Modeling Gastrointestinal Tract Wet Pool Size in Small Ruminants
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Set
2.2. Models’ Development
2.3. Model’s Evaluation
3. Results
3.1. Models’ Development
3.2. Model Evaluation
4. Discussion
4.1. Goats Fed with the Ad Libitum Suckling Diet
4.2. Animals Fed with Feeding Levels of Solid Diet
4.3. Animals Fed with an Ad Libitum Solid Diet
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clauss, M.; Schwarm, A.; Ortmann, S.; Streich, W.J.; Hummel, J. A Case of Non-Scaling in Mammalian Physiology? Body Size, Digestive Capacity, Food Intake, and Ingesta Passage in Mammalian Herbivores. Comp. Biochem. Phys. A 2007, 148, 249–265. [Google Scholar] [CrossRef]
- Chay-Canul, A.J.; Espinoza-Hernandez, J.C.; Ayala-Burgos, A.J.; Magaña-Monforte, J.G.; Aguilar-Perez, C.F.; Chizzotti, M.L.; Tedeschi, L.O.; Ku-Vera, J.C. Relationship of Empty Body Weight with Shrunken Body Weight and Carcass Weights in Adult Pelibuey Ewes at Different Physiological States. Small Rumin. Res. 2014, 117, 10–14. [Google Scholar] [CrossRef]
- Zinn, R.A. Influence of Time of Day of Live Weight Measurements for Feedlot Steers. J. Anim. Sci. 1990, 68, 915–918. [Google Scholar] [CrossRef]
- Waldo, D.R.; Smith, L.W. Gut Fill in Cattle and its Relation to Dietary Neutral Detergent Fibre. In Proceedings of the 2nd International Symposium on the Nutrition of Herbivores, Brisbane, Australia, 6–10 July 1987; p. 89. [Google Scholar]
- Kay, M.; Macdearmid, A.; MacLeod, N.A. Intensive Beef Production: 10. Replacement of Cereals with Chopped Straw. Anim. Prod. 1970, 12, 261–266. [Google Scholar] [CrossRef]
- Gindri, M.; Moraes, L.E.; Teixeira, I.A.M.A. Disentangling Digesta Passage Rate in The Gastrointestinal Tract of Male and Female Growing Goats Feed Varying Feeding Levels Using Multivariate and Metanalytical Approaches. Livest. Sci. 2021, 251, 104645–104659. [Google Scholar] [CrossRef]
- Macedo Junior, G.D.L.; Benevides, Y.I.; Campos, W.E.; Borges, I.; Rodriguez, N.M.; Ferreira, D.A. Nutrient Intake and Digestibility and Ruminal Rate Passage in Pregnant Ewes. Cienc. Anim. Bras. 2012, 13, 429–439. [Google Scholar]
- Owens, F.N.; Gill, D.R.; Secrist, D.S.; Coleman, S.W. Review of Some Aspects of Growth and Development of Feedlot Cattle. J. Anim. Sci. 1995, 73, 3152–3172. [Google Scholar] [CrossRef] [PubMed]
- Chilibroste, P.; Aguilar, C.; Garcia, F. Nutritional Evaluation of Diets. Simulation Model of Digestion and Passage of Nutrients Through the Rumen-Reticulum. Anim. Feed Sci. Technol. 1997, 68, 259–275. [Google Scholar] [CrossRef]
- Ahvenjärvi, S.; Rinne, M.; Heikkilä, T.; Huhtanen, P. The Effect of a Diet and Intrinsic Characteristics of Feed Particles on Passage Kinetics in Dairy Cows. In Proceedings of the 1st Nordic Feed Science Conference, Uppsala, Sweden, 22–23 June 2010; pp. 162–165. [Google Scholar]
- Huhtanen, P.; Ahvenjärvi, S.; Weisbjerg, M.R.; Nørgaard, P. Digestion and Passage of Fibre in Ruminants. In Ruminant Physiology: Digestion, Metabolism and Impact of Nutrition in Gene Impression, Immunology and Stress; Sejrsen, K., Hvelplund, T., Nielsen, M.O., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2006; pp. 87–135. [Google Scholar]
- Teixeira, I.A.M.A.; Gindri, M. Between-Individual Variability on Reticulorumen Digesta Passage Rate in Goats. Animal 2022, 13, 587–588. [Google Scholar] [CrossRef]
- Gindri, M.; Moraes, L.; Teixeira, I.A.M.A. Prediction Models of Reticulorumen Kinetics of Particles and Solutes in Goats. Animal 2022, 13, 534–535. [Google Scholar] [CrossRef]
- Lofgreen, G.P.; Hull, J.L.; Oxaaaki, K.K. Estimation of Empty Body Weight of Beef Cattle. J. Anim. Sci. 1962, 21, 20–24. [Google Scholar] [CrossRef]
- Garrett, W.N.; Hinman, N. Re-Evaluation of the Relationship Between Carcass Density and Body Composition of Beef Steers. J. Anim. Sci. 1969, 28, 1–5. [Google Scholar] [CrossRef]
- Fox, D.G.; Dockerty, T.R.; Johnson, R.R.; Preston, R.L. Relationship of Empty Body Weight to Carcass Weight in Beef Cattle. J. Anim. Sci. 1976, 43, 566–568. [Google Scholar] [CrossRef]
- Campos, L.M.; Almeida, A.K.; Biagioli, B.; Resende, K.T.; Teixeira, I.A.M.A. Predicting Empty Body Weight in Growing Goats: A Meta-Analytic Approach. Small Rum. Res. 2017, 155, 45–50. [Google Scholar] [CrossRef]
- Agricultural Research Council. The Nutrient Requirements of Ruminant Livestock; Commonwealth Agricultural Bureaux: Slough, UK, 1980; p. 351. [Google Scholar]
- Williams, C.B.; Keele, J.W.; Waldo, D.R. A Computer Model to Predict Empty Body Weight in Cattle from Diet and Animal Characteristics. J. Anim. Sci. 1992, 70, 3215–3222. [Google Scholar] [CrossRef]
- Schlecht, E.; Sangaré, M.; Becker, K. Seasonal Variations in Gastrointestinal Tract Fill of Grazing Zebu Cattle in the Sahel. J. Agric. Sci. 2003, 140, 461–468. [Google Scholar] [CrossRef]
- Bompadre, T.F.V.; Boaventura Neto, O.; Mendonça, A.N.; Souza, S.F.; Oliveira, D.; Fernandes, M.H.M.R.; Harter, C.J.; Almeida, A.K.; Resende, K.T.; Teixeira, I.A.M.A. Energy Requirements in Early Life are Similar for Male and Female Goat Kids. Asian-Austral. J. Anim. Sci. 2014, 27, 1712–1720. [Google Scholar] [CrossRef][Green Version]
- Medeiros, A.N.; Resende, K.T.; Teixeira, I.A.M.A.; Araújo, M.J.; Yañez, E.A.; Ferreira, A.C. Energy Requirements for Maintenance and Growth of Male Saanen Goat Kids. Asian-Austral. J. Anim. Sci. 2014, 27, 1293–1302. [Google Scholar] [CrossRef]
- Resende, K.T. Métodos de Estimativa da Composição Corporal e Exigências Nutricionais de Proteína, Energia e Macrominerais Inorgânicos de Caprinos em Crescimento. Ph.D. Thesis, Universidade Federal de Viçosa, Viçosa, Brazil, 1989. [Google Scholar]
- Ribeiro, S.D.A. Composição Corporal e Exigências em Energia, Proteína e Macrominerais de Caprinos Mestiços em Fase Inicial de Crescimento. Ph.D. Thesis, Universidade Estadual Paulista, Jaboticabal, Brazil, 1995. [Google Scholar]
- Teixeira, I.A.M.A.; Harter, C.J.; Pereira Filho, J.M.; Sobrinho, A.G.; Resende, K.T. Mineral Requirements for Growth and Maintenance of F1 Boer X Saanen Male Kids. J. Anim. Sci. 2015, 93, 2349–2356. [Google Scholar] [CrossRef]
- Almeida, A.K.; Resende, K.T.; St-Pierre, N.; Silva, S.P.; Soares, D.C.; Fernandes, M.H.M.R.; Souza, A.P.; Silva, N.C.D.; Lima, A.R.C.; Teixeira, I.A.M.A. Energy Requirements for Growth in Male and Female Saanen Goats. J. Anim. Sci. 2015, 93, 3932–3940. [Google Scholar] [CrossRef]
- Fernandes, M.H.M.R.; Resende, K.T.D.; Tedeshi, L.O.; Fernandes, J.S.; Silva, H.M.; Carstens, G.E.; Berchielli, T.T.; Teixeira, I.A.M.A.; Akinaga, L. Energy and Protein Requirements for Maintenance and Growth of Boer Crossbred Kids. J. Anim. Sci. 2007, 85, 1014–1023. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.C.D.; Yáñez, E.A.; Medeiros, A.N.; Resende, K.T.; Pereira Filho, J.M.; Fernandes, M.H.M.R.; Almeida, A.K.; Teixeira, I.A.M.A. Protein and Energy Requirements of Castrated Male Saanen Goats. Small Rumin. Res. 2015, 123, 88–94. [Google Scholar] [CrossRef]
- Figueiredo, F.O.M.; Leite, R.F.; St-Pierre, N.R.; Resende, K.T.; Almeida, A.K.; Souza, A.P.; Teixeira, I.A.M.A. Energy and Protein Requirements of Weaned Male and Female Saanen Goats. J. Anim. Physiol. Anim. Nutr. 2016, 101, 1014–1025. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, F.O.M.; Berchielli, T.T.; Resende, K.T.; Gomes, H.F.B.; Almeida, A.K.; Sakomura, N.K.; Teixeira, I.A.M.A. Energy Requirements for Growth of Pubertal Female Saanen Goats. J. Anim. Physiol. Anim. Nutr. 2016, 100, 294–300. [Google Scholar] [CrossRef]
- Gomes, H.F.B. Composição Corporal e Exigências Nutricionais de Caprinos Saanen Mahos dos 30 aos 45 Kg de Peso Vivo. Ph.D. Thesis, Universidade Estadual Paulista, Jaboticabal, Brazil, 2011. [Google Scholar]
- Härter, C.J.; Castagnino, D.D.S.; Rivera, A.R.D.; Lima, L.D.D.; Silva, H.G.D.O.; Mendonça, A.N.; Teixeira, I.A.M.A. Mineral Metabolism in Singleton and Twin-Pregnant Dairy Goats. Asian Australas. J. Anim. Sci. 2015, 28, 37–49. [Google Scholar] [CrossRef]
- Ribeiro, R.P. Determinação da Composição Corporal de Caprinos da Raça Saanen pelo Método de Absortometria de Raios-X de Dupla Energia (DXA). Master’s Thesis, Universidade Estadual Paulista, Jaboticabal, Brazil, 2019. [Google Scholar]
- Costa, M.R.G.F.; Pereira, E.S.; Silva, A.M.A.; Paulino, P.V.R.; Mizubuti, I.Y.; Pimentel, P.G.; Pinto, A.P.; Rocha Junior, J.N. Body Composition and Net Energy and Protein Requirements of Morada Nova Lambs. Small Rum. Res. 2013, 114, 206–213. [Google Scholar] [CrossRef][Green Version]
- Ribeiro do Nascimento, D.; Herbster, C.J.L.; Rocha, A.C.; Freitas, I.Q.; Justino, E.S.; Brito Neto, A.S.; Pereira, E.S. Efeito da Restrição Alimentar Sobre a Ingestão de Água e Disgestibilidade de Femêas Dorper X Santa Inês. In Proceedings of the XVI Congresso Nordestino de Produção Animal, Desmistificando a Produção Animal para a Sociedade, Fortaleza, Brazil, 30 November–2 December 2022. [Google Scholar]
- Macedo Junior, G.L. Exigências Nutricionais de Ovelhas Gestantes da Raça Santa Inês. Ph.D. Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, 2008. [Google Scholar]
- Mendes, M.S.; Souza, J.G.; Herbster, C.J.L.; Brito Neto, A.S.; Silva, L.P.; Rodrigues, J.P.P.; Marcondes, M.I.; Oliveira, R.L.; Bezerra, L.R.; Pereira, E.S. Maintenance and Growth Requirements in Male Dorper × Santa Ines Lambs. Front. Vet. Sci. 2021, 8, 676956. [Google Scholar] [CrossRef]
- de Oliveira, A.P.; Pereira, E.S.; Pinto, A.P.; Silva, A.M.A.; Carneiro, M.S.S.; Mizubuti, I.Y.; Ribeiro, E.L.A.; Campos, A.C.N.; Gadelha, C.R.F. Estimativas dos Requisitos Nutricionais e Utilização do Modelo Small Ruminant Nutrition System para Ovinos Deslanados em Condições Semiáridas. Semina Ci. Agr. 2014, 35, 1985–1998. [Google Scholar] [CrossRef]
- Pereira, G.M. Exigências de Proteína e Energia de Carneiros Santa Inês na Região Semiárida Brasileira. Master’s Thesis, Universidade Federal De Campina Grande, Patos, Brazil, 2011. [Google Scholar]
- Pereira, E.S.; Lima, F.W.R.; Marcondes, M.I.; Rodrigues, J.P.P.; Campos, A.C.N.; Silva, L.P.; Bezerra, L.R.; Pereira, M.W.F.; Oliveira, R.L. Energy and Protein Requirements of Santa Ines Lambs, a Breed of Hair Sheep. Animal 2017, 11, 2165–2174. [Google Scholar] [CrossRef]
- Pereira, E.S.; Pereira, M.; Marcondes, M.I.; Medeiros, A.N.; Oliveira, R.L.; Silva, L.P.; Mizubuti, I.Y.; Campos, A.C.; Heinzen, E.L.; Véras, A.S.; et al. Maintenance and Growth Requirements in Male and Female Hair Lambs. Small Rum. Res. 2017, 159, 75–83. [Google Scholar] [CrossRef]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1–145, 2020. Available online: https://CRAN.R-project.org/package=nlme (accessed on 18 October 2021).
- Bartoń, K. MuMIn: Multi-Model Inference, 2023. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 18 October 2021).
- Fox, J.; Weisberg, S.; Price, B.; Adler, D.; Bates, D.; Baud-Bovy, G.; Bolker, B.; Ellison, S.; Firth, D.; Friendly, M.; et al. Car: Companion to Applied Regression, 2019. Available online: https://CRAN.R-project.org/package=car (accessed on 18 October 2021).
- Gindri, M.; Moraes, L.; Teixeira, I.A.M.A. Prediction Models of Reticulorumen Particles and Solutes Passage Rate in Growing Goats. Animal, 2023; accepted for publication. [Google Scholar]
- Lin, L.I. A Concordance Correlation Coefficient to Evaluate Reproducibility. Biometrics 1989, 45, 255–268. [Google Scholar] [CrossRef]
- Ribeiro, P.R.; Gindri, M.; Biagioli, B.; Teixeira, I.A.M.A. Relationship Between Diet and Gastrointestinal Tract Wet Pool Size in Growing Goats: An Exploratory Approach. In Proceedings of the 56a Reunião da Sociedade Brasileira de Zootecnia, Challenges in Production and Sustainability, Florianópolis, Brazil, 16–20 August 2021. [Google Scholar]
- Gindri, M.; Leite, R.F.; Härter, C.J.; da Silva, S.P.; St-Pierre, N.; Fernandes, M.H.M.D.R.; Berchielli, T.T.; Teixeira, I.A.M.D.A. Body Weight and Sex Effects on Digesta Mean Retention Time in Growing Saanen Goats. Transl. Anim. Sci. 2020, 4, 883–900. [Google Scholar] [CrossRef] [PubMed]
- Van Soest, P.J. The Nutritional Ecology of the Ruminant, 2nd ed.; Cornell University Press: Ithaca, NY, USA, 1994; p. 488. [Google Scholar]
- Kleiber, M. The Fire of Life. An Introduction to Animal Energetics; Wiley: New York, NY, USA, 1961; p. 454. [Google Scholar]
- Thornton, R.; Minson, D. The Relationship Between Voluntary Intake and Mean Apparent Retention Time in the Rumen. Aust. J. Agric. Res. 1972, 23, 871–877. [Google Scholar] [CrossRef]
- Grovum, W.L.; Williams, V.J. Rate of Passage of Digesta in Sheep. Br. J. Nutr. 1977, 38, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Udén, P. The Effect of Grinding and Pelleting Hay on Digestibility, Fermentation Rate, Digesta Passage and Rumen and Particle Size in Cows. Anim. Feed Sci. Technol. 1988, 19, 145–157. [Google Scholar] [CrossRef]
- Müller, D.W.; Codron, D.; Meloro, C.; Munn, A.; Schwarm, A.; Hummel, J.; Clauss, M. Assessing the Jarman-Bell Principle: Scaling of Intake, Digestibility, Retention Time and Gut Fill with Body Mass in Mammalian Herbivores. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 164, 129–140. [Google Scholar] [CrossRef]
- Ulyatt, M.J.; Dellow, D.W.; John, A.; Reid, C.S.W.; Waghorn, G.C. Contribution of Chewing During Eating and Rumination as The Clearance of Digesta from the Ruminoreticulum. In Control of Digestion and Metabolism in Ruminants, Proceedings of the Sixth International Symposium on Ruminant Physiology, Banff, AB, Canada. 10–14 September 1984; Milligan, L.P., Grovum, W.L., Dobson, A., Eds.; Prentice-Hall: Englewood Cliffs, NJ, USA, 1986. [Google Scholar]
- Van Vuuren, A.M. Effect of Level of Hay Intake, Method of Marker Administration, and Stage of Lactation on Rate of Passage Through the Reticulorumen. Can. J. Anim. Sci. 1984, 64, 80–81. [Google Scholar] [CrossRef]
- Gross, J.E.; Demment, M.W.; Alkon, P.U.; Kotzman, M. Feeding and Chewing Behaviours of Nubian Ibex: Compensation for Sex-Related Differences in Body Size. Funct. Ecol. 1995, 9, 385–393. [Google Scholar] [CrossRef]
- Almeida, A.K.; Tedeschi, L.O.; de Resende, K.T.; Biagioli, B.; Cannas, A.; Teixeira, I.A.M.A. Prediction of Voluntary Dry Matter Intake in Stall Fed Growing Goats. Livest. Sci. 2019, 219, 1–9. [Google Scholar] [CrossRef]
- Andrade, M.E.B.; Härter, C.J.; Gindri, M.; Resende, K.T.; Teixeira, I.A.M.A. Visceral Organ Growth Patterns in Saanen Goats. J. Agric. Sci. 2019, 157, 721–742. [Google Scholar] [CrossRef]
- Cheeke, P.R.; Dierenfeld, E.S. Comparative Animal Nutrition and Metabolism; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Souza, A.P.; St-Pierre, N.R.; Fernandes, M.H.R.M.; Almeida, A.K.; Vargas, J.A.C.; Resende, K.T.; Teixeira, I.A.M.A. Sex Effects on Net Protein and Energy Requirements for Growth of Saanen Goats. J. Dairy Sci. 2017, 100, 4574–4586. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.P.; St-Pierre, N.R.; Fernandes, M.H.M.R.; Almeida, A.K.; Vargas, J.A.C.; Resende, K.T.; Teixeira, I.A.M.A. Energy Requirements and Efficiency of Energy Utilization in Growing Dairy Goats of Different Sexes. J. Dairy Sci. 2020, 103, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, R.R.; Stewart, D.R. Grazer or Browser: A Classification Based On The Stomach-Structure and Feeding Habits of East African Ruminants. Biology 1972, 36, 226–240. [Google Scholar] [CrossRef]
- Coppock, D.L.; Ellis, J.E.; Swift, D.M. Livestock Feeding Ecology and Resource Utilization in a Nomadic Pastoral Ecosystem. J. Appl. Ecol. 1986, 23, 573–583. [Google Scholar] [CrossRef]
- Hofmann, R.R. Evolutionary Steps of Ecophysiological Adaptation and Diversification of Ruminants: A Comparative View of Their Digestive System. Oecologia 1989, 78, 443–457. [Google Scholar] [CrossRef]
- Howe, J.C.; Barry, T.N.; Popay, A.I. Voluntary Intake and Digestion of Gorse (Ulex europaeus) by Goats and Sheep. J. Agric. Sci. 1988, 111, 107–114. [Google Scholar] [CrossRef]
- Macedo Júnior, G.L.; Ferreira, M.I.C.; Borges, I.; Silva, V.B.; Couto, J.R.L.; Cavalcanti, L.F.L. Consumo e Digestibilidade Aparente das Frações Fibrosas por Ovelhas Gestantes Submetidas ou Não à Restrição Nutricional. Braz. J. Anim. Health Produc. 2010, 11, 179–192. [Google Scholar] [CrossRef][Green Version]
- Forbes, J.M. The Voluntary Food Intake of Farm Animals; Butterworths: London, UK, 1986; p. 206. [Google Scholar]
- Tsiplakou, E.; Hadjigeorgiou, I.; Sotirakoglou, K.; Zervas, G.P. Differences in Mean Retention Time of Sheep and Goats Under Controlled Feeding Practices. Small Rumin. Res. 2011, 95, 48–53. [Google Scholar] [CrossRef]
- Huston, J.E.; Rector, B.S.; Ellis, W.C.; Allen, M.L. Dynamics of Digestion in Cattle, Sheep, Goats and Deer. J. Anim. Sci. 1986, 62, 208–215. [Google Scholar] [CrossRef]
- Lechner-Doll, M.; Rutagwenda, T.; Schwartz, H.J.; Schultka, W.; Engelhardt, W.V. Seasonal Changes of Ingesta Mean Retention Time and Fore-Stomach Fluid Volume in Indigenous Camels, Cattle, Sheep and Goats Grazing a Thorn Bush Savannah Pasture in Kenya. J. Agric. Sci. 1990, 115, 409–420. [Google Scholar] [CrossRef]
- Gherardi, S.G.; Black, J.L.; Colebrook, W.F. Effect of Palatability on Voluntary Feed Intake by Sheep II. The Effect of Altering the Palatability of a Wheaten Hay on Long-term Intake and Preference. Aust. J. Agric. Res. 1991, 42, 585–589. [Google Scholar] [CrossRef]
- Hadjipanayiotou, M. Fractional Outflow of Soybean Meal from the Rumen, Water Intake and Ruminal Fermentation Pattern in Sheep and Goats at Different Seasons and Age Groups. Small Rumin. Res. 1995, 17, 137–143. [Google Scholar] [CrossRef]
- Hadjigeorgiou, I.E.; Gordon, I.J.; Milne, J.A. Intake, Digestion and Selection of Roughage with Different Staple Lengths by Sheep and Goats. Small Rum. Res. 2003, 43, 117–132. [Google Scholar] [CrossRef]
- Silanikove, N. The Physiological Basis of Adaptation in Goats to Harsh Environments. Small Rum. Res. 2000, 35, 181–193. [Google Scholar] [CrossRef]
- Blaxter, K.L.; Graham, N.M.C.; Wainman, F.W. Some Observations on the Digestibility of Food by Sheep, and on Related Problems. Br. J. Nutr. 1956, 10, 69–91. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.A. Symposium on Forage Utilization: Nutritive Value of Forage as Affected by Physical Form. Part I. General Principles Involved with Ruminants and Effect of Feeding Pelleted or Wafered Forage to Dairy Cattle. J. Anim. Sci. 1964, 230, 238. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods of Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition. J. Dairy Sci. 1991, 74, 583–3597. [Google Scholar] [CrossRef]
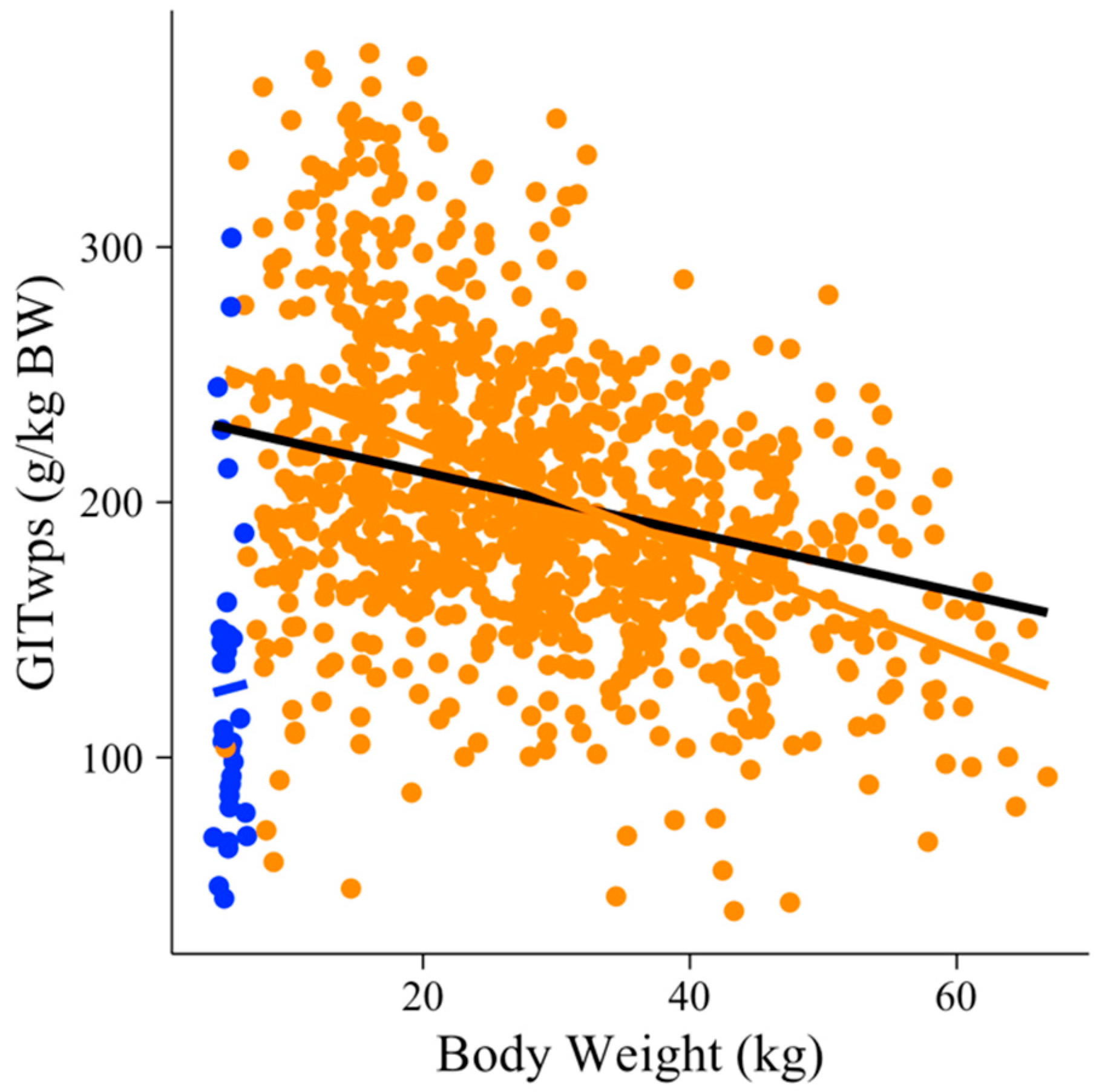
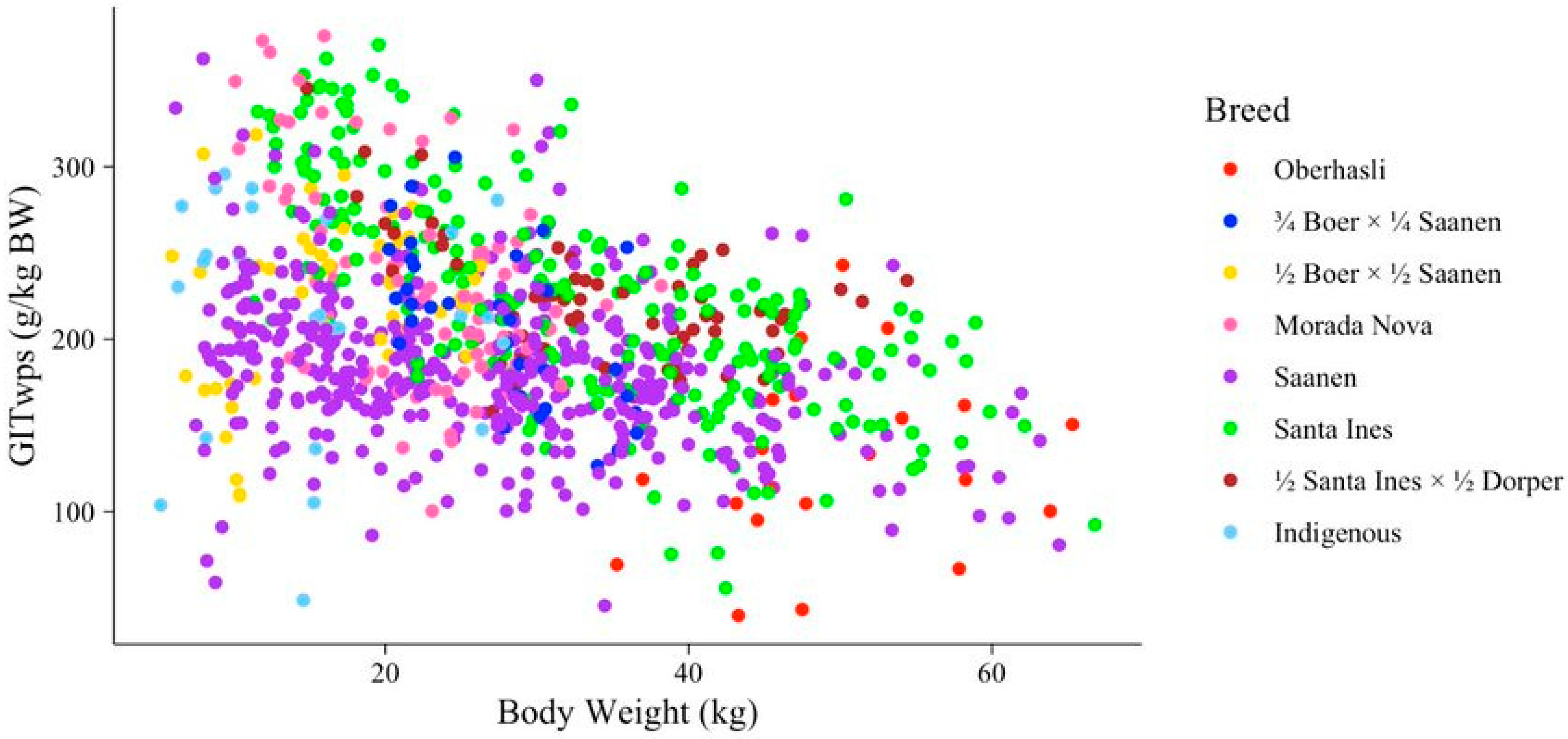

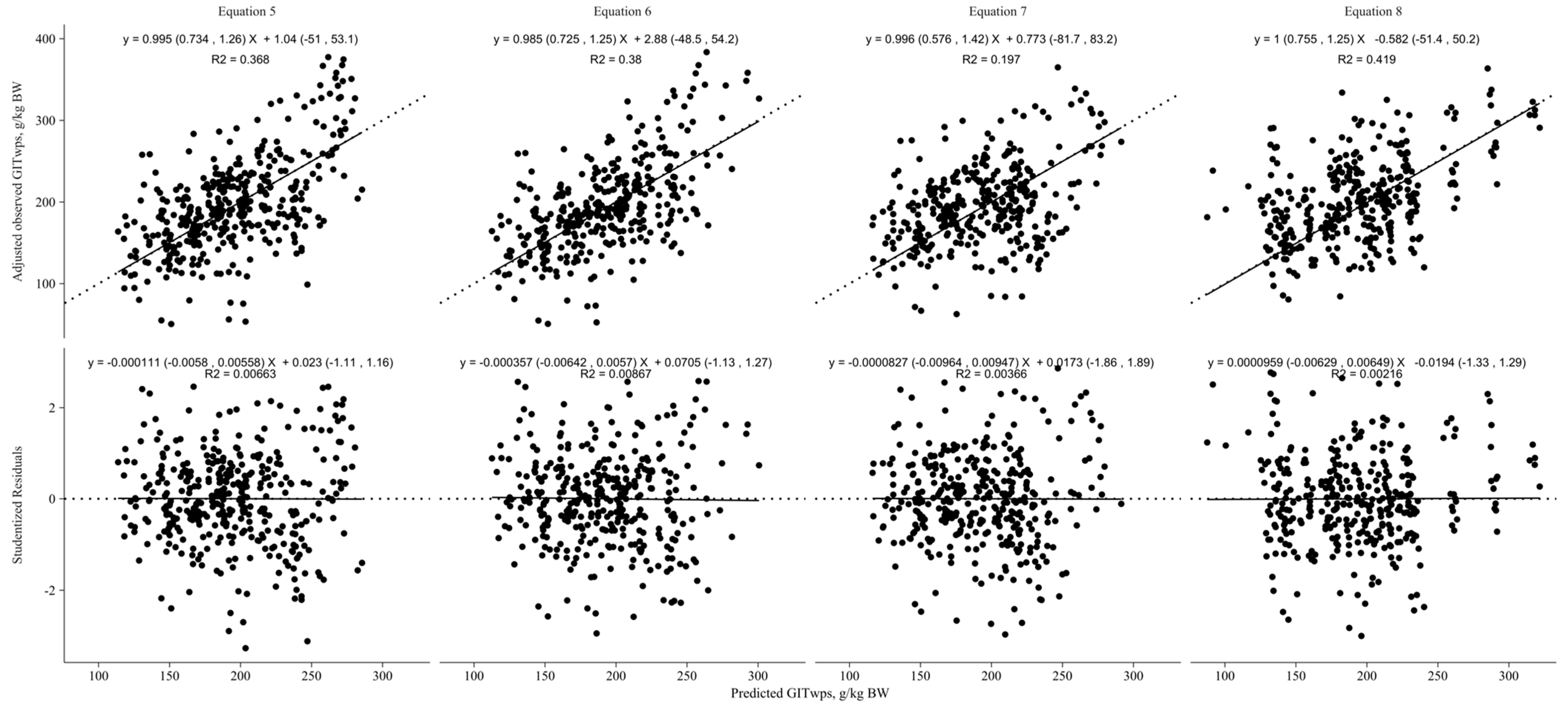

| Study | Sex 1 | Breed 2 | Species | Diet 3 | n 4 | BW 5 (kg) | GITwps 6 (g/kg BW) | DMI 7 (g/kg BW) | NDFI 8 (g/kg BW) | NDFI (g/g DMI) |
|---|---|---|---|---|---|---|---|---|---|---|
| [21] | FM | S | Goat | Suckling | 12 | 4.6–6.8 | 49.5–303.6 | – | – | – |
| [22] | M | S | Goat | Suckling | 8 | 5.1–5.7 | 44.7–105.9 | – | – | – |
| [23] | M | I | Goat | Suckling | 6 | 4.9–6.6 | 98.3–187.9 | – | – | – |
| [24] | M | I | Goat | Suckling | 4 | 4.9–5.1 | 144.9–146.2 | – | – | – |
| [25] | M | BS | Goat | Suckling | 6 | 4.3–6.7 | 68.7–160.8 | – | – | – |
| [26] | CFM | S | Goat | 100, 75, or 50% | 55 | 28–47 | 103.7–243.8 | 14.9–26 | 4.6–7.7 | 0.28–0.33 |
| [21] | CFM | S | Goat | 100, 75, or 50% | 46 | 6–16.7 | 163–362.8 | 12–26.9 | 3.9–8 | 0.31 |
| [27] | M | BBS | Goat | 100, 70, or 40% | 21 | 21–36.6 | 126.6–305.6 | 18–34 | 7–13 | 0.37–0.41 |
| [28] | C | S | Goat | 100, 70, or 40% | 27 | 21–35.5 | 100–286.6 | 21–41 | 8.9–17 | 0.41 |
| [29] | CFM | S | Goat | 100, 75, or 50% | 54 | 17–34 | 86–239 | 11- 30.5 | 3.7–10 | 0.33 |
| [30] | F | S | Goat | 100, 70, or 40% | 15 | 24.8–46 | 45.5–247.6 | 12–28 | 3.7–8.7 | 0.31 |
| [31] | M | S | Goat | 100, 70, or 40% | 18 | 29–51 | 103–319.8 | 15–31 | 4.7–9.8 | 0.31–0.34 |
| [32] 9 | F | S or OB | Goat | 100% | 42 | 33–65 | 39.8–243 | 15–31 | 6.4–13 | 0.42–0.46 |
| [22] | M | S | Goat | 100, 70, or 40% | 27 | 9.6–21.6 | 165.7–248.6 | 13–25.9 | 6.7–13 | 0.51 |
| [23] | M | I | Goat | 100%, or TM20 | 12 | 8.8–27.8 | 147.7–295.7 | 17–23.6 | 9–12.8 | 0.54 |
| [24] | M | I | Goat | 100%, or TM20 | 16 | 5–16.9 | 48.6–277 | 0.5–28 | 0.3–15 | 0.54 |
| [33] | C | S | Goat | 100%, or MT | 36 | 9.7–31 | 124.9–243 | 14–42.8 | 5–16 | 0.38 |
| [25] | M | BS | Goat | 100, 70, or 40% | 18 | 5.9–16 | 170–318.6 | 3.8–18.9 | 1.5–7 | 0.38 |
| [25] | M | BS | Goat | 100, 70, or 40% | 21 | 14.5–26 | 190–295 | 22–47.8 | 8.6–19 | 0.35–0.40 |
| [34] | M | MN | Sheep | 100% | 47 | 10–31.6 | 100–376 | 20.8–53 | 5.6–32 | 0.27–0.72 |
| [35] | F | SID | Sheep | 100, 70, or 40% | 24 | 20–42.6 | 167–307 | 15.8–32.9 | – | – |
| [36] 9 | F | SI | Sheep | 100% | 83 | 29–66.8 | 55.7–287 | 15–49.8 | 7–30.5 | 0.23–0.77 |
| [37] | M | SID | Sheep | 100, 70, or 40% | 30 | 27.6–54 | 174–251.7 | 16–32 | 8–15 | 0.46–0.50 |
| [38] | M | SI | Sheep | 100% | 34 | 11–33.5 | 167.5–370.9 | 19.9–33 | 6.6–20 | 0.26–0.73 |
| [39] | M | SI | Sheep | 100, 70, or 40% | 24 | 29.6–47 | 125.8–238.7 | 8.7–32.7 | 3.6–13.7 | 0.42 |
| [40] | CM | SI | Sheep | 100, 70, or 40% | 29 | 16–33 | 147–344 | 18–31 | 9–15 | 0.49 |
| [41] | CFM | MN | Sheep | 100, 70, or 40% | 35 | 13.7–38 | 137–276.8 | 19–32 | 8–14 | 0.44 |
| Equations | Species | Description 1 | n | RMSE 2 | RMSE% | CCC 3 |
|---|---|---|---|---|---|---|
| (5) | Goat | GITwps (g/kg BW) = 223.85 (±14.38) − 1.36 (±0.42) × BW (kg) | 390 | 43.01 | 22.37 | 0.60 |
| Sheep | GITwps (g/kg BW) = 309.22 (±21.47) − 2.77 (±0.58) × BW (kg) | |||||
| (6) | Goat | GITwps (g/kg BW) = 225.04 (±21.14) − 1.42 (±0.42) × BW (kg) + 0.10 (±0.66) × DMI (g/kg BW) | 365 | 41.12 | 21.39 | 0.65 |
| Sheep | GITwps (g/kg BW) = 399.55 (±33.69) − 3.07 (±0.62) × BW (kg) − 2.88 (±0.84) × DMI (g/kg BW) | |||||
| (7) | Goat and Sheep | GITwps (g/kg BW) = 200.77 (±16.7) − 1.15 (±0.35) × BW (kg) + 2.84 (±0.66) × NDFI (g/kg BW) | 357 | 41.96 | 21.82 | 0.62 |
| (8) | Goat and Sheep | GITwps (g/kg BW) = 110.11 (±17.46) − 0.5 (±0.33) × BW (kg) + 238.05 (±21.8) × NDFI (g/g DMI) | 357 | 36.77 | 19.12 | 0.74 |
| Equations | Species | Description 1 | n | RMSE 2 | RMSE% | CCC 3 |
|---|---|---|---|---|---|---|
| (9) | Goat | GITwps (g/kg BW) = 525.5 (±1.18) × BW (kg)−0.33 (±0.05) | 390 | 42.11 | 21.90 | 0.62 |
| Sheep | GITwps (g/kg BW) = 653.8 (±1.05) × BW (kg)−0.33 (±0.05) | |||||
| (10) | Goat | GITwps (g/kg BW) = 573.9 (±1.2) × BW (kg)−0.39 (±0.05) × e(0.00461 (±0.003) × DMI (g/kg BW)) | 365 | 40.22 | 20.94 | 0.66 |
| Sheep | GITwps (g/kg BW) = 1185.8 (±1.14) × BW (kg)−0.39 (±0.05) × e(−0.014145 (±0.005) × DMI (g/kg BW)) | |||||
| (11) | Goat | GITwps (g/kg BW) = 430.04 (±1.23) × BW (kg)−0.29 (±0.05) × e(0.00788 (±0.004) × NDFI (g/kg BW)) | 357 | 41.48 | 21.58 | 0.64 |
| Sheep | GITwps (g/kg BW) = 512.2 (±1.06) × BW (kg)−0.29 (±0.05) × e(0.00788 (±0.004) × NDFI (g/kg BW)) | |||||
| (12) | Goat | GITwps (g/kg BW) = 185.2 (±1.27) × BW (kg)−0.13 (±0.06) × e(1.01 (±0.14) × NDFI (g/g DMI)) | 357 | 36.2 | 18.83 | 0.75 |
| Sheep | GITwps (g/kg BW) = 200.55 (±1.08) × BW (kg)−0.13 (±0.06) × e(1.01 (±0.14) × NDFI (g/g DMI)) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, P.R.; Gindri, M.; Macedo Junior, G.L.; Herbster, C.J.L.; Pereira, E.S.; Biagioli, B.; Teixeira, I.A.M.A. Modeling Gastrointestinal Tract Wet Pool Size in Small Ruminants. Animals 2023, 13, 2909. https://doi.org/10.3390/ani13182909
Ribeiro PR, Gindri M, Macedo Junior GL, Herbster CJL, Pereira ES, Biagioli B, Teixeira IAMA. Modeling Gastrointestinal Tract Wet Pool Size in Small Ruminants. Animals. 2023; 13(18):2909. https://doi.org/10.3390/ani13182909
Chicago/Turabian StyleRibeiro, Paola R., Marcelo Gindri, Gilberto L. Macedo Junior, Caio J. L. Herbster, Elzania S. Pereira, Bruno Biagioli, and Izabelle A. M. A. Teixeira. 2023. "Modeling Gastrointestinal Tract Wet Pool Size in Small Ruminants" Animals 13, no. 18: 2909. https://doi.org/10.3390/ani13182909
APA StyleRibeiro, P. R., Gindri, M., Macedo Junior, G. L., Herbster, C. J. L., Pereira, E. S., Biagioli, B., & Teixeira, I. A. M. A. (2023). Modeling Gastrointestinal Tract Wet Pool Size in Small Ruminants. Animals, 13(18), 2909. https://doi.org/10.3390/ani13182909








