Relationship among Milk Conductivity, Production Traits, and Somatic Cell Score in the Italian Mediterranean Buffalo
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Data
2.2. Statistical Analyses
3. Results
3.1. Descriptive Statistics
3.2. Effect of Milk Conductivity on Milk Yield and Composition
3.3. Effect of Milk Conductivity on Somatic Cell Score
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azionale Allevatori Bufalini (ANASB). Available online: https://www.anasb.it/statistiche/ (accessed on 23 June 2022).
- Fagiolo, A.; Lai, O. Mastitis in Buffalo. Ital. J. Anim. Sci. 2007, 6, 200–206. [Google Scholar] [CrossRef]
- Salzano, A.; Manganiello, G.; Neglia, G.; Vinale, F.; De Nicola, D.; D’Occhio, M.; Campanile, G. A Preliminary Study on Metabolome Profiles of Buffalo Milk and Corresponding Mozzarella Cheese: Safeguarding the Authenticity and Traceability of Protected Status Buffalo Dairy Products. Molecules 2020, 25, 304. [Google Scholar] [CrossRef]
- Manuelian, C.L.; Visentin, G.; Boselli, C.; Giangolini, G.; Cassandro, M.; De Marchi, M. Short Communication: Prediction of Milk Coagulation and Acidity Traits in Mediterranean Buffalo Milk Using Fourier-Transform Mid-Infrared Spectroscopy. J. Dairy Sci. 2017, 100, 7083–7087. [Google Scholar] [CrossRef] [PubMed]
- Stevens, M.; Piepers, S.; De Vliegher, S. The Effect of Mastitis Management Input and Implementation of Mastitis Management on Udder Health, Milk Quality, and Antimicrobial Consumption in Dairy Herds. J. Dairy Sci. 2019, 102, 2401–2415. [Google Scholar] [CrossRef]
- Franzoi, M.; Manuelian, C.L.; Penasa, M.; De Marchi, M. Effects of Somatic Cell Score on Milk Yield and Mid-Infrared Predicted Composition and Technological Traits of Brown Swiss, Holstein Friesian, and Simmental Cattle Breeds. J. Dairy Sci. 2020, 103, 791–804. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Neglia, G.; Campanile, G.; De Marchi, M. Milk Somatic Cell Count and Its Relationship with Milk Yield and Quality Traits in Italian Water Buffaloes. J. Dairy Sci. 2020, 103, 5485–5494. [Google Scholar] [CrossRef]
- Sharma, N.; Pandey, V.; Sudhan, N.A. Comparison of some indirect screening tests for detection of subclinical mastitis in dairy cows. Bulg. J. Vet. Med. 2010, 1, 13. [Google Scholar]
- Costa, A.; Bovenhuis, H.; Penasa, M. Changes in Milk Lactose Content as Indicators for Longevity and Udder Health in Holstein Cows. J. Dairy Sci. 2020, 103, 11574–11584. [Google Scholar] [CrossRef]
- Bobbo, T.; Penasa, M.; Cassandro, M. Combining Total and Differential Somatic Cell Count to Better Assess the Association of Udder Health Status with Milk Yield, Composition and Coagulation Properties in Cattle. Ital. J. Anim. Sci. 2020, 19, 697–703. [Google Scholar] [CrossRef]
- Schwarz, D.; Santschi, D.E.; Durocher, J.; Lefebvre, D.M. Evaluation of the New Differential Somatic Cell Count Parameter as a Rapid and Inexpensive Supplementary Tool for Udder Health Management through Regular Milk Recording. Prev. Vet. Med. 2020, 181, 105079. [Google Scholar] [CrossRef]
- Bonestroo, J.; van der Voort, M.; Fall, N.; Emanuelson, U.; Klaas, I.C.; Hogeveen, H. Estimating the Nonlinear Association of Online Somatic Cell Count, Lactate Dehydrogenase, and Electrical Conductivity with Milk Yield. J. Dairy Sci. 2022, 105, 3518–3529. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lacasse, P. Mammary Tissue Damage during Bovine Mastitis: Causes and Control1. J. Anim. Sci. 2008, 86, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, B.J. Bovine Mastitis: Milk Compositional Changes and Related Diagnostic Tests. J. Dairy Res. 1981, 48, 167–188. [Google Scholar] [CrossRef] [PubMed]
- Norberg, E.; Hogeveen, H.; Korsgaard, I.R.; Friggens, N.C.; Sloth, K.H.M.N.; Løvendahl, P. Electrical Conductivity of Milk: Ability to Predict Mastitis Status. J. Dairy Sci. 2004, 87, 1099–1107. [Google Scholar] [CrossRef]
- Milner, P.; Page, K.L.; Walton, A.W.; Hillerton, J.E. Detection of Clinical Mastitis by Changes in Electrical Conductivity of Foremilk Before Visible Changes in Milk. J. Dairy Sci. 1996, 79, 83–86. [Google Scholar] [CrossRef]
- Díaz, J.R.; Romero, G.; Muelas, R.; Alejandro, M.; Peris, C. Effect of Intramammary Infection on Milk Electrical Conductivity in Murciano-Granadina Goats. J. Dairy Sci. 2012, 95, 718–726. [Google Scholar] [CrossRef]
- Khatun, M.; Clark, C.E.F.; Lyons, N.A.; Thomson, P.C.; Kerrisk, K.L.; García, S.C. Early Detection of Clinical Mastitis from Electrical Conductivity Data in an Automatic Milking System. Anim. Prod. Sci. 2017, 57, 1226. [Google Scholar] [CrossRef]
- Inzaghi, V.; Zucali, M.; Thompson, P.D.; Penry, J.F.; Reinemann, D.J. Changes in Electrical Conductivity, Milk Production Rate and Milk Flow Rate Prior to Clinical Mastitis Confirmation. Ital. J. Anim. Sci. 2021, 20, 1554–1561. [Google Scholar] [CrossRef]
- Moroni, P.; Sgoifo Rossi, C.; Pisoni, G.; Bronzo, V.; Castiglioni, B.; Boettcher, P.J. Relationships Between Somatic Cell Count and Intramammary Infection in Buffaloes. J. Dairy Sci. 2006, 89, 998–1003. [Google Scholar] [CrossRef]
- Špakauskas, V.; Klimien, I.; Matuseviius, A. A Comparison of Indirect Methods for Diagnosis of Subclinical Mastitis in Lactating Dairy Cows. Vet. Arhiv 2006, 76, 101–109. [Google Scholar]
- Ali, A.K.A.; Shook, G.E. An Optimum Transformation for Somatic Cell Concentration in Milk. J. Dairy Sci. 1980, 63, 487–490. [Google Scholar] [CrossRef]
- Pinheiro, J.; Douglas, B.; Deepayan, S.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. 2020. Available online: https://CRAN.R-project.org/package=nlme (accessed on 20 May 2022).
- R Core Team. R—A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning: With Applications in R; Springer Texts in Statistics; Springer: New York, NY, USA, 2013. [Google Scholar]
- Nakagawa, S.; Schielzeth, H. A General and Simple Method for Obtaining R2 from Generalized Linear Mixed-Effects Models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Lenth, R.V.; Buerkner, P.; Herve, M.; Love, J.; Miguez, F.; Riebl, H.; Singmann, H. emmeans: Estimated Marginal Means, Aka Least-Squares Means; R package version 1.8.0. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 23 May 2022).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Finocchiaro, A.; Conte, F. Conductivity of Donkey Milk. Ital. J. Food Saf. 2009, 1, 65. [Google Scholar] [CrossRef]
- Caria, M.; Chessa, G.; Murgia, L.; Todde, G.; Pazzona, A. Development and Test of a Portable Device to Monitor the Health Status of Sarda Breed Sheep by the Measurement of the Milk Electrical Conductivity. Ital. J. Anim. Sci. 2016, 15, 275–282. [Google Scholar] [CrossRef]
- Gelasakis, A.I.; Giannakou, R.; Valergakis, G.E.; Fortomaris, P.; Kominakis, A.; Arsenos, G. Prediction of Sheep Milk Chemical Composition Using Milk Yield, PH, Electrical Conductivity and Refractive Index. J. Dairy Res. 2018, 85, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Paudyal, S.; Melendez, P.; Manriquez, D.; Velasquez-Munoz, A.; Pena, G.; Roman-Muniz, I.N.; Pinedo, P.J. Use of Milk Electrical Conductivity for the Differentiation of Mastitis Causing Pathogens in Holstein Cows. Animal 2020, 14, 588–596. [Google Scholar] [CrossRef]
- Hogan, J.S.; Bogacz, V.L.; Thompson, L.M.; Romig, S.; Schoenberger, P.S.; Weiss, W.P.; Smith, K.L. Bacterial Counts Associated with Sawdust and Recycled Manure Bedding Treated with Commercial Conditioners. J. Dairy Sci. 1999, 82, 1690–1695. [Google Scholar] [CrossRef]
- Amiri, P.; Fallah Rad, A.H.; Heidarpour, M.; Azizzadeh, M.; Khoramian, B. Diagnostic Accuracy of Milk Oxidation Markers for Detection of Subclinical Mastitis in Early Lactation Dairy Cows. Comp. Clin. Pathol. 2020, 29, 95–101. [Google Scholar] [CrossRef]
- Pyörälä, S. Indicators of Inflammation in the Diagnosis of Mastitis. Vet. Res. 2003, 34, 565–578. [Google Scholar] [CrossRef]
- Nielen, M.; Deluyker, H.; Schukken, Y.H.; Brand, A. Electrical Conductivity of Milk: Measurement, Modifiers, and Meta Analysis of Mastitis Detection Performance. J. Dairy Sci. 1992, 75, 606–614. [Google Scholar] [CrossRef]
- Kasikçi, G.; Çetin, Ö.; Bingöl, E.B.; Gündüz, M.C. Relations between electrical conductivity, somatic cell count, California mastitis test and some quality parameters in the diagnosis of subclinical mastitis in dairy cows. Turk. J. Vet. Anim. Sci. 2012, 36, 49–55. [Google Scholar]
- Vilas Boas, D.F.; Vercesi Filho, A.E.; Pereira, M.A.; Roma Junior, L.C.; El Faro, L. Association between Electrical Conductivity and Milk Production Traits in Dairy Gyr Cows. J. Appl. Anim. Res. 2017, 45, 227–233. [Google Scholar] [CrossRef]
- Sadoon, A.S. Clinical and Subclinical Mastitis in Buffalue in Mosul Area, Iraq. Iraqi J. Vet. Sci. 2021, 36, 177–186. [Google Scholar] [CrossRef]
- Diaz, J.R.; Romero, G.; Muelas, R.; Sendra, E.; Pantoja, J.C.F.; Paredes, C. Analysis of the Influence of Variation Factors on Electrical Conductivity of Milk in Murciano-Granadina Goats. J. Dairy Sci. 2011, 94, 3885–3894. [Google Scholar] [CrossRef]
- Romero, G.; Pantoja, J.C.F.; Sendra, E.; Peris, C.; Díaz, J.R. Analysis of the Electrical Conductivity in Milking Fractions as a Mean for Detecting and Characterizing Mastitis in Goats. Small Rum. Res. 2012, 107, 157–163. [Google Scholar] [CrossRef]
- Tangorra, F.M.; Zaninelli, M.; Costa, A.; Agazzi, A.; Savoini, G. Milk Electrical Conductivity and Mastitis Status in Dairy Goats: Results from a Pilot Study. Small Rum. Res. 2010, 90, 109–113. [Google Scholar] [CrossRef]
- Romero, G.; Roca, A.; Alejandro, M.; Muelas, R.; Díaz, J.R. Relationship of Mammary Gland Health Status and Other Noninfectious Factors with Electrical Conductivity of Milk in Manchega Ewes. J. Dairy Sci. 2017, 100, 1555–1567. [Google Scholar] [CrossRef]
- Galfi, A.; Radinovic, M.; Milanov, D.; Bobos, S.; Pajic, M.; Savic, S.; Davidov, I. Electrical Conductivity of Milk and Bacteriological Findings in Cows with Subclinical Mastitis. Biotehnol. Anim. Husb. 2015, 31, 533–541. [Google Scholar] [CrossRef]
- Kuropatwińska, I.; Bogucki, M.; Miller, M.; Sawa, A. Relationships between Milk Electrical Conductivity, Daily Milk Yield, and Milking Ability of Primiparous and Multiparous Cows. J. Anim. Plant Sci. 2020, 30, 298–304. [Google Scholar] [CrossRef]
- Singh, M.; Yadav, P.; Sharma, A.; Garg, V.K.; Mittal, D. Estimation of Mineral and Trace Element Profile in Bubaline Milk Affected with Subclinical Mastitis. Biol. Trace Element Res. 2017, 176, 305–310. [Google Scholar] [CrossRef]
- Woolford, M.W.; Williamson, J.H.; Henderson, H.V. Changes in Electrical Conductivity and Somatic Cell Count between Milk Fractions from Quarters Subclinically Infected with Particular Mastitis Pathogens. J. Dairy Res. 1998, 65, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.; Buckley, F.; Berry, D.P.; Rath, M.; Pierce, K.; Byrne, N.; Dillon, P. Effects of Breed, Feeding System, and Parity on Udder Health and Milking Characteristics. J. Dairy Sci. 2007, 90, 5767–5779. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.L.; Cue, R.I.; Botaro, B.G.; Horst, J.A.; Valloto, A.A.; Santos, M.V. Milk Losses Associated with Somatic Cell Counts by Parity and Stage of Lactation. J. Dairy Sci. 2018, 101, 4357–4366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mabrook, M.F.; Petty, M.C. Effect of Composition on the Electrical Conductance of Milk. J. Food Eng. 2003, 60, 321–325. [Google Scholar] [CrossRef]
- Moussaoui, F.; Vangroenweghe, F.; Haddadi, K.; Le Roux, Y.; Laurent, F.; Duchateau, L.; Burvenich, C. Proteolysis in Milk During Experimental Escherichia Coli Mastitis. J. Dairy Sci. 2004, 87, 2923–2931. [Google Scholar] [CrossRef]
- Costa, A.; Negrini, R.; De Marchi, M.; Campanile, G.; Neglia, G. Phenotypic Characterization of Milk Yield and Quality Traits in a Large Population of Water Buffaloes. Animals 2020, 10, 327. [Google Scholar] [CrossRef]
- Mottram, T.; Rudnitskaya, A.; Legin, A.; Fitzpatrick, J.L.; Eckersall, P.D. Evaluation of a Novel Chemical Sensor System to Detect Clinical Mastitis in Bovine Milk. Biosens. Bioelectron. 2007, 22, 2689–2693. [Google Scholar] [CrossRef]
- Berghof, T.V.L.; Poppe, M.; Mulder, H.A. Opportunities to Improve Resilience in Animal Breeding Programs. Front. Genet. 2019, 9, 692. [Google Scholar] [CrossRef]
- Khatun, M.; Thomson, P.C.; Kerrisk, K.L.; Lyons, N.A.; Clark, C.E.F.; Molfino, J.; García, S.C. Development of a New Clinical Mastitis Detection Method for Automatic Milking Systems. J. Dairy Sci. 2018, 101, 9385–9395. [Google Scholar] [CrossRef]
- Bobbo, T.; Matera, R.; Pedota, G.; Ramirez-Diaz, J.; Manunza, A.; Stella, A.; Cotticelli, A.; Neglia, G.; Biffani, S. Machine learning to predict somatic cell count at the subsequent test-day record in the Italian Mediterranean Buffaloes. In Proceedings of the 12th World Congress of Genetics Applied to Livestock Production, Rotterdam, Netherlands, 2–7 July 2022; Available online: https://www.wageningenacademic.com/pb-assets/wagen/WCGALP2022/15_005.pdf (accessed on 27 July 2022).
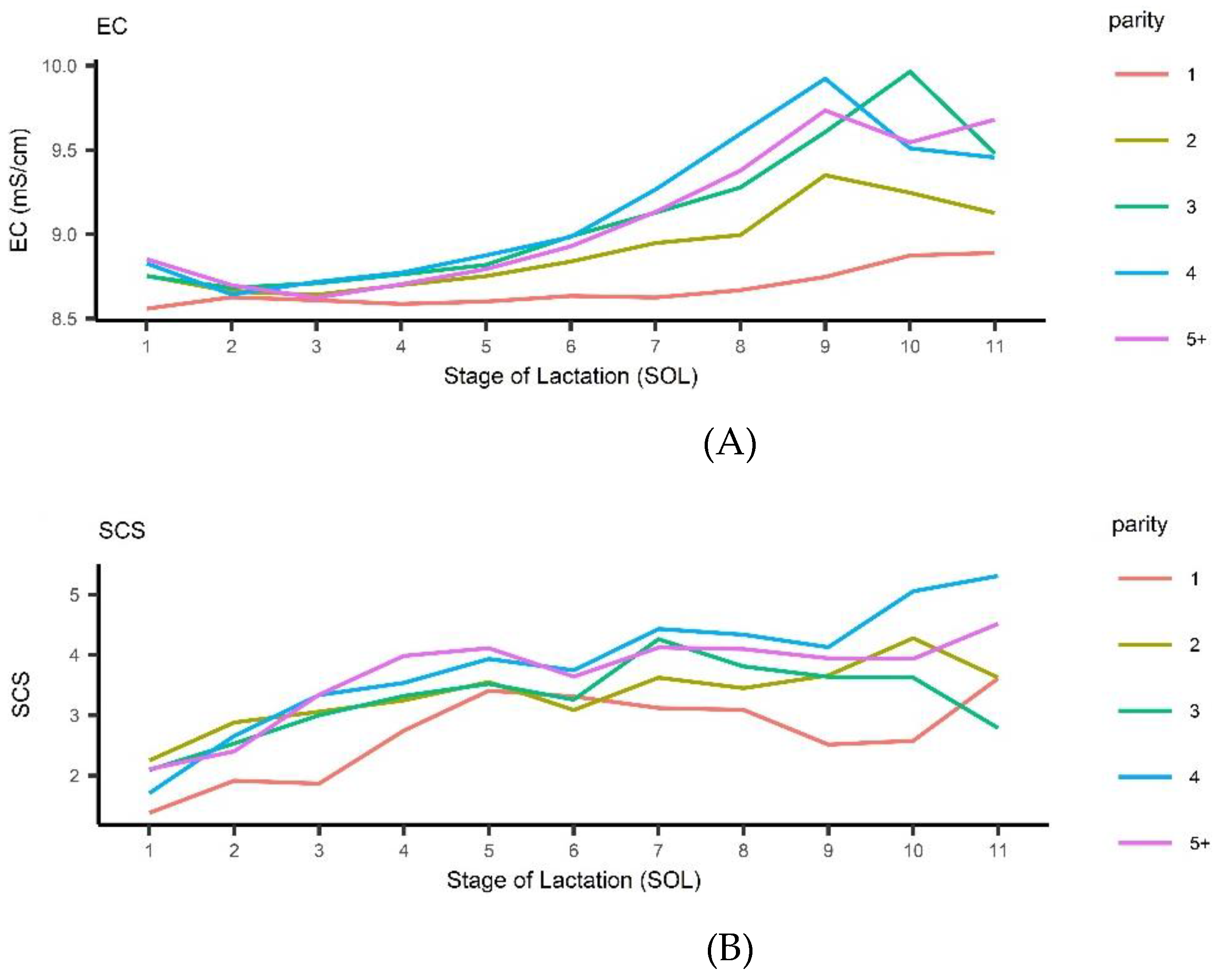
| Parity | Milk (kg/d) | Fat % | Protein % | SCS | EC (mS/cm) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Std. Dev. | Mean | Std. Dev. | Mean | Std. Dev. | Mean | Std. Dev. | Mean | Std. Dev. | |
| 1 | 8.96 | 3.39 | 9.30 | 1.93 | 4.83 | 0.37 | 2.73 | 2.15 | 8.64 | 0.50 |
| 2 | 9.97 | 4.06 | 8.88 | 1.81 | 4.73 | 0.36 | 3.24 | 2.18 | 8.82 | 0.84 |
| 3 | 10.84 | 4.43 | 8.77 | 1.78 | 4.63 | 0.39 | 3.26 | 2.08 | 8.91 | 0.96 |
| 4 | 10.42 | 4.32 | 8.68 | 1.79 | 4.65 | 0.38 | 3.58 | 2.19 | 9.05 | 1.07 |
| 5+ | 10.60 | 4.41 | 8.49 | 1.70 | 4.60 | 0.38 | 3.59 | 2.26 | 8.99 | 1.10 |
| Model a | Trait | YM b | SOL c | Par d | (SOL * Par) e | (EC * Par) f |
|---|---|---|---|---|---|---|
| 1 | MY (kg/day) | 279.3 *** | 318.2 *** | 32.3 *** | 151.3 *** | 159.9 *** |
| 1 | FP (%/day) | 46.1 *** | 437.1 *** | 1.9 ns | 65.9 ** | 5.7 ns |
| 1 | PP (%/day) | 323.3 *** | 407.6 *** | 4.5 ns | 88.4 *** | 15.3 ** |
| Parity | MY a | PP b | FP | |||
|---|---|---|---|---|---|---|
| b | p Value | b | p Value | b | p Value | |
| 1 | 0.0029 ± 0.17 | 0.99 | −0.0014 ± 0.02 | 0.9436 | −0.1308 ± 0.11 | 0.2452 |
| 2 | −0.4385 ± 0.08 | <0.0001 | −0.0101 ± 0.01 | 0.2906 | −0.0415 ± 0.05 | 0.4393 |
| 3 | −0.5541 ± 0.09 | <0.0001 | −0.0283 ± 0.01 | 0.0091 | −0.0425 ± 0.06 | 0.4784 |
| 4 | −0.5468 ± 0.09 | <0.0001 | −0.0089 ± 0.01 | 0.3948 | −0.0051 ± 0.06 | 0.9282 |
| 5+ | −0.6231 ± 0.08 | <0.0001 | −0.0243 ± 0.01 | 0.0095 | −0.0908 ± 0.05 | 0.0681 |
| Regression Parameter | YM a | SOL b | Par c | Milk d | (SOL * Par) e | (EC * Par) f |
|---|---|---|---|---|---|---|
| EC at milk recording | 77.5 *** | 41.6 *** | 16.3 *** | 91.2 *** | 1.2 ns | 22.9 *** |
| EC 3 days before milk recording | 77.2 *** | 41.2 *** | 16.1 *** | 90.9 *** | 1.1 ns | 17.2 *** |
| EC mean during 5 days before milk recording | 77.8 *** | 41.5 *** | 16.4 *** | 91.6 *** | 1.2 ns | 25.7 *** |
| EC standard deviation 5 days before milk recording | 77.3 *** | 40.9 *** | 16.1 *** | 90.0 *** | 1.1 ns | 18.9 *** |
| EC change (slope) 5 days before milk recording | 76.64 *** | 39.7 *** | 14.9 *** | 88.6 *** | 1.1 ns | 0.5 ns |
| Parity | EC at Milk Recording | EC 3 Days before Milk Recording | EC Mean During 5 Days before Milk Recording | EC Standard Deviation 5 Days before Milk Recording | ||||
|---|---|---|---|---|---|---|---|---|
| SCS | p Value | SCS | p Value | SCS | p Value | SCS | p Value | |
| 1 | 0.600 ± 0.14 | <0.0001 | 0.26 ± 0.10 | 0.01115 | 0.387 ± 0.13 | 0.0027 | 0.953 ± 0.21 | <0.0001 |
| 2 | 0.391 ± 0.07 | <0.0001 | 0.433 ± 0.07 | <0.0001 | 0.571 ± 0.08 | <0.0001 | 0.612 ± 0.12 | <0.0001 |
| 3 | 0.321 ± 0.08 | <0.0001 | 0.245 ± 0.07 | 0.00099 | 0.369 ± 0.09 | <0.0001 | 0.476 ± 0.13 | 0.00021 |
| 4 | 0.408 ± 0.07 | <0.0001 | 0.249 ± 0.06 | <0.0001 | 0.378 ± 0.08 | <0.0001 | 0.635 ± 0.13 | <0.0001 |
| 5+ | 0.277 ± 0.06 | <0.0001 | 0.309 ± 0.07 | <0.0001 | 0.409 ± 0.08 | <0.0001 | 0.530 ± 0.14 | 0.00018 |
| Regression Parameter | AIC | Marginal | Conditional |
|---|---|---|---|
| EC mean during 5 days before milk recording | 18,542 | 0.247 | 0.371 |
| EC 3 days before milk recording | 18,584 | 0.240 | 0.367 |
| EC at milk recording | 18,555 | 0.246 | 0.365 |
| EC standard deviation during 5 days before milk recording | 18,575 | 0.243 | 0.364 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matera, R.; Di Vuolo, G.; Cotticelli, A.; Salzano, A.; Neglia, G.; Cimmino, R.; D’Angelo, D.; Biffani, S. Relationship among Milk Conductivity, Production Traits, and Somatic Cell Score in the Italian Mediterranean Buffalo. Animals 2022, 12, 2225. https://doi.org/10.3390/ani12172225
Matera R, Di Vuolo G, Cotticelli A, Salzano A, Neglia G, Cimmino R, D’Angelo D, Biffani S. Relationship among Milk Conductivity, Production Traits, and Somatic Cell Score in the Italian Mediterranean Buffalo. Animals. 2022; 12(17):2225. https://doi.org/10.3390/ani12172225
Chicago/Turabian StyleMatera, Roberta, Gabriele Di Vuolo, Alessio Cotticelli, Angela Salzano, Gianluca Neglia, Roberta Cimmino, Danila D’Angelo, and Stefano Biffani. 2022. "Relationship among Milk Conductivity, Production Traits, and Somatic Cell Score in the Italian Mediterranean Buffalo" Animals 12, no. 17: 2225. https://doi.org/10.3390/ani12172225
APA StyleMatera, R., Di Vuolo, G., Cotticelli, A., Salzano, A., Neglia, G., Cimmino, R., D’Angelo, D., & Biffani, S. (2022). Relationship among Milk Conductivity, Production Traits, and Somatic Cell Score in the Italian Mediterranean Buffalo. Animals, 12(17), 2225. https://doi.org/10.3390/ani12172225








