Rabbit Enteropathies on Commercial Farms in the Iberian Peninsula: Etiological Agents Identified in 2018–2019
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Cases, Samples, and Isolates Studied
2.2. Bacteriological Culture
2.3. Extraction of Total Nucleic Acids from Clinical Samples
2.4. Extraction of DNA from E. coli Isolates
2.5. Amplification of Nucleic Acids
2.6. Software Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Commission. Overview Report of the Directorate-General for Health and Food Safety on Commercial Farming of Rabbits on the European Union; Publications Office of the European Union: Luxembourg, 2017. [Google Scholar] [CrossRef]
- El Sector Cunícola en Cifras: Principales Indicadores Económicos. Subdirección General de Productos Ganaderos, Dirección General de Producciones y Mercados Agrarios. Available online: https://publicacionesoficiales.boe.es/detail.php?id=479000319-0001 (accessed on 15 October 2019). (In Spanish).
- Marlier, D.; Dewree, R.; Delleur, V.; Licois, D.; Lassence, C.; Poulipoulis, A.; Vindevogel, H. Description des principales étiologies des maladies digestives chez le lapin européen (Oryctolagus cuniculus). Ann. Méd. Vét. 2003, 147, 385–392. (In Spanish) [Google Scholar]
- Boucher, S.; Leplat, A. De quoi meurent les lapins d’engraissement. In Proceedings of the Table Ronde, 11emes Journees de la Recherche Cunicole, Paris, France, 29–30 November 2005; Association Scientifique Française de Cuniculture: Paris, France. Available online: http://www.asfc-lapin.com/Docs/Activite/T-ronde-2005/Tables-rondes-01a.htm (accessed on 15 October 2019). (In French).
- Pohl, P.H.; Peeters, J.E.; Jacquemin, E.R.; Lintermans, P.F.; Mainil, J.G. Identification of eae sequences in enteropathogenic Escherichia coli strains from rabbits. Infect. Immunity 1993, 61, 2203–2206. [Google Scholar]
- Blanco, J.E.; Blanco, M.; Blanco, J.; Mora, A.; Balaguer, L.; Mourin, M.; Juarez, A.; Jansen, W.H. O Serogroups, biotypes, and eae genes in Escherichia coli strains isolated from diarrheic and healthy rabbits. J. Clin. Microbiol. 1996, 34, 3101–3107. [Google Scholar] [PubMed]
- Boullier, S.; Milon, A. Rabbit Colibacillosis. In Recent Advances in Rabbit Sciences; Maertens, L., Coudert, P., Eds.; Plot-it bvba: Merelbeke, Belgium, 2006; pp. 171–180. [Google Scholar]
- Licois, D. Pathologie d′origine bactérienne et parasitaire chez le lapin: Aports de la dernière décennie. In Proceedings of the 13èmes Journées de la Recherche cunicole, Le Mans, France, 17–18 November 2009; INRA: Nouzilly, France (In French). [Google Scholar]
- Franzin, F.M.; Sircili, M.P. Locus of Enterocyte Effacement: A pathogenicity island involved in the virulence of enteropathogenic and enterohemorrhagic Escherichia coli subjected to a complex network of gene regulation. BioMed Res. Int. 2015, 2015, 10. [Google Scholar] [CrossRef]
- Dow, M.A.; Tóth, I.; Alexa, P.; Davies, M.; Malik, A.; Oswal, E.; Nagy, B. Predominance of afr2 and ral fimbrial genes related to those encoding the K88 and CS31A fimbrial adhesins in enteropathogenic Escherichia coli isolates from rabbits with postweaning diarrhea in Central Europe. J. Clin. Microbiol. 2005, 43, 1366–1371. [Google Scholar] [CrossRef]
- Carman, R.J.; Borriello, S.P. Infectious nature of Clostridium spiroforme-mediated rabbit enterotoxaemia. Vet. Microbiol. 1984, 9, 497–502. [Google Scholar] [CrossRef]
- Agnoletti, F.; Lenarduzzi, M.; Ricci, A.; Marotta, A. Isolation of Salmonella spp. from Italian commercial rabbitries. In Proceedings of the 2nd International Conference on rabbit production in hot climates, Adana, Turkey, 7–9 September 1998; Testik, A., Baselga, M., Eds.; CIHEAM-IAMZ: Zaragoza, Spain; pp. 189–193. [Google Scholar]
- Lavazza, A.; Cerioli, M.; Martella, V.; Tittarelli, C.; Grilli, G.; Brivio, R.; Buonavoglia, C. Rotavirus in diarrheic rabbits: Prevalence and characterization of strains in Italian farms. In Proceedings of the 9th World Rabbit Congress, Verona, Italy, 10–13 June 2008; pp. 993–997. Available online: https://world-rabbit-science.com/WRSA-Proceedings/Congress-2008-Verona/Papers/P-Lavazza.pdf (accessed on 15 October 2019).
- Garcia, J.P.; Li, J.; Shrestha, A.; Freedman, J.C.; Beingesser, J.; McClane, B.; Uzal, F.A. Clostridium perfringens Type A enterotoxin damages the rabbit colon. Infect. Immun. 2014, 82, 2211. [Google Scholar] [CrossRef]
- Vela, A.I.; Fernández, A.; Moreno, B.; Casamayor, A.; Chacón, G.; Villa, A.; Comenge, J.; Fernández-Garayzábal, J.F. Isolation of Enterococcus hirae from suckling rabbits with diarrhoea. Vet. Rec. 2010, 167, 345–346. [Google Scholar] [CrossRef]
- Malo, M. Bacteroides fragilis enteropatógeno: Enfermedad emergente o hallagzo clinico? In Proceedings of the XLIV 44th Symposium de Cunicultura de ASESCU, Aranda de Duero, Spain, 5–6 June 2019; pp. 83–86. Available online: https://asescu.com/wp-content/uploads/2019/06/L_Actas-44-Symposium-ASESCU-Aranda-Duero.pdf (accessed on 15 October 2019). (In Spanish).
- Heberle, H.; Meirelles, G.V.; da Silva, F.R.; Telles, G.P.; Minghim, R. InteractiVenn: A web-based tool for the analysis of sets through Venn diagrams. BMC Bioinf. 2015, 16, 169. [Google Scholar] [CrossRef]
- Agnoletti, F. Update on rabbit enteric diseases: Despite improved diagnostic capacity, where does disease control and prevention stand? In Proceedings of the 10th Word Rabbit Congress, Sharm El Sheikh, Egypt, 3–6 September 2012; WRSA ed.: Sharm El Sheikh, Egypt; pp. 1113–1127. [Google Scholar]
- Kylie, J.; Brash, M.; Whiteman, A.; Tapscott, B.; Slavic, D.; Weese, J.S.; Turner, V. Biosecurity practices and causes of enteritis on Ontario meat rabbit farms. Can. Vet. J. 2017, 58, 571–578. [Google Scholar]
- Peeters, J.E.; Maertens, L.; Orsenigo, R.; Colin, M. Influence of dietary beet pulp on caecal VFA, experimental colibacillosis and iota-enterotoxaemia in rabbits. Anim. Feed Sci. Tech. 1995, 51, 123–139. [Google Scholar] [CrossRef]
- Songer, J.G. Clostridial enteric diseases of domestic animals. Clin. Microbiol. Rev. 1996, 9, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Lelkes, I. A review of rabbit enteric diseases: A new perspective. J. Appl. Rabbit Res. 1987, 10, 55–61. [Google Scholar]
- Milon, A.; Oswald, E.; De Rycke, J. Rabbit EPEC: A model for the study of enteropathogenic Escherichia coli. Vet. Res. 1999, 30, 203–219. [Google Scholar] [PubMed]
- García-Rubio, V.G.; Bautista-Gómez, L.G.; Martínez-Castañeda, J.S.; Romero-Núñez, C. Multicausal etiology of the enteric syndrome in rabbits from Mexico. Rev. Argent. Microbiol. 2017, 49, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Domingo, P.; Fernández, A.; Sanz, C.; Pueyo, R.; Borobia, M.; Muñoz, A.; Benito, A.; Chacón, G. Importancia de Rotavirus tipo A en conejo de cebo: Estudio comparativo entre animales con procesos entéricos y animales sanos. In Proceedings of the LV 40th Symposium de Cunicultura de ASESCU, Santiago de Compostela, España, 28–29 May 2015; pp. 162–165. Available online: https://asescu.com/wp-content/uploads/2015/06/40Symposium_Santiago2015.pdf (accessed on 15 October 2019). (In Spanish).
- Cerioli, M.; Lavazza, A. Viral enteritis of rabbits. In Recent Advances in Rabbit Sciences; Maertens, L., Coudert, P., Eds.; Plot-it bvba: Merelbeke, Belgium, 2006; pp. 181–186. [Google Scholar]
- Cocchi, M.; Drigo, I.; Bacchin, C.; Bano, L.; Marcon, B.; Agnoletti, F. Toxin-genotyping of Clostridium perfringens strains isolated from rabbits with enteric disease. In Proceedings of the 9th World Rabbit Congress, Verona, Italy, 10–13 June 2008; p. 278. Available online: https://world-rabbit-science.com/WRSA-Proceedings/Congress-2008-Verona/Papers/P-Cocchi.pdf (accessed on 15 October 2019).
- Cocchi, M.; Drigo, I.; Bacchin, C.; Guolo, A.; Bano, L.; Bonci, M.; Agnoletti, F. Characterization of Clostridium perfringens cpb2 gene in rabbit’s strains. In Proceedings of the 16th International Symposium on the Housing and Diseases of Rabbits, Furbearing Animals and Pet Animals, Celle, Germany, 11–13 May 2009; St Hoy: Celle, Germany; pp. 231–238. [Google Scholar]
- Myers, L.L.; Shoop, D.S.; Collins, J.E.; Bradbury, W.C. Diarrheal disease caused by enterotoxigenic Bacteroides fragilis in infant rabbits. J. Clin. Microbiol. 1989, 27, 2025–2030. [Google Scholar]
- Jin, D.X.; Zou, H.W.; Liu, S.Q.; Wang, L.Z.; Xue, B.; Wu, D.; Tian, G.; Cai, J.; Yan, T.H.; Wang, Z.S.; et al. The underlying microbial mechanism of epizootic rabbit enteropathy triggered by a low fiber diet. Sci. Rep. 2018, 8, 12489. [Google Scholar] [CrossRef]
- Saco, M.; Pérez de Rozas, A.; Aloy, N.; González, J.; Rosell, J.M.; Badiola, J.I. Salmonellosis in rabbits. Field and laboratory results during 1999–2011. In Proceedings of the 10th World Rabbit Congress, Sharm El-Sheikh, Egypt, 3–6 September 2012; WRSA ed.: Sharm El Sheikh, Egypt; pp. 1165–1168. [Google Scholar]
- Rosell, J.M.; De la Fuente, L.F.; Badiola, J.I.; Fernández de Luco, D.; Casal, J.; Saco, M. Study of urgent visits to commercial rabbit farms in Spain and Portugal during 1997–2007. World Rabbit Sci. 2009, 17, 127–136. [Google Scholar] [CrossRef]
- Baüerl, C.; Collado, M.C.; Zuñiga, M.; Blas, E.; Perez-Martinez, G. Changes in cecal microbiota and mucosal gene expression revealed new aspects of epizootic rabbit enteropathy. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Djukovic, A.; Garcia-Garcera, M.; Martinez-Paredes, E.; Isaac, S.; Artacho, A.; Martínez, J.; Ubeda, C. Gut colonization by a novel Clostridium species is associated with the onset of epizootic rabbit enteropathy. Vet. Res. 2018, 49, 123. [Google Scholar] [CrossRef]
- Bertó-Moran, A.; Palacio, I.; Serrano, E.; Moreno, S.; Rouco, C. Coccidian and nematode infections influence prevalence of antibody to myxoma and rabbit hemorrhagic disease viruses in European rabbits. J. Wild. Dis. 2013, 49, 10–17. [Google Scholar] [CrossRef]
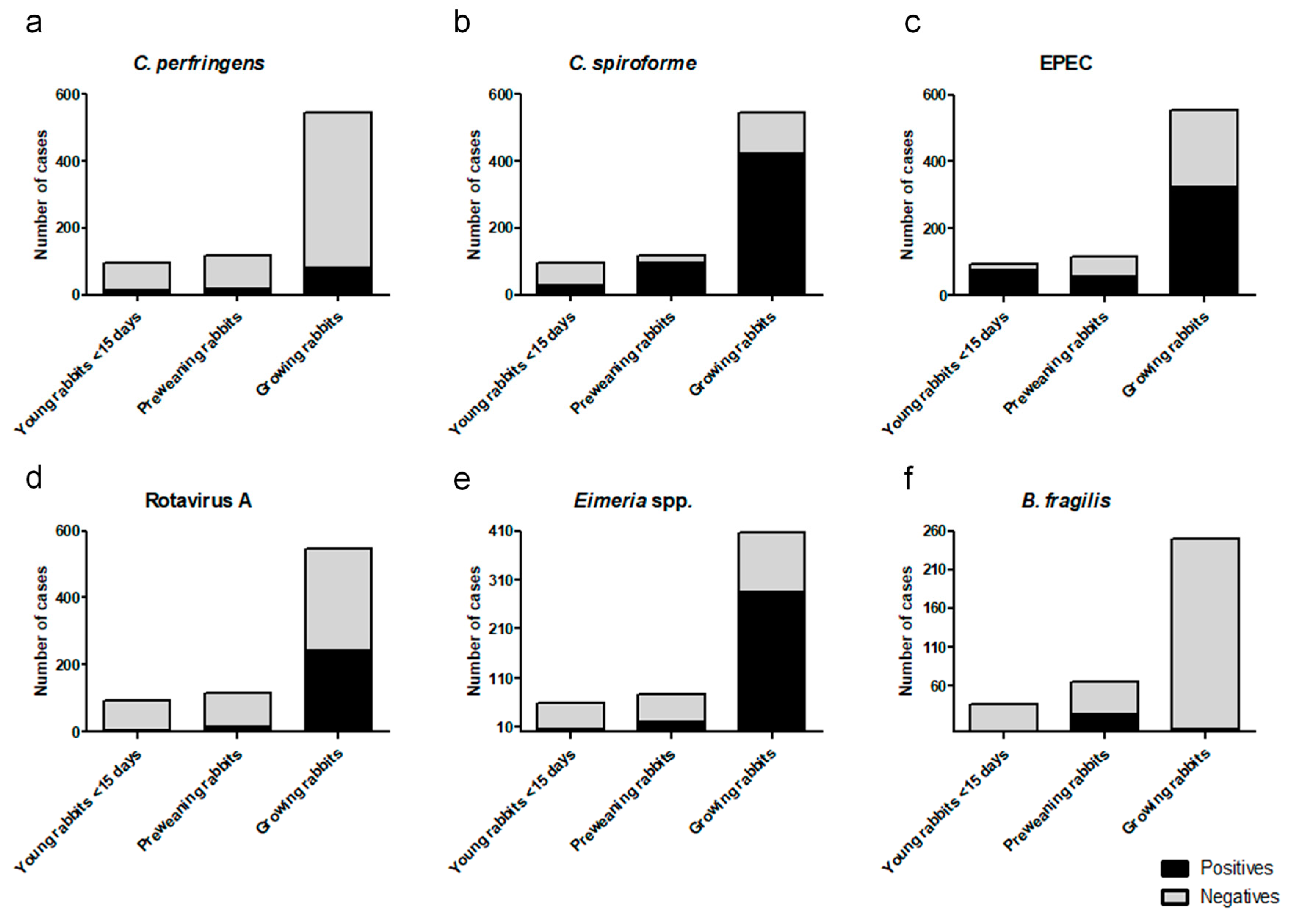
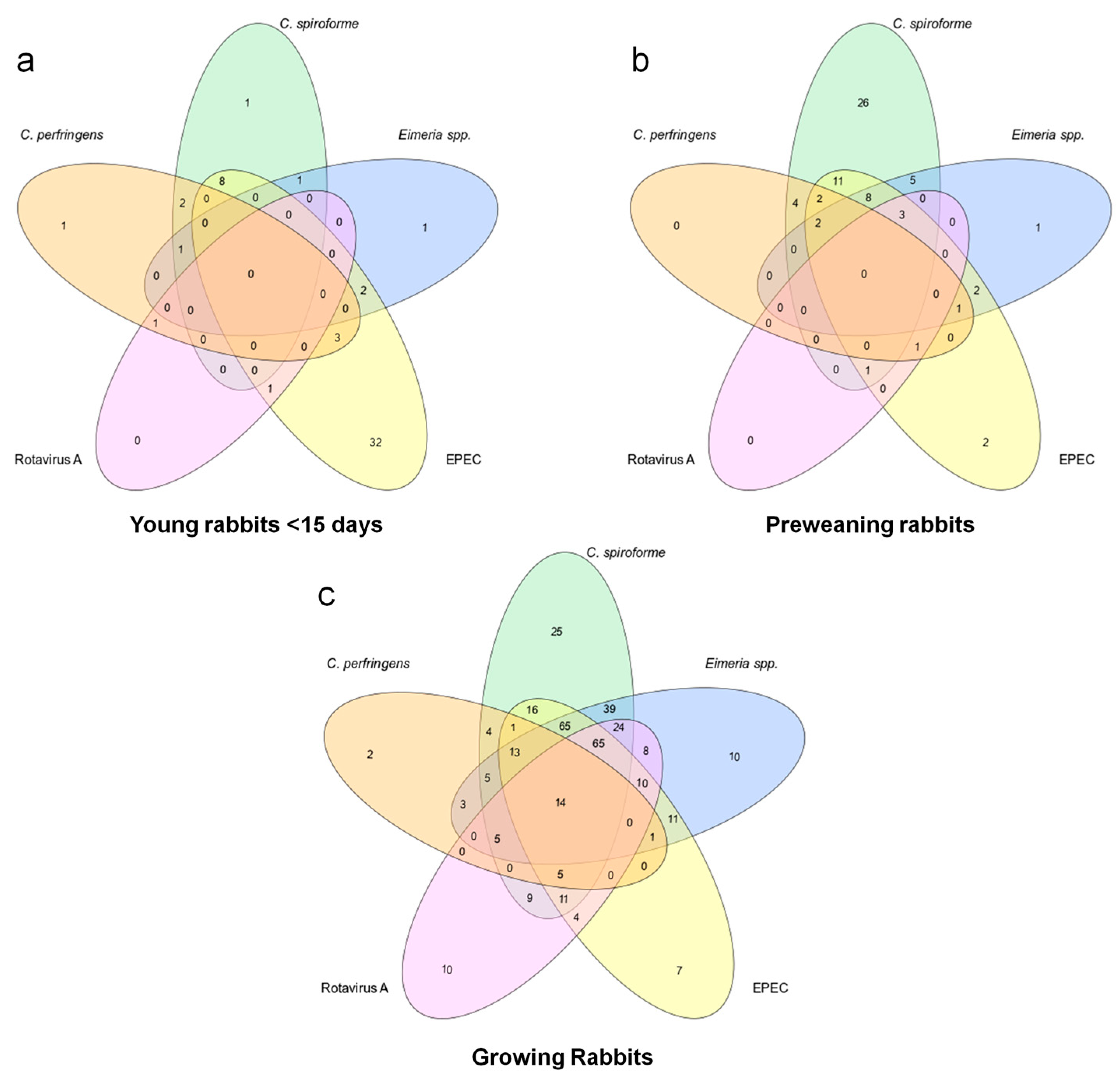
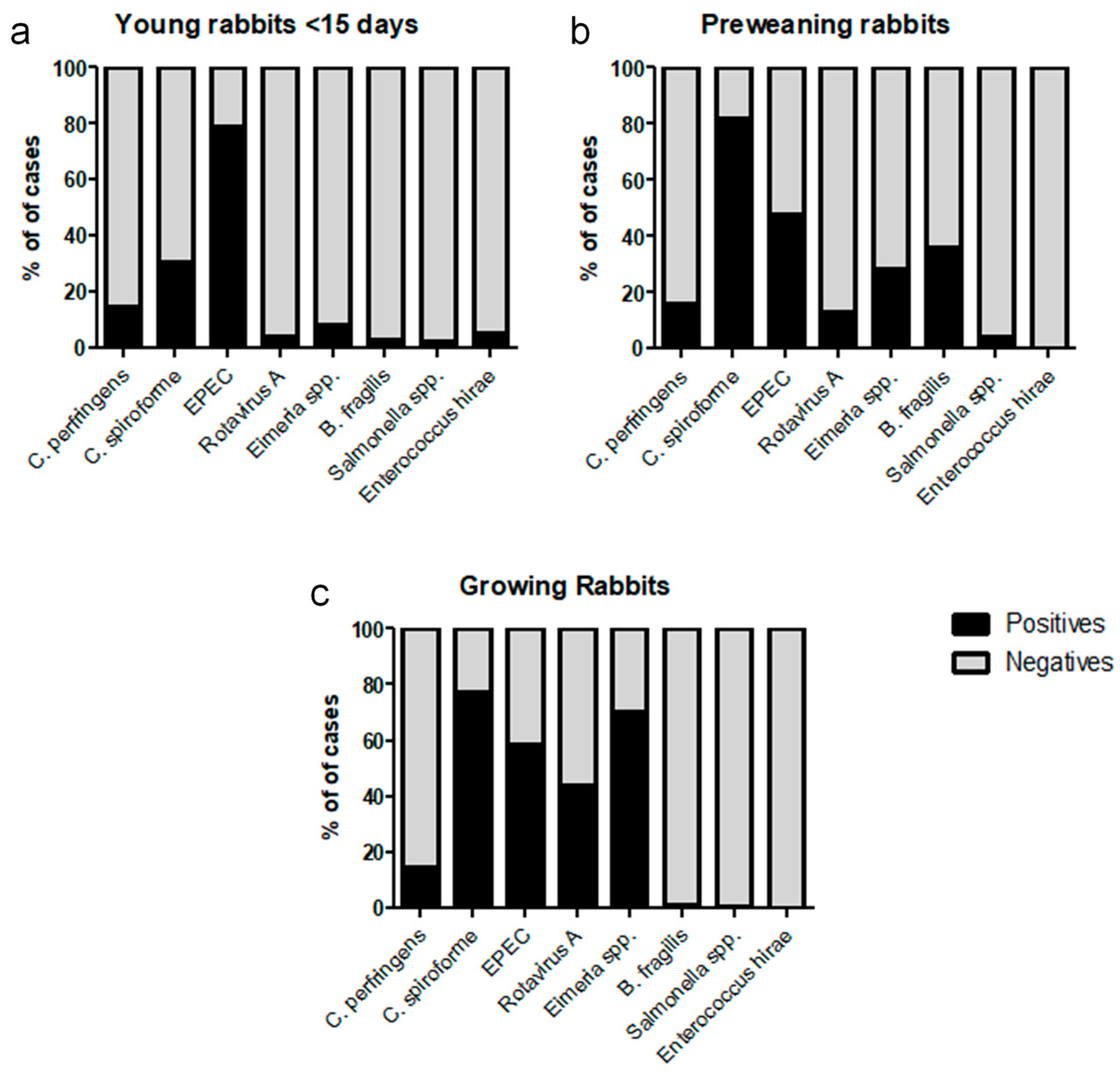
| Technique Used | Young Rabbits | Preweaning Rabbis | Growing Rabbits | Total Cases Studied |
|---|---|---|---|---|
| Microbiological culture | 95 | 117 | 545 | 757 |
| EPEC 1 qPCR 2 | 95 | 117 | 545 | 757 |
| C. spiroforme qPCR | 95 | 117 | 545 | 757 |
| C. perfringens qPCR | 95 | 117 | 545 | 757 |
| Rotavirus A qPCR | 95 | 117 | 545 | 757 |
| Eimeria spp. qPCR | 59 | 77 | 406 | 542 |
| B. fragilis3 qPCR | 35 | 64 | 251 | 349 |
| Etiological Agent | Gene Target | Coded Protein |
|---|---|---|
| EPEC | eae | Intimin |
| C. spiroforme | sbs | CSTb 1 |
| C. perfringens | cpa (plc) | Alpha toxin |
| Rotavirus A | gene7 | NSP3 2 |
| Enterotoxigenic B. fragilis | bftp | enterotoxin |
| Eimeria spp. | 16S | D16S rRNA |
| Isolated Agent | Young Rabbits (<15 days old) | Preweaning Rabbits | Growing Rabbits | Total Positive Cases (%) |
|---|---|---|---|---|
| Salmonella spp. | 2/95 | 5/117 | 2/545 | 9/757 (1.19%) |
| E. hirae | 2/95 | 0/117 | 0/545 | 2/757 (0.26%) |
| Genetic Attributes | Number of Isolates | ||||
|---|---|---|---|---|---|
| eae | afr2 | liftA | ral | paa | |
| - | - | - | - | - | 38 |
| + | - | + | + | + | 28 |
| + | - | + | - | - | 4 |
| + | - | + | + | - | 1 |
| + | - | - | + | + | 1 |
| + | + | - | - | + | 5 |
| + | + | - | - | - | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solans, L.; Arnal, J.L.; Sanz, C.; Benito, A.; Chacón, G.; Alzuguren, O.; Fernández, A.B. Rabbit Enteropathies on Commercial Farms in the Iberian Peninsula: Etiological Agents Identified in 2018–2019. Animals 2019, 9, 1142. https://doi.org/10.3390/ani9121142
Solans L, Arnal JL, Sanz C, Benito A, Chacón G, Alzuguren O, Fernández AB. Rabbit Enteropathies on Commercial Farms in the Iberian Peninsula: Etiological Agents Identified in 2018–2019. Animals. 2019; 9(12):1142. https://doi.org/10.3390/ani9121142
Chicago/Turabian StyleSolans, Luis, Jose L. Arnal, Celia Sanz, Alfredo Benito, Gema Chacón, Oihane Alzuguren, and Ana B. Fernández. 2019. "Rabbit Enteropathies on Commercial Farms in the Iberian Peninsula: Etiological Agents Identified in 2018–2019" Animals 9, no. 12: 1142. https://doi.org/10.3390/ani9121142
APA StyleSolans, L., Arnal, J. L., Sanz, C., Benito, A., Chacón, G., Alzuguren, O., & Fernández, A. B. (2019). Rabbit Enteropathies on Commercial Farms in the Iberian Peninsula: Etiological Agents Identified in 2018–2019. Animals, 9(12), 1142. https://doi.org/10.3390/ani9121142





