1. Introduction
Using different sources of growth promoters as dietary supplements to rabbit feed is a worldwide strategy to enhance the utilization of nutrients [
1,
2,
3,
4,
5]. Plant-derived supplements are used to maintain the growth performance of livestock animals [
6]. The active molecules in herbs or their oils can stimulate feed intake (FI), improve digestive enzyme secretion, and activate immune function, as well as promoting anthelmintic, antibacterial, antioxidant, and antiviral activities [
7]. After banning antibiotics as growth enhancers, scientists researched natural and safe alternatives. Recently, plant-derived growth enhancers are used globally, as reported by Ortserga et al. [
8].
Red pepper or
Capsicum annum is used widely as a condiment and shows an assortment of pharmacological and physiological features [
9,
10]. In Latin and Central America, Asia, and Africa, it is common for red pepper to be used to increase the spice level of food [
11]; however, it is well-known to be troublesome if consumed in excess [
12,
13]. Red pepper has many pharmacological and chemical properties like that of drugs’ classes which are capable of tissue deterioration inducing [
14]. Over the past few decades, it was experimentally confirmed that a number of the common spices can show beneficial physiological activities [
5]. The active compounds found in spices possess important roles in the promotion of physiological effects, including anti-oxidant and hypolipidemic activities [
7,
8,
15].
Capsaicin (8-methyl-
N-vanillyl-6-nonenamide), the active compound of pepper responsible for its spicy features, has captivated the interest of researchers for more than a century, who have proposed that it may have pharmacological and physiological effects [
14]. Red pepper oil (RPO) showed many properties like antimicrobial, anti-oxidant, antitumor, antifungal, and anti-inflammatory activities [
16,
17].
Black pepper (
Piper nigrum) is ordinarily consumed as a flavoring or used as a constituent in folk medicine. Remarkable uses for its active compound, termed ‘piperine,’ have been reported; for instance, it is used as a natural feed additive in livestock farming. Tatli et al. [
18] postulated that of all-natural products used in this regard, piperine can most easily be made in bulk at low cost. Interestingly, it was identified that piperine has massive advantages over other natural products: it produces no residues in the animal meat, increases cell recovery, and promotes anti-apoptotic and anti-oxidative activities. This suggests that piperine could be beneficial when used in the event of a compromised immune system, which is usually treated with antibiotics [
19,
20].
Piperine induces adipocytes to produce energy from lipids and hastens body energy expenditure [
21], as well as raising beta-endorphin production and serotonin level in the brain. Moreover, piperine can assuage gastrointestinal disturbances, cancer, and bronchitis by influencing gram positive bacteria and increasing the flow of digestive secretions in the stomach. However, there is inconsistent evidence about its effects on cancer [
22]. Some previous studies investigated impacts of black or red pepper powder as a dietary supplement for poultry, but studies on the use of their oils are very scarce. So, the present study aimed to evaluate the influences of using red or black pepper oil as a natural additive on the growth performance, carcasses, blood constituents, oxidative status and serum metabolites in rabbits.
4. Discussion
The analysis of RPO and BPO has been reported [
5,
6]. The Brazilian red pepper essential oil (RPEO) has abundant properties of interest, including antimicrobial, anti-inflammatory and antioxidant and activities, it can be used as a feed additive for weanling pigs [
8]. BPO is a powerful natural antioxidant that can slow down the oxidation of fats [
25]. Furthermore, BPO has a noteworthy antibacterial activity against both
Staphylococcus aureus and
Escherichia coli. BPO displayed significant anti-proliferative activity in dermal fibroblast cells. As well, BPO significantly inhibited the production of Collagen I and III as well as plasminogen activator inhibitor 1. At the gene level, BPO robustly modulated various genes and signaling pathways critical for tissue remodeling, metabolism, and cancer biology [
26].
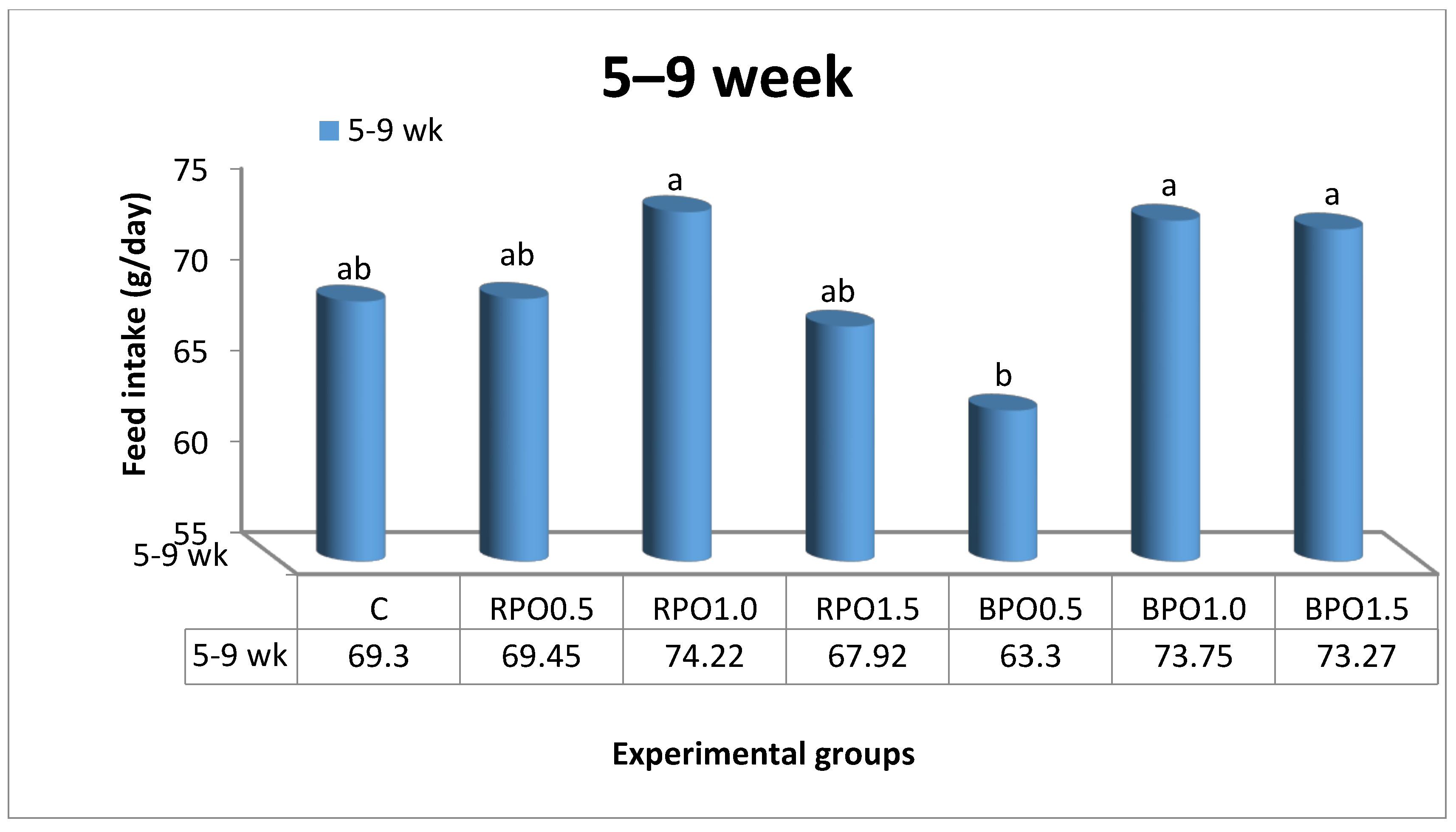
Values of LBW for rabbits fed either RPO- or BPO- enriched diets were higher (
p < 0.05) than those in the control group at nine weeks of age (
Table 2). The improvement in LBW and BWG by RPO or BPO as phytogenic additives may be due to the presence of some constituents that promote the digestion and absorption of nutrients. Additionally, Alagawany et al. [
27] suggested that it might be the biological activity of the components of RPO that enhance the FI and promote the growth rate. In fact, numerous in vitro and in vivo studies that have used active components (e.g., tannins, saponins, and flavonoids) extracted from herb extracts, have also reported antifungal, antimicrobial, anti-inflammatory, and anti-oxidant activities [
28]. Silva et al. [
29] confirmed that the supplementation of 4.0 g/kg RPO to broiler diets increased LBW and BWG compared to those did not receive antimicrobials and indicating that higher doses of RPO might be needed as a feed additive to achieve growth enhancing properties. Conversely, Cairo et al. [
30] stated that RPO did not impact growth of animals.
Results showed that supplementing the diet with BPO increased (
p < 0.05) the LBW compared to control (
Table 2). In parallel, Puvača et al. [
31] postulated that adding black pepper to broiler diets has a positive impact on growth rate of chickens; this is in accordance with the antecedent studies of Al-Kassie et al. [
22] and Valiollahi et al. [
32]. Additionally, Abou-Elkhair et al. [
33] showed that the addition of black pepper, or a mixture of turmeric powder and black pepper, to broiler chicken diets led to a higher final body weight of chickens during the fattening period of 35 days. The improvement in broiler body weight as a result of supplementation with black pepper powder was also observed and reported by Ghazalah et al. [
34]. Hosseini et al. [
35] observed that black pepper supplements can improve digestion and absorption of nutrients through increasing the secretion of digestive enzymes in the stomach and destroying infectious bacteria. The influence of black pepper is related to higher absorption, which augments the secretion of digestive enzymes and also decreases the velocity of material transit. The most active component in black pepper, piperine, promotes pancreatic digestive enzymes such as amylase, lipase, and protease, which play critical roles in digestion process [
36].
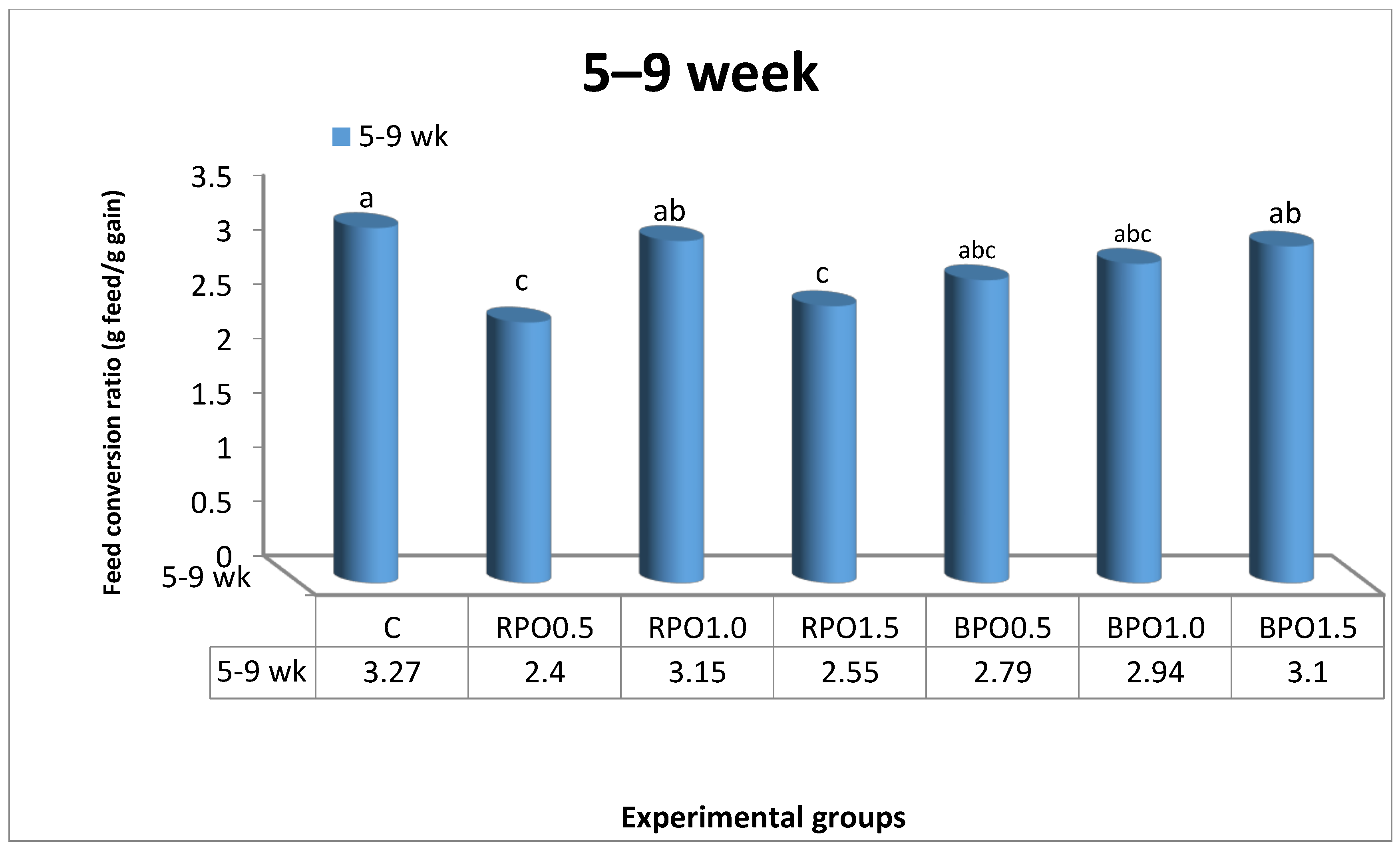
In accordance with our findings, Al-Kassie et al. [
22] demonstrated that the active compound capsaicin, which is rich in vitamin C, improves feed consumption and positively influences the body weight value. Moreover, Ghazalah et al. [
34] and Tollba et al. [
37] indicated that using a moderate level of black pepper stimulates feed consumption and causes the high piperazine citrate activity, which alters the flow of digestive fluids in the stomach. It has been reported that some botanicals such as RPO have the capacity to stimulate the endogenous production of enzymes, bile acids, or pancreatic juices, which improve nutrient digestibility, and thus, positively affect the FCR. In a previous study, Ipharraguerre et al. [
38] observed that red pepper supplementation improved dietary fat digestibility when supplemental fat was included at 4%. The findings of our study are similar to those of Al-Harthi [
39], who revealed that the supplementation of hot red pepper to chick diet improved FCR and induced digestion, because of its carminative property and antimicrobial attributes. The improvement in FCR in the present study may be due to the ability of black pepper to improve the digestibility of the feed which would improve the FCR and other growth parameters [
40]. However, this improvement was insignificant according to Al-Harthi [
39]. Conversely, Al-Kassie et al. [
22] and Abou-Elkhair et al. [
33] both reported that the use of black pepper powder in chicken feed did not have a positive influence on the FCR.
Results in
Table 3 showed that the majority of the carcass and edible organ percentages were not significantly affected by dietary treatments. In partial agreement with our findings, Cairo et al. [
30] observed no differences in the relative size of edible organs between animals fed diets supplemented with different quantities of RPO. Additionally, Costa et al. [
41], reported no differences in the organ weights of animals fed diets supplemented with essential oils of thyme, cinnamon, eucalyptus,
Melaleuca alternifolia,
Echinacea angustifolia, ginger and pepper extracts. Al-Kassie et al. [
22] stated that a mixture of red and black pepper powder did not exert any significant effects on the carcass or organ weights of broilers. In contrast with our results, Rahimian et al. [
42] postulated that liver relative weight was significantly increased (
p < 0.05) in broilers fed with black pepper compared to those in the control.
The supplementation with RPO decreased (
p < 0.01) the blood content of hemoglobin, mean corpuscular volume, and platelet count compared to those in the control (
Table 4). Al-Kassie et al. [
22] showed that broilers fed with a black and red pepper mixture had a significantly lower red blood cell count, packed cell volume, and hemoglobin level compared to those in the control group.
Our results showed better liver and kidney function in rabbits fed RPO or BPO than the control (
Table 5). Corduk et al. [
43] stated that the rapid metabolization of essential oils in the liver can damage it and consequently increase serum content of the liver enzymes (AST and ALT). However, our findings related to the serum AST and ALT levels showed that the levels of RPO and BPO were safe and useful in expressing renal and liver function. Similarly, Traesel et al. [
44] postulated that the prolonged use of high levels of supplementary essential oils could not cause nephritis or renal failure.
A significant increase (
p < 0.05) was reported in the serum TP and albumin as a response to RPO and BPO supplementation (
Table 5). This increase in serum TP and albumin may be due to the bioactive components in RPO and BPO. On the contrary, Dabbou et al. [
7] and Kovitvadhi et al. [
45] mentioned that dietary phytogenic supplementation did not affect the blood parameters (TP, globulin, and albumin) or humoral immune responses in growing rabbits.
Levels of serum TC, triglyceride, LDL were decreased (
p < 0.01) and serum HDL was increased (
p < 0.01) in RPO- and BPO-treated groups compared to those in the control. The reduction in blood cholesterol may be due to the decrease in the activity of enzyme synthesis, as hypothesized by Chowdhury et al. [
46]. On the other hand, Srinivasan and Satyanarayana [
47] indicated that capsaicin is considered the active component of red hot peppers and feeding female rats with red hot pepper depressed their serum triglyceride contents. In accordance with our results, Puvača et al. [
31] found that supplementation with 1.0 g/100 g of black pepper significantly (
p < 0.05) decreased the levels of triglycerides in the blood serum in broilers. The authors added that this result can be clarified by the conceivable decrease in acetyl-CoA enzyme synthesis that is important for the biosynthesis of fatty acids. In the present study, the addition of either RPO or BPO to rabbit diet decreased the serum LDL and increased the serum HDL. This influence could be elucidated by the possible mechanism of anti-oxidant and antiperoxide activity decreasing LDL, or the decline in hepatic production of very low-density lipoprotein (VLDL) which is a precursor of LDL in the bloodstream [
48]. Ghaedi et al. [
49] found that the addition of black pepper to the diet of broilers decreased triglycerides and total cholesterol, while the concentration of HDL increased. Moreover, Al-Kassie et al. [
22] reported that broilers fed with black pepper and red pepper mixture had significantly lowered cholesterol. Authors theorized that the addition of spices or herbs to animal feed can facilitate the activity of enzymes that participate in the transformation of cholesterol to bilious acids, and subsequently will result in a lower cholesterol concentration in the carcass. In disagreement with our results, Corduk et al. [
43] found no significant influences on the serum total protein, triglyceride, cholesterol, creatinine, and alanine aminotransferase enzyme contents after RPO addition to broiler diets. Similarly, Al-Harthi [
39] described that a combination of cumin, cardamom, and black and red pepper at 2 and 4 g/kg did not significantly influence the TC, TP, or ALT enzyme contents.
The enhancement of immune functions in treated groups (
Table 6) may imply herbal supplements are rich in flavonoids, which act as strong anti-oxidants [
50]. The supplementation of RPO and BPO may enhance the immune function via augmentation of the immunoglobulin (IgM) levels in rabbits fed phytogenic feed additives compared to those in the control group. The positive effects of RPO and BPO might be due to their antibacterial, antioxidant, and anti-inflammatory properties. These phytogenic additives are proposed to lower the growth and colonization of pathogenic and non-pathogenic species of bacteria in the gut of rabbits, and to balance microbial ecosystems in the gut, which all contribute to better feed utilization and metabolism [
51]. These findings are similar to the results of Alagawany et al. [
27], who reported that supplementation of 400 and 600 mg/kg diet of yucca powder showed greater levels of IgM and IgG in rabbit blood compared to that in the control group.
In the RPO- and BPO-treated groups, the levels of TAC and SOD were significantly (
p < 0.01) increased; while the highest (
p = 0.005) concentration of MDA was recorded in the control group. The SOD plays a major role in protecting cells from oxidative damage; this process requires specific nutrients to be present in the diet [
6]. The present conclusions are in accordance with those of Lin et al. [
52], who reported that herb intake caused an increase in serum antioxidant enzyme activities and a decrease in MDA levels. Generally, it was reported that essential oils of pepper enhance amino acid absorption, such as methionine or cysteine, in the gastro-intestinal tract which are the limiting factors of glutathione synthesis [
22,
30,
42]. The reduction in glutathione in treated groups might be due to the inhibition the formation of glutathione disulfide; thereby glutathione can protect cells from the attack of free radicals, prevent from the oxidative damage of macromolecules, and inhibit apolipoprotein B protein peroxidation [
27]. Alagawany et al. [
27] showed that dietary supplementation of herbs to rabbit diets had positive effects on both SOD and TAC activities. From these findings, it could be proposed that supplements with natural antioxidants could be practical in the future to enhance the health status of rabbits.