Relationships between Circulating Urea Concentrations and Endometrial Function in Postpartum Dairy Cows
Simple Summary
Abstract
1. Introduction
2. Experimental Section
2.1. Animals and Management
2.2. Blood Sampling and Metabolite Assays
2.3. Uterine Tissue Collection and RNA Isolation
2.4. Microarray, Correlation and Pathway Analysis
2.5. Histological Analysis
2.6. Primary Bovine Endometrial Cell Culture
2.7. Endometrial Cell Culture Validation
2.8. Cell Culture Treatment with Urea and LPS
2.9. Endometrial Cell Viability Assay
2.10. Quantitative Reverse Transcription PCR (qRT-PCR) Analysis and Data Normalization
| Gene Symbol | Primer Sequence 5′→3′ | Product Size (bp) * | GenBank Accession No. |
|---|---|---|---|
| RN18S1 | F: CGGCGACGACCCATTCGAAC | 99 | NR_036642.1 |
| R: AATCGAACCCTGATTCCCCGTC | |||
| S100A8 | F: TGCCATTAACTCCCTGATTGAC | 179 | NM_001113725.1 |
| R: TAATTCCACCATCCTGATTGAT | |||
| HSPA5 | F: GGTATTGAAACTGTGGGAGGTG | 119 | NM_001075148.1 |
| R: AAGGTGATTGTCTTTCGTCAGG | |||
| IGF1R | F: GATCCCGTGTTCTTCTACGTTC | 101 | NM_001244612.1 |
| R: AAGCCTCCCACTATCAACAGAA | |||
| IL17RB | F: AAAGCCACTTCCAGTCCTACAG | 179 | NM_001083467.1 |
| R: ACCGTCCTCATTCATATTTGC | |||
| BRINP1 | F: ACTGGAGCAATCAAGGTCACA | 173 | NM_001015669.1 |
| R: GCCGACTGGACGAACTTCT | |||
| CRISPLD2 | F: ACTGAAACGGACGACATGAAC | 175 | NM_001100299.1 |
| R: TGGACCCTTTACACTTGTCCTT |
2.11. Additional Data Analysis
3. Results
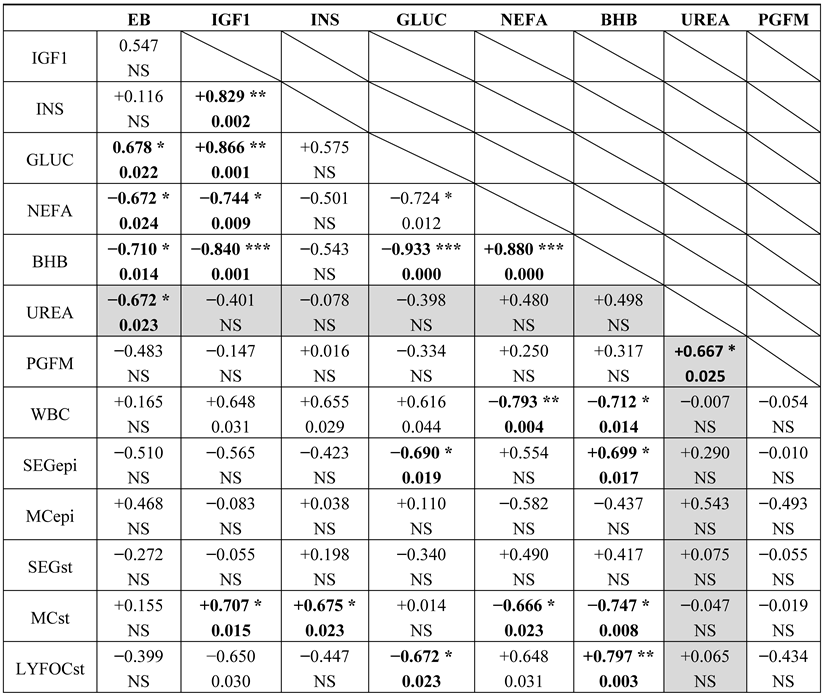
3.1. Study 1: in Vivo Microarray Experiment
| P Value | No. Genes | Disease Processes and Biological Functions |
|---|---|---|
| <0.001 | 32 | Endocrine system disorders |
| <0.001 | 28 | Haematological disease |
| <0.001 | 49 | Cellular function and maintenance |
| <0.001 | Cellular growth and proliferation | |
| Ratio a | Canonical Pathways | |
| 0.01 | 10/136 | Corticotrophin releasing hormone signalling |
| 0.016 | 5/38 | Coagulation system |
| 0.022 | 5/68 | Retinoic acid mediated apoptosis signalling |
| 0.026 | 10/131 | LXR/RXR Activation |
| Score | Focus Molecules | Network b |
| 43 | 29 | Endocrine system development, lipid metabolism, molecular transport |
| 40 | 27 | Cell-to-cell signaling and interaction, tissue development |
| 38 | 26 | Lipid metabolism, molecular transport, small molecule biochemistry |
| 38 | 26 | Cell-to-cell signaling and interaction, haematological system development and function, haematopoiesis |
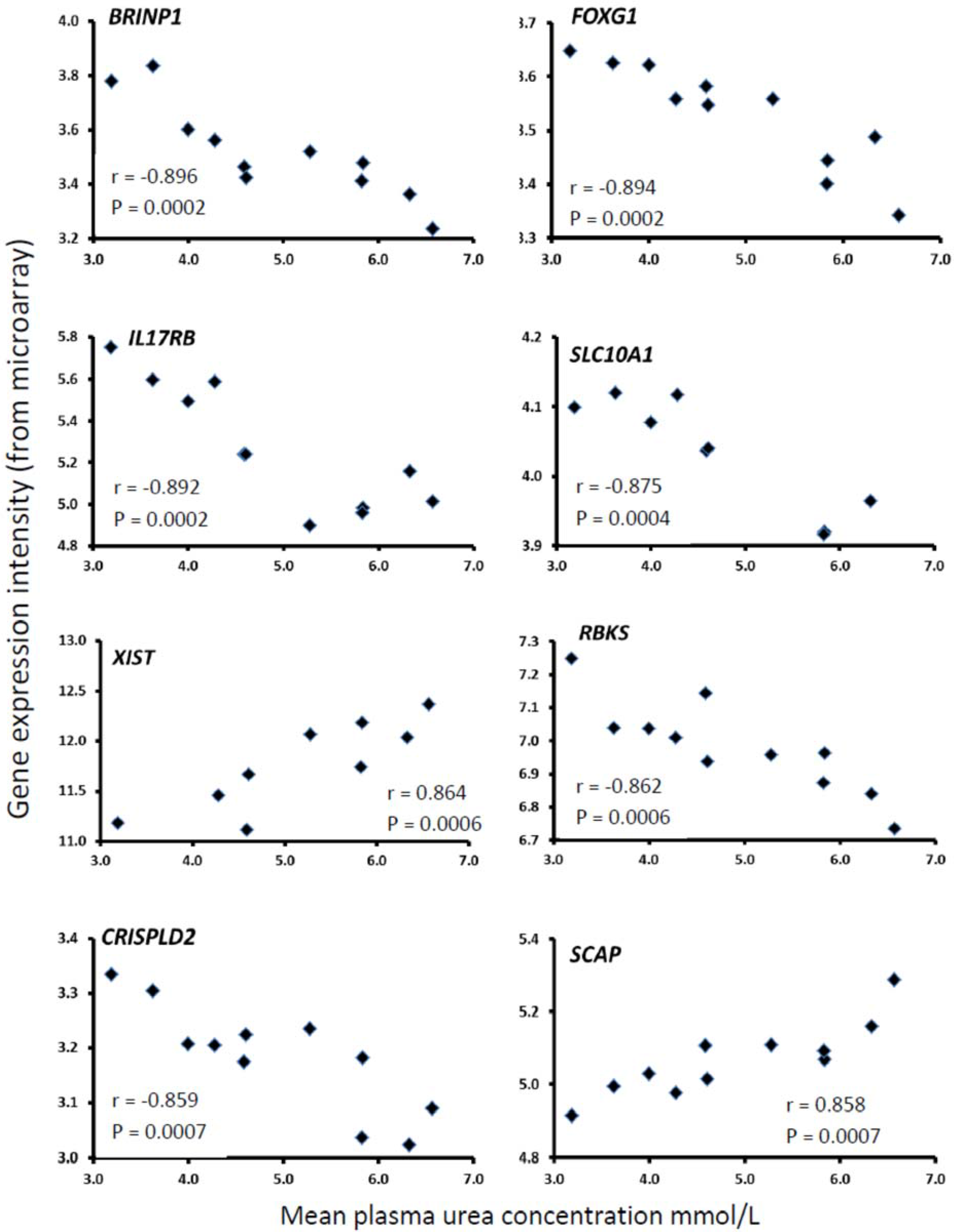
| Gene Symbol | Unigene ID | Gene Title | Entrez Gene | r | P |
|---|---|---|---|---|---|
| Top 10 Genes | |||||
| BRINP1 # | Bt.35142 | Bone morphogenetic protein/retinoic acid inducible neural-specific 1 | 538990 | −0.896 | 0.0002 |
| FOXG1 | Bt.66213 | Forkhead box G1 | 516947 | −0.894 | 0.0002 |
| IL17RB | Bt.24532 | Interleukin 17 receptor B | 533605 | −0.892 | 0.0002 |
| ACBD5 | Bt. 87957 | Acyl-coenzyme A binding domain containing 5 | 353160 | +0.883 | 0.0003 |
| SLC10A1 | Bt.51814 | Solute carrier family 10 (sodium/bile acid co-transporter family), member 1 | 532890 | −0.875 | 0.0004 |
| SRRM2 | Bt.22353 | Serine/arginine repetitive matrix 2 | 539515 | −0.869 | 0.0005 |
| XIST | --- | X (inactive)-specific transcript | 338325 | +0.864 | 0.0006 |
| RBKS | --- | Ribokinase | 513276 | −0.862 | 0.0006 |
| CRISPLD2 | Bt.55503 | Cysteine-rich secretory protein LCCL domain containing 2 | 505329 | −0.859 | 0.0007 |
| SCAP | Bt.18085 | SREBF chaperone | 507878 | +0.858 | 0.0007 |
| Additional Follow-up Genes Used in Study 2 | |||||
| S100A8 | Bt. 9360 | S100 calcium binding protein A8 | 616818 | +0.624 | 0.04 |
| HSPA5+ | Bt.65104 | Heat shock 70 kDa protein 5 (glucose-regulated protein, 78 kDa) | 415113 | +0.580 | 0.06 |
| IGF1R | Bt.12759 | Insulin-like growth factor 1 receptor | 281848 | +0.609 | 0.05 |
| Network | Molecules in Network |
|---|---|
| Endocrine system development, lipid metabolism, molecular transport | Alpha tubulin, ARPP19, ASCC2, CEBPB (includes EG:1051), CRTC1, CXCL5, DEFB1, FASTKD1, FASTKD5, HADHB, Ige, Interferon alpha, Lh, MATR3, MED17, MED22 (includes EG:20933), MED25 (includes EG:292889), MED26 (includes EG:306328), Mediator, MEIS1, MFSD2A, MLL2, NR3C1, PALB2, PCTP, POMC, RAD51AP1, RNA polymerase II, RNASE1, RPL12, SMC3, TMEM176A, TNS4, UBE2O, XIST |
| Cell-to-cell signaling and interaction, tissue development | Aconitase, ADAM9, APEH, CBR1, CDH16, CHMP3, CNKSR3, DOCK1, ELMO3, EPHA7, ERK1/2, FEZF1, FXN, HADH, HERC2, JAM, Laminin, Lfa-1, LGI1, LLGL2, Mac1, MLLT4, MVP, NAA15, PCBP2, RABAC1, Rap1, Rap, RAP1A, RAPGEF2, RASIP1, SNX15, TMEM176B, UBA5, VTA1 (includes EG:292640) |
| Lipid metabolism, molecular transport, small molecule biochemistry | ABCG4, AKR1C4, Angiotensin II receptor type 1, APOA5, C/ebp, CHST14, CLEC4G, CPN1, GALNT2, GC, HDL, HDL-cholesterol, HIVEP2, HNF1B, HSD11B1, IL17RB, IP6K2, IRAK1BP1, KPNA4, MAP4K2, N-cor, NCOR-LXR-Oxysterol-RXR-9 cis RA, NFkB (complex), NR1H2, OTUB2, PEPCK, PPP1R13L, RTKN, Rxr, SERPINF1, SERPINF2, SLC10A1, STMN2, TFG, ZFAND6 |
| Cell-to-cell signaling and interaction, haematological system development and function, haematopoiesis | ABI1, Akt, ASXL2, BATF, BBS5, BCOR, C1q, Complement component 1, Creatine Kinase, CREM, CYP19, Cytoplasmic Dynein, DYNLRB2, Fibrinogen, FKHR, FOXG1, GDF9, GRB10, HOXD10, MLLT1, MLLT10, MPDZ, NDEL1, NFIL3, PCGF1, PCM1, PHF1, PTX3, RAB40B, SERPING1, SPINK1, TDGF1, TFPI, Trypsin, TTC3 |
| Canonical Pathways | Molecules in Canonical Pathway |
| Corticotropin Releasing Hormone Signalling | ATF4, GNAS, GUCY2C, MAPK12, MEF2C, POMC, PRKCD, RAP1A, RAF1, VEGFA |
| Coagulation System | A2M, F3, PLAUR, SERPINF2, TFPI |
| Retinoic acid Mediated Apoptosis Signaling | CFLAR, IFNAR1, FADD, PARP10, TIPARP |
| LXR/RXR Activation | ABCG4, APOA5, C3, GC, HADH, IRF3, NR1H2, S100A8, SERPINF1, SERPINF2 |
3.2. Study 2: in Vitro Effects of Urea on Endometrial Gene Expression
| Gene | LPS | Urea (mmol/L) | COMB+ | |||
|---|---|---|---|---|---|---|
| 0 | 2.5 | 5.0 | 7.5 | |||
| S100A8 | - | 90 ± 34.8 | 90 ± 17.9 | 112 ± 47.8 | 86 ± 25.4 | 94 ± 15.2 |
| + | 545 ± 97.3 | 816 ± 242 | 795 ± 263 | 697 ± 225 | 713 ± 101 ** | |
| HSPA5 | - | 449 ± 110 | 547 ± 152 | 533 ± 210 | 517 ± 191 | 511 ± 76.7 |
| + | 533 ± 171 | 532 ± 192 | 523 ± 179 | 652 ± 285 | 560 ± 95.7 | |
| IGF1R | - | 40,686 ± 7122 | 51,365 ± 6018 | 40,512 ± 8384 | 47,800 ± 11,001 | 45,091 ± 3920 |
| + | 56,308 ± 12,006 | 41,976 ± 9441 | 40,360 ± 7702 | 46,944 ± 11,921 | 46,397 ± 4931 | |
| IL17RB | - | 1019 ± 471 | 1802 ± 283 | 2164 ± 923 | 2111 ± 337 | 1774 ± 278 |
| + | 3386 ± 898 | 1613 ± 795 | 1295 ± 505 | 2098 ± 486 | 2099 ± 373 | |
| BRINP1 | - | 334 ± 136 | 405 ± 142 | 289 ± 114 | 254 ± 119 | 320 ± 59.3 |
| + | 340 ± 141 | 370 ± 148 | 381 ± 202 | 224 ± 52.9 | 329 ± 67.3 | |
| CRISPLD2 | - | 3131 ± 1551 | 3403 ± 1470 | 3042 ± 1200 | 2418 ± 710 | 2998 ± 578 |
| + | 3261 ± 1511 | 2687 ± 1071 | 3390 ± 1105 | 1666 ± 359 | 2751 ± 519 | |
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bell, A.W. Regulation of organic nutrient metabolism during transition from late pregnancy to early lactation. J. Anim. Sci. 1995, 73, 2804–2819. [Google Scholar] [PubMed]
- Wathes, D.C.; Fenwick, M.; Cheng, Z.; Bourne, N.; Llewellyn, S.; Morris, D.G.; Kenny, D.; Murphy, J.; Fitzpatrick, R. Influence of negative energy balance on cyclicity and fertility in the high producing dairy cow. Theriogenology 2007, 68, S232–S241. [Google Scholar] [CrossRef] [PubMed]
- Bauman, D.E.; Currie, W.B. Partitioning of nutrients during pregnancy and lactation: A review of mechanisms involving homeostasis and homeorhesis. J. Dairy Sci. 1980, 63, 1514–1529. [Google Scholar] [CrossRef]
- Taylor, V.J.; Beever, D.E.; Wathes, D.C. Physiological Adaptations to Milk Production that Affect the Fertility of High Yielding Dairy Cows; British Society of Animal Science Occasional Publication No. 29; Nottingham University Press: Nottingham, UK, 2004; pp. 33–71. [Google Scholar]
- Wathes, D. Mechanisms linking metabolic status and disease with reproductive outcome in the dairy cow. Reprod. Dom. Anim. 2012, 47, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Leroy, J.L.; Vanholder, T.; van Knegsel, A.T.; Garcia-Ispierto, I.; Bols, P.E. Nutrient prioritization in dairy cows early postpartum: Mismatch between metabolism and fertility? Reprod. Domest. Anim. 2008, 43, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Laven, R.A.; Drew, S.B. Dietary protein and the reproductive performance of cows. Vet. Rec. 1999, 145, 687–695. [Google Scholar] [PubMed]
- Butler, W.R. Review: Effect of protein nutrition on ovarian and uterine physiology in dairy cattle. J. Dairy Sci. 1998, 81, 2533–2539. [Google Scholar] [CrossRef]
- Melendez, P.; Donovan, A.; Hernandez, J.; Barolome, J.; Risco, C.A.; Staples, C.; Thatcher, W.W. Milk, plasma, and blood urea nitrogen concentrations, dietary protein, and fertility in dairy cattle. J. Am. Vet. Med. Assoc. 2003, 223, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Wathes, D.C.; Bourne, N.; Cheng, Z.; Mann, G.E.; Taylor, V.J.; Coffey, M.P. Multiple correlation analyses of metabolic and endocrine profiles with fertility in primiparous and multiparous cows. J. Dairy Sci. 2007, 90, 1310–1325. [Google Scholar] [CrossRef]
- Van Saun, R.J. Metabolic profiles for evaluation of the transition period. Proc. Am. Assoc. Bov. Pract. 2006, 39, 130–138. [Google Scholar]
- Laven, R.A.; Scaramuzzi, R.J.; Wathes, D.C.; Peters, A.R.; Parkinson, T.J. Recent research on the effects of excess dietary nitrogen on the fertility of dairy cows. Vet. Rec. 2007, 160, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.E.; Cerri, R.L.; Sartori, R. Nutritional management of the donor cow. Theriogenology 2008, 69, 88–97. [Google Scholar] [CrossRef] [PubMed]
- O’Callahan, D.; Lozano, J.M.; Fahey, J.; Gath, V.; Snijders, S.; Boland, M.P. Relationships Between Nutrition and Fertility in Cattle; British Society of Animal Science Occasional Publication No. 26; Nottingham University Press: Edinburgh, UK, 2001; pp. 147–160. [Google Scholar]
- Moore, D.A.; Varga, G. BUN and MUN: Urea nitrogen testing in dairy cattle. Compendium 1996, 18, 712–720. [Google Scholar]
- Butler, W.R. Nutritional Effects on Resumption of Ovarian Cyclicity and Conception Rate in Postpartum Dairy Cows; British Society of Animal Science Occasional Publication 26; Nottingham University Press: Edinburgh, UK, 2001; pp. 133–145. [Google Scholar]
- Sinclair, K.D.; Kuran, M.; Gebbie, F.E.; Webb, R.; McEvoy, T.G. Nitrogen metabolism and fertility in cattle: II. Development of oocytes recovered from heifers offered diets differing in their rate of nitrogen release in the rumen. J. Anim. Sci. 2000, 78, 2670–2680. [Google Scholar] [PubMed]
- Rhoads, M.L.; Rhoads, R.P.; Gilbert, R.O.; Toole, R.; Butler, W.R. Detrimental effects of high plasma urea nitrogen levels on viability of embryos from lactating dairy cows. Anim. Reprod. Sci. 2006, 91, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Elrod, C.; van Amburgh, M.; Butler, W.R. Alterations of pH in response to increased dietary protein in cattle are unique to the uterus heifers. J. Anim. Sci. 1993, 71, 702–706. [Google Scholar] [PubMed]
- Rhoads, M.L.; Gilbert, R.O.; Lucy, M.C.; Butler, W.R. Effects of urea infusion on the uterine luminal environment of dairy cows. J. Dairy Sci. 2004, 87, 2896–2901. [Google Scholar] [CrossRef]
- McEvoy, T.G.; Robinson, J.J.; Aitken, R.P.; Findlay, P.A.; Robertson, I.S. Dietary excesses of urea influence the viability and metabolism of preimplantation sheep embryos and may affect fetal growth among survivors. Anim. Reprod. Sci. 1997, 47, 71–90. [Google Scholar] [CrossRef]
- Gunaretnam, I.; Pretheeban, T.; Rajamahendran, R. Effects of ammonia and urea in vitro on mRNA of candidate bovine endometrial genes. Anim. Reprod. Sci. 2013, 141, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Gier, H.T.; Marion, G.B. Uterus of the cow after parturition: Involutional changes. Am. J. Vet. Res. 1968, 29, 83–96. [Google Scholar] [PubMed]
- Leslie, K.E. The events of normal and abnormal postpartum endocrinology and uterine involution in dairy cows: A review. Can. Vet. J. 1983, 24, 67–71. [Google Scholar] [PubMed]
- Marion, G.B.; Gier, H.T. Histological and cytological changes in the bovine uterine epithelium. J. Anim. Sci. 1959, 18, 1552–1553. [Google Scholar]
- Llewellyn, S.; Fitzpatrick, R.; Kenny, D.A.; Patton, J.; Wathes, D.C. Endometrial expression of the insulin-like growth factor system during uterine involution in the postpartum dairy cow. Domest. Anim. Endocrinol. 2008, 34, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Wathes, D.C.; Cheng, Z.; Fenwick, M.A.; Fitzpatrick, R.; Patton, J. Influence of energy balance on the somatotrophic axis and matrix metalloproteinase expression in the endometrium of the postpartum dairy cow. Reproduction 2011, 141, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Földi, J.; Kulcsár, M.; Pécsi, A.; Huyghe, B.; de Sa, C.; Lohuis, J.A.; Cox, P.; Huszenicza, G. Bacterial complications of postpartum uterine involution in cattle. Anim. Reprod. Sci. 2006, 96, 265–281. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, I.M.; Cronin, J.; Goetze, L.; Donofrio, G.; Schuberth, H.J. Defining postpartum uterine disease and the mechanisms of infection and immunity in the female reproductive tract in cattle. Biol. Reprod. 2009, 81, 1025–1032. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.O.; Shin, S.T.; Guard, C.L.; Erb, H.N.; Frajblat, M. Prevalence of endometritis and its effects on reproductive performance of dairy cows. Theriogenology 2005, 64, 1879–1888. [Google Scholar] [CrossRef] [PubMed]
- Wathes, D.C.; Cheng, Z.; Chowdhury, W.; Fenwick, M.A.; Fitzpatrick, R.; Morris, D.G.; Patton, J.; Murphy, J.J. Negative energy balance alters global gene expression and immune responses in the uterus of postpartum dairy cows. Physiol. Genom. 2009, 39, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.O.; Messing, E.M.; Reeder, J.E. DBCCR1 mediates death in cultured bladder tumor cells. Oncogene 2004, 23, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Sung, S. Deleted in breast cancer 1 (DBC1) is a dynamically regulated protein. Neoplasma 2010, 57, 365–368. [Google Scholar] [PubMed]
- Lee, J.; Ho, W.H.; Maroka, M.; Corpuz, R.T.; Baldwin, D.T.; Foster, J.S.; Goddard, A.D.; Yansura, D.G.; Vandlen, R.L.; Wood, W.I.; et al. IL-17E, a novel proinflammatory ligand for the IL-17 receptor homolog IL-17Rh1. J. Biol. Chem. 2001, 276, 1660–1664. [Google Scholar] [CrossRef] [PubMed]
- Rickel, E.A.; Siegel, L.A.; Yoon, B.R.; Rottman, J.B.; Kugler, D.G.; Swart, D.A.; Anders, P.M.; Tocker, J.E.; Comeau, M.R.; Budelsky, A.L. Identification of functional roles for both IL-17RB and IL-17RA in mediating IL-25-induced activities. J. Immunol. 2008, 181, 4299–4310. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Xing, W.M.; Fan, H.H.; Wang, K.S.; Zhang, H.K.; Wang, Q.W.; Qi, J.; Yang, H.M.; Yang, J.; Ren, Y.N.; et al. The novel lipopolysaccharide-binding protein CRISPLD2 is a critical serum protein to regulate endotoxin function. J. Immunol. 2009, 183, 6646–6656. [Google Scholar] [CrossRef] [PubMed]
- Vásárhelyi, V.; Trexler, M.; Patthy, L. Both LCCL-domains of human CRISPLD2 have high affinity for lipid A. Biochimie 2014, 97, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Hendershot, L.M.; Valentine, V.A.; Lee, A.S.; Morris, S.W.; Shapiro, D.N. Localization of the gene encoding human BiP/GRP78, the endoplasmic reticulum cognate of the HSP70 family, to chromosome 9q34. Genomics 1994, 20, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Falahatpisheh, H.; Nanez, A.; Montoya-Durango, D.; Qian, Y.; Tiffany-Castiglioni, E.; Ramos, K.S. Activation profiles of HSPA5 during the glomerular mesangial cell stress response to chemical injury. Cell Stress Chaperones 2007, 12, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Guzel, E.; Basar, M.; Ocak, N.; Arici, A.; Kayisli, UA. Bidirectional interaction between unfolded-protein-response key protein HSPA5 and estrogen signaling in human endometrium. Biol. Reprod. 2011, 85, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Vandal, K.; Rouleau, P.; Boivin, A.; Ryckman, C.; Talbot, M.; Tessier, P.A. Blockade of S100A8 and S100A9 suppresses neutrophil migration in response to lipopolysaccharide. J. Immunol. 2003, 171, 2602–2609. [Google Scholar] [CrossRef] [PubMed]
- Swangchan-uthai, T.; Lavender, C.R.; Cheng, Z.; Fouladi-Nashta, A.A.; Wathes, D.C. Time course of defense mechanisms in bovine endometrium in response to lipopolysaccharide. Biol. Reprod. 2012, 87, 135. [Google Scholar] [CrossRef] [PubMed]
- Swangchan-Uthai, T.; Chen, Q.; Kirton, S.E.; Fenwick, M.A.; Cheng, Z.; Patton, J.; Fouladi-Nashta, A.A.; Wathes, D.C. Influence of energy balance on the antimicrobial peptides S100A8 and S100A9 in the endometrium of the post-partum dairy cow. Reproduction 2013, 145, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Patton, J.; Kenny, D.A.; Mee, J.F.; O’Mara, F.P.; Wathes, D.C.; Cook, M.; Murphy, J.J. Effect of milking frequency and diet on milk production, energy balance, and reproduction in dairy cows. J. Dairy Sci. 2006, 89, 1478–1487. [Google Scholar] [CrossRef]
- Jarrige, J. INRAtion V2.7: Microsoft Computer Program of Ration Formulation for Ruminant Livestock. In Logiciel de Rationnement des Ruminants; Agabriel, J., Champciaux, P., Espinasse, C., Eds.; CNERTA: Dijon, France, 1989. [Google Scholar]
- Fenwick, M.A.; Fitzpatrick, R.; Kenny, D.A.; Diskin, M.G.; Patton, J.; Murphy, J.J.; Wathes, D.C. Interrelationships between negative energy balance (NEB) and IGF regulation in liver of lactating dairy cows. Domest. Anim. Endocrinol. 2008, 34, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.S.; Pushpakumara, P.G.; Cheng, Z.; Peters, A.R.; Abayasekara, D.R.; Wathes, D.C. Effects of dietary polyunsaturated fatty acids on ovarian and uterine function in lactating dairy cows. Reproduction 2002, 124, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Bonnett, B.N.; Miller, R.B.; Etherington, W.G.; Martin, S.W.; Johnson, W.H. Endometrial biopsy in Holstein-Friesian dairy cows. I. Technique, histological criteria and results. Can. J. Vet. Res. 1991, 55, 155–161. [Google Scholar] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New capabilities and interfaces. Nucl. Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [PubMed]
- Louhelainen, J.P.; Hurst, C.D.; Pitt, E.; Nishiyama, H.; Pickett, H.A.; Knowles, M.A. DBC1 re-expression alters the expression of multiple components of the plasminogen pathway. Oncogene 2006, 25, 2409–2419. [Google Scholar] [CrossRef] [PubMed]
- Arden, K.C. FoxO: Linking new signaling pathways. Mol. Cell 2004, 14, 416–418. [Google Scholar] [CrossRef]
- Geyer, J.; Wilke, T.; Petzinger, E. The solute carrier family SLC10: More than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmiedebergs Arch. Pharmacol. 2006, 372, 413–431. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Lei, H.; Chen, Y.; Liu, Q.; Li, L.C.; Moorhead, J.F.; Varghese, Z.; Ruan, X.Z. Enhanced SCAP glycosylation by inflammation induces macrophage foam cell formation. PLoS ONE 2013, 8, e75650. [Google Scholar] [CrossRef] [PubMed]
- Eves, P.C.; Haycock, J.W. Melanocortin signalling mechanisms. Adv. Exp. Med. Biol. 2010, 681, 19–28. [Google Scholar] [PubMed]
- Delgado, M.; Ganea, D. Anti-inflammatory neuropeptides: A new class of endogenous immunoregulatory agents. Brain Behav. Immun. 2008, 22, 1146–1151. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.B.; Leoni, G.; Melendez, T.M.; Sampaio, A.L.; Perretti, M. Melanocortin control of cell trafficking in vascular inflammation. Adv. Exp. Med. Biol. 2010, 681, 88–106. [Google Scholar] [PubMed]
- Hammon, D.S.; Holyoak, G.R.; Dhiman, T.R. Association between blood plasma urea nitrogen levels and reproductive fluid urea nitrogen and ammonia concentrations in early lactation dairy cows. Anim. Reprod. Sci. 2005, 86, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.A.; Wills, J.R.; Kendall, N.R.; Green, M.J.; Muray, R.D.; Dobson, H. Energy metabolites in pre- and postpartum dairy cattle as predictors of reproductive disorders. Vet. Rec. 2011, 168, 562. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.D.; Galligan, D.T.; Blanchard, T.; Reeves, M. Serum urea nitrogen and conception rate: The usefulness of test information. J. Dairy Sci. 1993, 76, 3742–3746. [Google Scholar] [CrossRef]
- Butler, W.R.; Calaman, J.J.; Beam, S.W. Plasma and milk urea nitrogen in relation to pregnancy rate in lactating dairy cattle. J. Anim. Sci. 1996, 74, 858–865. [Google Scholar] [PubMed]
- Laven, R.A.; Biggadike, H.J.; Allison, R.D. The effect of pasture nitrate concentration and concentrate intake after turnout on embryo growth and viability in the lactating dairy cow. Reprod. Dom. Anim. 2002, 37, 111–115. [Google Scholar] [CrossRef]
- Ordonez, A.; Parkinson, T.J.; Matthew, C.; Holmes, C.W.; Miller, R.D.; Lopez-Villalobos, N.; Burke, J.; Brookes, I. Effects of application in spring of urea fertiliser on aspects of reproductive performance of pasture-fed dairy cows. N. Z. Vet. J. 2007, 55, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Law, R.A.; Young, F.J.; Patterson, D.C.; Kilpatrick, D.J.; Wylie, A.R.; Mayne, C.S. Effect of dietary protein content on the fertility of dairy cows during early and mid lactation. J. Dairy Sci. 2009, 92, 2737–2746. [Google Scholar] [CrossRef] [PubMed]
- Burg, M.B.; Ferraris, J.D.; Dmitrieva, N.I. Cellular response to hyperosmotic stresses. Physiol. Rev. 2007, 87, 1441–1474. [Google Scholar] [CrossRef] [PubMed]
- Maddock, A.L.; Westenfelder, C. Urea induces the heat shock response in human neuroblastoma cells. J. Am. Soc. Nephrol. 1996, 7, 275–282. [Google Scholar] [PubMed]
- Beck, F.X.; Grunbein, R.; Lugmayr, K.; Neuhofer, W. Heat shock proteins and the cellular response to osmotic stress. Cell Physiol. Biochem. 2000, 10, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Elrod, C.; Butler, W. Reduction of fertility and alteration of uterine pH in heifers fed excess ruminally degradable protein. J. Anim. Sci. 1993, 71, 694–701. [Google Scholar] [PubMed]
- Beltman, M.E.; Forde, N.; Furney, P.; Carter, F.; Roche, J.F.; Lonergan, P.; Crowe, M.A. Characterisation of endometrial gene expression and metabolic parameters in beef heifers yielding viable or non-viable embryos on Day 7 after insemination. Reprod. Fertil. Dev. 2010, 22, 987–999. [Google Scholar] [CrossRef] [PubMed]
- Rooke, J.A.; Ewen, M.; Mackie, K.; Staines, M.E.; Mcevoy, T.G.; Sinclair, K.D. Effect of ammonium chloride on the growth and metabolism of bovine ovarian granulosa cells and the development of ovine oocytes matured in the presence of bovine granulosa cells previously exposed to ammonium chloride. Anim. Reprod. Sci. 2004, 84, 53–71. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, S.J. Reproductive tract inflammatory disease in postpartum dairy cows. Animal 2014, 8, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.J.; Kim, S.H.; Heo, K.; Ou, C.Y.; Stallcup, M.R.; Kim, J.H. Reciprocal roles of DBC1 and SIRT1 in regulating estrogen receptor α activity and co-activator synergy. Nucl. Acids Res. 2011, 39, 6932–6943. [Google Scholar] [PubMed]
- Bekana, M.; Jonsson, P.; Kindahl, H. Intrauterine bacterial findings and hormonal profiles in post-partum cows with normal puerperium. Acta Vet. Scand. 1996, 37, 251–263. [Google Scholar] [PubMed]
- Bekana, M.; Odensvik, K.; Kindahl, H. Prostaglandin F2 alpha metabolite and progesterone profiles in post-partum cows with retained foetal membranes. Acta Vet. Scand. 1996, 37, 171–185. [Google Scholar] [PubMed]
- Singh, J.; Murray, R.D.; Mshelia, G.; Woldehiwet, Z. The immune status of the bovine uterus during the peripartum period. Vet. J. 2008, 175, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Wathes, D.C.; Clempson, A.M.; Pollott, G.E. Associations between lipid metabolism and fertility in the dairy cow. Reprod. Fertil. Dev. 2012, 25, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Bondurant, R.H. Inflammation in the bovine female reproductive tract. J. Anim. Sci. 1999, 77, 101–110. [Google Scholar] [PubMed]
- Dong, X.Y.; Tang, S.Q. Insulin-induced gene: A new regulator in lipid metabolism. Peptides 2010, 31, 2145–2150. [Google Scholar] [CrossRef] [PubMed]
- Ignatova, I.D.; Angdisen, J.; Moran, E.; Schulman, I.G. Differential regulation of gene expression by LXRs in response to macrophage cholesterol loading. Mol. Endocrinol. 2013, 27, 1036–1047. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Hylemon, P.B. Bile acids are nutrient signaling hormones. Steroids 2014, 86, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Sejersen, H.; Sørensen, M.T.; Larsen, T.; Bendixen, E.; Ingvartsen, K.L. Liver protein expression in dairy cows with high liver triglycerides in early lactation. J. Dairy Sci. 2012, 95, 2409–2421. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Z.; Oguejiofor, C.F.; Swangchan-Uthai, T.; Carr, S.; Wathes, D.C. Relationships between Circulating Urea Concentrations and Endometrial Function in Postpartum Dairy Cows. Animals 2015, 5, 748-773. https://doi.org/10.3390/ani5030382
Cheng Z, Oguejiofor CF, Swangchan-Uthai T, Carr S, Wathes DC. Relationships between Circulating Urea Concentrations and Endometrial Function in Postpartum Dairy Cows. Animals. 2015; 5(3):748-773. https://doi.org/10.3390/ani5030382
Chicago/Turabian StyleCheng, Zhangrui, Chike F. Oguejiofor, Theerawat Swangchan-Uthai, Susan Carr, and D. Claire Wathes. 2015. "Relationships between Circulating Urea Concentrations and Endometrial Function in Postpartum Dairy Cows" Animals 5, no. 3: 748-773. https://doi.org/10.3390/ani5030382
APA StyleCheng, Z., Oguejiofor, C. F., Swangchan-Uthai, T., Carr, S., & Wathes, D. C. (2015). Relationships between Circulating Urea Concentrations and Endometrial Function in Postpartum Dairy Cows. Animals, 5(3), 748-773. https://doi.org/10.3390/ani5030382