Cardiorespiratory Effects and Desflurane Requirement in Dogs Undergoing Ovariectomy after Administration Maropitant or Methadone
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Study Subjects
2.3. Anesthetic and Analgesic Protocol/Management
2.4. Surgical Procedure
2.5. Data Collection and Evaluation Times
2.6. Recovery Quality and Postoperative Analgesic Management
2.7. Statistical Analysis
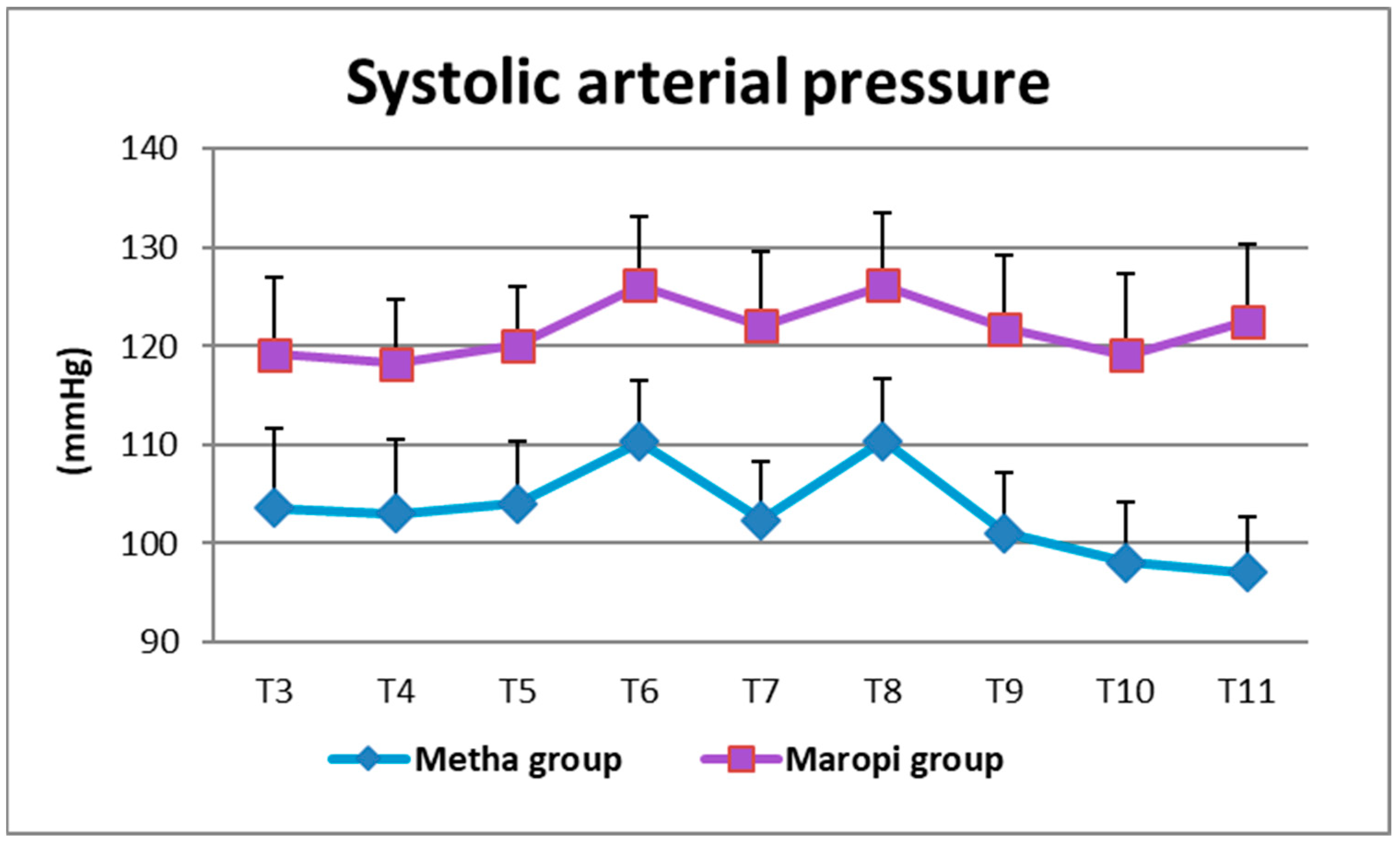
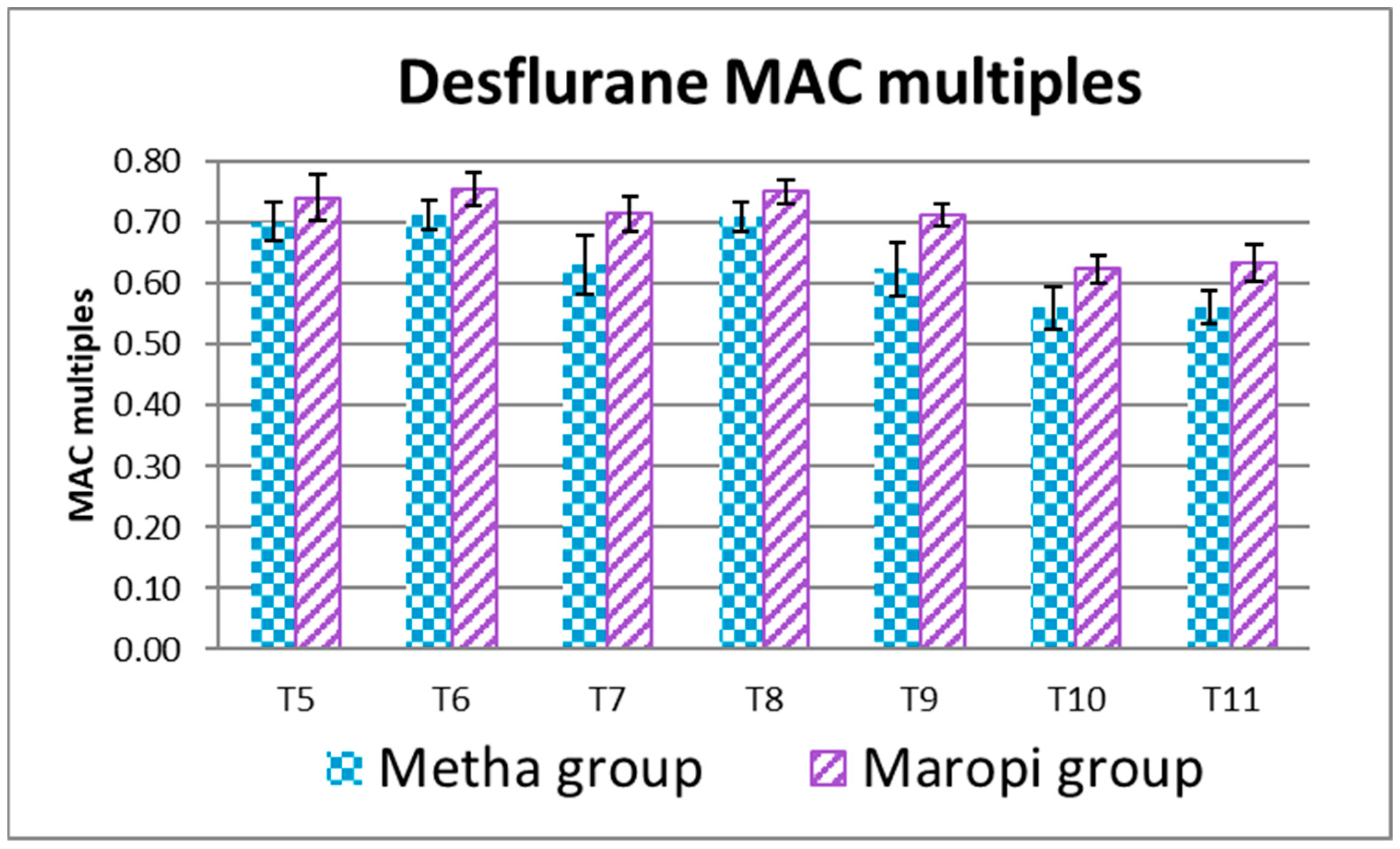
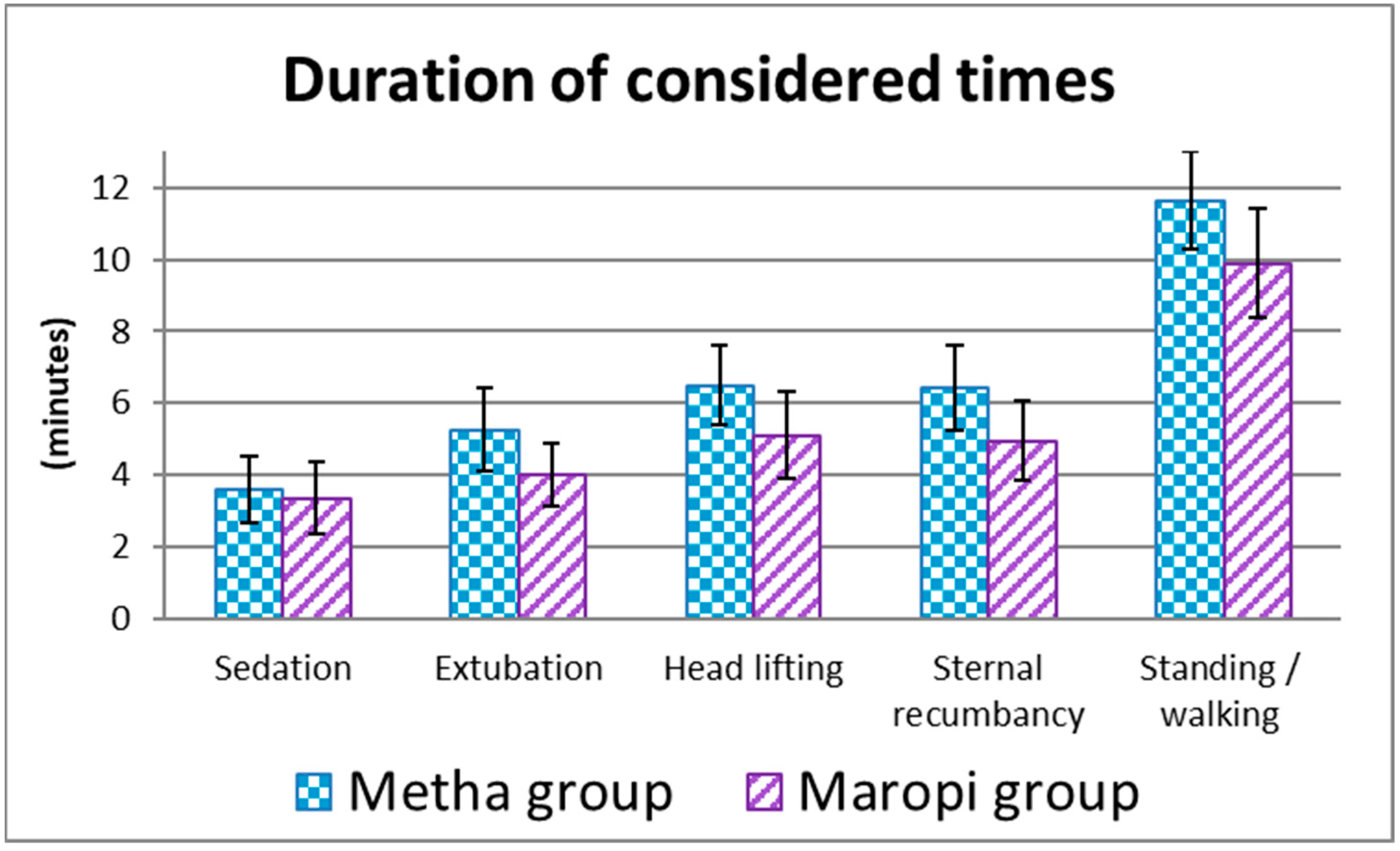
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lemmens, H.J.M. Pharmacokinetic–pharmacodynamic relationships for opioids in balanced anaesthesia. Clin. Pharmacokinet. 1995, 29, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Ilkiw, J.E. Balanced anesthetic techniques in dogs and cats. Clin. Tech. Small Anim. Pract. 1999, 14, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Columbano, N.; Secci, F.; Careddu, G.M.; Sotgiu, G.; Rossi, G.; Driessen, B. Effects of lidocaine constant rate infusion on sevoflurane requirement, autonomic responses, and postoperative analgesia in dogs undergoing ovariectomy under opioid-based balanced anesthesia. Vet. J. 2012, 193, 448–455. [Google Scholar] [CrossRef]
- Beverly, A.; Kaye, A.D.; Ljungqvist, O.; Urman, R.D. Essential Elements of Multimodal Analgesia in Enhanced Recovery After Surgery (ERAS) Guidelines. Anesthesiol. Clin. 2017, 35, e115–e143. [Google Scholar] [CrossRef]
- Fukui, S.; Ooyama, N.; Tamura, J.; Umar, M.A.; Ishizuka, T.; Itami, T.; Miyoshi, K.; Sano, T.; Yamashita, K. Interaction between maropitant and carprofen on sparing of the minimum alveolar concentration for blunting adrenergic response (MAC-BAR) of sevoflurane in dogs. J. Vet. Med. Sci. 2017, 79, 502–508. [Google Scholar] [CrossRef]
- Howard-Azzeh, M.; Pearl, D.L.; O’Sullivan, T.L.; Berke, O. The identification of risk factors contributing to accidental opioid poisonings in companion dogs using data from a North American poison control center (2006–2014). PLoS ONE 2020, 25, e0227701. [Google Scholar] [CrossRef]
- Hugonnard, M.; Leblond, A.; Keroack, S.; Cadoré, J.L.; Troncy, E. Attitudes and concerns of French veterinarians towards pain and analgesia in dogs and cats. Vet. Anaesth. Analg. 2004, 31, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Bradbrook, C.A.; Clark, L. State of the art analgesia- recent developments in pharmacological approaches to acute pain management in dogs and cats. Part 1. Vet. J. 2018, 238, 76–82. [Google Scholar] [CrossRef]
- Mwangi, W.E.; Mogoa, E.M.; Mwangi, J.N.; Mbuthia, P.G.; Mbugua, S.W. A systematic review of analgesia practices in dogs undergoing ovariohysterectomy. Vet. World 2018, 11, 1725–1735. [Google Scholar] [CrossRef]
- Casas-Alvarado, A.; Mota-Rojas, D.; Hernandez-Avalos, I.; Martinez-Burnes, J.; Rosas, M.E.; Miranda-Cortes, A.; Dominguez-Oliva, A.; Mora-Medina, P. Assessment of thermal response, cardiorespiratory parameters and post-operative analgesia in dogs undergoing ovariohysterectomy with defferent combination of epidural analgesia and isoflurane. J. Anim. Behav. Biometeorol. 2023, 11, e2023009. [Google Scholar] [CrossRef]
- Hernández-Avalos, I.; Alexander Valverde, A.; José Antonio Ibancovichi-Camarillo, J.A.; Sánchez-Aparicio, P.; Recillas-Morales, S.; Osorio-Avalos, J.; Rodríguez-Velázquez, D.; Miranda-Cortés, A.E. Clinical evaluation of postoperative analgesia, cardiorespiratory parameters and changes in liver and renal function tests of paracetamol compared to meloxicam and carprofen in dogs undergoing ovariohysterectomy. PLoS ONE 2020, 15, e0223697. [Google Scholar] [CrossRef]
- De la Puente-Redondo, V.A.; Siedek, E.M.; Benchaoui, H.A.; Tilt, N.; Rowan, T.G.; Clemence, R.G. The antiemetic efficacy of maropitant (Cerenia) in the treatment of ongoing emesis caused by a wide range of underlying clinical aetiologies in canine patients in Europe. J. Small Anim. Pract. 2007, 48, 93–98. [Google Scholar] [CrossRef]
- Conder, G.A.; Sedlacek, H.S.; Bouncher, J.F.; Clemence, R.G. Efficacy and safety of maropitant, a selective neurokinin 1 receptor antagonist, in two randomized clinical trials for prevention of vomiting due to motion sickness in dogs. J. Vet. Pharmacol. Ther. 2008, 31, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, D.S.; Kincaid, K.; Watkins, J.A.; Boucher, J.F.; Conder, G.A.; Eagleson, J.S.; Clemence, R.G. Safety and efficacy of injectable and oral maropitant, a selective neurokinin1 receptor antagonist, in a randomized clinical trial for treatment of vomiting in dogs. J. Vet. Pharmacol. Ther. 2008, 31, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Sedlacek, H.S.; Ramsey, D.S.; Boucher, J.F.; Eagleson, J.S.; Conder, G.A.; Clemence, R.G. Comparative efficacy of maropitant and selected drugs in preventing emesis induced by centrally or peripherally acting emetogens in dogs. J. Vet. Pharmacol. Ther. 2008, 31, 533–537. [Google Scholar] [CrossRef]
- Sakurada, T.; Katsumata, K.; Yogo, H.; Tan-No, K.; Sakurada, S.; Ohba, M.; Kisara, K. The neurokinin-1 receptor antagonist, sendide, exhibits antinociceptive activity in the formalin test. Pain 1995, 60, 175–180. [Google Scholar] [CrossRef]
- Laird, J.M.; Olivar, T.; Roza, C.; De Felipe, C.; Hunt, S.P.; Cervero, F. Deficits in visceral pain and hyperalgesia of mice with a disruption of the tachykinin NK1 receptor gene. Neuroscience 2000, 98, 345–352. [Google Scholar] [CrossRef]
- Ruggieri, M.R.; Filer-Maerten, S.; Hieble, J.P.; Hay, D.W. Role of neurokinin receptors in the behavioral effect of intravesical antigen infusion in guinea pig bladder. J. Urol. 2000, 164, 197–202. [Google Scholar] [CrossRef]
- Laird, J. Gut feelings about tachykinin NK1 receptor antagonists. Trends Pharmacol. Sci. 2001, 22, 169. [Google Scholar] [CrossRef]
- Okano, S.; Ikeura, Y.; Inatomi, N. Effects of tachykinin NK1 receptor antagonists on the viscerosensory response caused by colorectal distention in rabbits. J. Pharmacol. Exp. Ther. 2002, 300, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Greenwood-Van Meerveld, B.; Gibson, M.S.; Johnson, A.C.; Venkova, K.; Sutkowski-Markmann, D. NK1 receptor-mediated mechanisms regulate colonic hypersensitivity in the guinea pig. Pharmacol. Biochem. Behav. 2003, 74, 1005–1013. [Google Scholar] [CrossRef]
- Boscan, P.; Monnet, E.; Mama, K.; Twedt, D.C.; Congdon, J.; Steffey, E.P. Effect of maropitant, a neurokinin 1 receptor antagonist, on anesthetic requirements during noxious visceral stimulation of the ovary in dogs. Am. J. Vet. Res. 2011, 72, 1576–1579. [Google Scholar] [CrossRef]
- Plumb, D.C. Maropitant Citrate. In Plumb’s Veterinary Drug Handbook: Pocket, 7th ed.; Wiley-Blackwell: Ames, IA, USA, 2011; pp. 2192–2197. ISBN 978-0-4709-5965-7. Ebook ISBN: 978-0-9834-1810-8. [Google Scholar]
- Pang, D.S.J. Anesthetic and Analgesic Adjunctive Drugs. Section 2: Pharmacology. In Veterinary Anesthesia and Analgesia, 5th ed.; Lumb and Jones; Grimm, K.A., Lamont, L.A., Tranquilli, W.J., Greene, S.A., Robertson, S.A., Eds.; John Wiley & Sons: Hoboken, NJ, USA; Wiley-Blackwell: Ames, IA, USA, 2015; p. 248. ISBN 978-1-118-52620-0. [Google Scholar]
- Marquez, M.; Boscan, P.; Weir, H.; Vogel, P.; Twedt, D.C. Comparison of NK-1 Receptor Antagonist (Maropitant) to Morphine as a Pre-Anaesthetic Agent for Canine Ovariohysterectomy. PLoS ONE 2015, 10, e0140734. [Google Scholar] [CrossRef] [PubMed]
- Boscan, P.; Monnet, E.; Mama, K.; Twedt, D.C.; Congdon, J.; Eickhoff, J.C.; Steffey, E.P. A dog model to study ovary, ovarian ligament and visceral pain. Vet. Anaesth. Analg. 2011, 38, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Alvillar, B.M.; Boscan, P.; Mama, K.R.; Ferreira, T.H.; Congdon, J.; Twedt, D.C. Effect of epidural and intravenous use of the neurokinin-1 (NK-1) receptor antagonist maropitant on the sevoflurane minimum alveolar concentration (MAC) in dogs. Vet. Anaesth. Analg. 2012, 39, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Chi, T.T.; Kraus, B.L.H. The effect of intranenous maropitant on blood pressure in healthy awake and anesthetized dogs. PLoS ONE 2020, 15, e0229736. [Google Scholar] [CrossRef]
- Ko, J.C.; Fox, S.M.; Mandsager, R.E. Sedative and cardiorespiratory effects of medetomidine, medetomidine-butorphanol, and medetomidine-ketamine in dogs. J. Am. Vet. Med. Assoc. 2000, 216, 1578–1583. [Google Scholar] [CrossRef]
- Nishimura, L.T.; Auckburally, A.; Santilli, J.; Vieira, H.B.; Garcia, D.O.; Honsho, C.S.; de Mattos-Junior, E. Effects of dexmedetomidine combined with commonly administered opioids on clinical ariables in dogs. Am. J. Vet. Res. 2018, 79, 267–275. [Google Scholar] [CrossRef]
- Arenillas, M.; Canfràn, S.; Aguado, D.; Gomez de Segura, I.A. Sedative and analgesic effects of two subanaesthetic doses of ketamine in combination with methadone and a low dose of dexmedetomidine in healthy dogs. Vet. Anaesth. Analg. 2021, 48, 545–553. [Google Scholar] [CrossRef]
- Kersten, J.; Pagel, P.S.; Tessmer, J.P.; Roerig, D.L.; Schmeling, W.T.; Warltier, D.C. Dexmedetomidine alters the hemodynamic effects of desflurane and isoflurane in chronically instrumented dogs. Anesthesiology 1993, 79, 1022–1032. [Google Scholar] [CrossRef]
- Ingvast-Larsson, C.; Holgersson, A.; Bondesson, U.; Lagerstedt, A.-S.; Olsson, K. Clinical pharmacology of methadone in dogs. Vet. Anaesth. Analg. 2010, 37, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Amon, T.; Kästner, S.B.R.; Kietzmann, M.; Tünsmeyer, J. Plasma levels of a methadone constant rate infusion and their corresponding effects on thermal and mechanical nociceptive thresholds in dogs. BMC Vet. Res. 2021, 17, 1–13. [Google Scholar] [CrossRef] [PubMed]
- KuKanich, B.; Wiese, A.J. Opioids. In Veterinary Anesthesia and Analgesia, 5th ed.; Lumb and Jones; Grimm, K.A., Lamont, L.A., Tranquilli, W.J., Greene, S.A., Robertson, S.A., Eds.; John Wiley & Sons: Hoboken, NJ, USA; Wiley-Blackwell: Ames, IA, USA, 2015; pp. 218–219. ISBN 978-1-118-52623-1. [Google Scholar]
- Patel, S.S.; Goa, K.L. Desflurane. A review of its pharmacodynamic and pharmacokinetic properties and its efficacy in general anaesthesia. Drugs 1995, 50, 742–767. [Google Scholar] [CrossRef]
- Souza, A.P.; Guerrero, P.N.H.; Nishimori, C.T.; Danielli Parrilha, P.; Patto dos Santos, P.S.; Langenegger de Rezende, M.; Nunes, N. Cardiopulmonary and Acid–Base Effects of Desflurane and Sevoflurane in Spontaneously Breathing Cats. J. Feline Med. Surg. 2005, 7, 95–100. [Google Scholar] [CrossRef]
- Steffey, E.P.; Mama, K.R.; Brosnan, R.J. Inhalation anesthetics. In Veterinary Anesthesia and Analgesia, 5th ed.; Lumb and Jones; Grimm, K.A., Lamont, L.A., Tranquilli, W.J., Greene, S.A., Robertson, S.A., Eds.; John Wiley & Sons: Hoboken, NJ, USA; Wiley-Blackwell: Ames, IA, USA, 2015; pp. 297–322. ISBN 978-1-118-52623-1. [Google Scholar]
- Hammond, R.A.; Alibhai, H.I.K.; Walsh, K.P.; Clarke, K.W.; Holden, D.J.; White, R.N. Desflurane in the dog; Minimum alveolar concentration (MAC) alone and in combination with nitrous oxide. J. Vet. Anaesth. 1994, 21, 21–23. [Google Scholar] [CrossRef]
- Ebert, T.J.; Muzi, M. Sympathetic hyperactivity during desflurane anesthesia in healthy volunteers. A comparison with isoflurane. Anesthesiology 1993, 79, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Weiskopf, R.B.; Eger, E.I., 2nd; Noorani, M.; Daniel, M. Fentanyl, esmolol, and clonidine blunt the transient cardiovascular stimulation induced by desflurane in humans. Anesthesiology 1994, 81, 1350–1355. [Google Scholar] [CrossRef]
- Pacentine, G.G.; Muzi, M.; Ebert, T.J. Effects of fentanyl on sympathetic activation associated with the administration of desflurane. Anesthesiology 1995, 82, 823–831. [Google Scholar] [CrossRef]
- Grint, N.J.; Burford, J.; Dugdale, A.H.A. Does pethidine affect the cardiovascular and sedative effects of dexmedetomidine in dogs? J. Small Anim. Pract. 2009, 50, 62–66. [Google Scholar] [CrossRef]
- Wagner, M.C.; Hecker, K.G.; Pang, D.S.J. Sedation levels in dogs: A validation study. BMC Vet. Res. 2017, 13, 110. [Google Scholar] [CrossRef]
- Hampton, C.R.; Riebold, T.W.; Mandsager, R.E. Recovery characteristics of dogs following anesthesia induced with tiletamine-zolazepam, alfaxalone, ketamine-diazepam, or propofol and maintained with isoflurane. J. Am. Vet. Med. Assoc. 2019, 254, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Reid, J.; Nolan, A.M.; Hughes, J.M.L.; Lascelles, D.; Pawson, P.; Scott, E.M. Development of the short-form Glasgow Composite Measure Pain Scale (CMPS-SF) and derivation of an analgesic intervention score. Anim. Welf. 2007, 16, 97–104. [Google Scholar] [CrossRef]
- Della Rocca, G.; Colpo, R.; Reid, L.; Di Salvo, A.; Scott, M. Creation and validation of the italian version of the Glasgow Composite Measure Pain Scale—Short Form (ICMPS-SF). Vet. Ital. 2018, 54, 251–260. [Google Scholar] [CrossRef]
- Pypendop, B.H.; Ilkiw, J.E. Comparison of variability in cardiorespiratory measurements following desflurane anesthesia at a multiple of the minimum alveolar concentration for each dog versus a multiple of a single predetermined minimum alveolar concentration for all dogs in a group. Am. J. Vet. Res. 2006, 67, 1956–1961. [Google Scholar] [CrossRef]
- Feetham, C.H.; Barrett-Jolley, R. NK1-receptor-expressing paraventricular nucleus neurones modulate daily variation in heart rate and stress-induced changes in heart rate variability. Physiol. Rep. 2014, 2, e12207. [Google Scholar] [CrossRef]
- Zhou, J.X.; Liu, J. The effect of temperature on solubility of volatile anesthetics in human tissues. Anesth. Analg. 2001, 93, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Merin, R.G.; Bernard, J.M.; Doursout, M.F.; Cohen, M.; Chelly, J.E. Comparison of the effects of isoflurane and desflurane on cardiovascular dynamics and regional blood flow in the chronically instrumented dog. Anesthesiology 1991, 74, 568–574. [Google Scholar] [CrossRef]
- Eger, E.I., 2nd. New Inhaled Anesthetics. Anesthesiology 1994, 80, 906–922. [Google Scholar] [CrossRef]
- Murphy, K.M.; Rishniw, M.; Silverstein, D. Use of vasopressors for treatment of vasodilatory hypotension in dogs and cats by Diplomates of the American College of Veterinary Emergency and Critical Care. J. Vet. Emerg. Crit. Care 2022, 32, 714–722. [Google Scholar] [CrossRef]
- Ko, J.C.; Weil, A.B.; Inoue, T. Effects of carprofen and morphine on the minimum alveolar concentration of isoflurane in dogs. J. Am. Anim. Hosp. Assoc. 2009, 45, 19–23. [Google Scholar] [CrossRef]
- Ko, J.C.H.; Kriminis, R. Anesthetic monitoring devices to use e what the results mean. Today’s Vet. Pract. 2012, 2, 23–31. [Google Scholar]
- Noel-Morgan, J.; Muir, W.W. Anesthesia-associated relative hypovolemia: Mechanisms, monitoring, and treatment considerations. Front. Vet. Sci. 2018, 5, 53. [Google Scholar] [CrossRef]
- Luke, D.R.; Tomaszewski, K.; Damle, B.; Schlamm, H.T. Review of the basic and clinical pharmacology of sulfobutylether-β-cyclodextrin (SBECD). J. Pharm. Sci. 2010, 99, 3291–3301. [Google Scholar] [CrossRef]
- Wheeler, B.J.; Taylor, B.J. Successful management of allergy to the insulin excipient metacresol in a child with type 1 diabetes: A case report. J. Med. Case Rep. 2012, 6, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Rosseels, M.L.A.; Delaunois, A.G.; Hanon, E.; Guillaume, P.J.P.; Martin, F.D.C.; van den Dobbelsteen, D.J. Hydroxypropyl-β-cyclodextrin impacts renal and systemic hemodynamics in the anesthetized dog. Regul. Toxicol. Pharmacol. 2013, 67, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Keogh, T.P. Contact Points. J. Am. Dent. Assoc. 1999, 130, 467–468. [Google Scholar] [CrossRef] [PubMed]
- Modi, K.D.; Gadge, P.V.; Jain, P.; Pawar, S.; Shah, R.D.; Ingole, S.A.; Jain, R. Clinical challenges with excipients in insulin formulations and role of concentrated insulin. Int. J. Basic Clin. Pharmacol. 2019, 8, 821–826. [Google Scholar] [CrossRef]
- Kraus, B.L.H. Spotlight on the perioperative use of maropitant citrate. Vet. Med. 2017, 8, 41–51. [Google Scholar] [CrossRef]
- Wamaitha, M.N.; Mogoa, E.M.; Mande, J.D. Evaluation of anesthesia produced by ketofol in acepromazine- or medetomidine-sedated dogs. J. Adv. Vet. Anim. Res 2019, 6, 215–221. [Google Scholar] [CrossRef]
- Petrenko, A.B.; Yamakura, T.; Askalany, A.R.; Kohno, T.; Sakimura, K.; Baba, H. Effects of ketamine on acute somatic nociception in wild-type and N-methyl-D-aspartate (NMDA) receptor 31 subunit knockout mice. Neuropharmacology 2005, 50, 741–747. [Google Scholar] [CrossRef]
- Berry, S.H. Injectable anesthetics. In Veterinary Anethesia and Analgesia, 5th ed.; Lumb and Jones; Grimm, K.A., Lamont, L.A., Tranquilli, W.J., Greene, S.A., Robertson, S.A., Eds.; John Wiley & Sons: Hoboken, NJ, USA; Wiley Blackwell: Ames, IA, USA, 2015; p. 284. ISBN 978-1-118-52623-1. [Google Scholar]
- Soares, P.C.L.R.; Corrêa, J.M.X.; Niella, R.V.; de Oliveira, J.N.S.; Costa, B.A.; Silva Junior, A.C.; Sena, A.S.; Pinto, T.M.; Alexandre, D.; Munhoz, A.D.; et al. Continuous Infusion of Ketamine and Lidocaine Either with or without Maropitant as an Adjuvant Agent for Analgesia in Female Dogs Undergoing Mastectomy. Vet. Med. Int. 2021, 26, 4747301. [Google Scholar] [CrossRef] [PubMed]
- White, D.M.; Mair, A.R.; Martinez-Taboada, F. Opioid-free anaesthesia in three dogs. Open Vet. J. 2017, 7, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Beloeil, H. Opioid-free anesthesia. Best Pract. Res. Clin. Anaesthesiol. 2019, 33, 353–360. [Google Scholar] [CrossRef] [PubMed]






| Evaluation Time Points | |
|---|---|
| T0 | Baseline values |
| T1 | Ten minutes after administration of premedication |
| T2 | Shortly before induction |
| T3 | Immediately after intubation |
| T4 | Twenty minutes after EtDes 5.5% |
| T5 | Skin incision |
| T6 | Right ovarian ligament traction force |
| T7 | Right ovarian ligament resection |
| T8 | Left ovarian ligament traction force |
| T9 | Left ovarian ligament resection |
| T10 | Fascia suture |
| T11 | Skin suture |
| Category | Expressions | Score |
|---|---|---|
| Spontaneous posture | Standing | 0 |
| Tired but standing | 1 | |
| Lying but able to rise | 2 | |
| Lying but difficulty rising | 3 | |
| Unable to rise | 4 | |
| Palpebral Reflex | Brisk | 0 |
| Slow but with full corneal sweep | 1 | |
| Slow but with only partial corneal sweep | 2 | |
| Absent | 3 | |
| Eye position | Central | 0 |
| Rotated forwards/downwards but not obscured by third eyelid | 1 | |
| Rotated forwards/downwards and obscured by third eyelid | 2 | |
| Jaw and tongue relaxation | Normal jaw tone/(strong gag reflex) | 0 |
| Reduced tone, (but still moderate gag reflex) | 1 | |
| Much reduced tone, slight gag reflex | 2 | |
| Loss of jaw tone and no gag reflex | 3 | |
| Response to noise (handclap) | Normal startle reaction (head turn towards noise/cringe) | 0 |
| Reduced startle reaction (reduced head turn/minimal cringe) | 1 | |
| Minimal startle reaction | 2 | |
| Absent reaction | 3 | |
| Resistance when laid into lateral recumbency | Much struggling, perhaps not allowing this positioning | 0 |
| Some struggling, but allowing this positioning | 1 | |
| Minimal struggling/permissive | 2 | |
| No struggling | 3 | |
| General appearance attitude | Excitable | 0 |
| Awake and normal | 1 | |
| Tranquil | 2 | |
| Stuporous | 3 |
| Category | Expressions | Score |
|---|---|---|
| Excellent | Intubation successful in one attempt without physical reaction to intubation | 0 |
| Good | Intubation successful in one attempt with physical response to intubation | 1 |
| Satisfactory | Intubation successful after more than one attempt with or without physical response to intubation | 2 |
| Poor | Intubation impossible | 3 |
| Factors | Assessments | Score |
|---|---|---|
| Struggling | None | 0 |
| Transient, easily calmed by the investigator’s voice | 1 | |
| Prolonged (>1 min) | 2 | |
| Persistent (or requiring restraint) | 3 | |
| Excitement | None | 0 |
| Transient, easily calmed by the investigator’s voice | 1 | |
| Prolonged (>1 min) | 2 | |
| Persistent (or requiring restraint) | 3 | |
| Paddling | None | 0 |
| Transient, easily calmed by the investigator’s voice | 1 | |
| Prolonged (>1 min) | 2 | |
| Persistent (or requiring restraint) | 3 | |
| Flailing | None | 0 |
| Transient, easily calmed by the investigator’s voice | 1 | |
| Prolonged (>1 min) | 2 | |
| Persistent (or requiring restraint) | 3 | |
| Vocalization | None | 0 |
| Transient, easily calmed by the investigator’s voice | 1 | |
| Prolonged (>1 min) | 2 | |
| Persistent (or requiring restraint) | 3 | |
| Administration of rescue drugs | Not given | 0 |
| Given | 3 |
| SHORT FORM OF THE GLASGOW COMPOSITE PAIN SCALE | |||
| Dog’s name _______________________________________________ | |||
| Hospital Number _________ | Date / / | Time __________ | |
| Surgery Yes/No (delete as appropriate) | |||
| Procedure or Condition ___________________________________________ | |||
| In the sections below please circle the appropriate score in each list and sum these to give the total score | |||
| |||
| (i) | (ii) | ||
| Quiet | 0 | Ignoring any wound or painful area | 0 |
| Crying or whimpering | 1 | Looking at wound or painful area | 1 |
| Groaning | 2 | Licking wound or painful area | 2 |
| Screaming | 3 | Rubbing wound or painful area | 3 |
| Chewing wound or painful area | 4 | ||
| __________________________________________________________________________ | |||
| In the case of spinal, pelvic or multiple limb fractures, or where assistance is required to aid locomotion do not carry out section B and proceed to C Please tick if this is the case ◻ then proceed to C | |||
|
| ||
| When the dog rises/walks is it? | Does it? | ||
| (iii) | (iv) | ||
| Normal | 0 | Do nothing | 0 |
| Lame | 1 | Look round | 1 |
| Slow or reluctant | 2 | Flinch | 2 |
| Stiff | 3 | Growl or guard area | 3 |
| It refuses to move | 4 | Snap | 4 |
| Cry | 5 | ||
| __________________________________________________________________________ | |||
| |||
| Is the dog? | Is the dog? | ||
| (v) | (vi) | ||
| Happy and content or happy and bouncy | 0 | Comfortable | 0 |
| Quiet | 1 | Unsettled | 1 |
| Indifferent or non-responsive to surroundings | 2 | Restless | 2 |
| Nervous or anxious or fearful | 3 | Hunched or tense | 3 |
| Depressed or non-responsive to stimulation | 4 | Rigid | 4 |
| © University of Glasgow | Total Score (i + ii + iii + iv + v + vi) = _______ | ||
| Maropi Group | Metha Group | |||
|---|---|---|---|---|
| VT (mL) | VM (Lt) | VT (mL) | VM (Lt) | |
| T3 | 192.13 ± 31.78 | 1.93 ± 2.47 | 195.13 ± 34.81 | 1.90 ± 3.44 |
| T4 | 192.72 ± 30.74 | 1.90 ± 3.91 | 195.72 ± 33.77 | 1.87 ± 2.88 |
| T5 | 193.08 ± 29.63 | 1.88 ± 2.15 | 196.08 ± 32.66 | 1.85 ± 3.12 |
| T6 | 191.75 ± 26.08 | 1.95 ± 2.95 | 194.75 ± 29.11 | 1.92 ± 2.92 |
| T7 | 193.23 ± 31.78 | 1.90 ± 2.14 | 196.23 ± 34.81 | 1.87 ± 3.11 |
| T8 | 191.00 ± 37.14 | 1.97 ± 3.97 | 194.00 ± 30.17 | 1.94 ± 2.94 |
| T9 | 191.65 ± 31.78 | 1.91 ± 3.12 | 195.65 ± 34.81 | 1.88 ± 3.09 |
| T10 | 193.17 ± 30.78 | 1.89 ± 2.79 | 196.17 ± 34.81 | 1.86 ± 2.77 |
| T11 | 194.13 ± 24.34 | 1.88 ± 2.42 | 197.13 ± 27.37 | 1.86 ± 3.40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cubeddu, F.; Masala, G.; Sotgiu, G.; Mollica, A.; Versace, S.; Careddu, G.M. Cardiorespiratory Effects and Desflurane Requirement in Dogs Undergoing Ovariectomy after Administration Maropitant or Methadone. Animals 2023, 13, 2388. https://doi.org/10.3390/ani13142388
Cubeddu F, Masala G, Sotgiu G, Mollica A, Versace S, Careddu GM. Cardiorespiratory Effects and Desflurane Requirement in Dogs Undergoing Ovariectomy after Administration Maropitant or Methadone. Animals. 2023; 13(14):2388. https://doi.org/10.3390/ani13142388
Chicago/Turabian StyleCubeddu, Francesca, Gerolamo Masala, Giovanni Sotgiu, Alessandra Mollica, Sylvia Versace, and Giovanni Mario Careddu. 2023. "Cardiorespiratory Effects and Desflurane Requirement in Dogs Undergoing Ovariectomy after Administration Maropitant or Methadone" Animals 13, no. 14: 2388. https://doi.org/10.3390/ani13142388
APA StyleCubeddu, F., Masala, G., Sotgiu, G., Mollica, A., Versace, S., & Careddu, G. M. (2023). Cardiorespiratory Effects and Desflurane Requirement in Dogs Undergoing Ovariectomy after Administration Maropitant or Methadone. Animals, 13(14), 2388. https://doi.org/10.3390/ani13142388






