An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Adipocytes—The Main Compound of AT
3. Stromal Vascular Fraction—Interacting Cells Hosted by AT
3.1. Mechanism of Action
3.2. SVF Clinical Application for Various Conditions
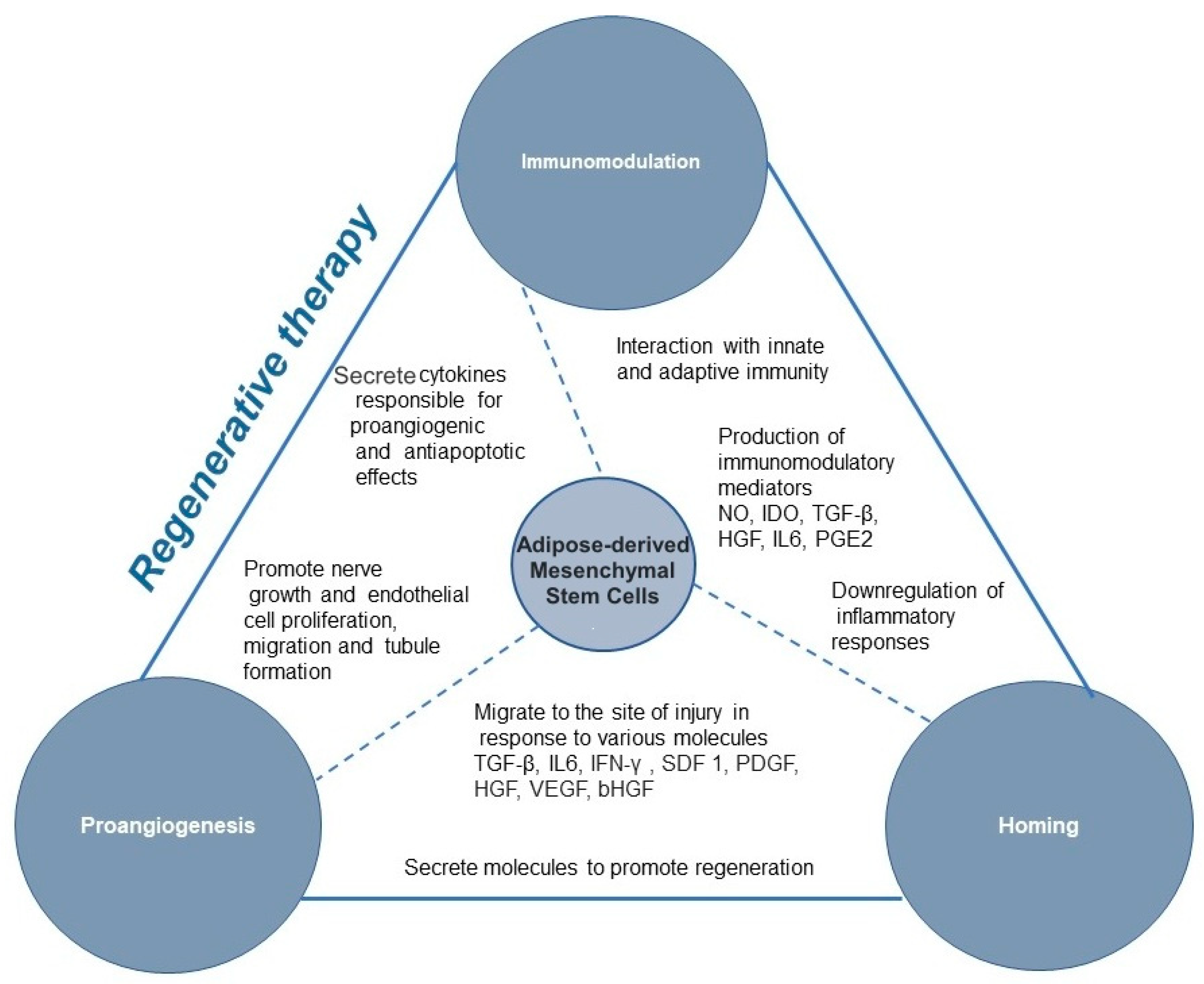
4. Adipose-Derived Mesenchymal Stem Cells—An Outstanding Component of the SVF
4.1. Mechanism of Action
4.1.1. Immunomodulation
4.1.2. Homing
4.1.3. Pro-Angiogenic and Anti-Apoptotic Mechanism of Action
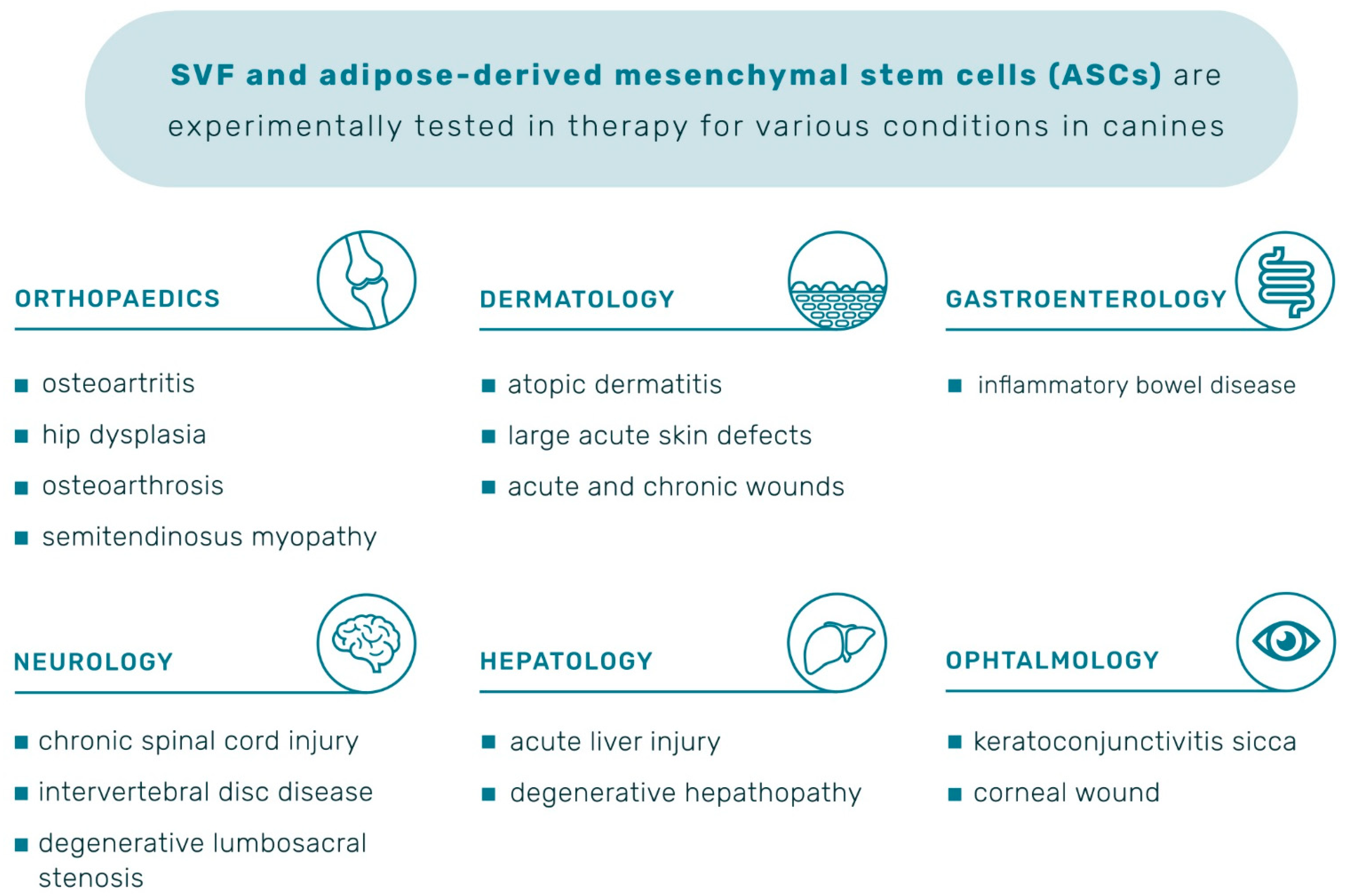
4.2. ASCs Clinical Application for Various Conditions
4.2.1. Orthopaedics
4.2.2. Neurology
4.2.3. Dermatology
4.2.4. Ophthalmology
4.2.5. Gastroenterology
4.2.6. Hepatology
5. The Importance of the Regulatory Considerations and Safety Aspects Using of Animal Cell-Based Products in Regenerative Therapy
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef] [PubMed]
- Esteve Ràfols, M. Adipose tissue: Cell heterogeneity and functional diversity. Endocrinol. y Nutr. (English Ed.) 2014, 61, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, V.M.; Boyd, N.L. The Adipose Stromal Vascular Fraction as a Complex Cellular Source for Tissue Engineering Applications. Tissue Eng.—Part B Rev. 2018, 24, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Billon, N.; Iannarelli, P.; Monteiro, M.C.; Glaviuex-Pardanaud, C.; Richardson, W.D.; Kessaris, N.; Dani, C.; Dupin, E. The generation of adipocytes by the neural crest. Development 2007, 134, 2283–2292. [Google Scholar] [CrossRef] [Green Version]
- Trevor, L.V.; Riches-Suman, K.; Mahajan, A.L.; Thornton, M.J. Adipose tissue: A source of stem cells with potential for regenerative therapies for wound healing. J. Clin. Med. 2020, 9, 2161. [Google Scholar] [CrossRef]
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front. Cardiovasc. Med. 2020, 7, 22. [Google Scholar] [CrossRef] [Green Version]
- Simpson, E.R. Sources of estrogen and their importance. J. Steroid Biochem. Mol. Biol. 2003, 86, 225–230. [Google Scholar] [CrossRef]
- Obradovic, M.; Sudar-Milovanovic, E.; Soskic, S.; Essack, M.; Arya, S.; Stewart, A.J.; Gojobori, T.; Isenovic, E.R. Leptin and Obesity: Role and Clinical Implication. Front. Endocrinol. (Lausanne) 2021, 12, 585887. [Google Scholar] [CrossRef]
- Trayhurn, P.; Beattie, J.H. Physiological role of adipose tissue: White adipose tissue as an endocrine and secretory organ. Proc. Nutr. Soc. 2001, 60, 329–339. [Google Scholar] [CrossRef] [Green Version]
- Bahamondes, F.; Flores, E.; Cattaneo, G.; Bruna, F.; Conget, P. Omental adipose tissue is a more suitable source of canine Mesenchymal stem cells. BMC Vet. Res. 2017, 13, 166. [Google Scholar] [CrossRef] [Green Version]
- Eurell, J.A.; Van Sickle, D.C. Connective and Supportive Tissues. In Dellmann’s Textbook of Veterinary Histology; Eurell, J.A., Freappier, B.L., Eds.; Blackwell Publishing: Ames, IA, USA, 2006; pp. 1–405. ISBN 9780781741484. [Google Scholar]
- Otto, T.C.; Lane, M.D. Adipose development: From stem cell to adipocyte. Crit. Rev. Biochem. Mol. Biol. 2005, 40, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.; Spiegelman, B.M. Cell biology of fat storage. Mol. Biol. Cell 2016, 27, 2523–2527. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.H.; Kokkotou, E.; Schulz, T.J.; Huang, T.L.; Winnay, J.N.; Taniguchi, C.M.; Tran, T.T.; Suzuki, R.; Espinoza, D.O.; Yamamoto, Y.; et al. New role of bone morphogenetic protein 7 in brown adipogenesis and energy expenditure. Nature 2008, 454, 1000–1004. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, A.; Oh, K.J.; Lee, S.C.; Kim, W.K.; Bae, K.H. The role of adipose tissue mitochondria: Regulation of mitochondrial function for the treatment of metabolic diseases. Int. J. Mol. Sci. 2019, 20, 4924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [Green Version]
- Bargut, T.C.L.; Souza-Mello, V.; Aguila, M.B.; Mandarim-De-Lacerda, C.A. Browning of white adipose tissue: Lessons from experimental models. Horm. Mol. Biol. Clin. Investig. 2017, 31, 1–13. [Google Scholar] [CrossRef]
- Kaisanlahti, A.; Glumoff, T. Browning of white fat: Agents and implications for beige adipose tissue to type 2 diabetes. J. Physiol. Biochem. 2019, 75, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.; Nguyen, A.; Banyard, D.A.; Fadavi, D.; Toranto, J.D.; Wirth, G.A.; Paydar, K.Z.; Evans, G.R.D.; Widgerow, A.D. Stromal vascular fraction: A regenerative reality? Part 2: Mechanisms of regenerative action. J. Plast. Reconstr. Aesthetic Surg. 2016, 69, 180–188. [Google Scholar] [CrossRef]
- Bora, P.; Majumdar, A.S. Adipose tissue-derived stromal vascular fraction in regenerative medicine: A brief review on biology and translation. Stem Cell Res. Ther. 2017, 8, 145. [Google Scholar] [CrossRef]
- Prunet-Marcassus, B.; Cousin, B.; Caton, D.; André, M.; Pénicaud, L.; Casteilla, L. From heterogeneity to plasticity in adipose tissues: Site-specific differences. Exp. Cell Res. 2006, 312, 727–736. [Google Scholar] [CrossRef]
- Hendawy, H.; Uemura, A.; Ma, D.; Namiki, R.; Samir, H.; Ahmed, M.F.; Elfadadny, A.; El-husseiny, H.M.; Chieh-jen, C.; Tanaka, R. Tissue harvesting site effect on the canine adipose stromal vascular fraction quantity and quality. Animals 2021, 11, 460. [Google Scholar] [CrossRef] [PubMed]
- Krešić, N.; Prišlin, M.; Vlahović, D.; Kostešić, P.; Ljolje, I.; Brnić, D.; Turk, N.; Musulin, A.; Habrun, B. The expression pattern of surface markers in canine adipose-derived mesenchymal stem cells. Int. J. Mol. Sci. 2021, 22, 7476. [Google Scholar] [CrossRef] [PubMed]
- Andia, I.; Maffulli, N.; Burgos-Alonso, N. Stromal vascular fraction technologies and clinical applications. Expert Opin. Biol. Ther. 2019, 19, 1289–1305. [Google Scholar] [CrossRef] [PubMed]
- van Dongen, J.A.; Stevens, H.P.; Parvizi, M.; van der Lei, B.; Harmsen, M.C. The fractionation of adipose tissue procedure to obtain stromal vascular fractions for regenerative purposes. Wound Repair Regen. 2016, 24, 994–1003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senesi, L.; De Francesco, F.; Farinelli, L.; Manzotti, S.; Gagliardi, G.; Papalia, G.F.; Riccio, M.; Gigante, A. Mechanical and enzymatic procedures to isolate the stromal vascular fraction from adipose tissue: Preliminary results. Front. Cell Dev. Biol. 2019, 7, 88. [Google Scholar] [CrossRef]
- Astor, D.E.; Hoelzler, M.G.; Harman, R.; Bastian, R.P. Patient factors influencing the concentration of stromal vascular fraction (SVF) for adipose-derived stromal cell (ASC) therapy in dogs. Can. J. Vet. Res. 2013, 77, 177–182. [Google Scholar]
- Lee, J.; Lee, K.S.; Kim, C.L.; Byeon, J.S.; Gu, N.Y.; Cho, I.S.; Cha, S.H. Effect of donor age on the proliferation and multipotency of canine adipose-derived mesenchymal stem cells. J. Vet. Sci. 2017, 18, 141–148. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Gahring, D.; Adams, C.; Aron, D.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of adipose-derived mesenchymal stem and regenerative cells on lameness in dogs with chronic osteoarthritis of the coxofemoral joints: A randomized, double-blinded, multicenter, controlled trial. Vet. Ther. 2007, 8, 272–284. [Google Scholar]
- Upchurch, D.A.; Renberg, W.C.; Roush, J.K.; Milliken, G.A.; Weiss, M.L. Effects of administration of adipose-derived stromal vascular fraction and platelet-rich plasma to dogs with osteoarthritis of the hip joints. Am. J. Vet. Res. 2016, 77, 940–951. [Google Scholar] [CrossRef] [Green Version]
- Black, L.L.; Gaynor, J.; Adams, C.; Dhupa, S.; Sams, A.E.; Taylor, R.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of intraarticular injection of autologous adipose-derived mesenchymal stem and regenerative cells on clinical signs of chronic osteoarthritis of the elbow joint in dogs. Vet. Ther. 2008, 9, 192–200. [Google Scholar]
- Marx, C.; Silveira, M.D.; Selbach, I.; Da Silva, A.S.; Braga, L.M.G.D.M.; Camassola, M.; Nardi, N.B. Acupoint injection of autologous stromal vascular fraction and allogeneic adipose-derived stem cells to treat hip dysplasia in dogs. Stem Cells Int. 2014, 2014, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemilew, J.; Sobczyńska-Rak, A.; Zylińska, B.; Szponder, T.; Nowicka, B.; Urban, B. The use of allogenic stromal vascular fraction (SVF) cells in degenerative joint disease of the spine in dogs. In Vivo (Brooklyn) 2019, 33, 1109–1117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caplan, A.I.; Dennis, J.E. Mesenchymal stem cells as trophic mediators. J. Cell. Biochem. 2006, 98, 1076–1084. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuk, P.A.; Min, Z.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human Adipose Tissue Is a Source of Multipotent Stem Cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Neupane, M.; Chang, C.-C.; Kiupel, M.; Yuzbasiyan-Gurkan, V. Isolation and Characterization of Canine Adipose–Derived Mesenchymal Stem Cells. Tissue Eng. Part A 2008, 14, 1007–1015. [Google Scholar] [CrossRef]
- Marcoccia, R.; Nesci, S.; Merlo, B.; Ballotta, G.; Algieri, C.; Pagliarani, A.; Iacono, E. Biological characteristics and metabolic profile of canine mesenchymal stem cells isolated from adipose tissue and umbilical cord matrix. PLoS ONE 2021, 16, e0247567. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Kulus, M.; Kulus, J.; Jankowski, M.; Borowiec, B.; Jeseta, M.; Bukowska, D.; Brüssow, K.P.; Kempisty, B.; Antosik, P. The use of mesenchymal stem cells in veterinary medicine. Med. J. Cell Biol. 2018, 6, 101–107. [Google Scholar] [CrossRef] [Green Version]
- Gugjoo, M.B.; Amarpal, A.; Sharma, G.T. Mesenchymal stem cell basic research and applications in dog medicine. J. Cell. Physiol. 2019, 234, 16779–16811. [Google Scholar] [CrossRef]
- Voga, M.; Adamic, N.; Vengust, M.; Majdic, G. Stem Cells in Veterinary Medicine—Current State and Treatment Options. Front. Vet. Sci. 2020, 7, 278. [Google Scholar] [CrossRef] [PubMed]
- Dias, I.E.; Pinto, P.O.; Barros, L.C.; Viegas, C.A.; Dias, I.R.; Carvalho, P.P. Mesenchymal stem cells therapy in companion animals: Useful for immune-mediated diseases? BMC Vet. Res. 2019, 15, 358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, F.; Chiu, S.M.; Motan, D.A.L.; Zhang, Z.; Chen, L.; Ji, H.L.; Tse, H.F.; Fu, Q.L.; Lian, Q. Mesenchymal stem cells and immunomodulation: Current status and future prospects. Cell Death Dis. 2016, 7, e2062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayala-Cuellar, A.P.; Kang, J.H.; Jeung, E.B.; Choi, K.C. Roles of mesenchymal stem cells in tissue regeneration and immunomodulation. Biomol. Ther. 2019, 27, 25–33. [Google Scholar] [CrossRef]
- Chow, L.; Johnson, V.; Coy, J.; Regan, D.; Dow, S. Mechanisms of Immune Suppression Utilized by Canine Adipose and Bone Marrow-Derived Mesenchymal Stem Cells. Stem Cells Dev. 2017, 26, 374–389. [Google Scholar] [CrossRef] [Green Version]
- Wheat, W.H.; Chow, L.; Kurihara, J.N.; Regan, D.P.; Coy, J.W.; Webb, T.L.; Dow, S.W. Suppression of Canine Dendritic Cell Activation/Maturation and Inflammatory Cytokine Release by Mesenchymal Stem Cells Occurs Through Multiple Distinct Biochemical Pathways. Stem Cells Dev. 2017, 26, 249–262. [Google Scholar] [CrossRef]
- Herrero, C.; Pérez-Simón, J.A. Immunomodulatory effect of mesenchymal stem cells. Brazilian J. Med. Biol. Res. 2010, 43, 425–430. [Google Scholar] [CrossRef]
- Marx, C.; Silveira, M.D.; Nardi, N.B. Adipose-Derived Stem Cells in Veterinary Medicine: Characterization and Therapeutic Applications. Stem Cells Dev. 2015, 24, 803–813. [Google Scholar] [CrossRef]
- Krešić, N.; Šimić, I.; Lojkić, I.; Bedeković, T. Mezenhimske matične stanice u veterinarskoj medicini. Vet. Stanica 2019, 50, 149–154. [Google Scholar]
- Ponte, A.L.; Marais, E.; Gallay, N.; Langonné, A.; Delorme, B.; Hérault, O.; Charbord, P.; Domenech, J. The In Vitro Migration Capacity of Human Bone Marrow Mesenchymal Stem Cells: Comparison of Chemokine and Growth Factor Chemotactic Activities. Stem Cells 2007, 25, 1737–1745. [Google Scholar] [CrossRef]
- Ullah, M.; Liu, D.D.; Thakor, A.S. Mesenchymal Stromal Cell Homing: Mechanisms and Strategies for Improvement. iScience 2019, 15, 421–438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sackstein, R. The lymphocyte homing receptors: Gatekeepers of the multistep paradigm. Curr. Opin. Hematol. 2005, 12, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.W.; Heo, S.C.; Jeong, G.O.; Yoon, J.W.; Mo, W.M.; Lee, M.J.; Jang, I.H.; Kwon, S.M.; Lee, J.S.; Kim, J.H. Tumor necrosis factor-α-activated mesenchymal stem cells promote endothelial progenitor cell homing and angiogenesis. Biochim. Biophys. Acta—Mol. Basis Dis. 2013, 1832, 2136–2144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al Delfi, I.R.; Sheard, J.J.; Wood, C.R.; Vernallis, A.; Innes, J.F.; Myint, P.; Johnson, W.E.B. Canine mesenchymal stem cells are neurotrophic and angiogenic: An in vitro assessment of their paracrine activity. Vet. J. 2016, 217, 10–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, C.; Zhou, Y.; Wang, H.; Deng, F.; Chen, Y.; Zhu, X.; Kong, Y.; Pan, L.; Xue, L.; Zhou, X.; et al. Adipose-derived stem cells alleviate radiation-induced dermatitis by suppressing apoptosis and downregulating cathepsin F expression. Stem Cell Res. Ther. 2021, 12, 447. [Google Scholar] [CrossRef]
- Brondeel, C.; Pauwelyn, G.; de Bakker, E.; Saunders, J.; Samoy, Y.; Spaas, J.H. Review: Mesenchymal Stem Cell Therapy in Canine Osteoarthritis Research: “Experientia Docet” (Experience Will Teach Us). Front. Vet. Sci. 2021, 8, 668881. [Google Scholar] [CrossRef]
- Yoon, H.-Y.; Lee, J.; Jeong, S. Long-term follow-up after implantation of autologous adipose tissue derived mesenchymal stem cells to treat a dog with stifle joint osteoarthrosis. J. Vet. Clin. 2012, 29, 82–86. [Google Scholar]
- Guercio, A.; Di Marco, P.; Casella, S.; Cannella, V.; Russotto, L.; Purpari, G.; Di Bella, S.; Piccione, G. Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol. Int. 2012, 36, 189–194. [Google Scholar] [CrossRef]
- Vilar, J.M.; Morales, M.; Santana, A.; Spinella, G.; Rubio, M.; Cuervo, B.; Cugat, R.; Carrillo, J.M. Controlled, blinded force platform analysis of the effect of intraarticular injection of autologous adipose-derived mesenchymal stem cells associated to PRGF-Endoret in osteoarthritic dogs. BMC Vet. Res. 2013, 9, 131. [Google Scholar] [CrossRef] [Green Version]
- Cuervo, B.; Rubio, M.; Sopena, J.; Dominguez, J.M.; Vilar, J.; Morales, M.; Cugat, R.; Carrillo, J.M. Hip osteoarthritis in dogs: A randomized study using mesenchymal stem cells from adipose tissue and plasma rich in growth factors. Int. J. Mol. Sci. 2014, 15, 13437–13460. [Google Scholar] [CrossRef]
- Vilar, J.M.; Batista, M.; Morales, M.; Santana, A.; Cuervo, B.; Rubio, M.; Cugat, R.; Sopena, J.; Carrillo, J.M. Assessment of the effect of intraarticular injection of autologous adipose-derived mesenchymal stem cells in osteoarthritic dogs using a double blinded force platform analysis. BMC Vet. Res. 2014, 10, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harman, R.; Carlson, K.; Gaynor, J.; Gustafson, S.; Dhupa, S.; Clement, K.; Hoelzler, M.; McCarthy, T.; Schwartz, P.; Adams, C. A prospective, randomized, masked, and placebo-controlled efficacy study of intraarticular allogeneic adipose stem cells for the treatment of osteoarthritis in dogs. Front. Vet. Sci. 2016, 3, 81. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Ku, S.K.; Kwon, Y.S. Adipose-derived mesenchymal stem cells and platelet-rich plasma synergistically ameliorate the surgical-induced osteoarthritis in Beagle dogs. J. Orthop. Surg. Res. 2016, 11, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilar, J.M.; Cuervo, B.; Rubio, M.; Sopena, J.; Domínguez, J.M.; Santana, A.; Carrillo, J.M. Effect of intraarticular inoculation of mesenchymal stem cells in dogs with hip osteoarthritis by means of objective force platform gait analysis: Concordance with numeric subjective scoring scales. BMC Vet. Res. 2016, 12, 223. [Google Scholar] [CrossRef] [Green Version]
- Kriston-Pál, É.; Czibula, Á.; Gyuris, Z.; Balka, G.; Seregi, A.; Sükösd, F.; Süth, M.; Kiss-Tóth, E.; Haracska, L.; Uher, F.; et al. Characterization and therapeutic application of canine adipose. Can. J. Vet. Res. 2017, 81, 73–78. [Google Scholar]
- Shah, K.; Drury, T.; Roic, I.; Hansen, P.; Malin, M.; Boyd, R.; Sumer, H.; Ferguson, R. Outcome of allogeneic adult stem cell therapy in dogs suffering from osteoarthritis and other joint defects. Stem Cells Int. 2018, 2018, 7. [Google Scholar] [CrossRef] [Green Version]
- Srzentić Dražilov, S.; Mrkovački, J.; Spasovski, V.; Fazlagić, A.; Pavlović, S.; Nikčević, G. The use of canine mesenchymal stem cells for the autologous treatment of osteoarthritis. Acta Vet. Hung. 2018, 66, 376–389. [Google Scholar] [CrossRef] [Green Version]
- Olsen, A.; Johnson, V.; Webb, T.; Santangelo, K.S.; Dow, S.; Duerr, F.M. Evaluation of Intravenously Delivered Allogeneic Mesenchymal Stem Cells for Treatment of Elbow Osteoarthritis in Dogs: A Pilot Study. Vet. Comp. Orthop. Traumatol. 2019, 32, 173–181. [Google Scholar] [CrossRef]
- Wits, M.I.; Tobin, G.C.; Silveira, M.D.; Baja, K.G.; Braga, L.M.M.; Sesterheim, P.; Camassola, M.; Nardi, N.B. Combining canine mesenchymal stromal cells and hyaluronic acid for cartilage repair. Genet. Mol. Biol. 2020, 43, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Brown, G.S.; Harman, J.R.; Black, L.L. Adipose-derived stem cell therapy for severe muscle tears in working German shepherds: Two case reports. Stem Cell Discov. 2012, 02, 41–44. [Google Scholar] [CrossRef] [Green Version]
- Gibson, M.A.; Brown, S.G.; Brown, N.O. Semitendinosus myopathy and treatment with adipose-derived stem cells in working German shepherd police dogs. Can. Vet. J. 2017, 58, 241–246. [Google Scholar] [PubMed]
- Escalhão, C.C.M.I.; Ramos, I.P.; Hochman-Mendez, C.; Brunswick, T.H.K.; Souza, S.A.L.; Gutfilen, B.; Dos Santos Goldenberg, R.C.; Coelho-Sampaio, T. Safety of allogeneic canine adipose tissue-derived mesenchymal stem cell intraspinal transplantation in dogs with chronic spinal cord injury. Stem Cells Int. 2017, 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Bach, F.S.; Rebelatto, C.L.K.; Fracaro, L.; Senegaglia, A.C.; Fragoso, F.Y.I.; Daga, D.R.; Brofman, P.R.S.; Pimpão, C.T.; Engracia Filho, J.R.; Montiani-Ferreira, F.; et al. Comparison of the Efficacy of Surgical Decompression Alone and Combined With Canine Adipose Tissue-Derived Stem Cell Transplantation in Dogs With Acute Thoracolumbar Disk Disease and Spinal Cord Injury. Front. Vet. Sci. 2019, 6, 383. [Google Scholar] [CrossRef] [PubMed]
- Mrkovački, J.; Srzentić Dražilov, S.; Spasovski, V.; Fazlagić, A.; Pavlović, S.; Nikčević, G. Case Report: Successful Therapy of Spontaneously Occurring Canine Degenerative Lumbosacral Stenosis Using Autologous Adipose Tissue-Derived Mesenchymal Stem Cells. Front. Vet. Sci. 2021, 8, 1055. [Google Scholar] [CrossRef]
- Chen, C.C.; Yang, S.F.; Wang, I.K.; Hsieh, S.Y.; Yu, J.X.; Wu, T.L.; Huang, W.J.; Su, M.H.; Yang, H.L.; Chang, P.C.; et al. The Long-Term Efficacy Study of Multiple Allogeneic Canine Adipose Tissue-Derived Mesenchymal Stem Cells Transplantations Combined With Surgery in Four Dogs With Lumbosacral Spinal Cord Injury. Cell Transplant. 2022, 31, 09636897221081487. [Google Scholar] [CrossRef]
- Zubin, E.; Conti, V.; Leonardi, F.; Zanichelli, S.; Ramoni, R.; Grolli, S. Regenerative therapy for the management of a large skin wound in a dog. Clin. Case Reports 2015, 3, 598–603. [Google Scholar] [CrossRef]
- Villatoro, A.J.; Hermida-Prieto, M.; Fernández, V.; Fariñas, F.; Alcoholado, C.; Isabel Rodríguez-García, M.; Mariñas-Pardo, L.; Becerra, J. Allogeneic adipose-derived mesenchymal stem cell therapy in dogs with refractory atopic dermatitis: Clinical efficacy and safety. Vet. Rec. 2018, 183, 654. [Google Scholar] [CrossRef]
- Enciso, N.; Avedillo, L.; Fermín, M.L.; Fragío, C.; Tejero, C. Cutaneous wound healing: Canine allogeneic ASC therapy. Stem Cell Res. Ther. 2020, 11, 261. [Google Scholar] [CrossRef]
- Kaur, G.; Ramirez, A.; Xie, C.; Clark, D.; Dong, C.; Maki, C.; Ramos, T.; Izadyar, F.; Najera, S.O.L.; Harb, J.; et al. A double-blinded placebo-controlled evaluation of adipose-derived mesenchymal stem cells in treatment of canine atopic dermatitis. Vet. Res. Commun. 2022, 46, 251–260, 10. [Google Scholar] [CrossRef]
- Villatoro, A.J.; Fernández, V.; Claros, S.; Rico-Llanos, G.A.; Becerra, J.; Andrades, J.A. Use of adipose-derived mesenchymal stem cells in keratoconjunctivitis sicca in a canine model. Biomed Res. Int. 2015, 2015, 527926. [Google Scholar] [CrossRef]
- Bittencourt, M.K.W.; Barros, M.A.; Martins, J.F.P.; Vasconcellos, J.P.C.; Morais, B.P.; Pompeia, C.; Bittencourt, M.D.; Evangelho, K.D.S.; Kerkis, I.; Wenceslau, C.V. Allogeneic Mesenchymal Stem Cell Transplantation in Dogs with Keratoconjunctivitis Sicca. Cell Med. 2016, 8, 63–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sgrignoli, M.R.; Silva, D.A.; Nascimento, F.F.; Sgrignoli, D.A.M.; Nai, G.A.; da Silva, M.G.; de Barros, M.A.; Bittencourt, M.K.W.; de Morais, B.P.; Dinallo, H.R.; et al. Reduction in the inflammatory markers CD4, IL-1, IL-6 and TNFα in dogs with keratoconjunctivitis sicca treated topically with mesenchymal stem cells. Stem Cell Res. 2019, 39, 101525. [Google Scholar] [CrossRef] [PubMed]
- Falcão, M.S.A.; Brunel, H.D.S.S.; Peixer, M.A.S.; Dallago, B.S.L.; Costa, F.F.; Queiroz, L.M.; Campbell, P.; Malard, P.F. Effect of allogeneic mesenchymal stem cells (MSCs) on corneal wound healing in dogs. J. Tradit. Complement. Med. 2020, 10, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Merino, E.M.; Usón-Casaús, J.M.; Zaragoza-Bayle, C.; Duque-Carrasco, J.; Mariñas-Pardo, L.; Hermida-Prieto, M.; Barrera-Chacón, R.; Gualtieri, M. Safety and efficacy of allogeneic adipose tissue-derived mesenchymal stem cells for treatment of dogs with inflammatory bowel disease: Clinical and laboratory outcomes. Vet. J. 2015, 206, 385–390. [Google Scholar] [CrossRef]
- Teshima, T.; Matsumoto, H.; Michishita, M.; Matsuoka, A.; Shiba, M.; Nagashima, T.; Koyama, H. Allogenic adipose tissue-derived mesenchymal stem cells ameliorate acute hepatic injury in dogs. Stem Cells Int. 2017, 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Gardin, C.; Ferroni, L.; Bellin, G.; Rubini, G.; Barosio, S.; Zavan, B. Therapeutic potential of autologous adipose-derived stem cells for the treatment of liver disease. Int. J. Mol. Sci. 2018, 19, 4064. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Fang, J.; Wen, X.; Teng, X.; Li, B.; Zhou, Z.; Peng, S.; Arisha, A.H.; Liu, W.; Hua, J. Therapeutic applications of adipose-derived mesenchymal stem cells on acute liver injury in canines. Res. Vet. Sci. 2019, 126, 233–239. [Google Scholar] [CrossRef]
- Anonymus. European Medicines Agency: Questions and Answers on Allogeneic Stem Cell-Based Products for Veterinary Use. 2019. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/questions-answers-allogeneic-stem-cell-based-products-veterinary-use-specific-questions-sterility_en.pdf (accessed on 31 January 2022).
- Anonymous. European Medicines Agency: Concept Paper on the Development and Data Requirements of Potency Tests for Cell-Based Therapy Products and the Relation to Clinical Efficacy. 2022. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/concept-paper-development-data-requirements-potency-tests-cell-based-therapy-products-relation_en.pdf (accessed on 3 February 2022).
- Lynne Boxer. Food and Drug Administration: Guidance for Industry Cell-Based Products for Animal Use. 2015. Available online: https://www.fda.gov/media/88925/download?source=govdelivery&utm_medium=email&utm_source=govdelivery (accessed on 21 December 2021).
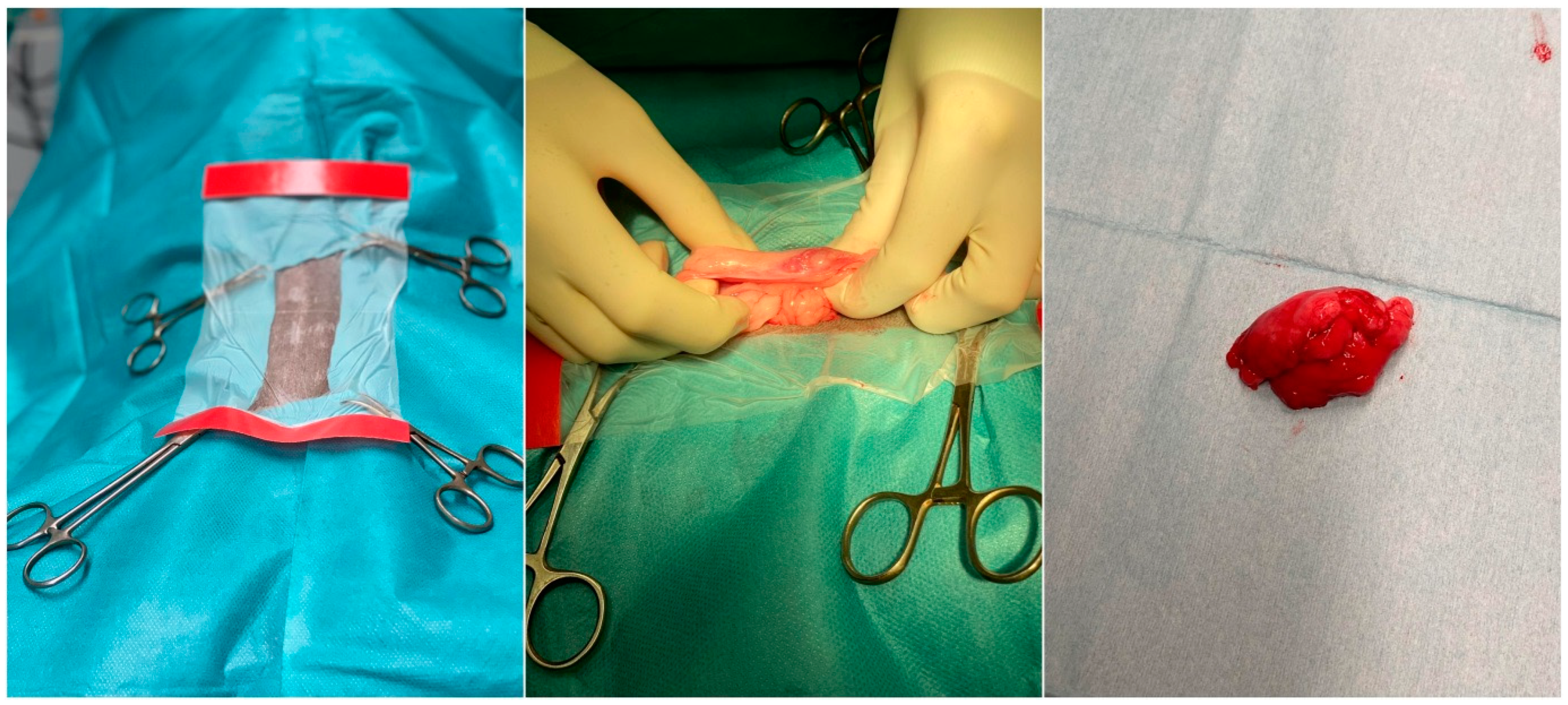
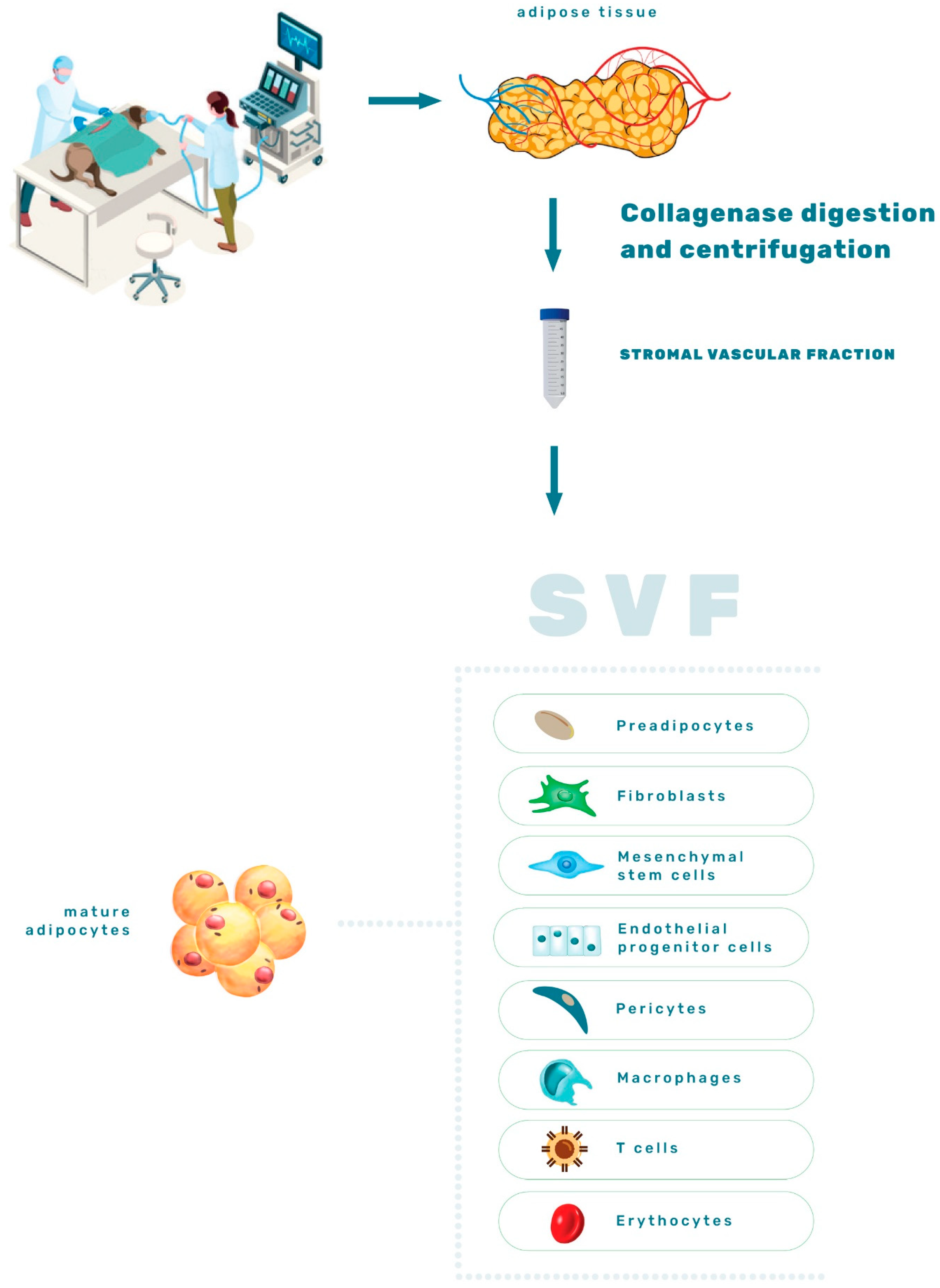
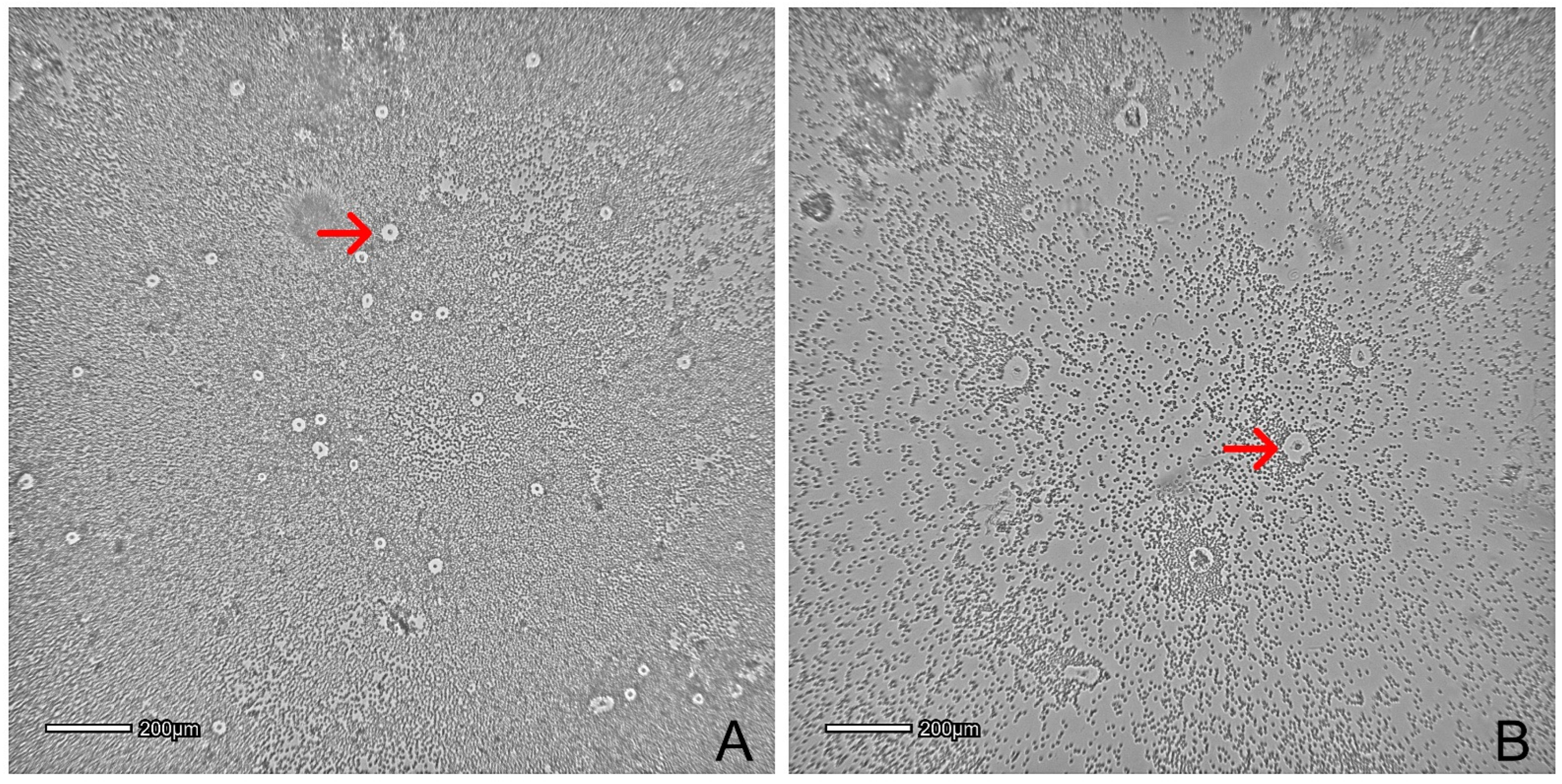
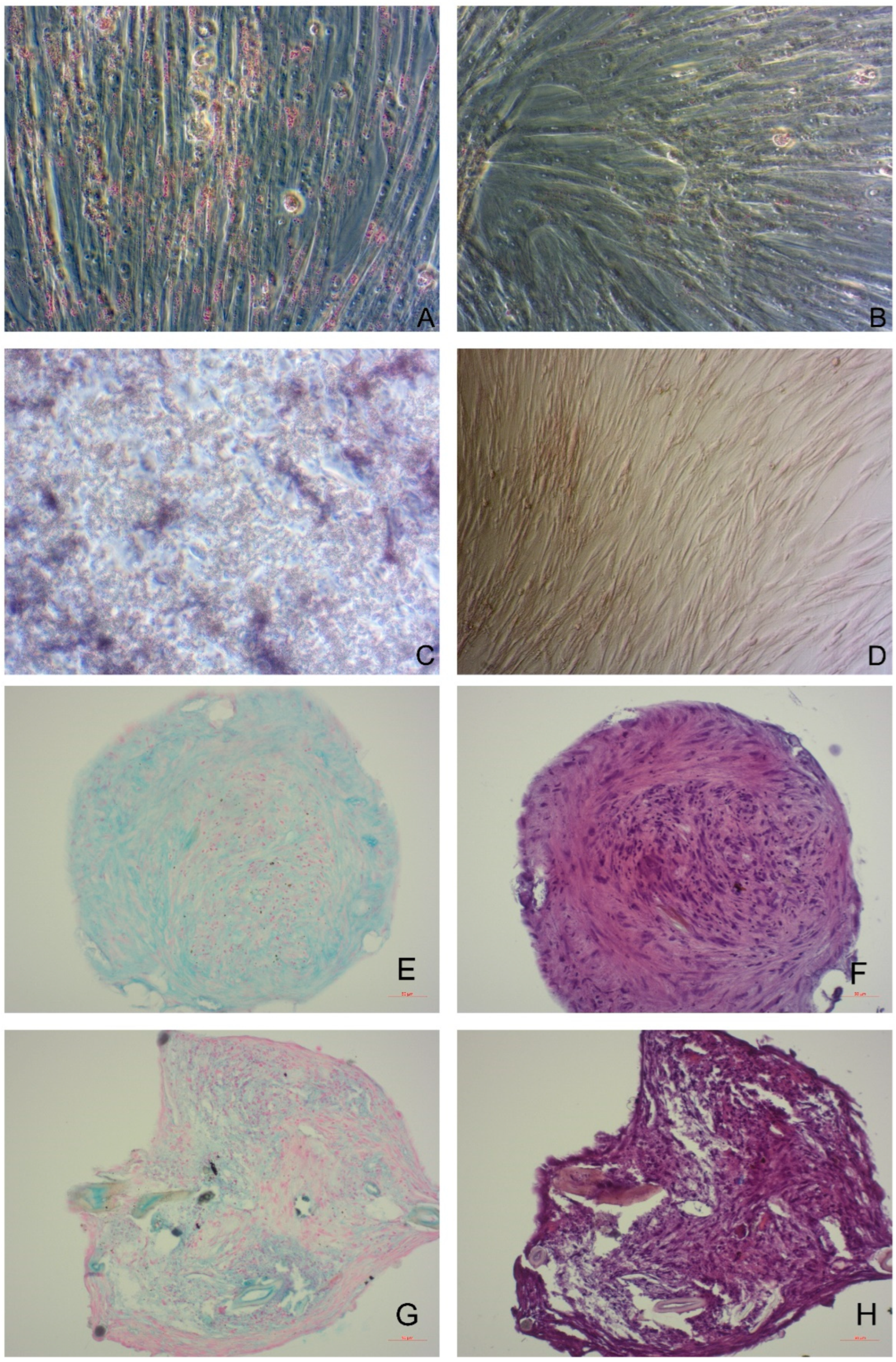
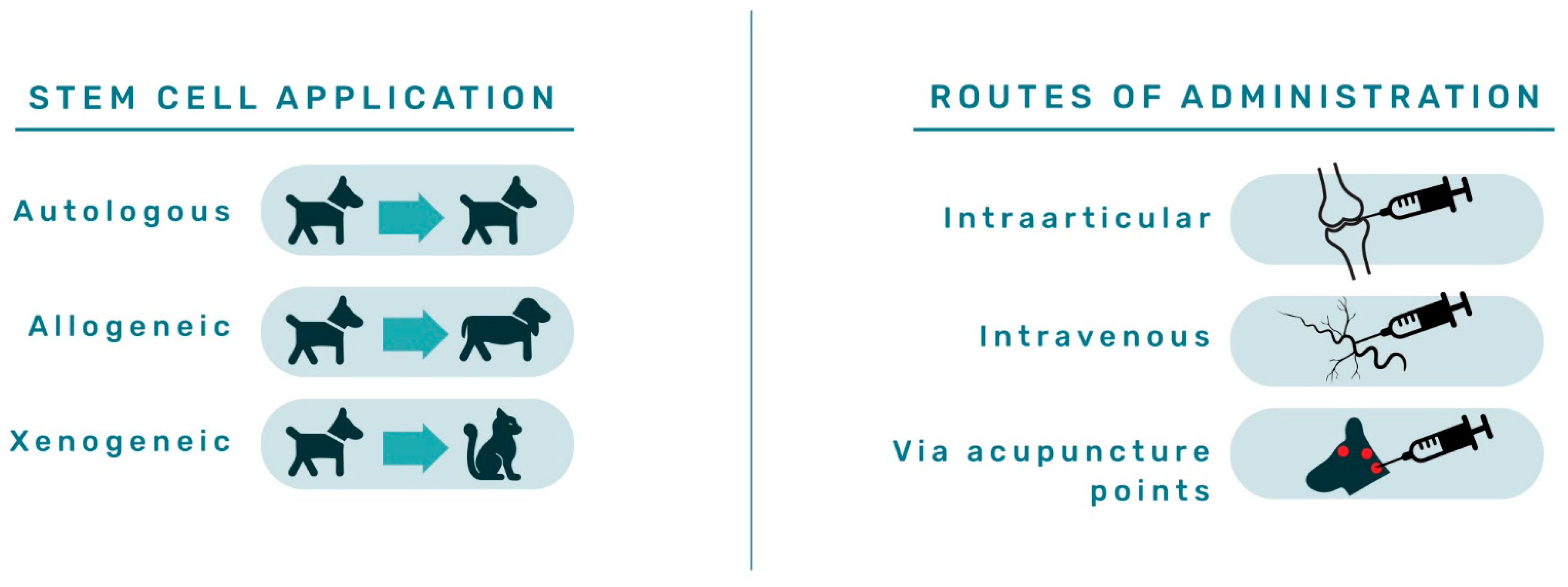
| Clinical Condition | Number of Canines Included | Type of Application | Route of Administration | Number of Cells (×106) | Reference | |
|---|---|---|---|---|---|---|
| Orthopaedics | Osteoarthritis of hip joints | 21 | Autologous SVF | Intraarticular | 4.2–5 | Black et al. (2007) [29] |
| Osteoarthritis of the elbow joint | 14 | Autologous SVF | Intraarticular | 3–5 | Black et al. (2008) [31] | |
| Stifle joint osteoarthrosis | 1 | Autologous ASCs + hyaluronic acid | intraarticular | 1 | Yoon et al. (2012) [58] | |
| Chronic osteoarthritis of the elbow joints | 4 | Autologous ASCs + hyaluronic acid/PRP | Intraarticular | 3–5 | Guercio et al. (2012) [59] | |
| Osteoarthritis of hip joints | 13 | Autologous ASCs + PRP | Intraarticular | 15 | Vilar et al. (2013) [60] | |
| Osteoarthritis of hip joints | 18 | Autologous ASCs | Intraarticular | 30 | Cuervo et al. (2014) [61] | |
| Hip dysplasia | SVF = 4 ASCs = 5 | Autologous SVF or allogeneic ASCs | Acupoint injection | SVF = 2–5 ASCs = 0.2–0.8 | Marx et al. (2014) [49] | |
| Osteoarthritis of hip joints | 15 | Autologous ASCs | Intraarticular | 15 | Vilar et al. (2014) [62] | |
| Osteoarthritis of hip joints | 22 | Autologous SVF + PRP | Intraarticular and intravenous | N/A | Upchurch et al. (2016) [30] | |
| Osteoarthritis of different joints | 74 | Allogeneic ASCs | Intraarticular | 12 | Harman et al. (2016) [63] | |
| Surgical-induced osteoarthritis in Beagle dogs | 24 | ASCs and/or PRP | Intraarticular | 10 | Yun et al. (2016) [64] | |
| Osteoarthritis of hip joints | 15 | Autologous ASCs | Intraarticular | 15 | Vilar et al. (2016) [65] | |
| Osteoarthritis of the elbow joint | 30 (39 elbows) | Allogeneic ASCs + hyaluronic acid | Intraarticular | 12 ± 3.2 | Kriston-Pal et al. (2017) [66] | |
| Osteoarthritis and other joint defects | 203 | Allogeneic ASCs | Intraarticular and/or intravenous | N/A | Shah et al. (2018) [67] | |
| Osteoarthritis of different joints | 10 | Autologous ASCs | Intraarticular | 15–30 | Srzentić Dražilov et al. (2018) [68] | |
| Osteoarthritis of the elbow joint | 13 | Allogeneic ASCs | Intravenous | 1–2/kg body weight | Olsen et al. (2019) [69] | |
| Osteoarthritis of hip joints | 12 (24 hips) | Allogeneic ASCs | Intraarticular | 5 | Wits et al. (2020) [70] | |
| Acute semitendinosus muscle injury | 2 | Autologous SVF | Intramuscular and intravenous | 4.7 | Brown et al. (2012) [71] | |
| Semitendinosus myopathy | 11 | Autologous ASCs | Intramuscular and intravenous | N/A | Gibson et al. (2017) [72] | |
| Neurology | Chronic spinal cord injury | 6 | Allogeneic ASCs | Intraspinal | N/A | Escalhao et al. (2017) [73] |
| Acute thoracolumbar disc disease and spinal cord injury | 22 | Allogeneic ASCs | Epidural | 10 | Bach et al. (2019) [74] | |
| Degenerative lumbosacral stenosis | 1 | Autologous ASCs | Paravertebral and intraarticular | Paravertebral = 30.6 Intraarticular = 15.3 | Mrkovački et al. (2021) [75] | |
| Lumbosacral spinal cord injury | 4 | Allogeneic ASCs + surgery | Nerve roots next to injury, intravenous and epidural | Nerve roots next to injury = 5 Intravenous = 4 Epidural = N/A | Chen et al. (2022) [76] | |
| Dermatology | Large skin wound | 1 | Autologous ASCs + PRP | Local dripping or spraying | N/A | Zubin et al. (2015) [77] |
| Atopic dermatitis | 26 | Allogeneic ASCs | Intravenous | 1.5 | Villatoro et al. (2018) [78] | |
| Acute and chronic skin wound | 24 | Allogeneic ASCs | Intradermal | 30 | Enciso et al. (2020) [79] | |
| Atopic dermatitis | 15 | Allogeneic ASCs | Subcutaneous | Low dose = 0.5/kg body weight High dose = 5/kg body weight | Kaur et al. (2022) [80] | |
| Ophthalmology | Keratoconjunctivitis sicca | 12 | Allogeneic ASCs | Around the lacrimal glands | 5 | Villatoro et al. (2015) [81] |
| Keratoconjunctivitis sicca | 15 (24 eyes) | Allogeneic ASCs | Intralacrimal | 1 | Bittencourt et al. (2016) [82] | |
| Keratoconjunctivitis sicca | 22 | Allogeneic ASCs | Topic in the conjunctival sac | 1 | Sgrignoli et al. (2019) [83] | |
| Corneal wound | 26 | Allogeneic ASCs | Sub-conjunctival | 3 | Falcao et al. (2020) [84] | |
| Gastroenterology | Inflammatory bowel disease | 11 | Allogeneic ASCs | Intravenous | 2/kg body weight | Perez-Merino et al. (2015) [85] |
| Hepatology | Acute liver injury | 9 | Allogeneic ASCs | Peripheral vein/splenic vein | 2 | Teshima et al. (2017) [86] |
| Degenerative hepatopathy | 10 | Autologous ASCs | Portal vein | 0.5/kg body weight | Gardin et al. (2018) [87] | |
| Acute liver injury | 6 | Allogeneic ASCs | Intravenous | 10 | Yan et al. (2019) [88] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prišlin, M.; Vlahović, D.; Kostešić, P.; Ljolje, I.; Brnić, D.; Turk, N.; Lojkić, I.; Kunić, V.; Karadjole, T.; Krešić, N. An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy. Animals 2022, 12, 1088. https://doi.org/10.3390/ani12091088
Prišlin M, Vlahović D, Kostešić P, Ljolje I, Brnić D, Turk N, Lojkić I, Kunić V, Karadjole T, Krešić N. An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy. Animals. 2022; 12(9):1088. https://doi.org/10.3390/ani12091088
Chicago/Turabian StylePrišlin, Marina, Dunja Vlahović, Petar Kostešić, Ivana Ljolje, Dragan Brnić, Nenad Turk, Ivana Lojkić, Valentina Kunić, Tugomir Karadjole, and Nina Krešić. 2022. "An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy" Animals 12, no. 9: 1088. https://doi.org/10.3390/ani12091088
APA StylePrišlin, M., Vlahović, D., Kostešić, P., Ljolje, I., Brnić, D., Turk, N., Lojkić, I., Kunić, V., Karadjole, T., & Krešić, N. (2022). An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy. Animals, 12(9), 1088. https://doi.org/10.3390/ani12091088








