Effects of the Vitamin D3 on Alleviating the Oxidative Stress Induced by Diquat in Wenchang Chickens
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Growth Performance
- ADFI = the total feed intake/(trial days × number of test animals)
- ADG = (final weight − initial weight)/measured days
- F/G = total feed consumption/(final weight − initial weight)
2.3. Sample Collection and Preparation
2.4. Determination of the Slaughter Performance and Organ Index
2.5. Meat Quality
2.6. Detection of the Biomarker in the Serum
2.7. Determination of Antioxidative Enzymes in the Serum, Liver, and Jejunum
2.8. Detection of the Inflammatory Cytokines in Serum
2.9. Evaluation of Morphology in the Duodenum, Jejunum, Ileum, and Liver Tissues
2.10. Determination of the Apoptotic Level of the Jejunum Epithelium
2.11. Detection of the Expression Level of Polysaccharides in the Jejunum Tissue
2.12. Statistical Analysis
3. Results
3.1. Effect of VD3 Addition on Growth Performance
3.2. Effect of VD3 Addition on Slaughter Performance and Organ Index
3.3. Effect of VD3 Addition on Meat Quality
3.4. Effect of VD3 Addition on the Intestines
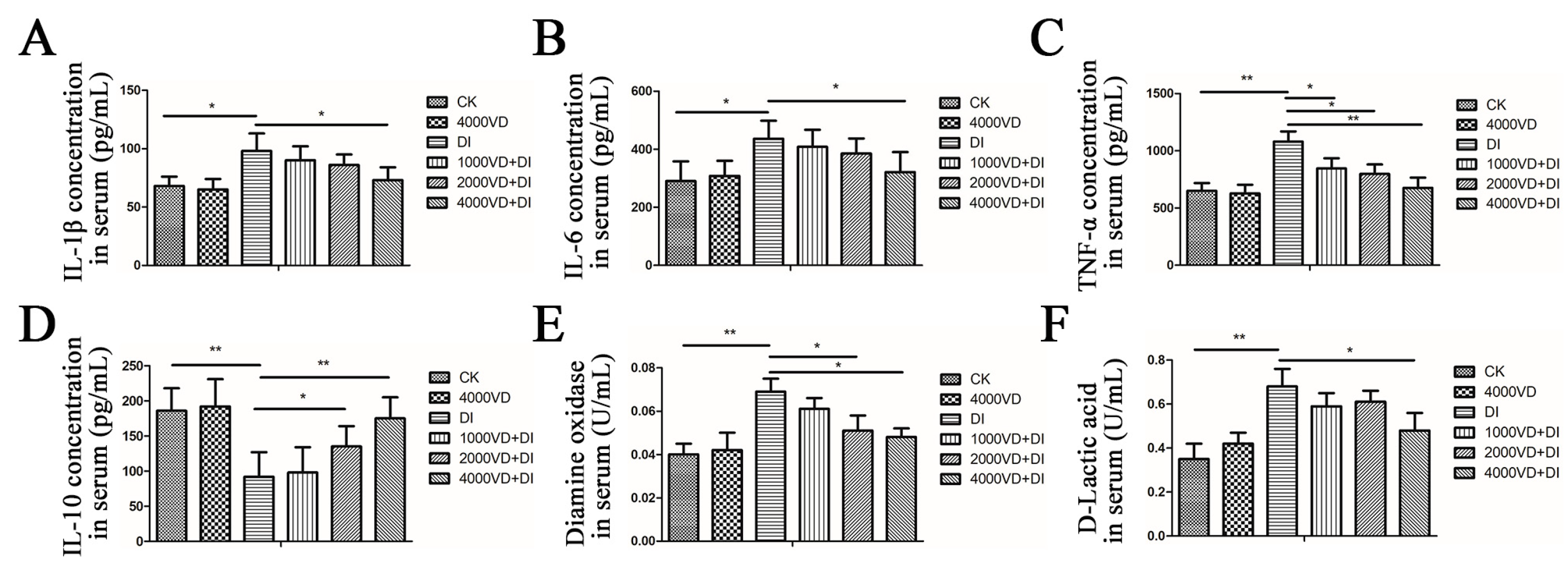
3.4.1. Effect of VD3 Addition on the Secretion of Inflammatory Cytokines in Serum
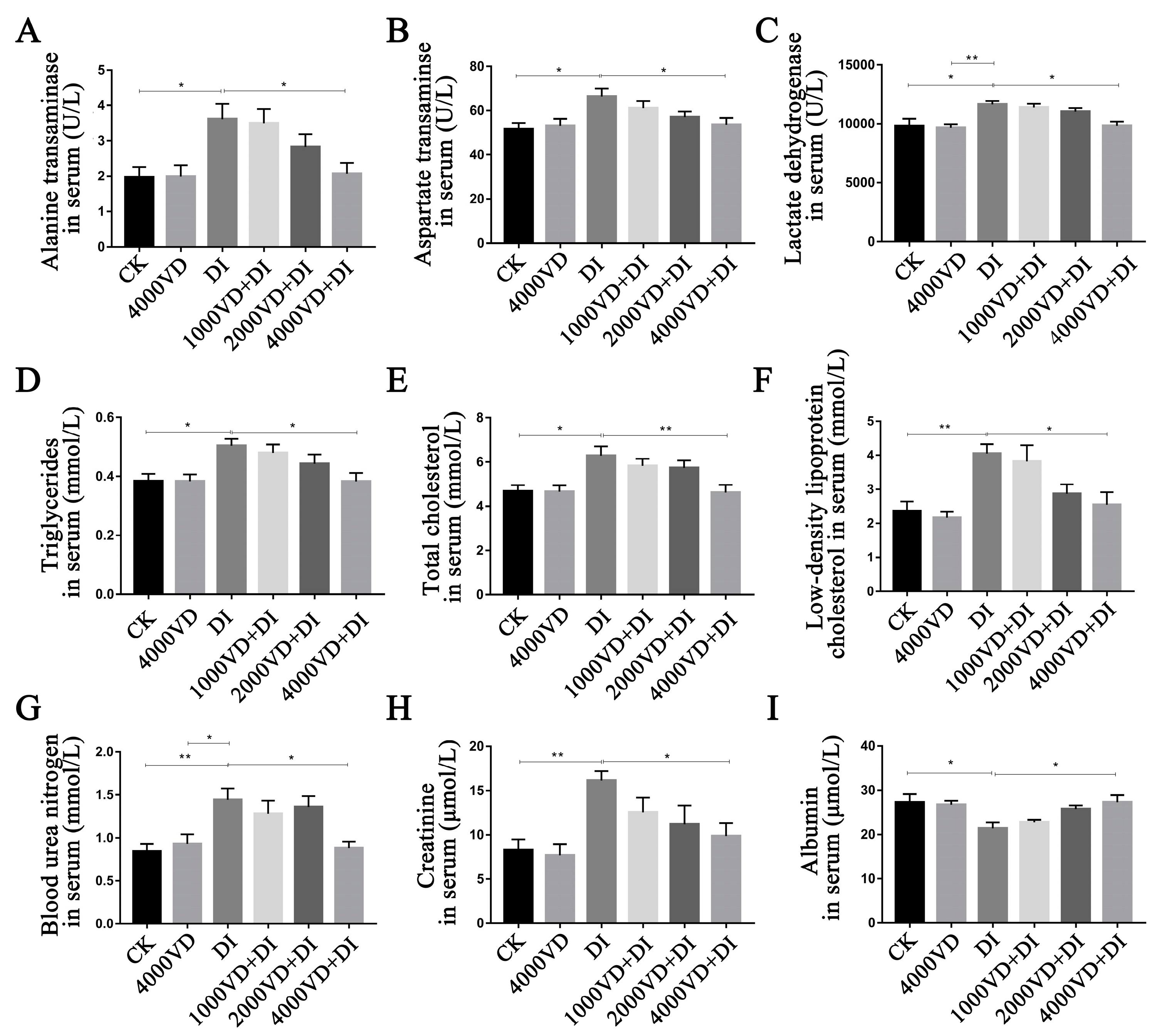
3.4.2. Determination of Biomarkers of Intestinal Injury in Serum
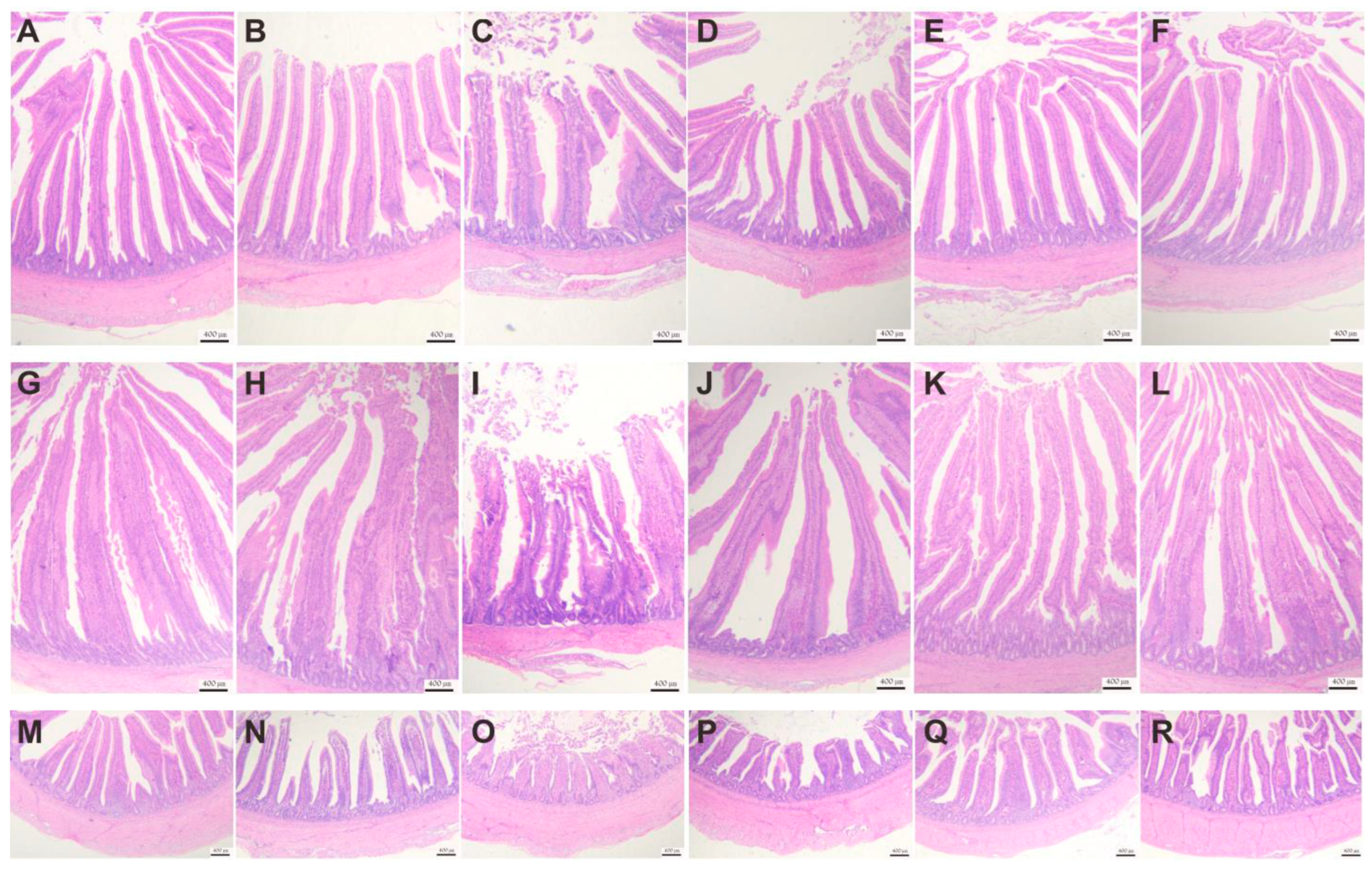
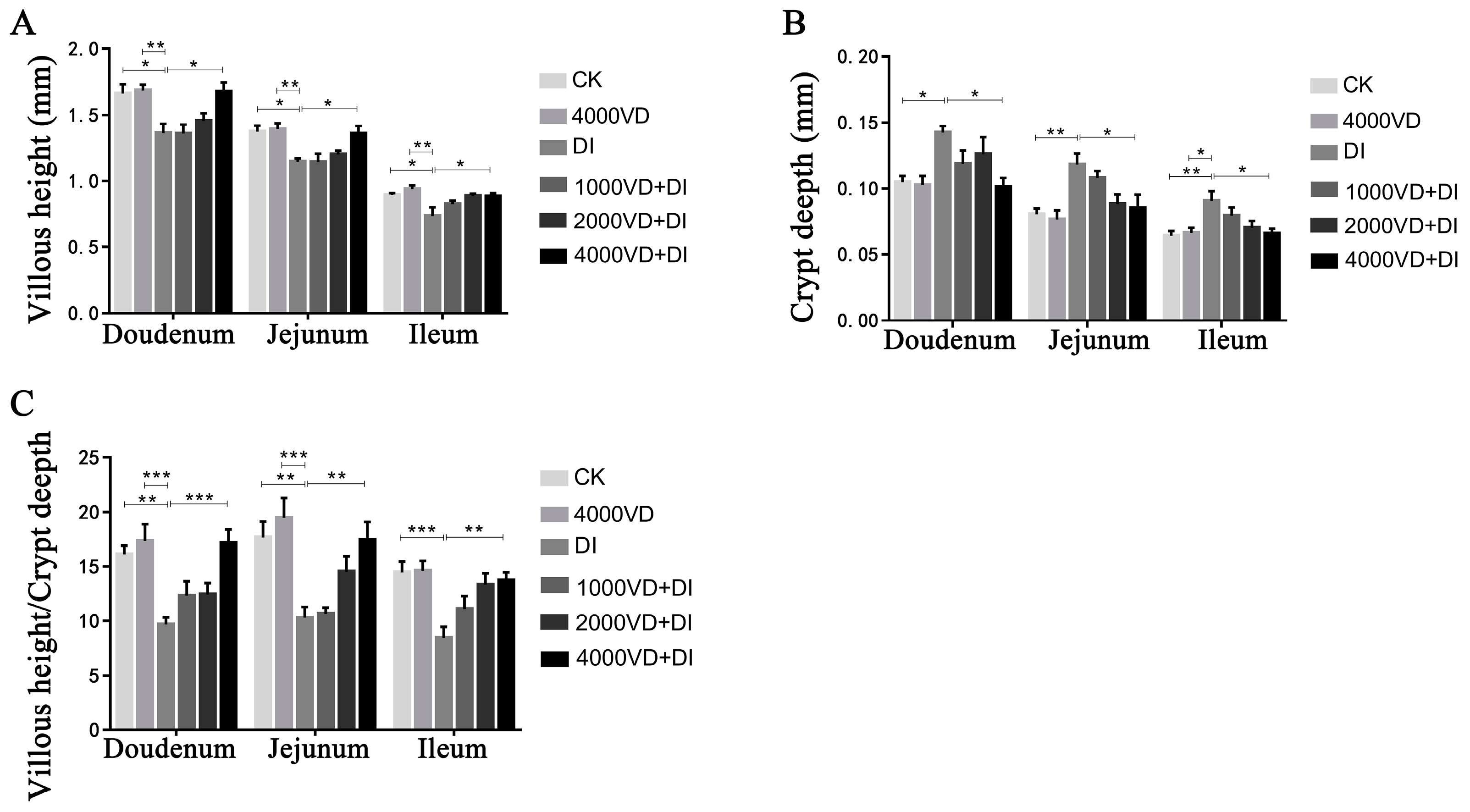
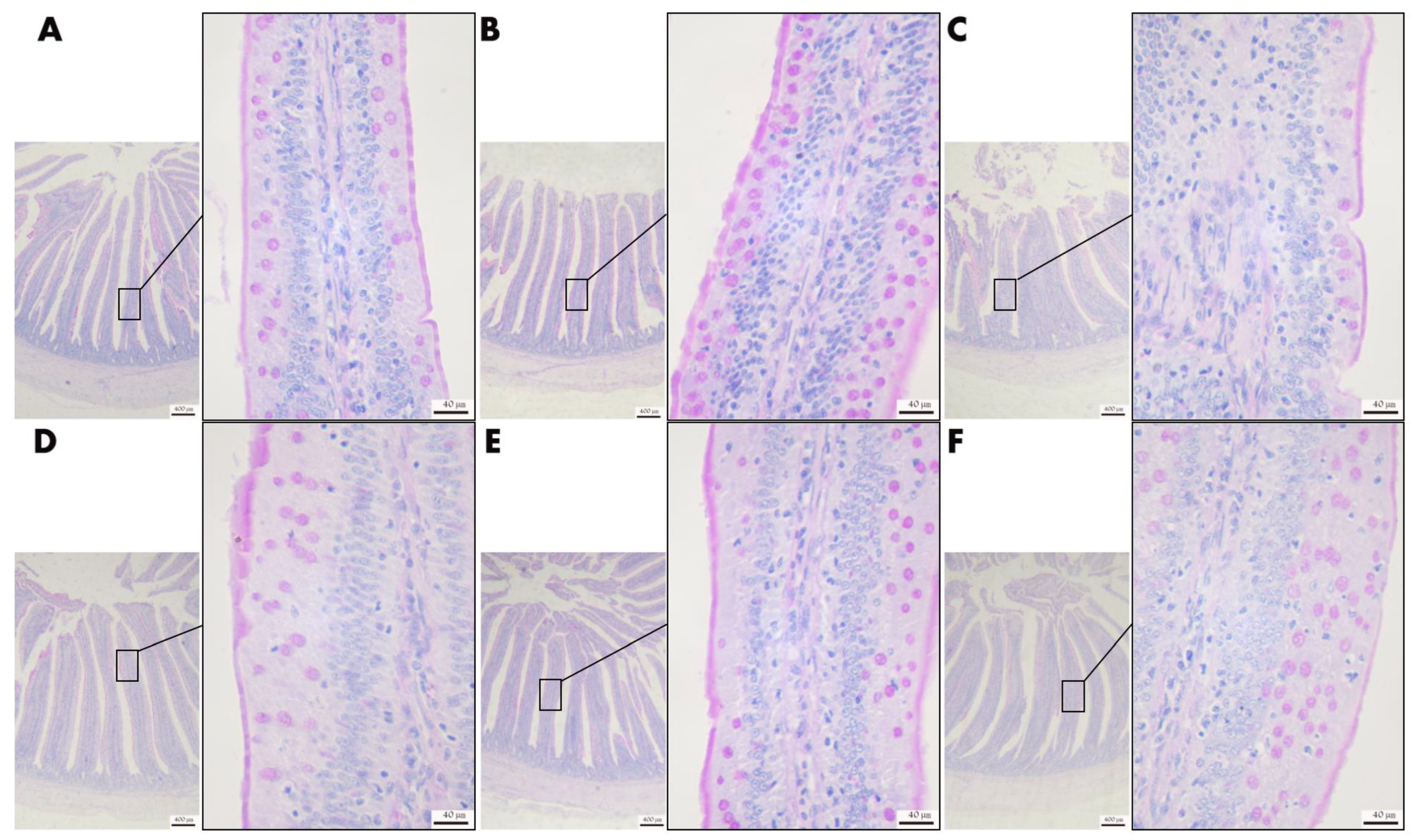
3.4.3. Effect of VD3 Addition on Intestinal Tissue Morphology
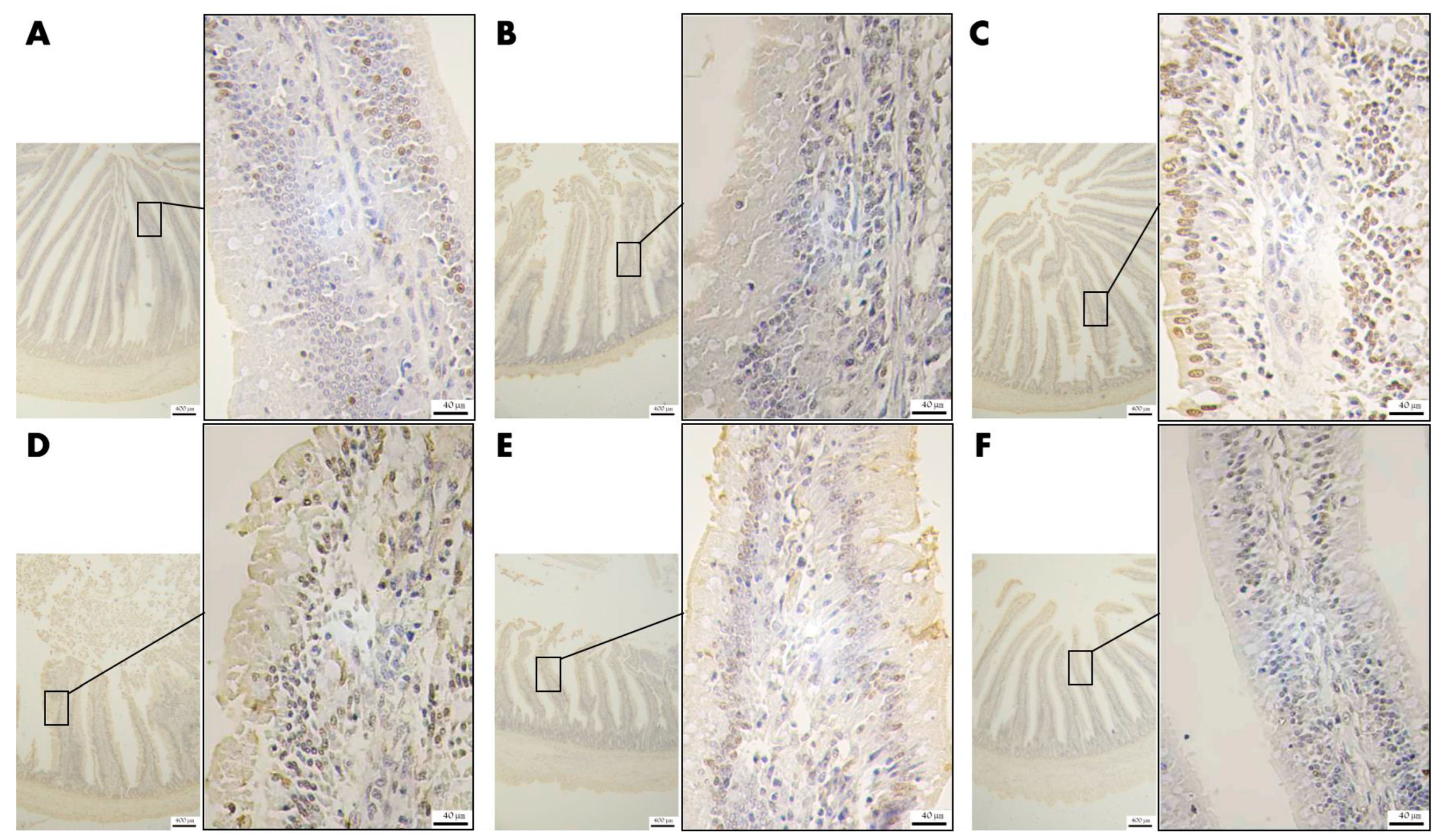
3.4.4. Effect of VD3 Addition on the Level of Apoptosis of Epithelial Cells in Jejunal Tissue
3.4.5. Effect of VD3 Addition on the Level of Polysaccharide Secretion in Goblet Cells of Jejunal Tissue
3.5. Effect of VD3 Addition on the Liver and Kidney Injury
3.5.1. Determination of Biomarkers of Liver and Kidney Injury in Serum
3.5.2. Effect of VD3 Addition on the Morphology of the Liver Tissues
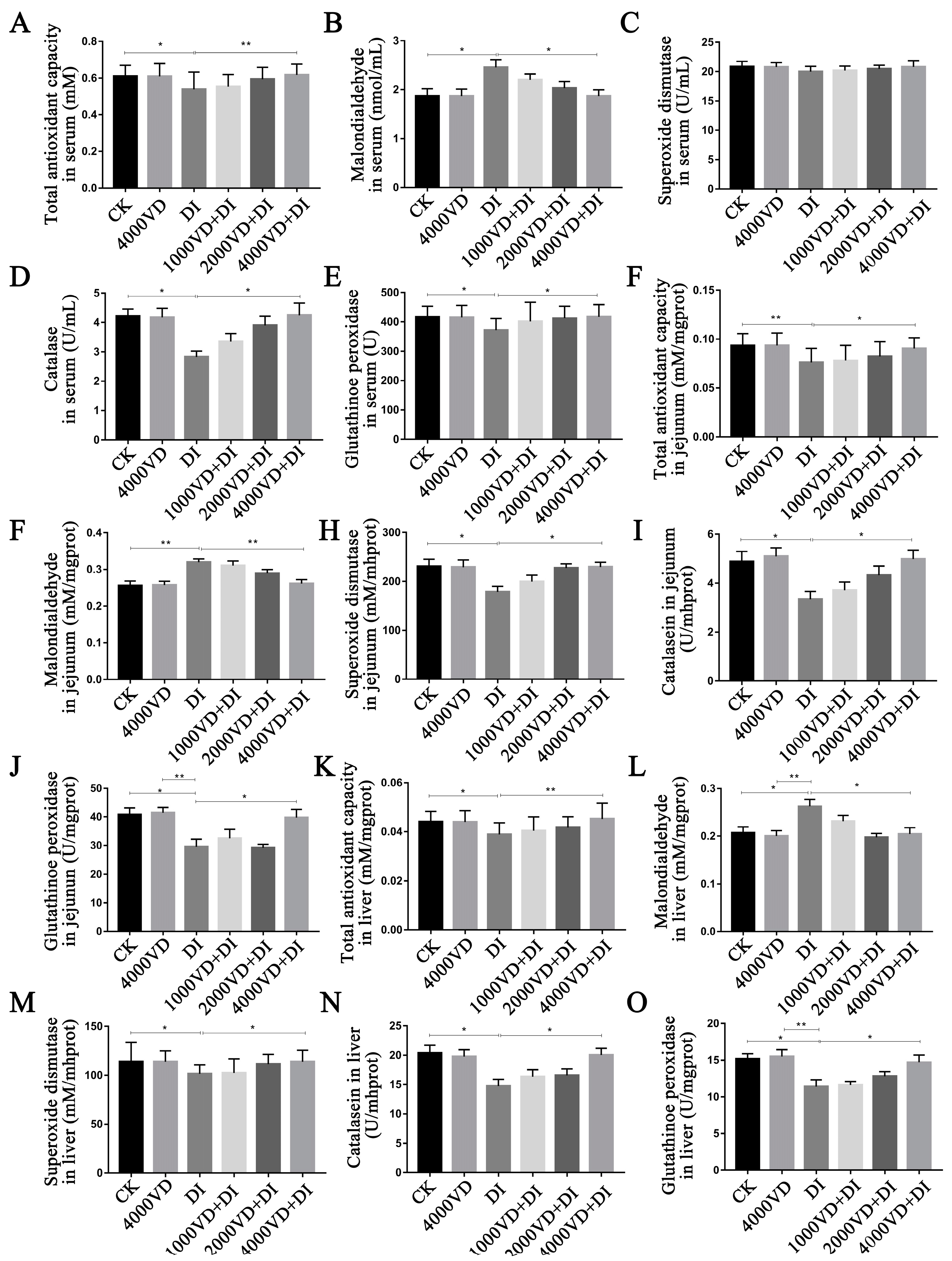
3.6. Effect of VD3 Addition on the Antioxidant-Related Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, Y.; Chen, Y.; Zhang, H.; Wang, T. Pterostilbene as a protective antioxidant attenuates diquat-induced liver injury and oxidative stress in 21-day-old broiler chickens. Poult. Sci. 2020, 99, 3158–3167. [Google Scholar] [CrossRef]
- Estévez, M. Oxidative damage to poultry: From farm to fork. Poult. Sci. 2015, 94, 1368–1378. [Google Scholar] [CrossRef]
- Chen, Y.P.; Gu, Y.F.; Zhao, H.R.; Zhou, Y.M. Dietary squalene supplementation alleviates diquat-induced oxidative stress and liver damage of broiler chickens. Poult. Sci. 2021, 100, 100919. [Google Scholar] [CrossRef]
- Miao, Z.Q.; Dong, Y.Y.; Qin, X.; Yuan, J.M.; Han, M.M.; Zhang, K.K.; Shi, S.R.; Song, X.Y.; Zhang, J.Z.; Li, J.H. Dietary supplementation of methionine mitigates oxidative stress in broilers under high stocking density. Poult. Sci. 2021, 100, 101231. [Google Scholar] [CrossRef]
- Hu, X.; Chi, Q.; Wang, D.; Chi, X.; Teng, X.; Li, S. Hydrogen sulfide inhalation-induced immune damage is involved in oxidative stress, inflammation, apoptosis and the Th1/Th2 imbalance in broiler bursa of Fabricius. Ecotoxicol. Environ. Saf. 2018, 164, 201–209. [Google Scholar] [CrossRef]
- Han, H.; Zhang, J.; Chen, Y.; Shen, M.; Yan, E.; Wei, C.; Yu, C.; Zhang, L.; Wang, T. Dietary taurine supplementation attenuates lipopolysaccharide-induced inflammatory responses and oxidative stress of broiler chickens at an early age. J. Anim. Sci. 2020, 98, skaa311. [Google Scholar] [CrossRef]
- Qiu, W.; Zhang, X.; Pang, X.; Huang, J.; Zhou, S.; Wang, R.; Tang, Z.; Su, R. Asiatic acid alleviates LPS-induced acute kidney injury in broilers by inhibiting oxidative stress and ferroptosis via activation of the Nrf2 pathway. Food Chem. Toxicol. 2022, 170, 113468. [Google Scholar] [CrossRef]
- Malekinejad, H.; Akbari, P.; Allymehr, M.; Hobbenaghi, R.; Rezaie, A. Cyclopiazonic acid augments the hepatic and renal oxidative stress in broiler chicks. Hum. Exp. Toxicol. 2011, 30, 910–919. [Google Scholar] [CrossRef]
- Kong, L.; Wang, Z.; Xiao, C.; Zhu, Q.; Song, Z. Glycerol monolaurate attenuated immunological stress and intestinal mucosal injury by regulating the gut microbiota and activating AMPK/Nrf2 signaling pathway in lipopolysaccharide-challenged broilers. Anim. Nutr. 2022, 10, 347–359. [Google Scholar] [CrossRef]
- Zheng, Y.W.; Zhang, J.Y.; Zhou, H.B.; Guo, Y.P.; Ma, Q.G.; Ji, C.; Zhao, L.H. Effects of dietary pyrroloquinoline quinone disodium supplementation on inflammatory responses, oxidative stress, and intestinal morphology in broiler chickens challenged with lipopolysaccharide. Poult. Sci. 2020, 99, 5389–5398. [Google Scholar] [CrossRef]
- Jones, G.M.; Vale, J.A. Mechanisms of toxicity, clinical features, and management of diquat poisoning: A review. J. Toxicol. Clin. Toxicol. 2000, 38, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Wu, H.; Wang, C.; Zhang, Q.; Jiao, L.; Lin, F.; Hu, C.H. Diquat-induced oxidative stress increases intestinal permeability, impairs mitochondrial function, and triggers mitophagy in piglets. J. Anim. Sci. 2018, 96, 1795–1805. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Guo, Q.; Wang, W.; Duan, Y.; Zhang, L.; Li, J.; He, S.; Chen, W.; Li, F. Taurine Alleviates Intestinal Injury by Mediating Tight Junction Barriers in Diquat-Challenged Piglet Models. Front. Physiol. 2020, 11, 449. [Google Scholar] [CrossRef]
- Van Vleet, T.R.; Schnellmann, R.G. Toxic nephropathy: Environmental chemicals. Semin. Nephrol. 2003, 23, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Koch, R.E.; Hill, G.E. An assessment of techniques to manipulate oxidative stress in animals. Funct. Ecol. 2017, 31, 9–21. [Google Scholar] [CrossRef]
- Han, J.C.; Wang, J.G.; Chen, G.H.; Zhang, J.L.; Zhang, N.; Qu, H.X.; Guo, Y.; Yan, Y.F.; Yang, X.J. 1α-Hydroxycholecalciferol improves the growth performance and up-regulates the mRNA expression of vitamin D receptor in the small intestine and kidney of broiler chickens. Poult. Sci. 2018, 97, 1263–1270. [Google Scholar] [CrossRef]
- Jones, G.; Strugnell, S.A.; Deluca, H.F. Current understanding of the molecular actions of vitamin D. Physiol. Rev. 1998, 78, 1193–1231. [Google Scholar] [CrossRef]
- L Bishop, E.; Ismailova, A.; Dimeloe, S.; Hewison, M.; White, J.H. Vitamin D and Immune Regulation: Antibacterial, Antiviral, Anti-Inflammatory. JBMR Plus 2021, 5, e10405. [Google Scholar] [CrossRef]
- Yang, P.; Ma, Y. Recent advances of vitamin D in immune, reproduction, performance for pig: A review. Anim. Health Res. Rev. 2021, 22, 85–95. [Google Scholar] [CrossRef]
- Charoenngam, N.; Shirvani, A.; Holick, M.F. Vitamin D for skeletal and non-skeletal health: What we should know. J. Clin. Orthop. Trauma 2019, 10, 1082–1093. [Google Scholar] [CrossRef]
- Baeke, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef]
- Roth, D.E.; Abrams, S.A.; Aloia, J.; Bergeron, G.; Bourassa, M.W.; Brown, K.H.; Calvo, M.S.; Cashman, K.D.; Combs, G.; De-Regil, L.M.; et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2018, 1430, 44–79. [Google Scholar] [CrossRef] [PubMed]
- Leal, L.K.A.M.; Lima, L.A.; de Aquino, P.E.A.; de Sousa, J.A.C.; Gadelha, C.V.J.; Calou, I.B.F.; Lopes, M.J.P.; Lima, F.A.V.; Neves, K.R.T.; de Andrade, G.M.; et al. Vitamin D (VD3) antioxidative and anti-inflammatory activities: Peripheral and central effects. Eur. J. Pharmacol. 2020, 879, 173099. [Google Scholar] [CrossRef]
- Geng, Y.; Ma, Q.; Wang, Z.; Guo, Y. Dietary vitamin D(3) supplementation protects laying hens against lipopolysaccharide-induced immunological stress. Nutr. Metab. 2018, 15, 58. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, H.; Okamatsu, M.; Ogasawara, H.; Tsugawa, N.; Isoda, N.; Matsuno, K.; Sakoda, Y. Oral Supplementation of the Vitamin D Metabolite 25(OH)D(3) Against Influenza Virus Infection in Mice. Nutrients 2020, 12, 2000. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, C.; Zhang, T.; Yan, L.; Qiu, L.; Yin, H.; Ding, X.; Bai, S.; Zeng, Q.; Mao, X.; et al. Dietary 25-hydroxyvitamin D improves intestinal health and microbiota of laying hens under high stocking density. Poult. Sci. 2021, 100, 101132. [Google Scholar] [CrossRef]
- Ramasundara, M.; Leach, S.T.; Lemberg, D.A.; Day, A.S. Defensins and inflammation: The role of defensins in inflammatory bowel disease. J. Gastroenterol. Hepatol. 2009, 24, 202–208. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, L.; Li, S.; Zhang, G.; Ouyang, L.; Robinson, K.; Tang, Y.; Zhu, Q.; Li, D.; Hu, Y.; et al. 1,25-Dihydroxyvitamin-D3 Induces Avian β-Defensin Gene Expression in Chickens. PLoS ONE 2016, 11, e0154546. [Google Scholar] [CrossRef]
- Jaime, J.; Vargas-Bermúdez, D.S.; Yitbarek, A.; Reyes, J.; Rodríguez-Lecompte, J.C. Differential immunomodulatory effect of vitamin D (1,25 (OH)(2) D(3)) on the innate immune response in different types of cells infected in vitro with infectious bursal disease virus. Poult. Sci. 2020, 99, 4265–4277. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.; Fang, X.; Gu, L.; Luo, C.; Chen, L.; Wang, Q. Vitamin D(3) Protects Mice from Diquat-Induced Oxidative Stress through the NF-κB/Nrf2/HO-1 Signaling Pathway. Oxid. Med. Cell. Longev. 2021, 2021, 6776956. [Google Scholar] [CrossRef]
- Almasmoum, H.; Refaat, B.; Ghaith, M.M.; Almaimani, R.A.; Idris, S.; Ahmad, J.; Abdelghany, A.H.; Basalamah, M.A.; El-Boshy, M. Protective effect of Vitamin D3 against lead induced hepatotoxicity, oxidative stress, immunosuppressive and calcium homeostasis disorders in rat. Environ. Toxicol. Pharmacol. 2019, 72, 103246. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D: A custodian of cell signaling stability in health and disease. Biochem. Soc. Trans. 2015, 43, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D deficiency: Infertility and neurodevelopmental diseases (attention deficit hyperactivity disorder, autism, and schizophrenia). Am. J. Physiol. Cell Physiol. 2018, 314, C135–C151. [Google Scholar] [CrossRef] [PubMed]
- Pitargue, F.M.; Kim, J.H.; Goo, D.; Delos Reyes, J.B.; Kil, D.Y. Effect of vitamin E sources and inclusion levels in diets on growth performance, meat quality, alpha-tocopherol retention, and intestinal inflammatory cytokine expression in broiler chickens. Poult. Sci. 2019, 98, 4584–4594. [Google Scholar] [CrossRef]
- Zhang, L.H.; He, T.F.; Hu, J.X.; Li, M.; Piao, X.S. Effects of normal and low calcium and phosphorus levels and 25-hydroxycholecalciferol supplementation on performance, serum antioxidant status, meat quality, and bone properties of broilers. Poult. Sci. 2020, 99, 5663–5672. [Google Scholar] [CrossRef]
- Duffy, S.K.; Kelly, A.K.; Rajauria, G.; Jakobsen, J.; Clarke, L.C.; Monahan, F.J.; Dowling, K.G.; Hull, G.; Galvin, K.; Cashman, K.D.; et al. The use of synthetic and natural vitamin D sources in pig diets to improve meat quality and vitamin D content. Meat Sci. 2018, 143, 60–68. [Google Scholar] [CrossRef]
- Leal, L.N.; Beltrán, J.A.; Marc, B.; Bello, J.M.; Den Hartog, L.A.; Hendriks, W.H.; Martín-Tereso, J. Supplementation of lamb diets with vitamin E and rosemary extracts on meat quality parameters. J. Sci. Food Agric. 2020, 100, 2922–2931. [Google Scholar] [CrossRef]
- Pion, S.J.; Van Heugten, E.; See, M.T.; Larick, D.K.; Pardue, S. Effects of vitamin C supplementation on plasma ascorbic acid and oxalate concentrations and meat quality in swine. J. Anim. Sci. 2004, 82, 2004–2012. [Google Scholar] [CrossRef]
- Vazquez, J.R.; Gómez, G.V.; López, C.C.; Cortés, A.C.; Díaz, A.C.; Fernández, S.R.T.; Rosales, E.M.; Avila, A.G. Effects of 25-hydroxycholecalciferol with two D(3) vitamin levels on production and immunity parameters in broiler chickens. J. Anim. Physiol. Anim. Nutr. 2018, 102, e493–e497. [Google Scholar] [CrossRef]
- Filgueiras, M.S.; Rocha, N.P.; Novaes, J.F.; Bressan, J. Vitamin D status, oxidative stress, and inflammation in children and adolescents: A systematic review. Crit. Rev. Food Sci. Nutr. 2020, 60, 660–669. [Google Scholar] [CrossRef]
- Wada, K.; Tanaka, H.; Maeda, K.; Inoue, T.; Noda, E.; Amano, R.; Kubo, N.; Muguruma, K.; Yamada, N.; Yashiro, M.; et al. Vitamin D receptor expression is associated with colon cancer in ulcerative colitis. Oncol. Rep. 2009, 22, 1021–1025. [Google Scholar]
- Asgharpour, P.; Dezfouli, M.R.M.; Nadealian, M.G.; Eftekhari, Z.; Borojeni, G.R.N. Effects of 1, 25-dihydroxy vitamin D3 on clinical symptoms, pro-inflammatory and inflammatory cytokines in calves with experimental pneumonia. Res. Vet. Sci. 2020, 132, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Castejon, G.; Brough, D. Understanding the mechanism of IL-1β secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.S.R.; Lima, V.C.O.; Piuvezam, G.; Azevedo, K.P.M.; Maciel, B.L.L.; Morais, A.H.A. Mechanisms of action of molecules with anti-TNF-alpha activity on intestinal barrier inflammation: A systematic review protocol. Medicine 2019, 98, e17285. [Google Scholar] [CrossRef]
- Saraiva, M.; O’garra, A. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 2010, 10, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Adelani, I.B.; Ogadi, E.O.; Onuzulu, C.; Rotimi, O.A.; Maduagwu, E.N.; Rotimi, S.O. Dietary vitamin D ameliorates hepatic oxidative stress and inflammatory effects of diethylnitrosamine in rats. Heliyon 2020, 6, e04842. [Google Scholar] [CrossRef]
- Zhang, H.; Majdeddin, M.; Gaublomme, D.; Taminiau, B.; Boone, M.; Elewaut, D.; Daube, G.; Josipovic, I.; Zhang, K.; Michiels, J. 25-hydroxycholecalciferol reverses heat induced alterations in bone quality in finisher broilers associated with effects on intestinal integrity and inflammation. J. Anim. Sci. Biotechnol. 2021, 12, 104. [Google Scholar] [CrossRef]
- Lauridsen, C. From oxidative stress to inflammation: Redox balance and immune system. Poult. Sci. 2019, 98, 4240–4246. [Google Scholar] [CrossRef]
- Liu, L.; Wu, C.; Chen, D.; Yu, B.; Huang, Z.; Luo, Y.; Zheng, P.; Mao, X.; Yu, J.; Luo, J.; et al. Selenium-Enriched Yeast Alleviates Oxidative Stress-Induced Intestinal Mucosa Disruption in Weaned Pigs. Oxid. Med. Cell. Longev. 2020, 2020, 5490743. [Google Scholar] [CrossRef]
- Bartsch, H.; Nair, J. Chronic inflammation and oxidative stress in the genesis and perpetuation of cancer: Role of lipid peroxidation, DNA damage, and repair. Langenbeck’s Arch. Surg. 2006, 391, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Alhotan, R.A.; Al Sulaiman, A.R.; Alharthi, A.S.; Abudabos, A.M. Protective influence of betaine on intestinal health by regulating inflammation and improving barrier function in broilers under heat stress. Poult. Sci. 2021, 100, 101337. [Google Scholar] [CrossRef] [PubMed]
- Laudadio, V.; Passantino, L.; Perillo, A.; Lopresti, G.; Passantino, A.; Khan, R.U.; Tufarelli, V. Productive performance and histological features of intestinal mucosa of broiler chickens fed different dietary protein levels. Poult. Sci. 2012, 91, 265–270. [Google Scholar] [CrossRef]
- Zhang, A.W.; Lee, B.D.; Lee, S.K.; Lee, K.W.; An, G.H.; Song, K.B.; Lee, C.H. Effects of yeast (Saccharomyces cerevisiae) cell components on growth performance, meat quality, and ileal mucosa development of broiler chicks. Poult. Sci. 2005, 84, 1015–1021. [Google Scholar] [CrossRef]
- Farré, R.; Fiorani, M.; Abdu Rahiman, S.; Matteoli, G. Intestinal Permeability, Inflammation and the Role of Nutrients. Nutrients 2020, 12, 1185. [Google Scholar] [CrossRef]
- Patankar, J.V.; Becker, C. Cell death in the gut epithelium and implications for chronic inflammation. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 543–556. [Google Scholar] [CrossRef]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran sulfate sodium (DSS)-induced colitis in mice. Curr. Protoc. Immunol. 2014, 104, 15.25.1–15.25.14. [Google Scholar] [CrossRef]
- Van Der Sluis, M.; De Koning, B.A.; De Bruijn, A.C.; Velcich, A.; Meijerink, J.P.; Van Goudoever, J.B.; Büller, H.A.; Dekker, J.; Van Seuningen, I.; Renes, I.B.; et al. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 2006, 131, 117–129. [Google Scholar] [CrossRef]
- Baradaran, A.; Samadi, F.; Ramezanpour, S.S.; Yousefdoust, S. Hepatoprotective effects of silymarin on CCl(4)-induced hepatic damage in broiler chickens model. Toxicol. Rep. 2019, 6, 788–794. [Google Scholar] [CrossRef]
- Parizadeh, S.M.; Rezayi, M.; Jafarzadeh-Esfehani, R.; Avan, A.; Ghazizadeh, H.; Emadzadeh, M.; Sahebi, R.; Ferns, G.A.; Ghayour-Mobarhan, M. Association of vitamin D status with liver and kidney disease: A systematic review of clinical trials, and cross-sectional and cohort studies. Int. J. Vitam. Nutr. Res. 2021, 91, 175–187. [Google Scholar] [CrossRef]
- Xing, J.; Chu, Z.; Han, D.; Jiang, X.; Zang, X.; Liu, Y.; Gao, S.; Sun, L. Lethal diquat poisoning manifesting as central pontine myelinolysis and acute kidney injury: A case report and literature review. J. Int. Med. Res. 2020, 48, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Razzaque, M.S. FGF23, klotho and vitamin D interactions: What have we learned from in vivo mouse genetics studies? Adv. Exp. Med. Biol. 2012, 728, 84–91. [Google Scholar] [PubMed]
- Tullet, J.M.A.; Green, J.W.; Au, C.; Benedetto, A.; Thompson, M.A.; Clark, E.; Gilliat, A.F.; Young, A.; Schmeisser, K.; Gems, D. The SKN-1/Nrf2 transcription factor can protect against oxidative stress and increase lifespan in C. elegans by distinct mechanisms. Aging Cell 2017, 16, 1191–1194. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Niu, Z.; Xue, Y.; Gao, J.; Zhang, M.; Li, M.; Peng, Y.; Zhang, S.; Li, W.; Zhang, Q.; et al. Chronic vitamin D(3) supplementation alleviates cognition impairment via inhibition of oxidative stress regulated by PI3K/AKT/Nrf2 in APP/PS1 transgenic mice. Neurosci. Lett. 2022, 783, 136725. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Meng, F.; Wang, S.; Xia, S.; Wang, R. Vitamin D(3) mitigates lipopolysaccharide-induced oxidative stress, tight junction damage and intestinal inflammatory response in yellow catfish, Pelteobagrus fulvidraco. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2021, 243, 108982. [Google Scholar] [CrossRef]


| Ingredient | 1~21 d | 22~38 d |
|---|---|---|
| Maize | 52.50 | 58.80 |
| Soybean meal | 40.00 | 33.80 |
| Soybean oil | 3.00 | 3.00 |
| Dicalcium phosphate | 1.9 | 1.8 |
| Limstore powder | 1.08 | 1.22 |
| Salt | 0.37 | 0.37 |
| Lysine | 0.05 | 0.03 |
| Methionine | 0.19 | 0.07 |
| Premix 1 | 0.80 | 0.80 |
| Choline chloride | 0.11 | 0.11 |
| Total | 100.00 | 100.00 |
| Nutritional levels 2 | ||
| Metabolisable energy (MJ/kg) | 12.42 | 12.62 |
| Crude protein | 21.77 | 19.65 |
| Calcium | 1.00 | 1.02 |
| Available phosphorus | 0.44 | 0.42 |
| Lysine | 1.34 | 1.15 |
| Methionine | 0.55 | 0.40 |
| Cystine | 0.40 | 0.36 |
| Item | Initial Weight (g) | Final Weight (g) | ADG (g) | ADFI (g) | F/G |
|---|---|---|---|---|---|
| CK | 109.33 ± 4.56 a | 565.84 ± 34.18 a | 16.30 ± 1.09 a | 30.29 ± 1.62 bc | 1.87 ± 0.18 a |
| 4000VD | 109.06 ± 5.07 a | 566.58 ± 47.79 a | 16.34 ± 1.47 a | 30.44 ± 1.50 bc | 1.87 ± 0.16 a |
| DI | 109.01 ± 6.90 a | 546.61 ± 38.87 c | 15.62 ± 1.10 a | 29.89 ± 1.82 c | 1.92 ± 0.19 a |
| 1000VD+DI | 108.39 ± 5.56 a | 547.04 ± 36.44 c | 15.66 ± 1.07 a | 29.51 ± 2.00 c | 1.89 ± 0.19 a |
| 2000VD+DI | 110.22 ± 6.58 a | 552.50 ± 38.27 bc | 15.80 ± 1.17 a | 28.75 ± 2.21 c | 1.82 ± 0.17 a |
| 4000VD+DI | 109.52 ± 6.79 a | 564.41 ± 36.73 ab | 16.25 ± 1.12 a | 31.83 ± 1.71 a | 1.96 ± 0.13 a |
| F | 0.828 | 4.766 | 0.808 | 3.176 | 0.759 |
| P | 0.530 | <0.001 | 0.549 | 0.014 | 0.584 |
| Item | Dressing Percentage | Percentage of Eviscerated Yield |
|---|---|---|
| CK | 92.66 ± 1.07 a | 77.39 ± 1.12 a |
| 4000VD | 92.68 ± 1.50 a | 77.27 ± 1.23 a |
| DI | 91.66 ± 0.80 b | 76.37 ± 1.24 b |
| 1000VD+DI | 91.91 ± 1.08 ab | 76.94 ± 1.28 ab |
| 2000VD+DI | 92.49 ± 0.96 a | 77.07 ± 1.22 ab |
| 4000VD+DI | 92.27 ± 1.11 ab | 76.94 ± 1.05 ab |
| F | 2.55 | 2.113 |
| P | 0.032 | 0.070 |
| Item | Liver Index | Bursa of Fabricius Index | Kidney Index | Spleen Index |
|---|---|---|---|---|
| CK | 22.70 ± 0.68 a | 4.00 ± 0.24 a | 2.41 ± 0.06 a | 2.69 ± 0.22 a |
| 4000VD | 22.73 ± 0.57 a | 4.02 ± 0.16 a | 2.43 ± 0.07 a | 2.67 ± 0.14 a |
| DI | 21.39 ± 0.43 a | 3.48 ± 0.15 a | 2.19 ± 0.07 b | 2.09 ± 0.16 b |
| 1000VD+DI | 21.72 ± 0.45 a | 3.57 ± 0.25 a | 2.29 ± 0.08 ab | 2.15 ± 0.14 b |
| 2000VD+DI | 21.92 ± 0.46 a | 3.83 ± 0.10 a | 2.34 ± 0.06 ab | 2.17 ± 0.14 b |
| 4000VD+DI | 22.07 ± 0.67 a | 3.87 ± 0.16 a | 2.42 ± 0.07 a | 2.28 ± 0.13 ab |
| F | 0.973 | 1.287 | 1.812 | 2.709 |
| P | 0.44 | 0.279 | 0.119 | 0.027 |
| Item | pH 1 h | pH 24 h | Cooking Loss | Drip Loss |
|---|---|---|---|---|
| CK | 6.68 ± 0.17 a | 5.78 ± 0.04 a | 10.91 ± 0.49 a | 5.91 ± 1.01 a |
| 4000VD | 6.64 ± 0.18 ab | 5.85 ± 0.02 a | 10.41 ± 0.77 a | 5.49 ± 1.76 a |
| DI | 6.55 ± 0.12 b | 5.76 ± 0.03 a | 12.01 ± 0.76 a | 8.13 ± 1.08 a |
| 1000VD+DI | 6.54 ± 0.12 b | 5.80 ± 0.03 a | 12.46 ± 0.58 a | 7.89 ± 1.13 a |
| 2000VD+DI | 6.56 ± 0.07 b | 5.76 ± 0.03 a | 12.54 ± 0.36 a | 6.35 ± 1.25 a |
| 4000VD+DI | 6.58 ± 0.13 ab | 5.82 ± 0.05 a | 10.84 ± 0.98 a | 6.12 ± 0.50 a |
| F | 2.334 | 0.966 | 1.73 | 0.45 |
| P | 0.051 | 0.445 | 0.141 | 0.81 |
| Item | pH 1 h | pH 24 h | Cooking Loss | Drip Loss |
|---|---|---|---|---|
| CK | 6.66 ± 0.13 a | 6.28 ± 0.16 a | 15.07 ± 1.67 a | 5.51 ± 0.38 a |
| 4000VD | 6.65 ± 0.13 a | 6.29 ± 0.17 a | 15.12 ± 1.32 a | 5.60 ± 0.52 a |
| DI | 6.54 ± 0.12 b | 6.11 ± 0.83 b | 16.56 ± 1.10 a | 6.61 ± 0.87 a |
| 1000VD+DI | 6.56 ± 0.09 ab | 6.13 ± 0.15 b | 16.39 ± 0.74 a | 6.32 ± 1.09 a |
| 2000VD+DI | 6.59 ± 0.12 ab | 6.17 ± 0.12 ab | 15.68 ± 0.96 a | 5.85 ± 0.96 a |
| 4000VD+DI | 6.63 ± 0.09 ab | 6.21 ± 0.24 ab | 15.34 ± 1.14 a | 5.81 ± 0.20 a |
| F | 2.461 | 3.023 | 0.274 | 0.173 |
| P | 0.041 | 0.016 | 0.925 | 0.97 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nong, K.; Liu, Y.; Fang, X.; Qin, X.; Liu, Z.; Zhang, H. Effects of the Vitamin D3 on Alleviating the Oxidative Stress Induced by Diquat in Wenchang Chickens. Animals 2023, 13, 711. https://doi.org/10.3390/ani13040711
Nong K, Liu Y, Fang X, Qin X, Liu Z, Zhang H. Effects of the Vitamin D3 on Alleviating the Oxidative Stress Induced by Diquat in Wenchang Chickens. Animals. 2023; 13(4):711. https://doi.org/10.3390/ani13040711
Chicago/Turabian StyleNong, Keyi, Youming Liu, Xin Fang, Xinyun Qin, Zhineng Liu, and Haiwen Zhang. 2023. "Effects of the Vitamin D3 on Alleviating the Oxidative Stress Induced by Diquat in Wenchang Chickens" Animals 13, no. 4: 711. https://doi.org/10.3390/ani13040711
APA StyleNong, K., Liu, Y., Fang, X., Qin, X., Liu, Z., & Zhang, H. (2023). Effects of the Vitamin D3 on Alleviating the Oxidative Stress Induced by Diquat in Wenchang Chickens. Animals, 13(4), 711. https://doi.org/10.3390/ani13040711





