The Spread of Peste Des Petits Ruminants Virus Lineage IV in West Africa
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
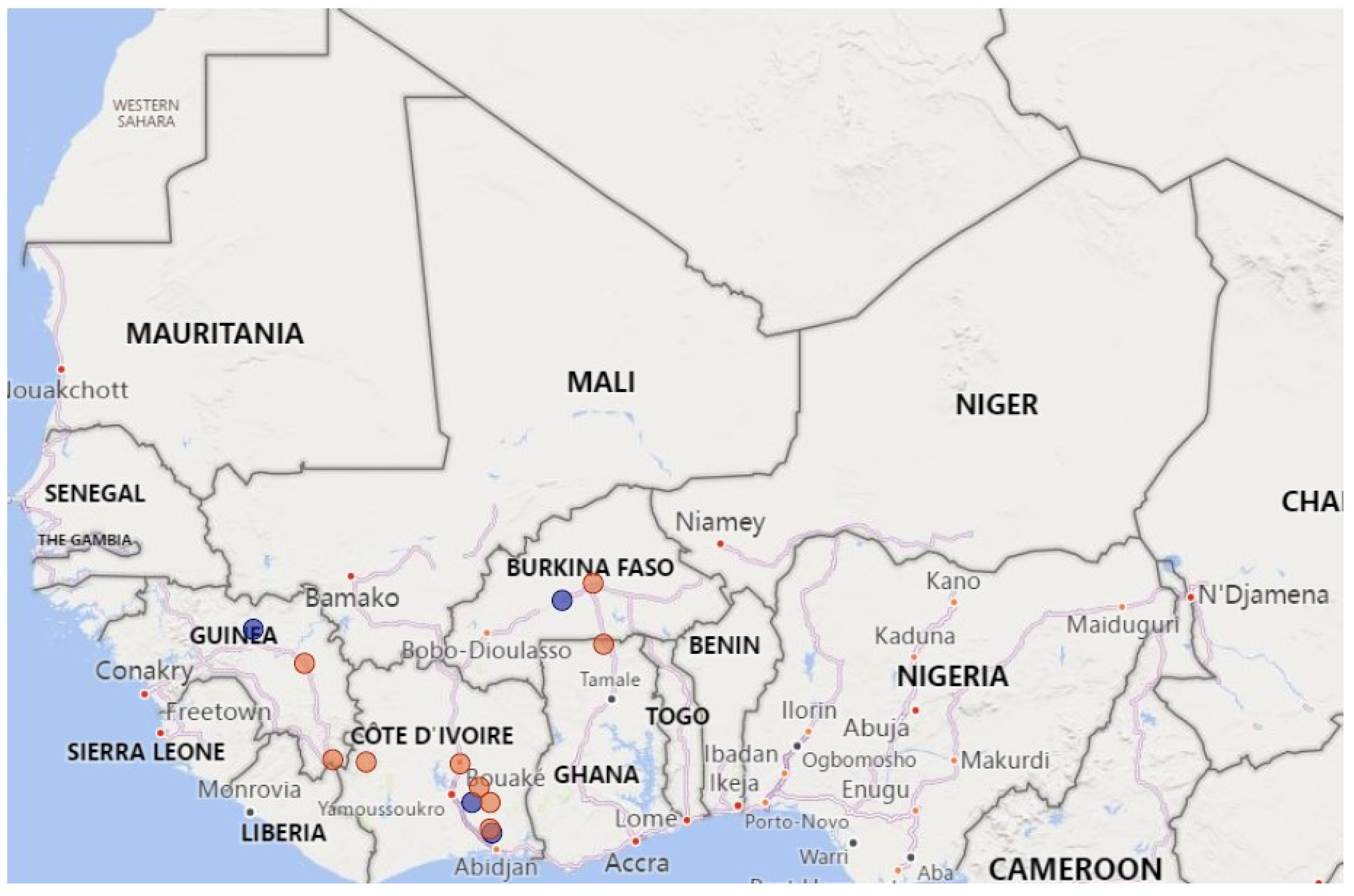
2.1. Sample Description and Processing
2.2. PCR Screening
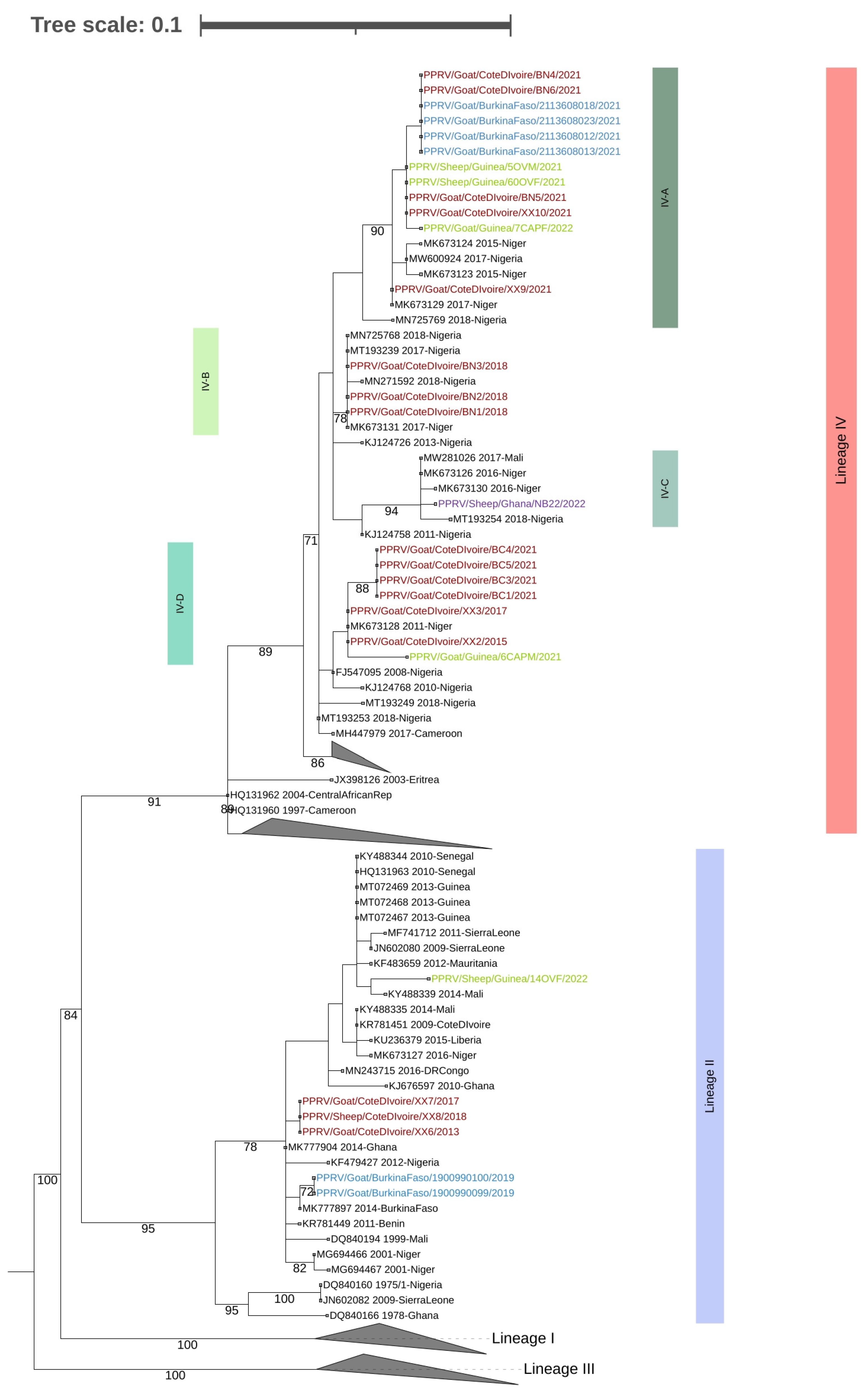
2.3. Sequencing and Phlyogenetic Analysis
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO OIE/WOAH. Peste des Petits Ruminants. Global Eradication Programme. Contributing to Food Security, Poverty Alleviation and Resilience. Five Years 2017–2021. 2016. Available online: http://www.fao.org/3/a-i6316e.pdf (accessed on 29 March 2023).
- Amarasinghe, G.K.; Bào, Y.; Basler, C.F.; Bavari, S.; Beer, M.; Bejerman, N.; Blasdell, K.R.; Bochnowski, A.; Briese, T.; Bukreyev, A.; et al. Taxonomy of the order Mononegavirales: Update. Arch. Virol. 2017, 162, 2493–2504. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, M.A.; Barrett, T. Evaluation of polymerase chain reaction for the detection and characterisation of rinderpest and peste des petits ruminants viruses for epidemiological studies. Virus Res. 1995, 39, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Couacy-Hymann, E.; Roger, F.; Hurard, C.; Guillou, J.P.; Libeau, G.; Diallo, A. Rapid and sensitive detection of peste des petits ruminants virus by a polymerase chain reaction assay. J. Virol. Methods 2002, 100, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Dundon, W.G.; Diallo, A.; Cattoli, G. Peste des petits ruminants in Africa: A review of currently available molecular epidemiological data, 2020. Arch. Virol. 2020, 165, 2147–2163. [Google Scholar] [CrossRef] [PubMed]
- Gargadennec, L.; Lalanne, A. Peste des petits ruminants. Bull. Serv. Zootech Epizoot Afr. Occident Fr. 1942, 5, 16–21. (In French) [Google Scholar]
- Woma, T.Y.; Adombi, C.M.; Yu, D.; Qasim, A.M.; Sabi, A.A.; Maurice, N.A.; Olaiya, O.D.; Loitsch, A.; Bailey, D.; Shamaki, D.; et al. Co-circulation of Peste-des-Petits-Ruminants Virus Asian lineage IV with Lineage II in Nigeria. Transbound. Emerg. Dis. 2016, 63, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Tounkara, K.; Bataille, A.; Adombi, C.M.; Maikano, I.; Djibo, G.; Settypalli, T.B.K.; Loitsch, A.; Diallo, A.; Libeau, G. First genetic characterization of Peste des Petits Ruminants from Niger: On the advancing front of the Asian virus lineage. Transbound Emerg. Dis. 2018, 65, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Souley, M.M.; Issa Ibrahim, A.; Sidikou, D.; Dundon, W.G.; Cattoli, G.; Abdou, A.; Soumana, F.; Yaou, B. Molecular epidemiology of peste des petits ruminants in Niger: An update. Transbound. Emerg. Dis. 2020, 67, 1388–1392. [Google Scholar] [CrossRef]
- Mantip, S.; Sigismeau, A.; Shamaki, D.; Woma, T.Y.; Kwiatek, O.; Libeau, G.; Farougou, S.; Bataille, A. Molecular epidemiology of peste des petits ruminants virus in Nigeria: An update. Transbound. Emerg. Dis. 2022, 69, 1634–1640. [Google Scholar] [CrossRef]
- Dundon, W.G.; Adombi, C.; Waqas, A.; Otsyina, H.R.; Arthur, C.T.; Silber, R.; Loitsch, A.; Diallo, A. Full genome sequence of a peste des petits ruminants virus (PPRV) from Ghana. Virus Genes 2014, 49, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Chard, L.S.; Bailey, D.S.; Dash, P.; Banyard, A.C.; Barrett, T. Full genome sequences of two virulent strains of peste-des-petits ruminants virus, the Côte d’Ivoire 1989 and Nigeria 1976 strains. Virus Res. 2008, 136, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Tounkara, K.; Kwiatek, O.; Niang, M.; Abou Kounta Sidibe, C.; Sery, A.; Dakouo, M.; Salami, H.; Lo, M.M.; Ba, A.; Diop, M.; et al. Genetic Evidence for Transboundary Circulation of Peste Des Petits Ruminants Across West Africa. Front Vet Sci. 2019, 6, 275. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA 6: Molecular Evolution Genetics Analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree of Life (ITOL) v5: An Online Tool for Phylogenetic Tree Display and Annotation. Nucleic Acids Res. 2021, 49, W293–W296. [Google Scholar] [CrossRef] [PubMed]
- Apolloni, A.; Corniaux, C.; Coste, C.; Lancelot, R.; Touré, I. Livestock Mobility in West Africa and Sahel and Transboundary Animal Diseases. In Transboundary Animal Diseases in Sahelian Africa and Connected Regions; Kardjadj, M., Diallo, A., Lancelot, R., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Couacy-Hymann, E.; Berete, K.; Odoom, T.; Zerbo, L.H.; Mathurin, K.Y.; Kouakou, V.K.; Doumbouya, M.I.; Balde, A.; Ababio, P.T.; Ouoba, L.B.; et al. The Spread of Peste Des Petits Ruminants Virus Lineage IV in West Africa. Animals 2023, 13, 1268. https://doi.org/10.3390/ani13071268
Couacy-Hymann E, Berete K, Odoom T, Zerbo LH, Mathurin KY, Kouakou VK, Doumbouya MI, Balde A, Ababio PT, Ouoba LB, et al. The Spread of Peste Des Petits Ruminants Virus Lineage IV in West Africa. Animals. 2023; 13(7):1268. https://doi.org/10.3390/ani13071268
Chicago/Turabian StyleCouacy-Hymann, Emmanuel, Kouramoudou Berete, Theophilus Odoom, Lamouni Habibata Zerbo, Koffi Yao Mathurin, Valère Kouame Kouakou, Mohamed Idriss Doumbouya, Aminata Balde, Patrick Tetteh Ababio, Lalidia Bruno Ouoba, and et al. 2023. "The Spread of Peste Des Petits Ruminants Virus Lineage IV in West Africa" Animals 13, no. 7: 1268. https://doi.org/10.3390/ani13071268
APA StyleCouacy-Hymann, E., Berete, K., Odoom, T., Zerbo, L. H., Mathurin, K. Y., Kouakou, V. K., Doumbouya, M. I., Balde, A., Ababio, P. T., Ouoba, L. B., Guigma, D., Drobo, A. D.-t., Guitti, M., Johnson, S. A. M., Blavo, D. L. M., Cattoli, G., Lamien, C. E., & Dundon, W. G. (2023). The Spread of Peste Des Petits Ruminants Virus Lineage IV in West Africa. Animals, 13(7), 1268. https://doi.org/10.3390/ani13071268





