Effect of Dietary Replacement of Fishmeal by Insect Meal on Growth Performance, Blood Profiles and Economics of Growing Pigs in Kenya
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. BSF Larval Meal (BSFLM) and Experimental Diets
2.2. Proximate, Amino Acids and Mineral Composition of Experimental Diets
2.3. Experimental Animals and Housing
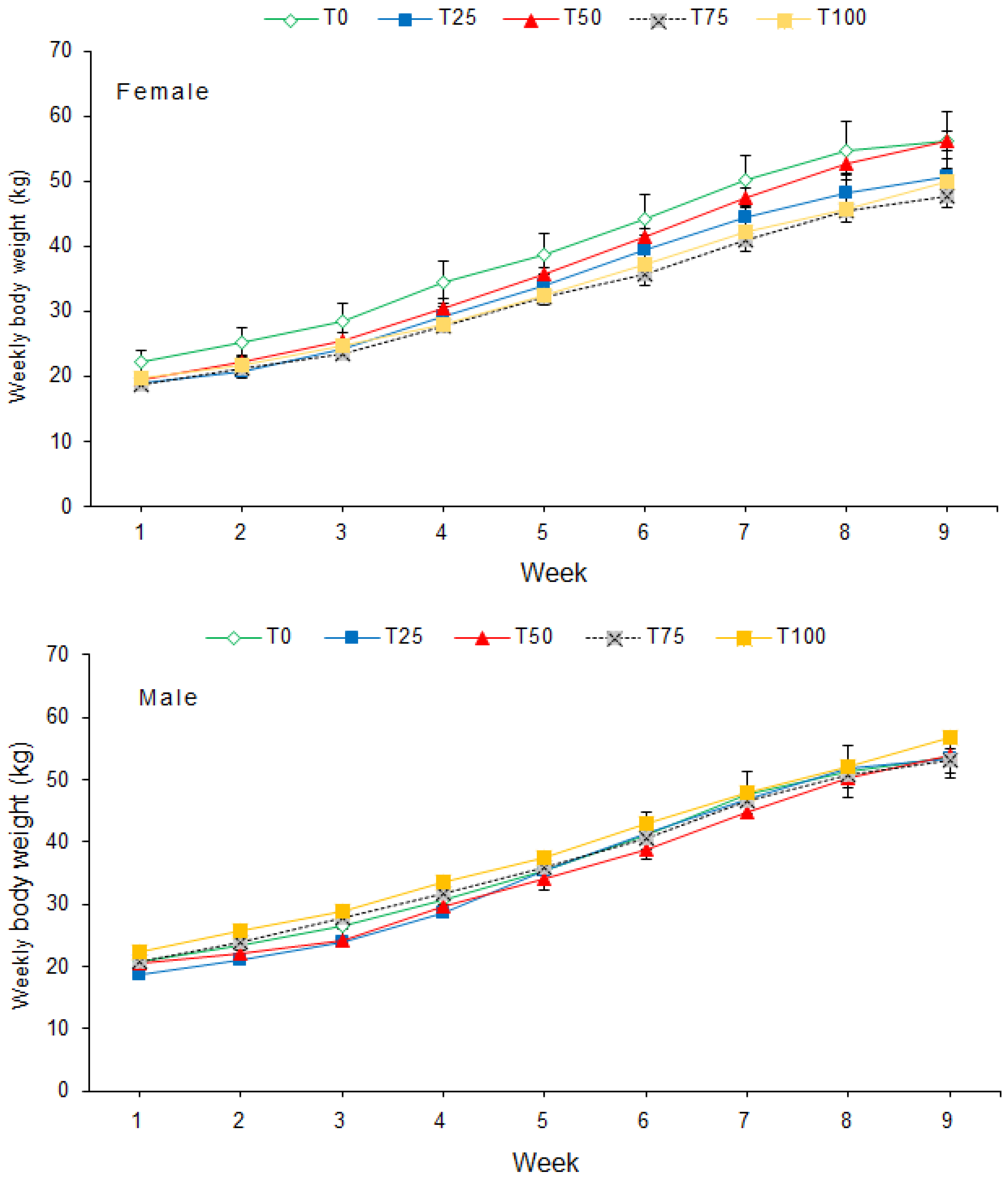
2.4. Growth Performance
2.5. Blood Characteristics
2.6. Hematological and Serum Lipid Parameters
2.7. Economic Analysis
2.8. Statistical Analysis
2.9. Ethical Approval
3. Results
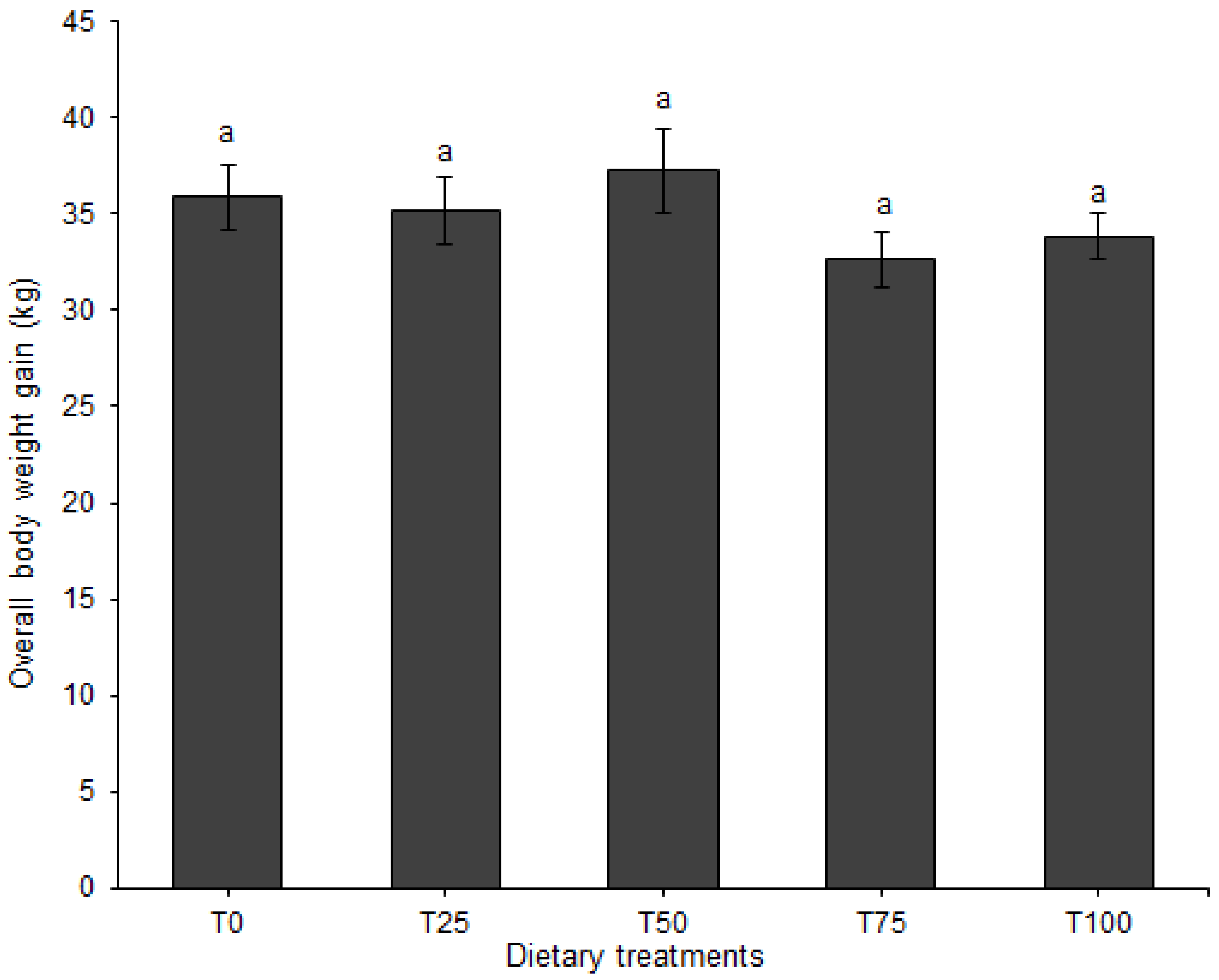
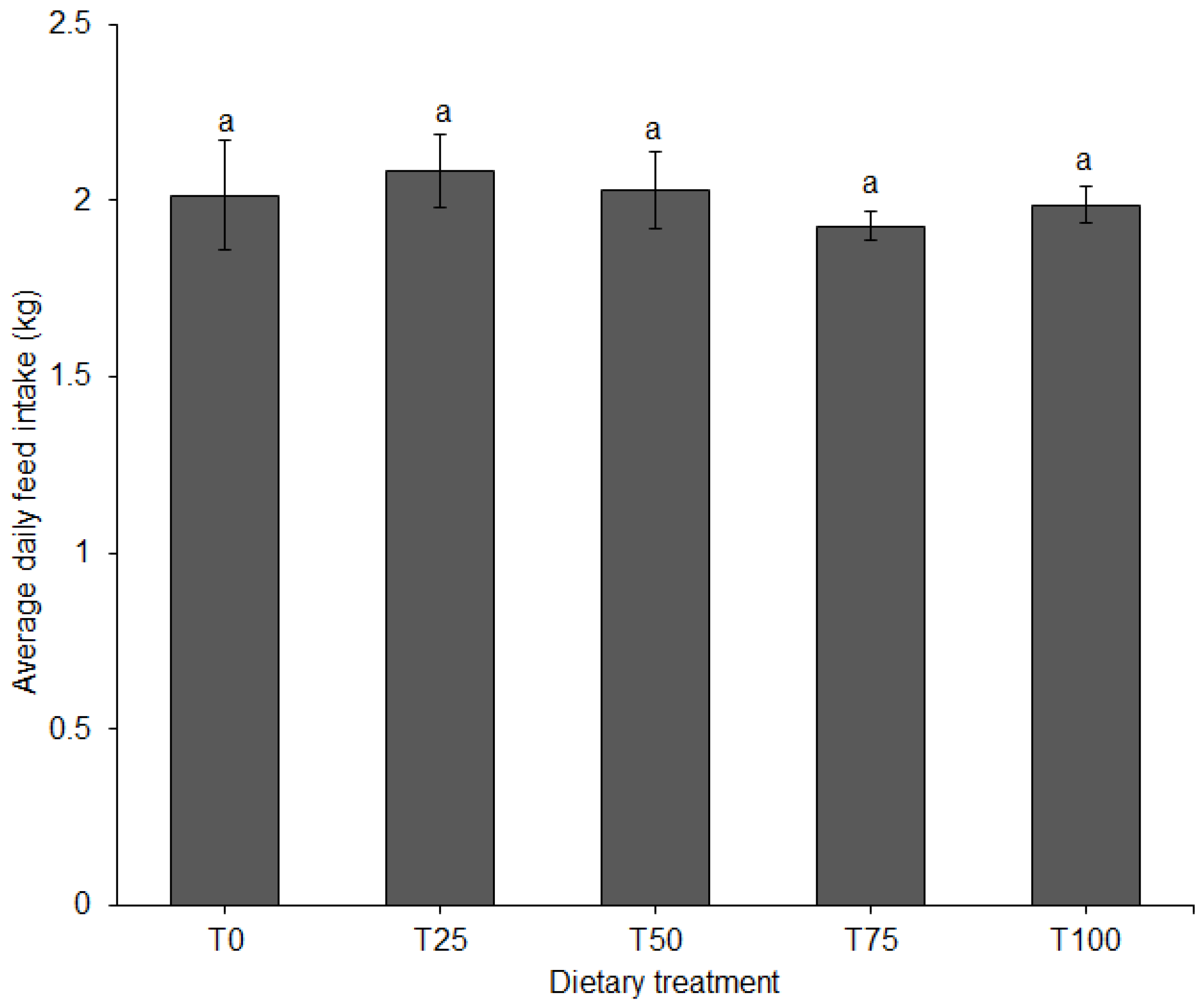
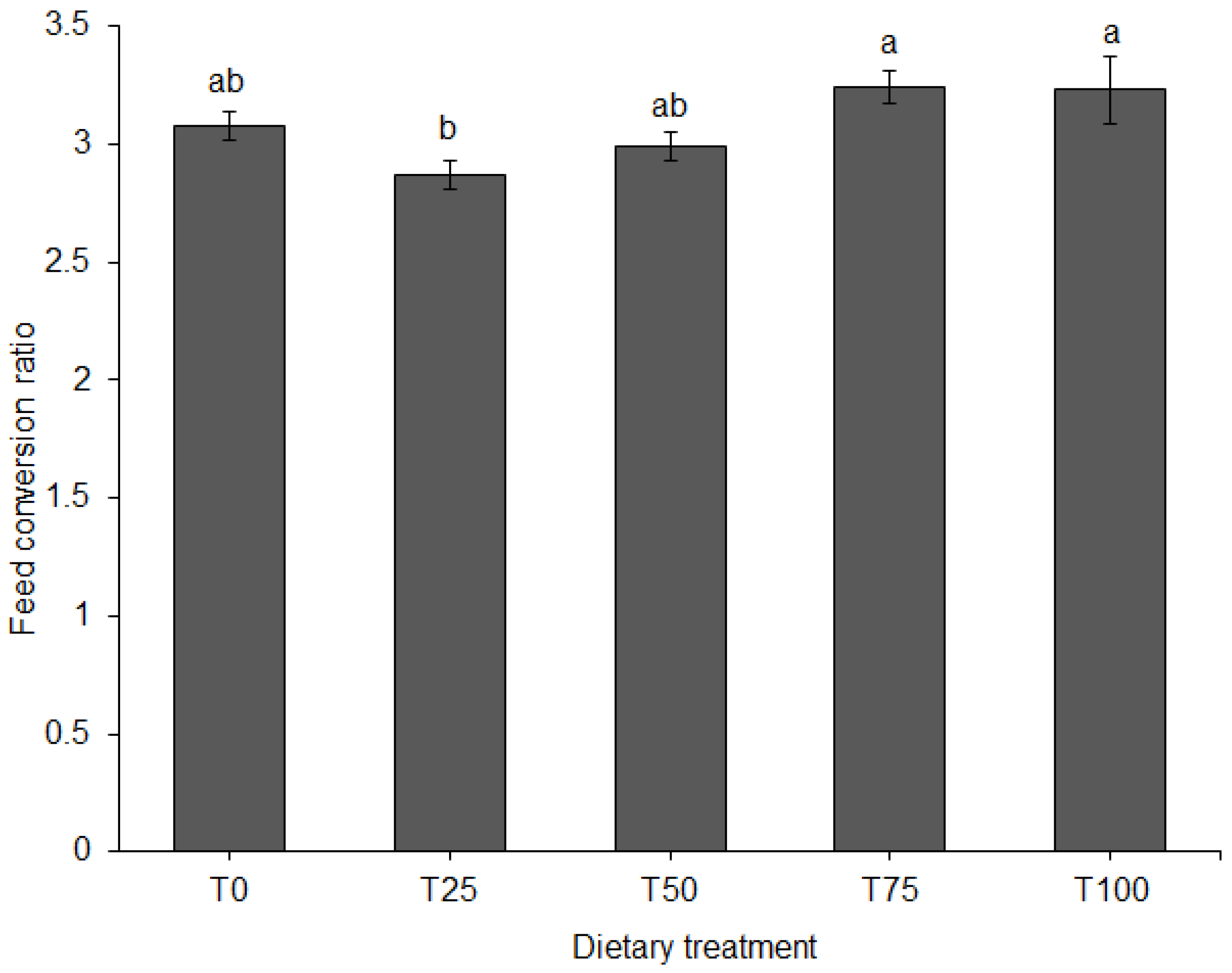
3.1. Growth Performance and Feed Conversion
3.2. Hematological and Serum Lipid Parameters
3.3. Economic Analyses of the Inclusion of Black Soldier Fly Larva Meal in Pig Diets
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Githigia, S.M.; Okuthe, S.; Diop, B. Pig Sector Kenya. FAO Animal Production and Health Livestock Country Reviews. No. 3. FAO, Rome. 2012. Available online: http://www.fao.org/3/a-i2566e.pdf (accessed on 25 March 2019).
- Serem, J.K.; Wahome, R.G.; Gakuya, D.W.; Kiama, S.G.; Gitao, G.C.; Onyango, D.W. Growth performance, feed conversion efficiency and blood characteristics of growing pigs fed on different levels of Moringa oleifera leaf meal. J. Vet. Med. Animal Health 2017, 9, 327–333. [Google Scholar] [CrossRef]
- Onsongo, V.O.; Osuga, I.M.; Gachuiri, C.K.; Wachira, A.M.; Miano, D.M.; Tanga, C.M.; Ekesi, S.; Nakimbugwe, D.; Fiaboe, K.K.M. Insects for income generation through animal feed: Effect of dietary replacement of soybean and fish meal with black soldier fly meal on broiler growth and economic performance. J. Econ. Entomol. 2018, 111, 1966–1973. [Google Scholar] [CrossRef] [PubMed]
- Etim, N.N.; Offiong, E.E.A.; Williams, M.E.; Asuquo, L.E. Influence of nutrition on blood parameters of pigs. Am. J. Biol. Life Sci. 2014, 2, 46–52. [Google Scholar]
- Etim, N.N. Hematological parameters and factors affecting their values. Agric. Sci. 2014, 2, 37–47. [Google Scholar] [CrossRef]
- Akpabio, U.; Etim, N.A.N.; Okpongete, R.O.; Offiong, E.E.A. Do diets affect hematological parameters of poultry? Br. J. Appl. Sci. Technol. 2014, 4, 1952–1965. [Google Scholar] [CrossRef]
- Tacon, A.G.; Metian, M. Global overview on the use of fish meal and fish oil in industrially compounded aquafeeds: Trends and future prospects. Aquaculture 2008, 285, 146–158. [Google Scholar] [CrossRef]
- Masuda, T.; Goldsmith, P.D. World soybean production: Area harvested, yield, and long-term projections. Int. Food Agribus. Man. 2009, 12, 143–162. [Google Scholar]
- Van Huis, A.; Van Itterbeeck, J.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects: Future Prospects for Food and Feed Security; FAO: Rome, Italy, 2013. [Google Scholar]
- Rumpold, B.A.; Schlüter, O.K. Nutritional composition and safety aspects of edible insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef]
- Bosch, G.; Zhang, S.; Oonincx, D.G.A.B.; Hendriks, W.H. Protein quality of insects as potential ingredients for dog and cat foods. J. Nutr. Sci. 2014, 3, e29. [Google Scholar] [CrossRef]
- Veldkamp, T.; Van Duinkerken, G.; Van Huis, A.; Lakemond, C.M.M.; Ottevanger, E.; Bosch, G.; Van Boekel, M.A.J.S. Insects as a Sustainable Feed Ingredient in Pig and Poultry Diets—A Feasibility Study; Report 638; Wageningen UR Livestock Research: Wageningen, The Netherlands, 2012; Available online: http://library.wur.nl/WebQuery/wurpubs/fulltext/234247 (accessed on 24 March 2019).
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed. Sci. Tech. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Sheppard, D.C.; Newton, G.L.; Thompson, S.A.; Savage, S. A value added manure management system using the black soldier fly. Bioresour. Technol. 1994, 50, 275–279. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Influence of resources on Hermetia illucens (Diptera: Stratiomyidae) larval development. J. Med. Entomol. 2013, 50, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Myers, H.M.; Tomberlin, J.K.; Lambert, B.D.; Kattes, D. Development of black soldier fly (Diptera: Stratiomyidae) larvae fed dairy manure. Environ. Entomol. 2008, 37, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Meneguz, M.; Schiavone, A.; Gai, F.; Dama, A.; Lussiana, C.; Renna, M.; Gasco, L. Effect of rearing substrate on growth performance, waste reduction efficiency and chemical composition of black soldier fly (Hermetia illucens) larvae. J. Sci. Food Agric. 2018, 98, 5776–5784. [Google Scholar] [CrossRef] [PubMed]
- Rehman, K.U.; Cai, M.; Xiao, X.; Zheng, L.; Wang, H.; Soomro, A.A.; Zhou, Y.; Li, W.; Yu, Z.; Zhang, J. Cellulose decomposition and larval biomass production from the co-digestion of dairy manure and chicken manure by mini-livestock (Hermetia illucens L.). J. Environ. Manag. 2017, 196, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zheng, L.; Cai, H.; Garza, E.; Yu, Z.; Zhou, S. From organic waste to biodiesel: Black soldier fly, Hermetia illucens, makes it feasible. Fuel 2011, 90, 1545–1548. [Google Scholar] [CrossRef]
- Salomone, R.; Saija, G.; Mondello, G.; Giannetto, A.; Fasulo, S.; Savastano, D. Environmental impact of food waste bioconversion by insects: Application of life cycle assessment to process using Hermetia illucens. J. Clean. Prod. 2017, 140, 890–905. [Google Scholar] [CrossRef]
- Chia, S.Y.; Tanga, C.M.; Osuga, I.M.; Mohamed, S.A.; Khamis, F.M.; Salifu, D.; Fiaboe, K.K.; Niassy, S.; van Loon, J.J.; Dicke, M.; et al. Effects of waste stream combinations from brewing industry on performance of black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). PeerJ 2018, 6, e5885. [Google Scholar] [CrossRef] [PubMed]
- Pinotti, L.; Giromini, C.; Ottoboni, M.; Tretola, M.; Marchis, D. Review: Insects and former foodstuffs for upgrading food waste biomasses/streams to feed ingredients for farm animals. Animal 2019, 13, 1365–1375. [Google Scholar] [CrossRef]
- Liu, Q.L.; Tomberlin, J.K.; Brady, J.A.; Sanford, M.R.; Yu, Z.N. Black soldier fly (Diptera: Stratiomyidae) larvae reduce Escherichia coli in dairy manure. Environ. Entomol. 2008, 37, 1525–1530. [Google Scholar] [CrossRef]
- Erickson, M.C.; Islam, M.; Sheppard, C.; Liao, J.; Doyle, M.P. Reduction of Escherichia coli O157:H7 and Salmonella enterica serovar Enteritidis in chicken manure by larvae of the black soldier fly. J. Food Prot. 2004, 67, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Barragan-Fonseca, K.B.; Dicke, M.; van Loon, J.J.A. Nutritional value of the black soldier fly (Hermetia illucens L.) and its suitability as animal feed—A review. J. Insects Food Feed 2017, 3, 105–120. [Google Scholar] [CrossRef]
- Newton, G.L.; Sheppard, D.C.; Watson, D.W.; Burtle, G.J.; Dove, C.R.; Tomberlin, J.K.; Thelen, E.E. The black soldier fly, Hermetia illucens, as a manure management/resource recovery tool. Symp. State Sci. Anim. Manure Waste Manag. 2005. Available online: https://pdfs.semanticscholar.org/5aa5/81bf66145ee15551c0a86df6436b9183fd05.pdf (accessed on 25 March 2019).
- Dierenfeld, E.; King, J. Digestibility and mineral availability of phoenix worms, Hermetia illucens, ingested by mountain chicken frogs, Leptodactylus fallax. J. Herpetol. Med. Surg. 2008, 18, 100–105. [Google Scholar] [CrossRef]
- Finke, M.D. Complete nutrient content of four species of feeder insects. Zoo Biol. 2013, 32, 27–36. [Google Scholar] [CrossRef]
- Dumas, A.; Raggi, T.; Barkhouse, J.; Lewis, E.; Weltzien, E. The oil fraction and partially defatted meal of black soldier fly larvae (Hermetia illucens) affect differently growth performance, feed efficiency, nutrient deposition, blood glucose and lipid digestibility of rainbow trout (Oncorhynchus mykiss). Aquaculture 2018, 492, 24–34. [Google Scholar] [CrossRef]
- Devic, E.; Leschen, W.; Murray, F.; Little, D.C. Growth performance, feed utilization and body composition of advanced nursing Nile tilapia (Oreochromis niloticus) fed diets containing black soldier fly (Hermetia illucens) larvae meal. Aquacult. Nutr. 2018, 24, 416–423. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Bruni, L.; Randazzo, B.; Vargas, A.; Gioacchini, G.; Truzzi, C.; Annibaldi, A.; Riolo, P.; Parisi, G.; Cardinaletti, G.; et al. Partial dietary inclusion of Hermetia illucens (black soldier fly) full-fat prepupae in Zebrafish feed: Biometric, histological, biochemical, and molecular implications. Zebrafish 2018, 15, 519–532. [Google Scholar] [CrossRef]
- Cummins, V.C.; Rawles, S.D.; Thompson, K.R.; Velasquez, A.; Kobayashi, Y.; Hager, J.; Webster, C.D. Evaluation of black soldier fly (Hermetia illucens) larvae meal as partial or total replacement of marine fish meal in practical diets for Pacific white shrimp (Litopenaeus vannamei). Aquaculture 2017, 473, 337–344. [Google Scholar] [CrossRef]
- Sealey, W.M.; Gaylord, T.G.; Barrows, F.T.; Tomberlin, J.K.; McGuire, M.A.; Ross, C.; St-Hilaire, S. Sensory analysis of rainbow trout, Oncorhynchus mykiss, fed enriched black soldier fly pre-pupae, Hermetia illucens. J. World Aquac. Soc. 2011, 42, 34–45. [Google Scholar] [CrossRef]
- Li, S.; Ji, H.; Zhang, B.; Tian, J.; Zhou, J.; Yu, H. Influence of black soldier fly (Hermetia illucens) larvae oil on growth performance, body composition, tissue fatty acid composition and lipid deposition in juvenile Jian carp (Cyprinus carpio var. Jian). Aquaculture 2016, 465, 43–52. [Google Scholar] [CrossRef]
- Magalhães, R.; Sánchez-López, A.; Leal, R.S.; Martínez-Llorens, S.; Oliva-Teles, A.; Peres, H. Black soldier fly (Hermetia illucens) pre-pupae meal as a fish meal replacement in diets for European seabass (Dicentrarchus labrax). Aquaculture 2017, 476, 79–85. [Google Scholar] [CrossRef]
- Kroeckel, S.; Harjes, A.G.E.; Roth, I.; Katz, H.; Wuertz, S.; Susenbeth, A.; Schulz, C. When a turbot catches a fly: Evaluation of a pre-pupae meal of the black soldier fly (Hermetia illucens) as fish meal substitute—Growth performance and chitin degradation in juvenile turbot (Psetta maxima). Aquaculture 2012, 364–365, 345–352. [Google Scholar] [CrossRef]
- Schiavone, A.; Cullere, M.; De Marco, M.; Meneguz, M.; Biasato, I.; Bergagna, S.; Dezzutto, D.; Gai, F.; Dabbou, S.; Gasco, L.; et al. Partial or total replacement of soybean oil by black soldier fly larvae (Hermetia illucens L.) fat in broiler diets: Effect on growth performances, feed-choice, blood traits, carcass characteristics and meat quality. Ital. J. Anim. Sci. 2017, 16, 93–100. [Google Scholar] [CrossRef]
- Maurer, V.; Holinger, M.; Amsler, Z.; Früh, B.; Wohlfahrt, J.; Stamer, A.; Leiber, F. Replacement of soybean cake by Hermetia illucens meal in diets for layers. J. Insects Food Feed 2015, 2, 83–90. [Google Scholar] [CrossRef]
- Newton, G.L.; Booram, C.V.; Barker, R.W.; Hale, O.M. Dried Hermetia illucens larvae meal as a supplement for Swine. J. Anim. Sci. 1977, 44, 395–400. [Google Scholar] [CrossRef]
- Driemeyer, H. Evaluation of Black Soldier Fly (Hermetia illucens) Larvae as an Alternative Protein Source in Pig Creep Diets in Relation to Production, Blood and Manure Microbiology Parameters. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, December 2016. [Google Scholar]
- Spranghers, T.; Michiels, J.; Vrancx, J.; Ovyn, A.; Eeckhout, M.; De Clercq, P.; De Smet, S. Gut antimicrobial effects and nutritional value of black soldier fly (Hermetia illucens L.) pre-pupae for weaned piglets. Anim. Feed Sci. Technol. 2018, 235, 33–42. [Google Scholar] [CrossRef]
- Biasato, I.; Renna, M.; Gai, F.; Dabbou, S.; Meneguz, M.; Perona, G.; Martinez, S.; Lajusticia, A.C.; Bergagna, S.; Sardi, L.; et al. Partially defatted black soldier fly larva meal inclusion in piglet diets: Effects on the growth performance, nutrient digestibility, blood profile, gut morphology and histological features. J. Anim. Sci. Biotechnol. 2019, 10, 12. [Google Scholar] [CrossRef]
- Wilson, G.D.A.; Harvey, D.G.; Snook, C.R. A review of factors affecting blood biochemistry in the pig. Br. Vet. J. 1972, 128, 596–610. [Google Scholar] [CrossRef]
- Ameen, S.A.; Adedeji, O.S.; Akingbade, A.A.; Olayemi, T.B.; Oyedapo, L.O.; Aderinola, A. The effect of different feeding regimes on hematological parameters and immune status of commercial broilers in derived Savannah Zone of Nigeria. In Proceedings of the 32nd Annual Conference Nigerian Society Animal Production (NSAP), Calabar, Nigeria, 18–21 March 2007; pp. 176–178. [Google Scholar]
- Shanmugam, A.A.; Muliya, S.K.; Deshmukh, A.; Suresh, S.; Nath, A.; Kalaignan, P.; Venkatramappa, M.; Jose, L. Baseline hematology and serum biochemistry results for Indian leopards (Panthera pardus fusca). Vet. World 2017, 10, 818–824. [Google Scholar] [CrossRef]
- Spring, P. The challenge of cost effective poultry and animal nutrition: Optimizing existing and applying novel concepts. Lohmann Inf. 2013, 48, 38–46. [Google Scholar]
- Federation of Animal Science Societies (FASS). Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching, 3rd ed.; FASS: Champaign, IL, USA, 2010. [Google Scholar]
- National Research Council. Nutrient Requirements of Swine: Eighth Revised Edition; National Academies Press: Washington, DC, USA, 1979. [Google Scholar] [CrossRef][Green Version]
- Núñez-Sánchez, N.; Marín, A.L.; Hernández, M.P.; Carrion, D.; Castro, G.G.; Alba, L.M. Faecal near infrared spectroscopy (NIRS) as a tool to assess rabbit’s feed digestibility. Livest. Sci. 2012, 150, 386–390. [Google Scholar] [CrossRef]
- Rosales, A.; Galicia, L.; Oviedo, E.; Islas, C.; Palacios-Rojas, N. Near-infrared reflectance spectroscopy (NIRS) for protein, tryptophan, and lysine evaluation in quality protein maize (QPM) breeding programs. J. Agric. Food Chem. 2011, 59, 10781–10786. [Google Scholar] [CrossRef] [PubMed]
- Powell, C.D.; Chowdhury, M.K.; Bureau, D.P. Assessing the bioavailability of L-methionine and a methionine hydroxy analogue (MHA-Ca) compared to DL-methionine in rainbow trout (Oncorhynchus mykiss). Aquac. Res. 2017, 48, 332–346. [Google Scholar] [CrossRef]
- Windham, W.R. AOAC official method 994.12, amino acids in feeds, alternative III, acid hydrolysis method. In Official methods of analysis of AOAC Intemational, 16th ed.; Cunniff, P., Ed.; AOAC International: Rockville, MD, USA, 1995; Volume 1. [Google Scholar]
- Llames, C.R.; Fontaine, J. Determination of amino acids in feeds: Collaborative study. J. Assoc. Off. Anal. Chem. 1994, 77, 1362–1402. [Google Scholar]
- Zampiga, M.; Laghi, L.; Petracci, M.; Zhu, C.; Meluzzi, A.; Dridi, S.; Sirri, F. Effect of dietary arginine to lysine ratios on productive performance, meat quality, plasma and muscle metabolomics profile in fast-growing broiler chickens. J. Anim. Sci. Biotechnol. 2018, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Al-Sagan, A.A.; Khalil, S.; Smith, M.P. The benefit of L-threonine supplementation on growth performance, carcass characteristics, intestinal morphology and litter quality of broilers. Braz. J. Poult. Sci. 2018, 20, 753–758. [Google Scholar] [CrossRef]
- Slump, P.; Bos, K.D. Determination of methionine in feed concentrates. Poult. Sci. 1985, 64, 705–707. [Google Scholar] [CrossRef]
- Płotka-Wasylka, J.; Frankowski, M.; Simeonov, V.; Polkowska, Ż.; Namieśnik, J. Determination of metals content in wine samples by inductively coupled plasma-mass spectrometry. Molecules 2018, 23, 2886. [Google Scholar] [CrossRef]
- Sreenivasulu, V.; Kumar, N.S.; Dharmendra, V.; Asif, M.; Balaram, V.; Zhengxu, H.; Zhen, Z. Determination of boron, phosphorus, and molybdenum content in biosludge samples by microwave plasma atomic emission spectrometry (MP-AES). Appl. Sci. 2017, 7, 264. [Google Scholar] [CrossRef]
- Santos, É.J.; Baika, L.M.; Herrmann, A.B.; Kulik, S.; Sato, C.S.; Santos, A.B.; Curtius, A.J. Fast assessment of mineral constituents in grass by inductively coupled plasma optical emission spectrometry. Braz. Arch. Biol. Technol. 2012, 55, 457–464. [Google Scholar] [CrossRef]
- Barałkiewicz, D.; Kanecka-Hanc, A.; Gramowska, H. ICP slurry introduction for simple and rapid determination of Pb, Mg and Ca in plant roots. Cent. Eur. J. Chem. 2007, 5, 1148–1157. [Google Scholar] [CrossRef]
- Aok, Y.M.D. Effect of Feed Enzymes and Energy Level on Broiler Chicken (Gallus domesticus) Performance in Kenya. Master’s Thesis, University of Nairobi, Nairobi, Kenya, July 2012. [Google Scholar]
- Wallace, P.A.; Nyameasem, J.K.; Adu-Aboagye, G.A.; Affedzie-Obresi, S.; Nkegbe, E.K.; Karbo, N.; Murray, F.; Leschen, W.; Maquart, P. Impact of black soldier fly larval meal on growth performance, apparent digestibility, hematological and blood chemistry indices of guinea fowl starter keets under tropical conditions. Trop. Anim. Health Prod. 2017, 49, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Lalander, C.; Diener, S.; Zurbrügg, C.; Vinnerås, B. Effects of feedstock on larval development and process efficiency in waste treatment with black soldier fly (Hermetia illucens). J. Clean. Prod. 2019, 208, 211–219. [Google Scholar] [CrossRef]
- Pieterse, E.; Erasmus, S.W.; Uushona, T.; Hoffman, L.C. Black soldier fly (Hermetia illucens) pre-pupae meal as a dietary protein source for broiler production ensures a tasty chicken with standard meat quality for every pot. J. Sci. Food Agric. 2018, 99, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Elorduy, J.; González, E.A.; Hernández, A.R.; Pino, J.M. Use of Tenebrio molitor (Coleoptera: Tenebrionidae) to recycle organic wastes and as feed for broiler chickens. J. Econ. Entomol. 2002, 95, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Xie, C.; Long, C.; Li, J.; Zhou, X.; Fan, Z.; Blachier, F.; Yin, Y. Effects of a daily three-meal pattern with different dietary protein contents on pig growth performance, carcass and muscle quality traits. J. Sci. Food Agric. 2018, 98, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Medhi, D. Effects of enzyme supplemented diet on finishing crossbred pigs at different levels of silk worm pupae meal in diet. Indian J. Field Vet. 2011, 7, 24–26. [Google Scholar]
- Medhi, D.; Math, N.C.; Sharma, D.N. Effect of silk worm pupae meal and enzyme supplementation on blood constituents in pigs. Indian Vet. J. 2009, 86, 433–434. [Google Scholar]
- Medhi, D.; Nath, N.C.; Gohain, A.K.; Bhuyan, R. Effect of silk worm pupae meal on carcass characteristics and composition of meat in pigs. Indian Vet. J. 2009, 86, 816–818. [Google Scholar]
- Newton, L.A.; Sheppard, C.R.; Watson, D.W.; Burtle, G.A.; Dove, R.O. Using the Black Soldier Fly, Hermetia illucens, as a Value-Added Tool for the Management of Swine Manure; Animal and Poultry Waste Management Center, North Carolina State University: Raleigh, NC, USA, 2005. [Google Scholar]
- Coll, J.F.; Crespi, M.P.; Itagiba, M.G.; Souza, J.C.; Gomes, A.V.; Donatti, F.C. Utilization of silkworm pupae meal (Bombyx mori L.) as a source of protein in the diet of growing-finishing pigs. Rev. Bras. Zootec. 1992, 21, 378–383. [Google Scholar]
- Adeniji, A.A. The feeding value of rumen content-maggot meal mixture in the diets of early weaned piglets. Asian J. Anim. Vet. Adv. 2008, 3, 115–119. [Google Scholar] [CrossRef][Green Version]
- Cullere, M.; Tasoniero, G.; Giaccone, V.; Miotti-Scapin, R.; Claeys, E.; De Smet, S.; Zotte, A.D. Black soldier fly as dietary protein source for broiler quails: Apparent digestibility, excreta microbial load, feed choice, performance, carcass and meat traits. Animal 2016, 10, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, Q. The Evaluation of Larvae of Musca domestica (Common House Fly) as Protein Source for Broiler Production. Master’s Thesis, Stellenbosch University, Matieland, South Africa, March 2011. [Google Scholar]
- Alegbeleye, W.O.; Obasa, S.O.; Olude, O.O.; Otubu, K.; Jimoh, W. Preliminary evaluation of the nutritive value of the variegated grasshopper (Zonocerus variegatus L.) for African catfish Clarias gariepinus (Burchell. 1822) fingerlings. Aquacult. Res. 2012, 43, 412–420. [Google Scholar] [CrossRef]
- Klaphake, E. A fresh look at metabolic bone diseases in reptiles and amphibians. Vet. Clin. North Am. Exot. Anim. Pract. 2010, 13, 375–392. [Google Scholar] [CrossRef]
- Marono, S.; Loponte, R.; Lombardi, P.; Vassalotti, G.; Pero, M.E.; Russo, F.; Gasco, L.; Parisi, G.; Piccolo, G.; Nizza, S.; et al. Productive performance and blood profiles of laying hens fed Hermetia illucens larvae meal as total replacement of soybean meal from 24 to 45 weeks of age. Poult. Sci. 2017, 96, 1783–1790. [Google Scholar] [CrossRef] [PubMed]
- Loponte, R.; Nizza, S.; Bovera, F.; De Riu, N.; Fliegerova, K.; Lombardi, P.; Vassalotti, G.; Mastellone, V.; Nizza, A.; Moniello, G. Growth performance, blood profiles and carcass traits of Barbary partridge (Alectoris barbara) fed two different insect larvae meals (Tenebrio molitor and Hermetia illucens). Res. Vet. Sci. 2017, 115, 183–188. [Google Scholar] [CrossRef]
- Olugbemi, T.S.; Mutayoba, S.K.; Lekule, F.P. Effect of Moringa (Moringa oleifera) inclusion in cassava based diets fed to broiler chickens. Int. J. Poul. Sci. 2010, 9, 363–367. [Google Scholar] [CrossRef]
- Martin, J.F.; Trowbridge, E.A.; Salmon, G.; Plumb, J. The biological significance of platelet volume: Its relationship to bleeding time, platelet thromboxane B2 production and megakaryocyte nuclear DNA concentration. Thromb. Res. 1983, 32, 443–460. [Google Scholar] [CrossRef]
- American Association for Clinical Chemistry. White Blood Cell (WBC) Differential. 2019. Available online: https://labtestsonline.org/tests/white-blood-cell-wbc-differential (accessed on 11 July 2019).
- Konlan, S.P.; Karikari, P.K.; Ansah, T. Productive and blood indices of dwarf rams fed a mixture of rice straw and groundnut haulms alone or supplemented with concentrates containing different levels of shea nut cake. Pak. J. Nutr. 2012, 11, 566–571. [Google Scholar]
- Nathan, C. Neutrophils and immunity: Challenges and opportunities. Nat. Rev. Immunol. 2006, 6, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Skrivanova, E.; Marounek, M.; Benda, V.; Brezina, P. Susceptibility of Escherichia coli, Salmonella sp. and Clostridium perfringens to organic acids and monolaurin. Vet. Med. 2006, 51, 81–88. [Google Scholar] [CrossRef]
- Ahlante, B.O.; Afiukwa, J.N.; Ajay, O.I. The dietary effects of coconut oil on the leucocytes and neutrophils count in rabbits. J. Ecophysiol. Occup. Health 2010, 10, 143–148. [Google Scholar]
- Dudek, K.; Sliwa, E.; Tatara, M.R. Changes in blood leukocyte pattern in piglets from sows treated with garlic preparations. Bull. Vet. Inst. Pulawy 2006, 50, 263–267. [Google Scholar]
- David, R.; Ngulube, P.; Dube, A. A cost–benefit analysis of document management strategies used at a financial institution in Zimbabwe: A case study. J. Inf. Manag. 2013, 15, 1–10. [Google Scholar] [CrossRef]
- Wang, Y.S.; Shelomi, M. Review of black soldier fly (Hermetia illucens) as animal feed and human food. Foods 2017, 6, 91. [Google Scholar] [CrossRef] [PubMed]
| Parameter | BSFLM | Dietary Treatment | ||||
|---|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | ||
| Ingredient (kg) | ||||||
| Maize meal | - | 12.0 | 14.0 | 14.0 | 14.0 | 15.0 |
| Wheat pollard | - | 52.0 | 35.0 | 33.0 | 35.2 | 34.2 |
| Rice polishing | - | 22.0 | 30.5 | 32.0 | 29.8 | 28.3 |
| Fishmeal | - | 10.0 | 7.5 | 5.0 | 2.5 | - |
| BSFLM | - | - | 9.0 | 12 | 14.5 | 18.5 |
| Lysine | - | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Methionine | - | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Salt | - | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Bone meal | - | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Limestone | - | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Vitamin and mineral premix a | - | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Dry matter (DM; %) | 94.9 | 92.6 | 94.0 | 92.4 | 93.2 | 94.0 |
| Crude protein (% DM) | 46.6 | 15.4 | 15.3 | 15.0 | 15.7 | 14.8 |
| Essential amino acids (% DM) | ||||||
| Lysine | 2.9 | 1.0 | 0.8 | 0.9 | 1.0 | 0.9 |
| Methionine | 0.8 | 0.5 | 0.4 | 0.5 | 0.5 | 0.5 |
| Methionine + Cystine | 1.1 | 0.7 | 0.6 | 0.8 | 0.7 | 0.7 |
| Isoleucine | 1.8 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Leucine | 2.9 | 1.0 | 1.1 | 0.9 | 1.0 | 0.9 |
| Threonine | 1.7 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Phenylalanine | 2.2 | 0.6 | 0.6 | 0.5 | 0.6 | 0.6 |
| Valine | 2.6 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 |
| Arginine | 2.2 | 0.9 | 0.9 | 0.8 | 0.8 | 0.8 |
| Histidine | 1.3 | 0.3 | 0.4 | 0.3 | 0.3 | 0.3 |
| Nonessential amino acids (% DM) | ||||||
| Alanine | 3.0 | 0.8 | 0.9 | 0.8 | 0.9 | 0.8 |
| Aspartic acid | 3.9 | 1.1 | 1.2 | 1.1 | 1.1 | 1.0 |
| Cystine | 0.4 | 0.3 | 0.3 | 0.2 | 0.2 | 0.2 |
| Glutamic acid | 4.9 | 2.3 | 2.3 | 2.0 | 2.1 | 2.0 |
| Glycine | 2.5 | 0.8 | 0.8 | 0.7 | 0.8 | 0.7 |
| Proline | 2.4 | 0.8 | 0.9 | 0.8 | 0.8 | 0.8 |
| Serine | 1.8 | 0.6 | 0.6 | 0.6 | 0.6 | 0.6 |
| Parameter | Dietary Treatment | ||||
|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | |
| Boron (ppm) | 4.2 | 2.8 | 2.3 | 2.7 | 3.1 |
| Molybdenum (ppm) | 1.3 | 1.1 | 0.5 | 1.0 | 1.2 |
| Iron (ppm) | 750.5 | 731.0 | 632.6 | 518.4 | 529.2 |
| Copper (ppm) | 22.6 | 501.1 | 80.5 | 16.7 | 22.8 |
| Zinc (ppm) | 121.4 | 110.6 | 114.1 | 102.3 | 112.6 |
| Cobalt (ppm) | 3.4 | 0.5 | 0.4 | 0.5 | 0.4 |
| Manganese (ppm) | 295.4 | 257.0 | 264.1 | 243.0 | 222.9 |
| Sodium (ppm) | 2439.8 | 1648.6 | 1097.8 | 1138.8 | 998.9 |
| Sulphur (%) | 0.4 | 0.6 | 0.3 | 0.3 | 0.3 |
| Magnesium (%) | 0.8 | 0.7 | 0.7 | 0.6 | 0.6 |
| Potassium (%) | 0.9 | 0.9 | 0.9 | 0.9 | 0.9 |
| Phosphorus (%) | 0.9 | 0.7 | 0.8 | 0.8 | 0.7 |
| Calcium (%) | 2.5 | 2.8 | 2.1 | 1.7 | 1.8 |
| Sugar (%) | 4.5 | 5.1 | 6.2 | 6.5 | 8.7 |
| Starch (%) | 29.2 | 25.3 | 27.6 | 20.7 | 26.9 |
| Ash (%) | 10.5 | 10.6 | 10.1 | 10.1 | 8.9 |
| Acid detergent fiber (%) | 15.3 | 19.0 | 17.0 | 20.7 | 15.4 |
| Neutral detergent fiber (%) | 37.7 | 42.8 | 41.6 | 47.0 | 39.7 |
| Digestibility (NCGD) (%) | 79.3 | 73.5 | 75.0 | 70.0 | 76.9 |
| Oil (%) | 5.0 | 8.1 | 8.9 | 9.8 | 12.2 |
| Net energy (MJ/kg) | 9.5 | 9.8 | 10.5 | 9.9 | 12.0 |
| Parameter | Sex | Dietary Treatment | p Value (GLM) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | Diet | Sex | Diet × Sex | ||
| Initial weight, kg | F | 19.5 ± 1.73 | 16.8 ± 0.63 | 17.1 ± 0.66 | 17.0 ± 0.87 | 18.5 ± 0.71 | |||
| M | 18.8 ± 1.56 | 17.1 ± 0.85 | 18.5 ± 0.89 | 18.5 ± 0.79 | 20.8 ± 0.83 | ||||
| Overall | 19.1 ± 1.09 | 17.0 ± 0.49 | 17.8 ± 0.57 | 17.8 ± 0.61 | 19.6 ± 0.66 | 0.080 | 0.152 | 0.628 | |
| Final weight, kg | F | 56.3 ± 4.3 | 50.8 ± 2.65 | 56.3 ± 1.45 | 47.8 ± 1.76 | 50.0 ± 1.43 | |||
| M | 53.8 ± 3.56 | 53.5 ± 3.68 | 53.9 ± 4.90 | 53.0 ± 1.97 | 56.9 ± 0.99 | ||||
| Overall | 55.0 ± 2.63 | 52.1 ± 2.16 | 55.1 ± 2.41 | 50.4 ± 1.58 | 53.4 ± 1.53 | 0.474 | 0.295 | 0.395 | |
| Average daily gain, kg | F | 0.61 ± 0.04 | 0.56 ± 0.04 | 0.62 ± 0.04 | 0.50 ± 0.03 | 0.54 ± 0.03 | |||
| M | 0.59 ± 0.04 | 0.57 ± 0.04 | 0.62 ± 0.04 | 0.57 ± 0.04 | 0.59 ± 0.03 | ||||
| Overall | 0.60 ± 0.03 | 0.57 ± 0.02 | 0.62 ± 0.02 | 0.53 ± 0.02 | 0.57 ± 0.02 | 0.126 | 0.408 | 0.641 | |
| Parameter | Dietary Treatment | p Value (GLM) | Normal Range | ||||
|---|---|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | |||
| RBC (×1012 /L) | 5.4 ± 2.3 | 7.0 ± 0.2 | 7.4 ± 0.1 | 6.8 ± 0.6 | 6.9 ± 0.1 | 0.725 | 5.0–8.00 |
| Hb (g/dL) | 10.2 ± 4.3 | 13.3 ± 0.2 | 13.8 ± 0.4 | 12.8 ± 1.0 | 13.5 ± 0.3 | 0.719 | 10.7–16.7 |
| Hct (%) | 35.5 ± 15 | 48.0 ± 1.0 | 47.6 ± 1.7 | 45.2 ± 3.2 | 47.0 ± 2.1 | 0.704 | 32.0–50.0 |
| MCV (fl) | 64.4 ± 1.7 | 69.1 ± 3.0 | 64.4 ± 3.2 | 66.5 ± 1.8 | 68.1 ± 2.4 | 0.593 | 50.0–68.0 |
| MCH (pg) | 19.3 ± 0.5 | 19.1 ± 0.8 | 18.6 ± 0.8 | 18.8 ± 0.4 | 19.6 ± 0.3 | 0.803 | 17.0–21.0 |
| MCHC (g/dL) | 30.0 ± 1.5 | 27.8 ± 0.1 | 28.9 ± 0.2 | 28.3 ± 0.8 | 28.8 ± 0.7 | 0.449 | 30.0–34.0 |
| RDW (%) | 20.5 ± 1.7 | 21.8 ± 0.2 | 22.0 ± 0.5 | 21.6 ± 1.0 | 22.2 ± 0.4 | 0.706 | 15.0–27.0 |
| Platelet (K/uL) | 382 ± 7.0 1 a | 209 ± 49 c | 328 ± 33 ab | 229 ± 27 bc | 251 ± 28 bc | 0.042 | 300–700 |
| Parameter | Dietary Treatment | p Value (GLM) | Normal Range | ||||
|---|---|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | |||
| WBC (k/L) | 17.3 ± 1.6 | 24.5 ± 2.7 | 20.6 ± 2.5 | 24.0 ± 3.6 | 24.8 ± 5.0 | 0.463 | 11.0–22.0 |
| Differential count (%) | |||||||
| Neutrophils | 24.2 ± 1.0 c | 24.7 ± 0.4 c | 27.1 ± 1.1 bc | 29.9 ± 2.9 ab | 31.8 ± 0.4 a | 0.019 | 28.0–51.0 |
| Lymphocytes | 69.4 ± 1.4 | 66.8 ± 0.8 | 64.8 ± 1.2 | 62.4 ± 4.0 | 60.9 ± 1.2 | 0.092 | 39.0–62.0 |
| Monocytes | 3.5 ± 0.7 | 5.6 ± 0.5 | 4.8 ± 0.9 | 4.1 ± 0.8 | 4.3 ± 1.2 | 0.496 | 2.00–10.0 |
| Eosinophils | 2.8 ± 0.5 | 3.0 ± 0.1 | 3.3 ± 0.3 | 3.5 ± 0.3 | 2.9 ± 0.5 | 0.684 | 0.50–11.0 |
| Basophils | 0.13 ± 0.03 | 0.08 ± 0.01 | 0.07 ± 0.03 | 0.15 ± 0.03 | 0.10 ± 0.0 | 0.585 | 0.00–2.00 |
| Blood serum indices (mmol/L) | |||||||
| Total Chol | 1.90 ± 0.16 | 2.11 ± 0.13 | 2.11 ± 0.20 | 2.50 ± 0.12 | 2.19 ± 0.12 | 0.185 | 1.68–5.81 |
| Total Trig | 0.69 ± 0.08 | 1.03 ± 0.23 | 0.99 ± 0.17 | 0.89 ± 0.03 | 0.89 ± 0.07 | 0.479 | 0.11–1.13 |
| LDL | 0.67 ± 0.11 | 0.88 ± 0.08 | 0.75 ± 0.12 | 0.71 ± 0.11 | 0.65 ± 0.04 | 0.50 | <3.00 |
| HDL | 1.23 ± 0.09 | 1.23 ± 0.07 | 1.37 ± 0.09 | 1.77 ± 0.23 | 1.53 ± 0.09 | 0.066 | >1.00 |
| Parameter | Dietary Treatment | p Value (GLM) | ||||
|---|---|---|---|---|---|---|
| T0 | T25 | T50 | T75 | T100 | ||
| Cost feed (US $/kg) | 0.5 | 0.52 | 0.52 | 0.51 | 0.51 | - |
| Cost of protein ingredient in feed (%) | 0.24 | 0.31 | 0.31 | 0.3 | 0.3 | - |
| Total feed consumed (kg/pig) | 126.6 | 128.5 | 127.9 | 121.6 | 126 | - |
| Cost of feed consumed per pig (US$), Cp | 62.89 | 67.29 | 66.14 | 61.97 | 64.43 | - |
| Final body weight of pig (kg) | 55.0 ± 2.63 | 52.1 ± 2.16 | 55.1 ± 2.41 | 50.4 ± 1.58 | 53.4 ± 1.53 | 0.474 |
| Amount at final weight (US$/live weight), Sp | 165.0 ± 7.89 | 156.4 ± 6.49 | 165.2 ± 7.22 | 151.7 ± 4.42 | 160.1 ± 4.58 | 0.477 |
| Profit, Pr | 102.1 ± 7.89 | 89.1 ± 6.49 | 99.0 ± 7.22 | 89.7 ± 4.42 | 95.9 ± 4.58 | 0.499 |
| Cost–benefit ratio, CBR | 2.6 ± 0.13 | 2.3 ± 0.10 | 2.5 ± 0.11 | 2.5 ± 0.07 | 2.5 ± 0.07 | 0.318 |
| Return on investment, RoI | 162.4 ± 13 | 132.4 ± 9.64 | 149.7 ± 11 | 144.8 ± 7.13 | 148.8 ± 7.11 | 0.318 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chia, S.Y.; Tanga, C.M.; Osuga, I.M.; Alaru, A.O.; Mwangi, D.M.; Githinji, M.; Subramanian, S.; Fiaboe, K.K.M.; Ekesi, S.; van Loon, J.J.A.; et al. Effect of Dietary Replacement of Fishmeal by Insect Meal on Growth Performance, Blood Profiles and Economics of Growing Pigs in Kenya. Animals 2019, 9, 705. https://doi.org/10.3390/ani9100705
Chia SY, Tanga CM, Osuga IM, Alaru AO, Mwangi DM, Githinji M, Subramanian S, Fiaboe KKM, Ekesi S, van Loon JJA, et al. Effect of Dietary Replacement of Fishmeal by Insect Meal on Growth Performance, Blood Profiles and Economics of Growing Pigs in Kenya. Animals. 2019; 9(10):705. https://doi.org/10.3390/ani9100705
Chicago/Turabian StyleChia, Shaphan Y., Chrysantus M. Tanga, Isaac M. Osuga, Alphonce O. Alaru, David M. Mwangi, Macdonald Githinji, Sevgan Subramanian, Komi K. M. Fiaboe, Sunday Ekesi, Joop J. A. van Loon, and et al. 2019. "Effect of Dietary Replacement of Fishmeal by Insect Meal on Growth Performance, Blood Profiles and Economics of Growing Pigs in Kenya" Animals 9, no. 10: 705. https://doi.org/10.3390/ani9100705
APA StyleChia, S. Y., Tanga, C. M., Osuga, I. M., Alaru, A. O., Mwangi, D. M., Githinji, M., Subramanian, S., Fiaboe, K. K. M., Ekesi, S., van Loon, J. J. A., & Dicke, M. (2019). Effect of Dietary Replacement of Fishmeal by Insect Meal on Growth Performance, Blood Profiles and Economics of Growing Pigs in Kenya. Animals, 9(10), 705. https://doi.org/10.3390/ani9100705





