Dynamic Analysis and Optimal Control of Fractional Order African Swine Fever Models with Media Coverage
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Models Formulation
2.2. Methods
- (i)
- The next generation matrix method is used to obtain the basic reproduction number.
- (ii)
- The Descartess rule of signs is used to determine the existence of a positive equilibrium.
- (iii)
- The eigenvalue method, Routh-Hurwitz criteria and LaSalle’s invariance principle are used to prove the stability of two equilibriums.
- (iv)
- The Pontryagin’s maximum principle is used to derive the formula for the optimal solution of System (2).
- (v)
- The Adams-type predictor corrector method and MATLAB software are used for the numerical simulations.
3. Results
3.1. Qualitative Analysis Results for System (1)
- (i)
- If , for , then is non-decreasing for each .
- (ii)
- If , for , then is non-increasing for each .
- (i)
- When , the endemic equilibrium is locally asymptotically stable, provided that
- (ii)
- When , the above conditions are sufficient but not necessary for the local asymptotic stability of the endemic equilibrium . In fact, is still locally asymptotically stable if all eigenvalues of Equation (A4) satisfy
3.2. Examples and Numerical Simulation Results for System (1)
- (i)
- (ii)
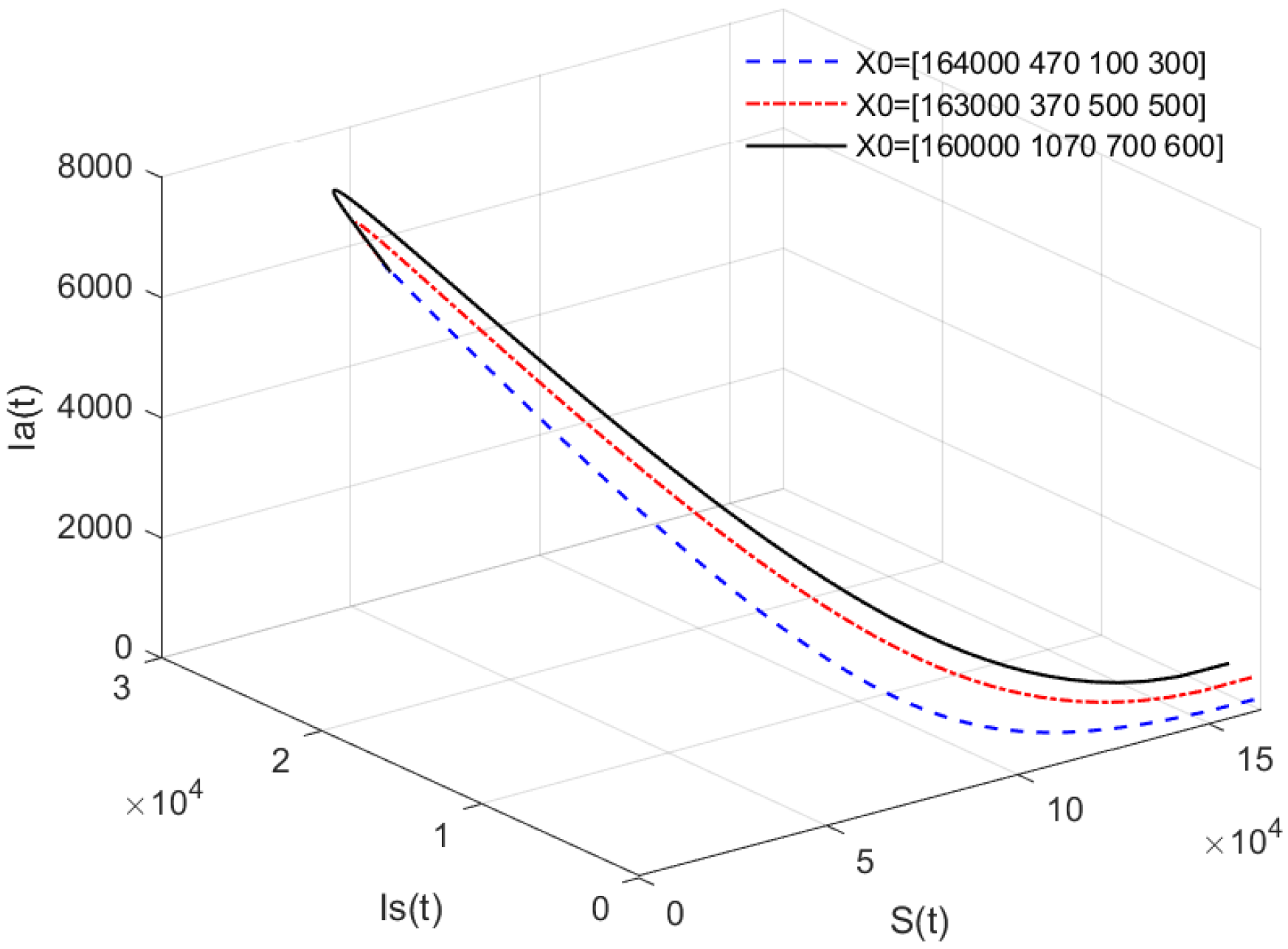
- In Figure 2, the value of α is fixed to , and different initial values are taken. = [164,000, 470, 100, 300], [164,000, 400, 170, 300], [163,000, 370, 500, 500], [160,000, 1070, 700, 600]. Figure 2 indicates that different initial values do not affect the stability of the disease-free equilibrium of system (1).
- (i)
- (ii)
- (i)
- (ii)
- (i)
- Figure 1 and Figure 2 show that if , then the disease-free equilibrium is always stable. If the basic reproduction number , that is, the number of healthy pigs infected by a diseased pig during its average disease period does not exceed 1, then the disease will eventually disappear, and this result is consistent with reality. The value of α can affect the speed towards the equilibrium. The initial values will not affect the stability, which is in line with Theorems 1 and 4.
- (ii)
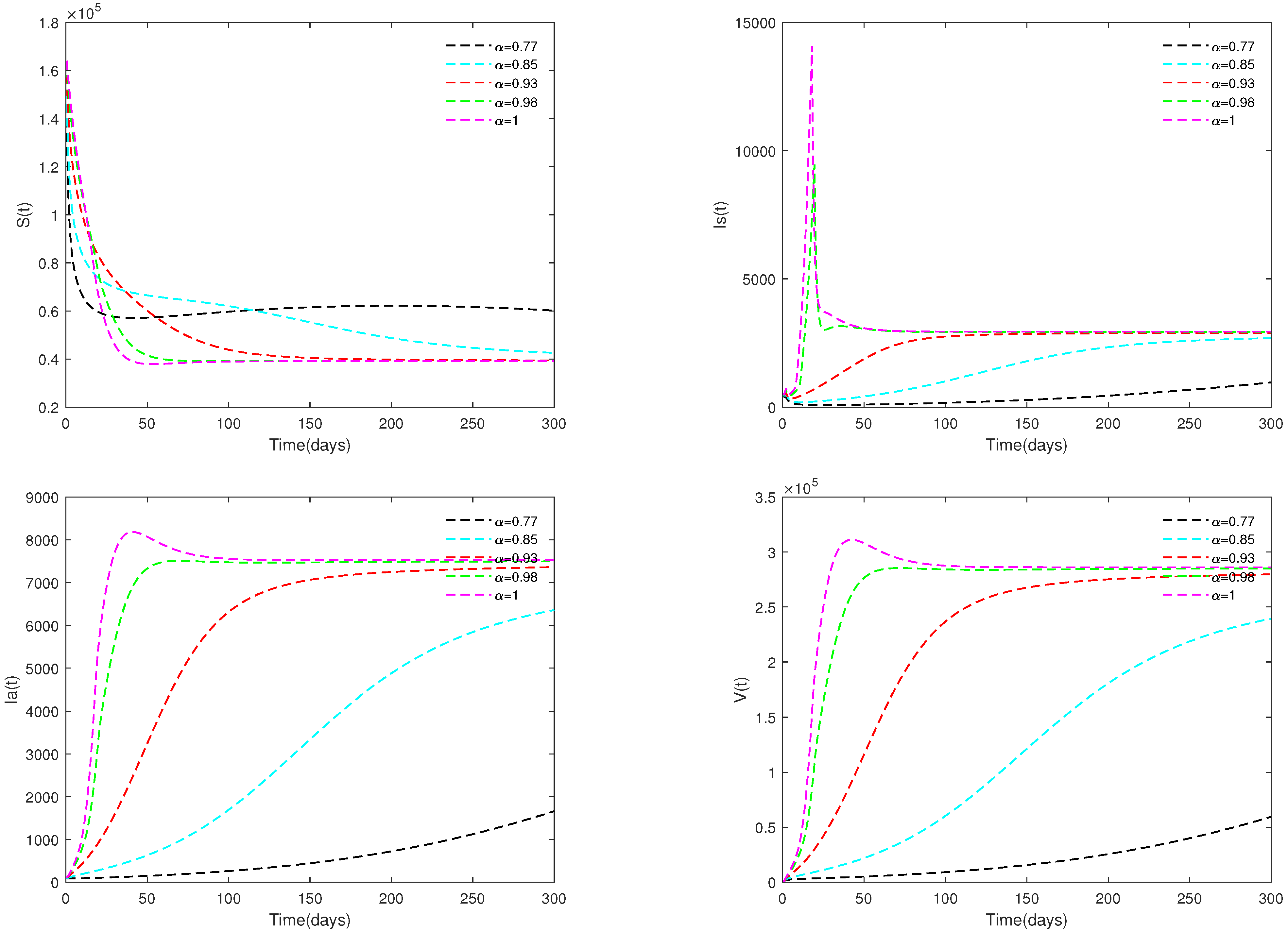
- Figure 3 and Figure 4 indicate that if , then the disease-free equilibrium is unstable and the endemic equilibrium exists. If the basic reproduction number , that is, if the number of healthy pigs infected by a diseased pig during its average disease period is more than 1, then the disease will break out in this region and become an endemic. The value of α will affect the speed towards the endemic equilibrium . The initial values will not affect the stability, which is in accordance with Theorems 1 and 2.
- (iii)
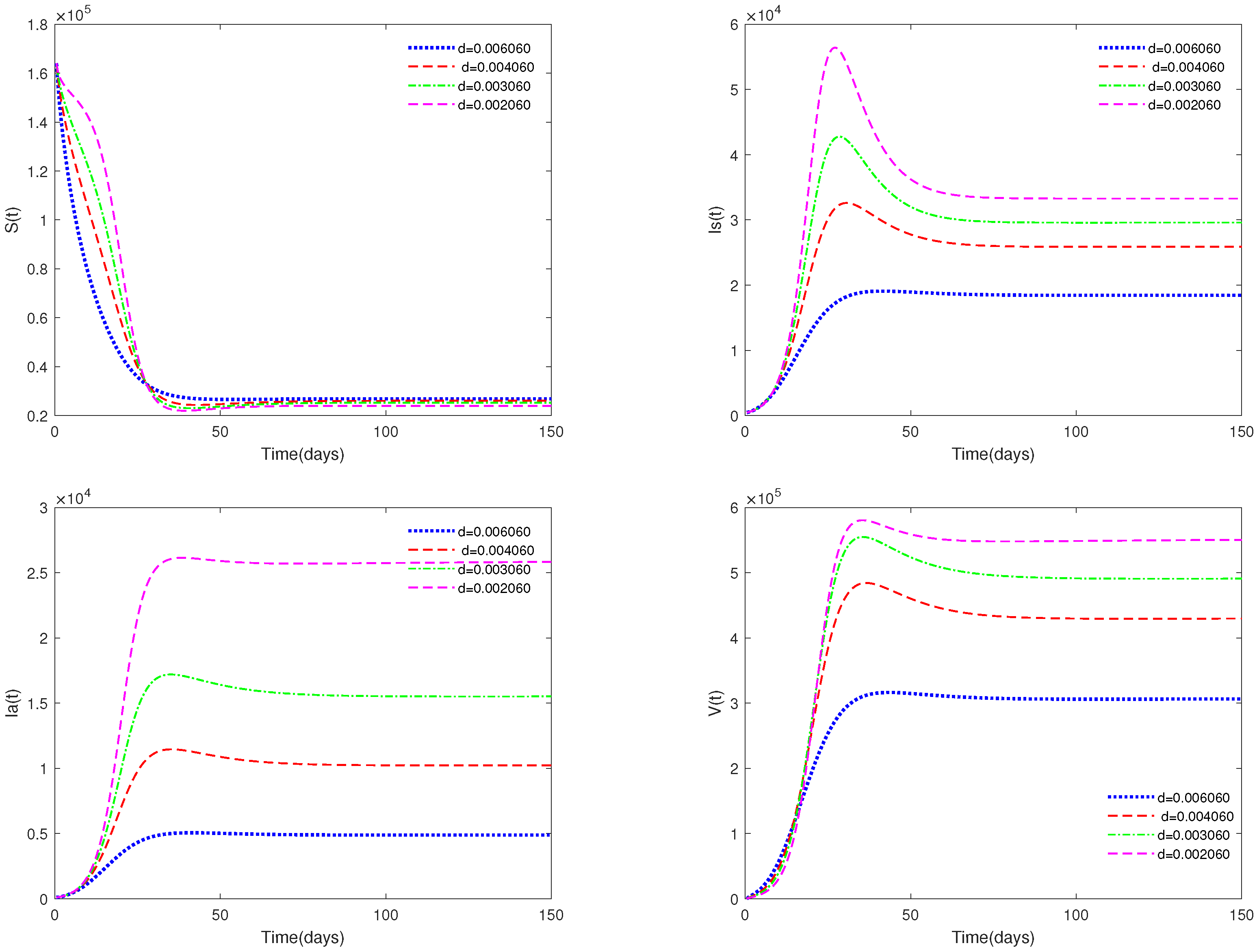
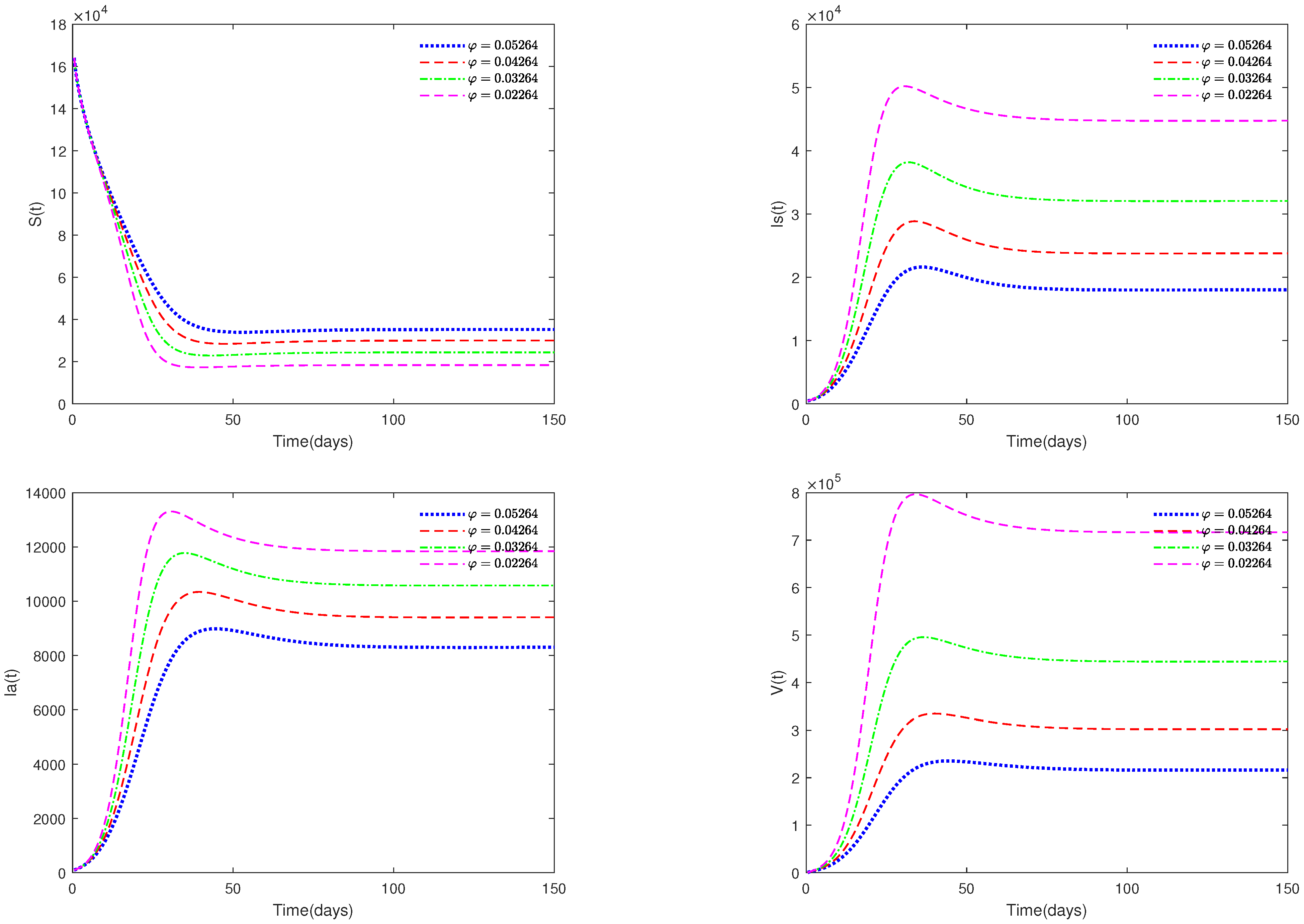
- Figure 5 and Figure 6 show the sensitivity analysis for parameters d and φ. Through observation, it can be observed that the mortality rate d of pigs and the clearance rate φ of viruses have a significant impact on system (1). Therefore, it is reasonable for us to consider specific control measures in system (2) as removing diseased pigs and strengthening the disinfection and sterilization of pig breeding environments.
- (iv)
3.3. Qualitative Analysis Results for System (2)
3.4. Examples and Numerical Simulation Results for System (2)
- (i)
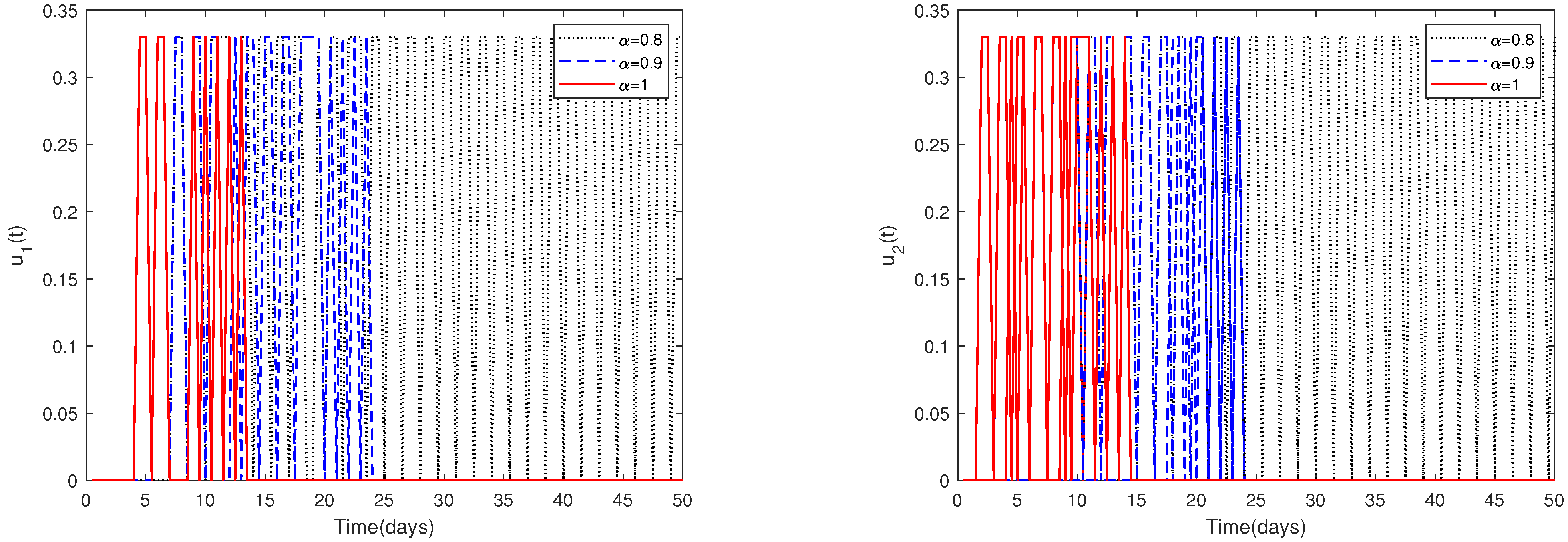
- In Figure 10, the initial value is fixed to = [164,000, 470, 100, 300]. For different values of α, this figure demonstrates the optimal solution of and when the upper limit of , is relatively small (realistically reasonable).
- (ii)
- In Figure 11, the initial value is fixed to = [164,000, 470, 100, 300]. For different values of α, this figure shows the optimal solution of and when the upper limit of , is relatively larger (realistically unreasonable).
- (i)
- When , Figure 8 shows that for all , the corresponding optimal solution tend to be stable with different speeds. This indicates that under the values of Example 3, ASF will outbreak in a certain region, and it will gradually become an endemic.
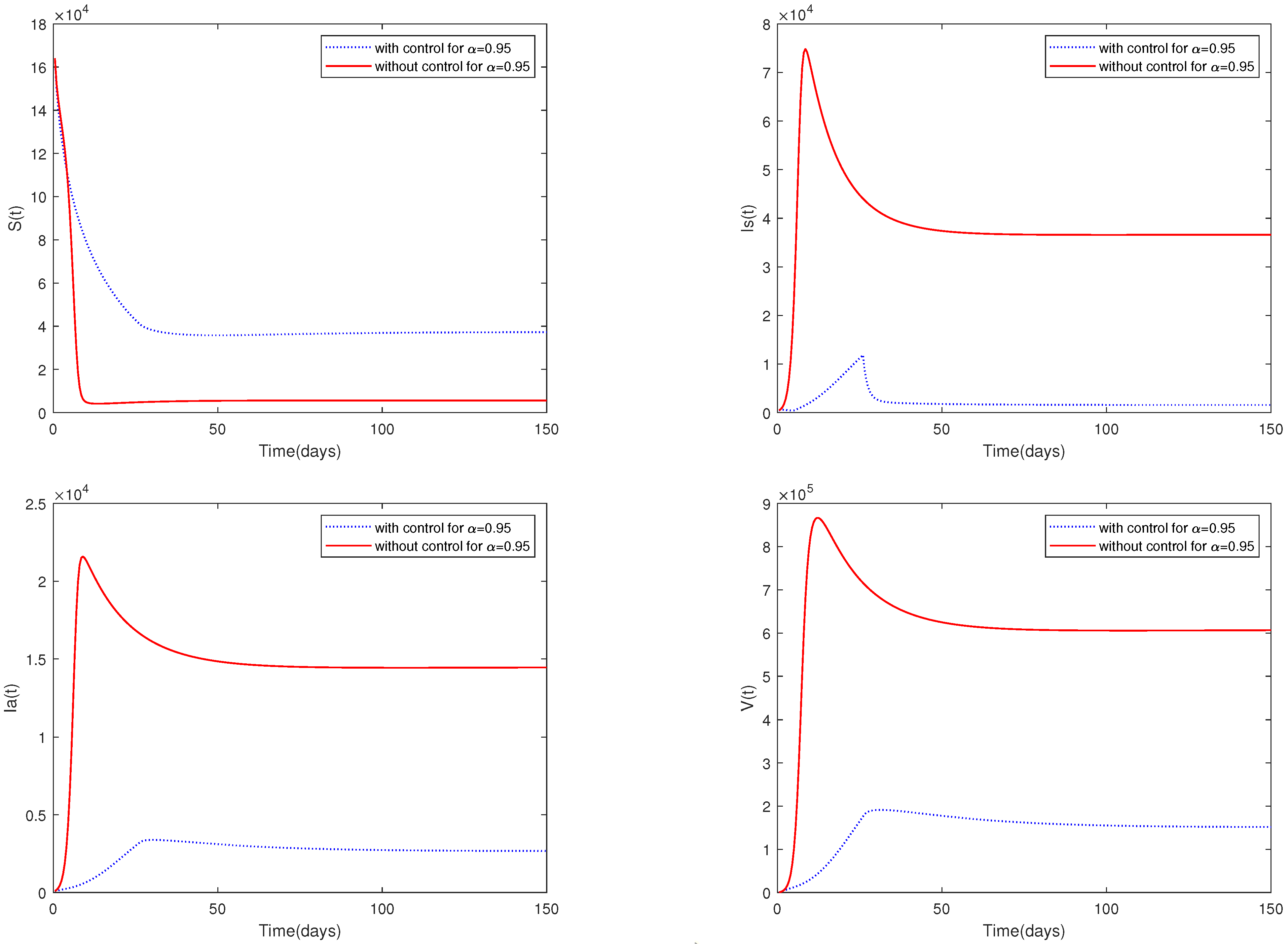
- (ii)
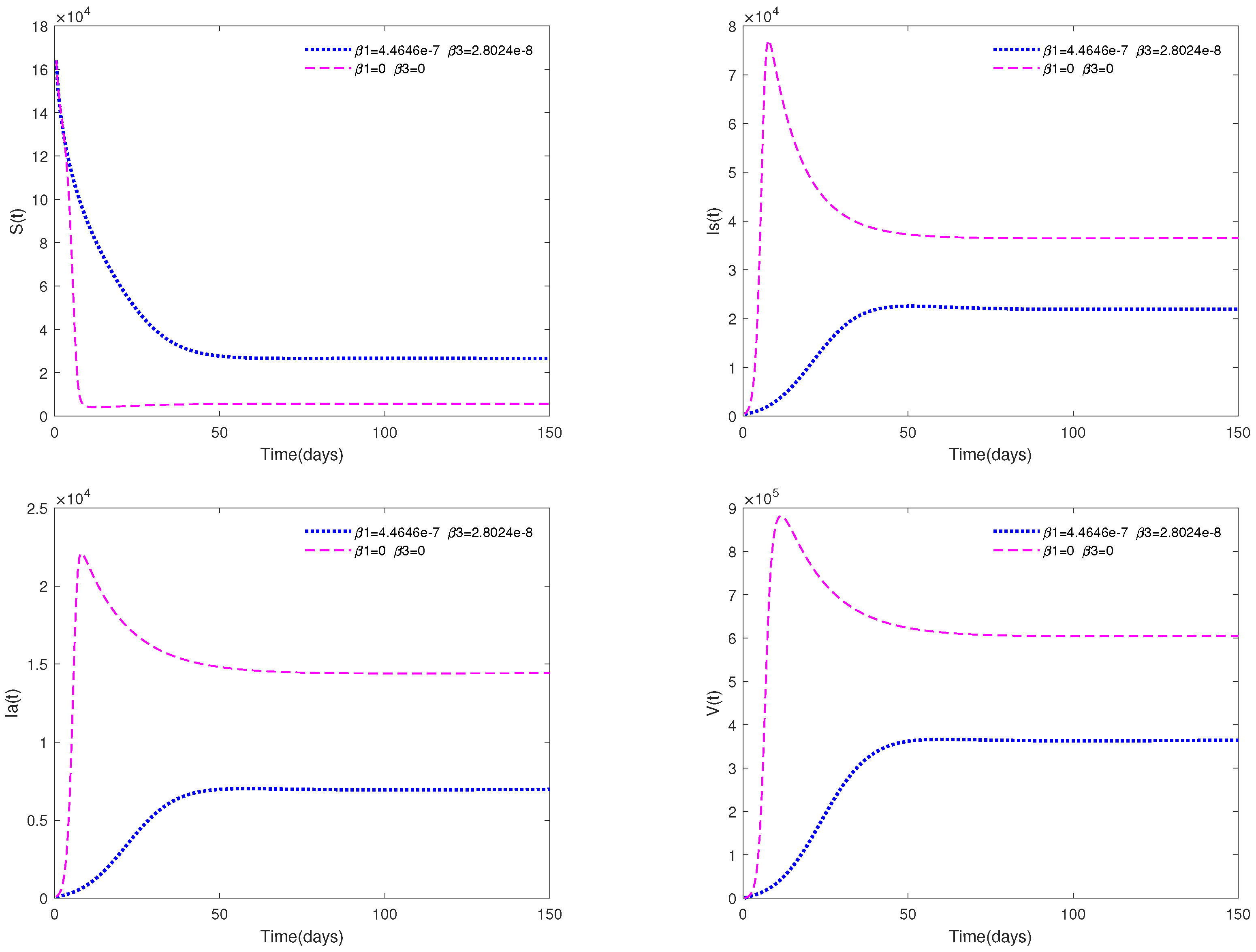
- A comparison between Figure 7 and Figure 9 shows that media coverage combined with control measures can suppress the spread of ASF more effectively. That is to say, if pig farmers take timely measures to eliminate suspected infected pigs and disinfect the environment of pig farms on a large scale after receiving media reports of the outbreak of ASF in the local area, they can greatly reduce the infection and help to prevent the spread of the epidemic.
- (iii)
- Since the magnitudes change dramatically for different parameters, choosing a suitable upper limit of , is important.Figure 10 shows that if the upper limit of , is relatively small, then the optimal control solutions and can be suitably solved. However, if the upper limit of , is relatively big, then the optimal control solutions and cannot be solved suitably, as shown in Figure 11. In fact, obvious and chaotic oscillations occur.
4. Discussion
- There always exists a unique positive solution for any positive initial value, and the set is positively invariant for this system. This conclusion is essential from a biological perspective.
- The basic reproduction number is obtained.
- The sufficient conditions for the existence and stability of the disease-free equilibrium and endemic equilibrium are derived.
- From Figure 1, Figure 2, Figure 3 and Figure 4, it can be observed that the initial value is not crucial and it does not affect the stability. This means that the initial value of susceptible pigs and diseased pigs is not a key factor. However, the value of is important, and it will affect the speed towards a stable state. This result indicates that the fractional order system is different from its corresponding integer order system.
- Figure 5 and Figure 6 show that both parameters d and are sensitive. In fact, d and have a significant effect on the basic reproduction number . In practice, we can reduce the value of by increasing the mortality rate of diseased pigs or increasing disinfection measures in pig houses, thereby achieving the goal of preventing the continued spread of the disease.
- Figure 7 shows that media coverage is a very useful measure to control the disease.
- The formula of the optimal control solution and is obtained by using the Pontryagin’s maximum principle.
- Figure 9 indicates that media coverage combined with control measures (such as disinfection and sterilization) can suppress the spread of the disease more effectively.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Proof of Theorem 1
Appendix B. Proof of Theorem 3
Appendix C. Proof of Theorem 5
Appendix D. Proof of Theorem 6
References
- Galindo, I.; Alonso, C. African swine fever virus: A Review. Viruses 2017, 9, 103. [Google Scholar] [CrossRef]
- Wang, Y.; Kang, W.; Yang, W.; Zhang, J.; Zheng, H. Structure of African swine fever virus and associated molecular mechanisms underlying infection and immunosuppression: A Review. Front. Immunol. 2021, 12, 715582. [Google Scholar] [CrossRef] [PubMed]
- Barongo, M.B.; Karl, S.; Bernard, B.; Richard, P.B.; Eric, M.F.; Tony, A.; Edward, O.; Charles, M.; Darryn, K.; Amos, S.; et al. Estimating the basic reproductive number (R0) for African swine fever virus (ASFV) transmission between pig herds in uganda. PLoS ONE 2015, 10, e0125842. [Google Scholar] [CrossRef]
- Portugal, R.; Coelho, J.; Hoper, D.; Little, N.S.; Smithson, C.; Upton, C.; Martins, C.; Leitao, A.; Keil, G.M. Related strains of African swine fever virus with different virulence: Genome comparison and analysis. J. Gen. Virol. 2015, 96, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Halasa, T.; Btner, A.; Mortensen, S.; Christensen, H.; Boklund, A. Simulating the epidemiological and economic effects of an African swine fever epidemic in industrialized swine populations. Vet. Microbiol. 2016, 193, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Halasa, T.; Btner, A.; Mortensen, S.; Christensen, H.; Toft, N.; Boklund, A. Control of African swine fever epidemics in industrialized swine populations. Vet. Microbiol. 2016, 197, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Dione, M.M.; Dohoo, I.; Ndiwa, N.; Poole, J.; Ouma, E.; Amia, W.C.; Wieland, B. Impact of participatory training of smallholder pig farmers on knowledge, attitudes and practices regarding biosecurity for the control of African swine fever in Uganda. Transbound. Emerg. Dis. 2020, 67, 2269–3076. [Google Scholar] [CrossRef] [PubMed]
- Stoffel, C.; Buholzer, P.; Fanelli, A.; Nardi, M.D. Analysis of the drivers of ASF introduction into the officially approved pig compartments in South Africa and implications for the revision of biosecurity standards. Porc. Health Manag. 2022, 8, 43. [Google Scholar] [CrossRef]
- Boinas, F.S.; Wilson, A.J.; Hutchings, G.H.; Martins, C.; Dixon, L.J. The persistence of African swine fever virus in field-infected ornithodoros erraticus during the ASF endemic period in Portugal. PLoS ONE 2011, 6, 20383. [Google Scholar] [CrossRef]
- Jori, F.; Vial, L.; Penrith, M.; Pérez-Sánchez, R.; Etter, E.; Albina, E.; Michaud, V.; Roger, F. Review of the sylvatic cycle of African swine fever in sub-Saharan Africa and the Indian ocean. Virus Res. 2013, 173, 212–227. [Google Scholar] [CrossRef]
- Frant, M.; Woźniakowski, G.; Pejsak, Z. African swine fever (ASF) and ticks. No risk of tick-mediated ASF spread in Poland and Baltic states. J. Vet. Res. 2017, 61, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Kouidere, A.; Balatif, O.; Rachik, M. Analysis and optimal control of a mathematical modeling of the spread of African swine fever virus with a case study of South Korea and cost-effectiveness. Chaos Soliton Fract. 2021, 146, 110867. [Google Scholar] [CrossRef]
- Barongo, M.B.; Bishop, R.P.; Fèvre, E.M.; Knobel, D.L.; Ssematimba, A. A mathematical model that simulates control options for African swine fever virus (ASFV). PLoS ONE 2016, 11, e0158658. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Rong, X.; Li, J.; Fan, M.; Zhu, H. Modeling the outbreak and control of African swine fever virus in large-scale pig farms. J. Theor. Biol. 2021, 526, 110798. [Google Scholar] [CrossRef]
- Song, H.; Li, J.; Jin, Z. Nonlinear dynamic modelling and analysis of African swine fever with culling in China. Commun. Nonlinear Sci. 2023, 117, 106915. [Google Scholar] [CrossRef]
- Shi, R.; Li, Y.; Wang, C. Stability analysis and optimal control of a fractional-order model for African swine fever. Virus Res. 2020, 288, 198111. [Google Scholar] [CrossRef]
- Buonomo, B.; D’Onofrio, A.; Lacitignola, D. Globally stable endemicity for infectious diseases with information-related changes in contact patterns. Appl. Math. Lett. 2012, 25, 1056–1060. [Google Scholar] [CrossRef]
- D’Onofrio, A.; Manfredi, P. Information-related changes in contactpatterns may trigger oscillations in the endemic prevalence of infectious diseases. J. Theor. Biol. 2009, 256, 473–478. [Google Scholar] [CrossRef]
- Vargas-De-Leon, C.; D’Onofrio, A. Global stability of infectious disease models with contact rate as a function of prevalence index. Math. Biosci. Eng. 2017, 14, 1019–1033. [Google Scholar] [CrossRef]
- Singh, S.; Chandra, P.; Shukla, J.B. Modeling and analysis of the spread of carrier dependent infectious diseases with environmental effects. J. Biol. Syst. 2008, 11, 325–335. [Google Scholar] [CrossRef]
- Gani, S.R.; Halawar, S.V. Optimal control for the spread of infectious disease: The role of awareness programs by media and antiviral treatment. Optim. Control Appl. Methods 2018, 39, 1407–1430. [Google Scholar] [CrossRef]
- Misra, A.K.; Sharma, A.; Shukla, J.B. Modeling and analysis of effects of awareness programs by media on the spread of infectious diseases. Math. Comput. Model. 2011, 53, 1221–1228. [Google Scholar] [CrossRef]
- Zuo, L.; Liu, M.; Wang, J. The impact of awareness programs with recruitment and delay on the spread of an epidemic. Math. Probl. Eng. 2015, 2015, 235935. [Google Scholar] [CrossRef]
- Juher, D.; Kiss, I.Z.; Salda, A.J. Analysis of an epidemic model with awareness decay on regular random networks. J. Theor. Biol. 2015, 365, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Sun, Y.; Zhu, H. The impact of media on the control of infectious diseases. J. Dyn. Differ. Equ. 2008, 20, 31–53. [Google Scholar] [CrossRef]
- Tchuenche, J.M.; Dube, N.; Bhunu, C.P.; Smith, R.J.; Bauch, C.T. The impact of media coverage on the transmission dynamics of human influenza. BMC Public Health 2011, 11, S5. [Google Scholar] [CrossRef]
- Zhao, H.; Zhao, M. Global Hopf bifurcation analysis of an susceptible-infective-removed epidemic model incorporating media coverage with time delay. J. Biol. Dynam. 2017, 11, 8–24. [Google Scholar] [CrossRef]
- Huang, C.; Cai, L.; Cao, J. Linear control for synchronization of a fractional-order time-delayed chaotic financial system. Chaos Soliton Fract. 2018, 113, 326–332. [Google Scholar] [CrossRef]
- Rihan, F.A.; Abdel Rahman, D.H.; Lakshmanan, S.; Alkhajeh, A.S. A time delay model of tumour-immune system interactions: Global dynamics, parameter estimation, sensitivity analysis. Appl. Math. Comput. 2014, 232, 606–623. [Google Scholar] [CrossRef]
- Vvatsala, A.L. Basic theory of fractional differential equations. Nonlinear Anal. Theor. Methods Appl. 2008, 69, 2677–2682. [Google Scholar]
- Rakkiyappan, R.; Velmurugan, G.; Cao, J. Stability analysis of fractional-order complex-valued neural networks with time delays. Chaos Soliton Fract. 2015, 78, 297–316. [Google Scholar] [CrossRef]
- Alidousti, J.; Ghaziani, R.K. Spiking and bursting of a fractional order of the modified FitzHugh-Nagumo neuron model. Math. Models Comput. Simul. 2017, 9, 390–403. [Google Scholar] [CrossRef]
- Djordjevic, V.D.; Jaric, J.; Fabry, B.; Fredberg, J.J. Fractional derivatives embody essential features of cell rheological behavior. Ann. Biomed. Eng. 2003, 31, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture and Rural Affairs of the People’s Republic of China. 2022. Available online: http://www.moa.gov.cn/ztzl/fzzwfk/yqxx/ (accessed on 20 March 2022).
- Gao, D.; Ruan, S. An SIS patch model with variable transmission coefficients. Math. Biosci. 2011, 232, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Yang, W.; Arino, J.; Khan, K. Effect of media-induced social distancing on disease transmission in a two patch setting. Math. Biosci. 2011, 230, 87–95. [Google Scholar] [CrossRef]
- Capasso, V.; Serio, G. A generalization of the Kermack-McKendrick deterministic epidemic model. Math. Biosci. 1978, 42, 43–61. [Google Scholar] [CrossRef]
- Diethelm, K. Monotonicity of functions and sign changes of their Caputo derivatives. Fract. Calc. Appl. Anal. 2016, 19, 561–566. [Google Scholar] [CrossRef]
- Wei, L. Global existence theory and chaos control of fractional differential equations. J. Math. Anal. Appl. 2007, 332, 709–726. [Google Scholar]
- Driessche, P. Reproduction numbers of infectious disease models. Infect. Dis. Model. 2017, 2, 288–303. [Google Scholar] [CrossRef]
- Akman Yıldız, T.; Arshad, S.; Baleanu, D. New observations on optimal cancer treatments for a fractional tumor growth model with and without singular kernel. Chaos Soliton Fract. 2018, 117, 226–239. [Google Scholar] [CrossRef]
- Ahmed, E.; Ama, E.S.; Haa, E.S. On some Routh-Hurwitz conditions for fractional order differential equations and their applications in Lorenz, Rssler, Chua and Chen systems. Phys. Lett. A 2006, 358, 1–4. [Google Scholar] [CrossRef]
- Pinto, C.; Carvalho, A. A latency fractional order model for HIV dynamics. J. Comput. Appl. Math. 2017, 312, 240–256. [Google Scholar] [CrossRef]
- Göllmann, L.; Kern, D.; Maurer, H. Optimal control problems with delays in state and control variables subject to mixed control–Cstate constraints. Optim. Control Appl. Methods 2010, 30, 341–365. [Google Scholar] [CrossRef]
- Catalano, S.; La Morgia, V.; Molinar Min, A.R.; Fanelli, A.; Meneguz, P.G.; Tizzani, P. Gastrointestinal Parasite Community and Phenotypic Plasticity in Native and Introduced Alien Lagomorpha. Animals 2022, 12, 1287. [Google Scholar] [CrossRef] [PubMed]
- Viani, A.; Orusa, T.; Borgogno-Mondino, E.; Orusa, R. Snow Metrics as Proxy to Assess Sarcoptic Mange in Wild Boar: Preliminary Results in Aosta Valley (Italy). Life 2023, 13, 987. [Google Scholar] [CrossRef] [PubMed]

| Density of the susceptible population | ||||
| Density of the symptomatic infectious population | ||||
| Density of the asymptomatic infectious population | ||||
| Density of ASFV in the environment | ||||
| The recruitment rate of population | 670 | [12] | ||
| ASFV transmission rate with direct contact of infectious population | [15] | |||
| Virus transmission rate of contaminated pig products and materials | [15] | |||
| m | The half-saturation constant | 30 | [27] | |
| Reduced rate by asymptomatic population | 0.7001 | [15] | ||
| p | The proportion of symptomatic infectious population | 0.7899 | [15] | |
| Natural and disease related death rate of population | 0.006040 | |||
| d | Natural death rate of population | 0.004060 | ||
| h | The release rate of virus from symptomatic infectious population | 10.0575 | [15] | |
| k | The release rate of virus from asymptomatic infectious population | 299.6462 | [15] | |
| Virus clearance rate | [15] | |||
| Measures to eliminate suspected disease population | [0, 1] | |||
| Measures for disinfection and sterilization with disinfectant | [0, 1] |
| Number and Sign of Roots | ||||
|---|---|---|---|---|
| 3 negative | + | + | + | + |
| 3 positive | + | − | + | − |
| 1 positive | + | − | − | − |
| 1 positive | + | + | − | − |
| 1 positive | + | + | + | − |
| 1 positive | − | + | + | + |
| 2 positive | + | − | + | + |
| 2 positive | + | − | − | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, R.; Zhang, Y.; Wang, C. Dynamic Analysis and Optimal Control of Fractional Order African Swine Fever Models with Media Coverage. Animals 2023, 13, 2252. https://doi.org/10.3390/ani13142252
Shi R, Zhang Y, Wang C. Dynamic Analysis and Optimal Control of Fractional Order African Swine Fever Models with Media Coverage. Animals. 2023; 13(14):2252. https://doi.org/10.3390/ani13142252
Chicago/Turabian StyleShi, Ruiqing, Yihong Zhang, and Cuihong Wang. 2023. "Dynamic Analysis and Optimal Control of Fractional Order African Swine Fever Models with Media Coverage" Animals 13, no. 14: 2252. https://doi.org/10.3390/ani13142252
APA StyleShi, R., Zhang, Y., & Wang, C. (2023). Dynamic Analysis and Optimal Control of Fractional Order African Swine Fever Models with Media Coverage. Animals, 13(14), 2252. https://doi.org/10.3390/ani13142252






