Betaine and Antioxidants Improve Growth Performance, Breast Muscle Development and Ameliorate Thermoregulatory Responses to Cyclic Heat Exposure in Broiler Chickens
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval and Consent to Participate
2.2. Birds and Husbandry Housing
2.3. Experimental Treatments
2.4. Physiological Responses
2.5. Tissue Sampling and Euthanasia
2.6. Intestinal Barrier Integrity and Permeability
2.7. Glutathione Peroxidase Assay
2.8. Statistical Analysis
3. Results
3.1. Performance
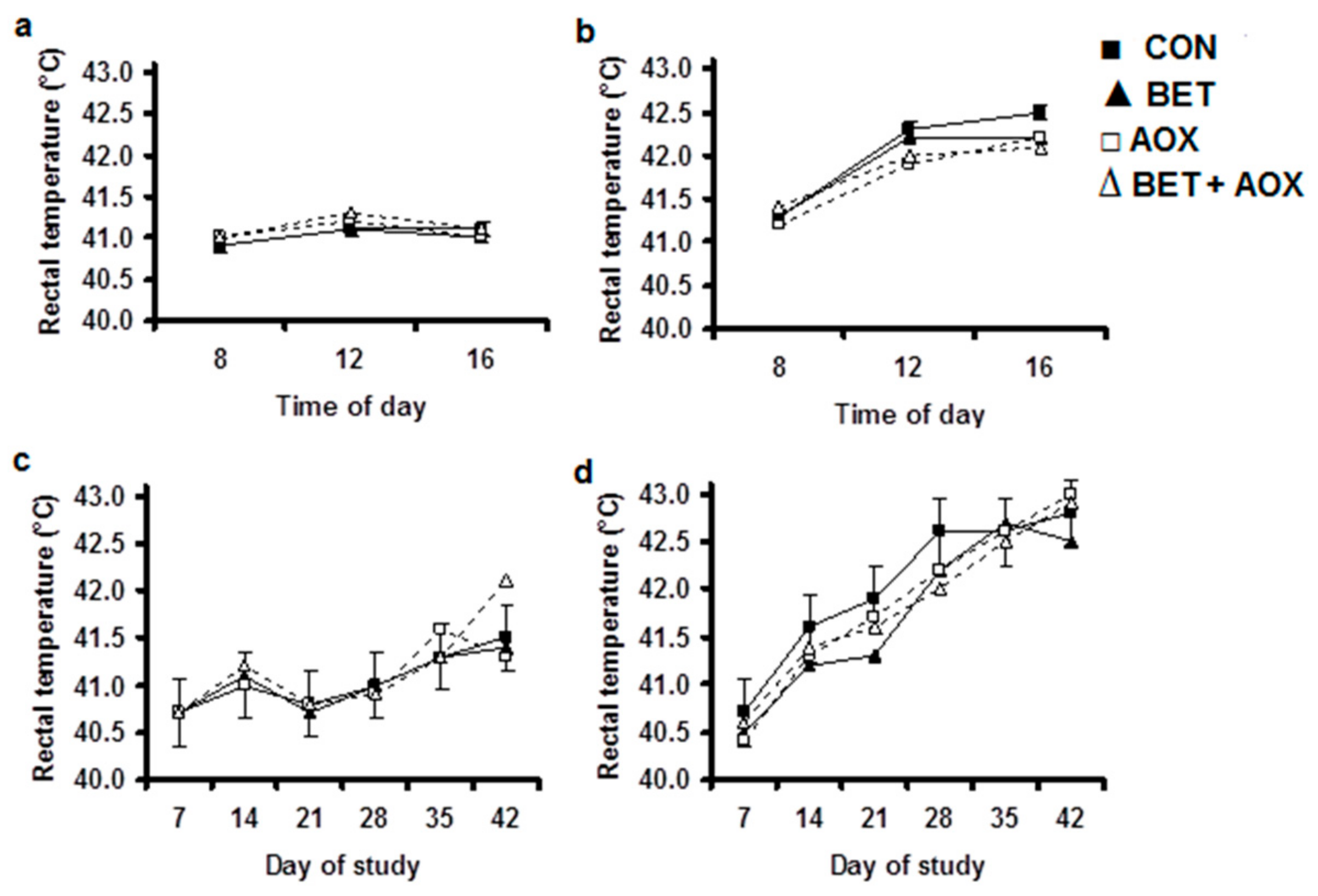
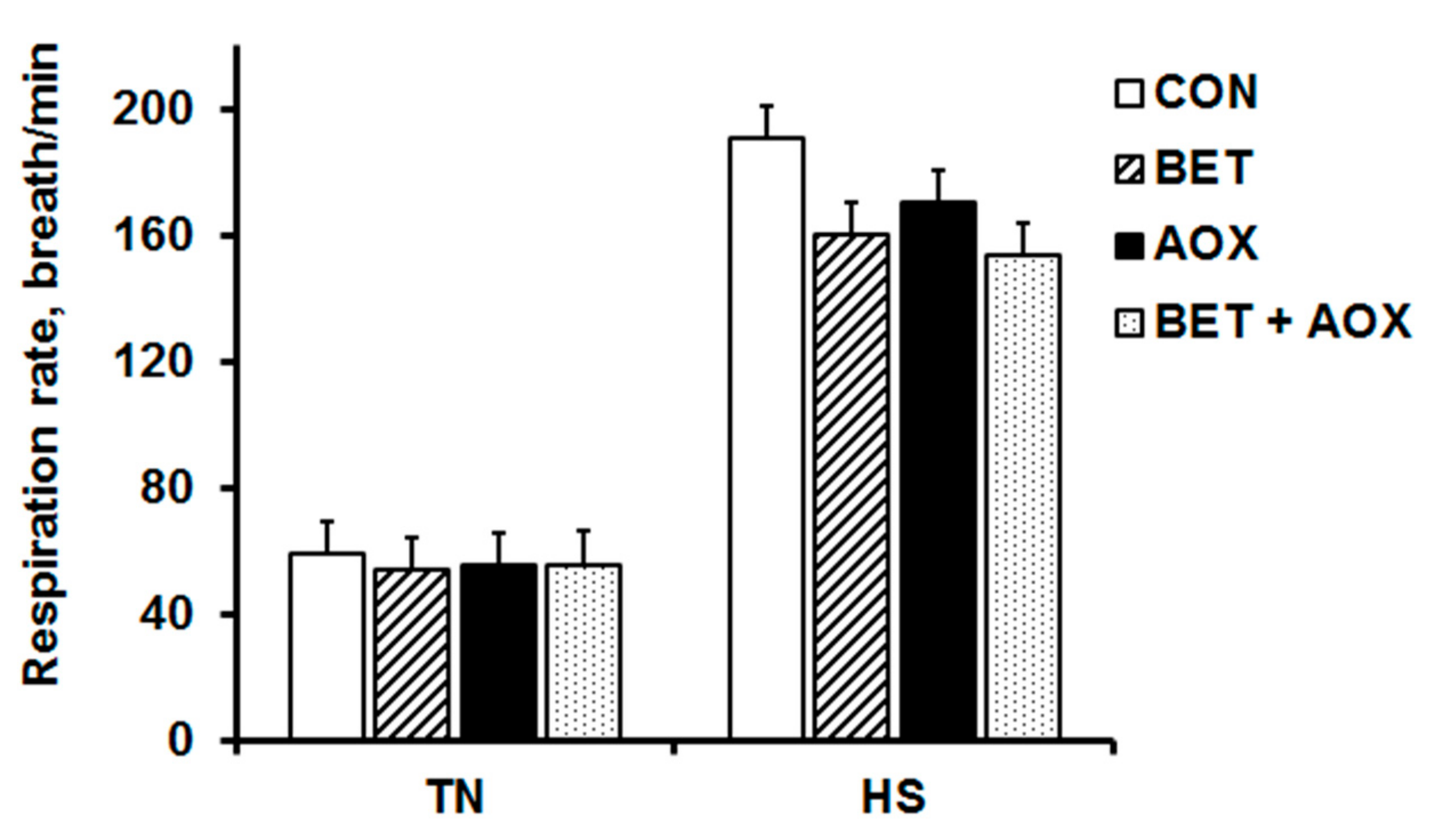
3.2. Physiological Responses
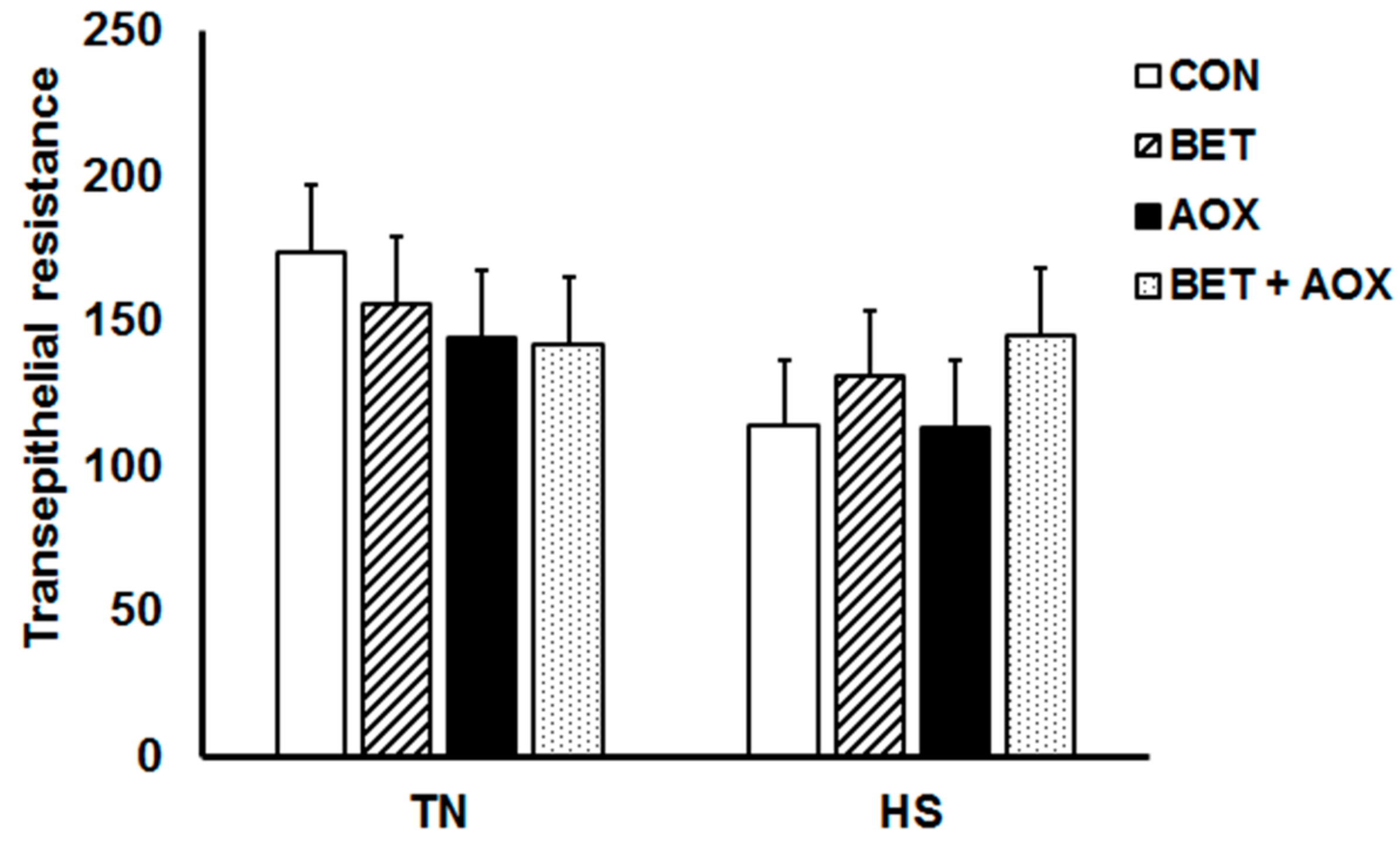
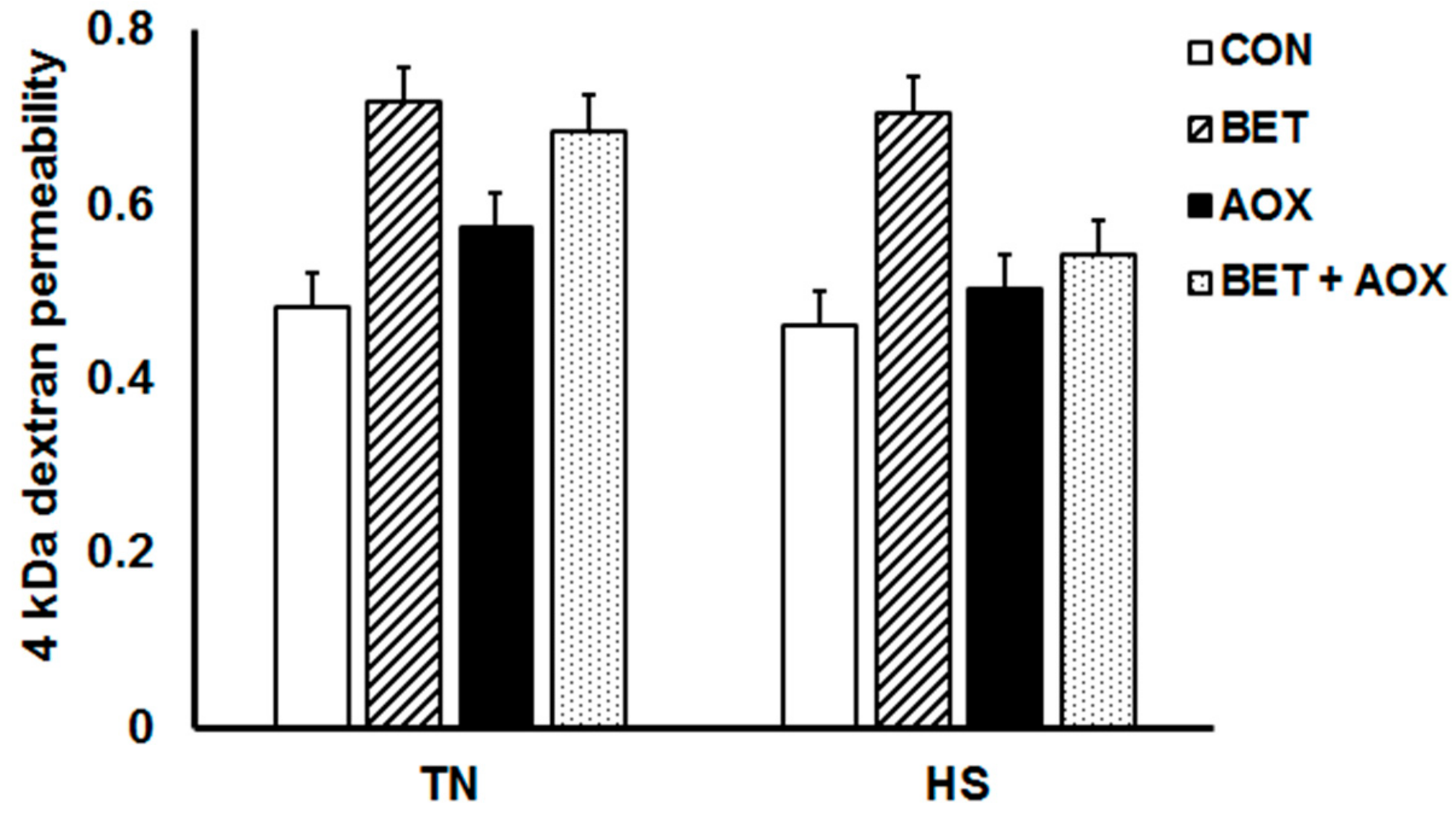
3.3. Intestinal Epithelial Barrier Integrity
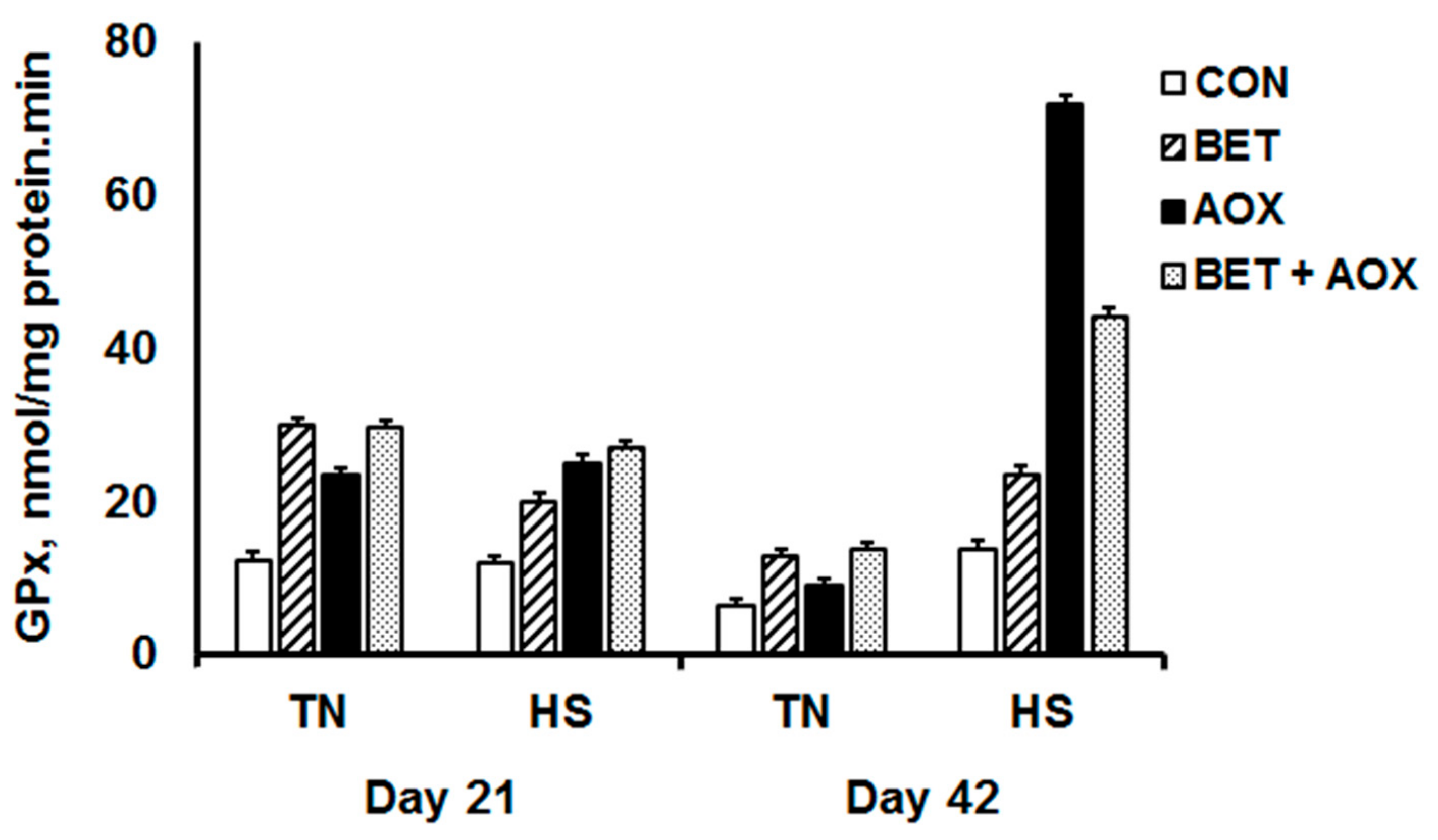
3.4. Glutathione Peroxidase Activity
4. Discussion
4.1. Effects of Betaine, Selenium, and Vitamin E on Growth Performance and Physiological Responses
4.2. Effects of Betaine, Selenium, and Vitamin E on Gut Permeability and Integrity
4.3. Effects of Betaine, Selenium, and Vitamin E on Glutathione Peroxidase Activity
4.4. Betaine, Selenium, and Vitamin E Functions During Heat Stress
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Syafwan, S.; Kwakkel, R.; Verstegen, M. Heat stress and feeding strategies in meat-type chickens. World’s Poult. Sci. J. 2011, 67, 653–674. [Google Scholar] [CrossRef]
- Garriga, C.; Hunter, R.R.; Amat, C.; Planas, J.M.; Mitchell, M.A.; Moretó, M. Heat stress increases apical glucose transport in the chicken jejunum. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R195–R201. [Google Scholar] [CrossRef] [PubMed]
- Quinteiro-Filho, W.M.; Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Sakai, M.; Sá, L.R.M.; Palermo-Neto, J. Heat stress impairs performance parameters, induces intestinal injury, and decreases macrophage activity in broiler chickens. Poult. Sci. 2010, 89, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- Lambert, G.P. Stress-induced gastrointestinal barrier dysfunction and its inflammatory effects. J. Anim. Sci. 2009, 87, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Keita, Å.V.; Söderholm, J.D. The intestinal barrier and its regulation by neuroimmune factors. Neurogastroenterol. Motil. 2010, 22, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Dunshea, F.R.; Leury, B.J.; Fahri, F.T.; Digiacomo, K.; Hung, A.; Chauhan, S.S.; Clarke, I.J.; Collier, R.J.; Little, S.; Baumgard, L.H.; et al. Amelioration of thermal stress impacts in dairy cows. Anim. Prod. Sci. 2013, 53, 965–975. [Google Scholar] [CrossRef]
- Moeckel, G.W.; Shadman, R.; Fogel, J.M.; Sadrzadeh, S.M. Organic osmolytes betaine, sorbitol and inositol are potent inhibitors of erythrocyte membrane ATPases. Life Sci. 2002, 71, 2413–2424. [Google Scholar] [CrossRef]
- Tsai, M.T.; Chen, C.Y.; Pan, Y.H.; Wang, S.H.; Mersmann, H.J.; Ding, S.T. Alleviation of carbon-tetrachloride-induced liver injury and fibrosis by betaine supplementation in chickens. Evid. Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Cronjé, P. Heat stress in livestock—The role of the gut in its aetiology and a potential role for betaine in its alleviation. Recent Adv. Anim. Nutr. Aust. 2005, 15, 107–122. [Google Scholar]
- Kettunen, H.; Tiihonen, K.; Peuranen, S.; Saarinen, M.T.; Remus, J.C. Dietary betaine accumulates in the liver and intestinal tissue and stabilizes the intestinal epithelial structure in healthy and coccidia-infected broiler chicks. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 130, 759–769. [Google Scholar] [CrossRef]
- Matthews, J.; Southern, L. The effect of dietary betaine in Eimeria acervulina-infected chicks. Poult. Sci. 2000, 79, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Babazadeh, D.; Naveed, M.; Arain, M.A.; Hassan, F.U.; Chao, S. Reconsidering betaine as a natural anti-heat stress agent in poultry industry: A review. Trop. Anim. Health Prod. 2017, 49, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.A.; Li, J.X.; Xu, Z.R.; Zhao, R.Q. Effects of methionine and betaine supplementation on growth performance, carcase composition and metabolism of lipids in male broilers. Br. Poult. Sci. 2006, 47, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Attia, Y.A.; Hassan, R.A.; Qota, E.M.A. Recovery from adverse effects of heat stress on slow-growing chicks in the tropics 1: Effect of ascorbic acid and different levels of betaine. Trop. Anim. Health Prod. 2009, 41, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Hassan, R.A.; Attia, Y.A.; El-Ganzory, E.H. Growth, carcass quality and serum constituents of slow growing chicks as affected by betaine addition to diets containing 1. Different levels of choline. Int. J. Poult. Sci. 2005, 4, 840–850. [Google Scholar]
- Krstić, B.; Jokić, Z.; Pavlović, Z.; Zivković, D. Options for the production of selenized chicken meat. Biol. Trace Elem. Res. 2012, 146, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Dale, N. National research council nutrient requirements of poultry–ninth revised edition (1994). J. Appl. Poult. Res. 1994, 3, 101. [Google Scholar] [CrossRef]
- Yang, Y.R.; Meng, F.C.; Wang, P.; Jiang, Y.B.; Yin, Q.Q.; Chang, J.; Zuo, R.Y.; Zheng, Q.H.; Liu, J.X. Effect of organic and inorganic selenium supplementation on growth performance, meat quality and antioxidant property of broilers. Afr. J. Biotechnol. 2012, 11, 3031–3036. [Google Scholar]
- Niu, Z.; Liu, F.; Yan, Q.; Li, L. Effects of different levels of selenium on growth performance and immunocompetence of broilers under heat stress. Arch. Anim. Nutr. 2009, 63, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Habibian, M.; Sadeghi, G.; Ghazi, S.; Moeini, M.M. Selenium as a feed supplement for heat-stressed poultry: A review. Biol. Trace Elem. Res. 2015, 165, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Uglietta, R.; Doyle, P.T.; Walker, G.P.; Heard, J.W.; Leddin, C.M.; Stockdale, C.R.; Mcintosh, G.H.; Young, G.P.; Dunshea, F.R. Bioavailability of selenium from selenium-enriched milk assessed in the artificially reared neonatal pig. Nutr. Diet. 2008, 65 (Suppl. S3), 37–40. [Google Scholar] [CrossRef]
- Packer, L.; Weber, S.U.; Rimbach, G. Molecular aspects of α-tocotrienol antioxidant action and cell signalling. J. Nutr. 2001, 131, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Villacorta, L.; Graça-Souza, A.V.; Ricciarelli, R.; Zingg, J.M.; Azzi, A.L. α-Tocopherol induces expression of connective tissue growth factor and antagonizes tumor necrosis factor-α–mediated downregulation in human smooth muscle cells. Circ. Res. 2003, 92, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Lin, H.; Wang, X.J.; Song, Z.G.; Jiao, H.C. Vitamin E supplementation alleviates the oxidative stress induced by dexamethasone treatment and improves meat quality in broiler chickens. Poult. Sci. 2010, 89, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Kucuk, O. Effects of vitamin C and vitamin E on performance, digestion of nutrients and carcass characteristics of Japanese quails reared under chronic heat stress (34 °C). J. Anim. Physiol. Anim. Nutr. 2001, 85, 335–341. [Google Scholar] [CrossRef]
- Hosseini-Mansoub, N.; Chekani-Azar, S.; Tehrani, A.; Lotfi, A.; Manesh, M. Influence of dietary vitamin E and zinc on performance, oxidative stability and some blood measures of broiler chickens reared under heat stress (35 °C). J. Agrobiol. 2010, 27, 103–110. [Google Scholar] [CrossRef]
- Hashizawa, Y.; Kubota, M.; Kadowaki, M.; Fujimura, S. Effect of dietary vitamin E on broiler meat qualities, color, water-holding capacity and shear force value, under heat stress conditions. Anim. Sci. J. 2013, 84, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Kamely, M.; Karimi Torshizi, M.A.; Rahimi, S. Incidence of ascites syndrome and related hematological response in short-term feed-restricted broilers raised at low ambient temperature. Poult. Sci. 2015, 94, 2247–2256. [Google Scholar] [CrossRef] [PubMed]
- Aviagen. Ross Broiler Management Handbook; Aviagen: Huntsville, AL, USA, 2014; pp. 11–53. [Google Scholar]
- Liu, F.; Cottrell, J.J.; Furness, J.B.; Rivera, L.R.; Kelly, F.W.; Wijesiriwardana, U.; Leury, B.J. Selenium and vitamin E together improve intestinal epithelial barrier function and alleviate oxidative stress in heat-stressed pigs. Exp. Physiol. 2016, 101, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Pearce, S.C.; Mani, V.; Boddicker, R.L.; Johnson, J.S.; Weber, T.E.; Ross, J.W.; Gabler, N.K. Heat stress reduces intestinal barrier integrity and favors intestinal glucose transport in growing pigs. PLoS ONE. 2013, 8, 70215. [Google Scholar] [CrossRef] [PubMed]
- SAS Institute Inc. SAS/Graph 9.1 Reference; SAS Institute Inc.: Cary, NC, USA, 2004. [Google Scholar]
- Zhang, Z.Y.; Jia, G.Q.; Zuo, J.J.; Zhang, Y.; Lei, J.; Ren, L.; Feng, D.Y. Effects of constant and cyclic heat stress on muscle metabolism and meat quality of broiler breast fillet and thigh meat. Poult. Sci. 2012, 91, 2931–2937. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, H.A.G.; Khan, M.S.; Khan, M.A.; Rabbani, M.; Pervez, K.; Khan, J.A. Evaluation of betaine and vitamin C in alleviation of heat stress in broilers. Int. J. Agric. Biol. 2005, 5, 744–746. [Google Scholar]
- Teeter, R.G.; Smith, M.O.; Owens, F.N.; Arp, S.C.; Sangiah, S.; Breazile, J.E. Chronic heat stress and respiratory alkalosis: Occurrence and treatment in broiler chicks. Poult. Sci. 1985, 64, 1060–1064. [Google Scholar] [CrossRef] [PubMed]
- Alirezaei, M.; Gheisari, H.R.; Ranjbar, V.R.; Hajibemani, A. Betaine: A promising antioxidant agent for enhancement of broiler meat quality. Br. Poult. Sci. 2012, 53, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Nofal, M.E.; Magda, A.G.; Mousa, S.M.M.; Doaa, M.M.; Bealsh, A.M.A. Effect of dietary betaine supplementation on productive, physiology and immunological performance and carcass characteristic of growing deleloped chicks under the condition of heat stress. Egypt. Poult. Sci. J. 2015, 35, 237–259. [Google Scholar]
- Swan, J.; Boles, J. Processing characteristics of beef roasts made from high and normal pH bull inside rounds. Meat Sci. 2002, 62, 399–403. [Google Scholar] [CrossRef]
- He, S.; Zhao, S.; Dai, S.; Liu, D.; Bokhari, S.G. Effects of dietary betaine on growth performance, fat deposition and serum lipids in broilers subjected to chronic heat stress. Anim. Sci. J. 2015, 86, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Craig, S.A. Betaine in human nutrition. Am. J. Clin. Nutr. 2004, 80, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Petričević, V.; Pavlovski, Z.; Škrbić, Z.; Lukić, M. The effect of genotype on production and slaughter properties of broiler chickens. Biotech. Anim. Husbandry 2011, 27, 171–181. [Google Scholar] [CrossRef]
- Dal Bosco, A.; Simona, M.; Silvia, R.; Cecilia, M.; Cesare, C. Effect of slaughtering age in different commercial chicken genotypes reared according to the organic system: 1. Welfare, carcass and meat traits. Ital. J. Anim. Sci. 2014, 13, 467–472. [Google Scholar]
- Attia, Y.A.; Al-Harthi, M.A.; Sh Elnaggar, A. Productive, physiological and immunological responses of two broiler strains fed different dietary regimens and exposed to heat stress. Ital. J. Anim. Sci. 2018, 17, 686–697. [Google Scholar] [CrossRef]
- Garcia Neto, M.; Pesti, G.M.; Bakalli, R.I. Influence of dietary protein level on the broiler chicken’s response to methionine and betaine supplements. Poult. Sci. 2000, 79, 1478–1484. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.V.; Raju, M.V.L.N.; Panda, A.K.; Saharia, P.; Sunder, G.S. Effect of supplementing betaine on performance, carcass traits and immune responses in broiler chicken fed diets containing different concentrations of methionine. Asian-Aust. J. Anim. Sci. 2011, 24, 662–669. [Google Scholar] [CrossRef]
- Kettunen, H.; Peuranen, S.; Tiihonen, K. Betaine aids in the osmoregulation of duodenal epithelium of broiler chicks, and affects the movement of water across the small intestinal epithelium in vitro. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 129, 595–603. [Google Scholar] [CrossRef]
- Egbuniwe, I.C.; Ayo, J.O.; Kawu, M.U.; Mohammed, A. Effects of betaine and ascorbic acid on tonic immobility, superoxide dismutaser and glutathione peroxidase in broiler chickens during the hot-dry season. J. Vet. Behav. 2016, 12, 60–65. [Google Scholar] [CrossRef]
- Gabler, N.; Frouel, S.; Awati, A.; Owusu-Asiedu, A.; Amerah, A.; Patridge, G.; Dunshea, F.R. Betaine mitigates intestinal permeability in growing pigs induced by heat stress. In Proceedings of the Manipulating Pig Production XIV, Melbourne, Australia, 24–27 November 2013; Pluske, J.R., Pluske, J.M., Eds.; Australian Pig Science Association: Melbourne, Australia, 2013. [Google Scholar]
- Ibrahim, M.T.; Eljack, B.H.; Fadlalla, I.M.T. Selenium supplementation to broiler diets. Anim. Sci. J. 2011, 2, 12–17. [Google Scholar]
- Tellez, J.G.; Tellez-Isaias, G.; Dridi, S. Heat stress and gut health in broilers: Role of tight junction proteins. Adv. Food Technol. Nutr. Sci. Open J. 2017, 3, 1–4. [Google Scholar] [CrossRef]
- Liu, S.K.; Niu, Z.Y.; Min, Y.N.; Wang, Z.P.; Zhang, J.; He, Z.F.; Li, H.L.; Sun, T.T.; Liu, F.Z. Effects of dietary crude protein on the growth performance, carcass characteristics and serum biochemical indexes of Lueyang black-boned chickens from seven to twelve weeks of age. Rev. Bras. Cienc. Avic. 2015, 17, 103–108. [Google Scholar] [CrossRef]
- Dunshea, F.R.; Cadogan, D.; Partridge, G. Dietary betaine and ractopamine combine to increase lean tissue deposition in finisher pigs, particularly gilts. Anim. Prod. Sci. 2008, 49, 65–70. [Google Scholar] [CrossRef]
- Zulkifli, I.; Mysahra, S.A.; Jin, L.Z. Dietary supplementation of betaine (Betafin®) and response to high temperature stress in male broiler chickens. Asian-Aust. J. Anim. Sci. 2004, 17, 244–249. [Google Scholar] [CrossRef]
- DiGiacomo, K.; Simpson, S.; Leury, B.J.; Dunshea, F.R. Dietary Betaine Impacts the Physiological Responses to Moderate Heat Conditions in a Dose Dependent Manner in Sheep. Animals 2016, 6, 51. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.S.; Celi, P.; Leury, B.J.; Clarke, I.J.; Dunshea, F.R. Dietary antioxidants at supranutritional doses improve oxidative status and reduce the negative effects of heat stress in sheep. J. Anim. Sci. 2014, 92, 3364–3374. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.A.; Kronfeld, D.S.; Hess, T.M.; Saker, K.E.; Waldron, J.N.; Crandell, K.M.; Harris, P.A. Antioxidant supplementation and subsequent oxidative stress of horses during an 80-km endurance race 1. J. Anim. Sci. 2004, 82, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; De Vos, D.; Decuypere, E.; Buyse, J. Dynamic changes in parameters of redox balance after mild heat stress in aged laying hens (Gallus gallus domesticus). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2008, 147, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Decuypere, E.; Buyse, J. Acute heat stress induces oxidative stress in broiler chickens. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2006, 144, 11–17. [Google Scholar] [CrossRef] [PubMed]
- De Vega, R.G.; Fernández-Sánchez, M.L.; Fernández, J.C.; Menéndez, F.V.; Sanz-Medel, A. Selenium levels and glutathione peroxidase activity in the plasma of patients with type II diabetes mellitus. J. Trace Elem. Med. Biol. 2016, 37, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Altan, Ö.; Pabuçcuoğlu, A.; Altan, A.; Konyalioğlu, S.; Bayraktar, H. Effect of heat stress on oxidative stress, lipid peroxidation and some stress parameters in broilers. Br. Poult. Sci. 2003, 44, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.; Kikusato, M.; Maekawa, T.; Shirakawa, H.; Toyomizu, M. Metabolic characteristics and oxidative damage to skeletal muscle in broiler chickens exposed to chronic heat stress. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2010, 155, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.Y.; Yang, L.; Fu, Y.Q.; Feng, J.H.; Zhang, M.H. Effects of different acute high ambient temperatures on function of hepatic mitochondrial respiration, antioxidative enzymes, and oxidative injury in broiler chickens. Poult. Sci. 2010, 89, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Hao, Y.; Wang, X. Overexpression of heat shock protein 70 and its relationship to intestine under acute heat stress in broilers: 2. Intestinal oxidative stress. Poult. Sci. 2012, 91, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.J.; Wu, C.X.; Gong, L.M.; Song, T.; Wu, H.; Zhang, L.Y. Effects of nano-selenium on performance, meat quality, immune function, oxidation resistance, and tissue selenium content in broilers. Poult. Sci. 2012, 91, 2532–2539. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, Y. Influence of dietary nano elemental selenium on growth performance, tissue selenium distribution, meat quality, and glutathione peroxidase activity in Guangxi Yellow chicken. Poult. Sci. 2011, 90, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Harsini, S.G.; Habibiyan, M.; Moeini, M.M.; Abdolmohammadi, A.R. Effects of dietary selenium, vitamin E, and their combination on growth, serum metabolites, and antioxidant defense system in skeletal muscle of broilers under heat stress. Biol. Trace Elem. Res. 2012, 148, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.D.; Baker, S.S.; LaRosa, K.; Whitney, C.; Newburger, P.E. Selenium regulation of glutathione peroxidase in human hepatoma cell line Hep3B. Arch. Biochem. Biophys. 1993, 304, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Kheradmand, A.; Alirezaei, M.; Dezfoulian, O. Cadmium-induced oxidative stress in the rat testes: Protective effects of betaine. Int. J. Pept. Res. Ther. 2013, 19, 337–344. [Google Scholar] [CrossRef]
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Suster, D.; Leury, B.J.; King, R.H.; Mottram, M.; Dunshea, F.R. Interrelationships between porcine somatotropin (pST), betaine, and energy level on body composition and tissue distribution of finisher boars. Aust. J. Agric. Res. 2004, 55, 983–990. [Google Scholar] [CrossRef]
- Sakomura, N.K.; Longo, F.A.; Oviedo-Rondon, E.O.; Boa-Viagem, C.; Ferraudo, A. Modeling energy utilization and growth parameter description for broiler chickens. Poult. Sci. 2005, 84, 1363–1369. [Google Scholar] [CrossRef] [PubMed]
- Nunes, V.A.; Gozzo, A.J.; Cruz-Silva, I.; Juliano, M.A.; Viel, T.A.; Godinho, R.O.; Meirelles, F.V.; Sampaio, M.U.; Sampaio, C.A.; Araujo, M.S. Vitamin E prevents cell death induced by mild oxidative stress in chicken skeletal muscle cells. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2005, 141, 225–240. [Google Scholar] [CrossRef] [PubMed]
- McDowell, L.R. Vitamins in Animal Nutrition: Comparative Aspects to Human Nutrition; Academic Press: San Diego, CA, USA, 1989; pp. 98–101. [Google Scholar]
- Perić, L.; Milošević, N.; Žikić, D.; Kanački, Z.; Džinić, N.; Nollet, L.; Spring, P. Effect of selenium sources on performance and meat characteristics of broiler chickens. J. Appl. Poult. Res. 2009, 18, 403–409. [Google Scholar] [CrossRef]
- Zduńczyk, Z.; Gruzauskas, R.; Semaskaite, A.; Juskiewicz, J.; Raceviciute-Stupeliene, A.; Wroblewska, M. Fatty acid profile of breast muscle of broiler chickens fed diets with different levels of selenium and vitamin, E. Arch Geflugelkd. 2011, 75, 264–267. [Google Scholar]
- Habibian, M.; Ghazi, S.; Moeini, M.M. Effects of dietary selenium and vitamin E on growth performance, meat yield, and selenium content and lipid oxidation of breast meat of broilers reared under heat stress. Biol. Trace Elem. Res. 2016, 169, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Yoon, I.; Werner, T.M.; Butler, J.M. Effect of source and concentration of selenium on growth performance and selenium retention in broiler chickens. Poult. Sci. 2007, 86, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Ramnath, V.; Rekha, P.; Sujatha, K. Amelioration of heat stress induced disturbances of antioxidant defense system in chicken by Brahma Rasayana. Evid. Based Complement. Altern. Med. 2008, 5, 77–84. [Google Scholar] [CrossRef] [PubMed]
| Ingredients % | Starter | Grower | Finisher |
|---|---|---|---|
| Wheat | 57.86 | 60.82 | 63.61 |
| Soybean meal 48% | 31.13 | 25.28 | 19.69 |
| Canola meal 38% | 4.00 | 6.00 | 8.00 |
| Soya oil | 3.21 | 4.55 | 5.68 |
| Dicalcium phosphate | 1.27 | 0.99 | 0.81 |
| Limestone | 1.12 | 1.04 | 0.96 |
| Sodium bicarbonate | 0.25 | 0.25 | 0.31 |
| Lysine | 0.25 | 0.23 | 0.20 |
| Celite | 0.20 | 0.20 | 0.20 |
| DL-methionine | 0.28 | 0.23 | 0.19 |
| Salt | 0.20 | 0.20 | 0.16 |
| Starter premix | 0.10 | 0.10 | 0.10 |
| Threonine | 0.11 | 0.09 | 0.06 |
| Axtra XB 201 † | 0.01 | 0.01 | 0.01 |
| Axtra Phy TPT 10,000 ‡ | 0.01 | 0.01 | 0.01 |
| Total | 100 | 100 | 100 |
| Calculate analysis | |||
| AME 1 (kcal/kg) | 2950.00 | 3100.00 | 3200.00 |
| Crude protein (%) | 22.19 | 20.54 | 19.16 |
| Sodium (%) | 0.16 | 0.16 | 0.16 |
| Fat (%) | 5.04 | 6.46 | 7.35 |
| Salt (%) | 0.27 | 0.28 | 0.24 |
| Calcium (%) | 0.96 | 0.87 | 0.79 |
| Chloride (%) | 0.23 | 0.23 | 0.20 |
| Lysine (%) | 1.37 | 1.23 | 1.10 |
| Methionine (%) | 0.61 | 0.54 | 0.48 |
| Starch (%) | 35.54 | 37.18 | 38.90 |
| p available | 0.48 | 0.44 | 2.00 |
| Temperature (TEMP) | Thermoneutral | Heat Stress | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antioxidants 2 (AOX) | Control | Supplemental | Control | Supplemental | p-Values | |||||||
| Betaine (g/kg) | 0 | 1.0 | 0 | 1.0 | 0 | 1.0 | 0 | 1.0 | SE 3 | TEMP | AOX | BET |
| Days and items * | ||||||||||||
| 0 to 21 days | ||||||||||||
| ADFI (g/d) | 59.2 | 56.7 | 54.4 | 60.1 | 62.0 | 58.4 | 57.4 | 55.3 | 2.82 | 0.64 | 0.12 | 0.66 |
| ADG (g/d) | 36.6 | 36.3 | 35.2 | 35.2 | 34.1 | 36.4 | 35.5 | 36.3 | 1.58 | 0.87 | 0.57 | 0.49 |
| FCR | 1.62 | 1.56 | 1.55 | 1.71 | 1.82 | 1.61 | 1.62 | 1.43 | 0.10 | 0.52 | 0.35 | 0.34 |
| 21 to 42 days | ||||||||||||
| ADFI (g/d) | 162 | 173 | 167 | 169 | 161 | 155 | 153 | 159 | 6.30 | 0.001 | 0.82 | 0.30 |
| ADG 4 (g/d) | 93.5 | 102 | 91.8 | 91.4 | 80.5 | 86.9 | 79.6 | 89.3 | 3.59 | <0.001 | 0.13 | 0.001 |
| FCR 5 | 1.74 | 1.69 | 1.83 | 1.86 | 1.99 | 1.80 | 1.93 | 1.78 | 0.09 | 0.04 | 0.36 | 0.04 |
| 0 to 42 days | ||||||||||||
| ADFI (g/d) | 111 | 115 | 111 | 115 | 111 | 109 | 105 | 107 | 3.92 | 0.01 | 0.45 | 0.49 |
| ADG 6 (g/d) | 65.0 | 69.3 | 63.5 | 63.3 | 57.6 | 61.6 | 57.5 | 62.8 | 1.90 | <0.001 | 0.09 | 0.001 |
| FCR 7 | 1.71 | 1.66 | 1.75 | 1.81 | 1.93 | 1.74 | 1.83 | 1.70 | 0.06 | 0.03 | 0.60 | 0.02 |
| Final weight (g) | 2769 | 2949 | 2704 | 2699 | 2457 | 2627 | 2454 | 2675 | 161.9 | <0.001 | 0.20 | 0.003 |
| Breast weight 8(g) | 598 | 746 | 612 | 584 | 500 | 587 | 498 | 575 | 66.2 | <0.001 | 0.17 | <0.001 |
| Fat weight 9 (g) | 31.9 | 38.3 | 42.7 | 35.2 | 28.9 | 34.0 | 37.2 | 33.7 | 9.31 | 0.13 | 0.10 | 0.95 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakeri, M.; Cottrell, J.J.; Wilkinson, S.; Ringuet, M.; Furness, J.B.; Dunshea, F.R. Betaine and Antioxidants Improve Growth Performance, Breast Muscle Development and Ameliorate Thermoregulatory Responses to Cyclic Heat Exposure in Broiler Chickens. Animals 2018, 8, 162. https://doi.org/10.3390/ani8100162
Shakeri M, Cottrell JJ, Wilkinson S, Ringuet M, Furness JB, Dunshea FR. Betaine and Antioxidants Improve Growth Performance, Breast Muscle Development and Ameliorate Thermoregulatory Responses to Cyclic Heat Exposure in Broiler Chickens. Animals. 2018; 8(10):162. https://doi.org/10.3390/ani8100162
Chicago/Turabian StyleShakeri, Majid, Jeremy James Cottrell, Stuart Wilkinson, Mitchell Ringuet, John Barton Furness, and Frank Rowland Dunshea. 2018. "Betaine and Antioxidants Improve Growth Performance, Breast Muscle Development and Ameliorate Thermoregulatory Responses to Cyclic Heat Exposure in Broiler Chickens" Animals 8, no. 10: 162. https://doi.org/10.3390/ani8100162
APA StyleShakeri, M., Cottrell, J. J., Wilkinson, S., Ringuet, M., Furness, J. B., & Dunshea, F. R. (2018). Betaine and Antioxidants Improve Growth Performance, Breast Muscle Development and Ameliorate Thermoregulatory Responses to Cyclic Heat Exposure in Broiler Chickens. Animals, 8(10), 162. https://doi.org/10.3390/ani8100162







