Development and Implementation of Baseline Welfare Assessment Protocol for Captive Breeding of Wild Ungulate—Punjab Urial (Ovis vignei punjabiensis, Lydekker 1913)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Development of Welfare Assessment Protocol
3.1.1. Lack of Prolonged Appetite
3.1.2. Lack of Prolonged Thirst
3.1.3. Lack of Minerals
3.1.4. Thermal Ease
3.1.5. Easiness in Movement
3.1.6. Standard Enclosures
3.1.7. Lack of Injuries
3.1.8. Lack of Disease
3.1.9. Displaying Social Behavior
3.1.10. Group Dynamics
3.1.11. Display of Other Behavior
3.1.12. Good Human–Animal Affiliations
3.2. Application of Punjab Urial Welfare Assessment Protocol
3.2.1. Lack of Prolonged Appetite
3.2.2. Lack of Prolonged Thirst
3.2.3. Lack of Minerals
3.2.4. Thermal Ease
3.2.5. Easiness in Movement
3.2.6. Standard Enclosures
3.2.7. Lack of Injuries
3.2.8. Lack of Diseases
3.2.9. Displaying Social Behavior
3.2.10. Group Dynamics
3.2.11. Display of Other Behavior
3.2.12. Good Human–Animal Affiliations
3.3. Overall Comparison
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Salas, M.; Temple, D.; Abáigar, T.; Cuadrado, M.; Delclaux, M.; Enseñat, C.; Almagro, V.; Martínez-Nevado, E.; Quevedo, M.Á.; Carbajal, A.; et al. Aggressive behavior and hair cortisol levels in captive Dorcas gazelles (Gazella dorcas) as animal-based welfare indicators. Zoo Biol. 2016, 35, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Kleiman, D.; Allen, M.; Thompson, K.; Lumpkin, S. Wild mammals in captivity: Principles and techniques. Biol. Conserv. 1998, 2, 232. [Google Scholar]
- Witzenberger, K.A.; Hochkirch, A. Ex situ conservation genetics: A review of molecular studies on the genetic consequences of captive breeding programmes for endangered animal species. Biodivers. Conserv. 2011, 20, 1843–1861. [Google Scholar] [CrossRef]
- Williams, S.E.; Hoffman, E.A. Minimizing genetic adaptation in captive breeding programs: A review. Biol. Conserv. 2009, 142, 2388–2400. [Google Scholar] [CrossRef]
- Gusset, M.; Dick, G. Building a Future for Wildlife? Evaluating the contribution of the world zoo and aquarium community to in situ conservation. Int. Zoo Yearb. 2010, 44, 183–191. [Google Scholar] [CrossRef]
- Liz, P. Beyond Captive Breeding: Re-Introducing Endangered Mammals to the Wild: Zoological Society of London Symposia 62; Gipps, J.H.W., Ed.; Clarendon Press: Oxford, UK, 1991; Volume 26, p. 56. ISBN 0-19-854019-1. [Google Scholar]
- Benefiel, A.C.; Dong, W.K.; Greenough, W.T. Mandatory “Enriched” Housing of Laboratory Animals: The Need for Evidence-based Evaluation. ILAR J. 2005, 46, 95. [Google Scholar] [CrossRef]
- World Association of Zoos and Aquariums. Ethics and Animal Welfare. In Building a Future for Wildlife—The World Zoo and Aquarium Conservation Strategy; Olney, P.J.S., Ed.; WAZA Executive Office: Bern, Switzerland, 2005; pp. 59–64. ISBN 303300427X. [Google Scholar]
- Gosling, S.D. From mice to men: What can we learn about personality from animal research? Psychol. Bull. 2001, 127, 45. [Google Scholar] [CrossRef]
- Salas, M.; Manteca, X.; Abáigar, T.; Delclaux, M.; Enseñat, C.; Martínez-Nevado, E.; Quevedo, M.; Fernández-Bellon, H. Using farm animal welfare protocols as a base to assess the welfare of wild animals in captivity Case study: Dorcas gazelles (Gazella dorcas). Animals 2018, 8, 111. [Google Scholar] [CrossRef]
- Welfare Quality®. Welfare Quality® Assessment Protocol for Cattle; Welfare Quality® Consortium: Lelystad, The Netherlands, 2009; ISBN 9789078240044. [Google Scholar]
- Ayaz, S.M.; Jamil, A.A.; Khan, M.; Ayaz Qamar, M.F. Behaviour and biology of Ovis orientalis (urial) in Kotal Wildlife Park and Borraka Wildlife sanctuary in Kohat. J. Anim. Pl. Sci. 2012, 22, 29–31. [Google Scholar]
- Damm, G.R.; Franco, N.S. CIC Caprinae Atlas of the World; CIC International Council for Game and Wildlife Conservation: Budakeszi, Hungary, 2014. [Google Scholar]
- Roberts, T.J. The Mammals of Pakistan; Earnst Benn Limited: London, UK, 1977; pp. 206–208. [Google Scholar]
- International Union for Conservation of Nature and Natural Resources. International Red List of Threatened Species 2008; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2008. [Google Scholar]
- Sheikh, K.; Molur, S. Status and Red List of Pakistan Mammals, Based on Conservation Assessment and Management Plan for Mammals; International Union for Conservation of Nature and Natural Resources: Islamabad, Pakistan, 2005; p. 344. [Google Scholar]
- Awan, G.A.; Ahmad, T.A.H.I.R.A.; Festa-Bianchet, M.A.R.C.O. Disease spectrum and mortality of Punjab urial (Ovis vignei punjabiensis) in Kalabagh Game Reserve. Pak. J. Zool. 2005, 37, 175. [Google Scholar]
- International Union for Conservation of Nature and Natural Resources. International Red List of Threatened Species 2002; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2002. [Google Scholar]
- Dwyer, C.; Ruiz, R.; Beltran de Heredia, I.; Canali, E.; Barbieri, S.; Zanella, A. AWIN Welfare Assessment Protocol for Sheep; Animal Welfare Indicators (AWIN): Edinburgh, UK, 2015. [Google Scholar] [CrossRef]
- Mirza, Z.B. Study of Morphology, Distribution, Population, Behaviour, Food and Habitat of Punjab Urial (Ovis orientalis punjabiensis), in Salt Range and Kala Chitta Range, Punjab, Pakistan; Pakistan Museum of Natural History: Islamabad, Pakistan, 1980.
- Botreau, R.; Veissier, I.; Butterworth, A.; Bracke, M.B.; Keeling, L.J. Definition of criteria for overall assessment of animal welfare. Anim. Welf. 2007, 16, 698–705. [Google Scholar]
- Schaller, G.B.; Mirza, Z. On the behaviour of Punjab urial (Ovis orientalis punjabiensis). In The Behaviour of Ungulates and Its Relation to Management; The University Of Calgary: Calgary, AB, Canada, 1974; pp. 306–323. [Google Scholar]
- Fred, K.; Kumarasinghe, J.C. Remarks on body growth and phenotypes in Asian elephant Elephas maximus. Acta Theriol. Suppl. 1998, 5, 135–153. [Google Scholar]
- Audigé, L.; Wilson, P.; Morris, R. A body condition score system and its use for farmed red deer hinds. N. Z. J. Agric. Res. 1998, 41, 545–553. [Google Scholar] [CrossRef]
- Hernandez, A.; Berg, C.; Eriksson, S.; Edstam, L.; Orihuela, A.; Leon, H.; Galina, C. The Welfare Quality® assessment protocol: How can it be adapted to family farming dual purpose cattle raised under extensive systems in tropical conditions? Anim. Welf. 2017, 26, 177–184. [Google Scholar] [CrossRef]
- Michell, A. Sodium and research in farm animals: Problems of requirement, deficit, and excess. Outlook Agric. 1985, 14, 179–182. [Google Scholar] [CrossRef]
- Mason, G.; Rushen, J. Stereotypic Animal Behaviour: Fundamentals and Applications to Welfare; Centre for Agriculture and Bioscience International: Wallingford, UK, 2006. [Google Scholar]
- Underwood, E.J. The mineral nutrition of livestock. Vet. J. 1993, 161, 70–71. [Google Scholar]
- Shackleton, D.M. Social maturation and productivity in bighorn sheep: Are young males incompetent? Appl. Anim. Behav. Sci. 1991, 29, 173–184. [Google Scholar] [CrossRef]
- Roberts, T.J. The Mammals of Pakistan; Oxford University Press: Karachi, Pakistan, 1997; p. 299. [Google Scholar]
- Parrini, F.; Grignolio, S.; Luccarini, S.; Bassano, B.; Apollonio, M. Spatial behaviour of adult male Alpine ibexCapra ibex ibex in the Gran Paradiso National Park, Italy. Acta Theriol. 2003, 48, 411–423. [Google Scholar] [CrossRef]
- Carlstead, K.; Shepherdson, D. Alleviating stress in zoo animals with environmental enrichment. In The Biology of Animal Stress: Basic Principles and Implications for Animal Welfare; Centre for Agriculture and Bioscience International: Wallingford, UK, 2000; pp. 337–354. [Google Scholar]
- America, Z.A. Animal Care & Enclosure Standards and Related Policies. 2016. Available online: http://www.zaa.org (accessed on 12 September 2019).
- Espinosa, J.; López-Olvera, J.R.; Cano-Manuel, F.J.; Fandos, P.; Pérez, J.M.; López-Graells, C.; Ráez-Bravo, A.; Mentaberre, G.; Romero, D.; Soriguer, R.C.; et al. Guidelines for managing captive Iberian ibex herds for conservation purposes. J. Nat. Conserv. 2017, 40, 24–32. [Google Scholar] [CrossRef]
- Jordan, B. Science-based assessment of animal welfare: Wild and captive animals. Rev. Sci. Tech. Off. Int. Epizoot. 2005, 24, 515. [Google Scholar] [CrossRef]
- Popp, J.W. Interspecific aggression in mixed ungulate species exhibits. Zoo Biol. 1984, 3, 211–219. [Google Scholar] [CrossRef]
- Boever, W. Artiodactylids (Artiodactyla): Noninfectious diseases. In Zoo and Wild Animal Medicine, 2nd ed.; Fowler, M.E., Ed.; WB Saunders & Co.: Philadelphia, PA, USA, 1986; pp. 962–964. ISBN 0721610137. [Google Scholar]
- Carpenter, N.S.C.; Helmick, K.; Meehan, T.; Murray, M.; Smith, J.; Wyatt, J. Guidelines for Zoo and Aquarium Veterinary Medical Programs and Veterinary Hospital, 6th ed.; American Association of Zoo Veterinarians: Yulee, FL, USA, 2016. [Google Scholar]
- Kaurivi, Y.; Laven, R.; Hickson, R.; Stafford, K.; Parkinson, T. Identification of Suitable Animal Welfare Assessment Measures for Extensive Beef Systems in New Zealand. Agriculture 2019, 9, 66. [Google Scholar] [CrossRef]
- Battini, M.; Vieira, A.; Barbieri, S.; Ajuda, I.; Stilwell, G.; Mattiello, S. Invited review: Animal-based indicators for on-farm welfare assessment for dairy goats. J. Dairy Sci. 2014, 97, 6625–6648. [Google Scholar] [CrossRef] [PubMed]
- Lovari, S.; Fattorini, N.; Boesi, R.; Bocci, A. Male ruff colour as a rank signal in a monomorphic-horned mammal: Behavioural correlates. Sci. Nat. 2015, 102, 39. [Google Scholar] [CrossRef] [PubMed]
- Mason, G.J. Stereotypies: A critical review. Anim. Behav. 1991, 41, 1015–1037. [Google Scholar] [CrossRef]
- Loijens, L.W.S.; Schouten, W.G.P.; Wiepkema, P.R.; Wiegant, V.M. Brain Opioid Receptor Density Relates to Stereotypies in Chronically Stressed Pigs. Stress. Int. J. Biol. Stress 1999, 3, 17–26. [Google Scholar] [CrossRef]
- Manteca, X.; Salas, M.; Temple, D. Animal-based indicators to assess welfare in zoo animals. CAB Rev. 2016, 11, 1–10. [Google Scholar] [CrossRef]
- Manteca, X.; Salas, M. Concepto de Bienestar Animal; Zawec Zoo Animal Welfare Education Center: Barcelona, Spain, 2015. [Google Scholar]
- Abáigar Ancín, T.; López Jiménez de Rueda, L. Husbandry Guidelines for Captive Breeding and Management of Saharawi Dorcas gazelle (Gazella dorcas neglecta); Spanish National Research Council: Madrid, Spain, 2013; pp. 1–345. [Google Scholar]
- Mononen, J.; Møller, S.H.; Hansen, S.W.; Hovland, A.L.; Koistinen, T.; Lidfors, L.; Malmkvist, J.; Vinke, C.M.; Ahola, L. The Development of on-farm welfare assessment protocols for foxes and mink: The WelFur project. Anim. Welf. UFAW J. 2012, 21, 363. [Google Scholar] [CrossRef]
- Reade, L.S.; Waran, N.K. The modern zoo: How do people perceive zoo animals? Appl. Anim. Behav. Sci. 1996, 47, 109–118. [Google Scholar] [CrossRef]
- Sherwen, S.L.; Hemsworth, P.H. The visitor effect on zoo animals: Implications and opportunities for zoo animal welfare. Animals 2019, 9, 366. [Google Scholar] [CrossRef]
- Palomares, F.; Caro, T.M. Interspecific killing among mammalian carnivores. Am. Nat. 1999, 153, 492–508. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, F.; Sforzi, A.; Lovari, S. Behavioural interference between ungulate species: Roe are not on velvet with fallow deer. Behav. Ecol. Sociobiol. 2010, 65, 875–887. [Google Scholar] [CrossRef]
- Fattorini, N.; Brunetti, C.; Baruzzi, C.; Macchi, E.; Pagliarella, M.C.; Pallari, N.; Lovari, S.; Ferretti, F. Being “hangry”: Food depletion and its cascading effects on social behaviour. Biol. J. Linn. Soc. 2018, 125, 640–656. [Google Scholar] [CrossRef]
- Clegg, I.; Borgerturner, J.; Eskelinen, H. C-Well: The development of a welfare assessment index for captive bottlenose dolphins (Tursiops truncatus). Anim. Welf. 2015, 24, 267–282. [Google Scholar] [CrossRef]
| Welfare Principles | Criteria | Indicators |
|---|---|---|
| Good feeding | 1. Lack of prolonged appetite | 1.1 Body conditions * |
| 2. Lack of prolonged thirst | 2.1 Number of water sources | |
| 2.2 Water availability | ||
| 2.3 Cleanliness of water sources | ||
| 3. Lack of minerals | 3.1 Availability of salt licks | |
| 3.2 Licking objects | ||
| Good housing | 4. Thermal ease | 4.1 Shelter availability |
| 4.2 Shade availability | ||
| 5. Easiness in movement | 5.1 Total area of enclosure | |
| 5.2 Space (m2) offered per animal | ||
| 6. Standard enclosures | 6.1 Fence conditions | |
| 6.2 Fence substratum | ||
| 6.3 Availability of quarantine | ||
| 6.4 Number of quarantines | ||
| Good health | 7. Lack of injuries | 7.1 Integument deformities * |
| 7.2 Lameness * | ||
| 8. Lack of disease | 8.1 Ophthalmic discharge * | |
| 8.2 Nasal discharge * | ||
| 8.3 Labored breathing * | ||
| 8.4 Diarrhea * | ||
| 8.5 Availability of veterinarian | ||
| 8.6 Availability of veterinary facility | ||
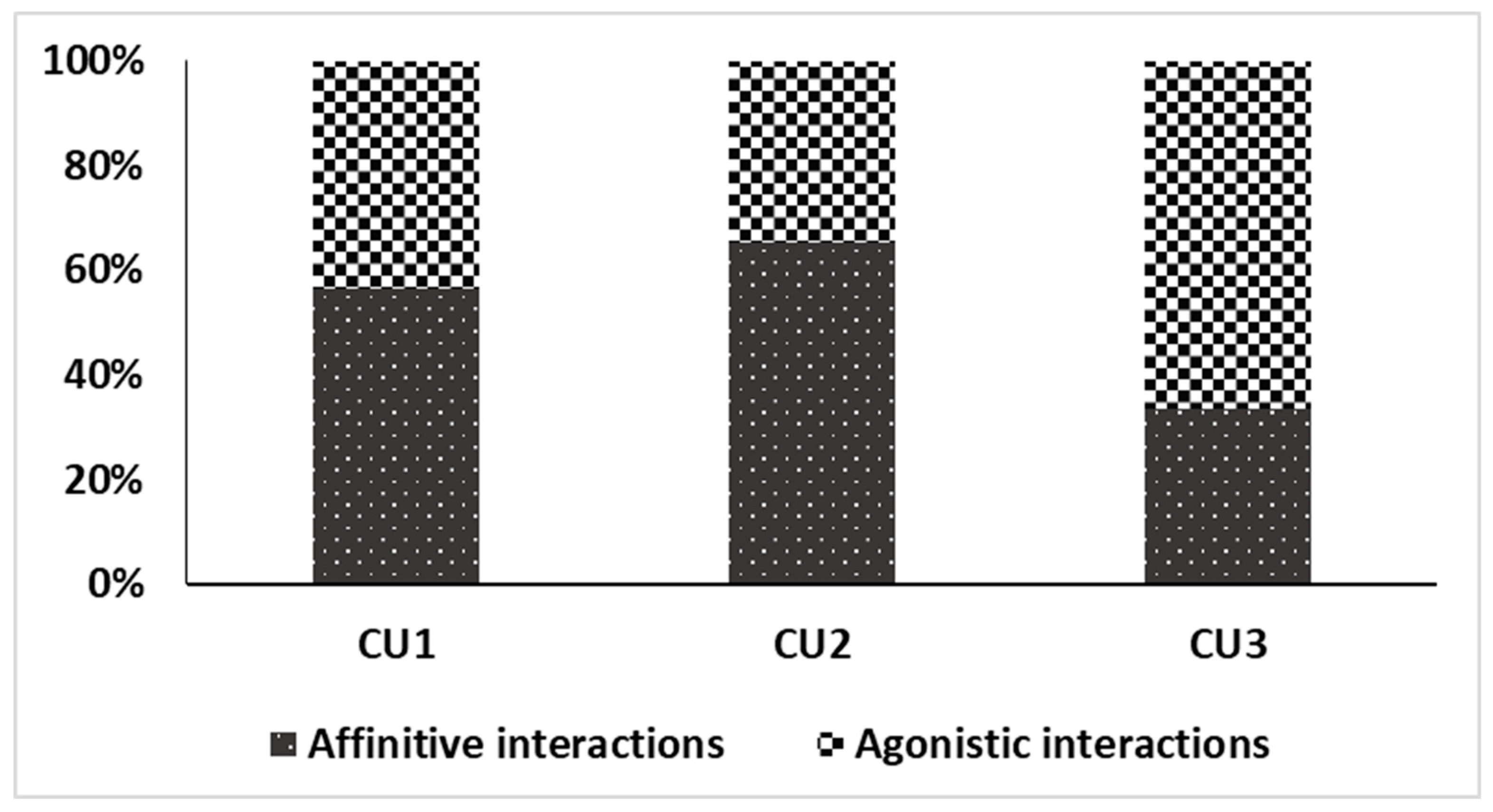
| Appropriate behavior | 9. Displaying social behavior | 9.1 Affinitive interactions * |
| 9.2 Agonistic interactions * | ||
| 10. Group dynamics | 10.1 Herd size | |
| 10.2 Herd composition | ||
| 10.3 Number of animals (other species) | ||
| 11. Display of other behavior | 11.1 Stereotypic behavior * | |
| 11.2 Environmental enrichment programs | ||
| 12.Good human–animal affiliations | 12.1 Medical training program | |
| 12.2 Capturing, handling, immobilization, and translocation |
| Behavior Pattern | Description of Behavior |
|---|---|
| Mutual grooming | When an animal brushes another animal with its muzzle on any part of the body with exception to the anal region. If the actor animal stops brushing for 10 s and starts again, it is to be counted as a new bout, regardless of whether the actor brushes the same receiver or another. If the actor receives reversal brushing from the receiver, it should also be counted as a new bout (AFI). |
| Licking | One animal licks any part of another animal with the tongue with the exception of anal region or urine. If the actor animal stops licking for 10 s and starts again, it is to be counted as a new bout, regardless of whether the actor licks the same receiver or another. If the actor receives reversal brushing from the receiver, it should also be counted as a new bout (AFI). |
| Smelling | One animal smells any part of another animal with the exception of the anal region or urine (Flehmen response). If the actor animal stops smelling for 10 s and starts again, it is to be counted as a new bout, regardless of whether the actor smells the same receiver or another. If an actor receives reversal smelling from the receiver, it should also be counted as a new bout (AFI). |
| Play | Physical contact of two animals by rubbing bodies, horning, head to head play, or rubbing horns against the neck or other parts of the body, with no signs of aggression or taking advantage. If the actor stops for more than 10 s and then resumes with the same receiver or another, it should be counted as a new bout (AFI). |
| Chase | One animal running behind another animal (receiver), causing the receiver to flee from its previous position. The animal also shows strong aggression toward the receiver (AGI). |
| Block | One animal (actor) runs after another (receiver), stands broadly in front of the receiver, and prevents approaching the opposite sex (AGI). |
| Parallel walk | Two animals at the same time walking parallel with heads bent down and maintaining a distance of 10–20 m. If the animals scratch the ground with their feet, stop for 10 s or more, and resume, it is to be counted as a new bout (AGI). |
| Fighting and thrusting | Two animals raise their front legs and strike their heads and horns, or push one another back (head-to-head or horns’ base) planting legs on the ground with great force. One animal hits others with a kick or strong butting, or if any animal thrusts vegetation or another object with signs of aggression. If any animal displays such behavior and stops for 10 s or more and then resumes, it should be counted as a new bout (AGI). |
| Facility | Number of Animals | Total Area of Enclosure (Area) (m2) | Space Per Animal (m2/Animal) |
|---|---|---|---|
| CU1 (Cherat Wildlife Park) | 23 | 14,299 | 621 |
| CU2 (Manglot Wildlife Park) | 6 | 3530 | 588 |
| CU3 (Manglot Wildlife Park) | 22 a | 15,793 | 717 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khattak, R.H.; Liu, Z.; Teng, L. Development and Implementation of Baseline Welfare Assessment Protocol for Captive Breeding of Wild Ungulate—Punjab Urial (Ovis vignei punjabiensis, Lydekker 1913). Animals 2019, 9, 1102. https://doi.org/10.3390/ani9121102
Khattak RH, Liu Z, Teng L. Development and Implementation of Baseline Welfare Assessment Protocol for Captive Breeding of Wild Ungulate—Punjab Urial (Ovis vignei punjabiensis, Lydekker 1913). Animals. 2019; 9(12):1102. https://doi.org/10.3390/ani9121102
Chicago/Turabian StyleKhattak, Romaan Hayat, Zhensheng Liu, and Liwei Teng. 2019. "Development and Implementation of Baseline Welfare Assessment Protocol for Captive Breeding of Wild Ungulate—Punjab Urial (Ovis vignei punjabiensis, Lydekker 1913)" Animals 9, no. 12: 1102. https://doi.org/10.3390/ani9121102
APA StyleKhattak, R. H., Liu, Z., & Teng, L. (2019). Development and Implementation of Baseline Welfare Assessment Protocol for Captive Breeding of Wild Ungulate—Punjab Urial (Ovis vignei punjabiensis, Lydekker 1913). Animals, 9(12), 1102. https://doi.org/10.3390/ani9121102





