Effect of Different Levels of Extruded Coffee (Coffea arabica) Pulp Flour on the Productive Performance and Intestinal Morphometry of Cobb 500 Broiler Chickens
Simple Summary
Abstract
1. Introduction
2. Methodology
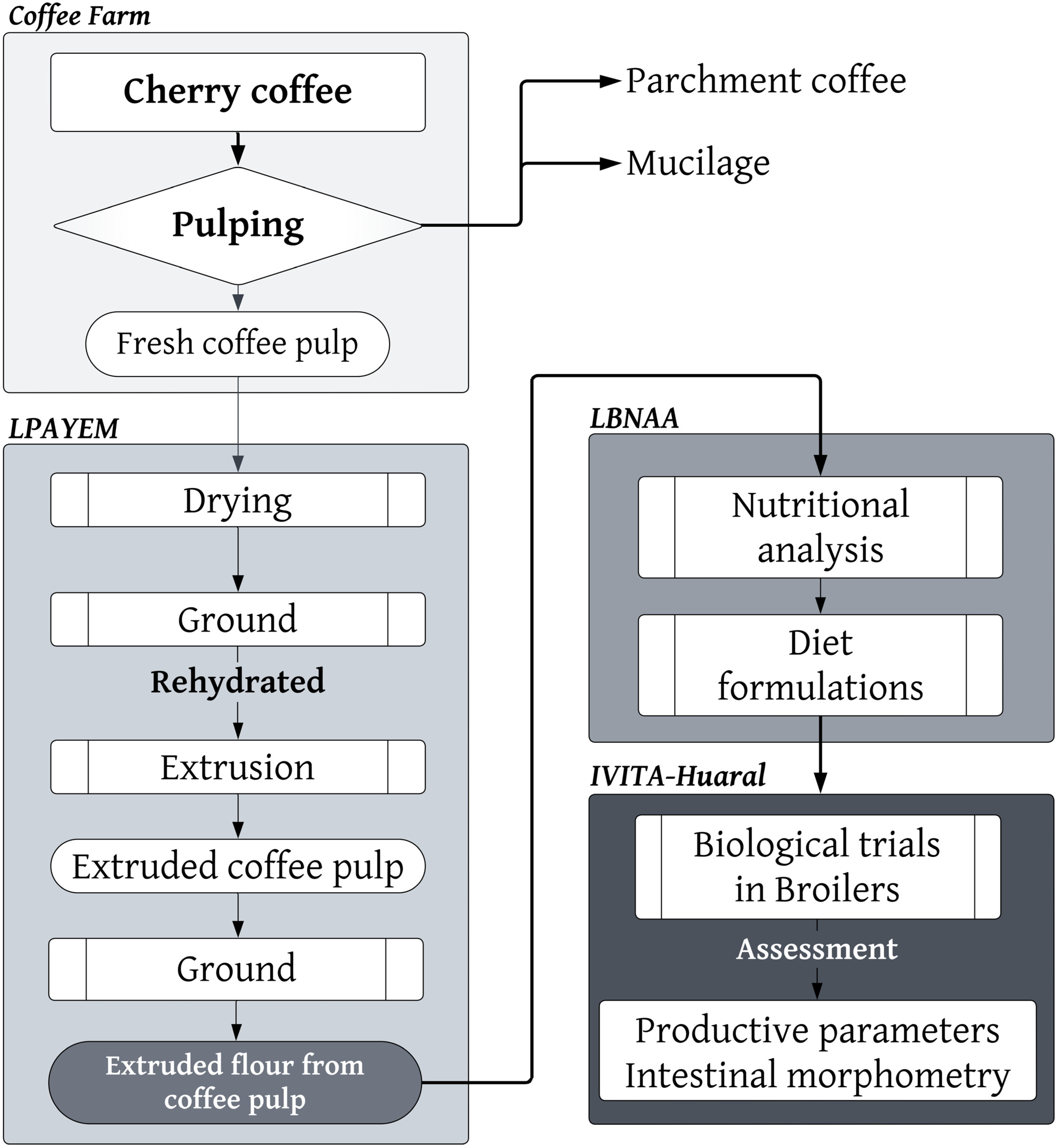
2.1. Area of Study
2.2. Extruded Coffee Pulp Flour (ECPF)
2.3. Diets
2.4. Biological Testing on Chickens
Variables Assessed
2.5. Statistical Analysis
3. Results
3.1. Feed Intake
3.2. Morphometric Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United States (FAO). FAOSTAT: Statistical Database; FAO: Rome, Italy, 2021; Available online: http://www.fao.org/faostat/en/#data/FBS (accessed on 20 October 2023).
- Díaz, C.; Willems, M. Línea de Base Del Sector Café En El Perú; Ministerio de Desarrollo Agrario y Riego. 2017. Available online: https://www.midagri.gob.pe/portal/pncafe-publicaciones/20118-linea-de-base-del-sector-cafe-en-el-peru (accessed on 20 October 2023).
- Blinová, L.; Sirotiak, M.; Bartošová, A.; Soldán, M. Review: Utilization of Waste From Coffee Production. Res. Pap. Fac. Mater. Sci. Technol. Slovak Univ. Technol. 2017, 25, 91–101. [Google Scholar] [CrossRef]
- Pandey, A.; Soccol, C.R.; Nigam, P.; Brand, D.; Mohan, R.; Roussos, S. Biotechnological potential of coffee pulp and coffee husk for bioprocesses. Biochem. Eng. J. 2000, 6, 153–162. [Google Scholar] [CrossRef]
- Montilla, J.; Arcila, J.; Aristizábal, M.; Montoya, E.C.; Puerta, G.I.; Oliveros, C.E.; Cadena, G. Caracterización de algunas propiedades físicas y factores de conversión del café durante el proceso de beneficio húmedo tradicional. Cenicafé 2008, 59, 120–142. [Google Scholar]
- Hernandez, C.E.; Chen, H.-H.; Chang, C.-I.; Huang, T.-C. Direct lipase-catalyzed lipophilization of chlorogenic acid from coffee pulp in supercritical carbon dioxide. Ind. Crop. Prod. 2009, 30, 359–365. [Google Scholar] [CrossRef]
- Torres-Valenzuela, L.S.; Martínez, K.G.; Serna-Jimenez, J.A.; Hernández, M.C. Drying of coffee pulp: Process parameters, mathematical model and its effect over physicochemical properties. Inf. Tecnol. 2019, 30, 189–200. [Google Scholar] [CrossRef]
- Serna-Jiménez, J.A.; Siles, J.A.; de los Ángeles Martín, M.; Chica, A.F. A Review on the Applications of Coffee Waste Derived from Primary Processing: Strategies for Revalorization. Processes 2022, 10, 2436. [Google Scholar] [CrossRef]
- Arpi, N.; Muzaifa, M.; Sulaiman, M.I.; Andini, R.; Kesuma, S.I. Chemical Characteristics of Cascara, Coffee Cherry Tea, Made of Various Coffee Pulp Treatments. In Proceedings of the IOP Conference Series: Earth and Environmental Science, 7th International Conference on Sustainable Agriculture, Food and Energy, Phuket, Thailand, 18–21 October 2019; Volume 709. [Google Scholar] [CrossRef]
- Arellano-González, M.A.; Ramírez-Coronel, M.A.; Torres-Mancera, M.T.; Pérez-Morales, G.G.; Saucedo-Castañeda, G. Antioxidant activity of fermented and nonfermented coffee (Coffea arabica) pulp extracts. Food Technol Biotechnol. 2011, 49, 374–378. [Google Scholar]
- Lin, Z.; Wei, J.; Hu, Y.; Pi, D.; Jiang, M.; Lang, T. Caffeine Synthesis and Its Mechanism and Application by Microbial Degradation, A Review. Foods 2023, 12, 2721. [Google Scholar] [CrossRef]
- Machado, M.; Santo, L.E.; Machado, S.; Lobo, J.C.; Costa, A.S.G.; Oliveira, M.B.P.P.; Ferreira, H.; Alves, R.C. Bioactive Potential and Chemical Composition of Coffee By-Products: From Pulp to Silverskin. Foods 2023, 12, 2354. [Google Scholar] [CrossRef]
- Lemos, M.F.; Salustriano, N.d.A.; Costa, M.M.d.S.; Lirio, K.; da Fonseca, A.F.A.; Pacheco, H.P.; Endringer, D.C.; Fronza, M.; Scherer, R. Chlorogenic acid and caffeine contents and anti-inflammatory and antioxidant activities of green beans of conilon and arabica coffees harvested with different degrees of maturation. J. Saudi Chem. Soc. 2022, 26, 101467. [Google Scholar] [CrossRef]
- Roshan, H.; Nikpayam, O.; Sedaghat, M.; Sohrab, G. Effects of green coffee extract supplementation on anthropometric indices, glycaemic control, blood pressure, lipid profile, insulin resistance and appetite in patients with the metabolic syndrome: A randomised clinical trial. Br. J. Nutr. 2018, 119, 250–258. [Google Scholar] [CrossRef]
- Cañas, S.; Rebollo-Hernanz, M.; Cano-Muñoz, P.; Aguilera, Y.; Benítez, V.; Braojos, C.; Gila-Díaz, A.; Rodríguez-Rodríguez, P.; Cobeta, I.M.; Pablo, Á.L.L.d.; et al. Critical Evaluation of Coffee Pulp as an Innovative Antioxidant Dietary Fiber Ingredient: Nutritional Value, Functional Properties, and Acute and Sub-Chronic Toxicity. Proceedings 2021, 70, 65. [Google Scholar] [CrossRef]
- Mazzafera, P. Degradation of caffeine by microorganisms and potential use of decaffeinated coffee husk and pulp in animal feeding. Sci. Agricola 2002, 59, 815–821. [Google Scholar] [CrossRef]
- Noriega Salazar, A.; Acuña, R.S.; García De Salcedo, M. Composición química de la pulpa de café a diferentes tiempos de ensilaje para su uso potencial en la alimentación animal. Zootec. Trop. 2009, 27, 135–141. Available online: http://ve.scielo.org/scielo.php?script=sci_arttext&pid=S0798-72692009000200004 (accessed on 23 October 2023).
- Rojas, J.U.; Verreth, J.; Amato, S.; Huisman, E. Biological treatments affect the chemical composition of coffee pulp. Bioresour. Technol. 2003, 89, 267–274. [Google Scholar] [CrossRef]
- Gualtieri, M.J.; Villalta, C.; Díaz, L.E.; Medina, G.; Lapenna, E.; Rondón, M.E. Producción de biomasa de Saccharomyces cerevisiae y Candida utilis usando residuos de pulpa de Coffea arabica L. Rev. Del Inst. Nac. Hig. Rafael Rangel 2007, 38, 31–37. [Google Scholar]
- Garcia, I.F.F.; Perez, J.R.O.; Teixeira, J.C.; Barbosa, C.M.P. Desempenho de cordeiros Texel x Bergamácia, Texel x Santa Inês e Santa Inês puros, terminados em confinamento, alimentados com casca de café como parte da dieta. Rev. Bras. Zootec. 2000, 29, 564–572. [Google Scholar] [CrossRef]
- Rios, T.S.; Torres, T.S.; Cerrilla, M.E.O.; Hernández, M.S.; Cruz, A.D.; Bautista, J.H.; Cuéllar, C.N.; Huerta, H.V. Changes in composition, antioxidant content, and antioxidant capacity of coffee pulp during the ensiling process. Rev. Bras. Zootec. 2014, 43, 492–498. [Google Scholar] [CrossRef]
- Salinas-Rios, T.; de Oaxaca, U.A.B.J.; Sánchez-Torres-Esqueda, M.T.; Díaz-Cruz, A.; Cordero-Mora, J.L.; Leon, M.C.; Hernández-Bautista, J.; Nava-Cuellar, C.; Aquino, R.N.; de Postgraduados, C.; et al. Oxidative status and fertility of ewes supplemented coffee pulp during estrous synchronization and early pregnancy. Rev. Colomb. Cienc. Pecu. 2016, 29, 255–263. [Google Scholar] [CrossRef]
- Hernández-Bautista, J.; Rodríguez-Magadán, H.M.; Villegas-Sánchez, J.A.; Salinas-Rios, T.; Ortiz-Muñoz, I.Y.; Aquino-Cleto, M.; Lozano-Trejo, S. Health status and productivity of sheep fed coffee pulp during fattening. Austral J. Veter-Sci. 2018, 50, 95–99. [Google Scholar] [CrossRef]
- Donkoh, A.; Atuahene, C.; Kese, A.; Mensah-Asante, B. The nutritional value of dried coffee pulp (DCP) in broiler chickens’ diets. Anim. Feed. Sci. Technol. 1988, 22, 139–146. [Google Scholar] [CrossRef]
- Gutiérrez-Prado, L.K.; Sánchez-Torres-Esqueda, M.T.; Salinas-Ríos, T.; Figueroa-Velasco, J.L.; Martínez-Aispuro, J.A.; Contreras-Caro-Del-Castillo, D.A. Coffee pulp supplementation prior to breeding improves oxidative status without affecting fertility of primiparous ewes. Rev. Colomb. Cienc. Pecu. 2019, 32, 261–273. [Google Scholar] [CrossRef]
- Bressani, R.; González, J. Evaluación de la pulpa de café como posible sustituto del maíz en raciones para pollos de carne. Inst. De Nutr. De Cent. América Y Panamá (INCAP) 1978, 28, 208–221. [Google Scholar]
- Yoplac, I.; Yalta, J.; Vásquez, H.V.; Maicelo, J.L. Efecto de la Alimentación con Pulpa de Café (Coffea arabica) en los Índices Productivos de Cuyes (Cavia porcellus L.) Raza Perú. Rev. Investig. Vet. Del Peru 2017, 28, 549–561. [Google Scholar] [CrossRef]
- Pinto-Ruiz, R.; Guevara-Hernández, F.; Medina, J.A.; Hernández-Sánchez, D.; Coss, A.L.-D.; Guerra-Medina, E. Conducta ingestiva y preferencia bovina por el ensilaje de Pennisetum y pulpa de café. Agron. Mesoam. 2016, 28, 59. [Google Scholar] [CrossRef][Green Version]
- Estrada-López, Y.; Avilés-Nova, I.; Estrada-Flores, F.; Pedraza-Beltrán, J.G.; Yong-Angel, P.E. Estimación del consumo de pasto estrella (Cynodon plectostachyus k. Schum.) por vacas lecheras en pastoreo mediante las técnicas de N-alcanos, diferencia en masa forrajera y comportamiento al pastoreo. Trop. Subtrop. Agroecosystems 2014, 17, 463–477. Available online: http://www.redalyc.org/articulo.oa?id=93935728006 (accessed on 19 September 2023).
- Noguera, R.; Posada, S. Efecto del método de secado sobre la digestibilidad in situ de la pulpa de café (Coffea arabica). Livest. Res. Rural. Dev. 2017, 29, 1–6. Available online: https://www.lrrd.org/lrrd29/8/ricn29154.html (accessed on 20 September 2022).
- Pedraza-Beltrán, P.; Estrada-Flores, J.G.; Martínez-Campos, A.R.; Estrada-López, I.; Rayas-Amor, A.A.; Yong-Angel, G.; Figueroa-Medina, M.; Nova, F.A.; Castelán-Ortega, O.A. On-farm evaluation of the effect of coffee pulp supplementation on milk yield and dry matter intake of dairy cows grazing tropical grasses in central Mexico. Trop. Anim. Health. Prod. 2011, 44, 329–336. [Google Scholar] [CrossRef]
- Bautista, E.O.; Pernía, J.; Barrueta, D.; Useche, M. Pulpa ecológica de café ensilada en la alimentación de alevines del híbrido cachamay (Colossoma macropomum × Piaractus brachypomus). Rev. Científica 2005, 15, 33–40. [Google Scholar]
- Rasowo, J.; Ochieng, R.S. Nutrient utilization and growth response of the Nile Tilapia (Oreochromis niloticus) fed on varying levels of dietary raw and boiled coffee pulp as a replacement for rice bran. Discov. Innov. 2005, 17, 110–116. [Google Scholar] [CrossRef]
- Ulloa, R.J.B.; Verreth, J.A. Growth, feed utilization and nutrient digestibility in tilapia fingerlings (Oreochromis aureus Steindachner) fed diets containing bacteria-treated coffee pulp. Aquac. Res. 2002, 33, 189–195. [Google Scholar] [CrossRef]
- Alam, M.S.; Kaur, J.; Khaira, H.; Gupta, K. Extrusion and Extruded Products: Changes in Quality Attributes as Affected by Extrusion Process Parameters: A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, 445–473. [Google Scholar] [CrossRef] [PubMed]
- Crawford, D.E.; Casaban, J. Recent Developments in Mechanochemical Materials Synthesis by Extrusion. Adv. Mater. 2016, 28, 5747–5754. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.; Aldrich, G. Extruders and Expanders in Pet Food, Aquatic and Livestock Feeds; Agrimedia: Fort Worth, TX, USA, 2007; Available online: https://books.google.com.pe/books/about/Extruders_and_Expanders_in_Pet_Food_Aqua.html?id=_3qDZwEACAAJ&redir_esc=y (accessed on 22 October 2023).
- Tran, Q.D.; Hendriks, W.H.; van der Poel, A.F. Effects of extrusion processing on nutrients in dry pet food. J. Sci. Food Agric. 2008, 88, 1487–1493. [Google Scholar] [CrossRef]
- Dalbhagat, C.G.; Mahato, D.K.; Mishra, H.N. Effect of extrusion processing on physicochemical, functional and nutritional characteristics of rice and rice-based products: A review. Trends Food Sci. Technol. 2019, 85, 226–240. [Google Scholar] [CrossRef]
- Choton, S.; Gupta, N.; Bandral, J.D.; Anjum, N.; Choudary, A. Extrusion technology and its application in food processing: A review. Pharma Innov. 2020, 9, 162–168. [Google Scholar] [CrossRef]
- Garcia-Amezquita, L.E.; Tejada-Ortigoza, V.; Pérez-Carrillo, E.; Serna-Saldívar, S.O.; Campanella, O.H.; Welti-Chanes, J. Functional and compositional changes of orange peel fiber thermally-treated in a twin extruder. LWT 2019, 111, 673–681. [Google Scholar] [CrossRef]
- Villasante, J.; Pérez-Carrillo, E.; Heredia-Olea, E.; Metón, I.; Almajano, M.P. In Vitro Antioxidant Activity Optimization of Nut Shell (Carya illinoinensis) by Extrusion Using Response Surface Methods. Biomolecules 2019, 9, 883. [Google Scholar] [CrossRef]
- Ministerio de Desarrollo Agrario y Riego (MIDAGRI). Producción y Comercialización de Productos Avícolas; MIDAGRI: Lima, Perú, 2023.
- Instituto Nacional de Defensa de la Competencia y de la Protección de la Propiedad Intelectual (INDECOPI). Menu Alternativo Guia de Productos Sustitutos; INDECOPI: Lima, Perú, 2021. Available online: https://www.indecopi.gob.pe/documents/51084/2681482/MenuAlternativo_ProductosSustitutos_Indecopi_2021/7f99fce9-935a-4434-34b6-9df7babb0c5b (accessed on 22 October 2023).
- Asociación Peruana de Avicultura (APA). Boletin Julio 2022; APA: Lima, Perú, 2022. [Google Scholar]
- Chirinos, A.; Rodríguez, G.; Bonomie, M.E. Integración vertical de la cadena de valor del sector avícola en el estado Zulia. Rev. Venez. Análisis Coyunt. 2008, 14, 175–193. [Google Scholar]
- Collatto, D.C.; Petrusch, A.; Lacerda, D.P.; Piran, F.A.S.; Mansilha, R.B. Autonomation in broiler production: A comparative analysis. Int. Food Agribus. Manag. Rev. 2019, 22, 47–64. [Google Scholar] [CrossRef]
- Lima, N.D.d.S.; Nääs, I.d.A.; Garcia, R.G.; de Moura, D.J. Environmental impact of Brazilian broiler production process: Evaluation using life cycle assessment. J. Clean. Prod. 2019, 237, 117752. [Google Scholar] [CrossRef]
- Mansilha, R.B.; Collatto, D.C.; Lacerda, D.P.; Morandi, M.I.W.M.; Piran, F.S. Environmental externalities in broiler production: An analysis based on system dynamics. J. Clean. Prod. 2019, 209, 190–199. [Google Scholar] [CrossRef]
- AOAC International. Block Digestion Method Using Cooper Catalyst and Steam Distillation into Boric Acid. AOAC Official method 201111. 2001. Available online: https://img.21food.cn/img/biaozhun/20100108/177/11285182.pdf (accessed on 24 October 2023).
- Latimer, G. Fiber. In Official Methods of Analysis of AOAC International; Wendt Thiex, N.J., Latimer, G.W., Jr., Eds.; Oxford University Press: New York, NY, USA, 2023; pp. C4-44–C4-54. [Google Scholar] [CrossRef]
- Carré, B.; Lessire, M.; Juin, H. Prediction of the net energy value of broiler diets. Animal 2014, 8, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- ISO 20481: 2008; Coffee and Coffee Products–Determination of the Caffeine Content Using High Performance Liquid Chromatography (HPLC)–Reference Method. 2011. Available online: https://www.iso.org/standard/34185.html (accessed on 25 October 2023).
- Wu, Y.; Zhou, Y.; Lu, C.; Ahmad, H.; Zhang, H.; He, J.; Zhang, L.; Wang, T. Influence of Butyrate Loaded Clinoptilolite Dietary Supplementation on Growth Performance, Development of Intestine and Antioxidant Capacity in Broiler Chickens. PLoS ONE 2016, 11, e0154410. [Google Scholar] [CrossRef] [PubMed]
- Megías, M.; Molist, P.; Pombal, M. Atlas de Histología Vegetal y Animal. 2017. Available online: http://mmegias.webs.uvigo.es/cita-celula.php (accessed on 21 October 2023).
- Vallejos, D.; Carcelén, F.; Jiménez, R.; Perales, R.; Santillán, G.; Ara, M.; Quevedo, W.; Carzola, F. Efecto de la Suplementación de Butirato de Sodio en la Dieta de Cuyes (Cavia porcellus) de Engorde sobre el Desarrollo de las Vellosidades Intestinales y Criptas de Lieberkühn. Rev. Investig. Vet. Del Peru 2015, 26, 395. [Google Scholar] [CrossRef]
- Sakamoto, K.; Hirose, H.; Onizuka, A.; Hayashi, M.; Futamura, N.; Kawamura, Y.; Ezaki, T. Quantitative Study of Changes in Intestinal Morphology and Mucus Gel on Total Parenteral Nutrition in Rats. J. Surg. Res. 2000, 94, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Farah, A. Coffee: Emerging health effects and disease prevention. In Coffee Constituents; Jhon Wiley and Sons, Inc.: Hoboken, NJ, USA; Institute of Food Technologists: Hull, IA, USA, 2012; Volume 1, pp. 22–58. [Google Scholar]
- Hosseini-Vashan, S.J.; Manochehri, D.; Afzali, N.; Naeimipour Yonesi, H. Green coffee powder: Effect on growth performance, biochemical blood indices, and intestine morphology in broiler chickens. Res Sq. 2023, 1, 1–10. [Google Scholar] [CrossRef]
- Londoño-Hernandez, L.; Ruiz, H.A.; Ramírez, T.C.; Ascacio, J.A.; Rodríguez-Herrera, R.; Aguilar, C.N. Fungal detoxification of coffee pulp by solid-state fermentation. Biocatal. Agric. Biotechnol. 2020, 23, 101467. [Google Scholar] [CrossRef]
- Svihus, B. Function of the digestive system. J. Appl. Poult. Res. 2014, 23, 306–314. [Google Scholar] [CrossRef]
- Awad, W.A.; Ghareeb, K.; Abdel-Raheem, S.; Böhm, J. Effects of dietary inclusion of probiotic and synbiotic on growth performance, organ weights, and intestinal histomorphology of broiler chickens. Poult. Sci. 2009, 88, 49–55. [Google Scholar] [CrossRef]
- Sharifian, M.; Hosseini-Vashan, S.; Nasri, M.F.; Perai, A. Pomegranate peel extract for broiler chickens under heat stress: Its influence on growth performance, carcass traits, blood metabolites, immunity, jejunal morphology, and meat quality. Livest. Sci. 2019, 227, 22–28. [Google Scholar] [CrossRef]
- Maliza, R.; Yatalaththov, F.G.; Setiawan, H.; Utami, L.B. The Effect of Coffee Arabica (Coffea arabica L.) Fruit Skin Extracts on Small Intestine Morphometry of mice (Mus musculus L.) with Ethanol-Induced. Bioscience 2021, 5, 21–31. [Google Scholar] [CrossRef]
- Choct, M. Managing gut health through nutrition. Br. Poult. Sci. 2009, 50, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Puente, J.; Carcelén, F.; Ara, M.; Santillán, G.; Perales, R. Effect of supplementation with increasing levels of probiotics on the histomorphometry of the small intestine of Guinea pig (Cavia porcellus). Rev. Investig. Vet. Del Peru 2019, 30, 624–633. [Google Scholar] [CrossRef]
- Singh, A.K.; Kim, W.K. Effects of Dietary Fiber on Nutrients Utilization and Gut Health of Poultry: A Review of Challenges and Opportunities. Animals 2021, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Bamba, T.; Sasaki, M. Physiological and Anti-Inflammatory Roles of Dietary Fiber and Butyrate in Intestinal Functions. J. Parenter. Enter. Nutr. 1999, 23, S70–S73. [Google Scholar] [CrossRef]
- Almeida, A.A.P.; Farah, A.; Silva, D.A.M.; Nunan, E.A.; Glória, M.B.A. Antibacterial Activity of Coffee Extracts and Selected Coffee Chemical Compounds against Enterobacteria. J. Agric. Food Chem. 2006, 54, 8738–8743. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.; Ramos, L.; Moreno, C.; Zúñiga-Paredes, J.; Carlosama-Yepez, M.; Ruales, P. Antimicrobial activity of plant-food by-products: A review focusing on the tropics. Livest. Sci. 2016, 189, 32–49. [Google Scholar] [CrossRef]
- Iriondo-DeHond, A.; Casas, A.R.; del Castillo, M.D. Interest of Coffee Melanoidins as Sustainable Healthier Food Ingredients. Front. Nutr. 2021, 8, 730343. [Google Scholar] [CrossRef]
- Nakayama, T.; Oishi, K. Influence of coffee (Coffea arabica) and galacto-oligosaccharide consumption on intestinal microbiota and the host responses. FEMS Microbiol. Lett. 2013, 343, 161–168. [Google Scholar] [CrossRef]
- Bhandarkar, N.S.; Mouatt, P.; Majzoub, M.E.; Thomas, T.; Brown, L.; Panchal, S.K. Coffee Pulp, a By-Product of Coffee Production, Modulates Gut Microbiota and Improves Metabolic Syndrome in High-Carbohydrate, High-Fat Diet-Fed Rats. Pathogens 2021, 10, 1369. [Google Scholar] [CrossRef] [PubMed]
- Amat, C.; Planas, J.M.; Moreto, M. Kinetics of hexose uptake by the small and large intestine of the chicken. Am. J. Physiol. Integr. Comp. Physiol. 1996, 271, R1085–R1089. [Google Scholar] [CrossRef] [PubMed]
- Lilburn, M.S.; Loeffler, S. Review Early intestinal growth and development in poultry. Poult. Sci. 2015, 94, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cheng, J.; Yuan, Y.; Luo, R.; Zhu, Z. Age-related intestinal monosaccharides transporters expression and villus surface area increase in broiler and layer chickens. J. Anim. Physiol. Anim. Nutr. 2019, 104, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Turesky, R.J.; Richoz, J.; Constable, A.; Curtis, K.D.; Dingley, K.H.; Turteltaub, K.W. The effects of coffee on enzymes involved in metabolism of the dietary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine in rats. Chem. Biol. Interact. 2003, 145, 251–265. [Google Scholar] [CrossRef] [PubMed]
- Olthof, M.R.; Hollman, P.C.H.; Katan, M.B. Chlorogenic Acid and Caffeic Acid Are Absorbed in Humans. J. Nutr. 2001, 131, 66–71. [Google Scholar] [CrossRef]
- Girard, M.; Bee, G. Invited review: Tannins as a potential alternative to antibiotics to prevent coliform diarrhea in weaned pigs. Animal 2020, 14, 95–107. [Google Scholar] [CrossRef]
- Cañas, S.; Rebollo-Hernanz, M.; Braojos, C.; Benítez, V.; Ferreras-Charro, R.; Dueñas, M.; Aguilera, Y.; Martín-Cabrejas, M.A. Understanding the Gastrointestinal Behavior of the Coffee Pulp Phenolic Compounds under Simulated Conditions. Antioxidants 2022, 11, 1818. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients 2021, 13, 273. [Google Scholar] [CrossRef]

| Ingredients, % | Treatments | ||||
|---|---|---|---|---|---|
| T1 | T2 | T3 | T4 | T5 | |
| Corn | 56.62 | 55.31 | 54.01 | 52.70 | 51.40 |
| Soybean meal | 23.24 | 22.30 | 21.36 | 20.41 | 19.47 |
| Flour whole soybean | 6.69 | 8.71 | 10.73 | 12.75 | 14.77 |
| Extruded coffee pulp flour | 0.00 | 1.50 | 3.00 | 4.50 | 6.00 |
| Wheat bran | 6.00 | 4.50 | 3.00 | 1.50 | 0.00 |
| Soybean oil | 4.21 | 4.44 | 4.67 | 4.90 | 5.14 |
| Diammonium phosphate | 1.25 | 1.27 | 1.28 | 1.29 | 1.30 |
| Calcium carbonate | 0.78 | 0.76 | 0.75 | 0.74 | 0.72 |
| NaCl | 0.29 | 0.30 | 0.30 | 0.30 | 0.30 |
| Methionine | 0.24 | 0.25 | 0.25 | 0.25 | 0.26 |
| Lysine-HCL | 0.18 | 0.17 | 0.17 | 0.16 | 0.16 |
| Vitamin and trace mineral premix | 0.12 | 0.12 | 0.12 | 0.12 | 0.12 |
| Sodium bicarbonate | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Choline chloride | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Mycotoxin sequestrant | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Threonine | 0.07 | 0.07 | 0.07 | 0.07 | 0.07 |
| Total | 100 | 100 | 100 | 100 | 100 |
| Nutrient | Unit | Extruded Coffee Pulp Flour (Replacement Levels %) | ||||
|---|---|---|---|---|---|---|
| T1:0 | T2:25 | T3:50 | T4:75 | T5:100 | ||
| Metabolizable energy | KJ | 12,866 | 12,866 | 12,866 | 12,866 | 12,866 |
| Crude protein | % | 18.68 | 18.85 | 19.02 | 19.18 | 19.35 |
| Crude fiber | % | 3.50 | 3.50 | 3.50 | 3.50 | 3.50 |
| Calcium | % | 0.73 | 0.73 | 0.73 | 0.73 | 0.73 |
| Phosphorus available | % | 0.37 | 0.37 | 0.37 | 0.37 | 0.37 |
| Calcium/phosphorus | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 | |
| Sodium | % | 0.16 | 0.16 | 0.16 | 0.16 | 0.16 |
| Chlorine | % | 0.27 | 0.27 | 0.26 | 0.26 | 0.26 |
| Electrolyte balance | mEq/K | −5.80 | −5.30 | −4.90 | −4.40 | −4.00 |
| Digestible lysine | % | 1.01 | 1.01 | 1.01 | 1.01 | 1.01 |
| Digestible methionine | % | 0.51 | 0.52 | 0.52 | 0.52 | 0.52 |
| Digestible methionine + cysteine | % | 0.78 | 0.78 | 0.78 | 0.78 | 0.78 |
| Digestible threonine | % | 0.68 | 0.68 | 0.68 | 0.68 | 0.68 |
| Digestible tryptophan | % | 0.19 | 0.19 | 0.19 | 0.19 | 0.19 |
| Digestible arginine | % | 1.12 | 1.12 | 1.12 | 1.12 | 1.12 |
| Digestible valine | % | 0.78 | 0.78 | 0.78 | 0.78 | 0.78 |
| Linoleic acid | % | 4.28 | 4.54 | 4.80 | 5.07 | 5.33 |
| Choline | ppm | 520.74 | 520.74 | 520.74 | 520.74 | 520.74 |
| Phytic phosphorus | % | 0.28 | 0.27 | 0.25 | 0.24 | 0.23 |
| Extruded Coffee Pulp Flour (Replacement Levels %) | ANOVA p-Value | |||||
|---|---|---|---|---|---|---|
| T1:0 | T2:25 | T3:50 | T4:75 | T5:100 | ||
| Feed Intake (g) | 1320.00 a | 1320.00 a | 1290.00 a | 1209.00 b | 1151.00 c | <0.001 |
| Weight gain (g) | 809.80 ab | 834.61 a | 784.26 ab | 761.13 b | 662.15 c | <0.001 |
| Feed conversion ratio | 1.64 ab | 1.58 b | 1.65 ab | 1.59 b | 1.75 a | 0.0831 |
| Parameter | Extruded Coffee Pulp Flour (Replacement Levels %) | ANOVA | ||||
|---|---|---|---|---|---|---|
| T1:0 | T2:25 | T3:50 | T4:75 | T5:100 | p-Value | |
| Duodenum | ||||||
| VL | 1591.88 a | 1544.83 a | 1437.01 a | 1530.84 a | 1538.54 a | 0.55 |
| VW | 137.30 b | 136.22 b | 124.35 b | 136.92 b | 172.18 a | 0.0295 |
| CD | 173.77 c | 193.95 bc | 197.89 bc | 234.59 a | 227.18 ab | < 0.001 |
| Ratio V/C | 9.41 a | 8.17 ab | 7.25 b | 6.68 b | 6.84 b | < 0.001 |
| VSA (μm2) | 0.44 a | 0.40 a | 0.41 a | 0.38 a | 0.45 a | 0.19 |
| Jejunum | ||||||
| VL | 1380.64 a | 1321.42 ab | 1415.64 a | 1264.97 ab | 1191.04 b | 0.0279 |
| VW | 133.75 a | 161.79 a | 175.62 a | 145.88 a | 132.30 a | 0.699 |
| CD | 158.38 b | 199.51 a | 199.13 a | 218.34 a | 197.09 a | 0.0126 |
| Ratio V/C | 8.79 a | 6.85 b | 7.17 b | 5.90 b | 6.07 b | < 0.001 |
| VSA (μm2) | 0.58 a | 0.67 a | 0.76 a | 0.58 a | 0.50 a | 0.21 |
| Ileum | ||||||
| VL | 952.67 a | 869.71 a | 932.79 a | 916.65 a | 895.83 a | 0.692 |
| VW | 146.77 a | 149.57 a | 142.25 a | 130.73 a | 158.88 a | 0.896 |
| CD | 144.21 c | 185.02 b | 186.69 b | 228.56 a | 212.09 ab | < 0.001 |
| Ratio V/C | 6.89 a | 4.94 b | 5.06 b | 4.19 b | 4.24 b | 0.0028 |
| VSA (μm2) | 0.69 a | 0.67 a | 0.56 a | 0.66 a | 0.84 a | 0.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antúnez, S.; Fuentes, N.; Gutierrez, M.; Carcelén, F.; Trillo, F.; López, S.; Bezada, S.; Rivadeneira, V.; Pizarro, S.; Nuñez, J. Effect of Different Levels of Extruded Coffee (Coffea arabica) Pulp Flour on the Productive Performance and Intestinal Morphometry of Cobb 500 Broiler Chickens. Animals 2024, 14, 1170. https://doi.org/10.3390/ani14081170
Antúnez S, Fuentes N, Gutierrez M, Carcelén F, Trillo F, López S, Bezada S, Rivadeneira V, Pizarro S, Nuñez J. Effect of Different Levels of Extruded Coffee (Coffea arabica) Pulp Flour on the Productive Performance and Intestinal Morphometry of Cobb 500 Broiler Chickens. Animals. 2024; 14(8):1170. https://doi.org/10.3390/ani14081170
Chicago/Turabian StyleAntúnez, Steven, Nadia Fuentes, Marco Gutierrez, Fernando Carcelén, Fritz Trillo, Sofía López, Sandra Bezada, Virginia Rivadeneira, Samuel Pizarro, and Jimny Nuñez. 2024. "Effect of Different Levels of Extruded Coffee (Coffea arabica) Pulp Flour on the Productive Performance and Intestinal Morphometry of Cobb 500 Broiler Chickens" Animals 14, no. 8: 1170. https://doi.org/10.3390/ani14081170
APA StyleAntúnez, S., Fuentes, N., Gutierrez, M., Carcelén, F., Trillo, F., López, S., Bezada, S., Rivadeneira, V., Pizarro, S., & Nuñez, J. (2024). Effect of Different Levels of Extruded Coffee (Coffea arabica) Pulp Flour on the Productive Performance and Intestinal Morphometry of Cobb 500 Broiler Chickens. Animals, 14(8), 1170. https://doi.org/10.3390/ani14081170







