Measuring Faecal Glucocorticoid Metabolites to Assess Adrenocortical Activity in Reindeer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Reindeer (Both Regions) and ACTH Challenge Test (Only Tverrvatnet)
2.2. Study Area and Field Conditions
2.3. Faecal Pellet Collection
2.3.1. Tverrvatnet
2.3.2. Stjernevatn
2.4. Analysis of FCMs
2.5. DNA Analysis
2.5.1. DNA Extraction
2.5.2. PCR Amplification
2.6. Animal Welfare
2.7. Statistical Analysis
2.7.1. Samples from Tverrvatnet
2.7.2. Samples from Stjernevatn
3. Results
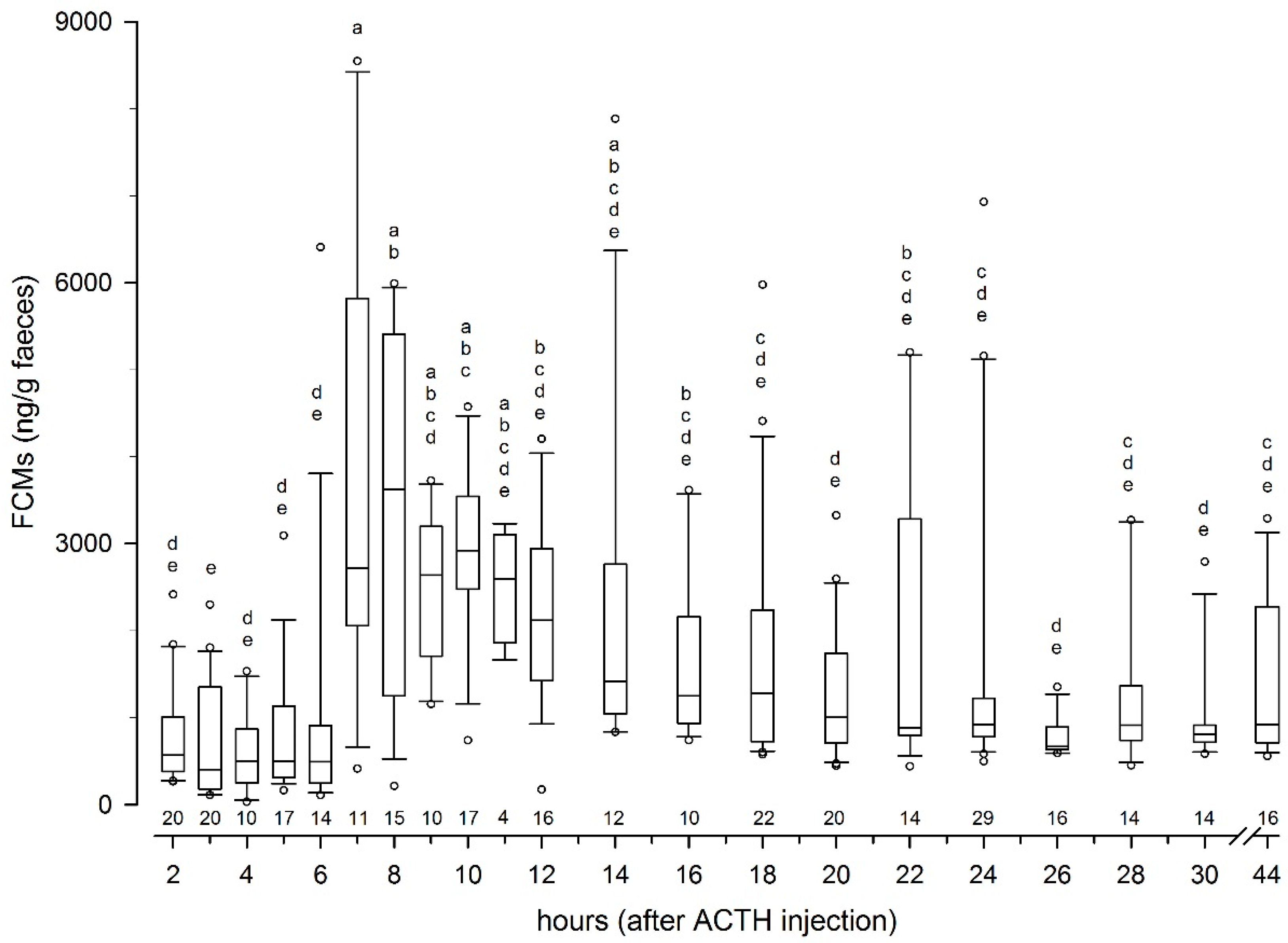
3.1. Controlled Experiment with ACTH Challenge on Eight Reindeer (Tverrvatnet)
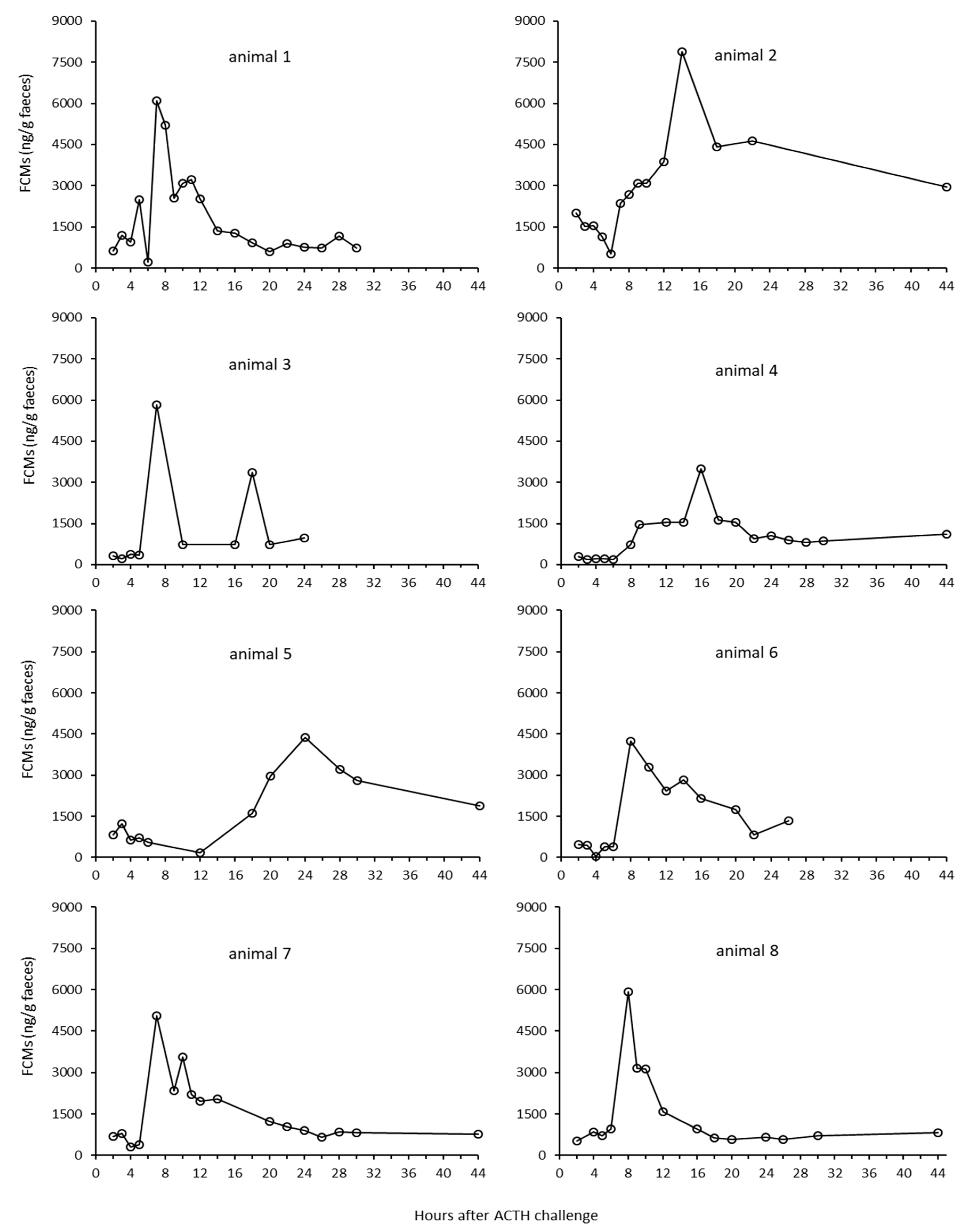
3.1.1. Identification of Individual Animals
3.1.2. Results from the ACTH Challenge Test
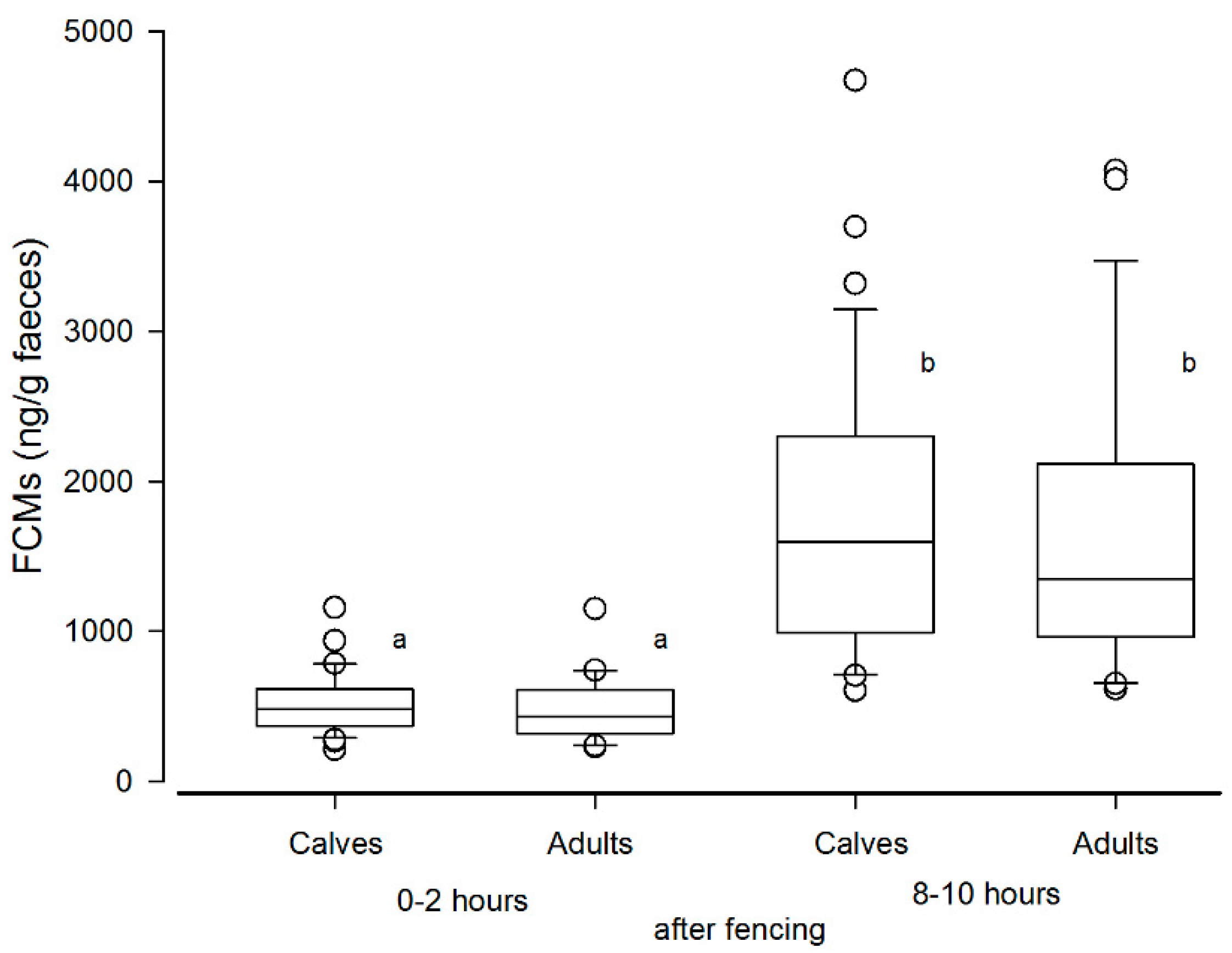
3.2. Results from the Biological Validation (Normal Handling and Calf Marking at Stjernevatn)
4. Discussion
4.1. Non-Invasive Versus Non-Disturbing
4.2. Peak FCM Levels
4.3. Effects of Sex and Individual
4.4. Identification of Unknown Samples
4.5. Ways Forward
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Axelrod, J.; Reisine, T.D. Stress hormones: Their interaction and regulation. Science 1984, 224, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Sheriff, M.J.; Dantzer, B.; Delehanty, B.; Palme, R.; Boonstra, R. Measuring stress in wildlife: Techniques for quantifying glucocorticoids. Oecologia 2011, 166, 869–887. [Google Scholar] [CrossRef]
- Palme, R. Monitoring stress hormone metabolites as a useful, non-invasive tool for welfare assessment in farm animals. Anim. Welf. 2012, 21, 331–337. [Google Scholar] [CrossRef]
- Möstl, E.; Palme, R. Hormones as indicators of stress. Domest. Anim. Endocrinol. 2002, 23, 67–74. [Google Scholar] [CrossRef]
- Palme, R. Non-invasive measurement of glucocorticoids: Advances and problems. Physiol. Behav. 2019, 199, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Palme, R.; Robia, C.; Messmann, S.; Hofer, J.; Möstl, E. Measurement of faecal cortisol metabolites in ruminants: A non-invasive parameter of adrenocortical function. Wien. Tierarztl. Monatsschr. 1999, 86, 237–241. [Google Scholar]
- Reimers, E.; Miller, F.L.; Eftestøl, S.; Colman, J.E.; Dahle, B. Flight by feral reindeer Rangifer tarandus tarandus in response to a directly approaching human on foot or on skis. Wildl. Biol. 2006, 12, 403–414. [Google Scholar] [CrossRef]
- Rehbinder, C. Management stress in reindeer. Rangifer 1990, 10, 267–288. [Google Scholar] [CrossRef]
- Wiklund, E. Pre-Slaughter Handling of Reindeer (Rangifer tarandus tarandus L.)-Effects on Meat Quality. Ph.D. Thesis, Swedish University of Agricultural Sciences (SLU), Uppsala, Sweden, 20 December 1996. [Google Scholar]
- Gregory, N.G.; Grandin, T. Animal Welfare and Meat Science; CABI Pub.: Wallingford, UK, 1998; p. 298. [Google Scholar]
- Jørgensen, G.H.M.; Mejdell, C.M.; Stubsjøen, S.M.; Özkan Gülzari, Ş.; Rødbotten, R.; Bårdsen, B.-J.; Rødven, R. Velferdskriterier i reindriften: En studie av velfersindikatorer og mulige kvalitetskriterier for rein og reinkjøtt. In Welfare Criteria in Reindeer Management: A Study of Welfare Indicators and Possible Quality Criteria for Reindeer and Reindeer Meat; Norwegian Institute of Bioeconomy Research (NIBIO): Akershus, Norway, 2017; Volume 3, p. 48. (In Norwegian) [Google Scholar]
- Touma, C.; Palme, R. Measuring fecal glucocorticoid metabolites in mammals and birds: The importance of validation. Ann. N. Y. Acad. Sci. 2005, 1046, 54–74. [Google Scholar] [CrossRef]
- Palme, R.; Rettenbacher, S.; Touma, C.; El-Bahr, S.; Möstl, E. Stress hormones in mammals and birds: Comparative aspects regarding metabolism, excretion, and noninvasive measurement in fecal samples. Ann. N. Y. Acad. Sci. 2005, 1040, 162–171. [Google Scholar] [CrossRef]
- Ashley, N.; Barboza, P.; Macbeth, B.; Janz, D.; Cattet, M.; Booth, R.; Wasser, S. Glucocorticosteroid concentrations in feces and hair of captive caribou and reindeer following adrenocorticotropic hormone challenge. Gen. Comp. Endocrinol. 2011, 172, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Flasko, A.; Manseau, M.; Mastromonaco, G.; Bradley, M.; Neufeld, L.; Wilson, P. Fecal DNA, hormones, and pellet morphometrics as a noninvasive method to estimate age class: An application to wild populations of Central Mountain and Boreal woodland caribou (Rangifer tarandus caribou). Canad. J. Zool. 2017, 95, 311–321. [Google Scholar] [CrossRef]
- Palme, R.; Touma, C.; Arias, N.; Dominchin, M.F.; Lepschy, M. Steroid extraction: Get the best out of faecal samples. Wien. Tierarztl. Mon. 2013, 100, 238–246. [Google Scholar]
- Palme, R. Measuring fecal steroids: Guidelines for practical application. Ann. N. Y. Acad. Sci. 2005, 1046, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Möstl, E.; Maggs, J.; Schrötter, G.; Besenfelder, U.; Palme, R. Measurement of cortisol metabolites in faeces of ruminants. Vet. Res. Comm. 2002, 26, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.; Strobeck, C.; Wu, L.; Coffin, J. Characterization of microsatellite loci in caribou Rangifer tarandus, and their use in other artiodactyls. Mol. Ecol. 1997, 6, 697–699. [Google Scholar] [CrossRef]
- Røed, K.; Midthjell, L. Microsatellites in reindeer, Rangifer tarandus, and their use in other cervids. Mol. Ecol. 1998, 7, 1773–1776. [Google Scholar] [CrossRef]
- Brownstein, M.J.; Carpten, J.D.; Smith, J.R. Modulation of non-templated nucleotide addition by Taq DNA polymerase: Primer modifications that facilitate genotyping. Biotechniques 1996, 20, 1004–1010. [Google Scholar] [CrossRef]
- Statistical Analysis System Institute Inc. SAS/STAT 9.3 for User’s Guide; SAS Institute: Cary, NC, USA, 2011. [Google Scholar]
- Moberg, G.P.; Mench, J.A. The Biology of Animal Stress: Basic Principles and Implications for Animal Welfare; CABI Pub.: Wallingford, UK, 2000; p. 377. [Google Scholar]
- Huber, S.; Palme, R.; Zenker, W.; Möstl, E. Non-invasive monitoring of the adrenocortical response in red deer. J. Wild. Manag. 2003, 67, 258–266. [Google Scholar] [CrossRef]
- Kleinsasser, C.; Graml, C.; Klobetz-Rassam, E.; Barth, K.; Waiblinger, S.; Palme, R. Physiological validation of a non-invasive method for measuring adrenocortical activity in goats. Wien. Tierärztl. Mon. 2010, 97, 259–262. [Google Scholar]
- Carlsson, A.; Mastromonaco, G.; Vandervalk, E.; Kutz, S. Parasites, stress and reindeer: Infection with abomasal nematodes is not associated with elevated glucocorticoid levels in hair or faeces. Conserv. Physiol. 2016, 4, cow058. [Google Scholar] [CrossRef] [PubMed]
- Palme, R.; Fischer, P.; Schildorfer, H.; Ismail, M.N. Excretion of infused 14C-steroid hormones via faeces and urine in domestic livestock. Anim. Reprod. Sci. 1996, 43, 43–63. [Google Scholar] [CrossRef]
- Ball, M.C.; Finnegan, L.; Manseau, M.; Wilson, P. Integrating multiple analytical approaches to spatially delineate and characterize genetic population structure: An application to boreal caribou (Rangifer tarandus caribou) in central Canada. Conserv. Genet. 2010, 11, 2131–2143. [Google Scholar] [CrossRef]
- Klütsch, C.F.; Manseau, M.; Trim, V.; Polfus, J.; Wilson, P.J. The eastern migratory caribou: The role of genetic introgression in ecotype evolution. Royal. Soc. Open Sci. 2016, 3, 150469. [Google Scholar] [CrossRef]
- Jenkins, D.A.; Yannic, G.; Schaefer, J.A.; Conolly, J.; Lecomte, N. Population structure of caribou in an ice-bound archipelago. Divers. Distrib. 2018, 24, 1092–1108. [Google Scholar] [CrossRef]
- Coppes, J.; Kämmerle, J.L.; Willert, M.; Kohnen, A.; Palme, R.; Braunisch, V. The importance of individual heterogeneity for interpreting faecal glucocorticoid metabolite levels in wildlife studies. J. Appl. Ecol. 2018, 55, 2043–2054. [Google Scholar] [CrossRef]
- Rehnus, M.; Palme, R. How genetic data improve the interpretation of results of faecal glucocorticoid metabolite measurements in a free-living population. PLoS ONE 2017, 12, e0183718. [Google Scholar] [CrossRef]
- Taberlet, P.; Luikart, G. Non-invasive genetic sampling and individual identification. Biol. J. Linn. Soc. 1999, 68, 41–55. [Google Scholar] [CrossRef]
- Fernando, P.; Vidya, T.; Rajapakse, C.; Dangolla, A.; Melnick, D. Reliable noninvasive genotyping: Fantasy or reality? J. Hered. 2003, 94, 115–123. [Google Scholar] [CrossRef]
- Ball, M.C.; Pither, R.; Manseau, M.; Clark, J.; Petersen, S.D.; Kingston, S.; Morrill, N.; Wilson, P. Characterization of target nuclear DNA from faeces reduces technical issues associated with the assumptions of low-quality and quantity template. Conserv. Genet. 2007, 8, 577–586. [Google Scholar] [CrossRef]
| Multiplex Assays | Microsatellite Marker | Primer Sequences (5′–3′) | Repeat Motif | PCR Concentration and Fluorescent Dye | Source | GeneBank Accession No. |
|---|---|---|---|---|---|---|
| 1 | NVHRT22 | F: GTATTCTTGCCAGGAAAAACC | CA | 0.1 µM, NED | Røed and Midthjell [20] | AF068208 |
| R: GTTGCTTCAGTGCTCTCAGAT | ||||||
| RT1 | F: TGCCTTCTTTCATCCAACAA | GT | 0.2 µM, FAM | Wilson et al. [19] | U90737 | |
| R: CATCTTCCCATCCTCTTTAC | ||||||
| RT20 | F: GCAGAAGAGTGAGTGTGAGT | GT | 0.2 µM, NED | Wilson et al. [19] | U90744 | |
| R: GTTTCTTGTTGTATTTTGGACCTTT | ||||||
| RT13 | F: GCCCAGTGTTAGGAAAGAAG | GT | 0.2 µM, FAM | Wilson et al. [19] | U90743 | |
| R: CATCCCAGAACAGGAGTGAG | ||||||
| 2 | NVHRT46 | F: CCGACTGAAGTGACCAAG | CA | 0.2 µM, FAM | Røed and Midthjell [20] | AF068213 |
| R: TGTTGAGAGGATTGATAAG | ||||||
| RT6 | F: TTCCTCTTACTCATTCTTGG | GT | 0.4 µM, NED | Wilson et al. [19] | U90739 | |
| R: CGGATTTTGAGACTGTTAC | ||||||
| RT30 | F: CACTTGGCTTTTGGACTTA | GT | 0.3 µM, NED | Wilson et al. [19] | U90749 | |
| R: CTGGTGTATGTATGCACACT | ||||||
| RT27 | F: CCAAAGACCCAACAGATG | GT | 0.2 µM, PET | Wilson et al. [19] | U90748 | |
| R: TTGTAACACAGCAAAAGCATT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Özkan Gülzari, Ş.; Jørgensen, G.H.M.; Eilertsen, S.M.; Hansen, I.; Hagen, S.B.; Fløystad, I.; Palme, R. Measuring Faecal Glucocorticoid Metabolites to Assess Adrenocortical Activity in Reindeer. Animals 2019, 9, 987. https://doi.org/10.3390/ani9110987
Özkan Gülzari Ş, Jørgensen GHM, Eilertsen SM, Hansen I, Hagen SB, Fløystad I, Palme R. Measuring Faecal Glucocorticoid Metabolites to Assess Adrenocortical Activity in Reindeer. Animals. 2019; 9(11):987. https://doi.org/10.3390/ani9110987
Chicago/Turabian StyleÖzkan Gülzari, Şeyda, Grete Helen Meisfjord Jørgensen, Svein Morten Eilertsen, Inger Hansen, Snorre Bekkevold Hagen, Ida Fløystad, and Rupert Palme. 2019. "Measuring Faecal Glucocorticoid Metabolites to Assess Adrenocortical Activity in Reindeer" Animals 9, no. 11: 987. https://doi.org/10.3390/ani9110987
APA StyleÖzkan Gülzari, Ş., Jørgensen, G. H. M., Eilertsen, S. M., Hansen, I., Hagen, S. B., Fløystad, I., & Palme, R. (2019). Measuring Faecal Glucocorticoid Metabolites to Assess Adrenocortical Activity in Reindeer. Animals, 9(11), 987. https://doi.org/10.3390/ani9110987





