The Formation and Invariance of Canine Nose Pattern of Beagle Dogs from Early Puppy to Young Adult Periods
Abstract
:Simple Summary
Abstract
1. Introduction
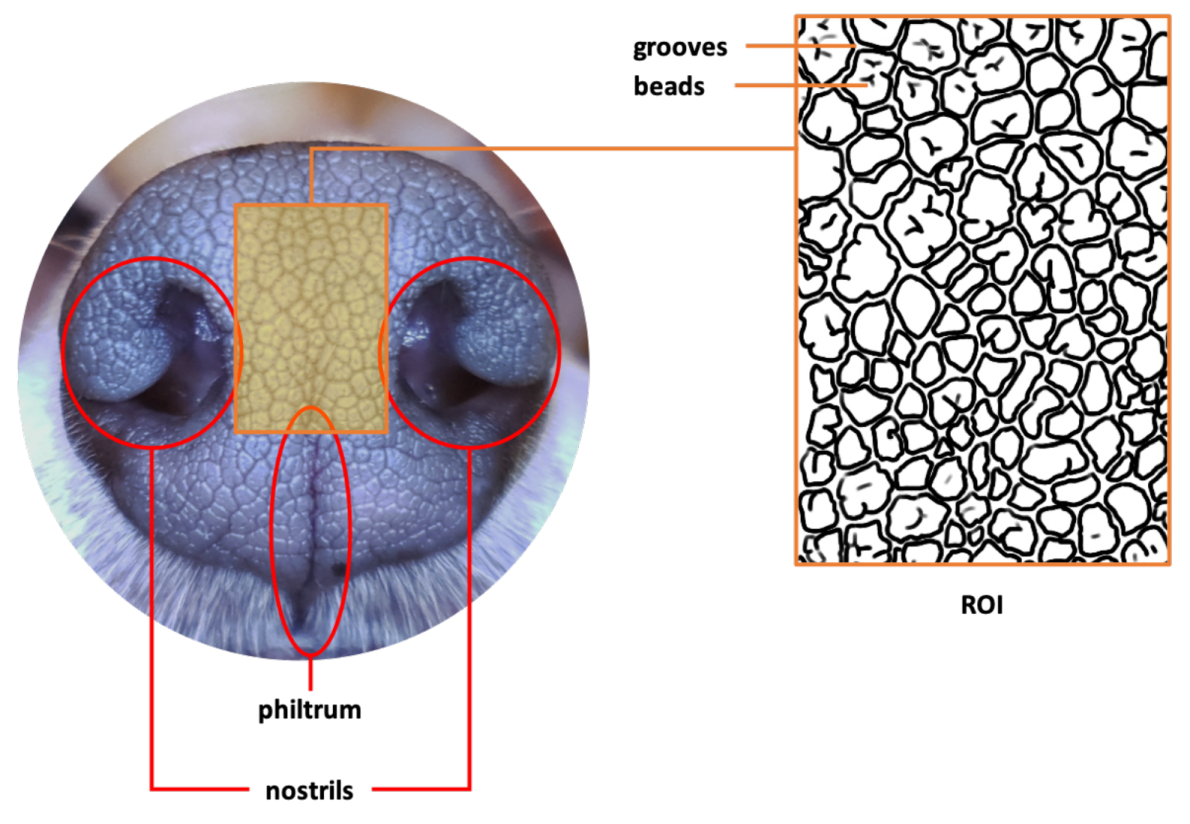
2. Canine Nose Pattern and its Biological Basis
3. Materials and Methods
- Birth of puppiesOn 6 Novemebr 2019, the staff of Konkuk University Veterinary Medical Teaching Hospital procured two pregnant female beagle dogs that gave birth to ten puppies on 19 November 2019. Six of the ten puppies were born of one dam and four of the other. Genetic identification of individual puppies was performed with the canine genotype panel 2.1 kit (ThermoFisher) using buccal swabs.
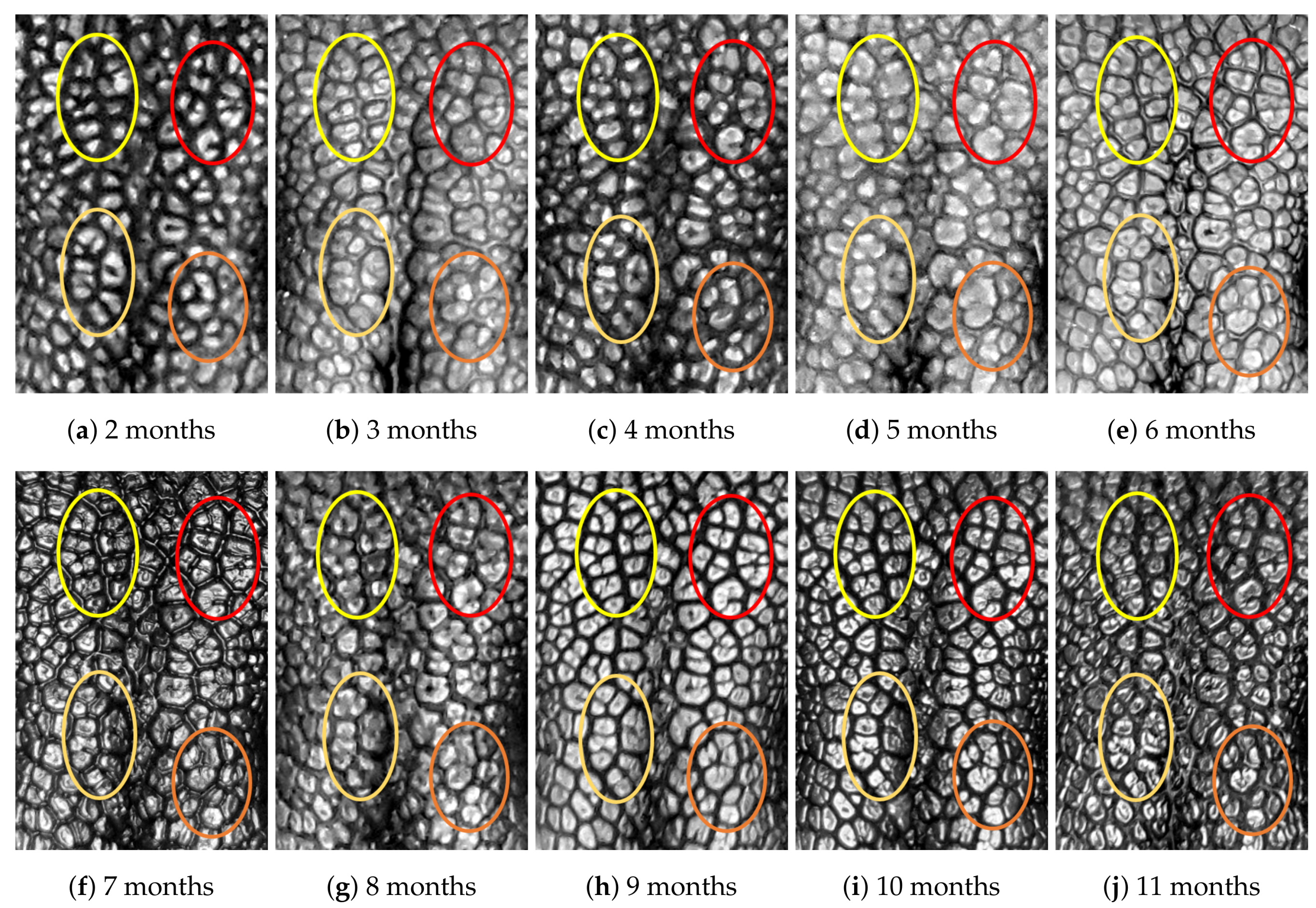
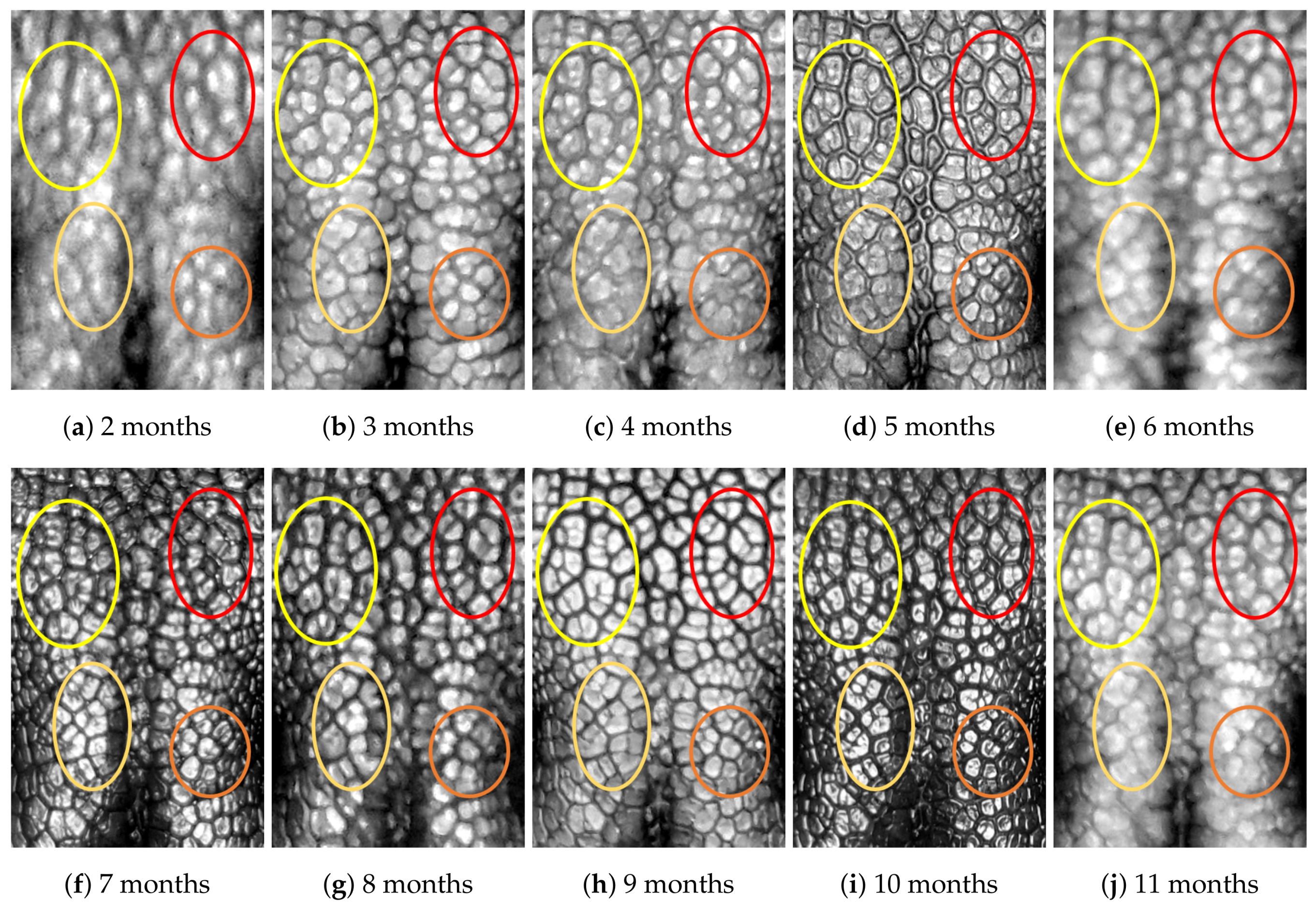
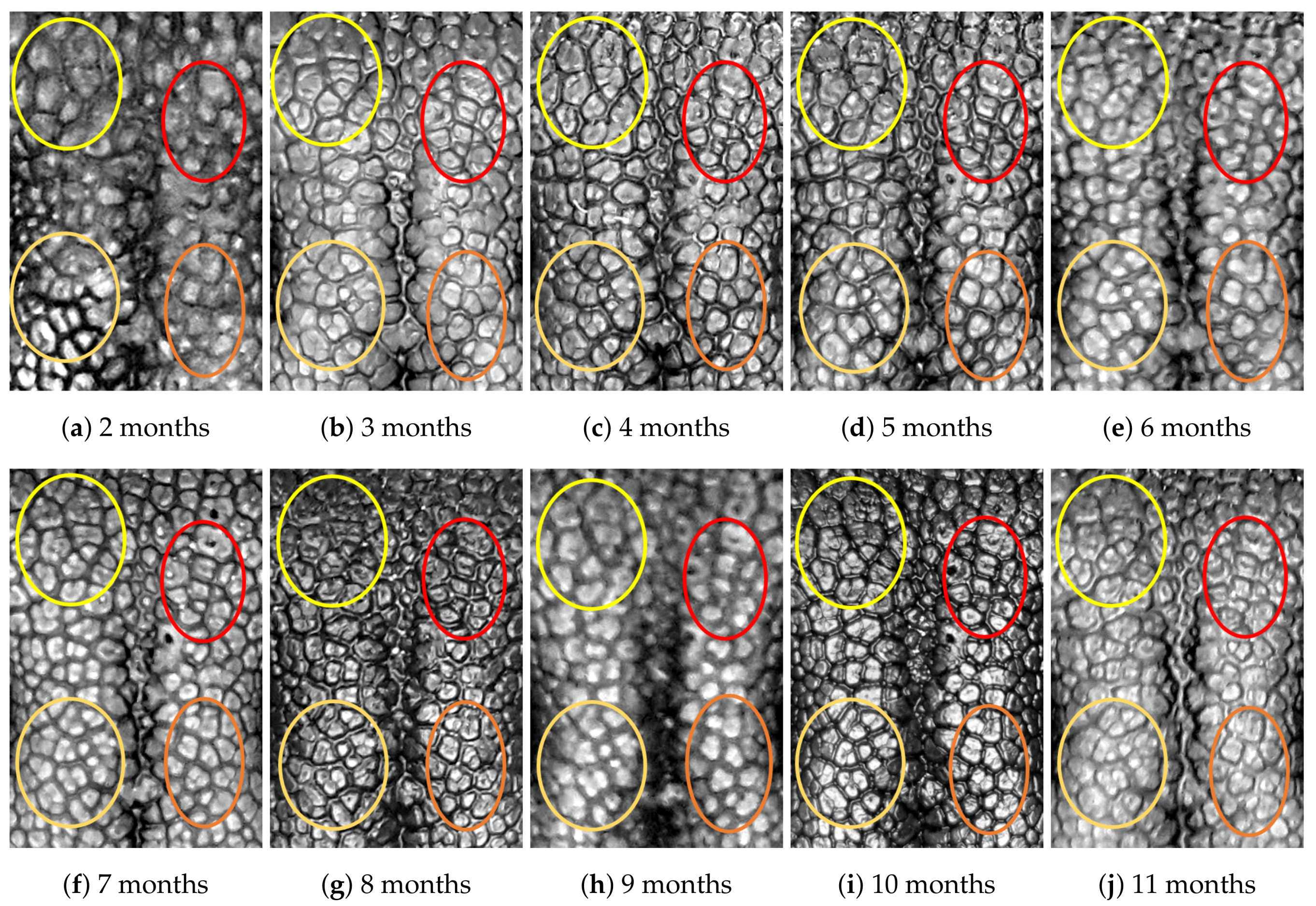
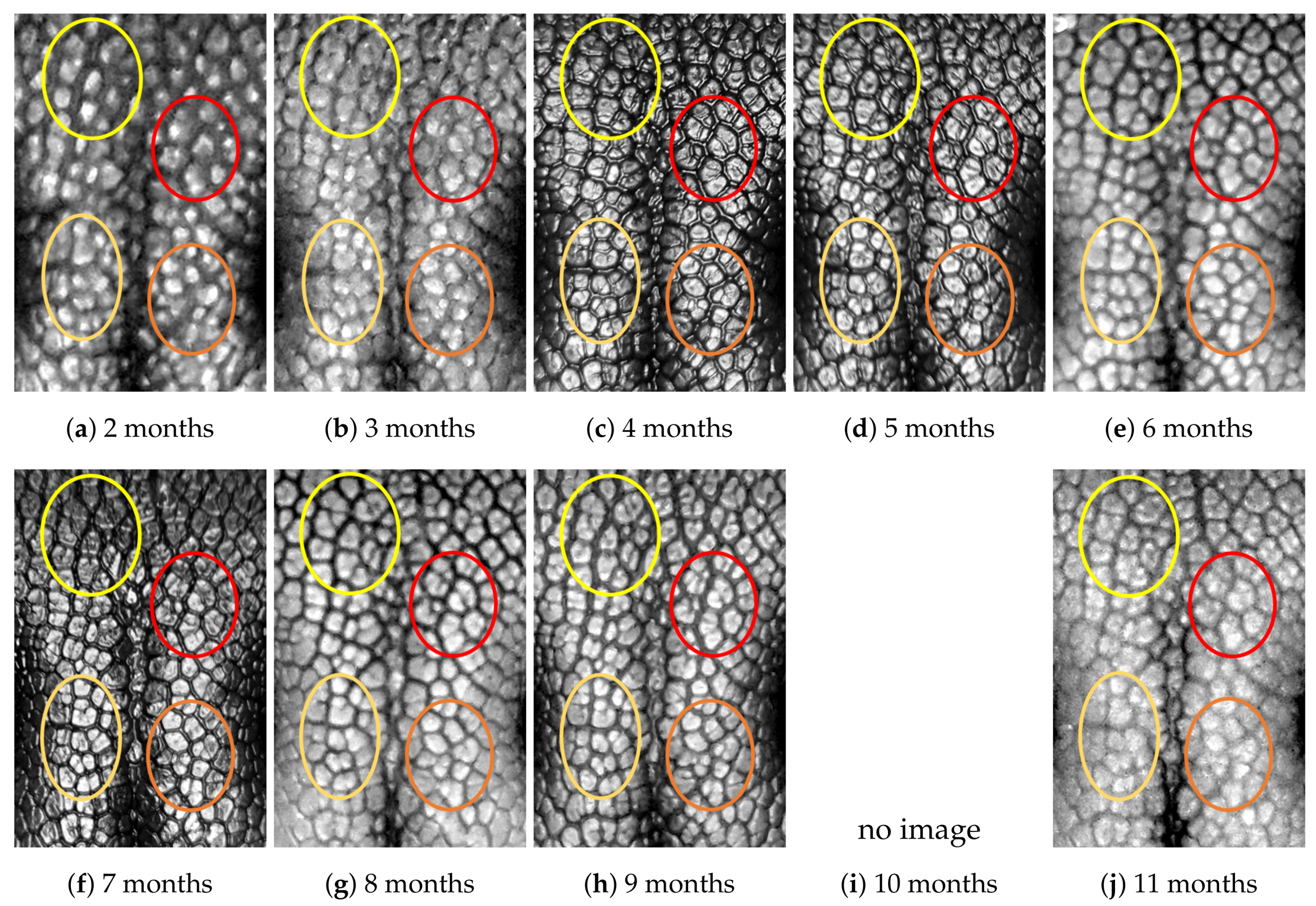
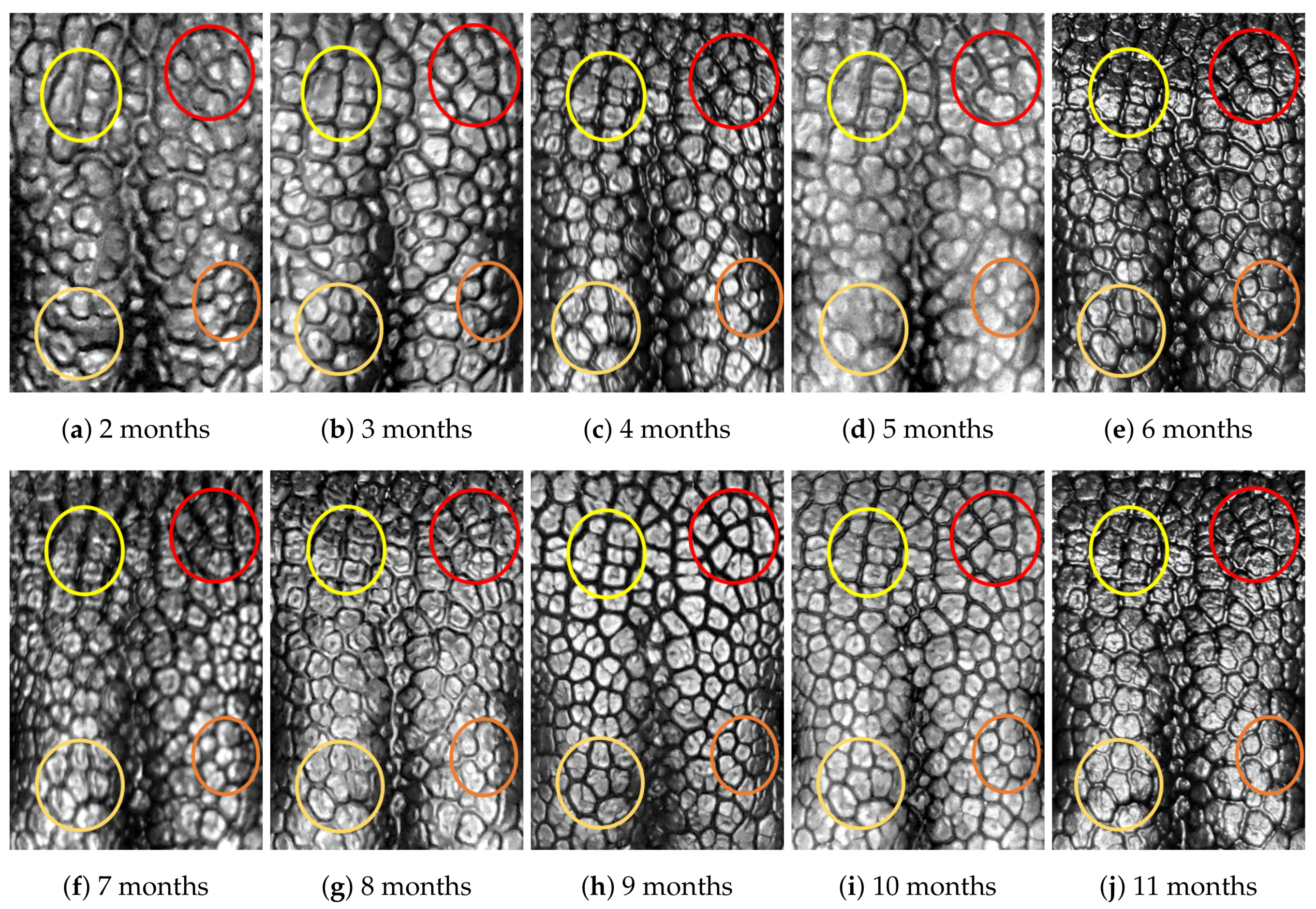
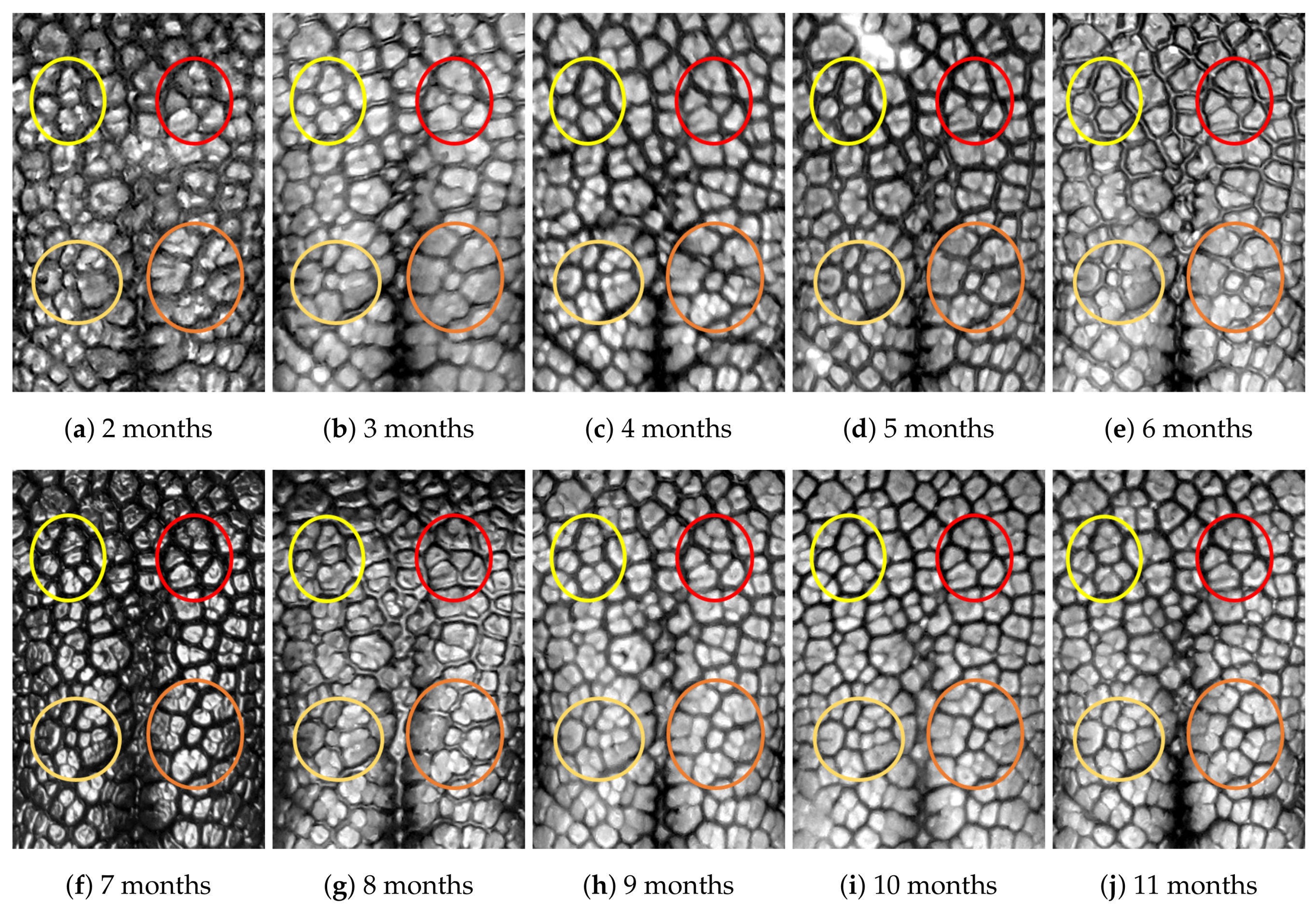
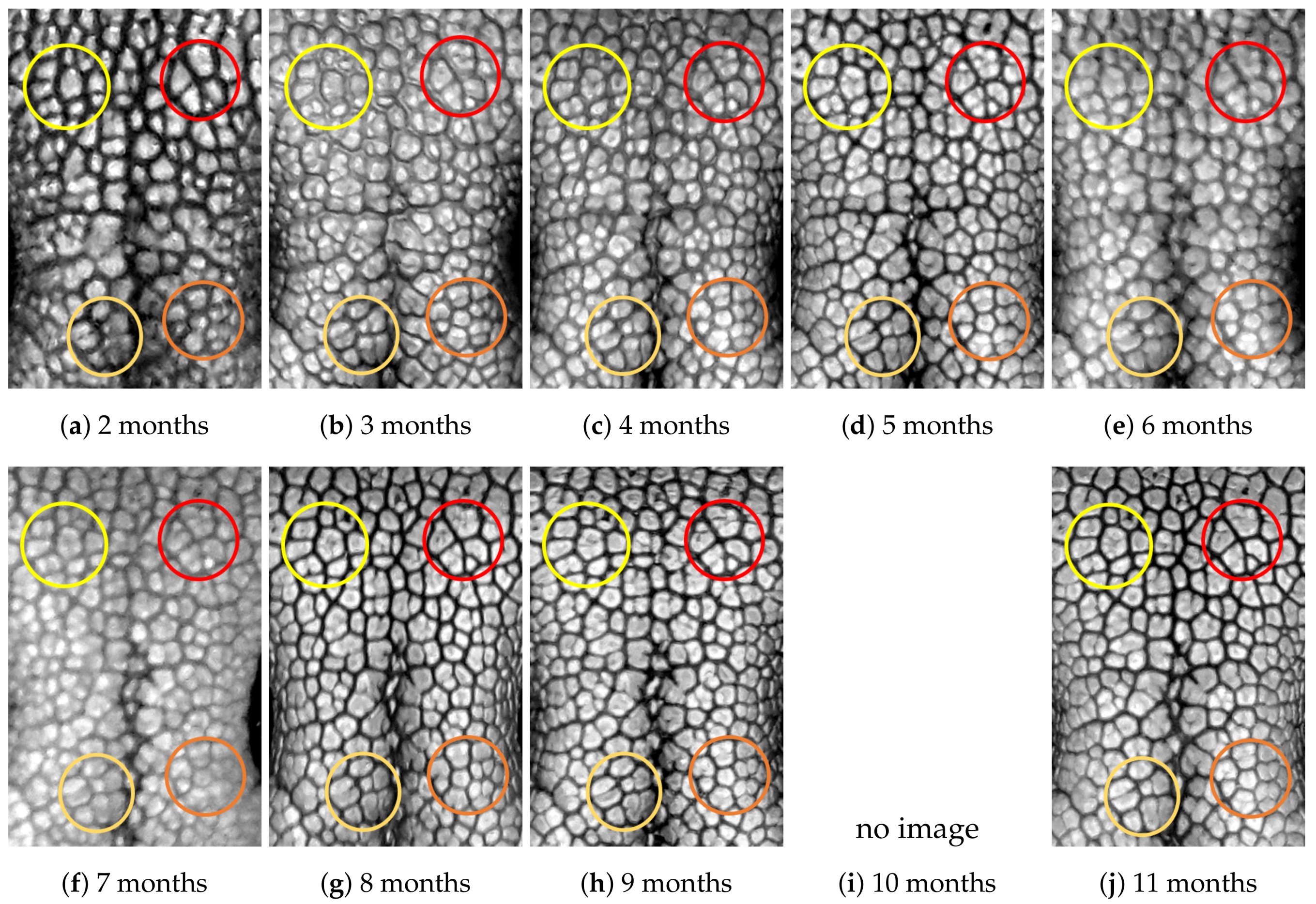
- Capturing nose imagesAfter taking care of necessary precautions such as vaccinations, the staff started to capture nose images of each of the ten puppies on 16 January 2019 (the second mensiversary). Afterwards, nose images of the ten puppies were taken every month for the ten-month period ending on 21 October 2020 (the eleventh mensiversary). The exact dates are recorded in Table 1.
4. Results
4.1. Visual Examination
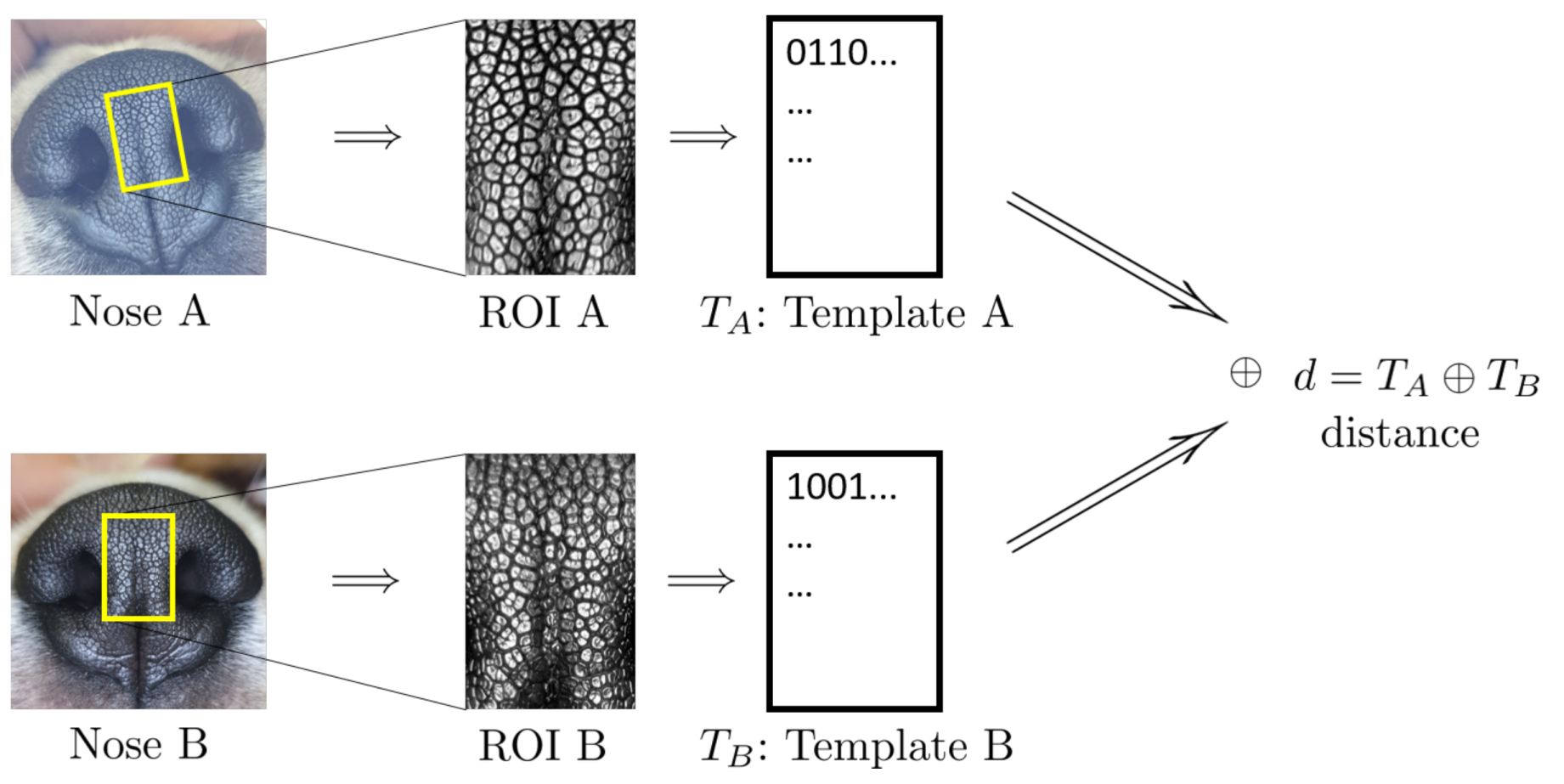
4.2. Algorithmic Verification
- (Genuine comparisons) the nose pattern of a dog formed at the second mensiversary remains the same throughout the dog’s life;
- (Impostor comparisons) the nose patterns of different dogs are always different regardless of when the images are taken.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Appendix A. Data




References
- Vascellari, M.; Mutinelli, F.; Cossettini, R.; Altinier, E. Liposarcoma at the site of an implanted microchip in a dog. Vet. J. 2004, 168, 188–190. [Google Scholar] [CrossRef]
- Vascellari, M.; Melchiotti, E.; Mutinelli, F. Fibrosarcoma with Typical Features of Postinjection Sarcoma at Site of Microchip Implant in a Dog: Histologic and Immunohistochemical Study. Vet. Pathol. 2006, 43, 545–548. [Google Scholar] [CrossRef] [Green Version]
- Legallet, C.; Mankin, K.T.; Spaulding, K.; Mansell, J. Granulomatous Inflammatory Response to a Microchip Implanted in a Dog for Eight Years. J. Am. Anim. Hosp. Assoc. 2017, 53, 227–229. [Google Scholar] [CrossRef]
- Daly, M.K.; Saba, C.F.; Crochik, S.S.; Howerth, E.W.; Kosarek, C.E.; Cornell, K.K.; Roberts, R.E.; Northrup, N.C. Fibrosarcoma adjacent to the site of microchip implantation in a cat. J. Feline Med. Surg. 2008, 10, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Carminato, A.; Vascellari, M.; Marchioro, W.; Melchiotti, E.; Mutinelli, F. Microchip-associated fibrosarcoma in a cat. Vet. Dermatol. 2011, 22, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Tillmann, T.; Kamino, K.; Dasenbrock, C.; Ernst, H.; Kohler, M.; Morawietz, G.; Campo, E.; Cardesa, A.; Tomatis, L.; Mohr, U. Subcutaneous soft tissue tumours at the site of implanted microchips in mice. Exp. Toxicol. Pathol. 1997, 49, 197–200. [Google Scholar] [CrossRef]
- Blanchard, K.T.; Barthel, C.; French, J.E.; Holden, H.E.; Moretz, R.; Pack, F.D.; Tennant, R.W.; Stoll, R.E. Transponder-Induced Sarcoma in the Heterozygous p53+/- Mouse. Toxicol. Pathol. 1999, 27, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Elcock, L.E.; Stuart, B.P.; Wahle, B.S.; Hoss, H.E.; Crabb, K.; Millard, D.M.; Mueller, R.E.; Hastings, T.F.; Lake, S.G. Tumors in long-term rat studies associated with microchip animal identification devices. Exp. Toxicol. Pathol. 2001, 52, 483–491. [Google Scholar] [CrossRef]
- Lapp, S.; Bube, A.; Colbatzky, F.A.; Ernst, H.; Kellner, R.; Nolte, T.; Rinke, M. Best Practice Approach for Assessment of Microchip-associated Tumors in Preclinical Safety Studies: Position of the Registry of Industrial Toxicology Animal-data (RITA). Toxicol. Pathol. 2018, 46, 728–734. [Google Scholar] [CrossRef]
- Platt, S.; Wieczorek, L.; Dennis, R.; Stefani, A.D. Spinal cord injury resulting from incorrect microchip placement in a cat. J. Feline Med. Surg. 2007, 9, 157–160. [Google Scholar] [CrossRef]
- Joslyn, S.K.; Witte, P.G.; Scott, H.W. Delayed spinal cord injury following microchip placement in a dog. Vet. Comp. Orthop. Traumatol. 2010, 23, 214–217. [Google Scholar] [CrossRef]
- Smith, T.J.; Fitzpatrick, N. Surgical removal of a microchip from a puppy’s spinal canal. Vet. Comp. Orthop. Traumatol. 2009, 22, 63–65. [Google Scholar] [CrossRef]
- Taylor-Brown, F.; Kenny, P.J.; Whiting, M. Microchipping very small dogs. Vet. Rec. 2016, 179, 151. [Google Scholar] [CrossRef] [PubMed]
- Swift, S. Keeping track of microchip adverse reactions. J. Small Anim. Pract. 2002, 43, 570. [Google Scholar] [PubMed]
- Petersen, W. The Identification of the Bovine by Means of Nose-Prints. J. Dairy Sci. 1922, 5, 249–258. [Google Scholar] [CrossRef]
- Baranov, A.S.; Graml, R.; Pirchner, F.; Schmid, D.O. Breed differences and intra-breed genetic variability of dermatoglyphic pattern of cattle. J. Anim. Breed. Genet. 1993, 110, 385–392. [Google Scholar] [CrossRef]
- Coldea, N. Nose prints as a method of identification in dogs. Vet. Q. 1994, 16, 60. [Google Scholar] [CrossRef]
- Horning, J.G.; McKee, A.J.; Keller, H.E.; Smith, K.K. Nose printing your cat and dog patient. Vet. Med. 1926, 21, 432–453. [Google Scholar]
- Evans, H.; de Lahunta, A. Miller’s Anatomy of the Dog, 4th ed.; Saunders: St. Louis, MI, USA, 2012. [Google Scholar]
- Budras, K.; McCarthy, P.; Fricke, W.; Richter, R.; Horowitz, A.; Berg, R. Anatomy of the Dog: An Illustrated Text, 5th ed.; Schluetersche: Hannover, Germany, 2007. [Google Scholar]
- Barry, B.; Gonzales-Barron, U.; Mcdonnell, K.; Butler, F.; Ward, S. Using Muzzle Pattern Recognition as a Biometric Approach for Cattle Identification. Trans. ASABE 2007, 50, 1073–1080. [Google Scholar] [CrossRef]
- Noviyanto, A.; Arymurthy, A. Automatic cattle identification based on muzzle photo using speed-up robust features approach. In Proceedings of the 3rd European Conference of Computer Science, Paris, France, 2–4 December 2012; pp. 110–114. [Google Scholar]
- Noviyanto, A.; Arymurthy, A.M. Beef cattle identification based on muzzle pattern using a matching refinement technique in the SIFT method. Comput. Electron. Agric. 2013, 99, 77–84. [Google Scholar] [CrossRef]
- Awad, A.I.; Hassanien, A.E.; Zawbaa, H.M. A Cattle Identification Approach Using Live Captured Muzzle Print Images. In Advances in Security of Information and Communication Networks; Awad, A.I., Hassanien, A.E., Baba, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 143–152. [Google Scholar]
- Awad, A.I.; Zawbaa, H.M.; Mahmoud, H.A.; Nabi, E.H.H.A.; Fayed, R.H.; Hassanien, A.E. A robust cattle identification scheme using muzzle print images. In Proceedings of the 2013 Federated Conference on Computer Science and Information Systems, Krakow, Poland, 8–11 September 2013; pp. 529–534. [Google Scholar]
- Tharwat, A.; Gaber, T.; Hassanien, A.E.; Hassanien, H.A.; Tolba, M.F. Cattle Identification Using Muzzle Print Images Based on Texture Features Approach. In Proceedings of the Fifth International Conference on Innovations in Bio-Inspired Computing and Applications IBICA 2014; Kömer, P., Abraham, A., Snášel, V., Eds.; Springer International Publishing: Cham, Switzerland, 2014; pp. 217–227. [Google Scholar]
- Tharwat, A.; Gaber, T.; Hassanien, A.E. Cattle Identification Based on Muzzle Images Using Gabor Features and SVM Classifier. In Advanced Machine Learning Technologies and Applications; Hassanien, A.E., Tolba, M.F., Taher Azar, A., Eds.; Springer International Publishing: Cham, Switzerland, 2014; pp. 236–247. [Google Scholar]
- Tharwat, A.; Hassanien, A.E. Two Biometric Approaches for Cattle Identification Based on Features and Classifiers Fusion. Int. J. Image Min. 2015, 1, 342–365. [Google Scholar] [CrossRef]
- Kücken, M.; Newell, A.C. Fingerprint formation. J. Theor. Biol. 2005, 235, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Budras, K.D. Bovine Anatomy an Illustrated Text, 2nd ed.; Schluetersche: Hannover, Germany, 2011. [Google Scholar]
- Jain, A.K.; Ross, A.A.; Nandakumar, K. Introduction to Biometrics; Springer Publishing Company, Incorporated: New York, NY, USA, 2011. [Google Scholar]
- Li, S.Z.; Jain, A. Encyclopedia of Biometrics; Springer Publishing Company, Incorporated: New York, NY, USA, 2015. [Google Scholar]
- Kumar, S.; Singh, S.K.; Singh, R.; Singh, A.K. Animal Biometrics; Springer: Singapore, 2017. [Google Scholar]
- Han, C.Y.; Kwon, S.H.; Choi, H.I.; Lee, S.J.; Wee, N.S. Seeded Ising model and the statistical nature of human iris templates. Phys. Rev. E 2018, 98, 032115. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.I.; Lee, S.; Moon, H.P.; Wee, N.S.; Kim, D.; Kwon, S.H. Seeded Ising Model and Distributed Biometric Template Storage and Matching. Entropy 2021, 23, 849. [Google Scholar] [CrossRef]

| Age in months | 2 M | 3 M | 4 M | 5 M | 6 M |
| date | 16 January 2020 | 20 February 2020 | 19 March 2020 | 23 April 2020 | 21 May 2020 |
| Age in months | 7 M | 8 M | 9 M | 10 M | 11 M |
| date | 18 June 2020 | 16 July 2020 | 18 August 2020 | 15 September 2020 | 21 October 2020 |
| # of Subjects | 10 |
| # of Images per subject | 9∼10 |
| Total # of Images | 98 |
| # of Genuine comparisons | 432 |
| # of Impostor comparisons | 4321 |
| Comparison Type | Min | Max | Mean | Std |
|---|---|---|---|---|
| genuine | 0.1265 | 0.3577 | 0.2324 | 0.0456 |
| impostor | 0.4254 | 0.5496 | 0.4894 | 0.01955 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, H.I.; Lee, Y.; Shin, H.; Lee, S.; Choi, S.S.; Han, C.Y.; Kwon, S.-H. The Formation and Invariance of Canine Nose Pattern of Beagle Dogs from Early Puppy to Young Adult Periods. Animals 2021, 11, 2664. https://doi.org/10.3390/ani11092664
Choi HI, Lee Y, Shin H, Lee S, Choi SS, Han CY, Kwon S-H. The Formation and Invariance of Canine Nose Pattern of Beagle Dogs from Early Puppy to Young Adult Periods. Animals. 2021; 11(9):2664. https://doi.org/10.3390/ani11092664
Chicago/Turabian StyleChoi, Hyeong In, Yoonsuk Lee, Hyunjin Shin, Sungjin Lee, Stephanie Sujin Choi, Chang Yong Han, and Song-Hwa Kwon. 2021. "The Formation and Invariance of Canine Nose Pattern of Beagle Dogs from Early Puppy to Young Adult Periods" Animals 11, no. 9: 2664. https://doi.org/10.3390/ani11092664
APA StyleChoi, H. I., Lee, Y., Shin, H., Lee, S., Choi, S. S., Han, C. Y., & Kwon, S.-H. (2021). The Formation and Invariance of Canine Nose Pattern of Beagle Dogs from Early Puppy to Young Adult Periods. Animals, 11(9), 2664. https://doi.org/10.3390/ani11092664






