In Vitro Evaluation of Different Dietary Methane Mitigation Strategies
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Treatments
2.1.1. Experiment 1
2.1.2. Experiment 2
2.2. In Vitro Incubations
2.3. In Vitro Gas Production Measurements and Sampling
2.4. Chemical Analysis
2.5. Calculations
2.6. Statistical Analysis
3. Results
3.1. Experiment 1
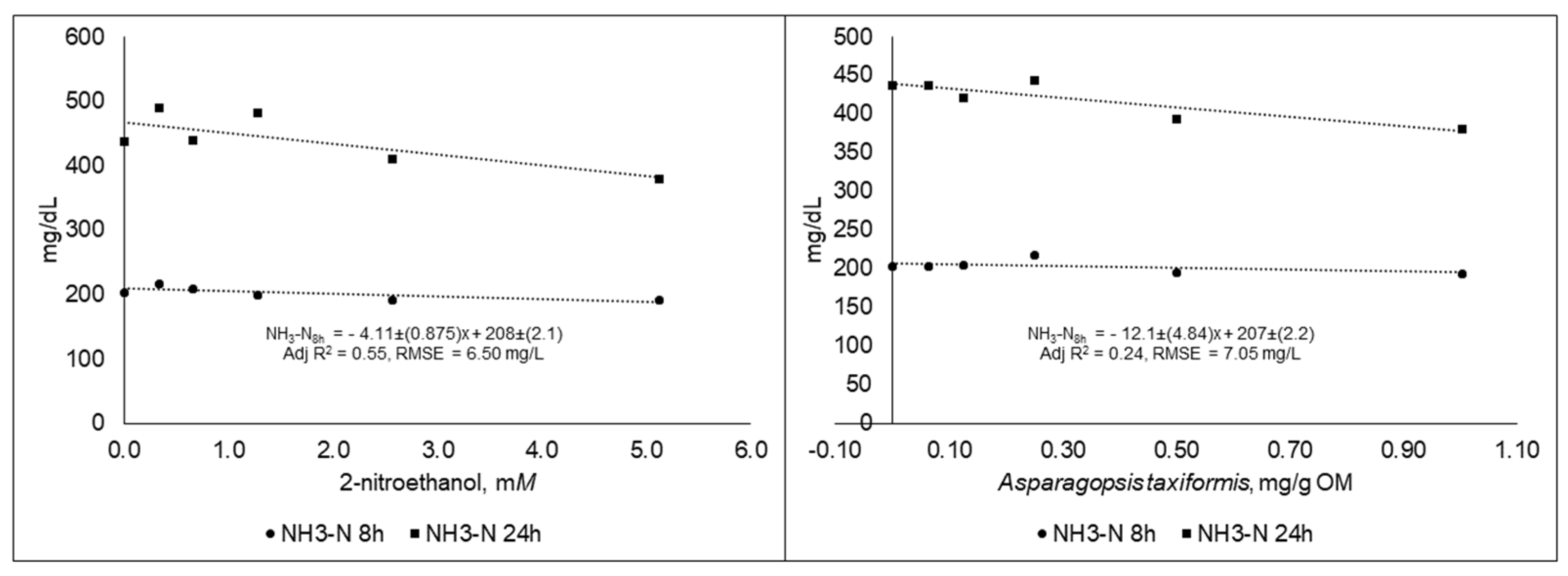
3.2. Experiment 2
4. Discussion
4.1. In Vitro Measurements of CH4 Production in Ruminants
4.2. Dietary Strategies to Decrease CH4 Production from Ruminants
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. Food and Agriculture Organization of the United Nations. The State of Food Security and Nutrition in the World 2018. Available online: http://www.fao.org/3/i9553en/i9553en.pdf (accessed on 10 August 2019).
- IPCC. Intergovernmental Panel on Climate Change. Climate Change 2013. Available online: https://www.ipcc.ch/site/assets/uploads/2018/03/WG1AR5_SummaryVolume_FINAL.pdf (accessed on 10 August 2019).
- Shindell, D. The social cost of atmospheric release. Clim. Chang. 2015, 130, 313–326. [Google Scholar] [CrossRef]
- Howarth, R.W. Ideas and perspectives: Is shale gas a major driver of recent increase in global atmospheric methane? Biogeosciences 2019, 16, 3033–3046. [Google Scholar] [CrossRef]
- Petrenko, V.V.; Smith, A.M.; Schaefer, H.; Riedel, K.; Brook, E.; Baggenstos, D.; Hart, C.; Hua, Q.; Buizert, C.; Schilt, A.; et al. Minimal geological methane emissions during the Younger Dryas-Preboreal abrupt warming event. Nature 2017, 548, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Henderson, G.; Cox, F.; Ganesh, S.; Jonker, A.; Young, W.; Global Rumen Census Collaborators; Janssen, P.H. Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Rep. 2015, 5, 14567. [Google Scholar] [CrossRef]
- Johnson, K.A.; Johnson, D.E. Methane emissions from cattle. J. Anim. Sci. 1998, 73, 2483–2492. [Google Scholar] [CrossRef]
- Ramin, M.; Huhtanen, P. Development of equations for predicting methane emissions from ruminants. J. Dairy Sci. 2013, 96, 2476–2493. [Google Scholar] [CrossRef]
- Knapp, J.R.; Laur, G.L.; Vadas, P.A.; Weiss, W.P.; Tricarico, W. Invited review: Enteric methane in dairy cattle production: Quantifying the opportunities and impact of reducing emissions. J. Dairy Sci. 2014, 97, 3231–3326. [Google Scholar] [CrossRef]
- Shindell, D.; Kuylenstierna, J.C.; Vignati, E.; van Dingenen, R.; Amann, M.; Klimont, Z.; Anenberg, S.C.; Muller, N.; Janssens-Maenhout, G.; Raes, F.; et al. Simultaneously mitigating near-term climate change and improving human health and food security. Science 2012, 335, 183–189. [Google Scholar] [CrossRef]
- IPCC. Intergovernmental Panel on Climate Change 2018. Available online: https://report.ipcc.ch/sr15/pdf/sr15_spm_final.pdf (accessed on 10 August 2019).
- Hristov, A.N.; Oh, J.; Giallongo, F.; Frederick, T.W.; Harper, M.T.; Weeks, H.L.; Brabco, A.F.; Moate, P.J.; Deighton, M.H.; Williams, R.O.; et al. An inhibitor persistently decreased enteric methane emission from dairy cows with no negative effect on milk production. Proc. Natl. Acad. Sci. USA 2015, 112, 10663–10668. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; de Nys, R.; Tomkins, N. Effects of marine and freshwater macroalgae on in vitro total gas and methane production. PLoS ONE 2014, 9, e85289. [Google Scholar] [CrossRef]
- Ramin, M.; Huhtanen, P. Development of an in vitro method for determination of methane production kinetics using a fully automated in vitro gas system—A modelling approach. Anim. Feed Sci. Technol. 2012, 174, 190–200. [Google Scholar] [CrossRef]
- Menke, K.H.; Steingass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–25. [Google Scholar]
- Cone, J.W.; Van Gelder, A.H.; Visscher, G.J.W.; Oudshoorn, L. Influence of rumen fluid and substrate concentration on fermentation kinetics measured with a fully automated time related gas production apparatus. Anim. Feed Sci. Technol. 1996, 61, 113–128. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005; pp. 24–56. [Google Scholar]
- Mertens, D.R. Gravimetric determination of amylase-treated neutral detergent fiber in feeds using refluxing in beakers or crucibles: Collaborative study. J. AOAC Int. 2002, 85, 1217–1240. [Google Scholar]
- Krizsan, S.J.; Rinne, M.; Nyholm, L.; Huhtanen, P. New recommendations for the ruminal in situ determination of indigestible neutral detergent fibre. Anim. Feed Sci. 2015, 205, 31–41. [Google Scholar] [CrossRef]
- Puhakka, L.; Jaakkola, S.; Simpura, I.; Kokkonen, T.; Vanhatalo, A. Effects of replacing rapeseed meal with fava bean at two concentrate crude protein levels on feed intake, nutrient digestion, and milk production in cows fed grass silage-based diets. J. Dairy Sci. 2016, 99, 7993–8006. [Google Scholar] [CrossRef]
- Roque, B.M.; Salwen, J.K.; Kinley, R.; Kebreab, E. Inclusion of Asparagopsis armata in lactating dairy cows’ diet reduces enteric methane emission by over 50 percent. J. Clean. Prod. 2019, 234, 132–138. [Google Scholar] [CrossRef]
- Broderick, G.A.; Huhtanen, P.; Ahvenjärvi, S.; Reynal, S.M.; Shingfield, J. Quantifying ruminal nitrogen metabolism using the omasal sampling technique in cattle—A meta-analysis. J. Dairy Sci. 2010, 93, 3216–3230. [Google Scholar] [CrossRef]
- Chang, J.; Peng, S.; Ciais, P.; Saunois, M.; Dangal, S.R.S.; Herrero, M.; Havlík, P.; Tian, H.; Bousquet, P. Revisiting enteric methane emissions from domestic ruminants and their δ13CCH4 source signature. Nat. Commun. 2019, 10, e3420. [Google Scholar] [CrossRef]
- Kingston-Smith, A.H.; Edwards, J.E.; Huws, S.A.; Kim, E.J.; Abberton, M. Plant-based strategies towards minimising ‘livestock’s long shadow’. Proc. Nutr. Soc. 2010, 69, 613–620. [Google Scholar] [CrossRef]
- Li, B.; Fikse, W.F.; Løvendahl, P.; Lassen, J.; Lidauer, M.H.; Mäntysaari, P.; Berglund, B. Genetic heterogeneity of feed intake, energy-corrected milk and body weight across lactation in Holstein, Nordic Red, and Jersey cows. J. Dairy Sci. 2018, 101, 10011–10021. [Google Scholar] [CrossRef] [PubMed]
- Grandl, F.; Furger, M.; Kreuzer, M.; Zehetmeier, M. Impact of longevity on greenhouse gas emissions and profitability of individual dairy cows analysed with different system boundaries. Animal 2019, 13, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Agnew, R.E.; Gordon, F.J.; Porter, M.G. Prediction of methane energy output in dairy and beef cattle offered grass silage-based diets. Livest. Prod. Sci. 2000, 64, 253–263. [Google Scholar] [CrossRef]
- Danielsson, R.; Ramin, M.; Bertilsson, J.; Lund, P.; Huhtanen, P. Evaluation of an in vitro system for predicting methane production in vivo. J. Dairy. Sci. 2017, 100, 8881–8894. [Google Scholar] [CrossRef] [PubMed]
- Hristov, N.; Oh, J.; Lee, C.; Meinen, R.; Montes, F.; Ott, T.; Firkins, J.; Rotz, A.; Dell, C.; Adesogan, A.; et al. Mitigation of Greenhouse Gas Emissions in Livestock Production—A Review of Technical Options for Non-CO2 emissions; FAO Animal Production and Health: Rome, Italy, 2013; pp. 10–60. [Google Scholar]
- Pinares-Patiño, C.S.; Ulyatt, M.J.; Lassey, K.R.; Barry, T.N.; Holmes, C.W. Rumen function and digestion parameters associated with differences between sheep in methane emissions when fed chaffed lucerne hay. J. Agric. Sci. 2003, 140, 205–214. [Google Scholar] [CrossRef]
- Pinares-Patiño, C.S.; Ebrahimi, S.H.; McEwan, J.C.; Clark, H.; Luo, D. Is rumen retention time implicated in sheep differences in methane emission? In Proceedings of the New Zealand Society of Animal Production, Wellington, New Zeland, 24–26 June 2011; Volume 71, pp. 219–222. [Google Scholar]
- Goopy, J.P.; Donaldson, A.; Hegarty, R.; Vercoe, P.E.; Haynes, F.; Barnett, M.; Oddy, V.H. Low-methane yield sheep have smaller rumens and shorter rumen retention time. Br. J. Nutr. 2014, 111, 578–585. [Google Scholar] [CrossRef]
- Bayat, A.R.; Tapio, I.; Vilkki, J.; Shingfield, K.J.; Leskinen, H. Plant oil supplements reduce methane emissions and improve milk fatty acid composition in dairy cows fed grass silage-based diets without affecting milk yield. J. Dairy Sci. 2018, 101, 1136–1151. [Google Scholar] [CrossRef]
- Franco, M.O.; Krizsan, S.J.; Ramin, M.; Spörndly, R.; Huhtanen, P. In vitro evaluation of agro-industrial by-products replacing soybean meal in two different basal diets for ruminants. In Proceedings of the 8th Nordic Feed Science Conference, Uppsala, Sweden, 13–14 June 2017; pp. 170613–170614. [Google Scholar]
- Fant, P.; Ramin, M.; Jaakkola, S.; Grimberg, Å.; Carlsson, A.S.; Huhtanen, P. Effects of different barley and oat varieties on methane production, digestibility and fermentation pattern in vitro. J. Dairy. Sci. 2019, in press. [Google Scholar] [CrossRef]
- O’Mara, F.P.; Fitzgerald, J.J.; Murphy, J.J.; Rath, M. The effect on milk production of replacing grass silage with maize silage in the diet of dairy cows. Livest. Prod. Sci. 1998, 55, 79–87. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; Kreuzer, M.; O’Mara, F.; McAllister, T.A. Nutritional management for enteric methane abatement: A review. Aust. J. Exp. Agric. 2008, 48, 21–27. [Google Scholar] [CrossRef]
- Haque, M.N. Dietary manipulation: A sustainable way to mitigate methane emissions from ruminants. J. Anim. Sci. Technol. 2018, 60, 15. [Google Scholar] [CrossRef] [PubMed]
- Vanhatalo, A.; Kuoppala, K.; Ahvenjärvi, S.; Rinne, M. Effects of feeding grass or red clover silage cut at two maturity stages in dairy cows. Nitrogen metabolism and supply of amino acids. J. Dairy Sci. 2009, 92, 5620–5633. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Villa, A.; O’Brian, M.; López, S.; Boland, T.M.; O’Kiely, P. In vitro rumen methane output of red clover and perennial ryegrass assayed using the gas production technique (GPT). Anim. Feed Sci. Tech. 2011, 168, 152–164. [Google Scholar] [CrossRef]
- Allison, M.J.; Reddy, C.A.; Cook, H.M. The effects of nitrate and nitrite on VFA and CH4 production by rumen microbes. J. Anim.l Sci. 1981, 53, 391–399. [Google Scholar]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; Nys, R.; Tomkins, N. Identification of bioactives from the red seaweed Asparagopsis taxiformis that promote antimethanogenic activity in vitro. J. Appl. Phycol. 2016, 28, 3117–3126. [Google Scholar] [CrossRef]
- Vucko, M.J.; Magnusson, M.; Kinley, R.D.; Villart, C.; Nys, R. The effects of processing on the in vitro antimethanogenic capacity and concentration of secondary metabolites of Asparagopsis taxiformis. J. Appl. Phycol. 2017, 29, 1577–1586. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; de Nys, R.; Tomkins, N. Dose-response effects of Asparagopsis taxiformis and Oedogonium sp. on in vitro fermentation and methane production. J. Appl. Phycol. 2016, 28, 1443–1452. [Google Scholar] [CrossRef]
- Li, X.; Norman, H.C.; Kinley, R.D.; Laurence, M.; Wilmot, M.; Bender, H.; Nys, R.; Tomkins, N. Asparagopsis taxiformis decreases enteric methane production from sheep. Anim. Prod. Sci. 2018, 58, 681–688. [Google Scholar] [CrossRef]
- Stefenoni, H.; Räisänen, S.; Melgar, A.; Lage, C.; Young, M.; Hristov, A. Dose-response effect of the macroalga Asparagopsis taxiformis on enteric methane emission in lactating dairy cows. In Proceedings of the American Dairy Science Association Annual Meeting, Cincinnati, OH, USA, 23–26 June 2019; pp. W163, 378. [Google Scholar]
- Zhang, D.F.; Yan, H.J. In vitro ruminal methanogenesis of a hay-rich substrate in response to different combination supplements of nitrocompounds; pyromellitic diimide and 2-bromoethanesulphonate. Anim. Feed Sci. Technol. 2011, 163, 20–23. [Google Scholar] [CrossRef]
- Andries, J.I.; Buysse, F.X.; DeBrabander, D.L.; Cottyn, B.G. Isoacids in ruminant nutrition: Their role in ruminal and intermediary metabolism and possible influences on performances—A review. Anim. Feed Sci. Technol. 1987, 18, 169–180. [Google Scholar] [CrossRef]
- Newbold, J.R.; van Zijderveld, S.M.; Hulshof, R.B.A.; Fokkink, W.B.; Leng, R.A.; Terencio, P.; Powers, W.J.; van Adrichem, P.S.J.; Paton, N.D.; Perdok, H.B. The effect of incremental levels of dietary nitrate on methane emissions in Holstein steers and performance in Nelore bulls. J. Anim. Sci. 2014, 92, 5032–5040. [Google Scholar] [CrossRef] [PubMed]
- Klop, G.; Hatew, B.; Bannink, A.; Dijkstra, J. Feeding nitrate and docosahexaenoic acid affects enteric methane production and milk fatty acid composition in lactating dairy cows. J. Dairy Sci. 2016, 99, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Olijhoek, D.W.; Hellwing, A.L.F.; Brask, M.; Weisbjerg, M.R.; Højberg, O.; Larsen, M.K.; Dijkstra, E.J.; Erlandsen, E.J.; Lund, P. Effect of dietary nitrate level on enteric methane production, hydrogen emission, rumen fermentation, and nutrient digestibility in dairy cows. J. Dairy Sci. 2016, 99, 6191–6205. [Google Scholar] [CrossRef] [PubMed]
- Department of Climate Change, Commonwealth Government of Australia. The Potential of Feeding Nitrate to Reduce Enteric Methane Production in Ruminants. Available online: http://www.penambulbooks.com/ (accessed on 14 September 2019).
- Zhou, Z.; Meng, Q.; Yu, Z. Effects of methanogenic inhibitors on methane production and abundances of methanogens and cellulolytic bacteria in in vitro ruminal cultures. Appl. Environ. Microbiol. 2011, 77, 2634–2639. [Google Scholar] [CrossRef]
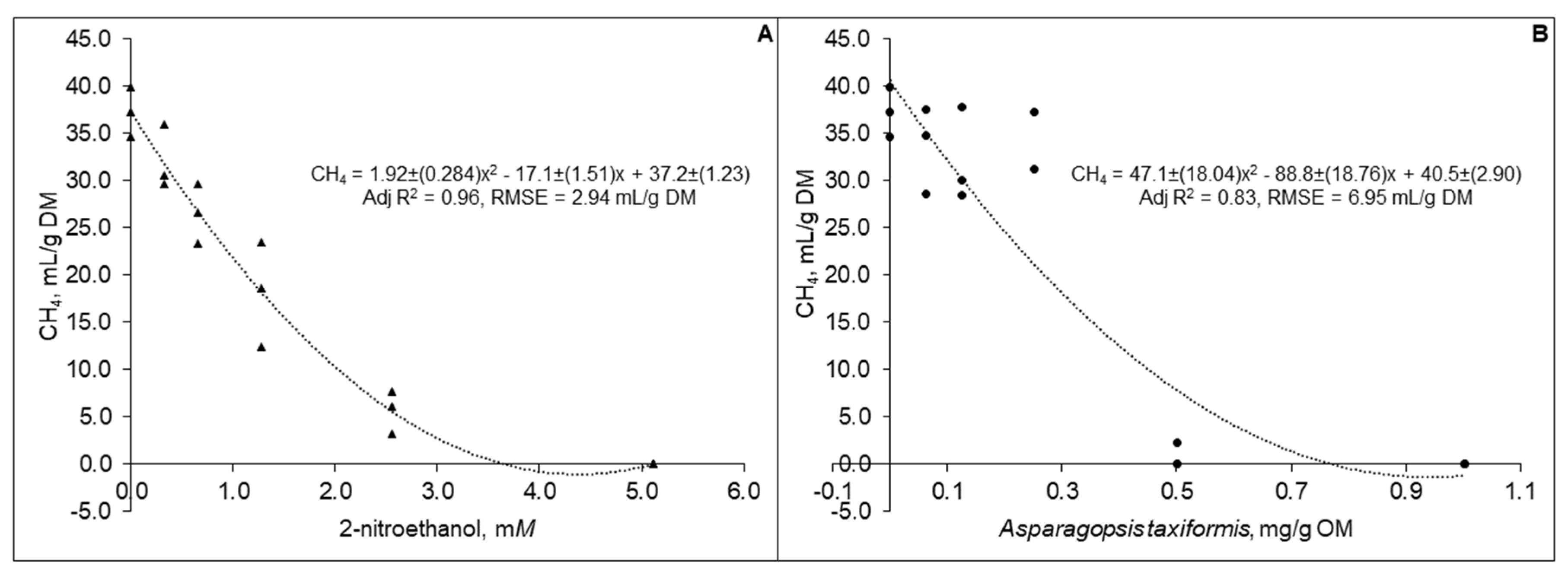
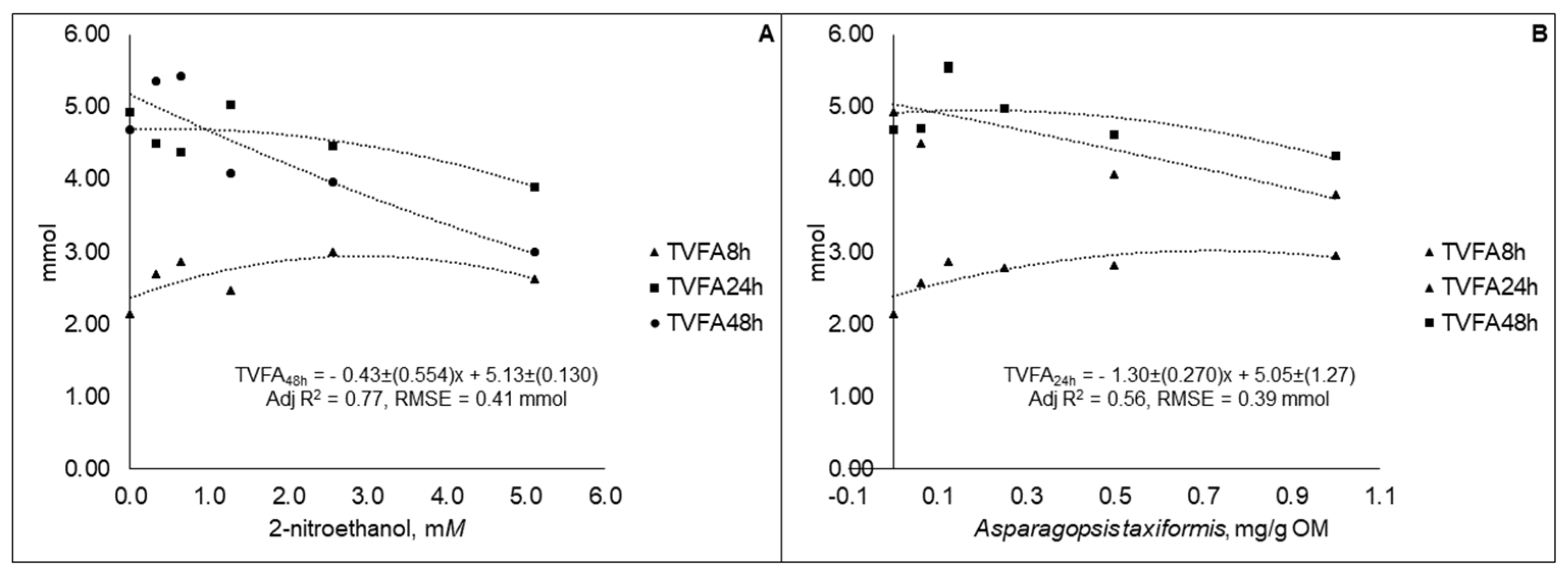
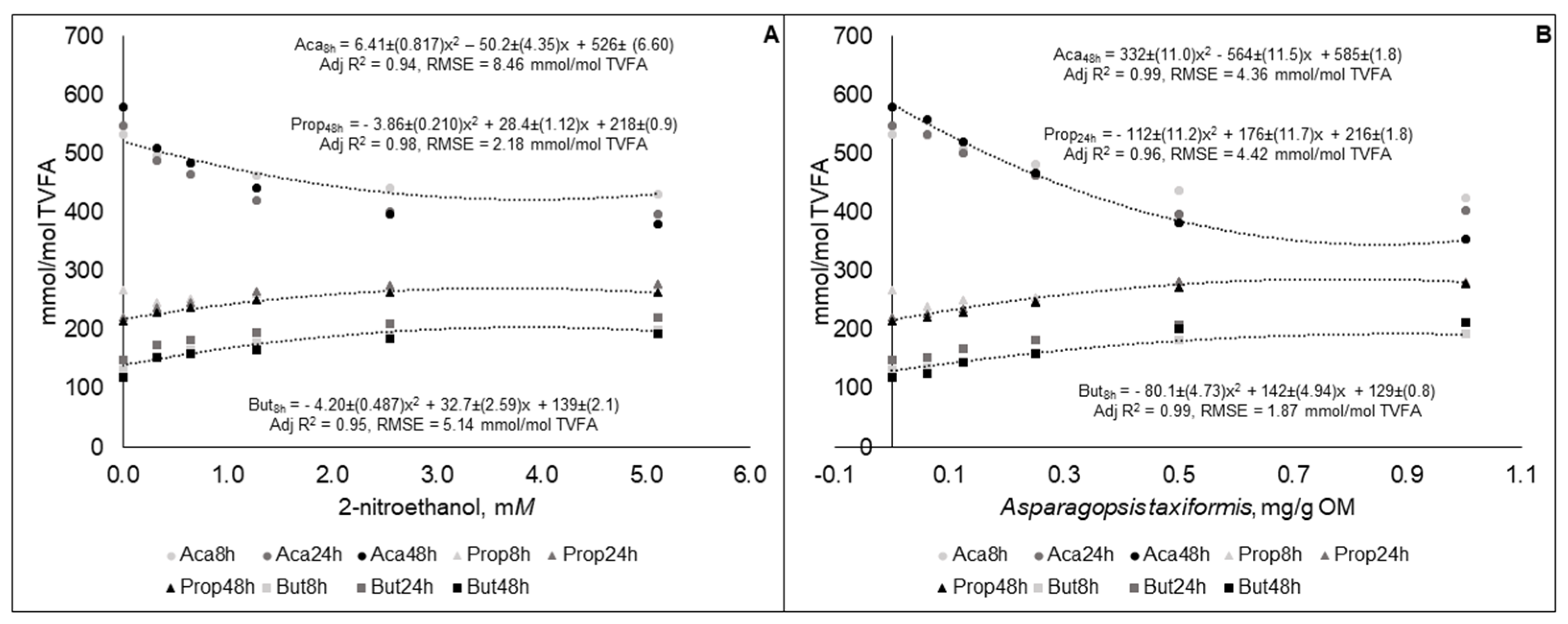
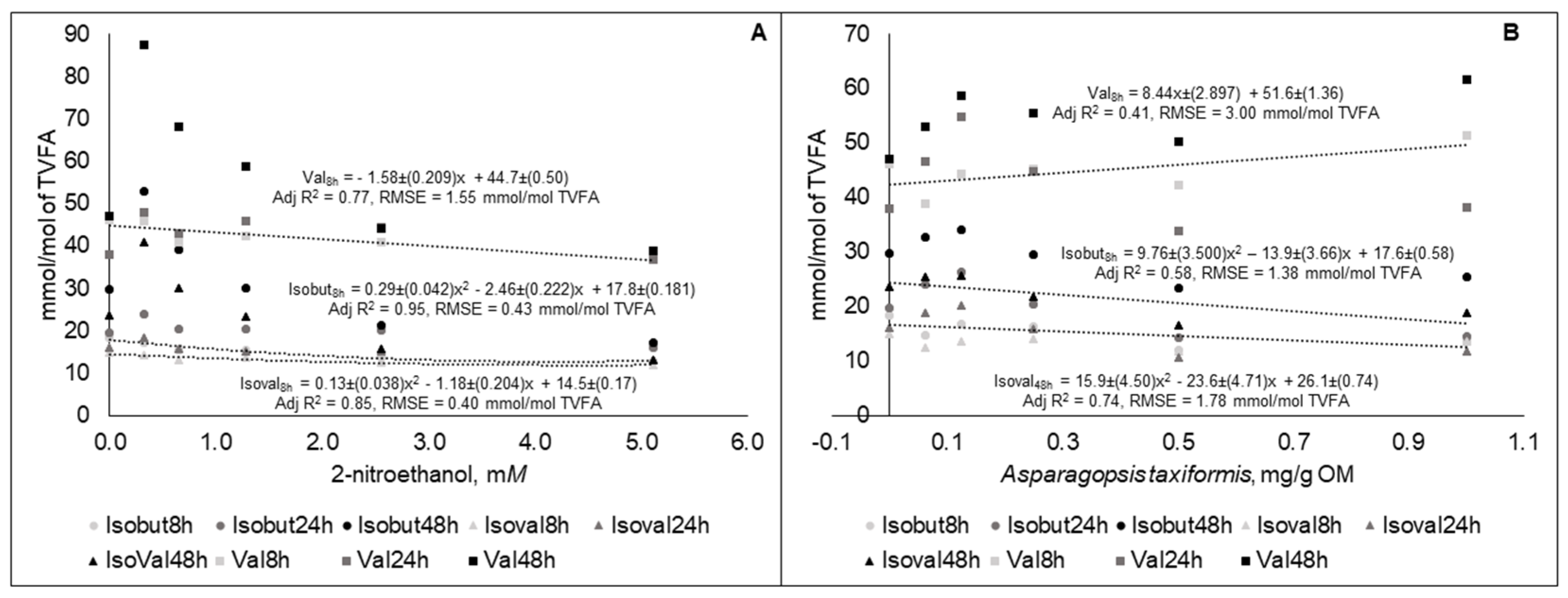
| Treatments | Levels | |
|---|---|---|
| Chemical compounds | ||
| 2-nitroethanol | 5 mM | 10 mM |
| Nitrate | None 1 | 21 g/kg DM 2 |
| Propynoic acid | 2 mM | 4 mM |
| Ferulic acid | 10 mM | 20 mM |
| p-Coumaric acid | 10 mM | 20 mM |
| Bromoform | 1.5 mg/g DM | 3 mg/g DM |
| Plant-derived treatments | ||
| Rowan berries | 50 g/kg DM | 100 g/kg DM |
| Fireweed | 50 g/kg DM | 100 g/kg DM |
| Asparagopsis taxiformis | 10 g/kg OM | 20 g/kg OM |
| Potentially CH4-reducing treatments | ||
| Rapeseed oil | 40 g/kg DM | 80 g/kg DM |
| Dried distiller’s grain | 90 g/kg DM | 180 g/kg DM |
| Barley:oat | 175:175 g/kg | 0:350 g/kg |
| Maize silage:grass | 275:275 g/kg 3 | 545:0 g/kg 4 |
| Red clover:grass | 275:275 g/kg | None |
| Lactic acid | 60 g/kg DM | 120 g/kg DM |
| Lactic acid + acetic acid | 80 + 30 g DM | 80 + 60 g DM |
| Treatment | Level | Organic Matter | Crude Protein | Neutral Detergent Fiber |
|---|---|---|---|---|
| Control diet | ----- | 944 | 160 | 387 |
| Rapeseed oil | 40 g/kg DM | 906 | 154 | 372 |
| Rapeseed oil | 80 g/kg DM | 869 | 149 | 356 |
| Dried distiller’s grain | 90 g/kg DM | 946 | 161 | 378 |
| Dried distiller’s grain | 180 g/kg DM | 946 | 181 | 366 |
| Barley: oat | 175:175 g/kg | 944 | 165 | 385 |
| Barley: oat | 0:350 g/kg | 944 | 170 | 383 |
| Maize silage: grass | 275:275 g/kg | 954 | 160 | 355 |
| Maize silage: grass | 545:0 g/kg | 963 | 160 | 323 |
| Red clover: grass | 275:275 g/kg | 932 | 171 | 345 |
| Lactic acid | 60 g/kg DM | 887 | 151 | 364 |
| Lactic acid | 120 g/kg DM | 831 | 143 | 341 |
| Lactic acid + acetic acid | 80 + 30 g/kg DM | 840 | 144 | 345 |
| Lactic acid + acetic acid | 80 + 60 g/kg DM | 812 | 140 | 333 |
| Treatment | CH4 | TOMD | TVFA | Acetate | Propionate | Butyrate | p-Value 1 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CCH4 | CTOMD | CTVFA | CAcetate | CPropionate | CButyrate | |||||||
| Control | 38.7 | 867 | 3.62 | 583 | 237 | 125 | – | – | – | – | – | – |
| 2-nitroethanol | 1.30 | 858 | 3.01 | 440 | 309 | 211 | <0.01 | 0.30 | 0.10 | <0.01 | <0.01 | <0.01 |
| Nitrate 2 | 21.3 | 874 | 2.96 | 619 | 250 | 87 | <0.01 | 0.82 | NA | NA | NA | NA |
| Propynoic acid | 13.9 | 839 | 2.57 | 476 | 297 | 209 | <0.01 | 0.25 | 0.01 | <0.01 | <0.01 | <0.01 |
| Ferulic acid | 27.5 | 859 | 3.54 | 597 | 229 | 109 | 0.06 | 0.71 | 0.82 | 0.62 | 0.68 | 0.32 |
| p-Coumaric acid | 24.2 | 763 | 3.01 | 492 | 176 | 121 | 0.01 | <0.01 | 0.10 | <0.01 | <0.01 | 0.82 |
| Bromoform | 2.00 | 822 | 2.30 | 436 | 270 | 261 | <0.01 | <0.01 | <0.01 | <0.01 | 0.08 | <0.01 |
| Fireweed | 38.1 | 858 | 4.01 | 583 | 226 | 140 | 0.34 | 0.69 | 0.24 | 0.99 | 0.55 | 0.34 |
| Rowan berries | 28.9 | 843 | 3.71 | 586 | 241 | 117 | 0.96 | 0.35 | 0.80 | 0.93 | 0.82 | 0.64 |
| A. taxiformis | 0.20 | 852 | 3.61 | 418 | 327 | 184 | <0.01 | 0.97 | 0.98 | <0.01 | <0.01 | <0.01 |
| Rapeseed oil | 38.2 | 896 | 4.04 | 600 | 217 | 128 | 0.82 | 0.04 | 0.24 | 0.56 | 0.28 | 0.83 |
| Dried distiller’s grain | 35.3 | 877 | 3.74 | 549 | 241 | 152 | 0.43 | 0.48 | 0.73 | 0.66 | 0.91 | 0.33 |
| Barley: oat | 37.4 | 863 | 3.66 | 596 | 225 | 124 | 0.73 | 0.73 | 0.89 | 0.65 | 0.51 | 0.98 |
| Maize silage: grass | 30.7 | 846 | 3.71 | 577 | 240 | 136 | 0.61 | 0.56 | 0.80 | 0.84 | 0.87 | 0.48 |
| Red clover: grass | 47.9 | 882 | 3.16 | 599 | 234 | 129 | 0.20 | 0.53 | 0.27 | 0.62 | 0.88 | 0.85 |
| Lactic acid | 34.1 | 866 | 3.65 | 516 | 271 | 158 | 0.25 | 0.54 | 0.93 | 0.03 | 0.07 | 0.05 |
| Lactic acid + acetic acid | 35.2 | 885 | 3.34 | 598 | 224 | 135 | 0.34 | 0.11 | 0.43 | 0.59 | 0.49 | 0.53 |
| SEM | 1.75 | 4.3 | 0.120 | 7.2 | 4.2 | 5.5 | – | – | – | – | – | – |
| Treatments | Isobutyrate | Isovalerate | Valerate | NH3-N | p-Value 1 | |||
|---|---|---|---|---|---|---|---|---|
| CIsobutyrate | CIsovalerate | CValerate | CNH3-N | |||||
| Control | 10.9 | 0.61 | 21.4 | 282 | – | – | – | – |
| 2-nitroethanol | 5.16 | 2.77 | 16.4 | 436 | 0.80 | <0.01 | 0.14 | 0.39 |
| Nitrate 2 | 6.63 | 4.01 | 16.43 | 270 | NA | NA | NA | NA |
| Propynoic acid | 8.34 | 2.59 | 0.77 | 311 | 0.91 | <0.01 | <0.01 | 0.25 |
| Ferulic acid | 14.4 | 5.23 | 20.0 | 320 | 0.88 | 0.03 | 0.67 | 0.98 |
| p-Coumaric acid | 165 | 4.23 | 22.6 | 263 | <0.01 | <0.01 | 0.73 | 0.81 |
| Bromoform | 0.00 | 0.00 | 28.7 | 302 | 0.64 | <0.01 | 0.03 | 0.17 |
| Fireweed | 10.8 | 5.97 | 18.5 | 304 | 0.99 | 0.20 | 0.38 | 0.78 |
| Rowan berries | 10.1 | 5.85 | 21.5 | 289 | 0.97 | 0.15 | 0.98 | 0.81 |
| A. taxiformis | 5.95 | 5.33 | 35.4 | 354 | 0.83 | 0.04 | 0.00 | 0.52 |
| Rapeseed oil | 13.3 | 8.32 | 19.1 | 319 | 0.92 | 0.08 | 0.49 | 0.25 |
| Dried distiller’s grain | 11.5 | 6.87 | 20.7 | 301 | 0.98 | 0.91 | 0.83 | 0.07 |
| Barley: oat | 12.5 | 6.98 | 18.5 | 359 | 0.95 | 0.98 | 0.38 | 0.75 |
| Maize: grass | 10.0 | 6.10 | 17.8 | 281 | 0.97 | 0.26 | 0.28 | 0.20 |
| Red clover: grass | 9.04 | 5.23 | 14.6 | 306 | 0.94 | 0.06 | 0.08 | 0.45 |
| Lactic acid | 9.30 | 5.29 | 21.3 | 323 | 0.94 | 0.03 | 0.97 | 0.86 |
| Lactic acid + acetic acid | 8.95 | 4.61 | 16.7 | 282 | 0.93 | <0.01 | 0.16 | 0.77 |
| SEM | 4.426 | 0.370 | 0.87 | 12.2 | – | – | – | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chagas, J.C.; Ramin, M.; Krizsan, S.J. In Vitro Evaluation of Different Dietary Methane Mitigation Strategies. Animals 2019, 9, 1120. https://doi.org/10.3390/ani9121120
Chagas JC, Ramin M, Krizsan SJ. In Vitro Evaluation of Different Dietary Methane Mitigation Strategies. Animals. 2019; 9(12):1120. https://doi.org/10.3390/ani9121120
Chicago/Turabian StyleChagas, Juana C., Mohammad Ramin, and Sophie J. Krizsan. 2019. "In Vitro Evaluation of Different Dietary Methane Mitigation Strategies" Animals 9, no. 12: 1120. https://doi.org/10.3390/ani9121120
APA StyleChagas, J. C., Ramin, M., & Krizsan, S. J. (2019). In Vitro Evaluation of Different Dietary Methane Mitigation Strategies. Animals, 9(12), 1120. https://doi.org/10.3390/ani9121120





