296–318. [Google Scholar]
- Anovitz, L.; Cole, D. Characterization and analysis of porosity and pore structures. Rev. Mineral. Geochem. 2015, 80, 61–164. [Google Scholar] [CrossRef]
- Ilavsky, J.; Zhang, F.; Andrews, R.N.; Kuzmenko, I.; Jemian, P.R.; Levine, L.E.; Allen, A.J. Development of combined microstructure and structure characterization facility for in situ and operando studies at the advanced photon source. J. Appl. Crystallogr. 2018, 51, 867–882. [Google Scholar] [CrossRef]
- Benedetti, A.; Ilavsky, J.; Segre, C.; Strumendo, M. Analysis of textural properties of CaO-based CO2 sorbents by ex situ USAXS. Chem. Eng. J. 2019, 355, 760–776. [Google Scholar] [CrossRef]
- Chupas, P.J.; Chapman, K.W.; Kurtz, C.; Hanson, J.C.; Lee, P.L.; Grey, C.P. A versatile sample-environment cell for non-ambient X-ray scattering experiments. J. Appl. Crystallogr. 2008, 41, 822–824. [Google Scholar] [CrossRef]
- Ilavsky, J.; Jemian, P.R.; Allen, A.J.; Zhang, F.; Levine, L.E.; Long, G.G. Ultrasmall-angle X-ray scattering at the Advanced Photon Source. J. Appl. Crystallogr. 2009, 42, 469–479. [Google Scholar] [CrossRef]
- Ilavsky, J.; Zhang, F.; Allen, A.J.; Levine, L.E.; Jemian, P.R.; Long, G.G. Ultrasmall-angle X-ray scattering instrument at the advanced photon source: History, recent development, and current status. Metall. Mater. Trans. A 2013, 44, 68–76. [Google Scholar] [CrossRef]
- Black, D.R.; Windover, D.; Henins, A.; Gil, D.; Filliben, J.; Cline, J.P. Certification of NIST standard reference material 640d. Powder Diffr. 2010, 25, 187–190. [Google Scholar] [CrossRef]
- Ilavsky, J.; Jemian, P.R. Irena: Tool suite for modeling and analysis of small-angle scattering. J. Appl. Crystallogr. 2009, 42, 347–353. [Google Scholar] [CrossRef]
- Ilavsky, J. Nika: Software for two-dimensional data reduction. J. Appl. Crystallogr. 2012, 45, 324–328. [Google Scholar] [CrossRef]
- Petch, H.E. The hydrogen positions in portlandite, Ca(OH)2, as indicated by the electron distribution. Acta Crystallogr. 1961, 14, 950–957. [Google Scholar] [CrossRef]
- Gadikota, G.; Zhang, F.; Allen, A. In Situ Angstrom-to-Micrometer Characterization of the Structural and Microstructural Changes in Kaolinite on Heating Using Ultrasmall-Angle, Small-Angle, and Wide-Angle X-ray Scattering (USAXS/SAXS/WAXS). Ind. Eng. Chem. Res. 2017, 56, 11791–11801. [Google Scholar] [CrossRef]
- Materic, V.; Smedley, S.I. High Temperature Carbonation of Ca(OH)2. Ind. Eng. Chem. Res. 2011, 50, 5927–5932. [Google Scholar] [CrossRef]
- Nikulshina, V.; Gálvez, M.E.; Steinfeld, A. Kinetic analysis of the carbonation reactions for the capture of CO2 from air via the Ca(OH)2-CaCO3-CaO solar thermochemical cycle. Chem. Eng. J. 2007, 129, 75–83. [Google Scholar] [CrossRef]
- Kamhi, S.R. On the structure of vaterite, CaCO3. Acta Crystallogr. 1963, 16, 770–772. [Google Scholar] [CrossRef]
- Dal Negro, A.; Ungaretti, L. Refinement of the Crystal Structure of Aragonite. Am. Mineral. 1971, 56, 768–772. [Google Scholar]
- Chessin, H.; Hamilton, W.C.; Post, B. Position and thermal parameters of oxygen atoms in calcite. Acta Crystallogr. 1965, 18, 689–693. [Google Scholar] [CrossRef]
- Christy, A.G. A review of the structures of vaterite: the impossible, the possible, and the likely. Cryst. Growth Des. 2017, 17, 3567–3578. [Google Scholar] [CrossRef]
- Maslen, E.N.; Streltsov, V.A.; Streltsova, N.R. X-ray study of the electron density in calcite, CaCO3. Acta Crystallogr. Sect. B 1993, 49, 636–641. [Google Scholar] [CrossRef]
- Richards, R.; Li, W.; Decker, S.; Davidson, C.; Koper, O.; Zaikovski, V.; Volodin, A.; Rieker, T.; Klabunde, K.J. Consolidation of metal oxide nanocrystals. Reactive pellets with controllable pore structure that represent a new family of porous, inorganic materials. J. Am. Chem. Soc. 2000, 122, 4921–4925. [Google Scholar] [CrossRef]
- Liang, J.Z.; Duan, D.R.; Tang, C.Y.; Tsui, C.P. Correlation between impact strength and fractal dimensions of fracture surface for PLLA/nano-CaCO3 composites. Polym. Test. 2014, 35, 109–115. [Google Scholar] [CrossRef]
- Diamond, S. Aspects of concrete porosity revisited. Cem. Concr. Res. 1999, 29, 1181–1188. [Google Scholar] [CrossRef]
- Liu, M.; Gadikota, G. Probing the Influence of Thermally Induced Structural Changes on the Microstructural Evolution in Shale using Multiscale X-ray Scattering Measurements. Energy Fuels 2018, 32, 8193–8201. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Gadikota, G. Phase Evolution and Textural Changes during the Direct Conversion and Storage of CO2 to Produce Calcium Carbonate from Calcium Hydroxide. Geosciences 2018, 8, 445. https://doi.org/10.3390/geosciences8120445
Liu M, Gadikota G. Phase Evolution and Textural Changes during the Direct Conversion and Storage of CO2 to Produce Calcium Carbonate from Calcium Hydroxide. Geosciences. 2018; 8(12):445. https://doi.org/10.3390/geosciences8120445
Chicago/Turabian StyleLiu, Meishen, and Greeshma Gadikota. 2018. "Phase Evolution and Textural Changes during the Direct Conversion and Storage of CO2 to Produce Calcium Carbonate from Calcium Hydroxide" Geosciences 8, no. 12: 445. https://doi.org/10.3390/geosciences8120445
APA StyleLiu, M., & Gadikota, G. (2018). Phase Evolution and Textural Changes during the Direct Conversion and Storage of CO2 to Produce Calcium Carbonate from Calcium Hydroxide. Geosciences, 8(12), 445. https://doi.org/10.3390/geosciences8120445




