Thymoquinone-PLGA-PVA Nanoparticles Ameliorate Bleomycin-Induced Pulmonary Fibrosis in Rats via Regulation of Inflammatory Cytokines and iNOS Signaling
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Drugs and Chemicals
2.2. TQ-PLGA-PVA Nano-Emulsion Designing
2.3. Animals
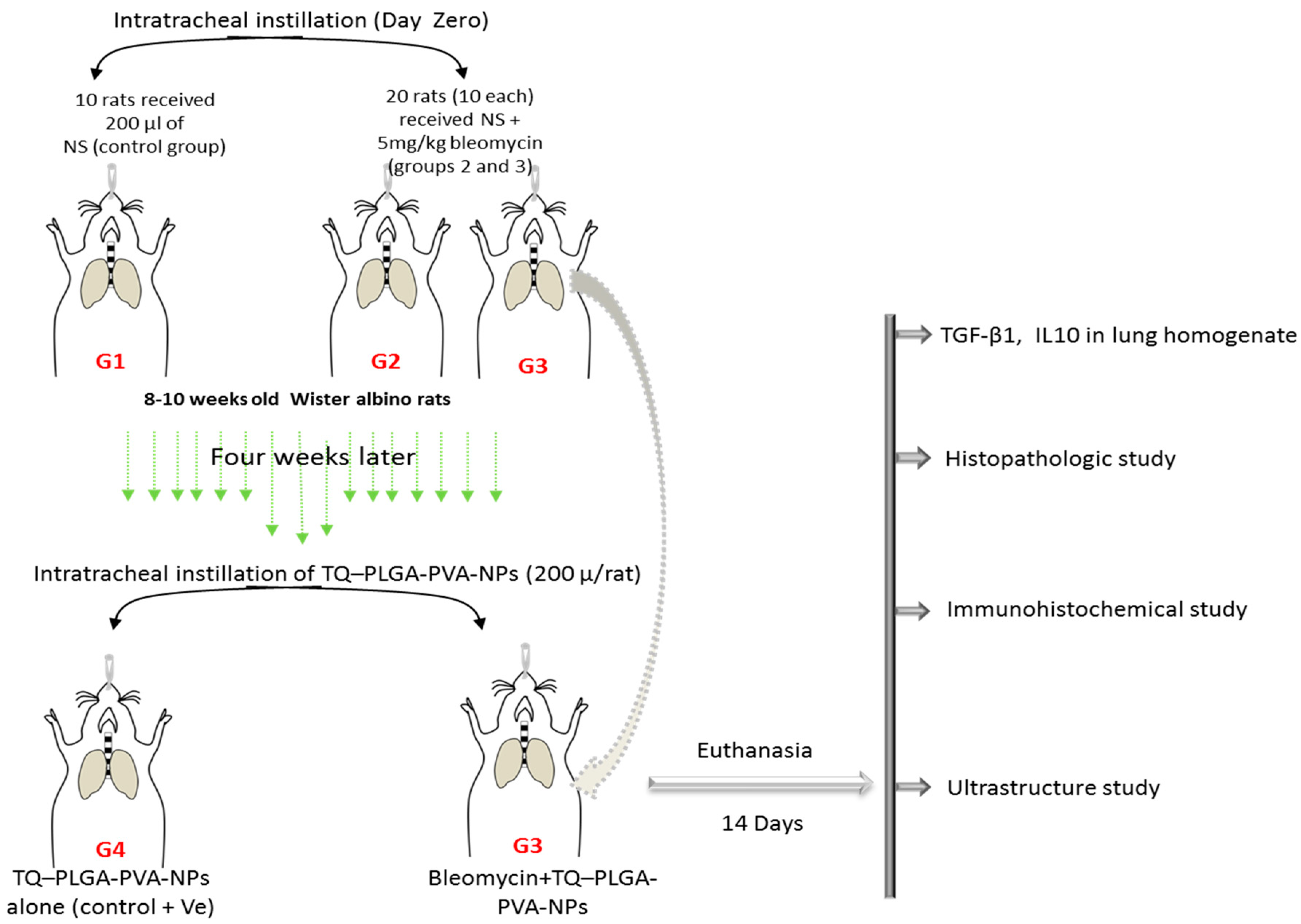
2.4. Experimental Design
2.5. Induction of Lung Fibrosis
2.6. Assessment of Levels of TGF-β1 and IL 10
2.7. Histopathologic Assessment
2.8. Immunohistochemical and Histo-Morphometric Assessment
2.9. Ultrastructure Assessment
2.10. Statistical Analysis
3. Results
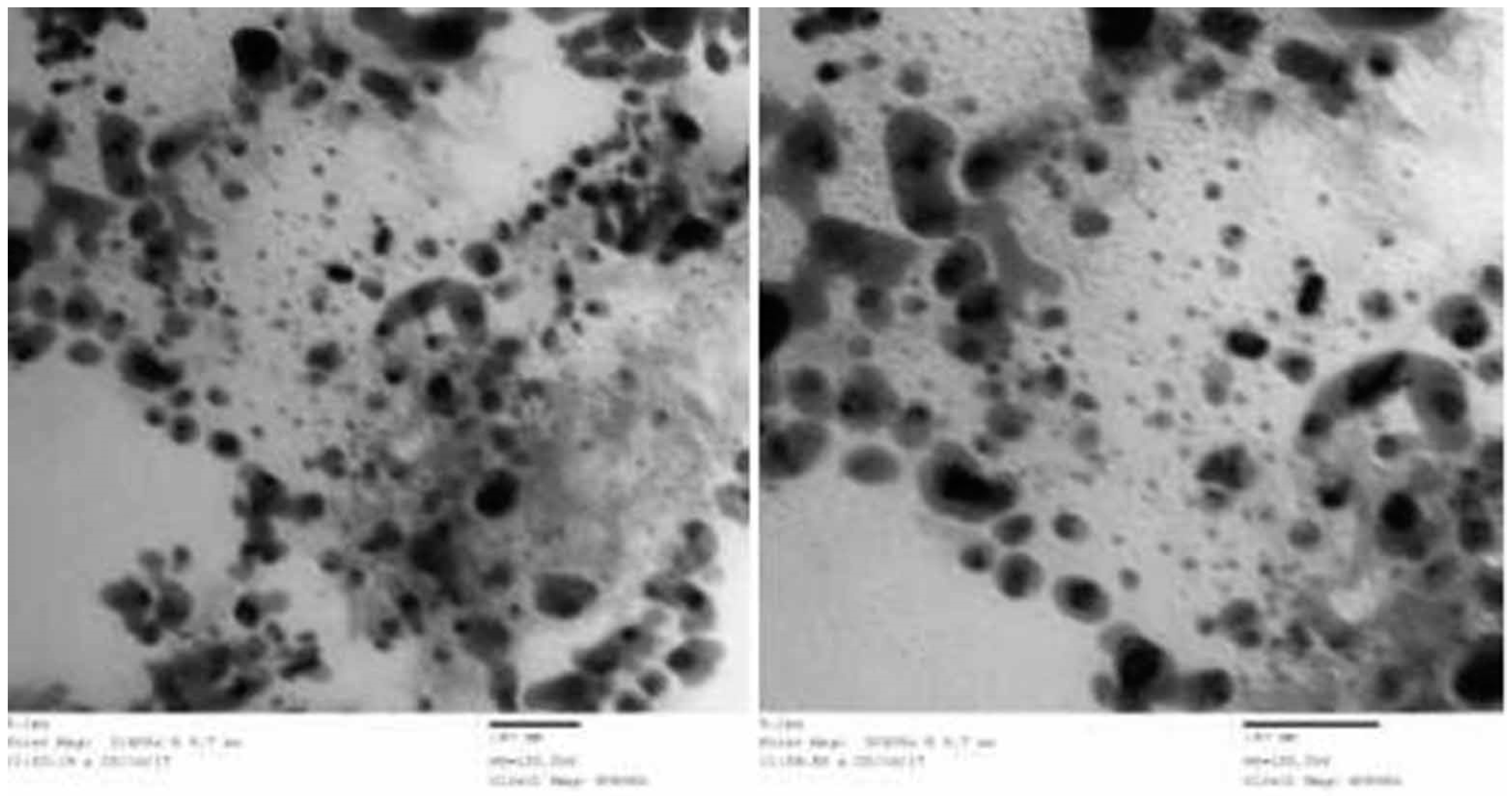
3.1. Characterization of Designed Nanoparticle
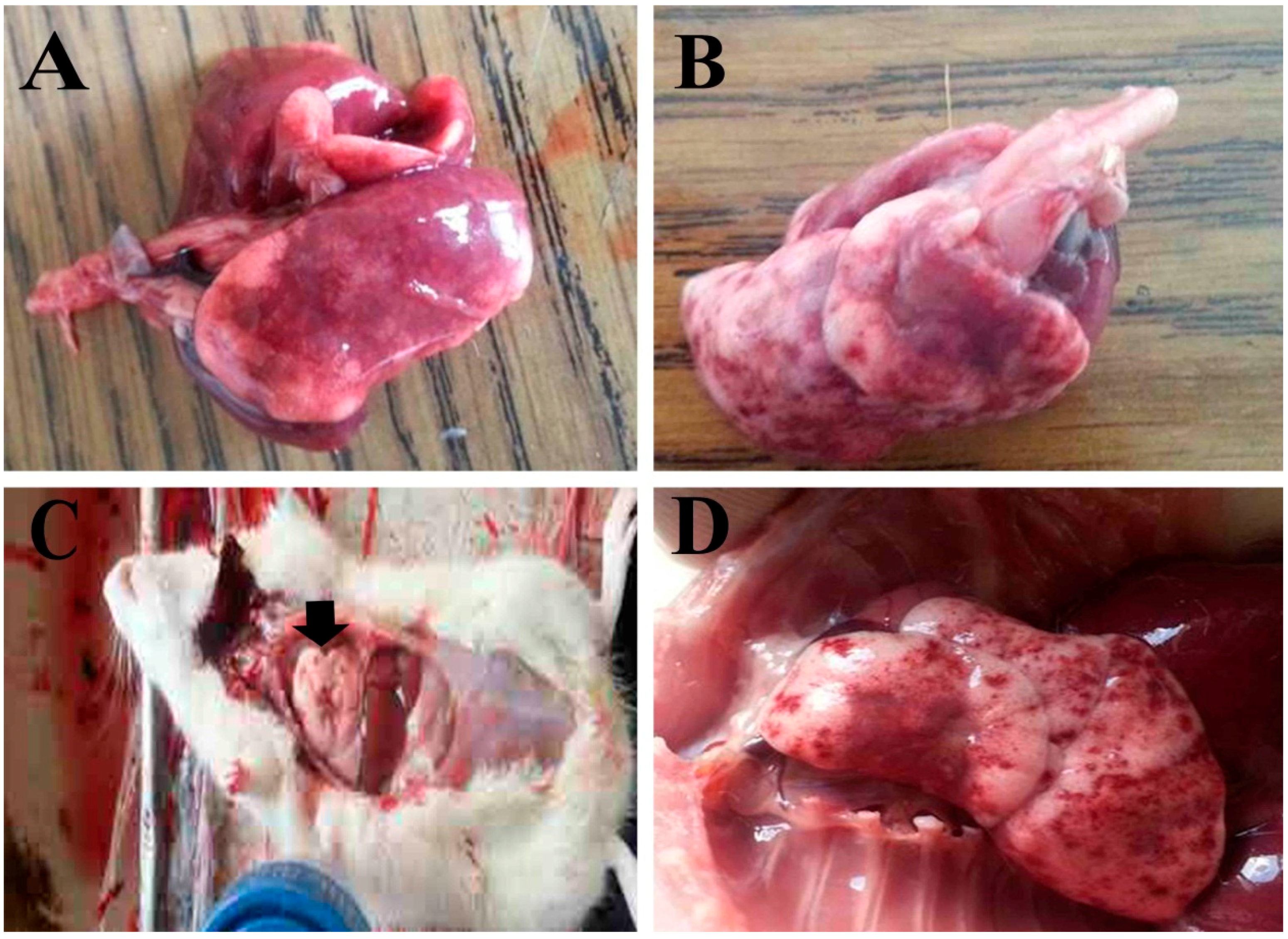
3.2. Gross Examination of Lung Tissues
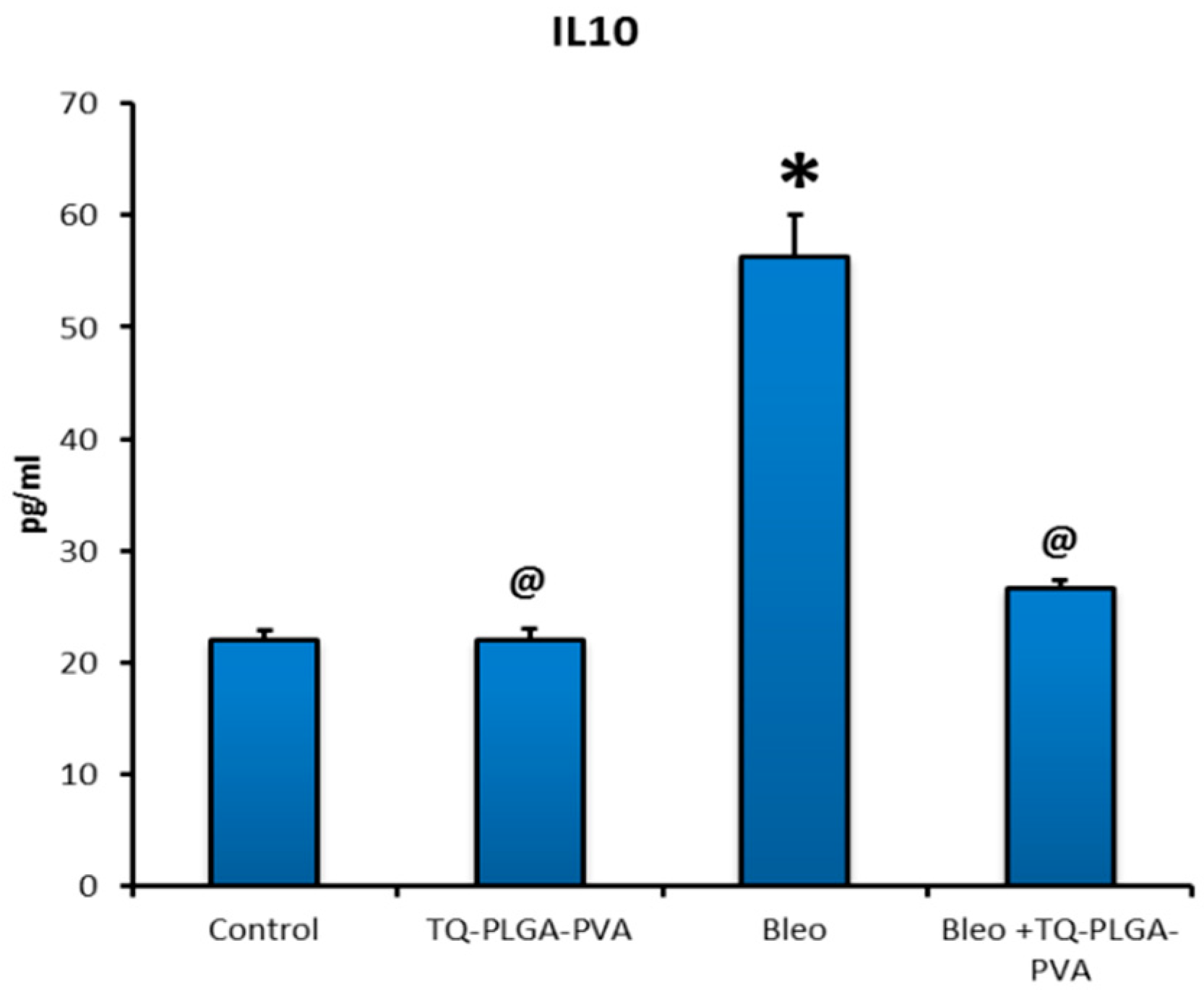
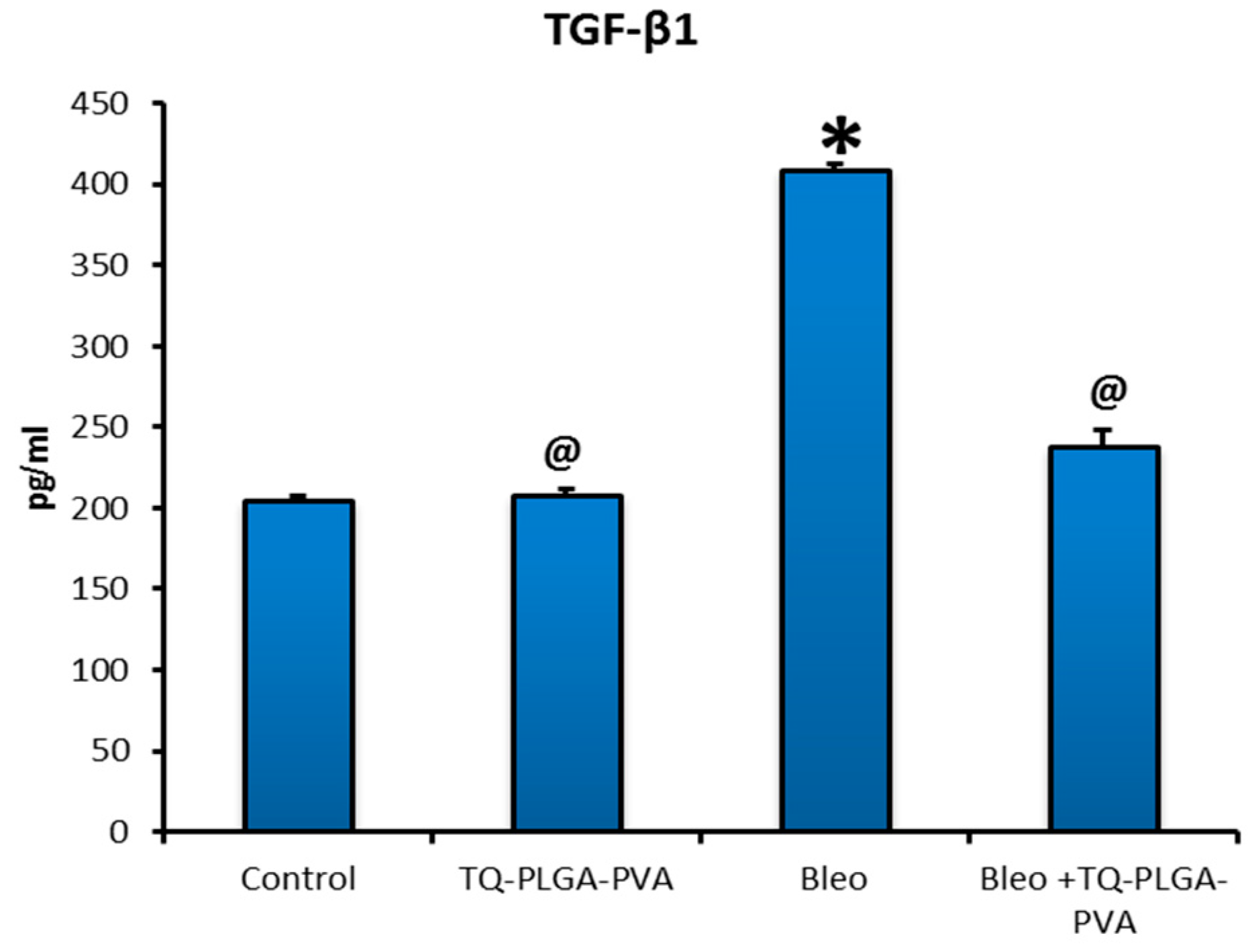
3.3. Levels of the Pulmonary TGF-β1 and IL 10
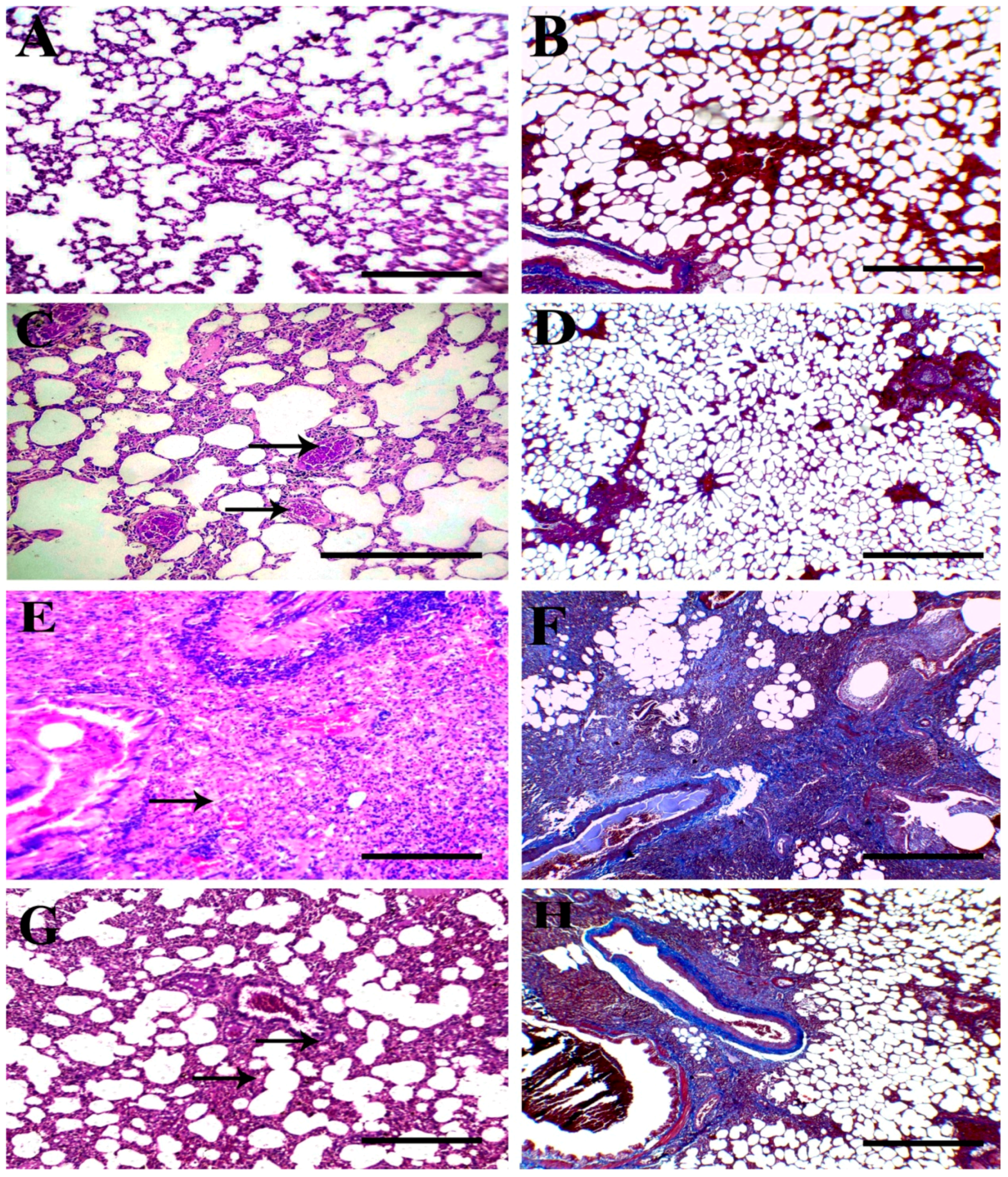
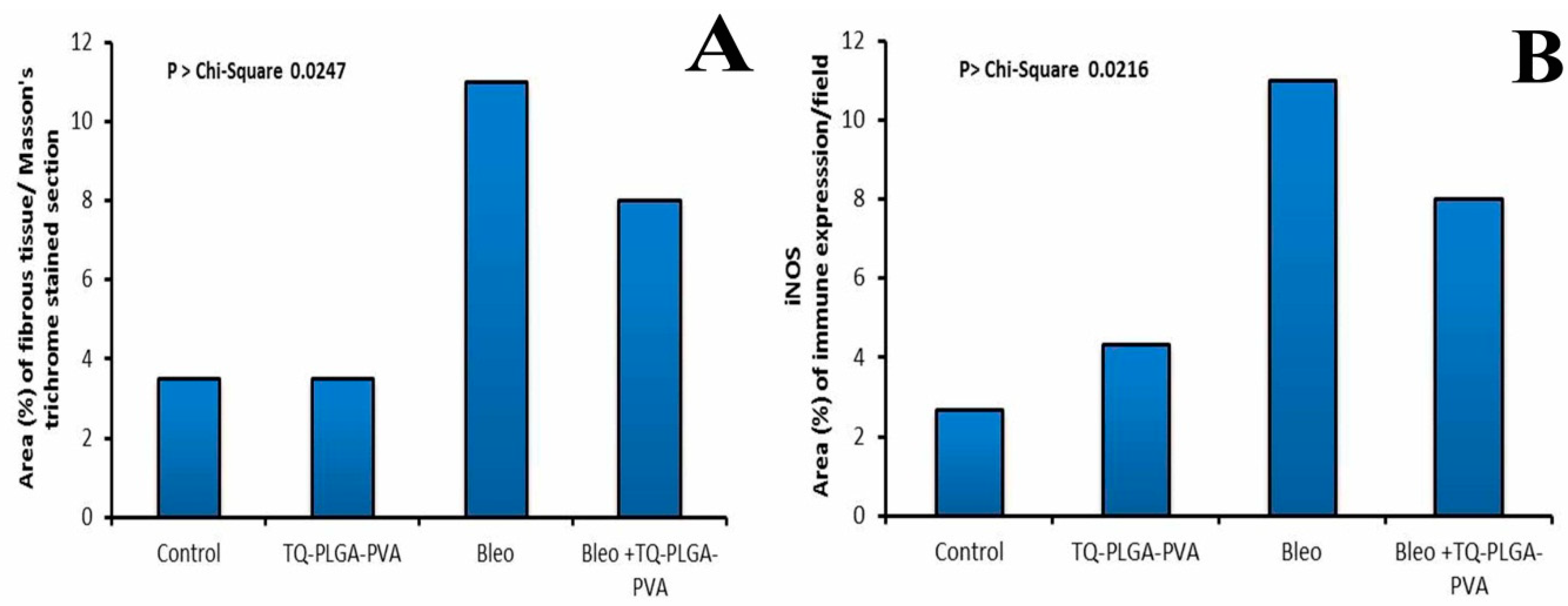
3.4. Histopathological Findings
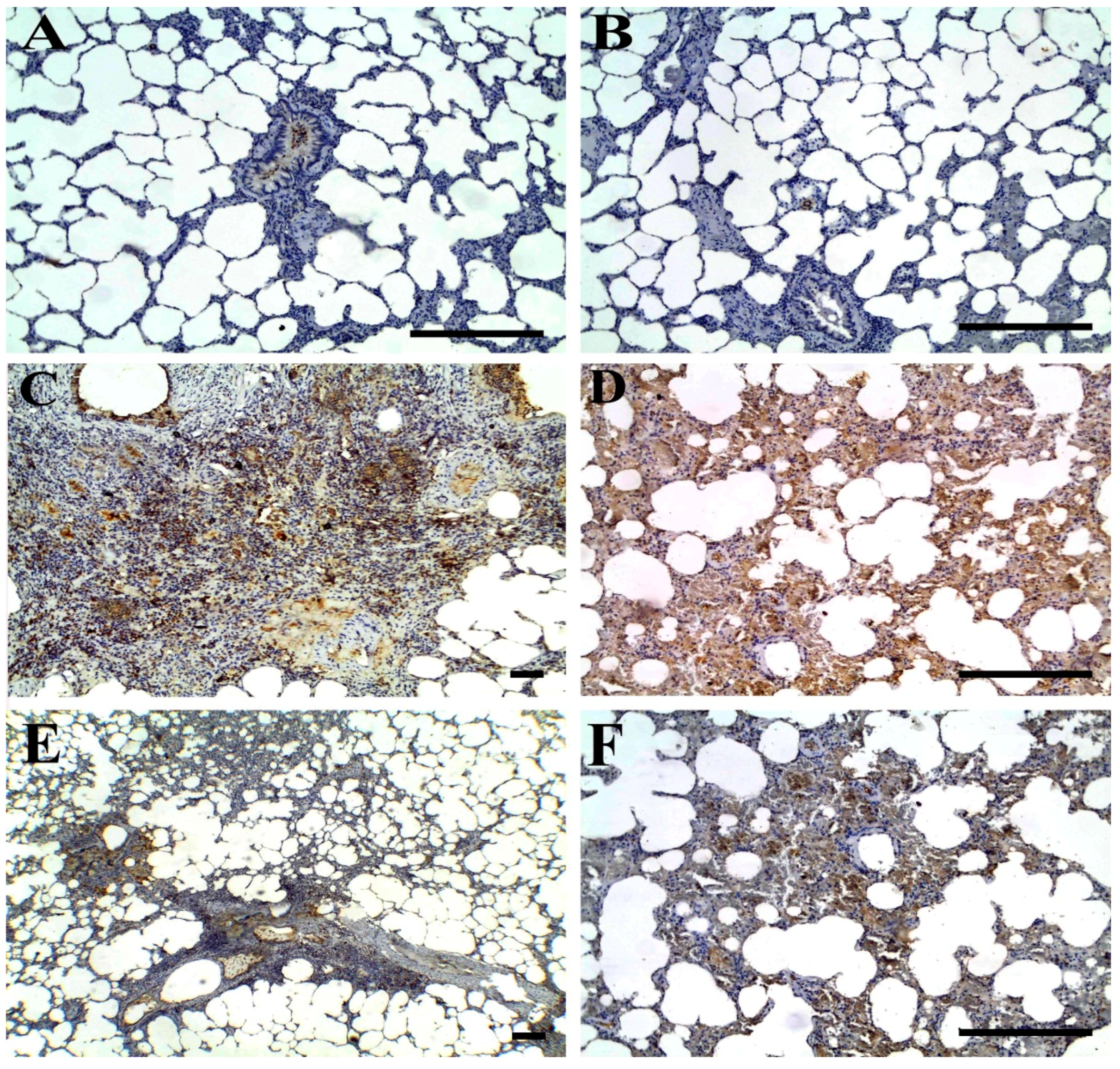
3.5. Immunohistochemical Findings
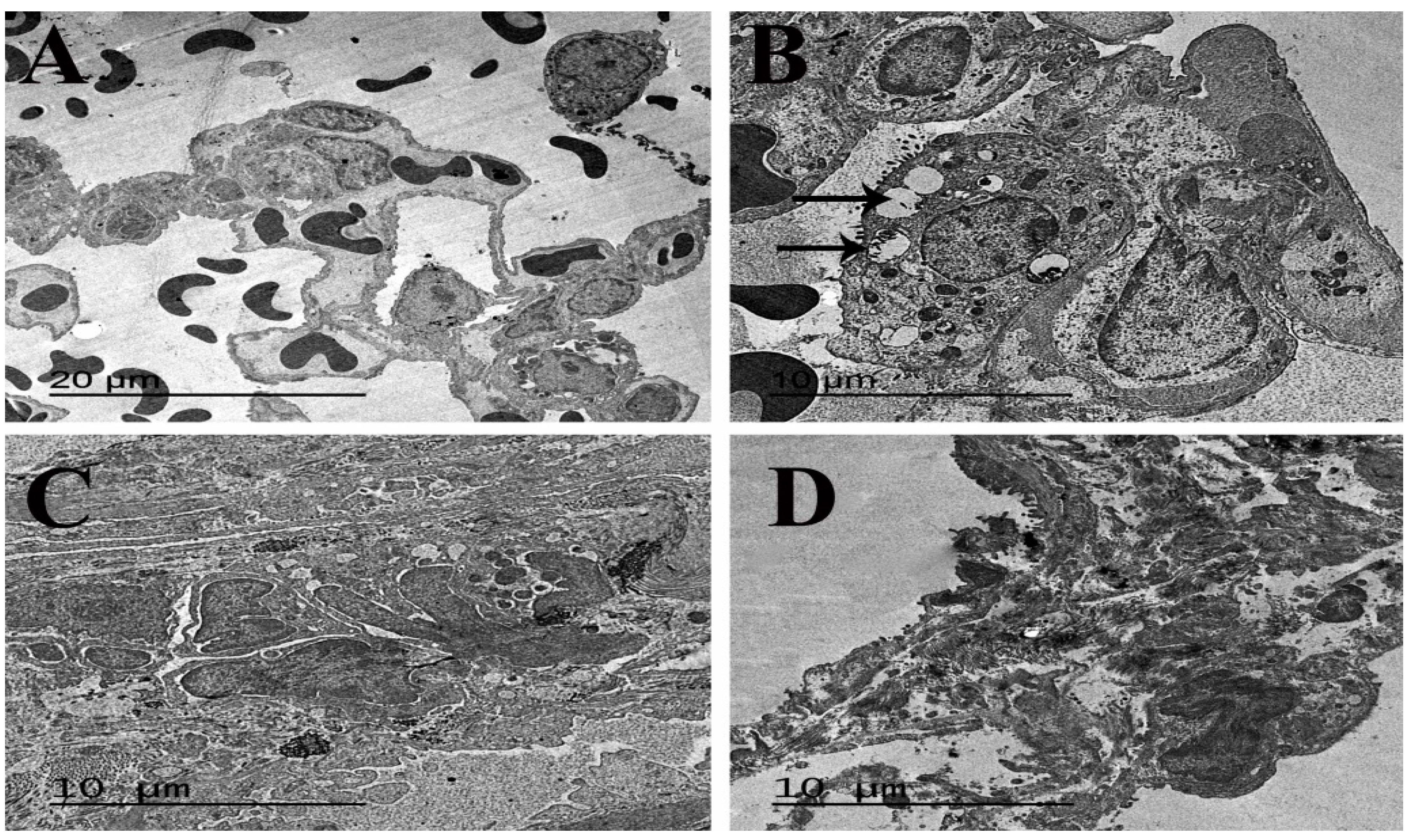
3.6. Ultrastructure Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ozyigit, L.P.; Aktas, E.C.; Senbas, Z.A.; Ozturk, A.B.; Ozturk, E.; Ergonul, M.O.; Deniz, G. The role of atopy in the pathogenesis of bleomycin pulmonary toxicity. Resp. Med. 2019, 155, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Sleijfer, S. Bleomycin-induced pneumonitis. Chest 2001, 120, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Kabel, A.M.; Estfanous, R.S.; Alrobaian, M.M. Targeting oxidative stress, proinflammatory cytokines, apoptosis and toll like receptor 4 by empagliflozin to ameliorate bleomycin-induced lung fibrosis. Resp. Physiol. Neurobiol. 2019, 7, 103316. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; De Los Santos, F.G.; Phan, S.H. The bleomycin model of pulmonary fibrosis. In Fibrosis; Springer Humana Press: New York, NY, USA, 2017; pp. 27–42. [Google Scholar]
- Zhou, X.; Zhao, Y.; He, C.C.; Li, J. Preventive effects of Citrus reticulata essential oil on bleomycin-induced pulmonary fibrosis in rats and the mechanism. Zhong Xi Yi Jie He Xue Bao = J. Chin. Integ. Med. 2012, 10, 200–209. [Google Scholar] [CrossRef]
- Nuovo, G.J.; Hagood, J.S.; Magro, C.M.; Chin, N.; Kapil, R.; Davis, L.; Folcik, V.A. The distribution of immunomodulatory cells in the lungs of patients with idiopathic pulmonary fibrosis. Modern Pathol. 2012, 25, 416. [Google Scholar] [CrossRef]
- Todd, N.W.; Luzina, I.G.; Atamas, S.P. Molecular and cellular mechanisms of pulmonary fibrosis. Fibrogen. Tissue Rep. 2012, 5, 11. [Google Scholar] [CrossRef]
- Tashiro, M.; Izumikawa, K.; Yoshioka, D.; Nakamura, S.; Kurihara, S.; Sakamoto, N.; Mukae, H. Lung fibrosis 10 years after cessation of bleomycin therapy. Tohoku J. Exp. Med. 2008, 216, 77–80. [Google Scholar] [CrossRef]
- Hobbs, A.J.; Higgs, A.; Moncada, S. Inhibition of nitric oxide synthase as a potential therapeutic target. Annu. Rev. Pharm. Toxicol. 1999, 39, 191–220. [Google Scholar] [CrossRef]
- Paredi, P.; Kharitonov, S.A.; Barnes, P.J. Analysis of expired air for oxidation products. Am. J. Resp. Crit. Care Med. 2002, 166, S31–S37. [Google Scholar] [CrossRef]
- Moncada, S. Nitric oxide: Physiology, pathophysiology, and pharmacology. Pharm. Rev. 1991, 43, 109–142. [Google Scholar]
- Nathan, C. Nitric oxide as a secretory product of mammalian cells. FASEB J. 1992, 6, 3051–3064. [Google Scholar] [CrossRef] [PubMed]
- Paredi, P.; Kharitonov, S.; Loukides, S.; Pantelidis, P.; Du Bois, R.; Barnes, P.J. Exhaled nitric oxide is increased in active fibrosing alveolitis. Chest 1999, 115, 1352–1356. [Google Scholar] [CrossRef] [PubMed]
- Alving, K.; Weitzberg, E.; Lundberg, J. Increased amount of nitric oxide in exhaled air of asthmatics. Eur. Resp. J. 1993, 6, 1368–1370. [Google Scholar]
- Kharitonov, S.A.; Wells, A.U.; O’Connor, B.J.; Cole, P.J.; Hansell, D.M.; Logan-Sinclair, R.B.; Barnes, P.J. Elevated levels of exhaled nitric oxide in bronchiectasis. Am. J. Resp. Crit. Care Med. 1995, 151, 1889–1893. [Google Scholar] [CrossRef] [PubMed]
- Baeuerle, P.A.; Henkel, T. Function and activation of NF-kappaB in the immune system. Annu. Rev. Immunol. 1994, 12, 141–179. [Google Scholar] [CrossRef]
- Saito, F.; Tasaka, S.; Inoue, K.I.; Miyamoto, K.; Nakano, Y.; Ogawa, Y.; Takano, H. Role of interleukin-6 in bleomycin-induced lung inflammatory changes in mice. Am. J. Resp. Cell Mol. Biol. 2008, 38, 566–571. [Google Scholar] [CrossRef]
- Jia, L.; Sun, P.; Gao, H.; Shen, J.; Gao, Y.; Meng, C.; Zhang, G. Mangiferin attenuates bleomycin-induced pulmonary fibrosis in mice through inhibiting TLR 4/p65 and TGF-β1/Smad2/3 pathway. J. Pharm. Pharm. 2019, 71, 1017–1028. [Google Scholar] [CrossRef]
- Nakagome, K.; Dohi, M.; Okunishi, K.; Tanaka, R.; Miyazaki, J.-I.; Yamamoto, K. In vivo IL-10 gene delivery attenuates bleomycin induced pulmonary fibrosis by inhibiting the production and activation of TGF-β in the lung. Thorax 2006, 61, 886–894. [Google Scholar] [CrossRef]
- Piguet, P.F.; Collart, M.A.; Grau, G.E.; Kapanci, Y.; Vassalli, P. Tumor necrosis factor/cachectin plays a key role in bleomycin-induced pneumopathy and fibrosis. J. Exp. Med. 1989, 170, 655–663. [Google Scholar] [CrossRef]
- Taniguchi, H.; Ebina, M.; Kondoh, Y.; Ogura, T.; Azuma, A.; Suga, M.; Takeuchi, M. Pirfenidone in idiopathic pulmonary fibrosis. Eur. Resp. J. 2010, 35, 821–829. [Google Scholar] [CrossRef]
- Ren, Y.-X.; Zhou, R.; Tang, W. (5R)-5-hydroxytriptolide (LLDT-8) protects against bleomycin-induced lung fibrosis in mice. Acta Pharm. Sin. 2007, 28, 518. [Google Scholar] [CrossRef] [PubMed]
- Pao, H.-P.; Liao, W.-I.; Wu, S.-Y.; Hung, K.-Y.; Huang, K.-L.; Chu, S.-J. PG490-88, a derivative of triptolide, suppresses ischemia/reperfusion-induced lung damage by maintaining tight junction barriers and targeting multiple signaling pathways. Int. Immunopharmacol. 2019, 68, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Giri, S.; Biring, I.; Nguyen, T.; Wang, Q.; Hyde, D. Abrogation of bleomycin-induced lung fibrosis by nitric oxide synthase inhibitor, aminoguanidine in mice. Nitric Oxide. 2002, 7, 109–118. [Google Scholar] [CrossRef]
- Bai, T.; Lian, L.-H.; Wu, Y.-L.; Wan, Y.; Nan, J.-X. Thymoquinone attenuates liver fibrosis via PI3K and TLR4 signaling pathways in activated hepatic stellate cells. Int. Immunopharmacol. 2013, 15, 275–281. [Google Scholar] [CrossRef]
- Asgharzadeh, F.; Bargi, R.; Beheshti, F.; Hosseini, M.; Farzadnia, M.; Khazaei, M. Thymoquinone restores liver fibrosis and improves oxidative stress status in a lipopolysaccharide-induced inflammation model in rats. Avicenna J. Phytomed. 2017, 7, 502. [Google Scholar]
- Pourgholamhossein, F.; Sharififar, F.; Rasooli, R.; Pourgholi, L.; Nakhaeipour, F.; Samareh-Fekri, H.; Mandegary, A. Thymoquinone effectively alleviates lung fibrosis induced by paraquat herbicide through down-regulation of pro-fibrotic genes and inhibition of oxidative stress. Environ. Toxicol. Pharm. 2016, 45, 340–345. [Google Scholar] [CrossRef]
- Hamed, K.; Debonnett, L. Tobramycin inhalation powder for the treatment of pulmonary Pseudomonas aeruginosa infection in patients with cystic fibrosis: a review based on clinical evidence. Ther. Adv. Resp. Dis. 2017, 11, 193–209. [Google Scholar] [CrossRef]
- Al-Nemrawi, N.; Alshraiedeh, N.A.; Zayed, A.; Altaani, B. Low molecular weight chitosan-coated PLGA nanoparticles for pulmonary delivery of tobramycin for cystic fibrosis. Pharmaceuticals 2018, 11, 28. [Google Scholar] [CrossRef]
- Nallamuthu, I.; Parthasarathi, A.; Khanum, F. Thymoquinone-loaded PLGA nanoparticles: Antioxidant and anti-microbial properties. Int. Curr. Pharm. J. 2013, 2, 202–207. [Google Scholar] [CrossRef]
- Ganea, G.M.; Fakayode, S.O.; Losso, J.N.; Van Nostrum, C.F.; Sabliov, C.M.; Warner, I.M. Delivery of phytochemical thymoquinone using molecular micelle modified poly (D, L lactide-co-glycolide)(PLGA) nanoparticles. Nanotechnology 2010, 21, 285104. [Google Scholar] [CrossRef]
- Xie, X.; Tao, Q.; Zou, Y.; Zhang, F.; Guo, M.; Wang, Y.; Wang, H.; Zhou, Q.; Yu, S. PLGA Nanoparticles improve the oral bioavailability of curcumin in rats: Characterizations and Mechanisms. J. Agric. Food Chem. 2011, 59, 9280–9289. [Google Scholar] [CrossRef] [PubMed]
- Vij, N.; Min, T.; Marasigan, R.; Belcher, C.N.; Mazur, S.; Ding, H.; Yong, K.-T.; Roy, I. Development of PEGylated PLGA nanoparticle for controlled and sustained drug delivery in cystic fibrosis. J. Nanobiotechnol. 2010, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.H.; Lazo, J.S. High Dose Continuous Infusion of Bleomycin in Mice: A New Model for Drug-Induced Pulmonary Fibrosis. J. Pharm. Exp. Ther. 1987, 243, 1185–1194. [Google Scholar]
- Al-Gabri Naif, A.; Qaid, M.M.; El-shaer, N.H.; Ali, M.H. Abudabos, A.M. Thymoquinone ameliorates pulmonary vascular damage induced by Escherichia coli–derived lipopolysaccharide via cytokine downregulation in rats. Environ. Sci. Poll. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Thomassen, M.J.; Buhrow, L.T.; Connors, M.J.; Takao Kaneko, F.; Erzurum, S.C.; Kavuru, M.S. Nitric Oxide Inhibits Inflammatory Cytokine Production by Human Alveolar Macrophages. Americ. J. Res. Cell Mol. Biol. 1997, 17, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Bancroft, J.D.; Gamble, M. The Hematoxylin and eosin. In Theory and Practice of Histological Techniques, 7th ed.; Suvarna, S.K., Layton, C., Bancroft, J.D., Eds.; Churchill Livingstone: Edinburgh, Scotland; New York, NY, USA, 2013; pp. 179–220. [Google Scholar]
- Crookham, J.; Dapson, R. Hazardous Chemicals in the Histopathology Laboratory, 2nd ed.; Anatech: Olivedal, Randburg, 1991. [Google Scholar]
- Khafaga, A.F. Exogenous phosphatidylcholine supplementation retrieve aluminum-induced toxicity in male albino rats. Environ. Sci. Pollut. Res. 2017, 24, 15589–15598. [Google Scholar] [CrossRef] [PubMed]
- Al-Gabri, N.A.; Ali, A.-M.; El-Sayed, A.-A.; Hamed, M. Pathological Study on the Role of Thymoquinone in Experimentally Induced Acute Lung Injury in Rats. Zag. Vet. J. 2017, 45, 228–237. [Google Scholar] [CrossRef]
- Hsu, S.; Raine, L.; Fanger, H. Use of biotin-avidin-peroxi dase conplex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody techniques. Am. J. Clin. Pathol. 1981, 75, 816–821. [Google Scholar] [CrossRef]
- Schneider, C.A., 1st; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Hayat, M.E. Basic Techniques for Transmission Electron Microscopy; Elsevier: Berlin, Germany, 2012. [Google Scholar]
- SAS. Statistical Analysis System. Version 9; SAS Institute Inc Cary: Cary, NC, USA, 2002. [Google Scholar]
- Mansour, M.A.; Nagi, M.N.; El-Khatib, A.S.; Al-Bekairi, A.M. Effects of thymoquinone on antioxidant enzyme activities, lipidperoxidation and DT-diaphorase in different tissues of mice: A possible mechanism of action. Cell Biochem. Func. 2002, 20, 143–151. [Google Scholar] [CrossRef]
- Umar, S.; Zargan, J.; Umar, K.; Ahmad, S.; Katiyar, C.K.; Khan, H.A. Modulation of the oxidative stress and inflammatory cytokine response by thymoquinone in the collagen induced arthritis in Wistar rats. Chem. Biol. Interac. 2012, 197, 40–46. [Google Scholar]
- Shewita, R.S.; Taha, A.E. Effect of dietary supplementation of different levels of black seed (Nigella Sativa L.) on growth performance, immunological, hematological and carcass parameters of broiler chicks. World Acad. Sci. Eng. Technol. 2011, 77, 788–794. [Google Scholar]
- Woo, C.C.; Kumar, A.P.; Sethi, G.; Tan, K.H.B. Thymoquinone: Potential cure for inflammatory disorders and cancer. Biochem. Pharm. 2012, 83, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Wahab, W.M. Protective effect of thymoquinone on sodium fluoride-induced hepatotoxicity and oxidative stress in rats. J. Basic Appl. Zool. 2013, 66, 263–270. [Google Scholar] [CrossRef]
- Harzallah, H.J.; Kouidhi, B.; Flamini, G.; Bakhrouf, A.; Mahjoub, T. Chemical composition, antimicrobial potential against cariogenic bacteria and cytotoxic activity of Tunisian Nigella sativa essential oil and thymoquinone. Food Chem. 2013, 129, 1469–1474. [Google Scholar] [CrossRef]
- El-Mahmoudy, A.; Matsuyama, H.; Borgan, M.A.; Shimizu, Y.; El-Sayed, M.G.; Minamoto, N.; Takewaki, T. thymoquinone suppresses expression of inducible nitric oxide synthase in rat macrophages. Int. Immunopharmacol. 2002, 2, 1603–1611. [Google Scholar] [CrossRef]
- Abidi, A.; Bahri, S.; Ben Khamsa, S.; Legrand, A.A. Comparative Study of Intratracheal and Aerosolization Instillations of Bleomycin Inducing Experimental Lung Fibrosis in Rat. Toxicol. Mech. Methods 2019, 29, 75–85. [Google Scholar] [CrossRef]
- Murphy, S.; Lim, R.; Dickinson, H.; Acharya, R.; Rosli, S.; Jenkin, G.; Wallace, E. Human Amnion Epithelial Cells Prevent Bleomycin-Induced Lung Injury and Preserve Lung Function. Cell Transplant. 2011, 20, 909–924. [Google Scholar] [CrossRef]
- Hosseini, S.; Imenshahidi, M.; Hosseinzadeh, H.; Karimi, G. Effects of plant extracts and bioactive compounds on attenuation of bleomycin-induced pulmonary fibrosis. Biomed. Pharm. 2018, 107, 1454–1465. [Google Scholar] [CrossRef]
- El-Khouly, D.; El-Bakly, W.M.; Awad, A.S.; El-Mesallamy, H.O.; El-Demerdash, E. Thymoquinone Blocks lung injury and fibrosis by attenuating bleomycin-induced oxidative stress and activation of nuclear factor kappa-b in rats. Toxicology 2012, 302, 106–113. [Google Scholar] [CrossRef]
- Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-beta: The master regulator of fibrosis. Nat. Rev. Nephrol. 2016, 12, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Khafaga, A.F.; Abu-Ahmed, H.M.; El-Khamary, A.N.; Elmehasseb, I.M.; Shaheen, H.M. Enhancement of Equid Distal Limb Wounds Healing by Topical Application of Silver Nanoparticles. J. Equine Vet. Sci. 2018, 61, 76–87. [Google Scholar] [CrossRef]
- Cheresh, P.; Kim, S.J.; Tulasiram, S.; Kamp, D.W. Oxidative stress and pulmonary fibrosis. Biochim. Biophys. Acta 2013, 1832, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, F.; Uchibori, R.; Sehara, Y.; Saga, Y.; Urabe, M.; Mizukami, H.; Hagiwara, K.; Kume, A. AAV6-Mediated IL-10 Expression in the lung ameliorates bleomycin-induced pulmonary fibrosis in mice. Hum. Gene Ther. 2018, 29, 1242–1251. [Google Scholar] [CrossRef]
- Rajput, S.; Kumar, B.P.; Banik, P.; Parida, S.; Mandal, M. Thymoquinone restores radiation-induced TGF-β expression and abrogates EMT in chemoradiotherapy of breast cancer cells. J. Cell. Physiol. 2015, 230, 620–629. [Google Scholar] [CrossRef]
- Abdelghany, A.H.; BaSalamah, M.A.; Idris, S.; Ahmad, J.; Refaat, B. The Fibrolytic Potentials of Vitamin D and Thymoquinone Remedial Therapies: Insights from Liver Fibrosis Established by CCl4 in Rats. J. Trans. Med. 2016, 14, 281. [Google Scholar] [CrossRef]
- Hadi, V.; Kheirouri, S.; Alizadeh, M.; Khabbazi, A.; Hosseini, H. Effects of Nigella Sativa oil extract on inflammatory cytokine response and oxidative stress status in patients with Rheumatoid Arthritis: A randomized, double-blind, placebo-controlled clinical trial. Avicenna J. Phytomed. 2016, 6, 34. [Google Scholar]
- Couper, K.N.; Blount, D.G. Riley EM. IL-10: The master regulator of immunity to infection. J. Immunol. 2008, 180, 5771–5777. [Google Scholar] [CrossRef]
- Kinnula, V.L.; Fattman, C.L.; Tan, R.J.; Oury, T.D. Oxidative stress in pulmonary fibrosis: A possible role for redox modulatory therapy. Am. J. Respir. Crit. Care Med. 2005, 172, 417–422. [Google Scholar] [CrossRef]
- Kliment, C.R.; Oury, T.D. Oxidative stress, extracellular matrix targets, and idiopathic pulmonary fibrosis. Free Radic. Biol. Med. 2010, 49, 707–717. [Google Scholar] [CrossRef]
- Fois, A.G.; Paliogiannis, P.; Sotgia, S.; Mangoni, A.A.; Zinellu, E.; Pirina, P.; Carru, C.; Zinellu, A. Evaluation of oxidative stress biomarkers in idiopathic pulmonary fibrosis and therapeutic applications: A systematic review. Respir. Res. 2018, 19, 51. [Google Scholar] [CrossRef] [PubMed]
- Metwally, M.M.M.; Al-Gabri, N.A. Remedial effects of exogenous surfactant in Pseudomonas aeruginosa LPS-induced acute lung injury in rat model. J. Entomol. Zool. Stud. 2017, 5, 1028–1035. [Google Scholar]
- Zolnik, B.S.; Leary, P.E.; Burgess, D.J. Elevated Temperature Accelerated Release Testing of PLGA Microspheres. J. Contr. Release 2006, 112, 293–300. [Google Scholar] [CrossRef] [PubMed]
| Scored Lesions | Experiment Groups | |||
|---|---|---|---|---|
| Bleomycin | Bleomycin + TQ-PLGA-PVA-NPs | TQ-PLGA-PVA-NPs | Control | |
| Fibrosis | 3 | 1 | 0 | 0 |
| Focal pneumonic areas | 3 | 1 | 0 | 0 |
| Hyperplasia and hypertrophy of pneumocytes I & II | 3 | 1 | 0 | 0 |
| Thickening of alveoli septal | 3 | 1 | 1 | 0 |
| Atelectasis | 2 | 0 | 1 | 0 |
| Alveolitis | 3 | 1 | 1 | 0 |
| Compensatory emphysema | 2 | 1 | 1 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saghir, S.A.M.; Al-Gabri, N.A.; Khafaga, A.F.; El-shaer, N.H.; Alhumaidh, K.A.; Elsadek, M.F.; Ahmed, B.M.; Alkhawtani, D.M.; Abd El-Hack, M.E. Thymoquinone-PLGA-PVA Nanoparticles Ameliorate Bleomycin-Induced Pulmonary Fibrosis in Rats via Regulation of Inflammatory Cytokines and iNOS Signaling. Animals 2019, 9, 951. https://doi.org/10.3390/ani9110951
Saghir SAM, Al-Gabri NA, Khafaga AF, El-shaer NH, Alhumaidh KA, Elsadek MF, Ahmed BM, Alkhawtani DM, Abd El-Hack ME. Thymoquinone-PLGA-PVA Nanoparticles Ameliorate Bleomycin-Induced Pulmonary Fibrosis in Rats via Regulation of Inflammatory Cytokines and iNOS Signaling. Animals. 2019; 9(11):951. https://doi.org/10.3390/ani9110951
Chicago/Turabian StyleSaghir, Sultan A. M., Naif A. Al-Gabri, Asmaa F. Khafaga, Nahla H. El-shaer, Khaled A. Alhumaidh, Mohamed F. Elsadek, Badreldin M. Ahmed, Daniyah M. Alkhawtani, and Mohamed E. Abd El-Hack. 2019. "Thymoquinone-PLGA-PVA Nanoparticles Ameliorate Bleomycin-Induced Pulmonary Fibrosis in Rats via Regulation of Inflammatory Cytokines and iNOS Signaling" Animals 9, no. 11: 951. https://doi.org/10.3390/ani9110951
APA StyleSaghir, S. A. M., Al-Gabri, N. A., Khafaga, A. F., El-shaer, N. H., Alhumaidh, K. A., Elsadek, M. F., Ahmed, B. M., Alkhawtani, D. M., & Abd El-Hack, M. E. (2019). Thymoquinone-PLGA-PVA Nanoparticles Ameliorate Bleomycin-Induced Pulmonary Fibrosis in Rats via Regulation of Inflammatory Cytokines and iNOS Signaling. Animals, 9(11), 951. https://doi.org/10.3390/ani9110951









