1. Introduction
Despite the existence of some fish and amphibian species that do not show any apparent behavioural signs of sleep [
1], it is widely accepted that sleep is an evolutionarily adaptive phenomenon, which has crucial importance for daily normal functioning. The importance of sleep is not only salient from the consequences of sleep deprivation (including hypertension, obesity or diabetes [
2]), but also from its link with cognitive (e.g., memory consolidation [
3,
4]) and affective [
5] functioning. The present study builds on results from this latter direction of research and explores in family dogs heart rate and heart rate variability measures during sleep preceded by positively versus negatively valenced social interactions.
Sleep can be divided into two distinct stages: rapid eye movement (REM) and non-rapid eye movement (non-REM) sleep. Non-REM sleep is also often called slow wave sleep, due to the apparent low frequency, high amplitude electroencephalogram (EEG) activity during the “deep sleep” part of this stage. REM sleep is also known as paradoxical sleep due to characteristic high frequency, low amplitude EEG activity paired with muscle atonia. Beside EEG, the activity of the autonomic nervous system (ANS) as indicated by heart rate (HR) and heart rate variability (HRV) are frequently used as physiological correlates of the level of activation along the sleep-wake axis [
6]. During a sleep cycle non-REM and REM sleep are modulated differently by the ANS. After sleep onset parasympathetic activity increases, resulting in a lower HR and higher HRV [
7]. This parasympathetic dominance prevails during the non-REM phase (with sympathetic activity reduced), while the REM phase is characterised by fluctuations in parasympathetic and sympathetic control [
8].
Heart rate has been used as an indicator of the physiological state in both animals and humans [
9], and it is generally assumed that increased levels of stress are reflected in increased HR. Due to the autonomic innervations of the heart, HRV can be a good physiological index of emotion regulation, since the central autonomic network (CAN) uses brain areas that are also involved in emotion regulation, like the anterior cingulum, ventral medial prefrontal cortex or the central nucleus of the amygdala [
10,
11]. In addition, CAN provides the parasympathetic fibres to the heart, therefore HRV can be a good indirect measure of physiological capacity for emotion regulation [
12].
Emotional experiences during the day can alter sleep quality and macrostructure in many ways [
13]. For example REM sleep has been reported to be enhanced [
14], shortened [
15] or unchanged due to different stressors. More fragmented sleep, longer sleep latency, shorter sleep duration also frequently occurs following stressful events [
16]. Insufficient and/or disturbed sleep (sleep deprivation), in turn, alters mood and emotion regulation [
17,
18,
19]. It appears that sleep deprivation diminishes the top–down control of the prefrontal cortex over the amygdala (the main structure in the processing of emotion-related information, especially aversive stimuli [
20]), and activation of the amygdala increases in response to negative pictures [
17].
While both the relationship between (awake) HR and emotions, as well as sleep and emotions are relatively well-studied separately, little is known about how HR and HRV change during sleep following an emotionally loaded event. The current paper will investigate this question in family dogs (
Canis familiaris), the most common pet animal around the world, that experience natural situations inducing both positive and negative affect in our anthropocentric environment. Dogs have been widely used as models of human behaviour due to the fact that during domestication this species has adapted to the same environmental challenges as humans [
21,
22,
23] and has thus developed several human-like socio-cognitive skills [
24,
25]. This makes them further valuable as a model system, since due to the increasing scientific interest in the species [
26], considerable amount of information has accumulated on their behaviour and neurocognition [
27]. Extensive data exists about dogs’ emotional processing, for example showing that they spontaneously distinguish between human emotions based on facial expressions [
28,
29,
30,
31]. Furthermore, dogs are able to base their in-test choices on human emotional expressions [
32,
33]. Dogs have also been shown to experience environmental stress [
34,
35], and they may develop phobias (e.g., thunderstorm-phobia [
36]). Some argue that dogs even possess secondary emotions like jealousy [
37] and guilt [
38,
39].
Several studies have found a relationship between dogs’ HR responses and stress in different situations [
40,
41,
42]. On the other hand, dogs’ HR and HRV have also been linked to positive emotions [
43,
44]. Research with shelter dogs has found that human-animal interactions not only alter HRV, but the magnitude of the HRV response is related to behavioural data [
45].
Recent studies also started to address the sleep physiology of family dogs, with a special interest in its relationship to dogs’ human-analogue social behaviours. It has been shown that dogs, despite displaying a polyphasic sleep structure, show several similarities to human sleep including the EEG spectra of the different sleep stages [
46], as well as the interrelatedness of sleep and memory consolidation [
47,
48]. Furthermore, recently it was found that significant differences can be observed in the macrostructure of sleep following negative (threatening approach, separation) or positive (playing, petting) pre-treatments [
49]. Data from these non-invasive studies is mostly in line with the decades of canine sleep research carried out with invasive methods, mainly focused on neurological conditions such as epilepsy [
50] and narcolepsy [
51]. Dogs are known to display polyphasic sleep [
52] with short sleep–wake cycles [
53]. A clear circadian, diurnal rhythm was also found in dogs [
54] similar to that in humans, and it was reported that dogs are most active after light onset and tend to have a rest during the afternoon [
55].
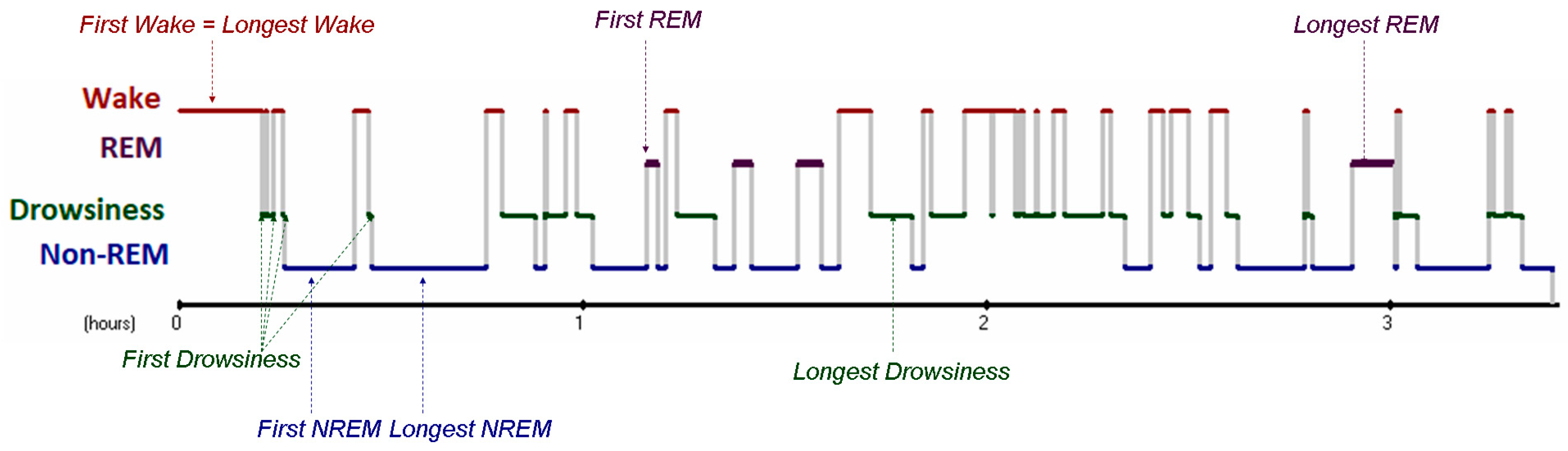
The present study capitalizes on these recent findings and explores the effect of positively versus negatively valenced social interactions on heart rate and heart rate variability during different stages of subsequent sleep in dogs. In order to do so, we first conduct a methodological study to establish heart rate measurements during sleep in dogs. Thus, Study I will provide descriptive data on dogs’ heart rate and heart rate variability during different stages of sleep after testing the effect of sampling length (20 s, 1 min, 5 min segments used for calculating heart rate data) as well as sample selection (longest period of the given sleep stage versus first occurrence of a given sleep stage). This is followed by Study II, testing the effect of pre-sleep positive and negative social interactions by analysing the ECG data from polysomnography recordings by [
49].
4. Discussion
This study was the first to quantitatively describe heart rate and heart rate variability during different stages of sleep in dogs. Our results show that there is a clear differentiation between ECG parameters during wakefulness versus sleep, while the differences between the distinct sleep stages are less pronounced. This finding is somewhat surprising given the fact that dogs’ sleep includes a so called drowsiness stage [
68,
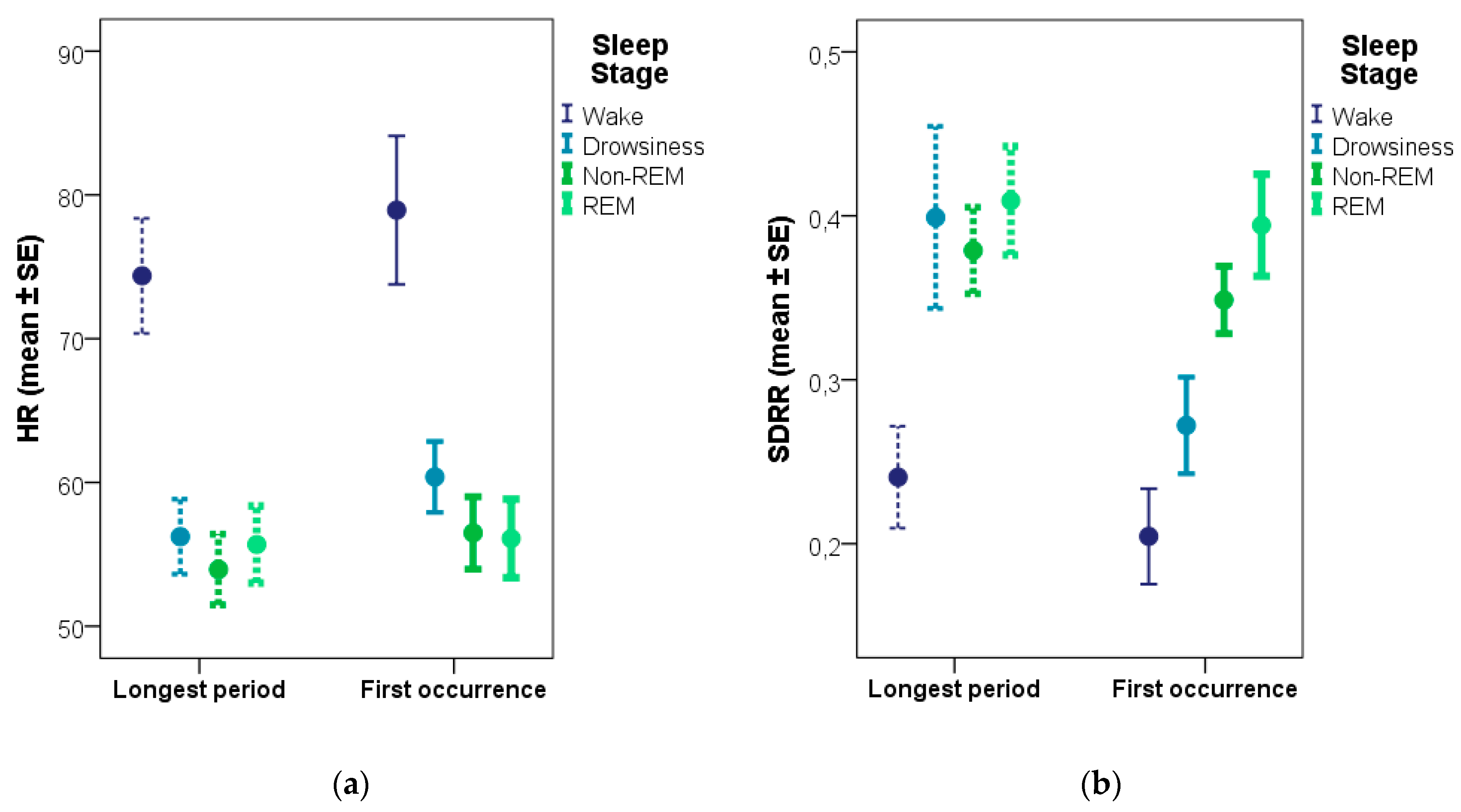
69] that, based on the EEG signal, acts like a transition between sleep and wake. Since the analysis of the first occurrence of each sleep stage (as opposed to the longest period of the given stage) found differences between not only wake and sleep, but also between drowsiness and sleep (non-REM, REM), we believe this sampling method to be the more reliable of the two.
Comparing our data to the literature, similar heart rate values have been found for resting and awake dogs, as in our study (74.4 and 78.9 bpm for the longest and the first wake period respectively). A study comparing dogs’ HR in different postures [
70] found approximately 90 bpm for lying, which overlaps with results [
71] from a group of resting (awake, un-anesthetized, lying) dogs that had a HR of 54–90 bpm, and is almost identical to a previous study from our group [
72] that found a mean ± SE HR of 89.47 ± 6.67 in resting awake dogs. Data from the current study are somewhat lower compared to the 90 bmp, which might suggest that dogs shortly before falling asleep have already lowered HR compared to simply lying down for a few minutes. Regarding HR during sleep, in studies using 24 h monitoring (without EEG-based sleep stage scoring) the minimum HR values fall close to our measures during both REM (55.7 bpm and 56.1 bpm) and non-REM (54.2 and 56.1 bpm): minimum mean 24 h heart rate (during behavioural sleep) was 43.12 ± 10.96 bpm in one study [
73], while in another study [
74] the 1 h periods with the lowest HR had 61 ± 3.5 bmp (4:00–5:00) and 62 ± 4.0 bmp (2:00–3:00). These data, together with our results following the expected tendencies between sleep stages (highest HR in wake, followed by drowsiness and then non-REM and REM sleep) [
8] are indicative of the validity of the methods used in the current study. The expected difference between drowsiness and the other sleep stages (non-REM, REM) is only apparent when contrasting samples from the first occurrence of a given stage (but not with samples from the longest periods) which might indicate that the validity of the ECG measures is more heavily compromised by using samples that are further away in time.
Comparing the three sampling lengths (20 s, 1 min and 5 min) some differences were found in the ECG measures suggesting that the 5 min sampling is necessary for properly measuring the HRV changes. Previous studies have often used 1 min samples with both dogs [
75] and humans [
76,
77] or even only a few seconds long recordings [
78]. Our current finding is in line with the outcome of a former human study that claimed the 5 min HRV results to be stable enough over time [
57].
Cardiac change is often used as a general psycho-physiological indicator, including in several domestic species [
62,
79,
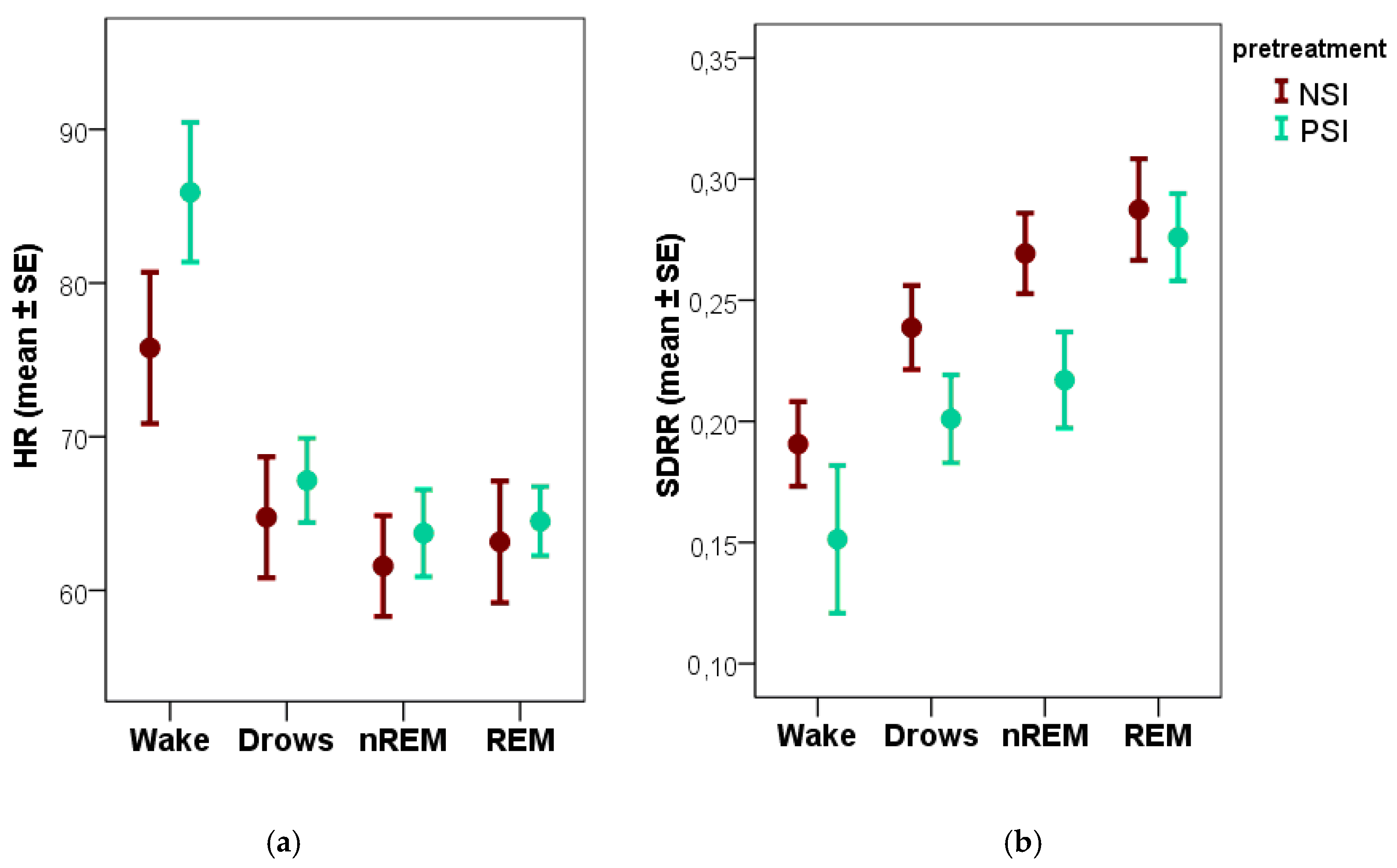
80]. Our results add to this literature by showing that changes in dogs’ HR and HRV are also noticeable after the positively versus negatively valenced social interactions. We should note, however, that such differences were only apparent during wakefulness, which is the very first stage in our recordings. Further studies should investigate if the non-significant trends found during non-REM sleep are indeed caused by the positive and negative social interactions that preceded the recordings.
Our results show that (during the wake phase) HR was higher, while the HRV measures were lower following the positive compared to the negative social interaction. This direction of change is contrary to what has been found in case of online ECG analysis, where negative interactions such as separation and threatening approach increased HR and decreased HRV [
65], while positive interactions such as petting by a human decreased HR [
66,
67]. Findings of the current study are instead more in line with the notion of the two-dimensional emotion model [
81] according to which not only valence (positivity/negativity), but also the arousal (low to high ‘activation’ or ‘intensity’) determines a given emotional experience. Based on the predictions of this model, intensity-dependent measures would react to a bigger extent to more intense emotions regardless of the valence (positivity or negativity) of the situation. There are some indications in the literature that (awake) heart rate data is one such intensity-dependent measure [
82]. Since due to ethical considerations the present study used an intense positive treatment, while only a mildly stressful negative treatment, the response pattern we found follows that of an intensity-dependent measure. This does not invalidate the previous finding, as low-key petting used as positive stimuli [
66] has been found to increase endogenous oxytocin release in dogs [
83], while intranasal oxytocin treatment, in turn, decreases dogs’ HR and increases HRV [
84]. In order to disentangle all possible hypotheses proposed by the two-dimensional emotion model, more research will be needed with positive and negative treatments of different intensity as well as a neutral baseline.
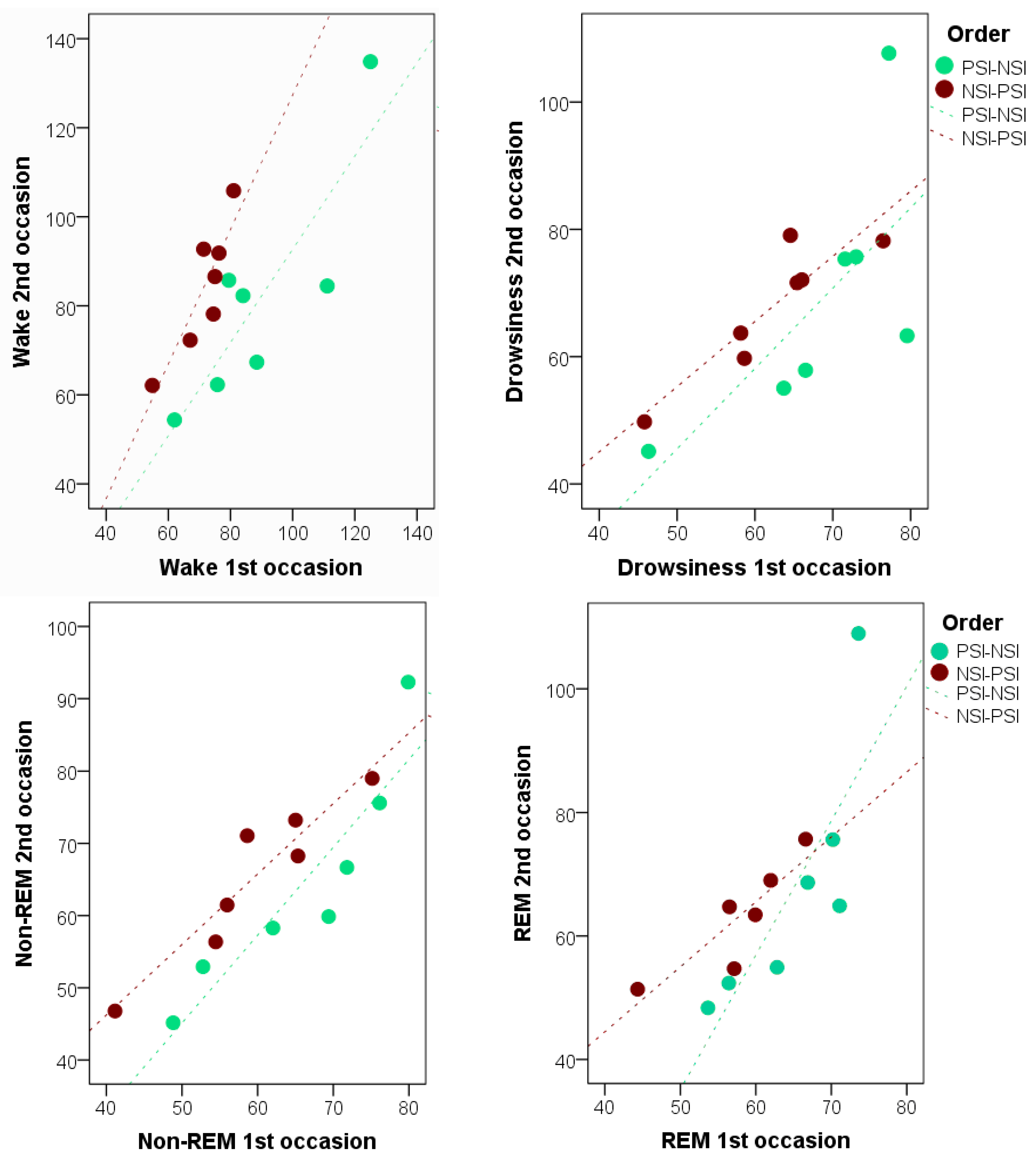
In addition, strong individual consistency was found between occasions in Study II. This is in line with the literature suggesting that several individual characteristics including age [
85], gender [
86], body mass [
87] and behavioural traits such as personality [
88] influence ECG parameters. It is unclear why in the current study some parameters showed within-individual consistency in certain sleep stages, while others did not, but the fact that several such within-individual correlations were found serves as a further methodological validation. Furthermore, this strong individual consistency together with the observed (and expected [
70]) high individual variation might have masked in some cases the effect of positive and negative emotional treatments, suggesting that studying more homogenous populations might be necessary in the future.