Description of a Sarcoptic Mange Outbreak in Alpine Chamois Using an Enhanced Surveillance Approach
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
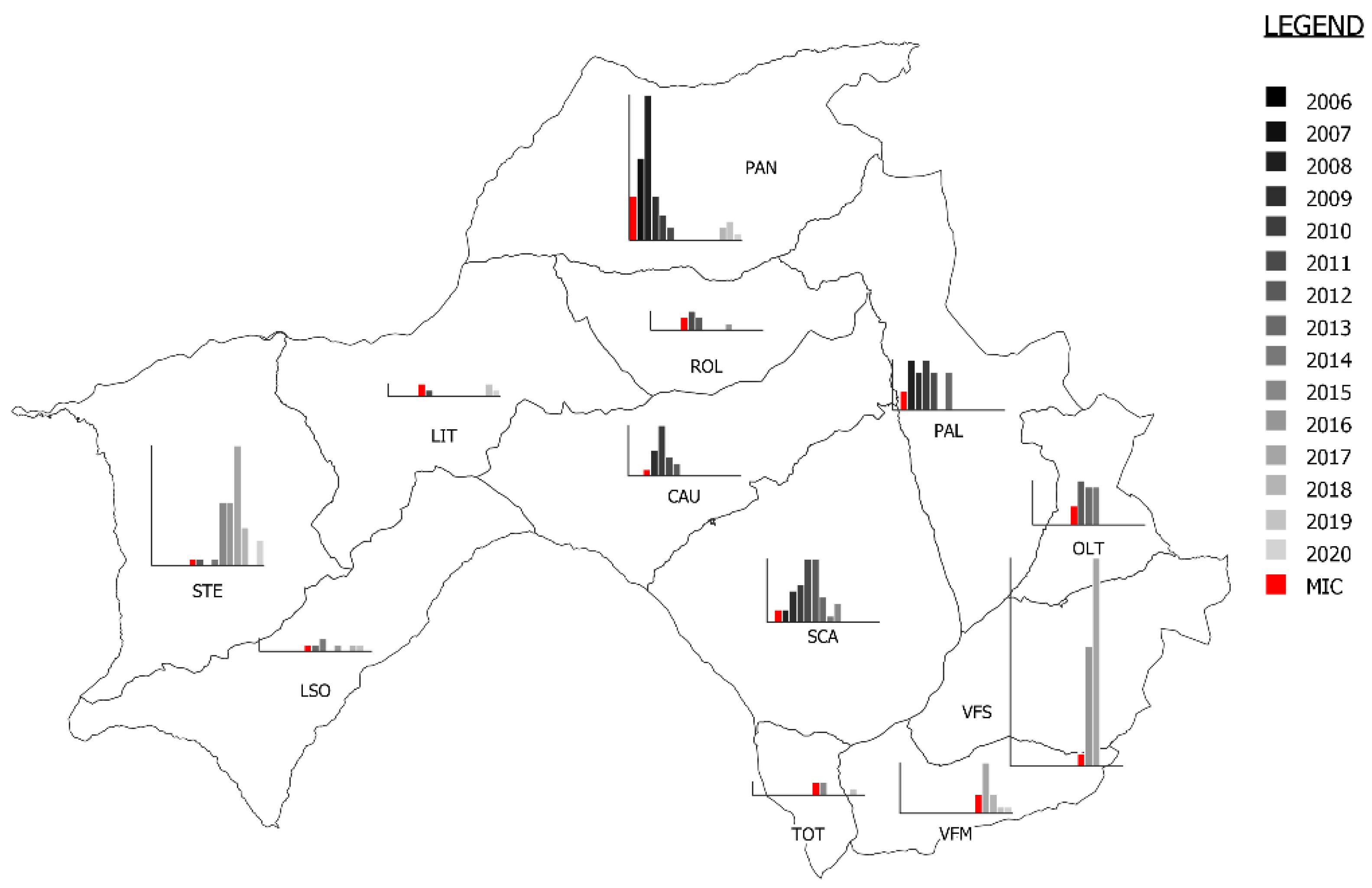
2.1. Study Area
- Mange management strategy in chamois populations with applications of seven types of hunting strategies (including hunting suspension), inside the mange zones, according to the epidemiological situation as described in Table S1;
- Definition of mange case: cadavers with typical skin lesions confirmed by a laboratory diagnosis of S. scabiei.
- A general systematic passive-surveillance (hereafter termed “ordinary surveillance”) protocol in order to estimate the impact of mange on chamois populations (details are provided in Section 2.3);
- Training of game wardens and local hunters for the detections of cases and the definition of protocols for the proper handling of mangy chamois;
- Deployment of standardized population censuses using the methodology of block count in order to estimate the demographic decrease (details are provided in Section 2.2).
2.2. Chamois Demographic Data
2.3. Ordinary Surveillance Data
- Mangy: typical skin lesions confirmed by laboratory diagnosis of S. scabiei;
- Not mangy: no skin lesions, or suspect lesion not confirmed at the laboratory as S. scabiei;
- Undetermined: poor conservation status—unreliable diagnosis.
2.4. Intensive Census and Enhanced Surveillance in CA
2.5. Data Analysis
3. Results
3.1. Description of the Mange Epidemic Using Ordinary Surveillance Data
3.2. Description of Mange Epidemic in the CA Using Enhanced Surveillance Data
3.3. Comparison between Ordinary and Enhanced Surveillance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gortázar, C.; Ferroglio, E.; Höfle, U.; Frölich, K.; Vicente, J. Diseases Shared between Wildlife and Livestock: A European Perspective. Eur. J. Wildl. Res. 2007, 53, 241–256. [Google Scholar] [CrossRef]
- Tompkins, D.M.; Carver, S.; Jones, M.E.; Krkošek, M.; Skerratt, L.F. Emerging Infectious Diseases of Wildlife: A Critical Perspective. Trends Parasitol. 2015, 31, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Escobar, L.E.; Carver, S.; Cross, P.C.; Rossi, L.; Almberg, E.S.; Yabsley, M.J.; Niedringhaus, K.D.; Van Wick, P.; Dominguez-Villegas, E.; Gakuya, F.; et al. Sarcoptic Mange: An Emerging Panzootic in Wildlife. Transbound. Emerg. Dis. 2021, 69, 927–942. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Tizzani, P.; Rambozzi, L.; Moroni, B.; Meneguz, P.G. Sanitary emergencies at the wild/domestic caprines interface in Europe. Animals 2019, 9, 922. [Google Scholar] [CrossRef] [PubMed]
- Pérez, J.M.; Ruiz-Martínez, I.; Granados, J.E.; Soriguer, R.C.; Fandos, P. The dynamics of sarcoptic mange in the ibex population of Sierra Nevada in Spain—Influence of climatic factors. J. Wildl. Res. 1997, 2, 86–89. [Google Scholar]
- León-Vizcaíno, L.; Ruíz de Ybáñez, M.R.; Cubero, M.J.; Ortíz, M.J.; Espinosa, J.; Pérez, L.; Simon, M.A.; Alonso, F. Sarcoptic mange in spanish ibex from Spain. J. Wildl. Dis. 1999, 35, 647–659. [Google Scholar] [CrossRef]
- Schaschl, E. Gamsräude; Österreichischer Jagd- und Fischereiverlag: Wien, Austria, 2003; p. 160. [Google Scholar]
- Rossi, L.; Fraquelli, C.; Vesco, U.; Permunian, R.; Sommavilla, G.M.; Carmignola, G.; Da Pozzo, R.; Meneguz, P.G. Descriptive Epidemiology of a Scabies Epidemic in Chamois in the Dolomite Alps, Italy. Eur. J. Wildl. Res. 2007, 53, 131–141. [Google Scholar] [CrossRef]
- Pence, D.B.; Ueckermann, E. Sarcoptic mange in wildlife. Rev. Sci. Tech. 2002, 21, 385–398. [Google Scholar] [CrossRef]
- Rossi, L.; Menzano, A.; Sommavilla, G.M.; De Martin, P.; Cadamuro, A.; Rodolfi, M.; Coleselli, A.; Ramanzin, M. Actions for the recovery of an alpine ibex herd affected by epidemic scabies: The Marmolada case, Italy. In Proceedings of the III. International Conference on Alpine ibex, Pontresina, Switzerland, 12–14 October 2006. [Google Scholar]
- Pérez-Barbería, F.J.; Palacios, B.; González-Quirós, P.; Cano, M.; Nores, C.; Díaz, A. La evolución de la población del rebeco en la cordillera Cantábrica. In El Rebeco Cantábrico (Rupicapra pyrenaica parva). In Conservación y Gestión de sus Poblaciones, 1st ed.; Pérez-Barbería, F.J., Palacios, B., Eds.; Ministerio de Medio Ambiente, Medio Rural y Marino, Naturaleza y Parques Nacionales: Madrid, Spain, 2009; pp. 106–125. [Google Scholar]
- Leon Vizcaino, L.; Astorga, R.; Escos, J.; Alonso, F.; Alados, C.; Contreras, A.; Cubero, M.J. Epidemiologıa de la sarna sarcoptica en el Parque Natural de las Sierras de Cazorla, Segura y Las Villas. In Proceedings of the International Congress on the Genus Capra in Europe, Ronda, Spain, 20–22 October 1992; Junta Rectora del Parque Natural Sierra de las Nieves, Consejerıa de Medio Ambiente, Junta de Andalucıa, Eds.; Junta de Andalucia: Sevilla, Spain, 1993. [Google Scholar]
- Rossi, L.; Meneguz, P.G.; De Martin, P.; Rodolfi, M. The Epizootiology of Sarcoptic Mange in Chamois, Rupicapra rupicapra, from the Italian Eastern Alps. Parassitologia 1995, 37, 233–240. [Google Scholar]
- Turchetto, S.; Obber, F.; Permunian, R.; Vendrami, S.; Lorenzetto, M.; Ferré, N.; Stancampiano, L.; Rossi, L.; Citterio, C.V. Spatial and Temporal Explorative Analysis of Sarcoptic Mange in Alpine Chamois (Rupicapra r. rupicapra). Hystrix 2014, 25, 25–30. [Google Scholar] [CrossRef]
- Pérez, J.M.; Granados, J.E.; Espinosa, J.; Ráez-Bravo, A.; López-Olvera, J.R.; Rossi, L.; Meneguz, P.G.; Angelone, S.; Fandos, P.; Soriguer, R.C. Biology and Management of Sarcoptic Mange in Wild Caprinae Populations. Mammal Rev. 2021, 51, 82–94. [Google Scholar] [CrossRef]
- Loison, A.; Appolinaire, J.; Jullien, J.M.; Dubray, D. How reliable are total counts to detect trends in population size of chamois Rupicapra rupicapra and R. pyrenaica? Wildl. Biol. 2006, 12, 77–88. [Google Scholar] [CrossRef]
- Corlatti, L.; Fattorini, L.; Nelli, L. The use of block counts, mark-resight and distance sampling to estimate population size of a mountain-dwelling ungulate. Popul. Ecol. 2015, 57, 409–419. [Google Scholar] [CrossRef]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. 2022. Available online: https://issues.qgis.org/projects/qgis/wiki/QGIS_Citation_Repository (accessed on 9 June 2022).
- Mustoni, A.; Alcione, T. Ungulati Delle Alpi: Biologia, Riconoscimento, Gestione; Nitida Immagine: Cles, Italy, 2002; ISBN 9788887439038. [Google Scholar]
- Monaco, A.; Lovari, S. Parametri Ambientali Determinanti La Dispersione e l’uso Dell’habitat Nel Camoscio Alpino (Rupicapra Rupicapra Rupicapra) nel Parco Naturale Paneveggio-Pale Di S.Martino. Relazione Conclusiva 1998–2003; Ente Parco Paneveggio Pale di San Martino: Predazzo, Italy, 2003. [Google Scholar]
- Hoinville, L.J.; Alban, L.; Drewe, J.A.; Gibbens, J.C.; Gustafson, L.; Häsler, B.; Saegerman, C.; Salman, M.; Stärk, K.D. Proposed terms and concepts for describing and evaluating animal-health surveillance systems. Prev. Vet. Med. 2013, 112, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, L.; Pedrotti, L.; Riga, F.; Toso, S. Banca Dati Ungulati: Status, Distribuzione, Consistenza, Gestione e Prelievo Venatorio Delle Popolazioni Di Ungulati in Italia. Rap-Porto 2001–2005; Istituto Superiore per la Protezione e la Ricerca Ambientale: Rome, Italy, 2009. [Google Scholar]
- Fernández-Morán, J.; Gómez, S.; Ballesteros, F.; Quirós, P.Q.; Benito, J.L.; Feliu, C.; Nieto, J.M. Epizootiology of Sarcoptic Mange in a Population of Cantabrian Chamois (Rupicapra Pyrenaica Parva) in Northwestern Spain. Vet. Parasitol. 1997, 73, 163–171. [Google Scholar] [CrossRef]
- Pioz, M.; Loison, A.; Gauthier, D.; Gibert, P.; Jullien, J.M.; Artois, M.; Gilot-Fromont, E. Diseases and Reproductive Success in a Wild Mammal: Example in the Alpine Chamois. Oecologia 2008, 155, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Serrano, E.; Colom-Cadena, A.; Gilot-Fromont, E.; Garel, M.; Cabezón, O.; Velarde, R.; Fernández-Sirera, L.; Fernández-Aguilar, X.; Rosell, R.; Lavín, S.; et al. Border Disease Virus: An Exceptional Driver of Chamois Populations Among Other Threats. Front. Microbiol. 2015, 6, 1307. [Google Scholar] [CrossRef]
- Rughetti, M.; Toïgo, C.; Von Hardenberg, A.; Rocchia, E.; Festa-Bianchet, M. Effects of an Exceptionally Snowy Winter on Chamois Survival. Acta Theriol. 2011, 56, 329–333. [Google Scholar] [CrossRef]
- Jonas, T.; Geiger, F.; Jenny, H. Mortality Pattern of the Alpine Chamois: The Influence of Snow-Meteorological Factors. Ann. Glaciol. 2008, 49, 56–62. [Google Scholar] [CrossRef]
- Gonzalez, G.; Crampe, J.-P. Mortality Patterns in a Protected Population of Isards (Rupicapra pyrenaica). Can. J. Zool. 2001, 79, 2072–2079. [Google Scholar] [CrossRef]
- Chirichella, R.; Stephens, P.A.; Mason, T.H.E.; Apollonio, M. Contrasting Effects of Climate Change on Alpine Chamois. J. Wildl. Manag. 2021, 85, 109–120. [Google Scholar] [CrossRef]
- Donini, V.; Pedrotti, L.; Ferretti, F.; Corlatti, L. Disentangling Demographic Effects of Red Deer on Chamois Population Dynamics. Ecol. Evol. 2021, 11, 8264–8280. [Google Scholar] [CrossRef] [PubMed]
- Turchetto, S.; Obber, F.; Rossi, L.; D’Amelio, S.; Cavallero, S.; Poli, A.; Parisi, F.; Lanfranchi, P.; Ferrari, N.; Dellamaria, D.; et al. Sarcoptic Mange in Wild Caprinae of the Alps: Could Pathology Help in Filling the Gaps in Knowledge? Front. Vet. Sci. 2020, 7, 193. [Google Scholar] [CrossRef] [PubMed]
- Ráez-Bravo, A.; Granados, J.E.; Serrano, E.; Dellamaria, D.; Casais, R.; Rossi, L.; Puigdemont, A.; Cano-Manuel, F.J.; Fandos, P.; Pérez, J.M.; et al. Evaluation of Three Enzyme-Linked Immunosorbent Assays for Sarcoptic Mange Diagnosis and Assessment in the Iberian Ibex, Capra Pyrenaica. Parasites Vectors 2016, 9, 558. [Google Scholar] [CrossRef] [PubMed]





| Mange Zone | Surface (ha) | Index Case | Pre-Epidemic Stock 1 | Post-Epidemic Stock 2 (Census Year) | Epidemic Wave Duration (Years 3) | Decline Rate | ||
|---|---|---|---|---|---|---|---|---|
| N Heads | Density | N Heads | Density | |||||
| PAN | 9504.737 | 2005 | 342 | 3.60 | 231 (2011) | 2.43 | 7 | 32% |
| PAL | 7579.657 | 2007 | 313 | 4.13 | 95 (2013) | 1.25 | 7 | 69% |
| SCA | 9285.647 | 2007 | 277 | 2.98 | 112 (2015) | 1.21 | 9 | 60% |
| CAU | 7154.198 | 2008 | 434 | 6.07 | 165 (2013) | 2.31 | 6 | 62% |
| ROL | 3909.907 | 2010 | 232 | 5.93 | 69 (2013) | 1.76 | 4 | 70% |
| LIT | 7434.002 | 2010 | 543 | 7.30 | 356 (2014) | 4.79 | 5 | 34% |
| STE | 8852.382 | 2011 | 639 | 7.22 | 397 (2020) | 4.48 | 10 | 38% |
| OLT | 2679.183 | 2011 | 172 | 6.42 | 133 (2014) | 4.96 | 4 | 23% |
| LSO | 6595.381 | 2012 | 288 | 4.37 | 232 (2015) | 3.52 | 4 | 19% |
| TOT | 1321.142 | 2014 | 54 | 4.09 | 35 (2014) | 2.65 | 1 | 35% |
| VFS 4 | 5282.734 | 2015 | 148 | 2.80 | 23 (2019) | 0.44 | 5 | 84% |
| VFM 4 | 3057.262 | 2016 | 173 | 5.66 | 41 (2018) | 1.34 | 3 | 76% |
| Mange Zone | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | TOTAL | Percentage of Mange Cases |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PAN | 8 (7) | 13 (13) | 26 (23) | 26 (7) | 5 (4) | 4 (2) | 0 | 1 | 0 | 0 | 0 | 0 | 3 (2) | 3 (3) | 2 (1) | 91 (62) | 68.1% |
| PAL | 1 | 4 (3) | 11 (8) | 25 (6) | 12 (8) | 10 (6) | 1 | 8 (6) | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 76 (37) | 48.6% |
| SCA | 10 | 4 (2) | 8 (2) | 17 (5) | 15 (6) | 16 (10) | 23 (10) | 7 (4) | 8 (1) | 3 (3) | 1 | 3 | 0 | 0 | 1 | 116 (43) | 37.1% |
| CAU | 6 | 1 | 9 (1) | 19 (4) | 18 (8) | 6 (3) | 10 (2) | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 74 (18) | 24.3% |
| ROL | 0 | 0 | 6 | 7 | 2 (2) | 3 (3) | 2 (2) | 2 | 0 | 0 | 1 (1) | 0 | 1 | 1 | 0 | 25 (8) | 32.0% |
| LIT | 0 | 0 | 0 | 0 | 2 (2) | 1 (1) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 (2) | 1 (1) | 6 (6) | 100% |
| STE | 0 | 0 | 0 | 0 | 0 | 1 (1) | 1 (1) | 0 | 1 (1) | 10 (10) | 10 (10) | 19 (19) | 6 (6) | 0 | 4 (4) | 52 (52) | 100% |
| OLT | 1 | 1 | 2 | 1 | 1 | 3 (3) | 8 (7) | 8 (6) | 8 (6) | 1 | 0 | 1 | 0 | 0 | 0 | 35 (22) | 62.8% |
| LSO | 2 | 0 | 0 | 0 | 0 | 0 | 1 (1) | 1 (1) | 2 (2) | 0 | 1 (1) | 1 | 1 (1) | 1 (1) | 2 | 12 (7) | 58.3% |
| TOT | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 2 | 2 (2) | 3 (2) | 0 | 2 | 0 | 1 (1) | 1 | 12 (5) | 41.6% |
| VFS | 0 | 2 | 2 | 1 | 0 | 1 | 2 | 0 | 2 | 5 (2) | 27 (19) | 38 (33) | 1 | 0 | 0 | 81 (54) | 66.6% |
| VFM | 1 | 0 | 2 | 3 | 1 | 1 | 0 | 1 | 0 | 1 | 4 (3) | 8 (8) | 3 (3) | 1 (1) | 3 (1) | 29 (16) | 55.2% |
| TOTAL | 29 (7) | 25 (18) | 66 (34) | 99 (22) | 57 (30) | 46 (29) | 48 (23) | 31 (17) | 25 (12) | 24 (17) | 45 (34) | 73 (60) | 17 (12) | 9 (8) | 15 (7) | 609 (330) | 54.1% |
| Year | Kids | Juveniles | Female Adults | Male Adults | Undetermined | Tot | Found Carcasses (Mangy- Not Mangy-Undetermined) |
|---|---|---|---|---|---|---|---|
| 2008 | 37 (1) | 21 | 60 (10) | 21 (4) | 15 (15) | 154 (30) | 34 (20-5-9) |
| 2009 | 24 | 5 | 34 | 15 | 0 | 78 | 34 (11-13-10) |
| 2010 | 10 | 10 | 15 | 9 | 0 | 44 | 5 (3-2-0) |
| 2011 | 8 | 4 | 11 | 12 | 0 | 35 | - |
| 2012 | 22 | 10 | 24 | 8 | 0 | 64 | - |
| 2013 | 13 | 10 | 18 | 15 | 0 | 56 | - |
| Mange Zone | Pre-Epidemic Stock | Post-Epidemic Stock | Epidemic Wave Duration | Number of Carcasses Found | Estimated Minimum Number of Dead Animals | Estimated Number of Animals Not Detected by Surveillance | Percentage of Dead Animals Not Detected by Surveillance |
|---|---|---|---|---|---|---|---|
| N Heads | N Heads | Years | |||||
| PAN | 342 | 231 | 7 | 82 | 111 | 29 | 26.1% |
| PAL | 313 | 95 | 3 | 40 | 149 | 109 | 73.2% |
| SCA | 277 | 112 | 9 | 101 | 165 | 64 | 38.8% |
| CAU | 434 | 165 | 6 | 63 | 269 | 206 | 76.6% |
| ROL | 232 | 69 | 4 | 9 | 163 | 154 | 94.5% |
| LIT | 543 | 356 | 5 | 3 | 187 | 184 | 98.4% |
| STE | 639 | 397 | 7 | 42 | 177 | 135 | 76.3% |
| OLT | 172 | 133 | 4 | 27 | 39 | 12 | 30.8% |
| LSO | 288 | 232 | 4 | 4 | 56 | 52 | 92.9% |
| TOT | 54 | 35 | 1 | 2 | 19 | 17 | 89.5% |
| VFS | 148 | 23 | 5 | 71 | 125 | 54 | 43.2% |
| VFM | 173 | 41 | 3 | 15 | 132 | 117 | 88.6% |
| Ordinary surveillance (All mange zones) | 3964 | 1889 | 459 | 2075 | 1616 | 77.9% | |
| Enhanced surveillance (CA) | 154 | 35 | 4 | 74 | 119 | 45 | 37.8% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obber, F.; Celva, R.; Libanora, M.; Da Rold, G.; Dellamaria, D.; Partel, P.; Ferraro, E.; Calabrese, M.S.; Morpurgo, L.; Pisano, S.R.R.; et al. Description of a Sarcoptic Mange Outbreak in Alpine Chamois Using an Enhanced Surveillance Approach. Animals 2022, 12, 2077. https://doi.org/10.3390/ani12162077
Obber F, Celva R, Libanora M, Da Rold G, Dellamaria D, Partel P, Ferraro E, Calabrese MS, Morpurgo L, Pisano SRR, et al. Description of a Sarcoptic Mange Outbreak in Alpine Chamois Using an Enhanced Surveillance Approach. Animals. 2022; 12(16):2077. https://doi.org/10.3390/ani12162077
Chicago/Turabian StyleObber, Federica, Roberto Celva, Martina Libanora, Graziana Da Rold, Debora Dellamaria, Piergiovanni Partel, Enrico Ferraro, Maria Santa Calabrese, Lia Morpurgo, Simone Roberto Rolando Pisano, and et al. 2022. "Description of a Sarcoptic Mange Outbreak in Alpine Chamois Using an Enhanced Surveillance Approach" Animals 12, no. 16: 2077. https://doi.org/10.3390/ani12162077
APA StyleObber, F., Celva, R., Libanora, M., Da Rold, G., Dellamaria, D., Partel, P., Ferraro, E., Calabrese, M. S., Morpurgo, L., Pisano, S. R. R., Citterio, C. V., & Cassini, R. (2022). Description of a Sarcoptic Mange Outbreak in Alpine Chamois Using an Enhanced Surveillance Approach. Animals, 12(16), 2077. https://doi.org/10.3390/ani12162077








