Bat Use of Hollows in California’s Old-Growth Redwood Forests: From DNA to Ecology
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement and Permits
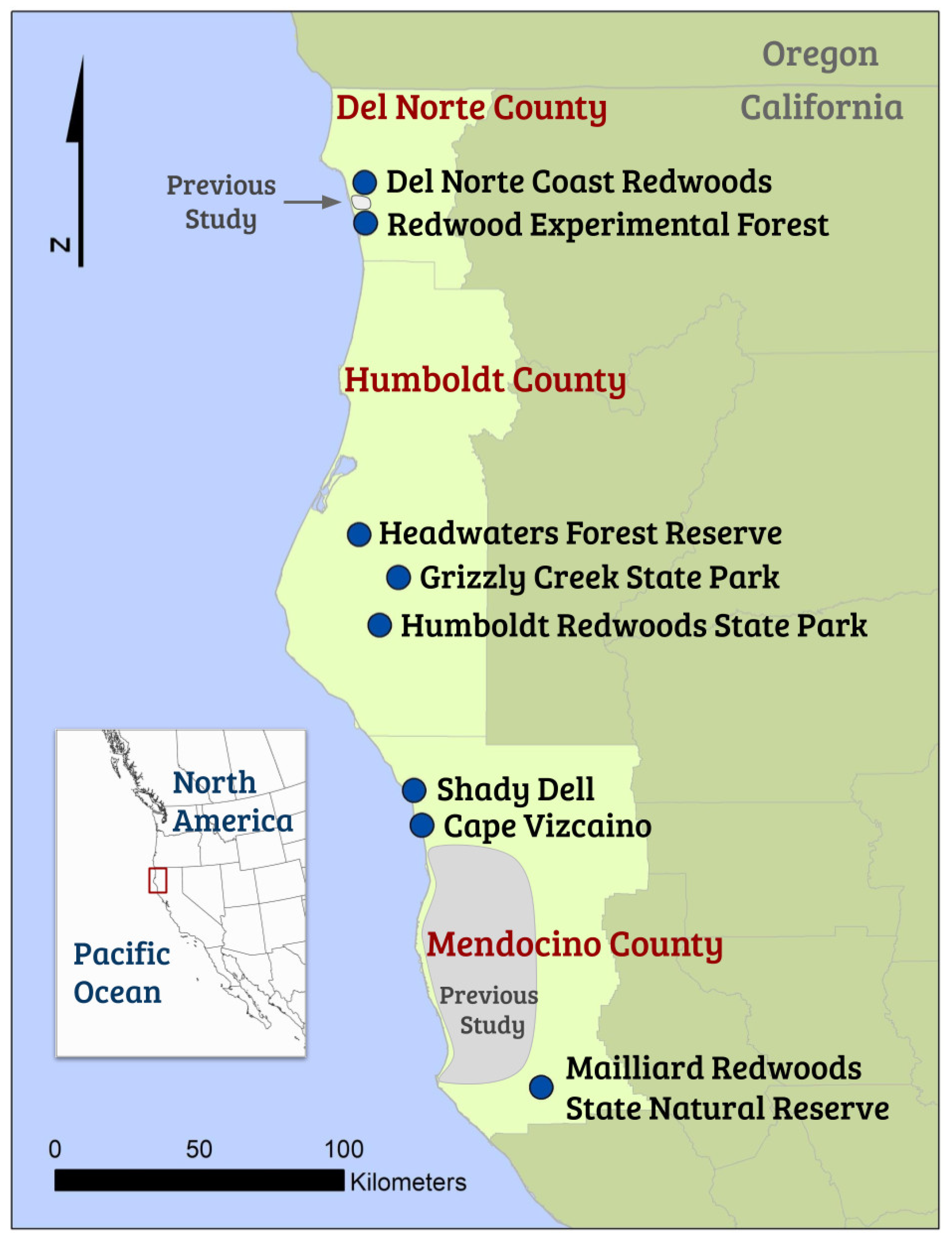
2.2. Study Site and Tree Hollow Selection
2.3. Guano Sampling
2.4. DNA Metabarcoding for Bat Species Identification
2.5. Vicinity Vegetation and Site Variables
2.6. Mixed-Effects Models
3. Results
3.1. Guano Collection and Evidence of Maternity Colonies
3.2. Bat Species Detected via DNA Metabarcoding
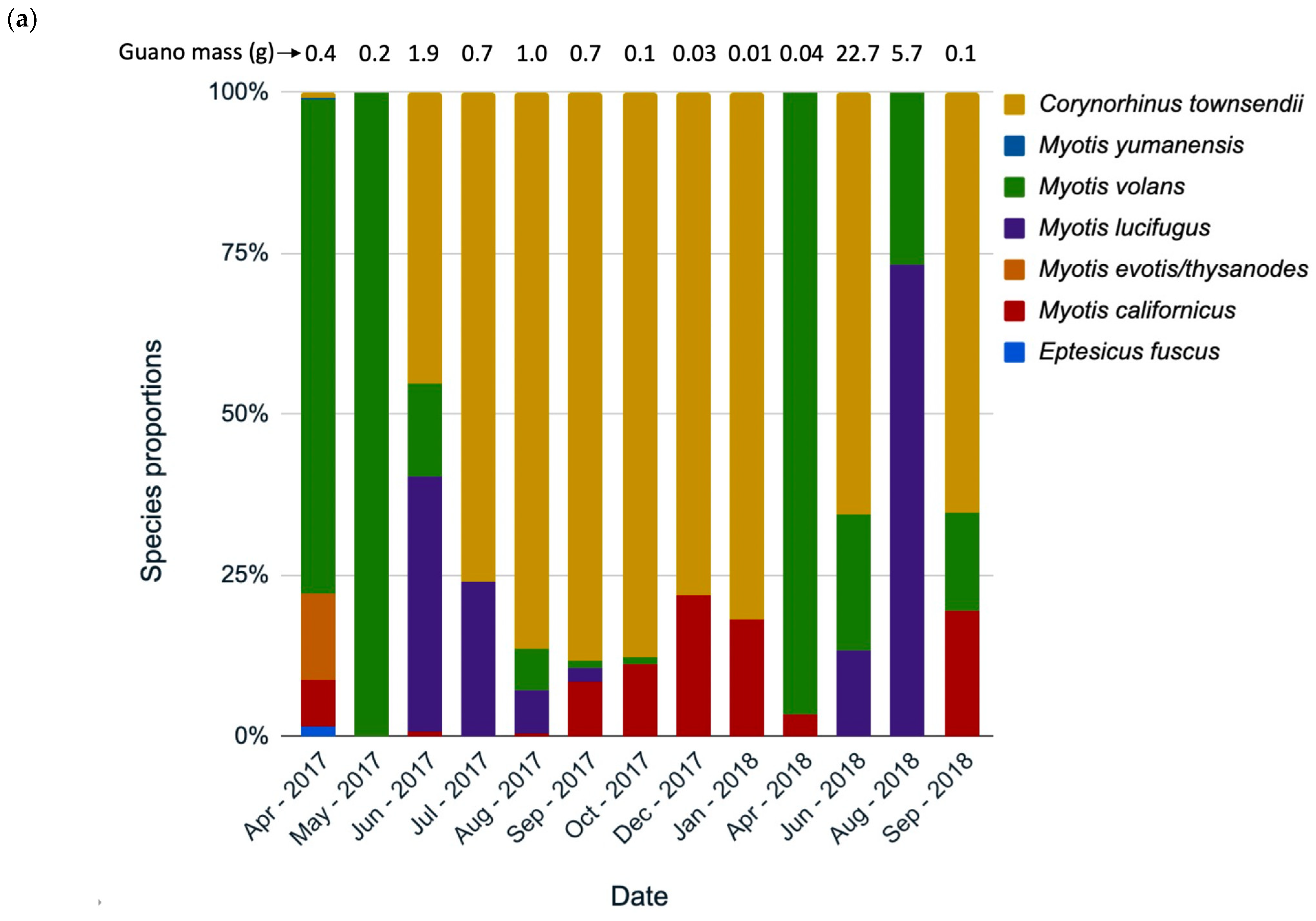
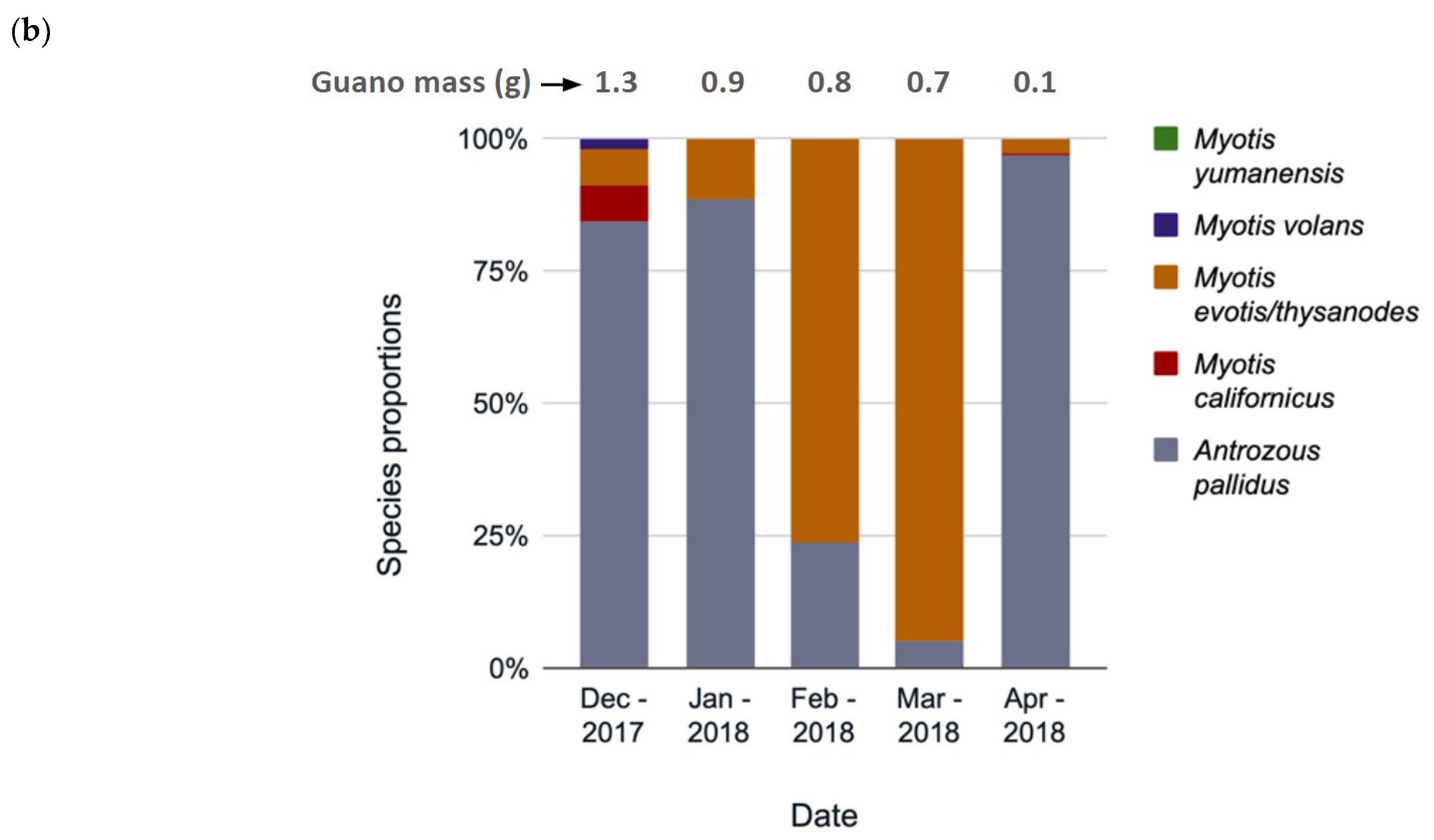
3.3. Species Composition Change over Time
3.4. Multimodel Inference of Tree Hollow Characteristics Important to Bats
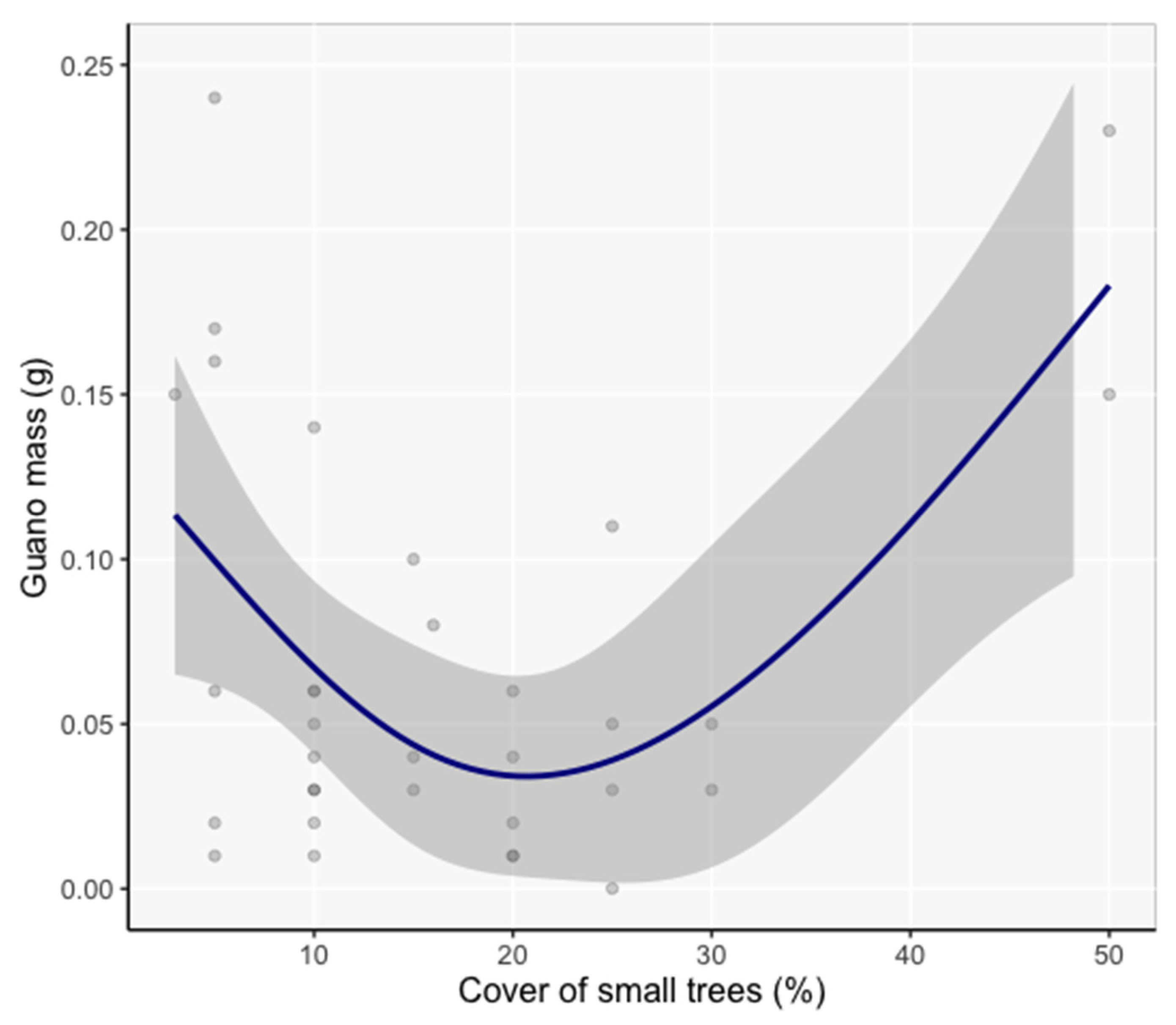
3.5. Model Inference of Vicinity and Site Characteristics Important to Bats
4. Discussion
4.1. Genetic Species Identification
4.2. Species Use by Month
4.3. Variables Affecting Hollow Use
4.3.1. Hollow Scale
4.3.2. Vicinity Scale
4.3.3. Site Scale
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boyles, J.G.; Cryan, P.M.; McCracken, G.F.; Kunz, T.H. Economic importance of bats in agriculture. Science 2011, 332, 41–42. [Google Scholar] [CrossRef] [PubMed]
- Weller, T.J.; Cryan, P.M.; O’Shea, T.J. Broadening the focus of bat conservation and research in the USA for the 21st century. Endanger. Species Res. 2009, 8, 129–145. [Google Scholar] [CrossRef]
- Reeder, D.M.; Moore, M.S. White-nose syndrome: A deadly emerging infectious disease of hibernating bats. In Bat Evolution, Ecology, and Conservation; Springer: New York, NY, USA, 2013; pp. 413–434. [Google Scholar]
- O’Shea, T.J.; Cryan, P.M.; Hayman, D.T.S.; Plowright, R.K.; Streicker, D.G. Multiple mortality events in bats: A global review. Mammal Rev. 2016, 46, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Hammerson, G.A.; Kling, M.; Harkness, M.; Ormes, M.; Young, B.E. Strong geographic and temporal patterns in conservation status of North American bats. Biol. Conserv. 2017, 212, 144–152. [Google Scholar] [CrossRef]
- Frick, W.; Kingston, T.; Flanders, J. A review of the major threats and challenges to global bat conservation. Ann. N. Y. Acad. Sci. 2019, 1469, 5–25. [Google Scholar] [CrossRef] [PubMed]
- Schaub, A.; Ostwald, J.; Siemers, B.M. Foraging bats avoid noise. J. Exp. Biol. 2008, 211, 3174–3180. [Google Scholar] [CrossRef]
- Chaverri, G.; Kunz, T.H. Response of a specialist bat to the loss of a critical resource. PLoS ONE 2011, 6, e28821. [Google Scholar]
- Fenton, M.B. Science and the conservation of bats. J. Mammal. 1997, 78, 1–14. [Google Scholar] [CrossRef]
- Hayes, J.P. Habitat ecology and conservation of bats in western coniferous forests. In Mammal Community Dynamics in Coniferous Forests of Western North America—Management and Conservation; Zabel, C.J., Anthony, R.G., Eds.; Cambridge University Press: Cambridge, UK, 2003; p. 81. [Google Scholar]
- Gellman, S.T.; Zielinski, W.J. Use by Bats of Old-Growth Redwood Hollows on the North Coast of California. J. Mammal. 1996, 77, 255–265. [Google Scholar] [CrossRef]
- Zielinski, W.J.; Gellman, S.T. Bat use of remnant old-growth redwood stands. Conserv. Biol. 1999, 13, 160–167. [Google Scholar] [CrossRef]
- Purdy, D. Bat Use of Old-Growth Redwood Basal Hollows: A Study of Capture Methods and Species Use of Redwoods. Master’s Thesis, California State University Humboldt, Arcata, CA, USA, 2002; p. 51. [Google Scholar]
- Kunz, T.H.; Lumsden, L.F. Ecology of cavity and foliage roosting bats. In Bat Ecology; Kunz, T.H., Fenton, M.B., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 3–89. [Google Scholar]
- Mazurek, M.J. A maternity roost of Townsend’s big-eared bats (Corynorhinus townsendii) in coast redwood basal hollows in northwestern California. Northwestern Nat. 2004, 85, 60–62. [Google Scholar] [CrossRef]
- Zielinski, W.J.; Mazurek, M.J.; Zinck, J. Identifying the species of bats roosting in redwood basal hollows using genetic methods. Northwest Sci. 2007, 81, 155–162. [Google Scholar] [CrossRef]
- Roberts, A. Bat Use of Old Growth Redwood Basal Hollows with Increasing Isolation in Contiguous, Remnant, and Legacy Forest Stands. Master’s Thesis, California State University, Arcata, CA, USA, 2008. [Google Scholar]
- Ruczyński, I.; Bogdanowicz, W. Summer roost selection by tree-dwelling bats Nyctalus noctula and N. leisleri: A multiscale analysis. J. Mammal. 2008, 89, 942–951. [Google Scholar] [CrossRef]
- Finney, M.A. Development of fire scar cavities on old-growth coast redwood. In Coast Redwood Forest Ecology and Management; Leblanc, J., Ed.; University of California: Berkeley, CA, USA, 1996; pp. 96–98. [Google Scholar]
- Mazurek, M.J.; Zielinski, W.J. Individual Legacy Trees Influence Vertebrate Wildlife Diversity in Commercial Forests. For. Ecol. Manag. 2004, 193, 321–334. [Google Scholar] [CrossRef]
- California Department of Fish and Wildlife (CDFW). California State Wildlife Action Plan, 2015 Update: A Conservation Legacy for Californians; Gonzales, A.G., Junko, H., Eds.; California Department of Fish and Wildlife: Sacramento, CA, USA, 2015. [Google Scholar]
- Kalcounis-Rüppell, M.C.; Psyllakis, J.M.; Brigham, R.M. Tree roost selection by bats: An empirical synthesis using meta-analysis. Wildl. Soc. Bull. 2005, 33, 1123–1132. [Google Scholar] [CrossRef]
- Lucas, J.S.; Loeb, S.C.; Jodice, P.G.R. Roost selection by Rafinesque’s big-eared bats (Corynorhinus Rafinesquii) in a pristine habitat at three spatial scales. Acta Chiropterologica 2015, 17, 131–141. [Google Scholar] [CrossRef]
- Fellers, G.M.; Pierson, E.D. Habitat use and foraging behavior of Townsend’s big-eared bat (Corynorhinus townsendii) in coastal California. J. Mammal. 2002, 83, 167–177. [Google Scholar] [CrossRef]
- O’Shea, T.J.; Cryan, P.M.; Bogan, M. United States bat species of concern: A synthesis. Proc. Calif. Acad. Sci. 2018, 65, 279. [Google Scholar]
- Perry, R.W.; Thill, R.E.; Leslie, D.M. Selection of roosting habitat by forest bats in a diverse forested landscape. For. Ecol. Manag. 2007, 238, 156–166. [Google Scholar] [CrossRef]
- Kunz, T.H.; Betke, M.; Hristov, N.I.; Vonhof, M.J. Methods for assessing colony size, population size, and relative abundance of bats. In Ecological and Behavioral Methods for the Study of Bats; Johns Hopkins University Press: Baltimore, MD, USA, 2009; pp. 133–157. [Google Scholar]
- Walker, F.M.; Williamson, C.H.; Sanchez, D.E.; Sobek, C.J.; Chambers, C.L. Species from feces: Order-wide identification of Chiroptera from guano and other non-invasive genetic samples. PLoS ONE 2016, 11, e0162342. [Google Scholar] [CrossRef] [PubMed]
- Walker, F.M.; Tobin, A.; Simmons, N.B.; Sobek, C.J.; Sanchez, D.E.; Chambers, C.L.; Fofanov, V.Y. A fecal sequel: Testing the limits of a genetic assay for bat species identification. PLoS ONE 2019, 14, e0224969. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.; Al-Ghalith, G.A.; Alm, E.J.; Arumugam, M.; Asnicar, F.; Bai, Y.; et al. Reproducible, interactive, scalable, and extensible microbiome data science using QIIME 2. Nat Biotechnol. 2019, 8, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–20. [Google Scholar] [CrossRef]
- Callahan, S.J.; Luyten, Y.A.; Gupta, Y.K.; Wilson, G.G.; Roberts, R.J.; Morgan, R.D.; Aggarwal, A.K. Structure of Type IIL Restriction-Modification Enzyme MmeI in Complex with DNA Has Implications for Engineering New Specificities. PLoS Biol. 2016, 14, e1002442. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Caproraso, J.G. Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- Benson, D.A.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Sayers, E.W. GenBank. Nucleic Acids Res. 2009, 37 (Suppl. 1), D26–D31. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Huson, D.H.; Auch, A.F.; Qi, J.; Schuster, S.C. MEGAN analysis of metagenomic data. Genome Res. 2007, 17, 377–386. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R.; Huyvaert, K.P. Erratum to: AIC Model selection and multimodel inference in behavioral ecology: Some background, observations, and comparisons. Behav. Ecol. Sociobiol. 2011, 65, 23–35. [Google Scholar] [CrossRef]
- Wood, S.N. Package mgcv: Mixed GAM Computation Vehicle with Automatic Smoothness Estimation. R Version 3.4.4. Available online: https://cran.r-project.org/web/packages/mgcv/index.html (accessed on 20 May 2021).
- Weller, T.J.; Stricker, C.A. Northern California redwood forests provide important seasonal habitat for migrant bats. Proceedings of Coast Redwood Forests in a Changing California: A Symposium for Scientists and Managers, Santa Cruz, CA, USA, 21–23 June 2011; Standiford, R.B., Weller, T.J., Piirto, D.D., Stuart, J.D., Eds.; Pacific Southwest Research Station, Forest Service, U.S. Department of Agriculture: Albany, CA, USA, 2012; pp. 447–457. [Google Scholar]
- Kennedy, J.-P.; Sillett, S.C.; Szewczak, J.M. Bat Activity Across the Vertical Gradient of an Old-Growth Sequoia Sempervirens Forest. Acta Chiropterologica 2014, 16, 53–63. [Google Scholar] [CrossRef]
- Zinck, J.M.; Duffield, D.A.; Ormsbee, P.C. Primers for identification and polymorphism assessment of Vespertilionid bats in the Pacific Northwest. Mol. Ecol. Notes 2004, 4, 239–242. [Google Scholar] [CrossRef]
- Rodriguez, R.M.; Ammerman, L.K. Mitochondrial DNA divergence does not reflect morphological differences between Myotis californicus and Myotis ciliolabrum. J. Mammal. 2004, 85, 842–851. [Google Scholar] [CrossRef]
- Lučan, R.K.; Hanák, V.; Horáček, I. Long-term re-use of tree roosts by European forest bats. For. Ecol. Manag. 2009, 258, 1301–1306. [Google Scholar] [CrossRef]
- Swift, S.M.; Racey, P.A. Resource partitioning in two species of vespertilionid bats (Chiroptera) occupying the same roost. J. Zool. 1983, 200, 249–259. [Google Scholar] [CrossRef]
- Ruczyński, I.; Nicholls, B.; MacLeod, C.D.; Racey, P.A. Selection of roosting habitats by Nyctalus noctula and Nyctalus leisleri in Białowieża Forest—Adaptive response to forest management? For. Ecol. Manag. 2010, 259, 1633–1641. [Google Scholar] [CrossRef]
- Jachowski, D.S.; Rota, C.T.; Dobony, C.A.; Ford, W.M.; Edwards, J.W. Seeing the forest through the trees: Considering roost-site selection at multiple spatial scales. PLoS ONE 2016, 11, e0150011. [Google Scholar] [CrossRef]
- Brigham, R.M.; Vonhof, M.J.; Barclay, R.M.R.; Gwilliam, J.C. Roosting behavior and roost-site preferences of forest-dwelling California bats (Myotis Californicus). J. Mammal. 1997, 78, 1231–1239. [Google Scholar] [CrossRef]
- Racey, P.A.; Swift, S.M. Variations in gestation length in a colony of pipistrelle bats (Pipistrellus pipistrellus) from year to year. J. Reprod. Fertil. 1981, 61, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Norberg, U. Evolutionary convergence in foraging niche and flight morphology in insectivorous aerial-hawking birds and bats. Ornis Scand. 1986, 17, 253–260. [Google Scholar] [CrossRef]
- Russell, W.; Jones, C. The effects of timber harvesting on the structure and composition of adjacent old-growth coast redwood forest, California, USA. Landsc. Ecol. 2001, 16, 731–741. [Google Scholar] [CrossRef]
- Bellamy, C.; Altringham, J. Predicting species distributions using record centre data: Multi-scale modelling of habitat suitability for bat roosts. PLoS ONE 2015, 10, e0128440. [Google Scholar]
- Jackson, B.K.; Stock, S.L.; Harris, L.S.; Szewczak, J.M.; Schofield, L.N.; Desrosiers, M.A. River food chains lead to riparian bats and birds in two mid-order rivers. Ecosphere 2020, 11, e03148. [Google Scholar] [CrossRef]
- Kühnert, E.; Schönbächler, C.; Arlettaz, R.; Christe, P. Roost selection and switching in two forest-dwelling bats: Implications for forest management. Eur. J. Wildl. Res. 2016, 62, 497–500. [Google Scholar] [CrossRef]
- Adams, R.A.; Thibault, K.M. Temporal resource partitioning by bats at water holes. J. Zool. 2006, 270, 466–472. [Google Scholar] [CrossRef]
- Lewis, S.E. Night roosting ecology of pallid bats (Antrozous pallidus) in Oregon. Am. Midl. Nat. 1994, 132, 219–226. [Google Scholar] [CrossRef]
- Bennett, A.; Lumsden, F.L.; Nicholls, A.O. Tree hollows as a resource for wildlife in remnant woodlands: Spatial and temporal patterns across the Northern Plains of Victoria, Australia. Pac. Conserv. Biol. 1994, 1, 222. [Google Scholar] [CrossRef]
- Ormsbee, P.C.; Kiser, J.D.; Perlmeter, S.I. Importance of night roosts to the ecology of bats. In Bats in Forests: Conservation and Management; Lacki, M.J., Hayes Allen Kurta, J.P., Eds.; Johns Hopkins University Press: Baltimore, MD, USA, 2007; p. 329. [Google Scholar]



| Hollow | Vicinity | Site |
|---|---|---|
| Diameter at Breast Height (DBH) | Cover | Distance to water |
| Aspect of opening | Canopy | Distance to road |
| Max. height of opening | Upper (tall trees) | Distance to clearing |
| Max. width of opening | Mid (small trees) | Elevation |
| Max. diameter-internal | Shrub | Number of hollows |
| Ceiling height above opening | Herbaceous | within 300 m |
| Max. diameter above opening | Within 30 m radius: | |
| Volume ( | Tree species | |
| Vegetation covering opening | Tree DBH (< or > 60 cm) | |
| Tree density |
| Del Norte Redwoods | Experimental Forest | Headwaters Forest | Grizzly Creek | Humboldt Redwoods | Shady Dell | Cape Vizcaino | Mailliard Redwoods | |
|---|---|---|---|---|---|---|---|---|
| Hollows per site | 15 | 10 | 24 | 25 | 26 | 13 | 12 | 9 |
| Number of species | 3 | 7 | 58 | 40 | 52 | 33 | 28 | 32 |
| Species percentage per site | ||||||||
| Myotis californicus | 33.3 | 42.9 | 29.3 | 17.5 | 34.6 | 21.2 | 42.9 | 21.9 |
| Myotis evotis/thysanodes | 33.3 | 28.6 | 13.8 | 20.0 | 25.0 | 9.1 | 3.6 | 31.3 |
| Corynorhinus townsendii * | 14.3 | 15.5 | 20.0 | 11.5 | 27.3 | 25.0 | 9.4 | |
| Myotis volans | 33.3 | 22.4 | 17.5 | 13.5 | 12.1 | 3.6 | 18.8 | |
| Eptesicus fuscus | 14.3 | 8.6 | 5.0 | 7.7 | 6.1 | 14.3 | ||
| Myotis yumanensis | 10.3 | 10.0 | 9.1 | 3.1 | ||||
| Myotis lucifugus ** | 10.0 | 15.2 | ||||||
| Lasionycteris noctivagans | 7.7 | 7.1 | ||||||
| Antrozous pallidus * | 15.6 | |||||||
| Lasiurus cinereus | 3.6 | |||||||
| Model | df | logLik | AICc | Delta AICc | Weight |
|---|---|---|---|---|---|
| CeilingHeight +MaxWidth | 7 | −73.93 | 152.23 | 0.00 | 0.22 |
| CeilingHeight +Volume | 7 | −73.48 | 152.30 | 0.07 | 0.21 |
| CeilingHeight +MaxHeight | 7 | −74.86 | 152.33 | 0.10 | 0.21 |
| CeilingHeight | 5 | −74.82 | 152.55 | 0.32 | 0.19 |
| CeilingHeight +MaxWidth +Volume | 9 | −73.19 | 153.82 | 1.59 | 0.10 |
| CeilingHeight +DBH | 7 | −74.56 | 154.31 | 2.08 | 0.08 |
| Null Model | 1 | −81.43 | 169.05 | 16.82 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Armstrong, A.J.; Walker, F.M.; Sobek, C.J.; Sanville, C.J.; Martin, S.L.; Szewczak, J.M. Bat Use of Hollows in California’s Old-Growth Redwood Forests: From DNA to Ecology. Animals 2022, 12, 2950. https://doi.org/10.3390/ani12212950
Armstrong AJ, Walker FM, Sobek CJ, Sanville CJ, Martin SL, Szewczak JM. Bat Use of Hollows in California’s Old-Growth Redwood Forests: From DNA to Ecology. Animals. 2022; 12(21):2950. https://doi.org/10.3390/ani12212950
Chicago/Turabian StyleArmstrong, Amon J., Faith M. Walker, Colin J. Sobek, Cheri J. Sanville, Stephanie L. Martin, and Joseph M. Szewczak. 2022. "Bat Use of Hollows in California’s Old-Growth Redwood Forests: From DNA to Ecology" Animals 12, no. 21: 2950. https://doi.org/10.3390/ani12212950
APA StyleArmstrong, A. J., Walker, F. M., Sobek, C. J., Sanville, C. J., Martin, S. L., & Szewczak, J. M. (2022). Bat Use of Hollows in California’s Old-Growth Redwood Forests: From DNA to Ecology. Animals, 12(21), 2950. https://doi.org/10.3390/ani12212950





