Cheese and Butter as a Source of Health-Promoting Fatty Acids in the Human Diet
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Methods
2.2.1. Lipid Extraction
2.2.2. Preparation of Fatty Acid Methyl Esters
2.2.3. Analysis of Fatty Acid Composition by GC Method
2.2.4. The Lipid Quality Indices
2.2.5. Statistical Analysis
3. Results and Discussion
3.1. Fatty Acid Composition and Lipid Quality Indices in the Fat from the Analyzed Products
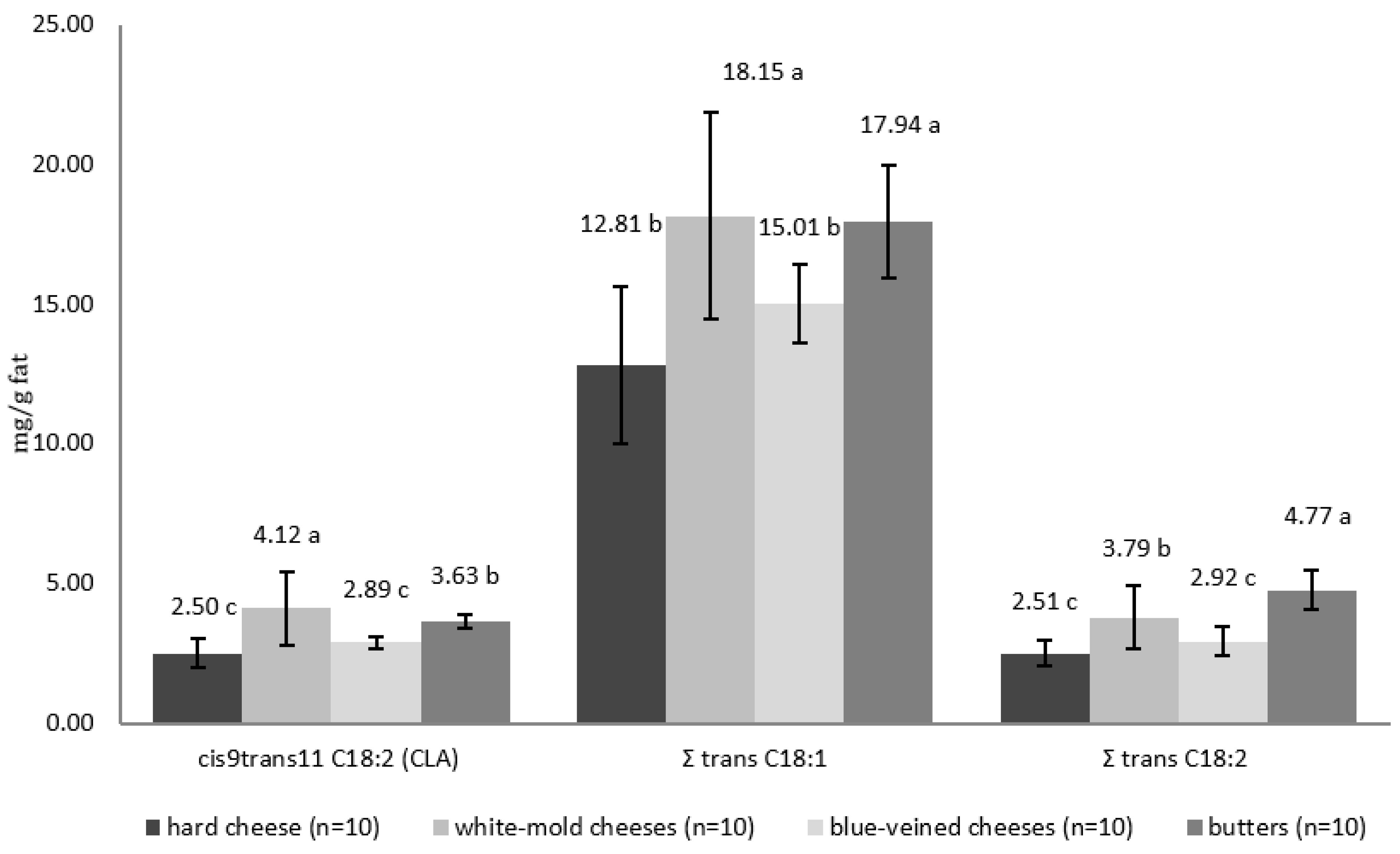
3.2. The Contents of cis9trans11 C18:2 (CLA) and trans C18:1 and C18:2 Fatty Acids in the Fat from the Analyzed Products
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pfeuffer, M.; Watzl, B. Nutrition and health aspects of milk and dairy products and their ingredients. Ernahr. Umschau Sci. Res. 2017, 65, 22–33. [Google Scholar]
- Górska-Warsewicz, H.; Rejman, K.; Laskowski, W.; Czeczotko, M. Milk and Dairy Products and Their Nutritional Contribution to the Average Polish Diet. Nutrients 2019, 11, 1771. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.G. The Composition of Bovine Milk Lipids: January 1995 to December 2000. J. Dairy Sci. 2002, 85, 295–350. [Google Scholar] [CrossRef] [PubMed]
- Månsson, H.L. Fatty acids in bovine milk fat. Food Nutr. Res. 2008, 52, 1821. [Google Scholar] [CrossRef] [PubMed]
- Hanuš, O.; Krížová, L.; Samková, E.; Špička, J.; Kučera, J.; Klimešová, M.; Roubal, P.; Jedelská, R. The effect of cattle bread, season and type of diet on the fatty acid profile of raw milk. Arch. Anim. Breed. 2016, 59, 373–380. [Google Scholar] [CrossRef]
- Sanjayaranj, I.; Lopez-Villalobos, N.; Blair, H.T.; Janssen, P.W.M.; Holroyd, S.E.; MacGibbon, A.K.H. Effect of Breed on the Fatty Acid Composition of Milk from Dairy Cows Milked Once and Twice a Day in Different Stages of Lactation. Dairy 2022, 3, 608–621. [Google Scholar] [CrossRef]
- Clarke, H.J.; Griffin, C.; Rai, D.K.; O’Callaghan, T.F.; O’Sullivan, M.G.; Kerry, J.P.; Kilcawley, K.N. Dietary compounds influencing the sensorial, volatile and phytochemical properties of bovine milk. Molecules 2019, 25, 26. [Google Scholar] [CrossRef]
- Capuano, E.; Gravink, R.; Boerrigter-Eenling, R.; van Ruth, S.M. Fatty acid and triglycerides profiling of retail organic, conventional and pasture milk: Implications for health and authenticity. Int. Dairy J. 2015, 42, 58–63. [Google Scholar] [CrossRef]
- Bär, C.; Sutter, M.; Kopp, C.; Neuhaus, P.; Portmann, R.; Egger, L.; Reidy, B.; Bisig, W. Impact of herbage proportion, animal breed, lactation stage and season on the fatty acid and protein composition of milk. Int. Dairy J. 2020, 109, 104785. [Google Scholar] [CrossRef]
- Magan, J.B.; O’Callaghan, T.F.; Kelly, A.L.; McCarthy, N.A. Compositional and functional properties of milk and dairy products derived from cows fed pasture or concentrate-based diets. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2769–2800. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, R.; Kang, R.; Meng, J.; Ao, C. Milk fatty acids profiles and milk production from dairy cows fed different forage quality diets. Anim. Nutr. 2016, 2, 329–333. [Google Scholar] [CrossRef]
- Frelich, J.; Šlachta, M.; Hanuš, O.; Špička, J.; Samková, E.; Węglarz, A.; Zapletal, P. Seasonal variation in fatty acid composition of cow milk in relation to the feeding system. Anim. Sci. Pap. Rep. 2012, 30, 219–229. [Google Scholar]
- Prado, L.A.; Schmidely, P.; Nozière, P.; Ferlay, A. Milk saturated fatty acids, odd- and branched-chain fatty acids and isomers of C18:1, C18:2, and C18:3 n-3 according to their duodenal flows in dairy cows: A meta-analysis approach. J. Dairy Sci. 2019, 102, 3053–3070. [Google Scholar] [CrossRef]
- Pipoyan, D.; Stepanyan, S.; Stepanyan, S.; Beglaryan, M.; Costantini, L.; Molinari, R.; Merendino, N. The Effect of Trans Fatty Acids on Human Health: Regulation and Consumption Patterns. Foods 2021, 10, 2452. [Google Scholar] [CrossRef]
- Oteng, A.B.; Kersten, S. Mechanisms of Action of trans Fatty Acids. Adv. Nutr. 2020, 11, 697–708. [Google Scholar] [CrossRef]
- Shingfield, K.J.; Chilliard, Y.; Toivonen, P.; Kairenius, P.; Givens, D.I. Trans fatty acids and bioactive lipids in ruminant milk. Adv. Exp. Med. Biol. 2008, 606, 3–65. [Google Scholar] [CrossRef]
- Piekut, M. Production and consumption of cheese in Poland. Dairy Rev. 2012, 7, 36–38. [Google Scholar]
- Walther, B.; Schmid, A.; Sieber, R.; Wehrmüller, K. Cheese in nutrition and health. Dairy Sci. Technol. 2008, 88, 389–405. [Google Scholar] [CrossRef]
- Zheng, X.; Shi, X.; Wang, B. A Review on the General Cheese Processing Technology, Flavor Biochemical Pathways and the Influence of Yeasts in Cheese. Front. Microbiol. 2021, 12, 703284. [Google Scholar] [CrossRef]
- Czechowska-Liszka, M. Jakość serów podpuszczkowych dojrzewających dostępnych w handlu detalicznym na terenie Krakowa. Zesz. Nauk. UEK 2007, 743, 135–147. [Google Scholar]
- Calzada, J.; del Olmo, A.; Picon, A.; Nuῆez, M. Effect of high-pressure-processing on lipolysis and volatile compounds of Brie cheese during ripening and refrigerated storage. Int. Dairy J. 2014, 39, 232–239. [Google Scholar] [CrossRef]
- Galli, B.D.; José Guilherme Prado Martin, J.G.; Moreira da Silva, P.P.; Porto, E.; Fillet Spoto, M.H. Sensory quality of Camembert-type cheese: Relationship between starter cultures and ripening molds. Int. J. Food Microbiol. 2016, 234, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Diezhandino, I.; Fernández, D.; Sacristán, N.; Combarros-Fuertes, P.; Prieto, B.; Fresno, J.M. Rheological, textural, colour and sensory characteristics of a Spanish blue cheese. LWT Food Sci. Technol. 2016, 65, 1118–1125. [Google Scholar] [CrossRef]
- McSweeney, P.L.H.; Sousa, M.J. Biochemical pathways for the production of flavour compounds in cheeses during ripening: A review. Lait 2000, 80, 293–324. [Google Scholar] [CrossRef]
- Bajek, E. Biała, szlachetna pleśń z charakterem. Forum Mlecz. Handel 2011, 2, 54–57. [Google Scholar]
- Dolganova, S.G.; Budaeva, A.B.; Khundanova, T.L.; Gomboeva, O.A.; Tsybikzhapov, A.D. Safety and quality assessment of cheeses with mold. IOP Conf. Ser. Earth Environ. Sci. 2020, 548, 082025. [Google Scholar] [CrossRef]
- PN-A-86155:1995; Mleko i Przetwory Mleczne. Masło. Polish Committee for Standardization: Warsaw, Poland, 1995.
- Chwastowska-Siwiecka, I.; Baryczka, M.J.; Mroziński, Ł. Analiza preferencji konsumenckich przy wyborze i zakupie masła. Przegl. Hod. 2019, 3, 26–31. [Google Scholar]
- Aguilar, C.; Toro-Mujica, P.; Vargas-Bello-Perez, E.; Vera, R.; Ugalde, C.; Rodriguez, S.; Briones, I. A comparative study of the fatty acid profiles in commercial sheep cheeses. Grasas Aceites 2014, 65, e048. [Google Scholar] [CrossRef][Green Version]
- Milewski, S.; Ząbek, K.; Antoszkiewicz, Z.; Tański, Z.; Błażejek, J. Walory prozdrowotne serów z mleka owczego i koziego wytwarzanych w gospodarstwach Warmii i Mazur. Przegl. Hodow. 2016, 2, 20–22. [Google Scholar]
- Kawęcka, A.; Radkowska, I.; Sikora, J. Concentrations of selected bioactive components in traditional cheeses made from goat’s, cow’s and sheep’s milk. J. Elem. 2020, 25, 431–442. [Google Scholar] [CrossRef]
- Talpur, F.N.; Bhanger, M.I.; Memon, N.N. Fatty acid composition with emphasis on conjugated linoleic acid (CLA) and cholesterol content of Pakistani dairy products. Pol. J. Food Nutr. Sci. 2008, 58, 313–320. [Google Scholar]
- Donmez, M.; Seckin, A.K.; Sagdic, O.; Simsek, B. Chemical characteristics, fatty acid compositions, conjugated linoleic acid contents and cholesterol levels of same traditional Turkish cheeses. Int. J. Food Sci. Nutr. 2005, 56, 157–163. [Google Scholar] [CrossRef]
- Rutkowska, J.; Sadowska, A.; Tabaszewska, M.; Stołyhwo, A. Fatty acid composition of hard cheeses from north, eastern and central region of Poland. Bromat. Chem. Toksykol. 2009, 42, 263–269. [Google Scholar]
- Prandini, A.; SigoLo, S.; Piva, G. A comparative study of fatty acid composition and CLA concentration in commercial cheeses. J. Food Compos. Anal. 2011, 24, 55–61. [Google Scholar] [CrossRef]
- Paszczyk, B.; Łuczyńska, J. Fatty acids profile, conjugated linoleic acid contents and fat quality in selected dairy products available on the Polish market. Czech J. Food Sci. 2020, 38, 109–114. [Google Scholar] [CrossRef]
- Paszczyk, B.; Łuczyńska, J. The Comparison of Fatty Acid Composition and Lipid Quality Indices in Hard Cow, Sheep, and Goat Cheeses. Foods 2020, 9, 1667. [Google Scholar] [CrossRef]
- Gürsoy, O.; Seckin, A.K.; Kinik, O.; Metin, M. Conjugated linoleic acid (CLA) content of most popular Turkish hard and soft cheeses. Milchwissenschaft 2003, 58, 622–623. [Google Scholar]
- Adamska, A.; Rasińska, E.; Rutkowska, J.; Antoniewska, A. Fatty acid profile of commercial Camembert- and Brie-type cheeses availableon the Polish market. CyTA J. Food 2017, 15, 639–645. [Google Scholar] [CrossRef]
- Paszczyk, B.; Borejszo, Z.; Łuczyńska, J. Conjugated linoleic acid (CLA) and trans isomers of C18:1 and C18:2 acids in mould cheeses. Pol. J. Nutr. Sci. 2012, 27, 93–101. [Google Scholar]
- Rutkowska, J.; Adamska, A. Fatty acid composition of butter originated from north-eastern region of Poland. Pol. J. Food Nutr. Sci. 2011, 61, 187–193. [Google Scholar] [CrossRef]
- Păduret, S. The Effect of Fat Content and Fatty Acids Composition on Color and Textural Properties of Butter. Molecules 2021, 26, 4565. [Google Scholar] [CrossRef] [PubMed]
- Beitz, D.C. Texture of butter from cows with different milk fatty acid compositions. J. Dairy Sci. 2003, 86, 3122–3127. [Google Scholar]
- Staniewski, B.; Ogrodowska, D.; Staniewska, K.; Kowalik, J. The effect of triacylglycerol and fatty acid composition on the rheological properties of butter. Int. Dairy J. 2021, 114, 104913. [Google Scholar] [CrossRef]
- Christie, W.W. (Ed.) The isolation of lipids from tissues. Recommended Procedures. Chloroform-methanol(2:1,v/v) extraction and “Folch” wash. In Lipid Analysis. Isolation, Separation, Identification and Structural Analysis of Lipids; Pergamon Press: Oxford, UK; New York, NY, USA; Toronto, ON, Canada; Tokyo, Japan; Sydney, Australia; Braunschweig, Germany, 1973; pp. 39–40. [Google Scholar]
- ISO 15884:2002 (IDF 182:2002); Milkfat: Preparation of Fatty Acid Methyl Esters. ISO: Geneva, Switzerland, 2002.
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- Osmari, E.K.; Cecato, U.; Macedo, F.A.F.; Souza, N.E. Nutritional quality indices of milk fat from goats on diets supplemented with different roughages. Small Rumin. Res. 2011, 98, 128–132. [Google Scholar] [CrossRef]
- Ivanova, A.; Hadzhinikolova, L. Evaluation of nutritional quality of common carp (Cyprinus carpio L.) lipidsthrough fatty acid ratios and lipid indices. Bulg. J. Agric. Sci. 2015, 21, 180–185. [Google Scholar]
- 50. STATISTICA Version 13.1 Software, StatsSoft: Kraków, Poland.
- Miciński, J.; Zwierzchowski, G.; Kowalski, I.M.; Szarek, J.; Pierożyński, B.; Raistenskis, J. The effects of bovine milk fat on human health. Pol. Ann. Med. 2012, 19, 170–175. [Google Scholar] [CrossRef]
- Unger, A.L.; Torres-Gonzalez, M.; Kraft, J. Dairy fat consumption and the risk of metabolic syndrome: An examination of the saturated fatty acids in dairy. Nutrients 2019, 11, 2200. [Google Scholar] [CrossRef]
- Lordan, R.; Zabetakis, I. Invited review: The anti-inflammatory properties of dairy lipids. J. Dairy Sci. 2017, 100, 4197–4212. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Mitra, B.; Zabetakis, I. Dairy fatsand cardiovascular disease: Do we really need to be concerned? Foods 2018, 7, 29. [Google Scholar] [CrossRef]
- Hanuš, O.; Samková, E.; Krížová, L.; Hasoňová, L.; Kala, R. Role of fatty acids in milk fat and the influence of selected factors on their variability—A Review. Molecules 2018, 23, 1636. [Google Scholar] [CrossRef]
- Gómez-Cortés, P.; Juárez, M.; Angel de la Fuente, M. Milk fat acids and potential health benefits: An update vision. Trends Food Sci. Technol. 2018, 81, 1–9. [Google Scholar] [CrossRef]
- Wongtangtintharn, S.; Oku, H.; Iwasaki, H.; Toda, T. Effect of branched-chain fatty acids on fatty acid biosynthesis of human breast cancer cells. J. Nutr. Sci. Vitaminol. 2004, 50, 137–143. [Google Scholar] [CrossRef]
- Adamska, A.; Rutkowska, J. Odd- and branched-chain fatty acids in milk fat—Characteristic and health properties. Postep. Hig. Med. Dosw. 2014, 68, 957–966. [Google Scholar] [CrossRef]
- Farag, M.A.; Gad, M.Z. Omega-9 fatty acids: Potential roles in inflammation and cancer management. J. Gen. Engin. Biotech. 2022, 20, 48. [Google Scholar] [CrossRef]
- Williams, C.M. Dietary fatty acids and human health. Ann. Zootech. 2000, 49, 165–180. [Google Scholar] [CrossRef]
- Haug, A.; Hostmark, A.T.; Harstad, O.M. Bovine milk in human nutrition: A review. Lipids Health Dis. 2007, 6, 25. [Google Scholar] [CrossRef]
- Arnould, V.M.-R.; Soyeurt, H. Genetic variability of milk fatty acids. J. Appl. Genet. 2009, 50, 29–39. [Google Scholar] [CrossRef]
- Wijendran, V.; Hayes, K.C. Dietary n-6 and n-3 fatty acid balance and cardiovascular health. Annu. Rev. Nutr. 2004, 24, 597–615. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharm. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Blaško, J.; Kubinec, R.; Górová, R.; Fábry, I.; Lorenz, W.; Soják, L. Fatty acid composition of summer and winter cows milk and butter. J. Food Nutr. Res. 2010, 49, 169–177. [Google Scholar]
- Wang, H.H.; Garruti, G.; Liu, M.; Portincasa, P.; Wang, D.Q.H. Cholesterol and Lipoprotein Metabolism and Atherosclerosis: Recent Advances in Reverse Cholesterol Transport. Ann. Hepatol. 2017, 16, s27–s42. [Google Scholar] [CrossRef] [PubMed]
- Lock, A.; Bauman, D. Modifying milk fat composition of dairy cows to enhance fatty acids beneficial to human health. Lipids 2006, 39, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Dachev, M.; Bryndová, J.; Jakubek, M.; Moučka, Z.; Urban, M. The Effects of Conjugated Linoleic Acids on Cancer. Processes 2021, 9, 454. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef]
- Yang, B.; Chen, H.; Stanton, C.; Ross, R.P.; Zhang, H.; Chen, Y.Q.; Chen, W. Review of the roles of conjugated linoleic acid in health and disease. J. Funct. Foods 2015, 15, 314–325. [Google Scholar] [CrossRef]
- Aydin, R. Conjugated linoleic acid: Structure, sources and biological properties. Turk. J. Vet. Anim. Sci. 2005, 29, 189–195. [Google Scholar]
- Park, Y. Conjugated linoleic acid (CLA): Good or bad trans fat? J. Food Comp. Anal. 2009, 22, S4–S12. [Google Scholar] [CrossRef]
- Kee, J.-I.; Ganesan, P.; Kwak, H.-S. Bioactive conjugated linoleic acid (CLA) in milk. Korean J. Food Sci. Anim. Resour. 2010, 30, 879–885. [Google Scholar] [CrossRef]
- Wahle, K.W.J.; Heys, S.D.; Rotundo, D. Conjugated linoleic acids: Are they beneficial or detrimental to health? Prog. Lipid Res. 2004, 43, 553–587. [Google Scholar] [CrossRef]
- Koba, K.; Yanagita, T. Health benefits of conjugated linoleic acid (CLA). Obes. Res. Clin. Pract. 2014, 8, e525–e532. [Google Scholar] [CrossRef]
- Jiang, J.; Björck, L.; Fondon, R. Production of conjugated linoleic acid by dairy starter cultures. J. Appl. Microbiol. 1998, 85, 95–102. [Google Scholar] [CrossRef]
- Kelly, M.L.; Berry, J.R.; Dwyer, D.A.; Griinari, J.M.; Chouinard, P.Y.; Van Amburgh, M.E.; Bauman, D.E. Dietary fatty acid sources affect conjugated linoleic acid concentrations in milk from lactating dairy cows. J. Nutr. Sci. 1998, 128, 881–885. [Google Scholar] [CrossRef]
- Chilliard, Y.; Ferlay, A.; Mansbridge, R.M.; Doreau, M. Ruminant milk fat plasticity: Nutritional control of saturated, polyunsaturated, trans and conjugated fatty acids. Anim. Res. 2000, 49, 181–205. [Google Scholar] [CrossRef]
- Kelsey, J.A.; Corl, B.A.; Collier, R.J.; Bauman, D.E. The Effect of Breed, Parity, and Stage of Lactation on Conjugated Linoleic Acid (CLA) in Milk Fat from Dairy Cows. J. Dairy Sci. 2003, 86, 2588–2597. [Google Scholar] [CrossRef]
- Collomb, M.; Schmid, A.; Sieber, R.; Wechsler, D.; Ryhänen, E.-L. Conjugated linoleic acids in milk fat: Variation and physiological effects. Int. Dairy J. 2006, 16, 1347–1361. [Google Scholar] [CrossRef]
- Zunong, M.; Hanada, M.; Aibibula, Y.; Okamoto, M.; Tanaka, K. Variations in Conjugated Linoleic Acid Concentrations in Cows Milk, Depending on Feeding Systems in Different Seasons. Asian Australas. J. Anim. Sci. 2008, 21, 1466–1472. [Google Scholar] [CrossRef]
- Żegarska, Z.; Paszczyk, B.; Rafałowski, R.; Borejszo, Z. Annual Changes in the Content of Unsaturated Fatty Acids with 18 Carbon Atoms, Including cis9trans11 C18:2 (CLA) Acid, in Milk Fat. Pol. J. Food Nutr. Sci. 2006, 56, 409–414. [Google Scholar]
- Kuczyńska, B.; Nałęcz-Tarwacka, T.; Puppel, K.; Gołębiewski, M.; Grodzki, H.; Slósarz, J. The content of bioactive components in milk depending on cow feeding model in certified ecological farms. J. Res. Appl. Agric. Eng. 2011, 56, 7–13. [Google Scholar]
- Murtaza, M.A.; Huma, N.; Hayat, Z.; Murtaza, M.S.; Meraj, A. Cheddar cheese from cow milk with elevated conjugated linoleic acid levels. J. Food Nutr. Res. 2014, 2, 506–509. [Google Scholar] [CrossRef]
- Kim, J.H.; Kwon, O.J.; Choi, N.J.; Oh, S.J.; Jeong, H.Y.; Song, M.K.; Jeong, I.; Kim, Y.J. Variations in conjugated linoleic acid (CLA) content of processed cheese by lactation time, feeding regimen, and ripening. J. Agric. Food Chem. 2009, 57, 3235–3239. [Google Scholar] [CrossRef] [PubMed]
- Sieber, R.; Collomb, M.; Aeschlimann, A.; Jelen, P.; Eyer, H. Impact of microbial cultures on conjugated linoleic acid in dairy products—A review. Int. Dairy J. 2004, 14, 1–15. [Google Scholar] [CrossRef]
- Seçkin, A.K.; Gursoy, O.; Kinik, O.; Akbulut, N. Conjugated linoleic acid (CLA) concentration, fatty acid composition and cholesterol content of same Turkish dairy products. LWT Food Sci. Technol. 2005, 38, 909–915. [Google Scholar] [CrossRef]
- Lin, H.; Boylston, T.D.; Chang, M.J.; Luedecke, L.O.; Shultz, T.D. Survey of the conjugated linoleic acid contents of dairy products. J. Diary Sci. 1995, 78, 2358–2365. [Google Scholar] [CrossRef]
- Białek, A.; Tokarz, A. Źródła pokarmowe oraz efekty prozdrowotne sprzężonych dienów kwasu linolowego (CLA). Biul. Wydz. Farm. WUM 1995, 1, 1–12. [Google Scholar] [CrossRef]
- Nunes, J.C.; Torres, A.G. Conjugated linoleic acid (CLA) in cheese: Analysis, composition, and dietary intake, (w:). In Handbook on Cheese; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2013; pp. 547–558. [Google Scholar]
- Lim, J.-N.; Oh, J.-J.; Wang, T.; Lee, J.-S.; Kim, S.-H.; Kim, Y.-H.; Lee, H.-G. trans-11 18:1 vaccenic acid (TVA) has a direct anti-carcinogenic effect on MCF-7 human mammary adenocarcinoma cells. Nutrients 2014, 6, 627–636. [Google Scholar] [CrossRef]
| Fatty Acid | Hard Cheeses | White-Mold Cheeses | Blue-Veined Cheeses | Butters | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | 10 | 10 | 10 | 10 | ||||||||
| Mean | ±SD | Min–Max | Mean | ±SD | Min–Max | Mean | ±SD | Min–Max | Mean | ±SD | Min–Max | |
| C4:0 | 17.55 | 3.27 | 14.66–24.56 | 19.69 | 2.52 | 15.92–23.22 | 17.44 | 1.61 | 16.18–20.47 | 33.09 | 2.21 | 30.78–38.67 |
| C6:0 | 12.72 | 2.16 | 10.45–17.35 | 13.45 | 1.26 | 11.74–15.42 | 12.51 | 1.23 | 11.82–14.88 | 22.72 | 1.48 | 20.86–26.26 |
| C8:0 | 7.75 | 1.51 | 5.63–10.86 | 8.41 | 0.67 | 7.69–9.55 | 7.91 | 0.82 | 7.39–9.48 | 14.09 | 0.93 | 12.79–15.95 |
| C10:0 | 18.82 | 3.14 | 12.97–24.75 | 19.58 | 1.54 | 18.22–22.46 | 18.82 | 1.91 | 17.51–22.46 | 32.04 | 2.55 | 31.78–36.33 |
| C10:1 | 1.89 | 0.34 | 1.41–2.58 | 2.15 | 0.14 | 1.95–2.36 | 2.01 | 0.18 | 1.89–2.36 | 3.20 | 0.27 | 2.91–3.74 |
| C12:0 | 21.81 | 4.06 | 15.63–28.58 | 23.13 | 1.85 | 21.75–26.65 | 22.30 | 2.25 | 20.66–26.57 | 36.09 | 3.17 | 31.17–41.60 |
| C12:1 | 0.49 | 0.08 | 0.36–0.63 | 0.25 | 0.04 | 0.19–0.32 | 0.20 | 0.02 | 0.17–0.22 | 0.30 | 0.04 | 0.26–0.35 |
| C13:0 iso | 0.57 | 0.09 | 0.46–0.72 | 0.51 | 0.04 | 0.45–0.56 | 0.47 | 0.05 | 0.44–0.57 | 0.95 | 0.10 | 0.80–1.10 |
| C13:0 | 0.67 | 0.10 | 0.56–0.84 | 0.62 | 0.09 | 0.54–0.79 | 0.57 | 0.05 | 0.53–0.66 | 2.02 | 0.21 | 1.68–2.31 |
| C14:0 iso | 0.74 | 0.18 | 0.51–1.17 | 0.91 | 0.13 | 0.69–1.06 | 0.77 | 0.06 | 0.71–0.86 | 1.09 | 0.15 | 0.77–1.34 |
| C14:0 | 76.27 | 8.14 | 93.87–93.43 | 79.25 | 7.11 | 71.14–92.01 | 75.08 | 7.16 | 70.21–88.78 | 107.83 | 6.19 | 103.30–123.16 |
| C15:0 iso | 1.46 | 0.33 | 1.10–2.20 | 1.87 | 0.26 | 1.49–2.14 | 1.55 | 0.12 | 1.41–1.70 | 2.24 | 0.22 | 1.92–2.55 |
| C15:0 aiso | 2.96 | 0.60 | 2.15–4.04 | 3.69 | 0.36 | 3.10–4.03 | 3.28 | 0.24 | 3.06–3.72 | 4.52 | 0.52 | 3.79–5.52 |
| C14:1 | 6.31 | 1.10 | 4.74–8.28 | 7.29 | 0.59 | 6.52–7.97 | 7.05 | 0.64 | 6.63–8.27 | 10.06 | 0.83 | 9.41–11.07 |
| C15:0 | 7.06 | 1.37 | 5.16–9.64 | 8.34 | 0.68 | 7.49–9.39 | 7.78 | 0.76 | 7.13–9.19 | 10.96 | 0.67 | 10.25–12.32 |
| C16:0 iso | 1.81 | 0.42 | 1.39–2.74 | 2.04 | 0.24 | 1.68–2.24 | 1.93 | 0.19 | 1.80–2.28 | 2.62 | 0.26 | 2.07–3.04 |
| C16:0 | 200.05 | 23.24 | 173.45–244.21 | 218.09 | 18.46 | 197.22–248.26 | 208.45 | 20.10 | 195.39–247.02 | 277.79 | 11.35 | 262.46–299.96 |
| C17:0 iso | 2.12 | 0.38 | 1.57–2.90 | 2.86 | 0.42 | 2.24–3.36 | 2.32 | 0.18 | 2.11–2.53 | 3.48 | 0.28 | 3.07–3.81 |
| C17:0 aiso | 1.05 | 0.19 | 0.81–1.45 | 1.37 | 0.24 | 1.04–1.70 | 1.22 | 0.10 | 1.15–1.41 | 3.81 | 0.47 | 3.19–4.66 |
| C16:1 | 9.65 | 2.07 | 7.30–14.51 | 13.65 | 0.96 | 12.63–15.34 | 13.34 | 1.44 | 12.19–16.04 | 15.26 | 1.41 | 13.41–17.83 |
| C17:0 | 4.31 | 0.97 | 3.67–6.01 | 0.51 | 0.23 | 0.30–0.83 | 0.35 | 0.03 | 0.33–0.40 | 6.44 | 0.59 | 5.61–7.18 |
| C17:1 | 1.42 | 0.25 | 1.08–1.95 | 1.79 | 0.14 | 1.58–1.99 | 1.67 | 0.14 | 1.57–1.93 | 2.29 | 0.46 | 1.61–2.76 |
| C18:0 | 55.60 | 6.81 | 41.73–62.28 | 70.82 | 8.00 | 55.03–76.51 | 65.84 | 5.90 | 62.18–77.19 | 76.75 | 5.83 | 69.47–85.65 |
| C18:1 trans6 − trans9 | 2.48 | 0.52 | 1.71–3.22 | 2.98 | 0.39 | 2.28–3.49 | 2.95 | 0.45 | 2.47–3.67 | 3.24 | 0.55 | 2.74–3.41 |
| C18:1 trans10 + trans11 | 7.09 | 1.63 | 5.10–10.16 | 11.16 | 2.94 | 7.73–16.19 | 8.20 | 0.59 | 7.55–9.13 | 10.20 | 0.98 | 8.64–11.89 |
| C18:1 trans 12 | 1.61 | 0.35 | 1.08–2.18 | 1.97 | 0.27 | 1.58–2.37 | 2.01 | 0.35 | 1.62–2.56 | 2.07 | 0.38 | 1.53–2.22 |
| C18:1 cis9 | 107.47 | 16.89 | 83.38–128.08 | 142.43 | 15.22 | 115.88–162.42 | 134.75 | 13.71 | 124.12–160.57 | 159.71 | 5.58 | 153.15–170.82 |
| C18:1 cis11 | 3.54 | 0.72 | 2.48–4.60 | 5.23 | 0.57 | 4.35–6.02 | 5.54 | 0.79 | 4.68–6.80 | 6.85 | 0.52 | 6.15-7.64 |
| C18:1 cis12 | 1.51 | 0.41 | 0.90–2.23 | 1.75 | 0.29 | 1.46–2.21 | 1.89 | 0.36 | 1.48–2.43 | 2.64 | 0.37 | 2.11-3.15 |
| C18:1 cis13 | 0.48 | 0.14 | 0.60–0.71 | 0.72 | 0.15 | 0.49–0.90 | 0.73 | 0.11 | 0.61–0.90 | 0.88 | 0.09 | 0.72-1.03 |
| C18:1 trans16 | 1.63 | 0.39 | 1.11–2.29 | 2.04 | 0.36 | 1.56–2.52 | 1.84 | 0.26 | 1.56–2.27 | 2.43 | 0.29 | 1.88-2.88 |
| C19:0 | 0.91 | 0.22 | 0.63–1.28 | 1.21 | 0.24 | 0.86–1.49 | 1.07 | 0.16 | 0.92–1.35 | 1.09 | 0.33 | 0.68-1.39 |
| C18:2 cis9trans13 | 1.09 | 0.26 | 0.77–1.66 | 1.31 | 0.33 | 0.95–1.86 | 1.15 | 0.18 | 0.96–1.45 | 1.57 | 0.36 | 0.78-2.12 |
| C18:2 cis9trans12 | 0.88 | 0.17 | 0.66–1.15 | 1.09 | 0.18 | 0.85–1.27 | 1.01 | 0.22 | 0.76–1.34 | 2.25 | 0.23 | 1.96-2.36 |
| C18:2 trans11cis15 | 0.54 | 0.24 | 0.33–1.04 | 1.28 | 0.58 | 0.75–2.40 | 0.65 | 0.03 | 0.63–0.72 | 0.95 | 0.23 | 0.85-1.18 |
| C18:2 cis9cis12 | 9.14 | 1.91 | 6.37–10.55 | 10.70 | 1.08 | 9.03–12.07 | 10.64 | 1.18 | 9.52–12.74 | 13.06 | 0.89 | 11.77-14.75 |
| C20:0 | 0.87 | 0.17 | 0.68–1.22 | 1.05 | 0.16 | 0.82–1.28 | 0.96 | 0.10 | 0.89–1.16 | 1.21 | 0.18 | 0.89-1.42 |
| C20:1 | 0.64 | 0.12 | 0.48–0.87 | 0.82 | 0.08 | 0.69–0.93 | 0.71 | 0.06 | 0.68–0.83 | 0.91 | 0.14 | 0.71-1.11 |
| C18:3 cis9cis12cis15 | 1.92 | 0.57 | 1.23–2.88 | 2.95 | 0.69 | 2.38–4.14 | 1.77 | 0.27 | 1.48–2.09 | 3.41 | 0.73 | 2.62-4.38 |
| C18:2 cis9trans11 (CLA) | 2.50 | 0.52 | 1.94–3.56 | 4.12 | 1.31 | 3.61–6.61 | 2.89 | 0.23 | 2.60–3.15 | 3.63 | 0.25 | 3.23-4.10 |
| Fatty Acids | Hard Cheeses | White-Mold Cheeses | Blue-Veined Cheeses | Butters | |
|---|---|---|---|---|---|
| n | 10 | 10 | 10 | 10 | |
| Σ SCFA 1 | Mean | 56.84 b | 61.12 b | 56.68 b | 101.93 a |
| SD | 9.5 | 5.68 | 5.51 | 6.61 | |
| Min–Max | 46.18–78.00 | 53.97–70.65 | 53.58–67.29 | 93.12–116.90 | |
| Σ BCFA 2 | Mean | 10.70 c | 13.24 b | 11.55 b,c | 18.70 a |
| SD | 2.13 | 1.51 | 0.84 | 1.84 | |
| Min–Max | 8.00–15.20 | 10.77–15.10 | 10.78–13.07 | 15.88–22.00 | |
| Σ OCFA 3 | Mean | 12.95 b | 11.03 c | 9.80 c | 20.51 a |
| SD | 2.62 | 1.17 | 1.03 | 1.41 | |
| Min–Max | 9.43–17.47 | 9.68–12.83 | 8.88–11.68 | 18.67–21.41 | |
| Σ SFA 4 | Mean | 356.44 c | 416.62 b | 394.31 b | 502.78 a |
| SD | 40.29 | 34.56 | 37.3 | 17.34 | |
| Min–Max | 307.23–430.54 | 366.88–471.64 | 370.02–465.87 | 475.39–524.61 | |
| Σ MUFA 5 | Mean | 146.21 c | 194.24 b | 182.88 b | 225.16 a |
| SD | 23.66 | 20.32 | 18.71 | 5.95 | |
| Min–Max | 101.43–175.77 | 159.56–221.47 | 168.08–217.97 | 216.81–237.00 | |
| Σ PUFA 6 | Mean | 16.07 c | 21.57 b | 18.22 c | 24.89 a |
| SD | 2.92 | 3.38 | 1.65 | 1.61 | |
| Min–Max | 12.50–21.02 | 17.13–26.56 | 16.99–21.34 | 21.90–28.06 | |
| n-3 | Mean | 1.92 b | 2.95 a | 1.77 b | 3.41 a |
| SD | 0.57 | 0.69 | 0.27 | 0.73 | |
| Min–Max | 1.73–2.30 | 2.38–4.14 | 1.48–2.09 | 2.70–4.50 | |
| n-6 | Mean | 9.14 c | 10.70 b | 10.64 b | 13.06 a |
| SD | 1.91 | 1.08 | 1.18 | 0.89 | |
| Min–Max | 7.82–13.01 | 9.03–12.07 | 9.52–12.74 | 11.80–15.05 | |
| n-6/n-3 | Mean | 5.05 a,b | 3.76 b | 6.17 a | 4.01 b |
| SD | 1.52 | 0.74 | 1.31 | 0.93 | |
| Min–Max | 3.60–8.00 | 2.42–4.47 | 4.56–7.41 | 2.83–5.00 | |
| AI 7 | Mean | 3.40 a | 2.69 c | 2.72 c | 3.09 b |
| SD | 0.43 | 0.17 | 0.04 | 0.15 | |
| Min–Max | 3.01–4.01 | 2.44–2.95 | 2.69–2.77 | 2.86–3.32 | |
| TI 8 | Mean | 4.02 a | 3.31 b | 3.42 b | 3.57 b |
| SD | 0.42 | 0.2 | 0.02 | 0.12 | |
| Min–Max | 3.65–4.54 | 2.94–3.53 | 3.39–3.44 | 3.40–3.80 | |
| HH 9 | Mean | 0.43 a | 0.26 b | 0.26 b | 0.46 a |
| SD | 0.05 | 0.02 | 0.01 | 0.02 | |
| Min–Max | 0.37–0.50 | 0.23–0.29 | 0.24–0.27 | 0.42–0.51 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paszczyk, B. Cheese and Butter as a Source of Health-Promoting Fatty Acids in the Human Diet. Animals 2022, 12, 3424. https://doi.org/10.3390/ani12233424
Paszczyk B. Cheese and Butter as a Source of Health-Promoting Fatty Acids in the Human Diet. Animals. 2022; 12(23):3424. https://doi.org/10.3390/ani12233424
Chicago/Turabian StylePaszczyk, Beata. 2022. "Cheese and Butter as a Source of Health-Promoting Fatty Acids in the Human Diet" Animals 12, no. 23: 3424. https://doi.org/10.3390/ani12233424
APA StylePaszczyk, B. (2022). Cheese and Butter as a Source of Health-Promoting Fatty Acids in the Human Diet. Animals, 12(23), 3424. https://doi.org/10.3390/ani12233424





