A Study of the Head during Prenatal and Perinatal Development of Two Fetuses and One Newborn Striped Dolphin (Stenella coeruleoalba, Meyen 1833) Using Dissections, Sectional Anatomy, CT, and MRI: Anatomical and Functional Implications in Cetaceans and Terrestrial Mammals
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Computed Tomography
2.3. Magnetic Resonance Imaging
2.4. Anatomic Evaluation
2.5. Gross Dissections
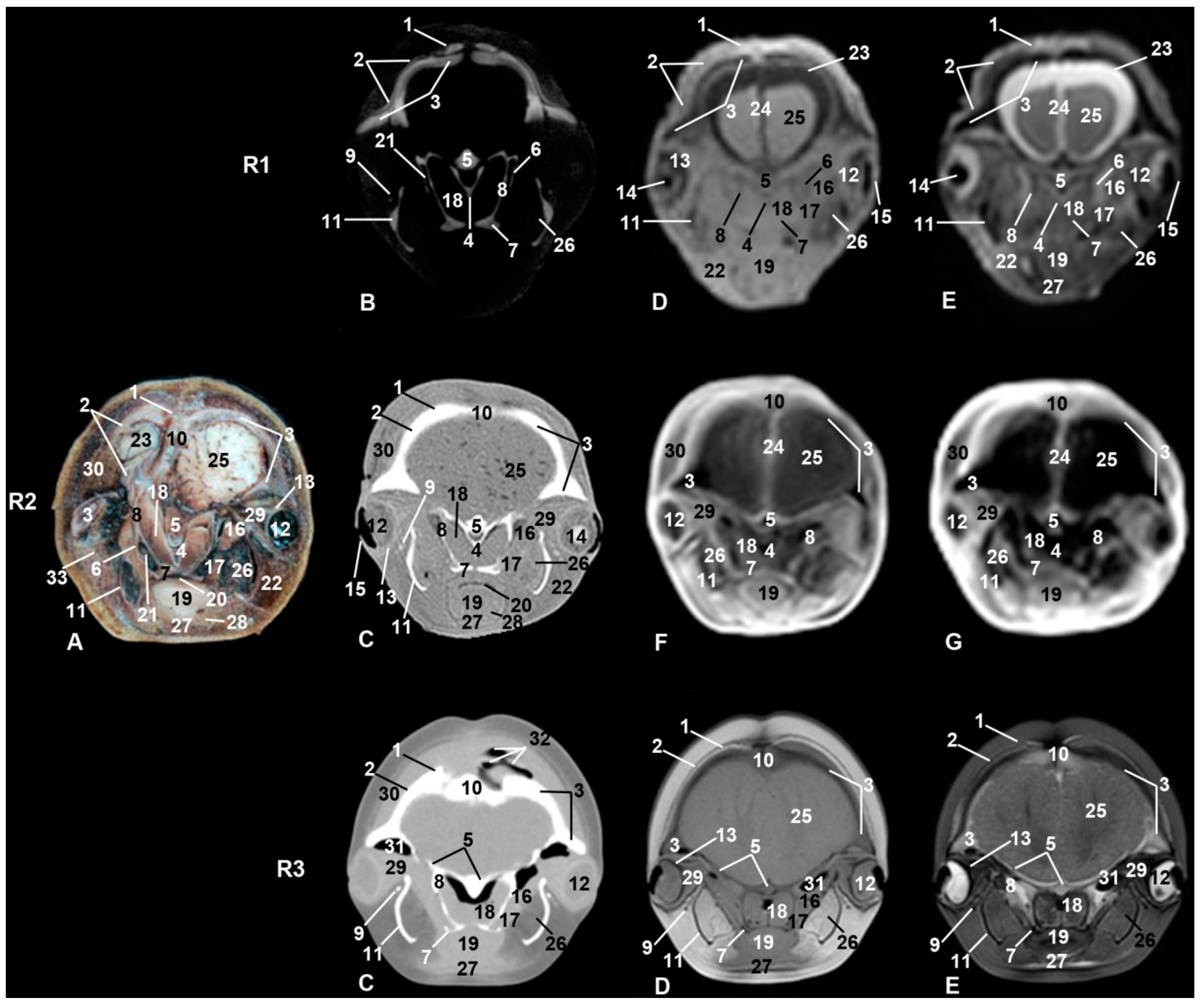
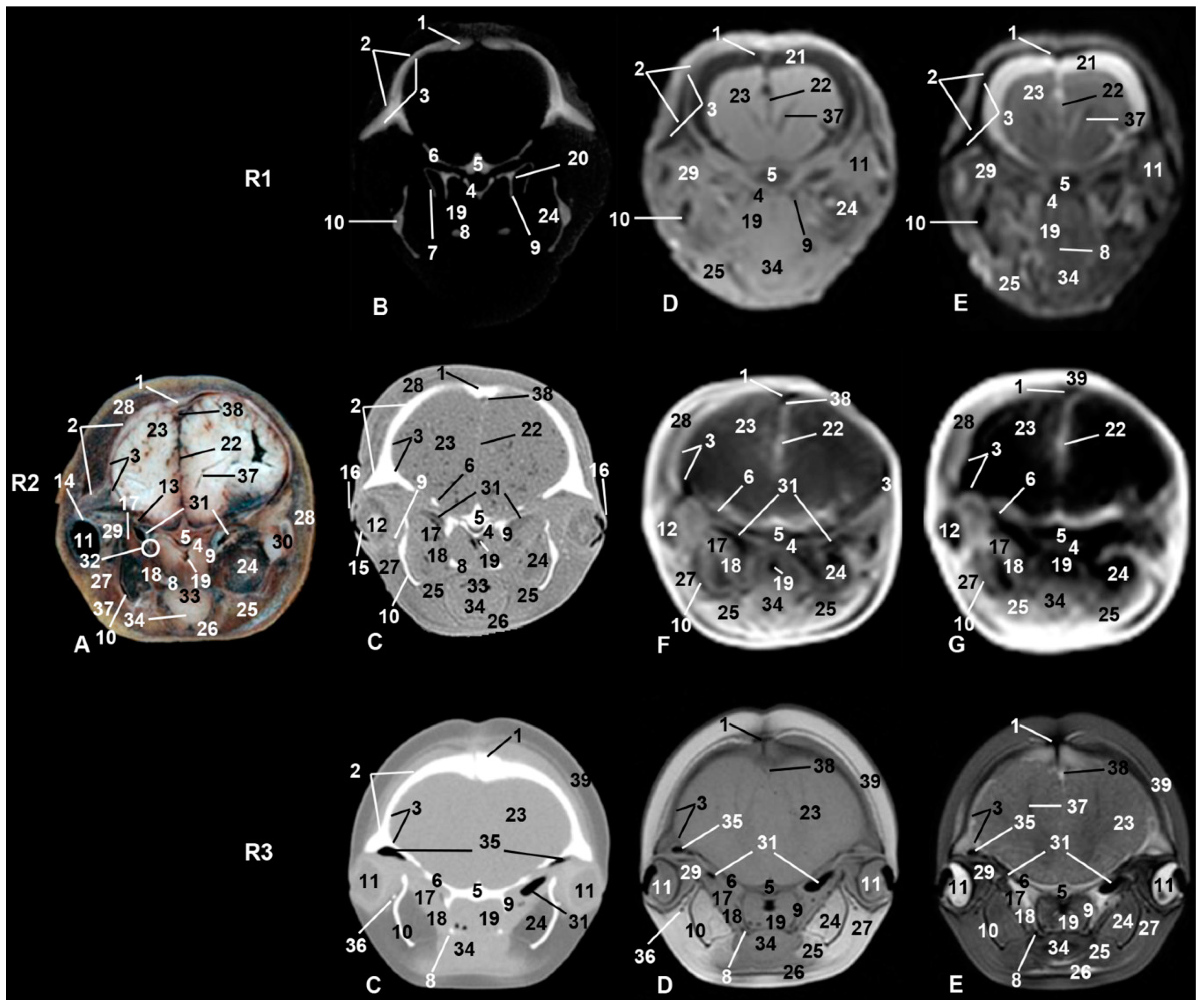
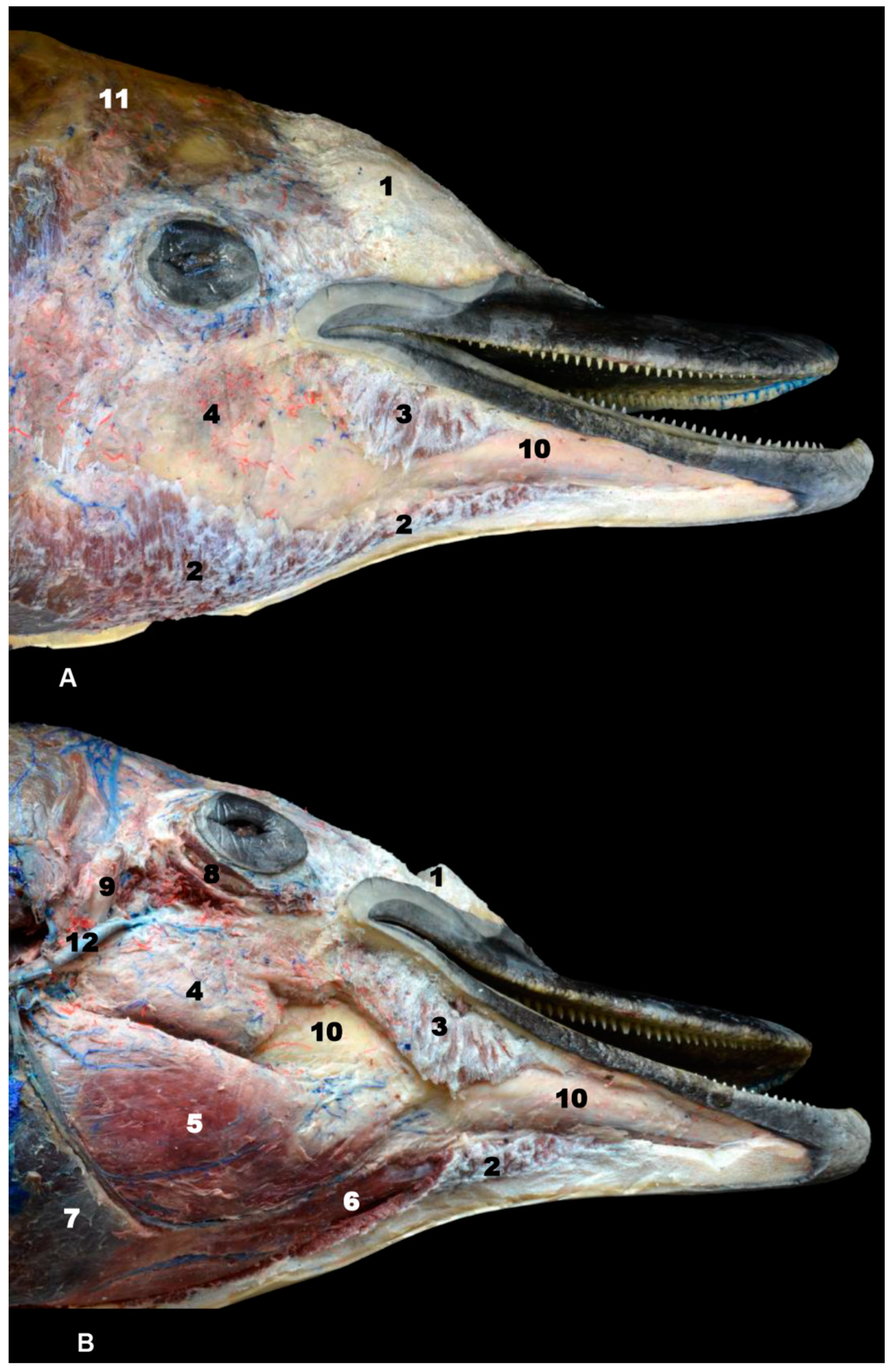
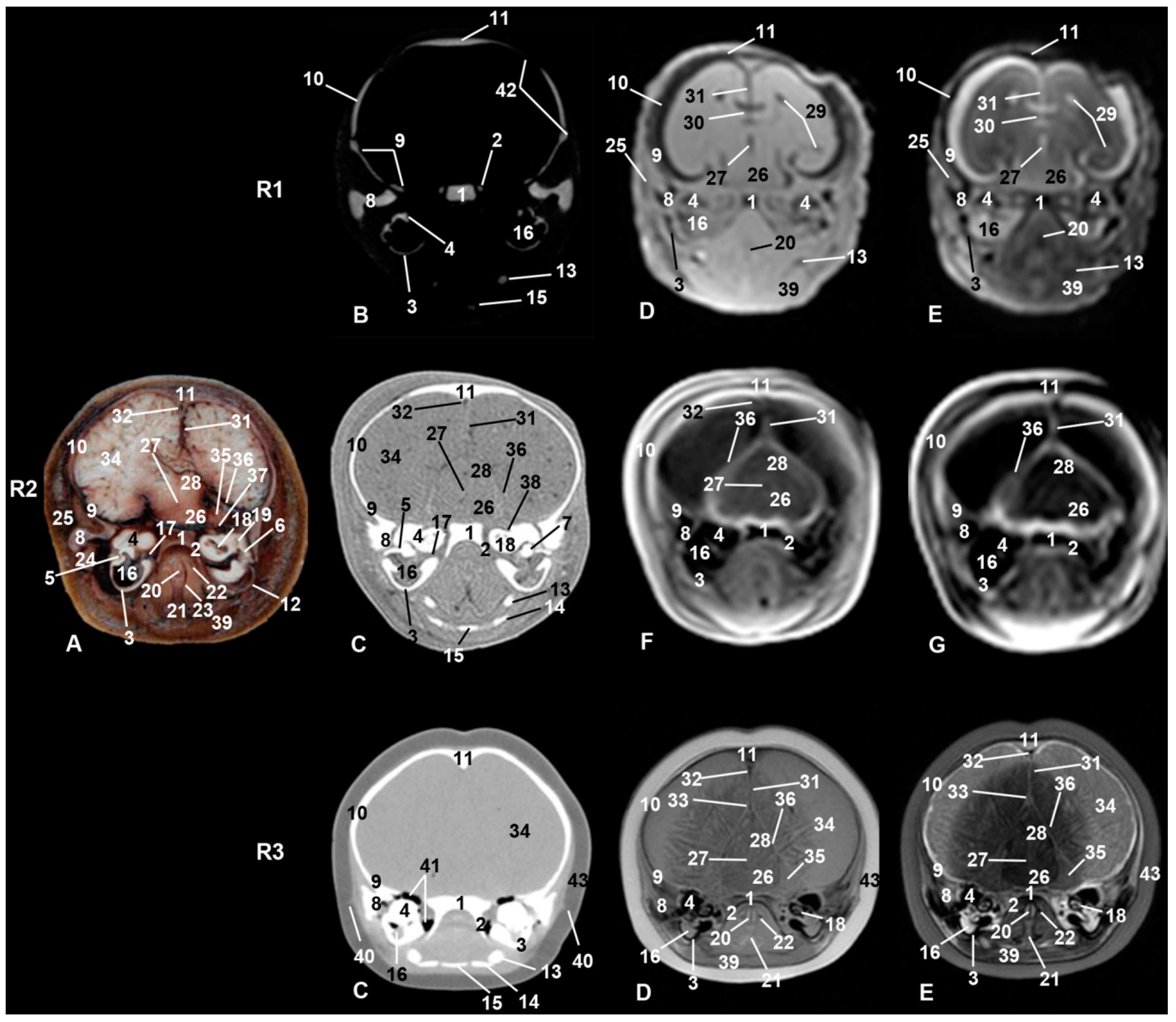
3. Results
3.1. Oral Cavity
3.2. Rostrum (Snout)
3.3. Melon
3.4. Nasal Cavity and Pasanasal Sinuses
3.5. Orbit and Eyeball
3.6. Central Nervous System
3.7. Ear
3.8. Larynx
3.9. Cranial Cavity
4. Discussion
4.1. Anatomical and Functional Considerations
4.1.1. Oral Cavity
4.1.2. Rostrum (Snout)
4.1.3. Melon
4.1.4. Nasal Cavity and Paranasal Sinuses
4.1.5. Orbit and Eyeball
4.1.6. Central Nervous System
4.1.7. Ear
4.1.8. Larynx
4.1.9. Cranial Cavity
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mead, J.G.; Fordyce, R.E. The therian skull. A lexicon with emphasis on the odontocetes. Smithson. Contr. Zool. 2009, 627, 1–248. [Google Scholar] [CrossRef]
- Berta, A.; Sumich, J.L.; Kovacs, K.M. Cetacean evolution and systematics. In Marine Mammal Evolutionary Biology, 2nd ed.; Academic Press: San Diego, CA, USA, 2005; pp. 165–209. [Google Scholar]
- Roston, R.A.; Roth, V.L. Cetacean Skull Telescoping Brings Evolution of Cranial Sutures into Focus. Anat. Rec. 2019. [Google Scholar] [CrossRef] [PubMed]
- Armfield, B.A.; George, J.C.; Vinyard, C.J.; Thewissen, J.G.M. Allometric patterns of fetal head growth in mysticetes and odontocetes: Comparison of Balaena mysticetus and Stenella attenuata. Marine Mammal Sci. 2011, 27, 819–827. [Google Scholar] [CrossRef]
- Sterba, O.; Klima, M.; Schildger, B. Embryology of dolphins. Staging and ageing of embryos and fetuses of some cetaceans. Adv. Anat. Embryol. Cell. Biol. 2000, 157, 1–133. [Google Scholar]
- Thewisen, J.G.M.; Heyning, J.E. Embryogenesis and development in Stenella atenuatta and other cetaceans. In Reproductive Biology and Phylogeny in Cetacea, Whales, Dolphins and Porpoises; Miller, D.L., Ed.; Science Publishers: Enflield, NH, USA, 2007; pp. 307–330. [Google Scholar]
- Reidenberg, J.S.; Laitman, J.T. Prenatal development in cetaceans. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: San Diego, CA, USA, 2009; pp. 220–230. [Google Scholar]
- Hosokawa, H.; Kamiya, T. Sections of the dolphin’s head (Stenella coeruleoalba). Sci. Rep. Whales Res. Inst. 1965, 19, 105–133. [Google Scholar]
- Alonso-Farre, J.M.; Gonzalo-Orden, M.; Barreiro-Vázquez, J.D.; Barreiro-Lois, A.; André Morell, M.; Llarena-Reino, M.; Monreal-Pawlowsky, T.; Degollada, E. Cross-sectional anatomy, computed tomography and magnetic resonance imaging of the head of common dolphin (Delphinus delphis) and striped dolphin (Stenella coeruleoalba). J. Vet. Med. Anat. Histol. Embryol. 2014, 43, 1–9. [Google Scholar] [CrossRef]
- Corpa, J.M.; Peris, B.; Palacio, J.; Liste, F.; Ribes, V. Hydrocephalus in a newborn bottlenosed dolphin (Tursiops truncatus). Vet. Rec. 2004, 155, 208–210. [Google Scholar] [CrossRef]
- Liste, F.; Palacio, J.; Ribes, V.; Alvarez-Clau, A.; Fernández, L.; Corpa, J.M. Anatomic and Computed tomography atlas of the head of the newborn bottlenosed dolphin (Tursiops truncatus). Vet. Radiol. Ultrasound. 2006, 47, 453–460. [Google Scholar] [CrossRef]
- Rauschmann, M.A.; Huggenberger, S.; Kossatz, L.S.; Oelschläger, H.H.A. Head morphology in perinatal dolphins: A window into phylogeny and ontogeny. J. Morphol. 2006, 267, 1295–1315. [Google Scholar] [CrossRef]
- Eales, N.B. The skull of the foetal narwhal Monodon monoceros L. Phil. Trans. R. Soc. 1950, 235, 1–33. [Google Scholar]
- Marino, L.; Murphy, T.L.; Lyad, G.; Johnson, J.I. Magnetic resonance imaging and three-dimensional reconstructions of the brain of a fetal common dolphin, Delphinus delphis. Anat. Embryol. 2001, 203, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Montie, E.W.; Schneider, G.E.; Ketten, D.R.; Marino, L.; Touhey, K.E.; Hahn, M.E. Neuroanatomy of the subadult and fetal brain of the atlantic white-sided dolphin (Lagenorhynchus acutus) from in situ magnetic resonance images. Anat. Rec. 2007, 290, 1459–1479. [Google Scholar] [CrossRef] [PubMed]
- Costidis, A.M.; Rommel, S.A. The extracranial arterial system in the heads of beaked whales, with implications on diving physiology and pathogenesis. J. Morphol. 2016, 277, 5–33. [Google Scholar] [CrossRef] [PubMed]
- Ridewood, W.G. Observations on the skull in foetal specimens of whales of the genera Megaptera and Balaenoptera. Phil. Trans. R. Soc. 1922, 211, 209–272. [Google Scholar] [CrossRef]
- Johnston, C.; Deméré, T.A.; Berta, A.; Yonas, J.; Leger, J. Observations on the musculoskeletal anatomy of the head of a neonate gray whale (Eschrichtius robustus). Mar. Mammal. Sci. 2010, 26, 186–194. [Google Scholar] [CrossRef]
- Schulte, H.W. The sei whale (Balaenoptera borealis, Lesson). Anatomy of a foetus of Balaenoptera borealis. Monographs of the Pacific Cetacea. Mem. Am. Mus. Nat. Hist. New Ser. 1916, 1, 389–499. [Google Scholar]
- O’Rahilly, R.; Müller, F. Developmental Stages in Human Embryos; Carnegie Institute: Washington, DC, USA, 1987; pp. 1–637. [Google Scholar]
- Perrin, W.F.; Holts, D.B.; Miller, R.B. Growth and reproduction of the eastern spinner dolphin, a geographical form of Stenella longirostris in the eastern tropical pacific. Fish Bull. 1977, 75, 725–750. [Google Scholar]
- Lacave, G.; Eggermont, M.; Verslycke, T.; Kinoshita, R. Prediction from ultrasonographic measurements of the expected delivery date in two species of bottlenose. Vet. Rec. 2004, 154, 228–233. [Google Scholar] [CrossRef]
- Hohn, A.A. Age estimation. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Pres: Cambridge, MA, USA, 2009; pp. 11–17. [Google Scholar]
- Schaller, O. Illustrated Veterinary Anatomical Nomenclature; Ferdinand Enke Verlag: Stuttgart, Germany, 1992; pp. 1–614. [Google Scholar]
- Roston, R.A.; Lickorish, D.; Buchholtz, E.A. Anatomy and age estimation of an early blue whale (Balaenoptera musculus) fetus. Anat. Rec. 2013, 296, 709–722. [Google Scholar] [CrossRef]
- Wilkie Tinker, S. Whales of the World; Brill, E.J., Ed.; Bess Pr Inc.: Leiden, The Netherlands, 1988; pp. 1–310. [Google Scholar]
- Armfield, B.A.; Zheng, Z.; Bajpai, S.; Vinyard, C.J.; Thewissen, J. Development and evolution of the unique cetacean dentition. PeerJ 2013, 1, e24. [Google Scholar] [CrossRef]
- Rommel, S.A.; Pabst, D.A.; McLellan, W.A. Skull anatomy. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: Cambridge, MA, USA, 2009; pp. 1033–1056. [Google Scholar]
- Huggenberger, S.; Oelschläger, H.; Cozzi, B. Atlas of the Anatomy of Dolphins and Whales; Academic Press, Elsevier: London, UK, 2019; pp. 1–513. [Google Scholar]
- Cozzi, B.; Huggenberger, S.; Oelschläger, H. Anatomy of Dolphins. Insights into Body Structure and Function; Academic Press: London, UK, 2017; pp. 1–438. [Google Scholar]
- Sandoval, J. Tratado de Anatomía Veterinaria. Tomo III: Cabeza y Sistemas Viscerales; Imprenta Sorles: León, Spain, 2000; pp. 1–457. [Google Scholar]
- Nickel, R.; Schummer, A.; Seiferle, E. The Anatomy of the Domestic Animals. The Locomotor System of the Domestic Mammals; Verlag Paul Parey: Berlin/Hamburg, Germany, 1986; Volume 1, pp. 1–499. [Google Scholar]
- Werth, A.J. Adaptations of the cetacean hyolingual apparatus for aquatic feeding and thermoregulation. Anat. Rec. 2007, 290, 546–568. [Google Scholar] [CrossRef] [PubMed]
- Heithaus, M.R.; Dill, L.M. Feeding strategies and tactics. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: San Diego, CA, USA, 2009; pp. 414–423. [Google Scholar]
- Pyenson, N.D.; Goldbogen, J.A.; Vogl, A.W.; Szathmary, G.; Drake, R.L.; Shadwick, R.E. Discovery of a sensory organ that coordinates lunge feeding in rorqual whales. Nature 2012, 485, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Lambertsen, R.H.; Hintz, R.J. Maxillomandibular cam articulation discovered in North Atlantic minke whale. J. Mammal. 2004, 85, 446–452. [Google Scholar] [CrossRef]
- Girard, G.; Lagacé, A.; Higgins, R.; Béland, P. Adenocarcinoma of the salivary gland in a beluga whale (Delphinapterus leucas). J. Vet. Diagn. Invest. 1991, 3, 264–265. [Google Scholar] [CrossRef] [PubMed]
- Harper, C.J.; McLellan, W.A.; Rommel, S.A.; Gay, D.M.; Dillaman, R.M.; Pabst, D.A. Morphology on the melon and its tendinous connections to the facial muscles in bottlenose dolphins (Tursiops truncatus). J. Morphol. 2008, 269, 820–839. [Google Scholar] [CrossRef] [PubMed]
- O´Corry-Crowe, G.M. Beluga whale. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: Cambridge, MA, USA, 2009; pp. 108–112. [Google Scholar]
- Behrmann, G. Funktion und Evolution der Delphinnase. Nat. Mus. 1983, 113, 71–78. [Google Scholar]
- Reidenberg, J.S.; Laitman, J.T. Sisters of the sinuse cetacean air sacs. Anat. Rec. 2008, 291, 1389–1396. [Google Scholar] [CrossRef]
- Lawrence, B.; Schevill, W.E. The Functional Anatomy of the Delphinid Nose. Bull. Mus. Comp. Zool. 1956, 114, 103–150. [Google Scholar]
- Schenkkan, E.J. The occurrence and position of the “connecting sac” in the nasal tract complex of small odontocetes (Mammalia, Cetacea). University of Amsterdam. Beaufortia 1971, 246, 37–43. [Google Scholar]
- Heyning, J.E. Comparative facial anatomy of beaked whales (Ziphiidae) and a systematic revision among the families of extant odontoceti. Nat. Hist. Mus. Los Angel. Cty. 1989, 405, 1–59. [Google Scholar]
- Cranford, T.W. Visualizing dolphin sonar signal generation using high speed video endoscopy. J. Acoust. Soc. Am. 1997, 102, 3123. [Google Scholar] [CrossRef]
- Cranford, T.W.; Amundin, M.; Kenneth, S.N. Functional morphology and homology in the Odontocete nasal complex: Implications for sound generation. J. Morph. 1996, 228, 223–285. [Google Scholar] [CrossRef]
- Reidenberg, J.S.; Laitman, J.T. Generation of sound in marine mammals. In Handbook of Mammalian Vocalization—An Integrative Neuroscience Approach; Brudzynski, S.M., Ed.; Academic Press/Elsevier: London, UK, 2010; pp. 451–465. [Google Scholar]
- Whitlow, W.L. Echolocation. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: Cambridge, MA, USA, 2009; pp. 348–357. [Google Scholar]
- Reidenberg, J.S.; Laitman, J.T. Cetacean prenatal development. In Encyclopedia of Marine Mammals, 2nd ed.; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: Cambridge, MA, USA, 2009; pp. 220–230. [Google Scholar]
- Racicot, A.; Berta, A. Comparative morphology of porpoise (Cetacea: Phocaenidae) pterygoid sinuses: Phylogenetic and functional implications. J. Morphol. 2013, 274, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Gatesy, J.; Geisler, J.H.; Chang, J.; Buell, C.; Berta, A.; Meredith, R.W.; Springer, M.S.; McGowen, M.R. A phylogenetic blueprint for a modern whale. Mol. Phylogenet. Evol. 2013, 66, 479–506. [Google Scholar] [CrossRef] [PubMed]
- Macleod, C.C.; Reidenberg, J.S.; Weller, M.; Santos, M.B.; Herman, J.; Goold, J.; Piercel, G.J. Breaking symmetry: The marine environment, prey size, and the evolution of asymmetry in cetacean skulls. Anat. Rec. 2007, 290, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, A.R. The history of the whales: Their adaptation to life in the water. Q. Rev. Biol. 1928, 3, 29–76. [Google Scholar] [CrossRef]
- Fraser, F.C.; Purves, P.E. Hearing in cetaceans-evolution of the accessory air sacs and the structure and function of the outer and middle ears in recent cetaceans. Bull. Br. Mus. 1960, 7, 1–140. [Google Scholar]
- Moran, M.M.; Numella, S.; Thewissen, J.G.M. Development of the skull of the pantropical spotted dolphin (Stenella attenuata). Anat. Rec. 2011, 294, 1743–1756. [Google Scholar] [CrossRef]
- McGeady, T.A.; Quinn, P.J.; Fitzpatrick, E.S.; Ryan, M.T.; Kilroy, D.; Lonergan, P. Veterinary Embryology; Wiley Blackwell: Oxford, UK, 2017; pp. 1–400. [Google Scholar]
- García Monterde, J.; Gil Cano, F. Embriología Veterinaria: Un Enfoque Dinámico del Desarrollo Animal; Inter-médica: Buenos Aires, Argentina, 2013; pp. 1–185. [Google Scholar]
- Houser, D.S.; Finneran, J.; Carder, D.; Van Bonn, W.; Smith, C.; Hoh, C.; Mattrey, R.; Ridgway, S. Structural and functional imaging of bottlenose dolphin (Tursiops truncatus) cranial anatomy. J. Exp. Biol. 2004, 207, 3657–3665. [Google Scholar] [CrossRef]
- Bouetel, V. Phylogenetic implications of skull structure and feeding behaviour in Balaenopterids (Cetacea, Mysticeti). J. Mammal. 2005, 86, 139–146. [Google Scholar] [CrossRef]
- Field, D.J.; Campbell-Malone, R.; Goldbogen, J.A.; Shadwick, R.E. Quantitative computed tomography of humpback whale (Megaptera novaeangliae) mandibles: Mechanical implications for rorqual lunge-feeding. Anat. Rec. 2010, 293, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Reidenberg, J.S.; Laitman, J.T. Anatomy of the hyoid apparatus in Odontoceti (toothed whales): Specializations of their skeleton and musculature compared with those of terrestrial mammals. Anat. Rec. 1994, 240, 598–624. [Google Scholar] [CrossRef] [PubMed]
- Kienle, S.S.; Ekdale, E.G.; Reidenberg, J.S.; Deméré, T.A. Tongue and Hyoid Musculature and Functional Morphology of a Neonate Gray Whale (Cetacea, Mysticeti, Eschrichtius robustus). Anat. Rec. 2015, 298, 660–674. [Google Scholar] [CrossRef] [PubMed]
- Hampe, O.; Franke, H.; Hipsley, C.A.; Kardjilov, N.; Muller, J. Prenatal cranial ossification of the humpback whale (Megaptera novaeangliae). J. Morph. 2015, 276, 564–582. [Google Scholar] [CrossRef]
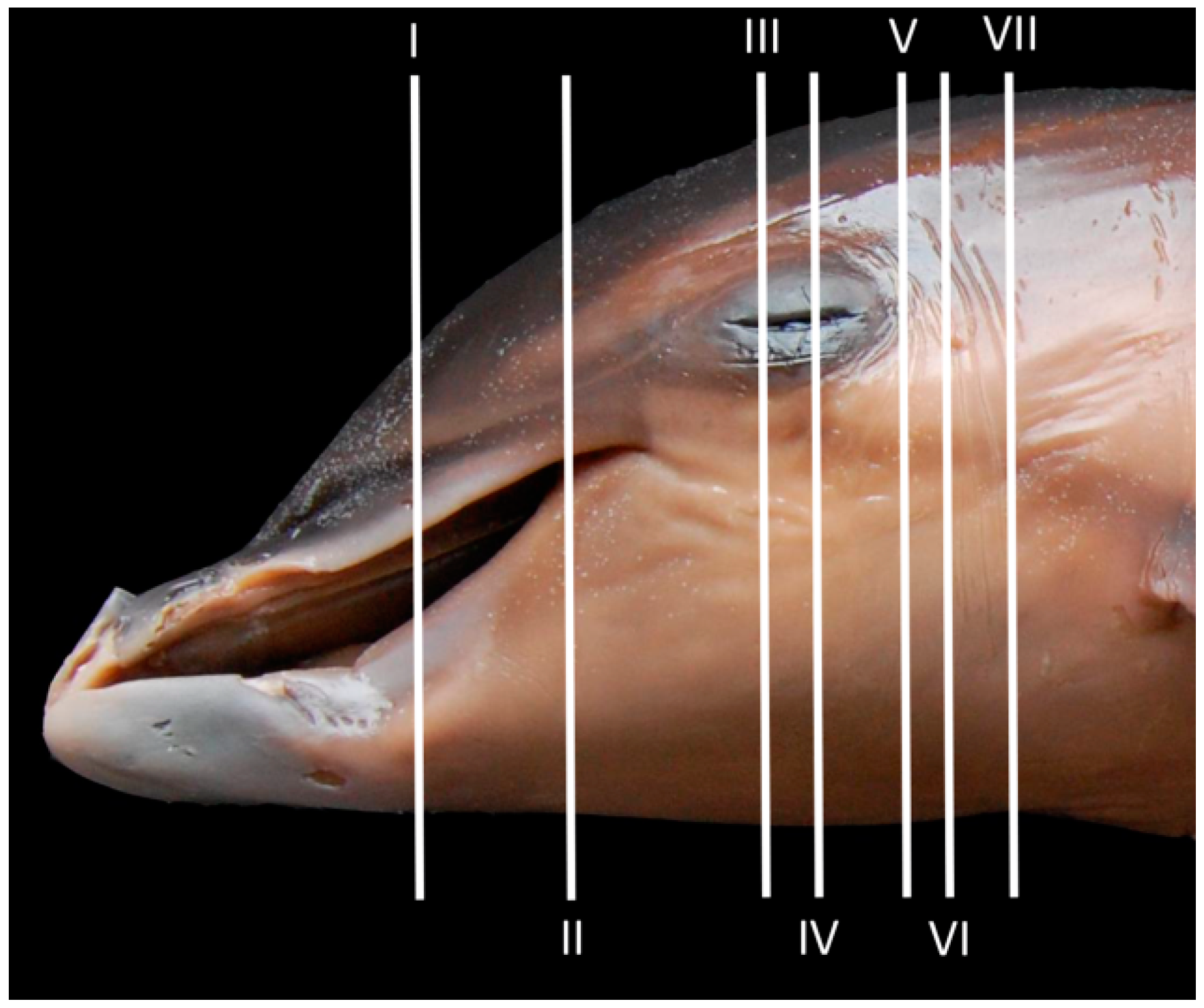
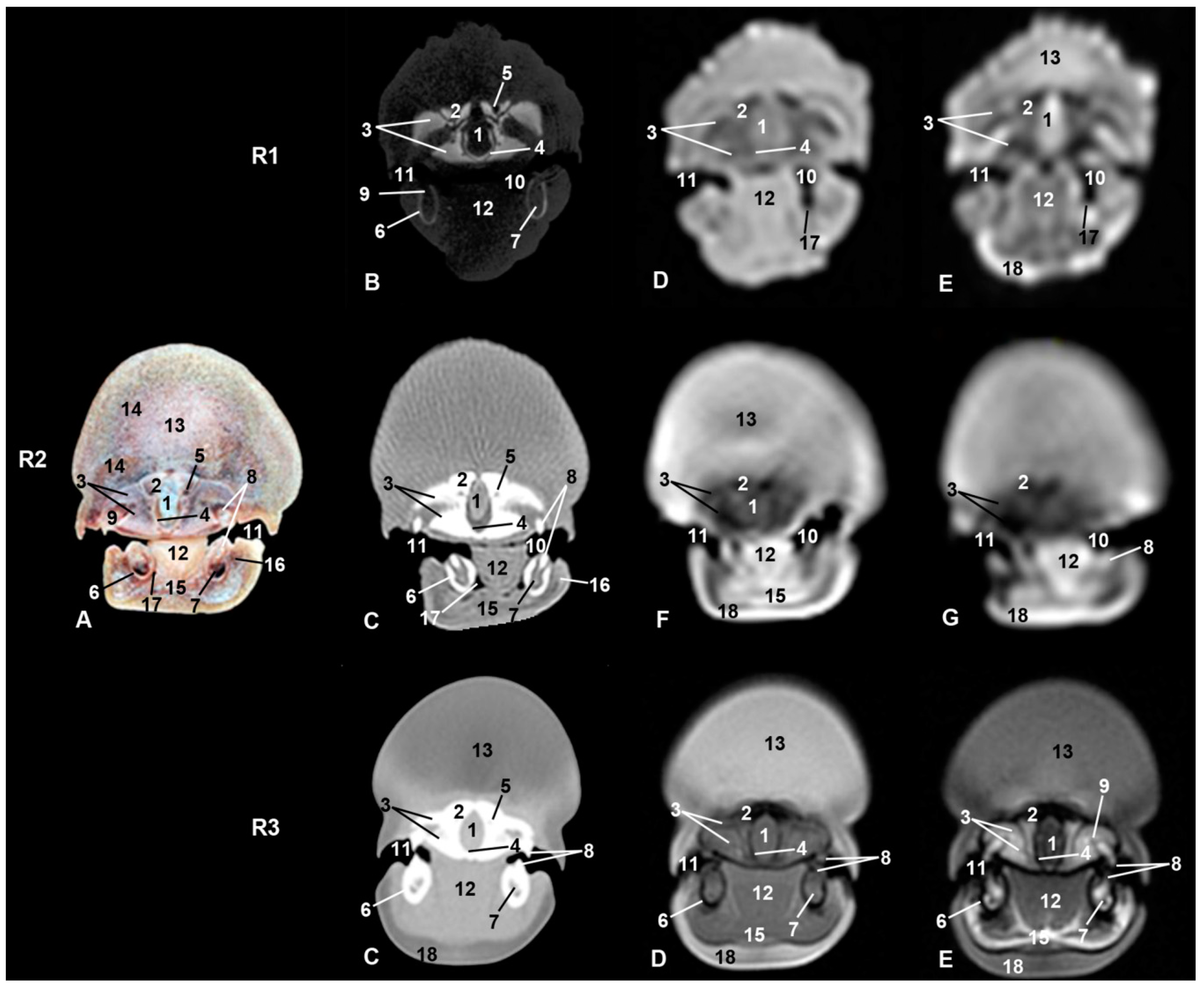
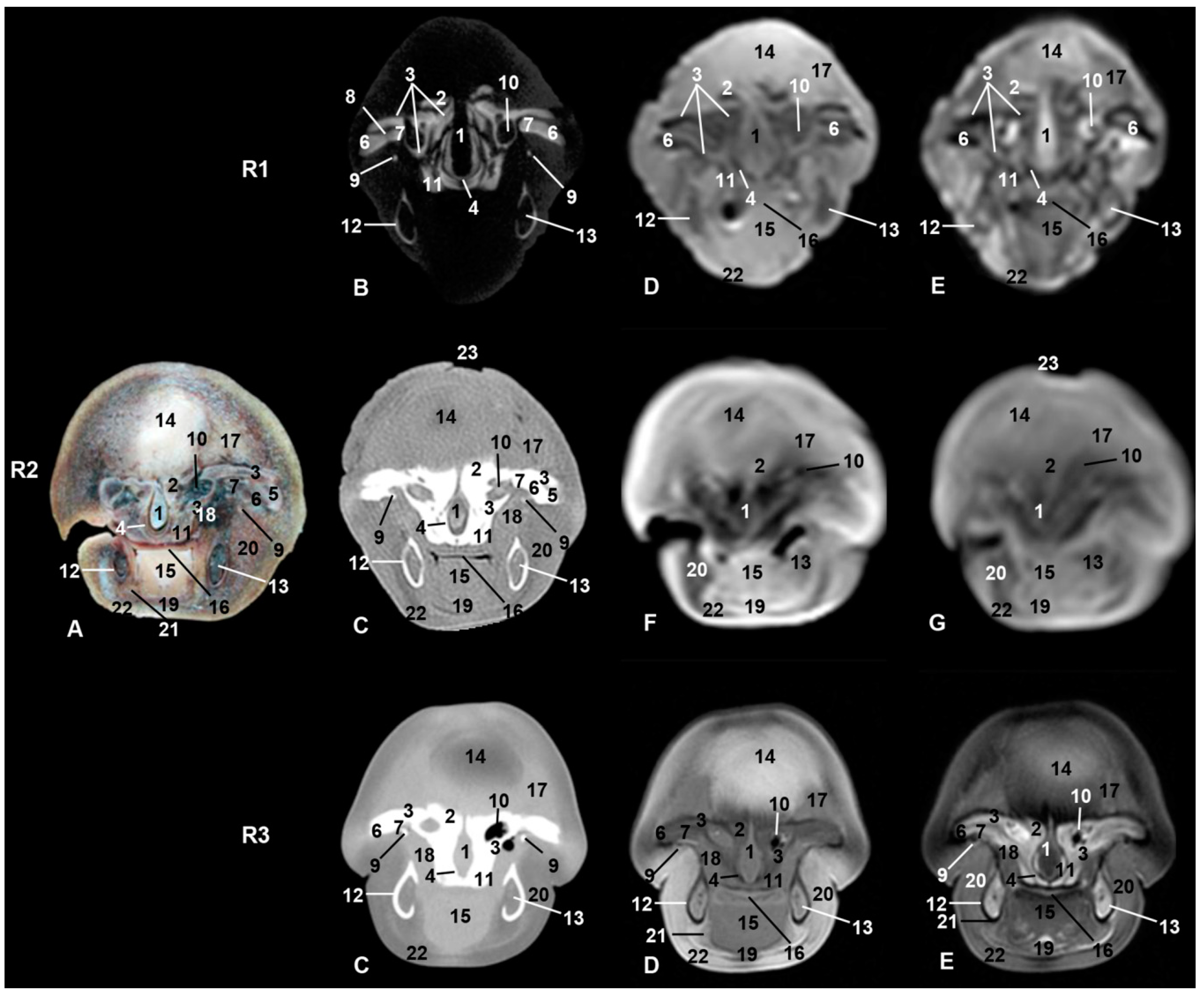
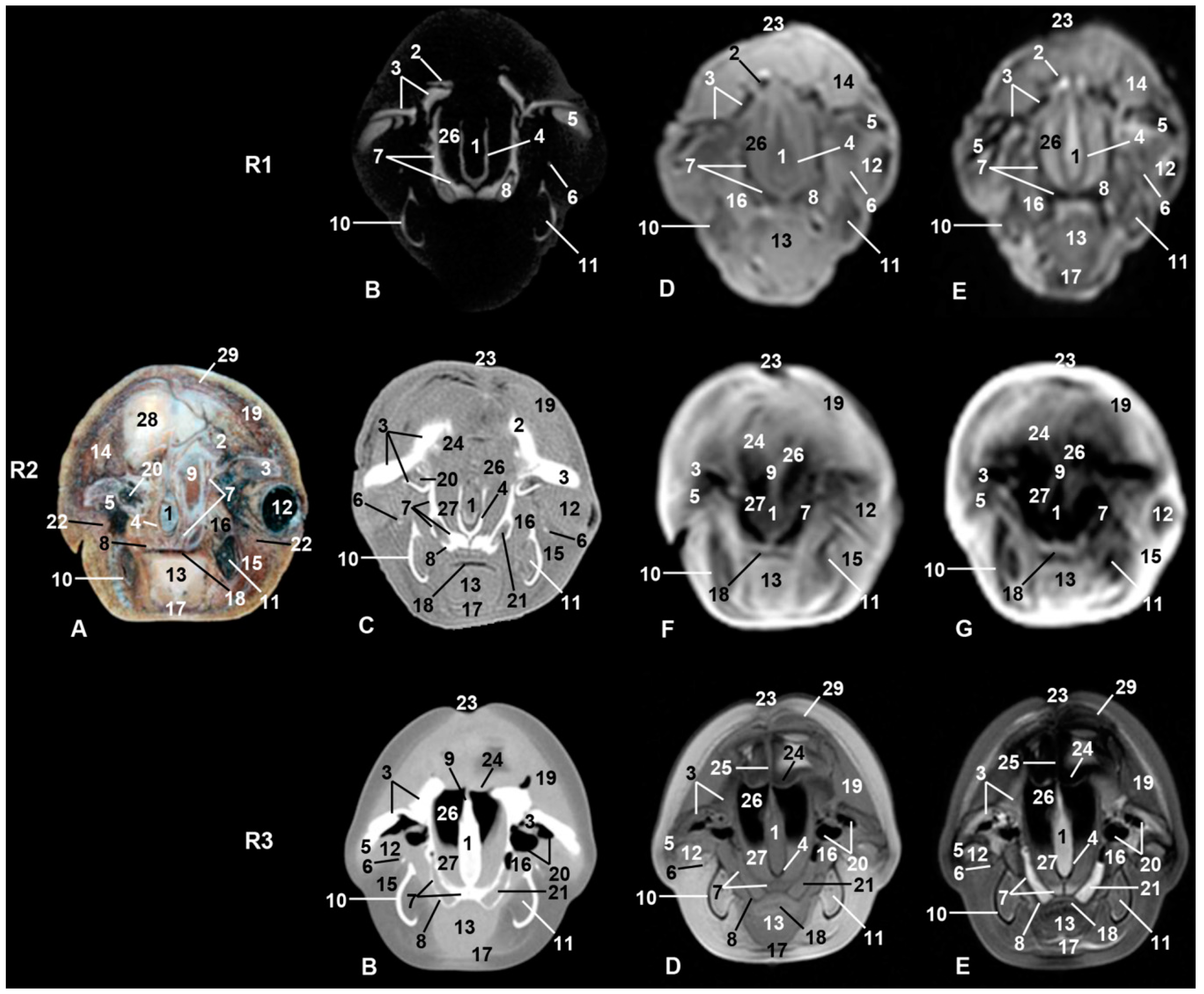
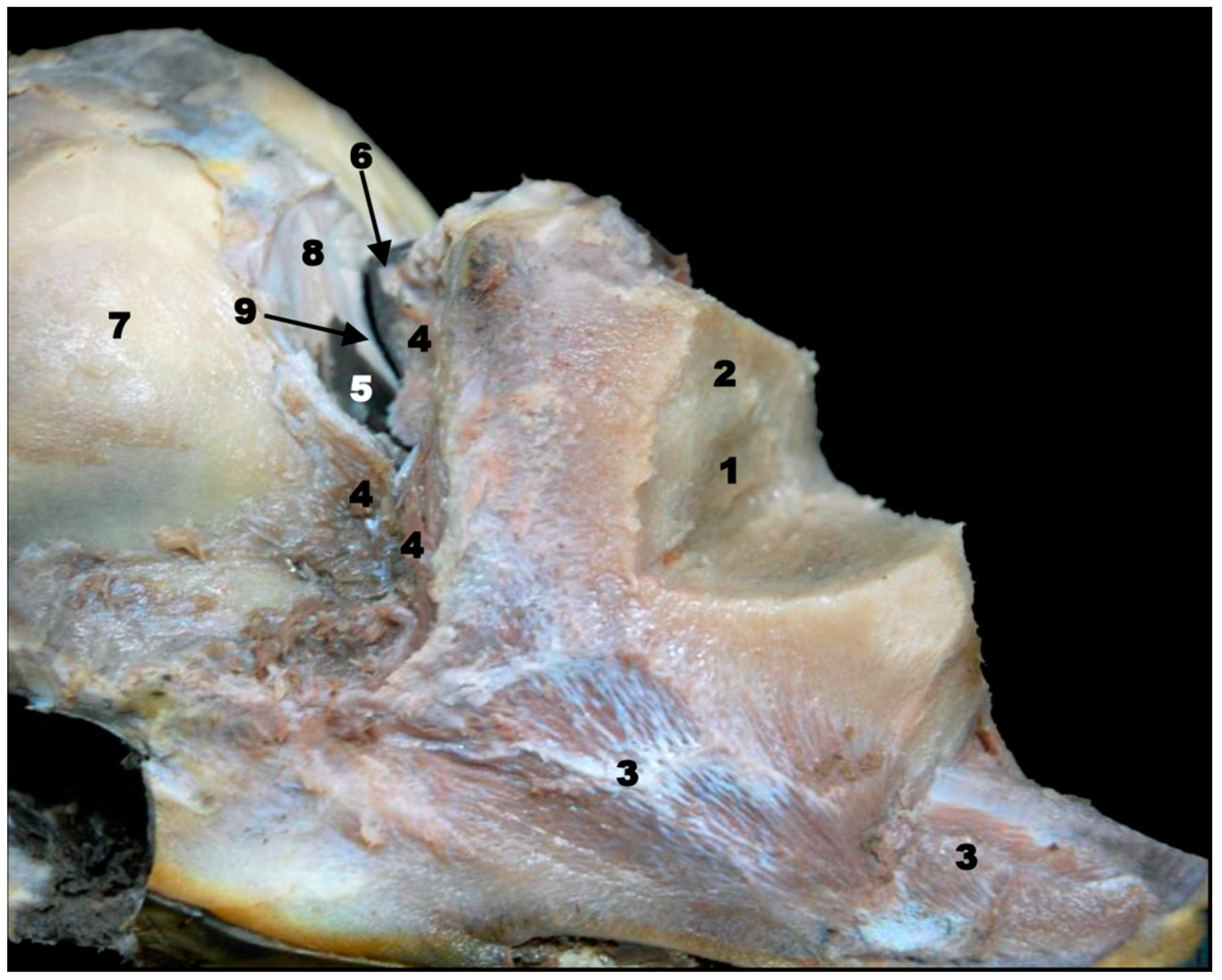
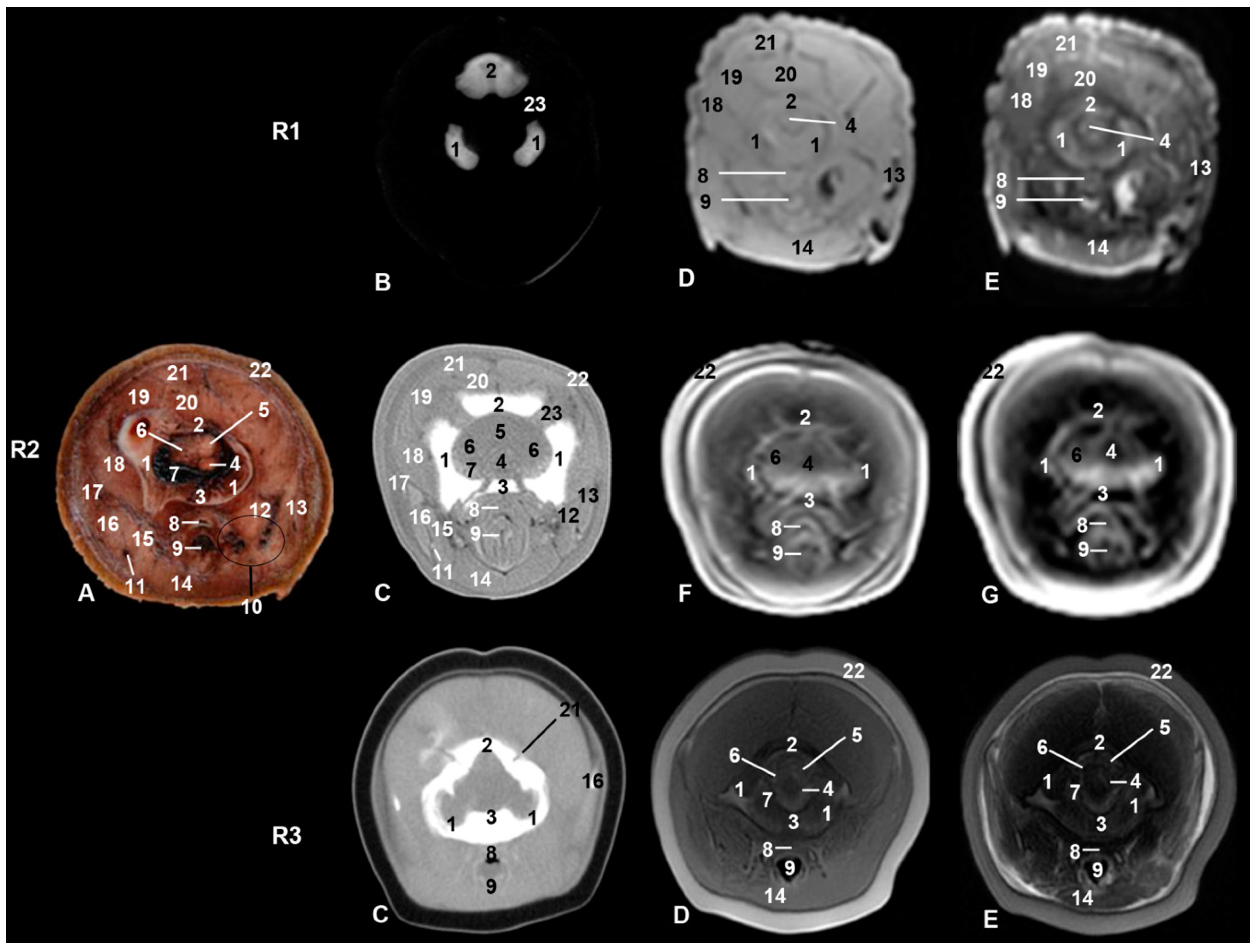
| Stranding Reference and Study Code | Sex, Length, Weight and Estimated Gestation Time [7,25,26] | Anatomical and Imaging Diagnostic Techniques | Preservation Techniques |
|---|---|---|---|
| SCOG | Female fetus, 32.5 cm, 508 g, 4.5 months | MRI, PET/SPECT/CT, | Fixation: formaldehyde 10% |
| CEMMA | |||
| sco1 | |||
| SCOCE1 | Male fetus, 48 cm, 1.535 kg, 7 months | MRI, CT, anatomical head sections | Fixation: formaldehyde 10% |
| CECAM | |||
| sco2 | |||
| SCOCE2 | Female newborn, 95 cm, 10.84 kg | MRI, CT | Freezing × 20 °C |
| CECAM | |||
| sco3 | |||
| SCOCE3 | Male newborn, 85 cm, 9.2 kg | Head dissection | Fixation: formaldehyde 10% |
| CECAM | |||
| sco4 | |||
| SCOMU | Adult female, 1.91 cm, 63.65 kg | Head dissection | Embalming: formaldehyde, glycerine, isopropyl alcohol, phenol |
| CRFS | |||
| sco5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García de los Ríos y Loshuertos, Á.; Arencibia Espinosa, A.; Soler Laguía, M.; Gil Cano, F.; Martínez Gomariz, F.; López Fernández, A.; Ramírez Zarzosa, G. A Study of the Head during Prenatal and Perinatal Development of Two Fetuses and One Newborn Striped Dolphin (Stenella coeruleoalba, Meyen 1833) Using Dissections, Sectional Anatomy, CT, and MRI: Anatomical and Functional Implications in Cetaceans and Terrestrial Mammals. Animals 2019, 9, 1139. https://doi.org/10.3390/ani9121139
García de los Ríos y Loshuertos Á, Arencibia Espinosa A, Soler Laguía M, Gil Cano F, Martínez Gomariz F, López Fernández A, Ramírez Zarzosa G. A Study of the Head during Prenatal and Perinatal Development of Two Fetuses and One Newborn Striped Dolphin (Stenella coeruleoalba, Meyen 1833) Using Dissections, Sectional Anatomy, CT, and MRI: Anatomical and Functional Implications in Cetaceans and Terrestrial Mammals. Animals. 2019; 9(12):1139. https://doi.org/10.3390/ani9121139
Chicago/Turabian StyleGarcía de los Ríos y Loshuertos, Álvaro, Alberto Arencibia Espinosa, Marta Soler Laguía, Francisco Gil Cano, Francisco Martínez Gomariz, Alfredo López Fernández, and Gregorio Ramírez Zarzosa. 2019. "A Study of the Head during Prenatal and Perinatal Development of Two Fetuses and One Newborn Striped Dolphin (Stenella coeruleoalba, Meyen 1833) Using Dissections, Sectional Anatomy, CT, and MRI: Anatomical and Functional Implications in Cetaceans and Terrestrial Mammals" Animals 9, no. 12: 1139. https://doi.org/10.3390/ani9121139
APA StyleGarcía de los Ríos y Loshuertos, Á., Arencibia Espinosa, A., Soler Laguía, M., Gil Cano, F., Martínez Gomariz, F., López Fernández, A., & Ramírez Zarzosa, G. (2019). A Study of the Head during Prenatal and Perinatal Development of Two Fetuses and One Newborn Striped Dolphin (Stenella coeruleoalba, Meyen 1833) Using Dissections, Sectional Anatomy, CT, and MRI: Anatomical and Functional Implications in Cetaceans and Terrestrial Mammals. Animals, 9(12), 1139. https://doi.org/10.3390/ani9121139





