Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status
Simple Summary
Abstract
1. Introduction
2. Fat Supplementation and Lipid Metabolism
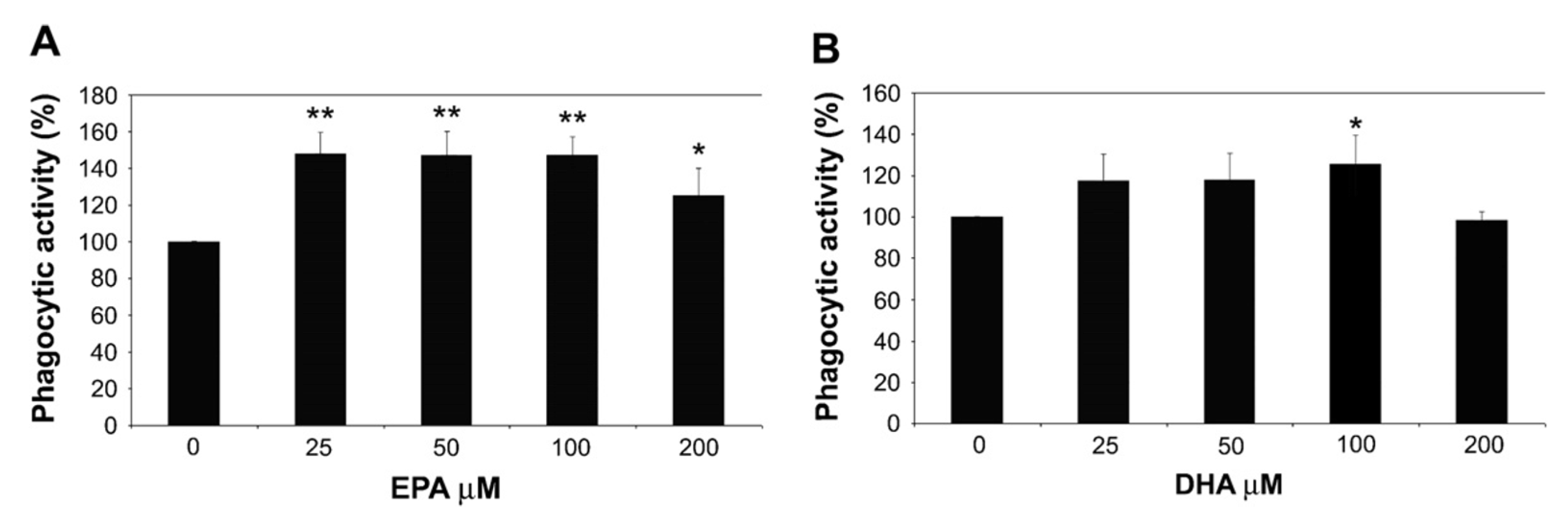
3. Fat Supplementation and Immune Response
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bhat, Z.; Bhat, H. Milk and dairy products as functional foods: A review. Int. J. Dairy Sci. 2011, 6, 1–12. [Google Scholar] [CrossRef]
- Park, Y.W. Bioactive Components in Goat Milk. In Bioactive Components in Milk and Dairy Products; Park, Y.W., Ed.; Wiley-Blackwell: Ames, IA, USA, 2009; pp. 43–81. [Google Scholar] [CrossRef]
- Park, Y.W.; Juárez, M.; Ramos, M.; Haenlein, G.F.W. Physico-chemical characteristics of goat and sheep milk. Small Rumin. Res. 2007, 68, 88–113. [Google Scholar] [CrossRef]
- Yadav, A.K.; Singh, J.; Yadav, S.K. Composition, nutritional and therapeutic values of goat milk: A review. Asian J. Dairy Food Res. 2016, 35, 7. [Google Scholar] [CrossRef]
- Clapham, W.M.; Foster, J.G.; Neel, J.P.; Fedders, J.M. Fatty acid composition of traditional and novel forages. J. Agric. Food Chem. 2005, 53, 10068–10073. [Google Scholar] [CrossRef] [PubMed]
- Chilliard, Y.; Glasser, F.; Ferlay, A.; Bernard, L.; Rouel, J.; Doreau, M. Diet, rumen biohydrogenation and nutritional quality of cow and goat milk fat. Eur. J. Lipid Sci. Technol. 2007, 109, 828–855. [Google Scholar] [CrossRef]
- Markiewicz-Kęszycka, M.; Czyżak-Runowska, G.; Lipińska, P.; Wójtowski, J. Fatty Acid Profile of Milk—A Review. Bull. Vet. Inst. Pulawy 2013, 57, 135–139. [Google Scholar] [CrossRef]
- Bickerstaffe, R.; Noakes, D.; Annison, E. Quantitative aspects of fatty acid biohydrogenation, absorption and transfer into milk fat in the lactating goat, with special reference to the cis-and trans-isomers of octadecenoate and linoleate. Biochem. J. 1972, 130, 607–617. [Google Scholar] [CrossRef]
- Mattos, W.; Palmquist, D.L. Biohydrogenation and availability of linoleic acid in lactating cows. J. Nutr. 1977, 107, 1755–1761. [Google Scholar] [CrossRef]
- Bauman, D.E.; Lock, A.L. Concepts in lipid digestion and metabolism in dairy cows. In Proceedings of the Tri-State Dairy Nutrition Conference, Ft. Wayne, IN, USA, 25–26 April 2006. [Google Scholar]
- Buccioni, A.; Decandia, M.; Minieri, S.; Molle, G.; Cabiddu, A. Lipid metabolism in the rumen: New insights on lipolysis and biohydrogenation with an emphasis on the role of endogenous plant factors. Anim. Feed Sci. Technol. 2012, 174, 1–25. [Google Scholar] [CrossRef]
- Conte, G.; Mele, M.; Chessa, S.; Castiglioni, B.; Serra, A.; Pagnacco, G.; Secchiari, P. Diacylglycerol acyltransferase 1, stearoyl-CoA desaturase 1, and sterol regulatory element binding protein 1 gene polymorphisms and milk fatty acid composition in Italian Brown cattle. J. Dairy Sci. 2010, 93, 753–763. [Google Scholar] [CrossRef]
- Morand-Fehr, P.; Fedele, V.; Decandia, M.; Le Frileux, Y. Influence of farming and feeding systems on composition and quality of goat and sheep milk. Small Rumin. Res. 2007, 68, 20–34. [Google Scholar] [CrossRef]
- O’Callaghan, T.F.; Hennessy, D.; McAuliffe, S.; Kilcawley, K.N.; O’Donovan, M.; Dillon, P.; Ross, R.P.; Stanton, C. Effect of pasture versus indoor feeding systems on raw milk composition and quality over an entire lactation. J. Dairy Sci. 2016, 99, 9424–9440. [Google Scholar] [CrossRef] [PubMed]
- Corazzin, M.; Romanzin, A.; Sepulcri, A.; Pinosa, M.; Piasentier, E.; Bovolenta, S. Fatty Acid Profiles of Cow’s Milk and Cheese as Affected by Mountain Pasture Type and Concentrate Supplementation. Animals 2019, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Serment, A.; Schmidely, P.; Giger-Reverdin, S.; Chapoutot, P.; Sauvant, D. Effects of the percentage of concentrate on rumen fermentation, nutrient digestibility, plasma metabolites, and milk composition in mid-lactation goats. J. Dairy Sci. 2011, 94, 3960–3972. [Google Scholar] [CrossRef] [PubMed]
- Sterk, A.; Johansson, B.E.; Taweel, H.Z.; Murphy, M.; van Vuuren, A.M.; Hendriks, W.H.; Dijkstra, J. Effects of forage type, forage to concentrate ratio, and crushed linseed supplementation on milk fatty acid profile in lactating dairy cows. J. Dairy Sci. 2011, 94, 6078–6091. [Google Scholar] [CrossRef]
- Butler, G.; Stergiadis, S.; Seal, C.; Eyre, M.; Leifert, C. Fat composition of organic and conventional retail milk in northeast England. J. Dairy Sci. 2011, 94, 24–36. [Google Scholar] [CrossRef]
- Lopez, A.; Vasconi, M.; Moretti, V.M.; Bellagamba, F. Fatty Acid Profile in Goat Milk from High- and Low-Input Conventional and Organic Systems. Animals 2019, 9, 452. [Google Scholar] [CrossRef]
- Boeckaert, C.; Vlaeminck, B.; Dijkstra, J.; Issa-Zacharia, A.; Van Nespen, T.; Van Straalen, W.; Fievez, V. Effect of dietary starch or micro algae supplementation on rumen fermentation and milk fatty acid composition of dairy cows. J. Dairy Sci. 2008, 91, 4714–4727. [Google Scholar] [CrossRef]
- Cattaneo, D.; Dell’Orto, V.; Varisco, G.; Agazzi, A.; Savoini, G. Enrichment in n-3 fatty acids of goat’s colostrum and milk by maternal fish oil supplementation. Small Rumin. Res. 2006, 64, 22–29. [Google Scholar] [CrossRef]
- Póti, P.; Pajor, F.; Bodnár, Á.; Penksza, K.; Köles, P. Effect of micro-alga supplementation on goat and cow milk fatty acid composition. Chil. J. Agric. Res. 2015, 75, 259–263. [Google Scholar] [CrossRef]
- Toral, P.G.; Frutos, P.; Hervas, G.; Gomez-Cortes, P.; Juarez, M.; de la Fuente, M.A. Changes in milk fatty acid profile and animal performance in response to fish oil supplementation, alone or in combination with sunflower oil, in dairy ewes. J. Dairy Sci. 2010, 93, 1604–1615. [Google Scholar] [CrossRef] [PubMed]
- Shingfield, K.J.; Chilliard, Y.; Toivonen, V.; Kairenius, P.; Givens, D.I. Trans fatty acids and bioactive lipids in ruminant milk. Adv. Exp. Med. Biol. 2008, 606, 3–65. [Google Scholar] [CrossRef] [PubMed]
- Cook, H.W.; McMaster, C.R. Chapter 7 Fatty acid desaturation and chain elongation in eukaryotes. In New Comprehensive Biochemistry; Elsevier: Amsterdam, The Netherlands, 2002; Volume 36, pp. 181–204. [Google Scholar]
- Lock, A.L.; Bauman, D.E. Modifying milk fat composition of dairy cows to enhance fatty acids beneficial to human health. Lipids 2004, 39, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Lanier, J.S.; Corl, B.A. Challenges in enriching milk fat with polyunsaturated fatty acids. J. Anim. Sci. Biotechnol. 2015, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Zymon, M.; Strzetelski, J.; Skrzyński, G. Aspects of appropriate feeding of cows for production of milk enriched in the fatty acids, EPA and DHA. A review. J. Anim. Feed Sci. 2014, 23, 109–116. [Google Scholar] [CrossRef]
- Kitessa, S.M.; Gulati, S.K.; Ashes, J.R.; Fleck, E.; Scott, T.W.; Nichols, P.D. Utilisation of fish oil in ruminants: II. Transfer of fish oil fatty acids into goats’ milk. Anim. Feed Sci. Technol. 2001, 89, 201–208. [Google Scholar] [CrossRef]
- Olloqui, E.J.; Castañeda-Ovando, A.; Contreras-López, E.; Hernandez-Sanchez, D.; Tapia-Maruri, D.; Piloni-Martini, J.; Añorve-Morga, J. Encapsulation of Fish Oil Into Low-Cost Alginate Beads and EPA-DHA Release in a Rumino-Intestinal In Vitro Digestion Model. Eur. J. Lipid Sci. Technol. 2018, 120, 1800036. [Google Scholar] [CrossRef]
- Castaneda-Gutierrez, E.; de Veth, M.J.; Lock, A.L.; Dwyer, D.A.; Murphy, K.D.; Bauman, D.E. Effect of supplementation with calcium salts of fish oil on n-3 fatty acids in milk fat. J. Dairy Sci. 2007, 90, 4149–4156. [Google Scholar] [CrossRef]
- Chilliard, Y.; Ferlay, A.; Rouel, J.; Lamberet, G. A review of nutritional and physiological factors affecting goat milk lipid synthesis and lipolysis. J. Dairy Sci. 2003, 86, 1751–1770. [Google Scholar] [CrossRef]
- Sanz Sampelayo, M.R.; Chilliard, Y.; Schmidely, P.; Boza, J. Influence of type of diet on the fat constituents of goat and sheep milk. Small Rumin. Res. 2007, 68, 42–63. [Google Scholar] [CrossRef]
- Fougere, H.; Delavaud, C.; Bernard, L. Diets supplemented with starch and corn oil, marine algae, or hydrogenated palm oil differentially modulate milk fat secretion and composition in cows and goats: A comparative study. J. Dairy Sci. 2018, 101, 8429–8445. [Google Scholar] [CrossRef] [PubMed]
- Toral, P.G.; Chilliard, Y.; Rouel, J.; Leskinen, H.; Shingfield, K.J.; Bernard, L. Comparison of the nutritional regulation of milk fat secretion and composition in cows and goats. J. Dairy Sci. 2015, 98, 7277–7297. [Google Scholar] [CrossRef] [PubMed]
- Luna, P.; Bach, A.; Juárez, M.; de la Fuente, M.A. Effect of a diet enriched in whole linseed and sunflower oil on goat milk fatty acid composition and conjugated linoleic acid isomer profile. J. Dairy Sci. 2008, 91, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Martínez Marín, A.L.; Gómez-Cortés, P.; Gómez Castro, A.G.; Juárez, M.; Pérez Alba, L.M.; Pérez Hernández, M.; de la Fuente, M.A. Animal performance and milk fatty acid profile of dairy goats fed diets with different unsaturated plant oils. J. Dairy Sci. 2011, 94, 5359–5368. [Google Scholar] [CrossRef] [PubMed]
- Gulliver, C.E.; Friend, M.A.; King, B.J.; Clayton, E.H. The role of omega-3 polyunsaturated fatty acids in reproduction of sheep and cattle. Anim. Reprod. Sci. 2012, 131, 9–22. [Google Scholar] [CrossRef]
- Mahla, A.S.; Chaudhari, R.K.; Verma, A.K.; Singh, A.K.; Singh, S.K.; Singh, G.; Sarkar, M.; Dutta, N.; Kumar, H.; Krishnaswamy, N. Effect of dietary supplementation of omega-3 polyunsaturated fatty acid (PUFA) rich fish oil on reproductive performance of the goat (Capra hircus). Theriogenology 2017, 99, 79–89. [Google Scholar] [CrossRef]
- Silvestre, F.T.; Carvalho, T.S.; Francisco, N.; Santos, J.E.; Staples, C.R.; Jenkins, T.C.; Thatcher, W.W. Effects of differential supplementation of fatty acids during the peripartum and breeding periods of Holstein cows: I. Uterine and metabolic responses, reproduction, and lactation. J. Dairy Sci. 2011, 94, 189–204. [Google Scholar] [CrossRef]
- Boeckaert, C.; Fievez, V.; Van Hecke, D.; Verstraete, W.; Boon, N. Changes in rumen biohydrogenation intermediates and ciliate protozoa diversity after algae supplementation to dairy cattle. Eur. J. Lipid Sci. Technol. 2007, 109, 767–777. [Google Scholar] [CrossRef]
- Zhao, T.; Ma, Y.; Qu, Y.; Luo, H.; Liu, K.; Zuo, Z.; Lu, X. Effect of dietary oil sources on fatty acid composition of ruminal digesta and populations of specific bacteria involved in hydrogenation of 18-carbon unsaturated fatty acid in finishing lambs. Small Rumin. Res. 2016, 144, 126–134. [Google Scholar] [CrossRef]
- Agazzi, A.; Invernizzi, G.; Campagnoli, A.; Ferroni, M.; Fanelli, A.; Cattaneo, D.; Galmozzi, A.; Crestani, M.; Dell’Orto, V.; Savoini, G. Effect of different dietary fats on hepatic gene expression in transition dairy goats. Small Rumin. Res. 2010, 93, 31–40. [Google Scholar] [CrossRef]
- Buccioni, A.; Pauselli, M.; Viti, C.; Minieri, S.; Pallara, G.; Roscini, V.; Rapaccini, S.; Marinucci, M.T.; Lupi, P.; Conte, G.; et al. Milk fatty acid composition, rumen microbial population, and animal performances in response to diets rich in linoleic acid supplemented with chestnut or quebracho tannins in dairy ewes. J. Dairy Sci. 2015, 98, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Tsiplakou, E.; Zervas, G. Changes in milk and plasma fatty acid profile in response to fish and soybean oil supplementation in dairy sheep. J. Dairy Res. 2013, 80, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, S.; Spener, F. Conjugated linoleic acids as functional food: An insight into their health benefits. Nutr. Metab. 2009, 6, 36. [Google Scholar] [CrossRef]
- Baumgard, L.H.; Matitashvili, E.; Corl, B.A.; Dwyer, D.A.; Bauman, D.E. trans-10, cis-12 conjugated linoleic acid decreases lipogenic rates and expression of genes involved in milk lipid synthesis in dairy cows. J. Dairy Sci. 2002, 85, 2155–2163. [Google Scholar] [CrossRef]
- Chilliard, Y.; Ferlay, A. Dietary lipids and forages interactions on cow and goat milk fatty acid composition and sensory properties. Reprod. Nutr. Dev. 2004, 44, 467–492. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Ma, Y.; Wang, H.; Loor, J.J.; Xu, H.; Shi, H.; Luo, J. Trans10, cis12 conjugated linoleic acid increases triacylglycerol accumulation in goat mammary epithelial cells in vitro. Anim. Sci. J. 2018, 89, 432–440. [Google Scholar] [CrossRef]
- Vargas-Bello-Perez, E.; Zhao, W.; Bionaz, M.; Luo, J.; Loor, J.J. Nutrigenomic Effect of Saturated and Unsaturated Long Chain Fatty Acids on Lipid-Related Genes in Goat Mammary Epithelial Cells: What Is the Role of PPARgamma? Vet. Sci. 2019, 6, 54. [Google Scholar] [CrossRef]
- Bernard, L.; Toral, P.G.; Chilliard, Y. Comparison of mammary lipid metabolism in dairy cows and goats fed diets supplemented with starch, plant oil, or fish oil. J. Dairy Sci. 2017, 100, 9338–9351. [Google Scholar] [CrossRef]
- Shingfield, K.J.; Bernard, L.; Leroux, C.; Chilliard, Y. Role of trans fatty acids in the nutritional regulation of mammary lipogenesis in ruminants. Animal 2010, 4, 1140–1166. [Google Scholar] [CrossRef]
- Bernard, L.; Bonnet, M.; Delavaud, C.; Delosière, M.; Ferlay, A.; Fougère, H.; Graulet, B. Milk Fat Globule in Ruminant: Major and Minor Compounds, Nutritional Regulation and Differences Among Species. Eur. J. Lipid Sci. Technol. 2018, 120, 1700039. [Google Scholar] [CrossRef]
- Loor, J.J.; Doreau, M.; Chardigny, J.M.; Ollier, A.; Sebedio, J.L.; Chilliard, Y. Effects of ruminal or duodenal supply of fish oil on milk fat secretion and profiles of trans-fatty acids and conjugated linoleic acid isomers in dairy cows fed maize silage. Anim. Feed Sci. Technol. 2005, 119, 227–246. [Google Scholar] [CrossRef]
- Toral, P.G.; Rouel, J.; Bernard, L.; Chilliard, Y. Interaction between fish oil and plant oils or starchy concentrates in the diet: Effects on dairy performance and milk fatty acid composition in goats. Anim. Feed Sci. Technol. 2014, 198, 67–82. [Google Scholar] [CrossRef]
- Toral, P.G.; Hervás, G.; Carreño, D.; Frutos, P. Does supplemental 18:0 alleviate fish oil-induced milk fat depression in dairy ewes? J. Dairy Sci. 2016, 99, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Vega, A.; Toral, P.G.; Gutiérrez-Gil, B.; Hervás, G.; Arranz, J.J.; Frutos, P. Elucidating fish oil-induced milk fat depression in dairy sheep: Milk somatic cell transcriptome analysis. Sci. Rep. 2017, 7, 45905. [Google Scholar] [CrossRef] [PubMed]
- Lecchi, C.; Invernizzi, G.; Agazzi, A.; Modina, S.; Sartorelli, P.; Savoini, G.; Ceciliani, F. Effects of EPA and DHA on lipid droplet accumulation and mRNA abundance of PAT proteins in caprine monocytes. Res. Vet. Sci. 2013, 94, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Rakhshandehroo, M.; Knoch, B.; Müller, M.; Kersten, S. Peroxisome Proliferator-Activated Receptor Alpha Target Genes. PPAR Res. 2010, 2010, 612089. [Google Scholar] [CrossRef]
- Mandard, S.; Muller, M.; Kersten, S. Peroxisome proliferator-activated receptor alpha target genes. Cell. Mol. Life Sci. 2004, 61, 393–416. [Google Scholar] [CrossRef]
- Bionaz, M.; Chen, S.; Khan, M.J.; Loor, J.J. Functional Role of PPARs in Ruminants: Potential Targets for Fine-Tuning Metabolism during Growth and Lactation. PPAR Res. 2013, 2013, 684159. [Google Scholar] [CrossRef]
- Grygiel-Gorniak, B. Peroxisome proliferator-activated receptors and their ligands: Nutritional and clinical implications—A review. Nutr. J. 2014, 13, 17. [Google Scholar] [CrossRef]
- Bionaz, M.; Thering, B.J.; Loor, J.J. Fine metabolic regulation in ruminants via nutrient-gene interactions: Saturated long-chain fatty acids increase expression of genes involved in lipid metabolism and immune response partly through PPAR-alpha activation. Br. J. Nutr. 2012, 107, 179–191. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Rajion, M.A.; Meng, G.Y.; Farjam, A.S.; Oskoueian, E.; Jafari, S. Diet high in α-linolenic acid up-regulate PPAR-α gene expression in the liver of goats. Electron. J. Biotechnol. 2015, 18, 210–214. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Rajion, M.A.; Jafari, S.; Faseleh Jahromi, M.; Oskoueian, E.; Qurni Sazili, A.; Goh, Y.M.; Ghaffari, M.H. Effects of dietary n-6: N-3 polyunsaturated fatty acid ratios on meat quality, carcass characteristics, tissue fatty acid profiles, and expression of lipogenic genes in growing goats. PLoS ONE 2018, 13, e0188369. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Rajion, M.A.; Goh, Y.M. Effects of oils rich in linoleic and alpha-linolenic acids on fatty acid profile and gene expression in goat meat. Nutrients 2014, 6, 3913–3928. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Rajion, M.A.; Goh, Y.M.; Sazili, A.Q.; Schonewille, J.T. Effect of linseed oil dietary supplementation on fatty acid composition and gene expression in adipose tissue of growing goats. BioMed Res. Int. 2013, 2013, 194625. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, G.; Modina, S.; Corbani, D.; Bronzo, V.; Pisani, L.F.; Caputo, J.M.; Agazzi, A.; Dell’Orto, V.; Savoini, G. Hepatic and subcutaneous adipose tissue variations in transition dairy goats fed saturated or unsaturated fat supplemented diets. Small Rumin. Res. 2016, 144, 211–219. [Google Scholar] [CrossRef]
- Ballou, M.A.; Gomes, R.C.; Juchem, S.O.; DePeters, E.J. Effects of dietary supplemental fish oil during the peripartum period on blood metabolites and hepatic fatty acid compositions and total triacylglycerol concentrations of multiparous Holstein cows. J. Dairy Sci. 2009, 92, 657–669. [Google Scholar] [CrossRef]
- Weiss, W.P.; Pinos-Rodríguez, J.M.; Wyatt, D.J. The value of different fat supplements as sources of digestible energy for lactating dairy cows. J. Dairy Sci. 2011, 94, 931–939. [Google Scholar] [CrossRef]
- Caputo, J.M.; Invernizzi, G.; Campagnoli, A.; Farina, G.; Ferroni, M.; Agazzi, A.; Savoini, G. Dietary fish oil and stearate action on adipose lipid metabolism transcriptomics in periparturient dairy goats. In Proceedings of the 21st ASPA Congress, Milan, Italy, 9–12 June 2015; p. 72. [Google Scholar]
- Caputo, J.M.; Invernizzi, G.; Ferroni, M.; Agazzi, A.; Loor, J.J.; Dell’Orto, V.; Savoini, G. Liver gene expression in periparturient dairy goats fed diets enriched with stearate or PUFA. In Proceedings of the 65th Annual Meeting of the European Federation of Animal Science, Copenhagen, Denmark, 25–29 August 2014; p. 402. [Google Scholar]
- Thering, B.J.; Graugnard, D.E.; Piantoni, P.; Loor, J.J. Adipose tissue lipogenic gene networks due to lipid feeding and milk fat depression in lactating cows. J. Dairy Sci. 2009, 92, 4290–4300. [Google Scholar] [CrossRef]
- Reue, K.; Brindley, D.N. Thematic Review Series: Glycerolipids. Multiple roles for lipins/phosphatidate phosphatase enzymes in lipid metabolism. J. Lipid Res. 2008, 49, 2493–2503. [Google Scholar] [CrossRef]
- Finck, B.N.; Gropler, M.C.; Chen, Z.; Leone, T.C.; Croce, M.A.; Harris, T.E.; Lawrence, J.C.; Kelly, D.P. Lipin 1 is an inducible amplifier of the hepatic PGC-1alpha/PPARalpha regulatory pathway. Cell Metab. 2006, 4, 199–210. [Google Scholar] [CrossRef]
- Meana, C.; Pena, L.; Lorden, G.; Esquinas, E.; Guijas, C.; Valdearcos, M.; Balsinde, J.; Balboa, M.A. Lipin-1 integrates lipid synthesis with proinflammatory responses during TLR activation in macrophages. J. Immunol. 2014, 193, 4614–4622. [Google Scholar] [CrossRef] [PubMed]
- Dewanckele, L.; Vlaeminck, B.; Hernandez-Sanabria, E.; Ruiz-González, A.; Debruyne, S.; Jeyanathan, J.; Fievez, V. Rumen Biohydrogenation and Microbial Community Changes Upon Early Life Supplementation of 22:6. Front. Microbiol. 2018, 9, 573. [Google Scholar] [CrossRef] [PubMed]
- Restelli, L.; Marques, A.T.; Savoini, G.; Invernizzi, G.; Carisetti, M.; Lecchi, C.; Bendixen, E.; Ceciliani, F. Saturated or unsaturated fat supplemented maternal diets influence omental adipose tissue proteome of suckling goat-kids. Res. Vet. Sci. 2019, 125, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Heberden, C.; Maximin, E. Epigenetic Effects of N-3 Polyunsaturated Fatty Acids. In Handbook of Nutrition, Diet, and Epigenetics; Patel, V., Preedy, V., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–15. [Google Scholar] [CrossRef]
- Jacometo, C.B.; Schmitt, E.; Pfeifer, L.F.; Schneider, A.; Bado, F.; da Rosa, F.T.; Halfen, S.; Del Pino, F.A.; Loor, J.J.; Correa, M.N.; et al. Linoleic and alpha-linolenic fatty acid consumption over three generations exert cumulative regulation of hepatic expression of genes related to lipid metabolism. Genes Nutr. 2014, 9, 405. [Google Scholar] [CrossRef] [PubMed]
- DeWille, J.W.; Fraker, P.J.; Romsos, D.R. Effects of Essential Fatty Acid Deficiency, and Various Levels of Dietary Polyunsaturated Fatty Acids, on Humoral Immunity in Mice. Nutr. J. 1979, 109, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D. Essential fatty acids and immune response. FASEB J. 1989, 3, 2052–2061. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P. Effects of Saturated Fatty Acids on Serum Lipids and Lipoproteins: A Systematic Review and Regression Analysis; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Lordan, R.; Tsoupras, A.; Mitra, B.; Zabetakis, I. Dairy Fats and Cardiovascular Disease: Do We Really Need to be Concerned? Foods 2018, 7, 29. [Google Scholar] [CrossRef]
- Pimpin, L.; Wu, J.H.; Haskelberg, H.; Del Gobbo, L.; Mozaffarian, D. Is Butter Back? A Systematic Review and Meta-Analysis of Butter Consumption and Risk of Cardiovascular Disease, Diabetes, and Total Mortality. PLoS ONE 2016, 11, e0158118. [Google Scholar] [CrossRef]
- Bauman, D.E.; Mather, I.H.; Wall, R.J.; Lock, A.L. Major advances associated with the biosynthesis of milk. J. Dairy Sci. 2006, 89, 1235–1243. [Google Scholar] [CrossRef]
- Lordan, R.; Zabetakis, I. Invited review: The anti-inflammatory properties of dairy lipids. J. Dairy Sci. 2017, 100, 4197–4212. [Google Scholar] [CrossRef]
- Haenlein, G.F.W. Goat milk in human nutrition. Small Rumin. Res. 2004, 51, 155–163. [Google Scholar] [CrossRef]
- Hollænder, P.L.; Ross, A.B.; Kristensen, M. Whole-grain and blood lipid changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2015, 102, 556–572. [Google Scholar] [CrossRef] [PubMed]
- Sacks Frank, M.; Lichtenstein Alice, H.; Wu Jason, H.Y.; Appel Lawrence, J.; Creager Mark, A.; Kris-Etherton Penny, M.; Miller, M.; Rimm Eric, B.; Rudel Lawrence, L.; Robinson Jennifer, G.; et al. Dietary Fats and Cardiovascular Disease: A Presidential Advisory From the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef]
- Calder, P.C. N-3 Polyunsaturated Fatty Acids, Inflammation, and Inflammatory Diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [CrossRef] [PubMed]
- De Santis, A.; Varela, Y.; Sot, J.; D’Errico, G.; Goni, F.M.; Alonso, A. Omega-3 polyunsaturated fatty acids do not fluidify bilayers in the liquid-crystalline state. Sci. Rep. 2018, 8, 16240. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.Y.; Fuentes, N.R.; Hou, T.Y.; Barhoumi, R.; Li, X.C.; Deutz, N.E.P.; Engelen, M.P.K.J.; McMurray, D.N.; Chapkin, R.S. Remodelling of primary human CD4+ T cell plasma membrane order by n-3 PUFA. Br. J. Nutr. 2018, 119, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, S.C.; Reding, A.M.; Patton, H.M.; Sullivan, T.P.; Stubbs, C.E.; Villalobos-Menuey, E.; Huber, S.A.; Newell, M.K. Endogenous versus exogenous fatty acid availability affects lysosomal acidity and MHC class II expression. J. Lipid Res. 2006, 47, 2525–2537. [Google Scholar] [CrossRef]
- Wong, S.W.; Kwon, M.J.; Choi, A.M.; Kim, H.P.; Nakahira, K.; Hwang, D.H. Fatty acids modulate Toll-like receptor 4 activation through regulation of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. J. Biol. Chem. 2009, 284, 27384–27392. [Google Scholar] [CrossRef]
- Hwang, D.H.; Kim, J.A.; Lee, J.Y. Mechanisms for the activation of Toll-like receptor 2/4 by saturated fatty acids and inhibition by docosahexaenoic acid. Eur. J. Pharmacol. 2016, 785, 24–35. [Google Scholar] [CrossRef]
- De Pascale, C.; Graham, V.; Fowkes, R.C.; Wheeler-Jones, C.P.; Botham, K.M. Suppression of nuclear factor-kappaB activity in macrophages by chylomicron remnants: Modulation by the fatty acid composition of the particles. FEBS J. 2009, 276, 5689–5702. [Google Scholar] [CrossRef]
- Zhao, Y.; Joshi-Barve, S.; Barve, S.; Chen, L.H. Eicosapentaenoic acid prevents LPS-induced TNF-alpha expression by preventing NF-kappaB activation. J. Am. Coll. Nutr. 2004, 23, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X.; Weylandt, K.H. Modulation of inflammatory cytokines by omega-3 fatty acids. Subcell. Biochem. 2008, 49, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Chiang, N.; Dalli, J. New pro-resolving n-3 mediators bridge resolution of infectious inflammation to tissue regeneration. Mol. Asp. Med. 2018, 64, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sordillo, L.M. Symposium review: Oxylipids and the regulation of bovine mammary inflammatory responses. J. Dairy Sci. 2018, 101, 5629–5641. [Google Scholar] [CrossRef]
- Maktoobian Baharanchi, E.; Moradi Sarabi, M.; Naghibalhossaini, F. Effects of Dietary Polyunsaturated Fatty Acids on DNA Methylation and the Expression of DNMT3b and PPARalpha Genes in Rats. Avicenna J. Med. Biotechnol. 2018, 10, 214–219. [Google Scholar]
- Agazzi, A.; Cattaneo, D.; Dell’Orto, V.; Moroni, P.; Bonizzi, L.; Pasotto, D.; Bronzo, V.; Savoini, G. Effect of administration of fish oil on aspects of cell-mediated immune response in periparturient dairy goats. Small Rumin. Res. 2004, 55, 77–83. [Google Scholar] [CrossRef]
- Bragaglio, A.; Braghieri, A.; Napolitano, F.; De Rosa, G.; Riviezzi, A.M.; Surianello, F.; Pacelli, C. Omega-3 supplementation, milk quality and cow immune-competence. Ital. J. Agron. 2015, 10, 9–14. [Google Scholar] [CrossRef]
- Kashiwazaki, Y.; Maede, Y.; Namioka, S. Transformation of bovine peripheral blood lymphocytes in the perinatal period. Nihon Juigaku Zasshi 1985, 47, 337–339. [Google Scholar] [CrossRef]
- Lacetera, N.; Scalia, D.; Bernabucci, U.; Ronchi, B.; Pirazzi, D.; Nardone, A. Lymphocyte functions in overconditioned cows around parturition. J. Dairy Sci. 2005, 88, 2010–2016. [Google Scholar] [CrossRef]
- Bronzo, V.; Puricelli, M.; Agazzi, A.; Invernizzi, G.; Ferroni, M.; Moroni, P.; Savoini, G. Effects of protected fish oil in the diet of periparturient dairy goats on phenotypic variation in blood and milk leukocytes. Animal 2010, 4, 1510–1517. [Google Scholar] [CrossRef]
- Thanasak, J.; Rutten, V.P.; Schonewille, J.T.; Hoek, A.; Beynen, A.C.; Noordhuizen, J.P.; Müller, K.E. Effect of a dietary n-6 polyunsaturated fatty acid supplement on distinct immune functions of goats. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2004, 51, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pisani, L.F.; Lecchi, C.; Invernizzi, G.; Sartorelli, P.; Savoini, G.; Ceciliani, F. In vitro modulatory effect of ω-3 polyunsaturated fatty acid (EPA and DHA) on phagocytosis and ROS production of goat neutrophils. Vet. Immunol. Immunopathol. 2009, 131, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Lecchi, C.; Invernizzi, G.; Agazzi, A.; Ferroni, M.; Pisani, L.F.; Savoini, G.; Ceciliani, F. In vitro modulation of caprine monocyte immune functions by ω-3 polyunsaturated fatty acids. Vet. J. 2011, 189, 353–355. [Google Scholar] [CrossRef] [PubMed]
- Wolins, N.E.; Brasaemle, D.L.; Bickel, P.E. A proposed model of fat packaging by exchangeable lipid droplet proteins. FEBS Lett. 2006, 580, 5484–5491. [Google Scholar] [CrossRef]
- Monson, E.A.; Crosse, K.M.; Das, M.; Helbig, K.J. Lipid droplet density alters the early innate immune response to viral infection. PLoS ONE 2018, 13, e0190597. [Google Scholar] [CrossRef]
- Farina, G.; Invernizzi, G.; Perricone, V.; Agazzi, A.; Cattaneo, D.; Loor, J.J.; Savoini, G. Fatty acid supplementation in transition goats: A transcriptional study related to inflammation. In Proceedings of the 68th EAAP Annual Meeting, Tallin, Estonia, 28 August–1 September 2017. [Google Scholar]
- Contreras, G.A.; Strieder-Barboza, C.; Raphael, W. Adipose tissue lipolysis and remodeling during the transition period of dairy cows. J. Anim. Sci. Biotechnol. 2017, 8, 41. [Google Scholar] [CrossRef]
| Time | Treatment 1 | p-Value 2 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| C | SD | FO | SD | ST | SD | Trt | Time | Trt × Time | |
| −7 | 3200.00 d | 1002 | 2877.69 d | 876 | 3088.80 d | 1010 | 0.60 | <0.01 | <0.01 |
| 7 | 1970.44 e | 523 | 1801.80 e | 548 | 2156.33 e | 718 | |||
| 21 | 1157.74 b,f | 216 | 1851.85 a,e | 638 | 1066.66 b,f | 269 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savoini, G.; Omodei Zorini, F.; Farina, G.; Agazzi, A.; Cattaneo, D.; Invernizzi, G. Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status. Animals 2019, 9, 917. https://doi.org/10.3390/ani9110917
Savoini G, Omodei Zorini F, Farina G, Agazzi A, Cattaneo D, Invernizzi G. Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status. Animals. 2019; 9(11):917. https://doi.org/10.3390/ani9110917
Chicago/Turabian StyleSavoini, Giovanni, Fabio Omodei Zorini, Greta Farina, Alessandro Agazzi, Donata Cattaneo, and Guido Invernizzi. 2019. "Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status" Animals 9, no. 11: 917. https://doi.org/10.3390/ani9110917
APA StyleSavoini, G., Omodei Zorini, F., Farina, G., Agazzi, A., Cattaneo, D., & Invernizzi, G. (2019). Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status. Animals, 9(11), 917. https://doi.org/10.3390/ani9110917





