Dietary Fish Meal Replacement with Hermetia illucens and Tenebrio molitor Larval Meals Improves the Growth Performance and Nutriphysiological Status of Ide (Leuciscus idus) Juveniles
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Meals and Diet Preparation
2.2. Animal Husbandry and Growth Trial
2.3. Sampling
2.4. Somatic Indices
2.5. Histological Analyses
2.6. Statistical Analyses
2.7. Ethics Statement
3. Results
3.1. Growth Performance and Feed Utilization
3.2. Condition and Somatic Indices
3.3. Chemical Composition of Fish
3.4. Histopathology of the Hepatopancreas and Histomorphology of Intestine
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Darwall, W.R.T.; Freyhof, J. Lost fishes, who is counting? The extent of the threat to freshwater fish biodiversity. In Conservation of Freshwater Fishes; Closs, G.P., Krkosek, M., Olden, J.D., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 1–36. ISBN 9781107040113. [Google Scholar]
- Cooke, S.J.; Schramm, H.L. Catch-and-release science and its application to conservation and management of recreational fisheries. Fish. Manag. Ecol. 2007, 14, 73–79. [Google Scholar] [CrossRef]
- Grift, R.E.; Buijse, A.D.; Van Densen, W.L.T.; Machiels, M.A.M.; Kranenbarg, J.; Klein Breteler, J.G.P.; Backx, J.J.G.M. Suitable habitats for 0-group fish in rehabilitated floodplains along the lower River Rhine. River Res. Appl. 2003, 19, 353–374. [Google Scholar] [CrossRef]
- Müller, K.; Berg, E. Spring migration of some anadromous freshwater fish species in the northern Bothnian Sea. Hydrobiologia 1982, 96, 161–168. [Google Scholar] [CrossRef]
- Rohtla, M.; Vilizzi, L.; Kováč, V.; Almeida, D.; Brewster, B.; Britton, J.R.; Głowacki, Ł.; Godard, M.J.; Kirk, R.; Nienhuis, S.; et al. Review and meta-analysis of the environmental biology and potential invasiveness of a poorly-studied cyprinid, the ide Leuciscus idus. Rev. Fish. Sci. Aquac. 2021, 29, 512–548. [Google Scholar] [CrossRef]
- Leung, P.; Lee, C.S.; O’Bryen, P.J. Species and System Selection for Sustainable Aquaculture; Wiley-Blackwell: Hoboken, NJ, USA, 2007; ISBN 9780813826912. [Google Scholar]
- Rahman, M.M. Role of common carp (Cyprinus carpio) in aquaculture production systems. Front. Life Sci. 2015, 8, 399–410. [Google Scholar] [CrossRef] [Green Version]
- Harzevili, A.S.; Vught, I.; Auwerx, J.; De Charleroy, D. Larval rearing of ide (Leuciscus idus (L.)) using decapsulated artemia. Arch. Polish Fish. 2012, 20, 219–222. [Google Scholar] [CrossRef]
- Kujawa, R.; Glińska-Lewczuk, K. The impact of regulation and hydrotechnical works in river channels on the rheophilic cyprinidae fish. Contemp. Probl. Manag. Environ. Prot. 2011, 7, 233–248. [Google Scholar]
- Śliwiński, J.; Ostaszewska, T.; Kamaszewski, M. Akwakultura jako narzȩdzie poprawy statusu karpiowa tych ryb reofilnych w Polsce-stan i perspektywy badań. Sci. Rev. Eng. Environ. Sci. 2016, 25, 239–248. (In Polish) [Google Scholar]
- Kamiński, R.; Wolnicki, J.; Sikorska, J.; Garcia, V. Effects of temperature on growth, survival and body composition in larvae of barbel, Barbus barbus (L.). Aquac. Int. 2013, 21, 829–841. [Google Scholar] [CrossRef]
- Kucharczyk, D.; Targońska, K.; Żarski, D.; Kujawa, R.; Mamcarz, A. A review of the reproduction biotechnology for fish from the genus leuciscus. Arch. Polish Fish. 2008, 16, 319–340. [Google Scholar] [CrossRef]
- Kupren, K.; Mamcarz, A.; Kucharczyk, D. Effects of temperature on survival, deformations rate and selected parameters of newly hatched larvae of three rheophilic cyprinids (Genus Leuciscus). Polish J. Nat. Sci. 2010, 25, 299–312. [Google Scholar] [CrossRef]
- Czerniawski, R.; Pilecka-Rapacz, M.; Domagała, J. Growth and survival of brown trout fry (Salmo trutta m. fario L.) in the wild, reared in the hatchery on different feed. Electron. J. Polish Agric. Univ. 2010, 13, art 04. [Google Scholar]
- Zakęś, Z.; Rożyński, M. Charakterystyka produkcji polskiej akwakultury zachowawczej w latach 2010-2019. In Akwakultura Jako Narzędzie Ochrony Ichtiofauny; Zakęś, Z., Demska-Zakęś, K., Eds.; Wydawnictwo Instytutu Rybactwa Śródlądowego: Olsztyn, Poland, 2021; pp. 11–25. ISBN 978-83-66805-02-6. (In Polish) [Google Scholar]
- Salin, K.R.; Arome Ataguba, G. Aquaculture and the Environment: Towards Sustainability. In Sustainable Aquaculture; Springer: Cham, Switzerland, 2018; pp. 1–62. [Google Scholar] [CrossRef]
- Nogales-Mérida, S.; Gobbi, P.; Józefiak, D.; Mazurkiewicz, J.; Dudek, K.; Rawski, M.; Kierończyk, B.; Józefiak, A. Insect meals in fish nutrition. Rev. Aquac. 2019, 11, 1080–1103. [Google Scholar] [CrossRef]
- Liland, N.S.; Araujo, P.; Xu, X.X.; Lock, E.J.; Radhakrishnan, G.; Prabhu, A.J.P.; Belghit, I. A meta-analysis on the nutritional value of insects in aquafeeds. J. Insects Food Feed. 2021, 7, 743–759. [Google Scholar] [CrossRef]
- Gebremichael, A.; Hancz, C.; Kucska, B. Effect of total or partial replacing of fishmeal with black solider fly (Hermetia illucens) meal on growth performance and body condition indices of common carp (Cyprinus carpio). AACL Bioflux 2021, 14, 2280–2286. [Google Scholar]
- Li, S.; Ji, H.; Zhang, B.; Zhou, J.; Yu, H. Defatted black soldier fly (Hermetia illucens) larvae meal in diets for juvenile Jian carp (Cyprinus carpio var. Jian): Growth performance, antioxidant enzyme activities, digestive enzyme activities, intestine and hepatopancreas histological structure. Aquaculture 2017, 477, 62–70. [Google Scholar] [CrossRef]
- Zhou, J.S.; Liu, S.S.; Ji, H.; Yu, H.B. Effect of replacing dietary fish meal with black soldier fly larvae meal on growth and fatty acid composition of Jian carp (Cyprinus carpio var. Jian). Aquac. Nutr. 2018, 24, 424–433. [Google Scholar] [CrossRef]
- Lu, R.; Chen, Y.; Yu, W.; Lin, M.; Yang, G.; Qin, C.; Meng, X.; Zhang, Y.; Ji, H.; Nie, G. Defatted black soldier fly (Hermetia illucens) larvae meal can replace soybean meal in juvenile grass carp (Ctenopharyngodon idellus) diets. Aquac. Rep. 2020, 18, 100520. [Google Scholar] [CrossRef]
- Lähteenmäki-Uutela, A.; Grmelová, N.; Hénault-Ethier, L.; Deschamps, M.H.; Vandenberg, G.W.; Zhao, A.; Zhang, Y.; Yang, B.; Nemane, V. Insects as food and feed: Laws of the European union, United States, Canada, Mexico, Australia, and China. Eur. Food Feed Law Rev. 2017, 12, 22–36. [Google Scholar]
- Rawski, M.; Mazurkiewicz, J.; Kierończyk, B.; Józefiak, D. Black soldier fly full-fat larvae meal as an alternative to fish meal and fish oil in siberian sturgeon nutrition: The effects on physical properties of the feed, animal growth performance, and feed acceptance and utilization. Animals 2020, 10, 2119. [Google Scholar] [CrossRef]
- Gasco, L.; Acuti, G.; Bani, P.; Dalle Zotte, A.; Danieli, P.P.; De Angelis, A.; Fortina, R.; Marino, R.; Parisi, G.; Piccolo, G.; et al. Insect and fish by-products as sustainable alternatives to conventional animal proteins in animal nutrition. Ital. J. Anim. Sci. 2020, 19, 360–372. [Google Scholar] [CrossRef] [Green Version]
- Gasco, L.; Józefiak, A.; Henry, M. Beyond the protein concept: Health aspects of using edible insects on animals. J. Insects as Food Feed 2021, 7, 715–741. [Google Scholar] [CrossRef]
- Józefiak, A.; Engberg, R.M. Insect proteins as a potential source of antimicrobial peptides in livestock production. A review. J. Anim. Feed Sci. 2017, 26, 87–99. [Google Scholar] [CrossRef]
- Finke, M.D. Estimate of chitin in raw whole insects. Zoo Biol. 2007, 26, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Gasco, L.; Gai, F.; Genovese, L.; Ragonese, S.; Bottari, T.; Caruso, G. Fishmeal alternative protein sources for aquaculture feeds. In Feeds for the Aquaculture Sector: Current Situation and Alternative Sources; Gasco, L., Gai, F., Maricchiolo, G., Genovese, L., Ragonese, S., Bottari, T., Caruso, G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–28. ISBN 978-3-319-77941-6. [Google Scholar]
- Janssen, R.H.; Vincken, J.P.; Van Den Broek, L.A.M.; Fogliano, V.; Lakemond, C.M.M. Nitrogen-to-protein conversion factors for three edible insects: Tenebrio molitor, Alphitobius diaperinus, and Hermetia illucens. J. Agric. Food Chem. 2017, 65, 2275–2278. [Google Scholar] [CrossRef]
- Ren, M.; Ji, K.; Liang, H.; Ge, X.; Mi, H. Dietary protein requirement of juvenile ide, Leuciscus idus in relation to growth performance, whole-body composition and plasma parameters. Isr. J. Aquac.-Bamidgeh 2017, 69, 20853. [Google Scholar] [CrossRef]
- Council, N.R. Nutrient Requirements of Fish and Shrimp; National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-47322-4. [Google Scholar]
- Takeuchi, T.; Satoh, S.; Kiron, V. Common carp, Cyprinus carpio. In Nutrient Requirements and Feeding of Finfish for Aquaculture; Webster, C.D., Lim, C., Eds.; CABI Publishing: New York, NY, USA, 2002; pp. 245–261. ISBN 0-85199-519-5. [Google Scholar]
- Kaushik, S.J.; Schrama, J.W. Bioenergetics. In Fish Nutrition; Hardy, R.W., Kaushik, S.J., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 17–48. ISBN 978-0-12-823159-3. [Google Scholar]
- Piccolo, G.; Iaconisi, V.; Marono, S.; Gasco, L.; Loponte, R.; Nizza, S.; Bovera, F.; Parisi, G. Effect of Tenebrio molitor larvae meal on growth performance, in vivo nutrients digestibility, somatic and marketable indexes of gilthead sea bream (Sparus aurata). Anim. Feed Sci. Technol. 2017, 226, 12–20. [Google Scholar] [CrossRef]
- Sakamoto, K.; Hirose, H.; Onizuka, A.; Hayashi, M.; Futamura, N.; Kawamura, Y.; Ezaki, T. Quantitative study of changes in intestinal morphology and mucus gel on total parenteral nutrition in rats. J. Surg. Res. 2000, 94, 99–106. [Google Scholar] [CrossRef]
- Peebua, P.; Kruatrachue, M.; Pokethitiyook, P.; Kosiyachinda, P. Histological effects of contaminated sediments in Mae Klong river tributaries, Thailand, on Nile tilapia, Oreochromis niloticus. ScienceAsia 2006, 32, 143–150. [Google Scholar] [CrossRef]
- Elia, A.C.; Capucchio, M.T.; Caldaroni, B.; Magara, G.; Dörr, A.J.M.; Biasato, I.; Biasibetti, E.; Righetti, M.; Pastorino, P.; Prearo, M.; et al. Influence of Hermetia illucens meal dietary inclusion on the histological traits, gut mucin composition and the oxidative stress biomarkers in rainbow trout (Oncorhynchus mykiss). Aquaculture 2018, 496, 50–57. [Google Scholar] [CrossRef]
- Takeuchi, T.; Watanabe, T.; Ogino, C. Optimum ratio of dietary energy to protein for carp. Nippon Suisan Gakkaishi 1979, 45, 983–987. [Google Scholar] [CrossRef]
- Ogino, C.; Chiou, J.Y.; Takeuchi, T. Protein nutrition in fish—Effects of dietary energy sources on the utilization of proteins by rainbow trout and carp. Nippon Suisan Gakkaishi 1976, 42, 213–218. [Google Scholar] [CrossRef]
- Chakraborty, S.C.; Ross, L.G.; Ross, B. Specific dynamic action and feeding metabolism in common carp, Cyprinus carpio L. Comp. Biochem. Physiol. Part A Physiol. 1992, 103, 809–815. [Google Scholar] [CrossRef]
- Kaushik, S.J. Nutrient requirements, supply and utilization in the context of carp culture. Aquaculture 1995, 129, 225–241. [Google Scholar] [CrossRef]
- Takeuchi, T.; Watanabe, T.; Ogino, C. Availability of carbohydrate and lipid as dietary energy sources for carp. Nippon Suisan Gakkaishi 1979, 45, 977–982. [Google Scholar] [CrossRef]
- Takeuchi, T.; Watanabe, T. Requirement of carp for essential fatty acids. Nippon Suisan Gakkaishi 1977, 43, 541–551. [Google Scholar] [CrossRef]
- Murai, T.; Akiyama, T.; Nose, T. Effects of glucose chain length of various carbohydrates and frequency of feeding on their utilization by fingerling carp. Nippon Suisan Gakkaishi 1983, 49, 1607–1611. [Google Scholar] [CrossRef]
- Takeuchi, T.; Jeong, K.S.; Watanabe, T. Availability of extruded carbohydrate ingredients to rainbow trout Oncorhynchus mykiss and carp Cyprinus carpio. Nippon Suisan Gakkaishi 1990, 56, 1839–1845. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Hemre, G.I.; Mommsen, T.P. Carbohydrates in fish nutrition: Digestion and absorption in postlarval stages. Aquac. Nutr. 2005, 11, 103–122. [Google Scholar] [CrossRef]
- Stone, D.A.J. Dietary carbohydrate utilization by fish. Rev. Fish. Sci. 2003, 11, 337–369. [Google Scholar] [CrossRef]
- Wolnicki, J.; Górny, W. Controlled rearing of ide (Leuciscus idus L.) larvae using live food and dry feed. Aquaculture 1995, 129, 255–256. [Google Scholar] [CrossRef]
- Turkowski, K.; Kucharczyk, D.; Kupren, K.; Hakuĉ-Bńałowska, A.; Targońska, K.; Zarski, D.; Kwiatkowski, M. Economic aspects of the experimental rearing of asp, Aspius aspius (L.), ide, Leuciscus idus (L.), and dace, Leuciscus leuciscus (L.), under controlled conditions. Arch. Polish Fish. 2008, 16, 397–411. [Google Scholar] [CrossRef]
- Austin, B.; Allen, D.A. Microbiology of laboratory-hatched brine shrimp (Artemia). Aquaculture 1982, 26, 369–383. [Google Scholar] [CrossRef]
- Chen, J.-C.; Lru, P.-C. Accumulation of heavy metals in the nauplii of Artemia salina. J. World Aquac. Soc. 1987, 18, 84–93. [Google Scholar] [CrossRef]
- Zulkifli, S.Z.; Aziz, F.Z.A.; Ajis, S.Z.M.; Ismail, A. Nauplii of brine shrimp (Artemia salina) as a potential toxicity testing organism for heavy metals contamination. In From Sources to Solution; Springer: Singapore, 2014; pp. 233–237. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Vidotto-Magnoni, A.P.; Carvalho, E.D. Aquatic insects as the main food resource of fish the community in aneotropical reservoir. Neotrop. Ichthyol. 2009, 7, 701–708. [Google Scholar] [CrossRef] [Green Version]
- Kowalska, J.; Rawski, M.; Homska, N.; Mikołajczak, Z.; Kierończyk, B.; Świątkiewicz, S.; Wachowiak, R.; Hetmańczyk, K.; Mazurkiewicz, J. The first insight into full-fat superworm (Zophobas morio) meal in guppy (Poecilia reticulata) diets: A study on multiple-choice feeding preferences and growth performance. Ann. Anim. Sci. 2021, 22, 371–384. [Google Scholar] [CrossRef]
- Rawski, M.; Józefiak, D.; Nogales-Mérida, S.; Józefiak, A.; Kierończyk, B.; Benzertiha, A.; Talibov, S.; Dudek, K.; Gobbi, P.; Noak, M.; et al. The preliminary study on live insects acceptance in common carp (Cyprinus carpio). In Proceedings of the XVth Conference of Young Researchers Physiology and Biochemistry in Animal Nutrition, Warsaw, Poland, 26–28 September 2018. [Google Scholar]
- Abdelghany, A.E.; Ahmad, M.H. Effects of feeding rates on growth and production of Nile tilapia, common carp and silver carp polycultured in fertilized ponds. Aquac. Res. 2002, 33, 415–423. [Google Scholar] [CrossRef]
- Goolish, E.M.; Adelman, I.R. Effects of ration size and temperature on the growth of juvenile common carp (Cyprinus carpio L.). Aquaculture 1984, 36, 27–35. [Google Scholar] [CrossRef]
- Yousefi, M.; Hoseini, S.M.; Vatnikov, Y.A.; Kulikov, E.V.; Drukovsky, S.G. Rosemary leaf powder improved growth performance, immune and antioxidant parameters, and crowding stress responses in common carp (Cyprinus carpio) fingerlings. Aquaculture 2019, 505, 473–480. [Google Scholar] [CrossRef]
- Naylor, R.L.; Hardy, R.W.; Buschmann, A.H.; Bush, S.R.; Cao, L.; Klinger, D.H.; Little, D.C.; Lubchenco, J.; Shumway, S.E.; Troell, M. A 20-year retrospective review of global aquaculture. Nature 2021, 591, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Rawski, M.; Mazurkiewicz, J.; Kierończyk, B.; Józefiak, D. Black soldier fly full-fat larvae meal is more profitable than fish meal and fish oil in siberian sturgeon farming: The effects on aquaculture sustainability, economy and fish git development. Animals 2021, 11, 604. [Google Scholar] [CrossRef] [PubMed]
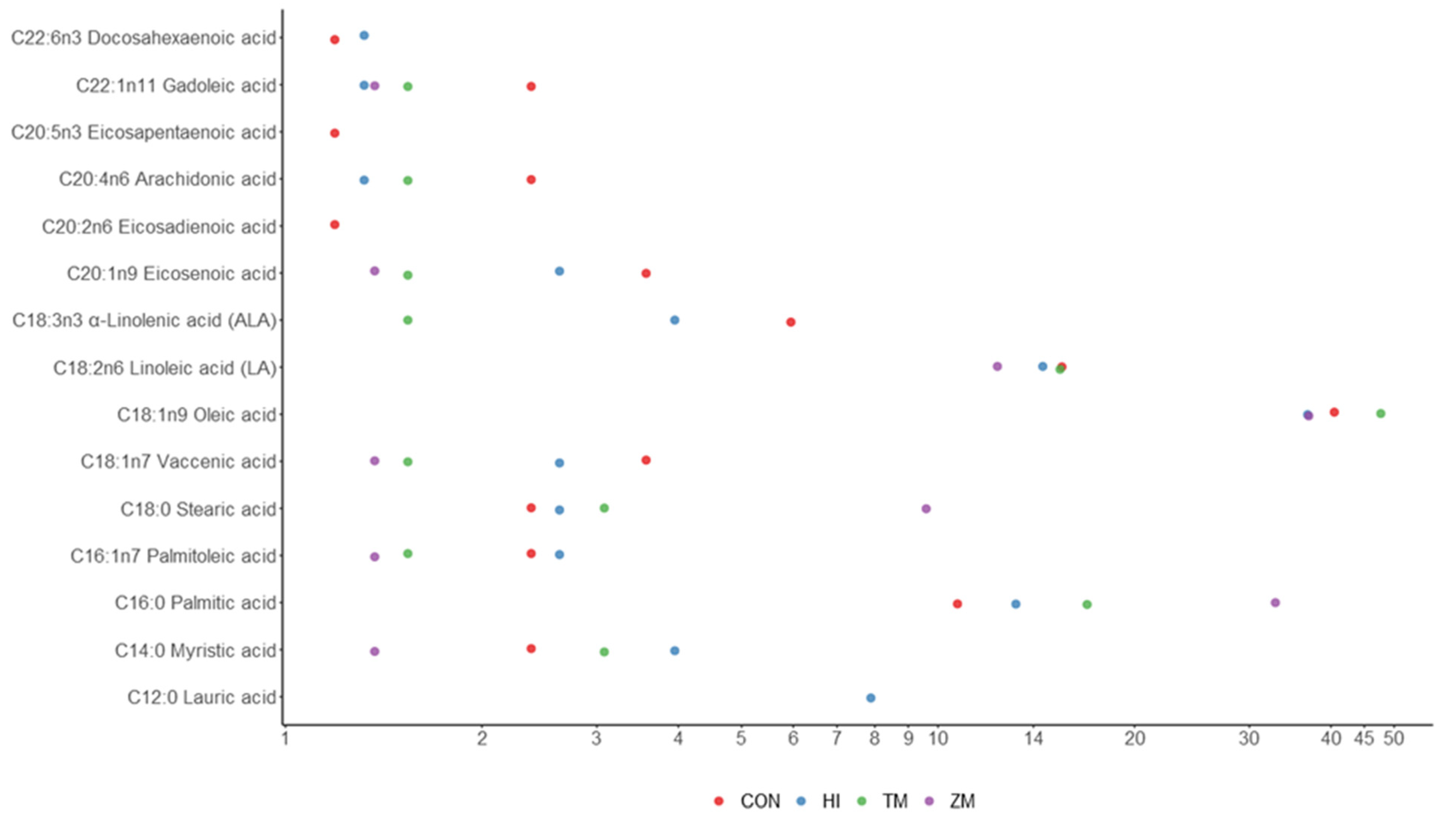
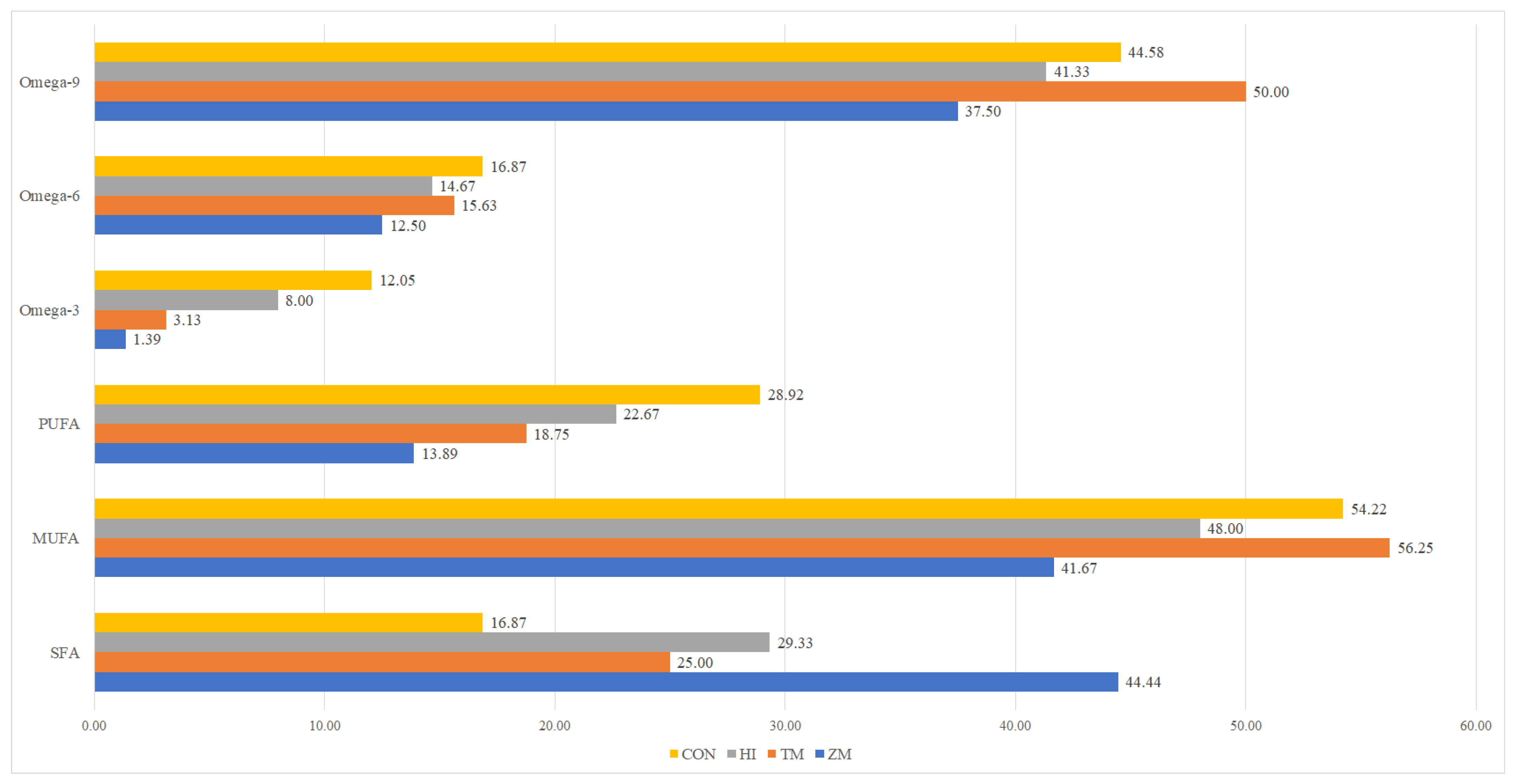
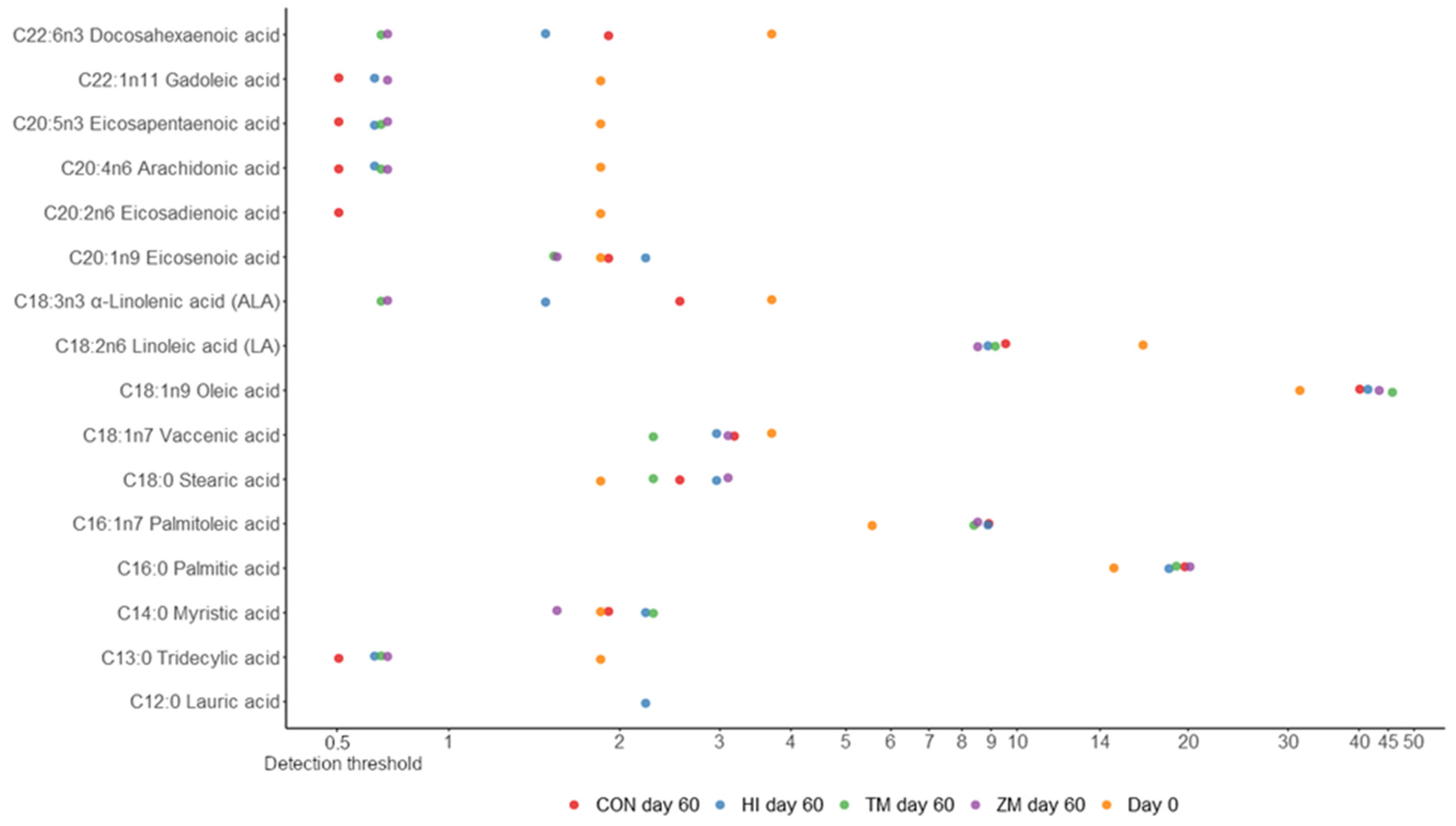
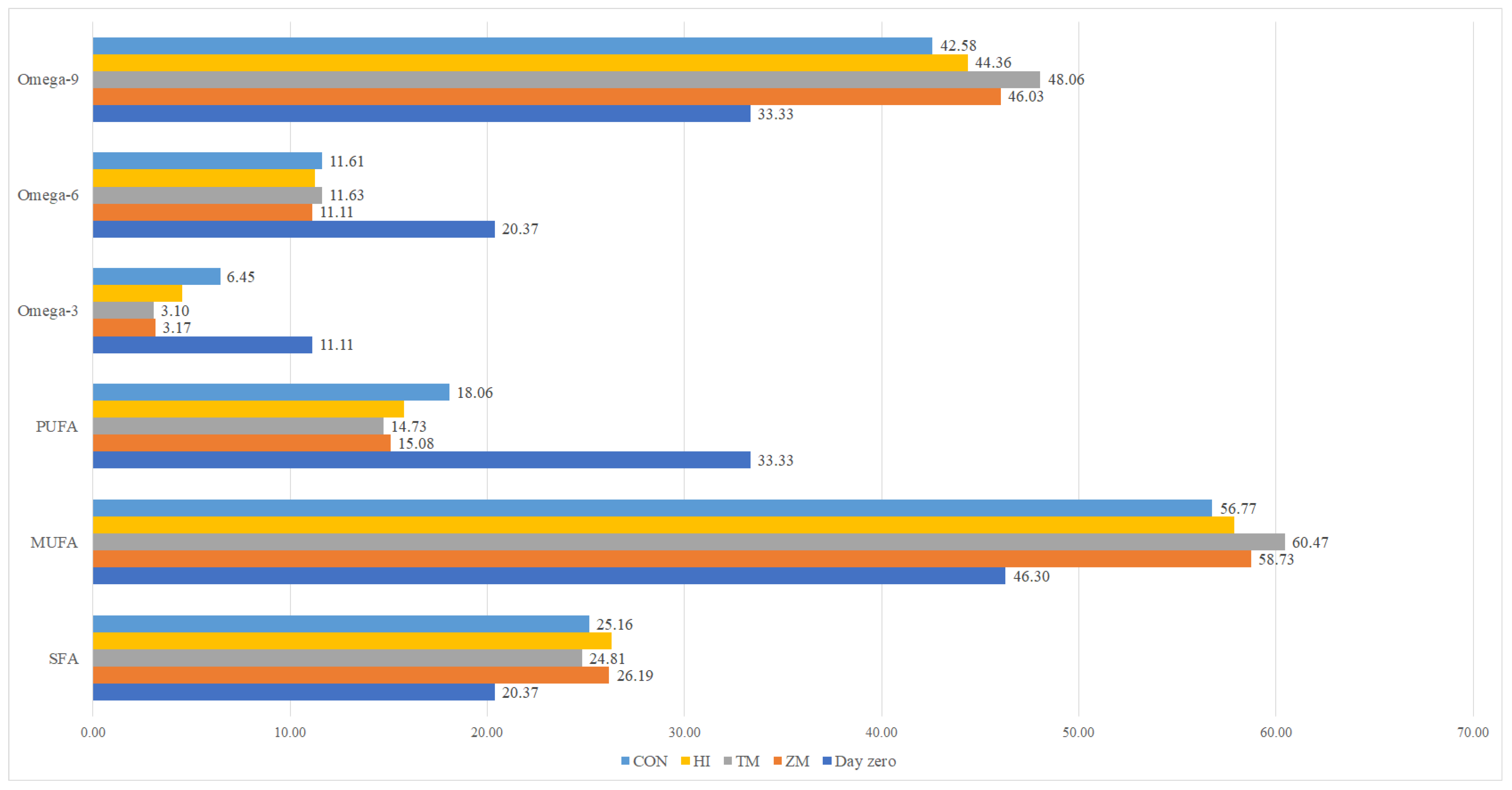
| Nutrient (% Fresh Matter) | Fish Meal | Hermetia illucens Meal | Tenebrio molitor Meal | Zophobas morio Meal |
|---|---|---|---|---|
| Dry matter | 91.10 | 94.97 | 95.20 | 96.90 |
| Crude fiber | 3.40 | 5.70 | 7.30 | 5.50 |
| Ash | 8.82 | 8.08 | 4.10 | 2.83 |
| Crude fat | 9.00 | 12.06 | 26.60 | 37.40 |
| Nitrogen-free extract | 16.58 | 16.29 | 2.5 | 2.77 |
| Crude protein (N × 6.25) | 53.30 | 52.84 | 54.70 | 48.40 |
| Kp | 6.25 | 5.12 | 4.91 | 5.77 |
| The crude protein, according to Kp | 53.30 | 45.58 | 44.81 | 46.13 |
| Amino acids (g 100 g protein−1 N × 6.25) | ||||
| Aspartic acid | 4.79 | 1.60 | 2.77 | 3.50 |
| Glutamic acid | 6.25 | 3.44 | 5.16 | 6.49 |
| Serine | 1.82 | 1.32 | 2.28 | 2.24 |
| Glycine | 2.04 | 1.75 | 2.77 | 2.58 |
| Histidine | 0.89 | 0.74 | 1.40 | 1.50 |
| Arginine | 2.66 | 1.32 | 2.37 | 2.45 |
| Threonine | 1.44 | 1.19 | 1.92 | 1.97 |
| Alanine | 1.87 | 3.01 | 4.41 | 3.88 |
| Proline | 2.03 | 1.98 | 3.32 | 2.94 |
| Tyrosine | 1.00 | 1.58 | 2.81 | 3.60 |
| Valine | 1.92 | 1.70 | 2.87 | 2.90 |
| Methionine | 0.11 | 0.49 | 0.65 | 0.60 |
| Cystine | 0.34 | 0.22 | 0.35 | 0.32 |
| Isoleucine | 1.76 | 1.25 | 2.08 | 2.15 |
| Leucine | 2.94 | 1.94 | 3.52 | 3.49 |
| Phenylalanine | 1.95 | 1.05 | 1.62 | 1.81 |
| Lysine | 2.25 | 1.36 | 2.36 | 2.28 |
| Ingredient (g kg−1) | Diets | |||
|---|---|---|---|---|
| CON | HI | TM | ZM | |
| Fish meal | 300 | 150 | 150 | 150 |
| Red blood cells | 90 | 90 | 90 | 90 |
| Insect meal | 0 | 200 | 200 | 200 |
| Soy protein isolate | 80 | 80 | 80 | 80 |
| Wheat gluten | 100 | 100 | 100 | 100 |
| Wheat meal | 125 | 125 | 125 | 125 |
| Corn starch | 196 | 155 | 186 | 201 |
| Fish oil | 61 | 50 | 15 | 0 |
| Soybean lecithin | 10 | 10 | 10 | 10 |
| Premix 1 | 15 | 15 | 15 | 15 |
| Vitamin premix 2 | 1 | 1 | 1 | 1 |
| Choline chloride | 2 | 2 | 2 | 2 |
| Fodder chalk | 20 | 22 | 26 | 26 |
| Vitamin C 3 | 0.5 | 0.5 | 0.5 | 0.5 |
| Nutrient (% Fresh Matter) | Diets | |||
|---|---|---|---|---|
| CON | HI | TM | ZM | |
| Dry matter | 91.2 | 91.0 | 91.8 | 91.6 |
| Crude protein | 42.3 | 44.0 | 44.7 | 43.5 |
| Amino acids (g 100 g protein−1) | ||||
| Aspartic acid | 6.52 | 6.25 | 6.29 | 6.16 |
| Glutamic acid | 16.24 | 15.48 | 15.55 | 15.66 |
| Serine | 4.23 | 4.32 | 4.41 | 4.23 |
| Glycine | 3.57 | 3.82 | 3.94 | 3.72 |
| Histidine | 2.65 | 2.75 | 2.77 | 2.71 |
| Arginine | 4.14 | 4.07 | 4.16 | 4.02 |
| Threonine | 2.86 | 3.05 | 3.06 | 2.99 |
| Alanine | 4.21 | 4.98 | 5.23 | 4.83 |
| Proline | 5.60 | 5.98 | 6.20 | 5.93 |
| Tyrosine | 2.22 | 2.86 | 2.75 | 2.99 |
| Valine | 3.90 | 4.14 | 4.18 | 4.09 |
| Methionine | 1.42 | 1.59 | 1.57 | 1.54 |
| Cystine | 0.66 | 0.64 | 0.65 | 0.60 |
| Isoleucine | 2.20 | 2.32 | 2.35 | 2.32 |
| Leucine | 6.97 | 7.00 | 7.23 | 7.10 |
| Phenylalanine | 4.44 | 4.41 | 4.32 | 4.34 |
| Lysine | 4.99 | 4.95 | 5.17 | 5.06 |
| Crude fat | 8.3 | 7.5 | 6.4 | 7.2 |
| Ash | 5.57 | 5.99 | 5.73 | 5.38 |
| Crude fiber | 1.4 | 2.4 | 2.1 | 1.6 |
| Nitrogen-free extract | 42.33 | 40.01 | 40.97 | 42.22 |
| Calcium | 1.24 | 1.20 | 1.28 | 1.27 |
| Phosphorus | 0.51 | 0.59 | 0.57 | 0.49 |
| Gross energy (MJ kg−1) | 18.77 | 18.57 | 18.48 | 19.05 |
| Energy/protein ratio (kJ g−1 protein) | 44.47 | 43.90 | 44.43 | 44.91 |
| Parameter | Diets | |||||
|---|---|---|---|---|---|---|
| CON | HI | TM | ZM | SEM | p Value | |
| Final individual body weight (g−1) | 6.94 b ± 0.37 | 7.88 a ± 0.38 | 7.99 a ± 0.25 | 6.59 b ± 0.50 | 0.1582 | 0.0000413 |
| Mean individual body weight gain (g−1) | 2.53 b ± 0.35 | 3.41 a ± 0.27 | 3.60 a ± 0.53 | 2.14 b ± 0.56 | 0.1658 | 0.000196 |
| Specific growth rate (SGR, % day−1) | 0.76 b ± 0.11 | 0.94 a ± 0.05 | 1.00 a ± 0.18 | 0.65 b ± 0.16 | 0.0423 | 0.00253 |
| Percent weight gain (PWG, %) | 57.9 b ± 9.8 | 76.4 a ± 5.1 | 83.3 a ± 19.0 | 48.5 b ± 14.4 | 4.1838 | 0.00238 |
| Feed conversion ratio (FCR) | 2.54 ab ± 0.39 | 2.02 b ± 0.14 | 2.01 b ± 0.35 | 3.08 a ± 0.87 | 0.1465 | 0.0127 |
| Protein efficiency ratio (PER) | 0.95 b ± 0.12 | 1.18 a ± 0.08 | 1.22 a ± 0.20 | 0.81 b ± 0.21 | 0.0512 | 0.00295 |
| Parameter | Diets | |||||
|---|---|---|---|---|---|---|
| CON | HI | TM | ZM | SEM | p Value | |
| CF | 1.58 a ± 0.09 | 1.55 ab ± 0.08 | 1.57 a ± 0.08 | 1.51 b ± 0.11 | 0.0075 | 0.0043 |
| VSI [%] | 11.65 a ± 1.32 | 10.96 b ± 1.53 | 11.08 ab ± 1.32 | 10.15 c ± 1.48 | 0.1186 | <0.0001 |
| RGL [%] | 104.02 ± 8.25 | 103.11 ± 6.27 | 102.89 ± 7.71 | 101.69 ± 10.51 | 0.6552 | 0.6600 |
| % Fresh Matter | Day Zero | Groups on Day 60 | |||
|---|---|---|---|---|---|
| CON | HI | TM | ZM | ||
| Dry matter | 24.5 | 35.2 | 33.2 | 33.6 | 33.8 |
| Crude protein | 16.1 | 17.0 | 17.1 | 16.6 | 17.2 |
| Crude fat | 5.4 | 15.5 | 13.3 | 12.9 | 12.6 |
| Ash | 3.53 | 3.13 | 3.24 | 3.13 | 3.48 |
| Calcium | 1.06 | 0.74 | 0.96 | 0.87 | 1.06 |
| Phosphorus | 0.58 | 0.51 | 0.54 | 0.54 | 0.69 |
| Parameters (0–4 Scale) | Groups | |||||
|---|---|---|---|---|---|---|
| CON | HI | TM | ZM | SEM | p Value | |
| Necrosis | 0.27 ± 0.59 | 0.27 ± 0.46 | 0.47 ± 0.64 | 0.13 ± 0.35 | 0.0676 | 0.3758 |
| Hepatocyte vacuolization | 1.07 ± 0.96 | 1.93 ± 0.88 | 1.93 ± 1.03 | 1.47 ± 1.06 | 0.1328 | 0.0489 |
| Congestion | 1.13 ± 1.06 | 0.73 ± 0.80 | 0.53 ± 0.64 | 0.6 ± 0.51 | 0.1026 | 0.3529 |
| Parenchymal eclipse | 0.27 ± 0.46 | 0.4 ± 0.63 | 0.27 ± 0.70 | 0.07 ± 0.26 | 0.0698 | 0.2948 |
| Fat vacuolization | 0.8 a ± 1.0 | 1.6 ab ± 1.12 | 2.0 b ± 0.93 | 1.0a ± 1.13 | 0.1462 | 0.0140 |
| Fibrosis | 0.0 a ± 0.0 | 0.07 ab ± 0.26 | 0.27 b ± 0.46 | 0.0ab ± 0.0 | 0.0360 | 0.0264 |
| Parameters | Groups | |||||
|---|---|---|---|---|---|---|
| CON | HI | TM | ZM | SEM | p Value | |
| Villi height (µm) | 286.64 ab ± 74.82 | 283.77 ab ± 54.06 | 308.36 a ± 38.42 | 239.41 b ± 51.68 | 7.7931 | 0.0052 |
| Villi width (µm) | 87.31 ± 10.65 | 91.06 ± 12.61 | 92.02 ± 10.30 | 84.88 ± 9.41 | 1.4095 | 0.2470 |
| Villi surface area (µm2) | 79798 ab ± 29578 | 82202 ab ± 24301 | 89715 a ± 18839 | 64205 b ± 17622 | 3144.4 | 0.0117 |
| Muscular layer thickness (µm) | 36.06 ± 8.69 | 34.27 ± 8.35 | 34.54 ± 5.03 | 33.91 ± 5.35 | 0.8940 | 0.8470 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Homska, N.; Kowalska, J.; Bogucka, J.; Ziółkowska, E.; Rawski, M.; Kierończyk, B.; Mazurkiewicz, J. Dietary Fish Meal Replacement with Hermetia illucens and Tenebrio molitor Larval Meals Improves the Growth Performance and Nutriphysiological Status of Ide (Leuciscus idus) Juveniles. Animals 2022, 12, 1227. https://doi.org/10.3390/ani12101227
Homska N, Kowalska J, Bogucka J, Ziółkowska E, Rawski M, Kierończyk B, Mazurkiewicz J. Dietary Fish Meal Replacement with Hermetia illucens and Tenebrio molitor Larval Meals Improves the Growth Performance and Nutriphysiological Status of Ide (Leuciscus idus) Juveniles. Animals. 2022; 12(10):1227. https://doi.org/10.3390/ani12101227
Chicago/Turabian StyleHomska, Natalia, Joanna Kowalska, Joanna Bogucka, Ewa Ziółkowska, Mateusz Rawski, Bartosz Kierończyk, and Jan Mazurkiewicz. 2022. "Dietary Fish Meal Replacement with Hermetia illucens and Tenebrio molitor Larval Meals Improves the Growth Performance and Nutriphysiological Status of Ide (Leuciscus idus) Juveniles" Animals 12, no. 10: 1227. https://doi.org/10.3390/ani12101227
APA StyleHomska, N., Kowalska, J., Bogucka, J., Ziółkowska, E., Rawski, M., Kierończyk, B., & Mazurkiewicz, J. (2022). Dietary Fish Meal Replacement with Hermetia illucens and Tenebrio molitor Larval Meals Improves the Growth Performance and Nutriphysiological Status of Ide (Leuciscus idus) Juveniles. Animals, 12(10), 1227. https://doi.org/10.3390/ani12101227







