Functional Neurorehabilitation in Dogs with an Incomplete Recovery 3 Months following Intervertebral Disc Surgery: A Case Series
Simple Summary
Abstract
1. Introduction
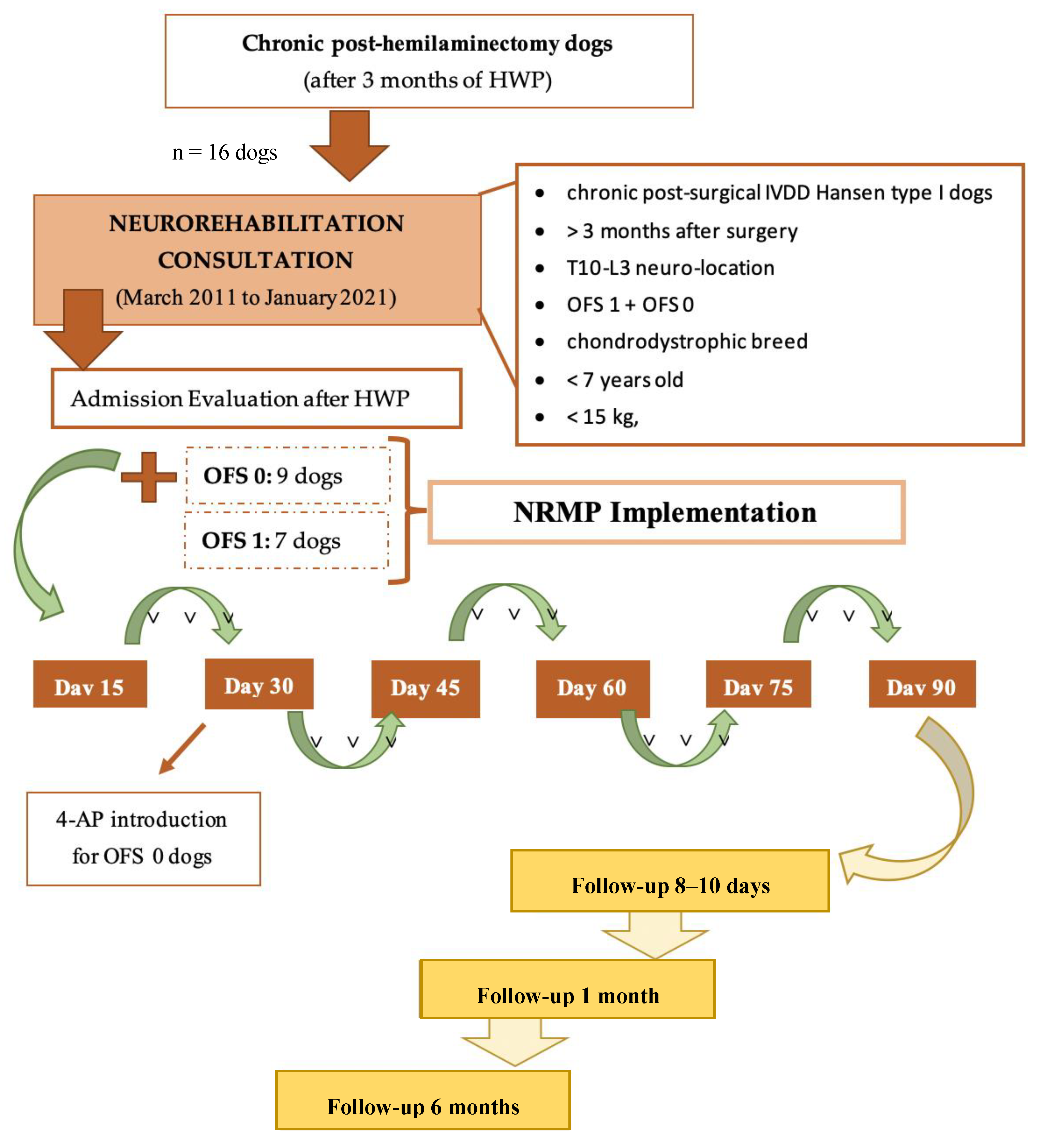
2. Materials and Methods
2.1. Participants
2.2. Interventions
2.2.1. Neurorehabilitation Multimodal Protocol
- Locomotor training
- Electrical Stimulation
- Pharmacological management
- Supportive care
2.2.2. Outcomes/Follow-Up
2.2.3. Statistical Analysis
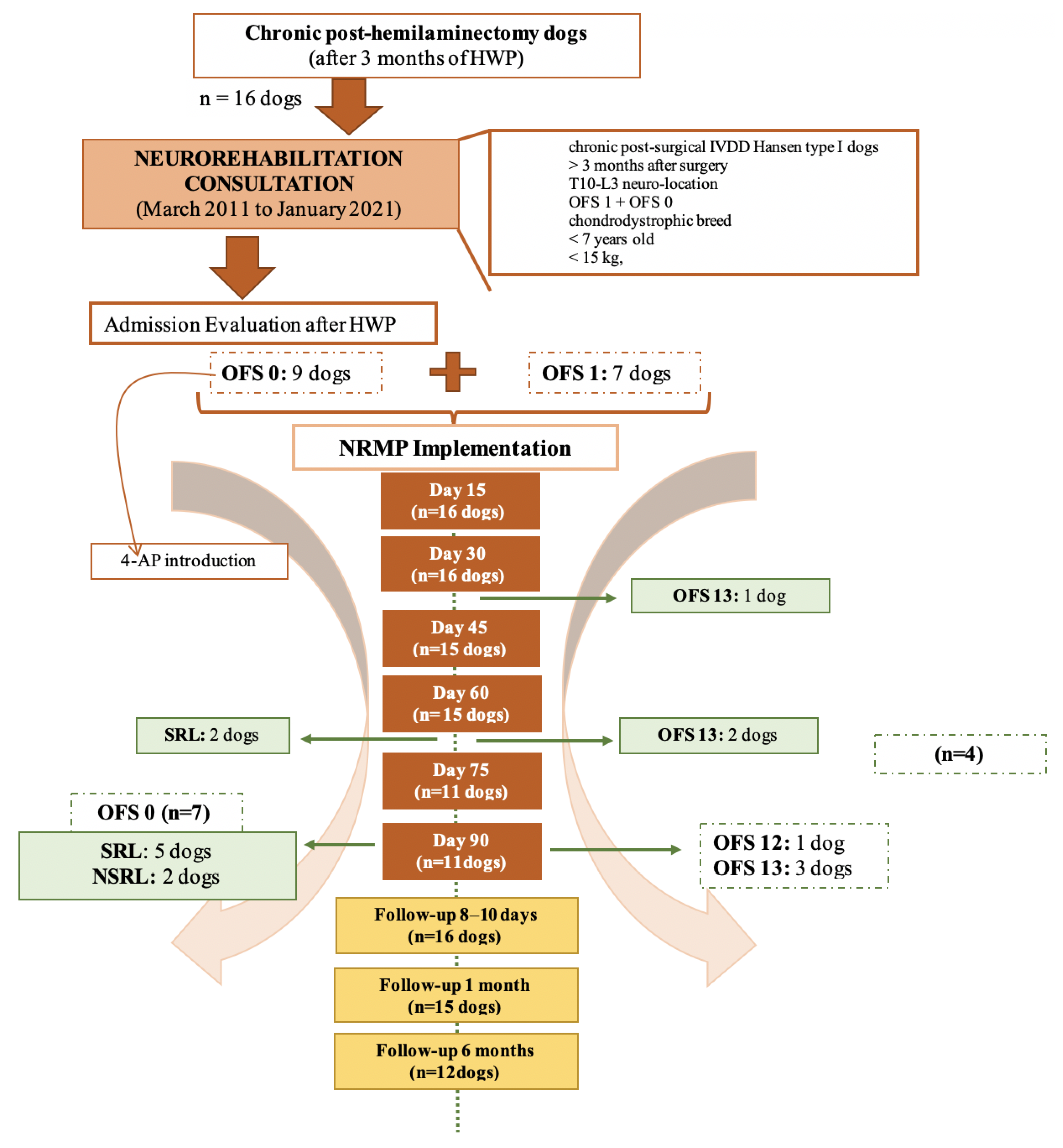
3. Results
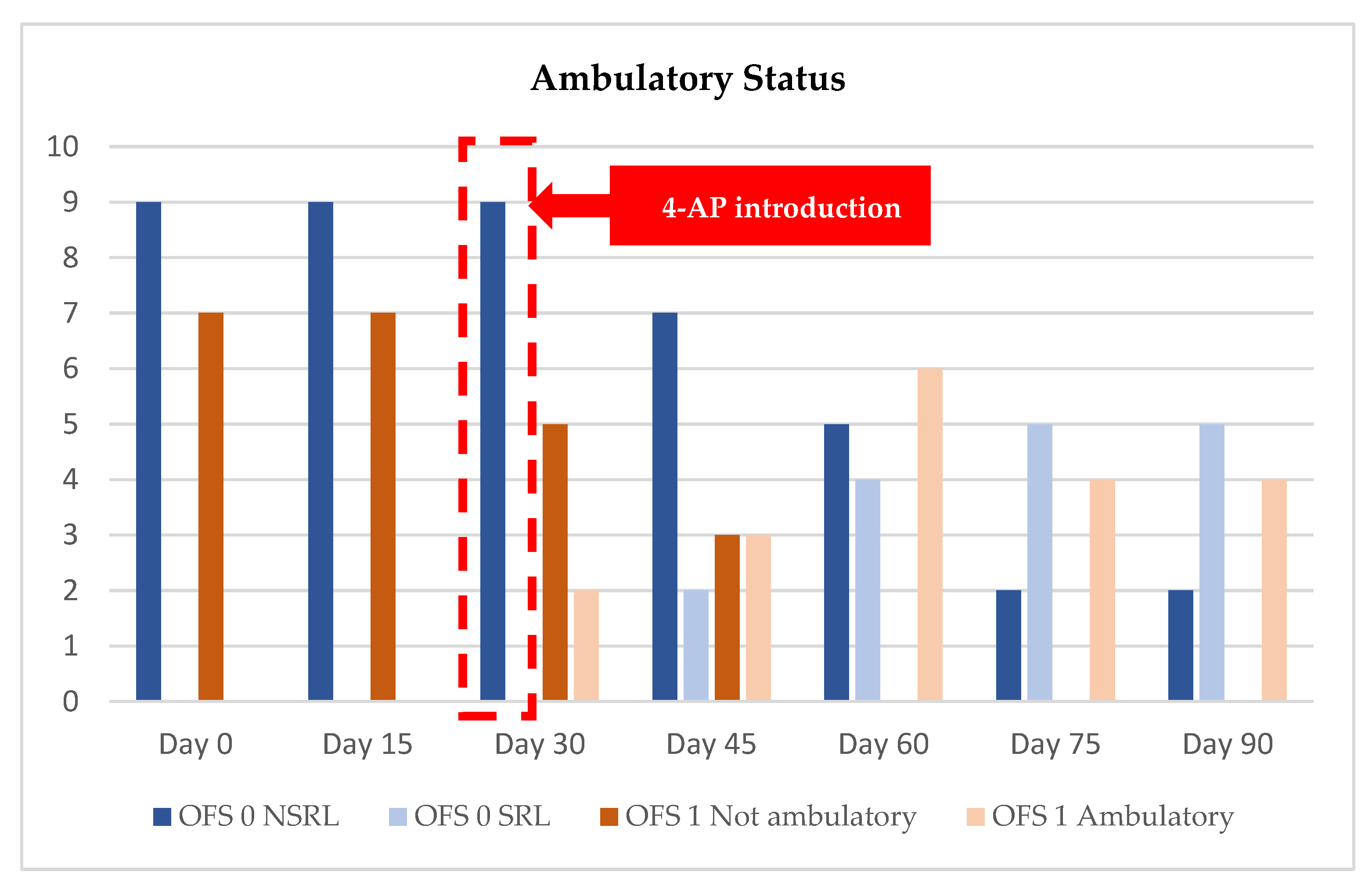
3.1. DPP− Dogs’ Results
3.2. DPP+ Dogs’ Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Olby, N.; Levine, J.; Harris, T.; Muñana, K.; Skeen, T.; Sharp, N. Long-term functional outcome of dogs with severe injuries of the thoracolumbar spinal cord:87 cases (1996–2001). J. Am. Vet. Med. Assoc. 2003, 222, 762–769. [Google Scholar] [CrossRef]
- Jeffery, N.D.; Barker, A.K.; Hu, H.Z.; Alcott, C.J.; Kraus, K.H.; Scanlin, E.M.; Granger, N.; Levine, J.M. Factors associated with recovery from paraplegia in dogs with loss of pain perception in the pelvic limbs following intervertebral disk herniation. J. Am. Vet. Med. Assoc. 2016, 248, 386–394. [Google Scholar] [CrossRef]
- Fingeroth, J.M.; Forterre, F.; Levine, J.M. Compressive and contusive spinal cord injury secondary to intervertebral disc displacement: A clinical perspective. In Advances in Intervertebral Disc Disease in Dogs and Cats; Fingeroth, J.M., Thomas, W.B., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 131–134. [Google Scholar]
- Ingram, E.A.; Kale, D.C.; Balfour, R.J. Hemilaminectomy for thoracolumbar hansen type I intervertebral disk disease in ambulatory dogs with or without neurologic deficits: 39 cases (2008–2010). Vet. Surg. 2013, 42, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Howard, J.F.; Olby, N.J. The relationship between trans-lesional conduction, motor neuron pool excitability, and motor function in dogs with incomplete recovery from severe spinal cord injury. J. Neurotrauma 2017, 34, 2994–3002. [Google Scholar] [CrossRef]
- Lewis, M.J.; Jeffery, N.D.; Olby, N.J. Ambulation in dogs with absent pain perception after acute thoracolumbar Spinal Cord Injury. Front. Vet. Sci. 2020, 7, 560–572. [Google Scholar] [CrossRef] [PubMed]
- Alluin, O.; Delivet-Mongrain, H.; Rossignol, S. Inducing hindlimb locomotor recovery in adult rat after complete thoracic spinal cord section using repeated treadmill training with perineal stimulation only. J. Neurophysiol. 2015, 114, 1031–1046. [Google Scholar] [CrossRef] [PubMed]
- Militskova, A.; Mukhametova, E.; Fatykhova, E.; Sharifullin, S.; Cuellar, C.A.; Calvert, J.S.; Grahn, P.J.; Baltina, T.; Lavrov, I. Supraspinal and Afferent Signaling Facilitate Spinal Sensorimotor Network Excitability After Discomplete Spinal Cord Injury: A Case Report. Front. Neurosci. 2020, 14, 552. [Google Scholar] [CrossRef]
- Olby, N.J.; Lim, J.H.; Wagner, N.; Zidan, N.; Early, P.J.; Mariani, C.L.; Muñana, K.R.; Laber, E. Time course and prognostic value of serum GFAP, pNFH, and S100β concentrations in dogs with complete spinal cord injury because of intervertebral disc extrusion. J. Vet. Intern. Med. 2019, 33, 726–734. [Google Scholar] [CrossRef]
- Fingeroth, J.M.; De Lahunta, A. Ascending/Descending myelomalacia secondary to Intervertebral Disc Herniation. In Advances in Intervertebral Disc Disease in Dogs and Cats; Fingeroth, J.M., Thomas, W.B., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 115–120. [Google Scholar]
- Moore, S.A.; Tipold, A.; Olby, N.J.; Stein, V.; Granger, N. Current approaches to the management of acute thoracolumbar disc extrusion in dogs. Front. Vet. Sci. 2020, 7, 610. [Google Scholar] [CrossRef]
- Tansey, K.E. Neural plasticity and locomotor recovery after spinal cord injury. PM R 2010, 2, 220–226. [Google Scholar] [CrossRef]
- Dimitrijevic, M.R. Residual motor function after spinal cord injury. In Restorative Neurology of Spinal Cord Injury; Dimitrejevic, M.R., Ed.; Oxford University Press: New York, NY, USA, 2012; pp. 1–9. [Google Scholar]
- Kakulas, B.A.; Kaelan, C. The neuropathological foundations for the restorative neurology of spinal cord injury. Clin. Neurol. Neurosurg. 2015, 129, 51–57. [Google Scholar] [CrossRef]
- Alisauskaite, N.; Spitzbarth, I.; Baumgartner, W.; Dziallas, P.; Kramer, S.; Dening, R.; Stein, V.M.; Tipold, A. Chronic post-traumatic intramedullary lesion in dogs, a translational model. PLoS ONE 2017, 12, e0187746. [Google Scholar] [CrossRef]
- Martins, Â.; Silva, C.M.; Gouveia, D.; Cardoso, A.; Coelho, T.; Gamboa, Ó.; Marcelino, E.; Ferreira, A. Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study. Animals 2021, 11, 1994. [Google Scholar] [CrossRef]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Viegas, I.; Gamboa, Ó.; Ferreira, A. A Comparison Between Body Weight-Supported Treadmill Training and Conventional Over-Ground Training in Dogs with Incomplete Spinal Cord Injury. Front. Vet. Sci. 2021, 8, 597949. [Google Scholar] [CrossRef]
- Solopova, I.A.; Selionov, V.A.; Sylos-Labini, F.; Gurfinkel, V.S.; Lacquaniti, F.; Ivanenko, Y.P. Tapping into rhythm generation circuitry in humans during simulated weightlessness conditions. Front. Syst. Neurosci. 2015, 18, 9–14. [Google Scholar] [CrossRef]
- Harness, E.T.; Yozbatiran, N.; Cramer, S.C. Effects of intense exercise in chronic spinal cord injury. Spinal Cord 2008, 46, 733–737. [Google Scholar] [CrossRef]
- Rossignol, S.; Bouyer, L. Adaptive Mechanisms of Spinal Locomotion in Cats. Integr. Comp. Biol. 2004, 44, 71–79. [Google Scholar] [CrossRef] [PubMed]
- De Leon, R.D.; Hodgson, J.A.; Roy, R.R.; Edgerton, V.R. Retention of hindlimb stepping ability in adult spinal cats after the cessation of step training. J. Neurophysiol. 1999, 81, 85–94. [Google Scholar] [CrossRef]
- Fouad, K.; Tetzlaff, W. Rehabilitative training and plasticity following spinal cord injury. Exp. Neurol. 2012, 235, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Kiehn, O. Locomotor circuits in the mammalian spinal cord. Annu. Rev. Neurosci. 2006, 29, 279–306. [Google Scholar] [CrossRef] [PubMed]
- Côté, M.P.; Murray, L.M.; Knikou, M. Spinal Control of locomotion: Individual neurons, their circuits and functions. Front. Physiol. 2018, 9, 784. [Google Scholar] [CrossRef]
- Guertin, P.A. Preclinical evidence supporting the clinical development of central pattern generator-modulating therapies for chronic spinal cord-injured patients. Front. Hum. Neurosci. 2014, 8, 272. [Google Scholar] [CrossRef] [PubMed]
- Barbeau, H.; Rossignol, S. Enhancement of locomotor recovery following spinal cord injury. Curr. Opin. Neurol. 1994, 7, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Wirz, M.; Zemon, D.H.; Rupp, R.; Anke, S.; Colombo, G.; Dietz, V.; Hornby, T.G. Effectiveness of automated locomotor training in patients with chronic incomplete spinal cord injury: A multicenter trial. Arch. Phys. Med. Rehabil. 2005, 86, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Barriére, G.; Leblond, H.; Provencher, J.; Rossignol, S. Prominent role of the spinal central pattern generator in the recovery of locomotion after partial spinal cord injuries. J. Neurosci. 2008, 28, 3976–3987. [Google Scholar] [CrossRef] [PubMed]
- Gossard, J.P.; Delivet-Mongrain, H.; Martinez, M.; Kundu, A.; Escalona, M.; Rossignol, S. Plastic changes in lumbar locomotor networks after a partial spinal cord injury in Cats. J. Neurosci. 2015, 35, 9446–9455. [Google Scholar] [CrossRef]
- Leech, K.A.; Kinnaird, C.R.; Holleran, C.L.; Kahn, J.; Hornby, T.G. Effects of locomotor exercise intensity on gait performance in individuals with incomplete spinal cord injury. Phys. Ther. 2016, 96, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S. Neural control of stereotypic limb movements. In Handbook of Physiology; Rowell, L.B., Sheperd, J.T., Eds.; Sect. 12. Exercise: Regulation and Integration of Multiple Systems; American Physiological Society: Oxford, UK, 1996; pp. 173–216. [Google Scholar]
- Edgerton, V.R.; Leon, R.D.; Harkema, S.J.; Hodgson, J.A.; London, N.; Reinkensmeyer, D.J.; Roy, R.R.; Talmadge, R.J.; Tillakaratne, N.J.; Timoszyk, W.; et al. Retraining the injured spinal cord. J. Physiol. 2001, 533, 15–22. [Google Scholar] [CrossRef] [PubMed]
- de Leon, R.D.; Kubasak, M.D.; Phelps, P.E.; Timoszyk, W.K.; Reinkensmeyer, D.J.; Roy, R.R.; Edgerton, V.R. Using robotics to teach the spinal cord to walk. Brain Res. Rev. 2002, 40, 267–273. [Google Scholar] [CrossRef]
- Ichiyama, R.M.; Broman, J.; Roy, R.R.; Zhong, H.; Edgerton, V.R.; Havton, L.A. Locomotor training maintains normal inhibitory influence on both alpha- and gamma-motoneurons after neonatal spinal cord transection. J. Neurosci. 2011, 31, 26–33. [Google Scholar] [CrossRef]
- Martinez, M.; Delivet-Mongrain, H.; Leblond, H.; Rossignol, S. Effect of locomotor training in completely spinalized cats previously submitted to a spinal hemisection. J. Neurosci. 2012, 32, 10961–10970. [Google Scholar] [CrossRef]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Gamboa, Ó.; Millis, D.; Ferreira, A. Nervous system modulation through electrical stimulation in companion animals. Acta Vet. Scand. 2021, 63, 22. [Google Scholar] [CrossRef]
- Thompson, A.K.; Wolpaw, J.R. The simplest motor skill: Mechanisms and applications of reflex operant conditioning. Exerc. Sport Sci. Rev. 2014, 42, 82–90. [Google Scholar] [CrossRef]
- Thompson, A.K.; Wolpaw, J.R. Operant conditioning of spinal reflexes: From basic science to clinical therapy. Front. Integr. Neurosci. 2014, 18, 8–25. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.A.; Spaich, E.G.; Serrao, M.; Andersen, O.K. Stimulation site and phase modulation of the withdrawal reflex during gait initiation. Clin. Neurophysiol. 2015, 126, 2282–2289. [Google Scholar] [CrossRef]
- Krueger, E.; Magri, L.M.S.; Botelho, A.S.; Bach, F.S.; Rebellato, C.L.K.; Fracaro, L.; Fragoso, F.Y.I.; Villanova, J.A.; Brofman, P.R.S.; Popović-Maneski, L. Effects of low-intensity electrical stimulation and adipose derived stem cells transplantation on the time-domain analysis-based electromyographic signals in dogs with SCI. Neurosci. Lett. 2019, 696, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Noonan, V.K.; Kopec, J.A.; Zhang, H.; Dvorak, M.F. Impact of associated conditions resulting from spinal cord injury on health status and quality of life in people with traumatic central cord syndrome. Arch. Phys. Med. Rehabil. 2008, 89, 1074–1082. [Google Scholar] [CrossRef] [PubMed]
- Taghavi, N.; Luecke, G.R.; Jeffery, N.D. A wearable body controlling device for application of functional electrical stimulation. Sensors 2018, 18, 1251. [Google Scholar] [CrossRef] [PubMed]
- Milinis, K.; Young, C.A.; On behalf of the Trajectories of Outcome in Neurological Conditions (TONiC) Study. Systematic review of the influence of spasticity on quality of life in adults with chronic neurological conditions. Disabil. Rehabil. 2015, 29, 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Sayenko, D.G.; Atkinson, D.A.; Floyd, T.C.; Gorodnichev, R.M.; Moshonkina, T.R.; Harkema, S.J.; Edgerton, V.R.; Gerasimenko, Y.P. Effects of paired transcutaneous electrical stimulation delivered at single and dual sites over lumbosacral spinal cord. Neurosci. Lett. 2015, 609, 229–234. [Google Scholar] [CrossRef][Green Version]
- Ladenbauer, J.; Minassian, K.; Hofstoetter, U.S.; Dimitrijevic, M.R.; Rattay, F. Stimulation of the human lumbar spinal cord with implanted and surface electrodes: A computer simulation study. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 637–645. [Google Scholar] [CrossRef]
- Edgerton, V.R.; Harkema, S. Epidural stimulation of the spinal cord in spinal cord injury: Current status and future challenges. Expert Rev. Neurother. 2011, 11, 1351–1353. [Google Scholar] [CrossRef] [PubMed]
- Harkema, S.; Gerasimenko, Y.; Hodes, J.; Burdick, J.; Angeli, C.; Chen, Y.; Ferreira, C.; Willhite, A.; Rejc, E.; Grossman, R.G.; et al. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: A case study. Lancet 2011, 377, 1938–1947. [Google Scholar] [CrossRef]
- Hofstoetter, U.S.; Krenn, M.; Danner, S.M.; Hofer, C.; Kern, H.; McKay, W.B.; Mayr, W.; Minassian, K. Augmentation of voluntary locomotor activity by transcutaneous spinal cord stimulation in motor-incomplete spinal cord-injured individuals. Artif. Organs 2015, 39, 176–186. [Google Scholar] [CrossRef]
- Minassian, K.; Hofstoetter, U.S.; Danner, S.M.; Mayr, W.; Bruce, J.A.; McKay, W.B.; Tansey, K.E. Spinal Rhythm Generation by Step-Induced Feedback and Transcutaneous Posterior Root Stimulation in Complete Spinal Cord-Injured Individuals. Neurorehabil. Neural Repair 2016, 30, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Hofstoetter, U.S.; Knikou, M.; Guertin, P.A.; Minassian, K. Probing the Human Spinal Locomotor Circuits by Phasic Step-Induced Feedback and by Tonic Electrical and Pharmacological Neuromodulation. Curr. Pharm. Des. 2017, 23, 1805–1820. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Granger, N.; Jeffery, N.D.; Canine Spinal Cord Injury Consortium (CANSORT-SCI). Emerging and Adjunctive Therapies for Spinal Cord Injury Following Acute Canine Intervertebral Disc Herniation. Front. Vet. Sci. 2020, 7, 579933. [Google Scholar] [CrossRef] [PubMed]
- Blight, A.R.; Toombs, J.P.; Bauer, M.S.; Widmer, W.R. The effects of 4-aminopyridine on neurological deficits in chronic cases of traumatic spinal cord injury in dogs: A phase i clinical trial. J. Neurotrauma 1991, 8, 103–119. [Google Scholar] [CrossRef]
- Lim, J.H.; Muguet-Chanoit, A.C.; Smith, D.T.; Laber, E.; Olby, N.J. Potassium channel antagonists 4-aminopyridine and the T-butyl carbamate derivative of 4-aminopyridine improve hind limb function in chronically nonambulatory dogs; a blinded, placebo-controlled trial. PLoS ONE 2014, 9, e116139. [Google Scholar] [CrossRef]
- Lewis, M.J.; Laber, E.; Olby, N.J. Predictors of response to 4-aminopyridine in chronic canine spinal cord injury. J. Neurotrauma 2019, 36, 1428–1434. [Google Scholar] [CrossRef]
- Karimi-Abdolrezaee, S.; Billakanti, R. Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol. Neurobiol. 2012, 46, 251–264. [Google Scholar] [CrossRef]
- Couillard-Despres, S.; Bieler, L.; Vogl, M. Pathophysiology of traumatic Spinal Cord Injury. In Neurological Aspects of Spinal Cord Injury; Weider, N., Rupp, R., Tansey, K.E., Eds.; Springer: Cham, Switzerland, 2017; pp. 503–528. [Google Scholar]
- Rolls, A.; Shechter, R.; Schwartz, M. The bright side of the glial scar in CNS repair. Nat. Rev. Neurosci. 2009, 10, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.L.; Gallo, V. The diversity and disparity of the glial scar. Nat. Neurosci. 2018, 21, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Raineteau, O.; Schwab, M.E. Plasticity of motor systems after incomplete spinal cord injury. Nat. Rev. Neurosci. 2001, 2, 263–273. [Google Scholar] [CrossRef]
- Wolpaw, J.R. Spinal cord plasticity in acquisition and maintenance of motor skills. Acta Physiol. 2007, 189, 155–169. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.; Hahn, C. Veterinary Neuroanatomy, A Clinical Approach, 1st ed.; Edwards, R., Leng, H., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2012; pp. 47–57. [Google Scholar]
- Uemura, E.E. Fundamentals of Canine Neuroanatomy and Neurophysiology, 1st ed.; Uemura, E.E., Ed.; Wiley Blackwell: Oxford, UK, 2015; pp. 128–155. [Google Scholar]
- Dietz, V.; Colombo, G.; Jensen, L.; Baumgartner, L. Locomotor capacity of spinal cord in paraplegic patients. Ann. Neurol. 1995, 37, 574–582. [Google Scholar] [CrossRef]
- Benito-Penalva, J.; Edwards, D.J.; Opisso, E.; Cortes, M.; Lopez-Blazquez, R.; Murillo, N.; Costa, U.; Tormos, J.M.; Vidal-Samsó, J.; Valls-Solé, J.; et al. Gait training in human spinal cord injury using electromechanical systems: Effect of device type and patient characteristic. Arch. Phys. Med. Rehabil. 2012, 93, 404–412. [Google Scholar] [CrossRef]
- Meyns, P.; Van de Crommert, H.W.A.A.; Rijken, H.; Van Kuppevelt, D.H.J.M.; Duysens, J. Locomotor training with body weight support in SCI: EMG improvement is more optimally expressed at a low testing speed. Spinal Cord 2014, 52, 887–893. [Google Scholar] [CrossRef]
- Battistuzzo, C.R.; Callister, R.J.; Callister, R.; Galea, M.P. A systematic review of exercise training to promote locomotor recovery in animal models of spinal cord injury. J. Neurotrauma 2012, 29, 1600–1613. [Google Scholar] [CrossRef]
- Cassilhas, R.C.; Tufik, S.; Mello, M.T. Physical exercise, neuroplasticity, spatial learning and memory. Cell Mol. Life Sci. 2016, 73, 975–983. [Google Scholar] [CrossRef]
- Maier, I.C.; Ichiyama, R.M.; Courtine, G.; Schnell, L.; Lavrov, I.; Edgerton, V.R.; Schwab, M.E. Differential effects of anti-Nogo-A antibody treatment and treadmill training in rats with incomplete spinal cord inury. Brain 2009, 132, 1426–1440. [Google Scholar] [CrossRef]
- Tillakaratne, N.; Duru, P.; Fujino, H.; Zhong, H.; Xiao, M.S.; Edgerton, V.R.; Roy, R.R. Identification of interneurons activated at different inclines during treadmill locomotion in adult rats. J. Neurosci. Res. 2014, 92, 1714–1722. [Google Scholar] [CrossRef] [PubMed]
- Muir, G.D.; Steeves, J.D. Sensorimotor stimulation to improve locomotor recovery after spinal cord injury. Trends Neurosci. 1997, 20, 72–77. [Google Scholar] [CrossRef]
- Engessor-Cesar, C.; Ichiyama, R.M.; Nefas, A.L.; Hill, M.A.; Edgerton, V.R.; Cotman, C.W.; Anderson, A.J. Wheel running following spinal cord injury improves locomotor recovery and stimulates serotonergic fiber growth. Eur. J. Neurosci. 2007, 25, 1931–1939. [Google Scholar] [CrossRef] [PubMed]
- Lang, C.E.; MacDonald, J.R.; Gnip, C. Counting repetitions: An observational study of outpatient therapy for people with hemiparesis post-stroke. J. Neurol. Phys. Ther. 2007, 31, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Dobkin, B.; Apple, D.; Barbeau, H.; Basso, M.; Behrman, A.; Deforge, D.; Ditunno, J.; Dudley, G.; Elashoff, R.; Fugate, L.; et al. Weight-supported treadmill vs over-ground training for walking after acute incomplete SCI. Neurology 2006, 66, 484–493. [Google Scholar] [CrossRef]
- Lang, C.E.; Lohse, K.R.; Birkenmeier, R.L. Dose and timing in neurorehabilitation: Prescribing motor therapy after stroke. Curr. Opin. Neurol. 2015, 28, 549–555. [Google Scholar] [CrossRef]
- Platt, S.R.; Olby, N.J. Nursing and Rehabilitation of the neurological patient. In Canine and Feline Neurology, 3rd ed.; Platt, S.R., Olby, N.J., Eds.; British Small Animal Veterinary Association: Quedgeley, Gloucester, UK, 2004; p. 542. [Google Scholar]
- Kralj, A.; Bajd, T.; Turk, R.; Krajnik, J.; Benko, H. Gait restoration in paraplegic patients: A feasibility demonstration using multichannel surface electrode FES. J. Rehabil. R D 1983, 20, 3–20. Available online: https://pubmed.ncbi.nlm.nih.gov/6887064/ (accessed on 11 June 2021).
- Holsheimer, J. Computer modelling of spinal cord stimulation and its contribution to therapeutic efficacy. Spinal Cord 1998, 36, 531–540. [Google Scholar] [CrossRef]
- Field-Fote, E.C. Combined use of body weight support, functional electric stimulation, and treadmill training to improve walking ability in individuals with chronic incomplete spinal cord injury. Arch. Phys. Med. Rehabil. 2001, 82, 818–824. [Google Scholar] [CrossRef]
- Field-Fote, E.C.; Lindley, S.D.; Sherman, A.L. Locomotor training approaches for individuals with spinal cord injury: A preliminary report of walking-related outcomes. J. Neurol. Phys. Ther. 2005, 29, 127–137. [Google Scholar] [CrossRef]
- Field-Fote, E.C.; Roach, K.E. Influence of a locomotor training approach on walking speed and distance in people with chronic spinal cord injury: A randomized clinical trial. Phys. Ther. 2010, 91, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, N.; Masani, K.; Craven, B.C.; Giangregorio, L.M.; Hitzing, S.L.; Richards, K.; Popovic, M.R. A randomized trial of functional electrical stimulation for walking in incomplete spinal cord injury: Effects on walking competency. J. Spinal Cord Med. 2014, 37, 511–524. [Google Scholar] [CrossRef] [PubMed]
- Levine, D.; Bockstahler, B. Electrical Stimulation. In Canine Rehabilitation and Physical Therapy, 2nd ed.; Millis, D.L., Levine, D., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2014; pp. 342–358. [Google Scholar]
- Gerasimenko, Y.; Sayenko, D.; Gad, P.; Kozesnik, J.; Moshonkina, T.; Grishin, A.; Pukhoy, A.; Moiseey, S.; Gorodnichev, R.; Selionov, V.; et al. Electrical spinal stimulation, and imaging of lower limb movements to modulate brain-spinal connectomes that control locomotor-like behavior. Front. Physiol. 2018, 9, 1196. [Google Scholar] [CrossRef]
- Savin, Z.; Lejbkowicz, I.; Glass-Marmor, L.; Lavi, I.; Rosenblum, S.; Miller, A. Effect of Fampridine-PR prolonged released 4-aminopyridine) on the manual functions of patients with Multiple Sclerosis. J. Neurol. Sci. 2016, 360, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Tseng, K.C.; Li, H.; Clark, A.; Sundem, L.; Zuscik, M.; Noble, M.; Elfar, J. 4-Aminopyridine promotes functional recovery and remyelination in acute peripheral nerve injury. EMBO Mol. Med. 2016, 8, 1409–1420. [Google Scholar] [CrossRef]
- Zörner, B.; Filli, L.; Reuter, K.; Kapitza, S.; Lörincz, L.; Sutter, T.; Weller, D.; Farkas, M.; Easthope, C.S.; Czaplinski, A.; et al. Prolonged-release fampridine in multiple sclerosis: Improved ambulation effected by changes in walking pattern. Mult. Scler. J. 2016, 22, 1463–1475. [Google Scholar] [CrossRef]
- Olby, N.J.; De Risio, L.; Muñana, K.R.; Wosar, M.A.; Skeen, T.M.; Sharp, N.J.; Keene, B.W. Development of a functional scoring system in dogs with acute spinal cord injuries. Am. J. Vet. Res. 2001, 62, 1624–1628. [Google Scholar] [CrossRef]
- Dickinson, P.J.; Bannasch, D.L. Current Understanding of the Genetics of Intervertebral Disc Degeneration. Front. Vet. Sci. 2020, 7, 431. [Google Scholar] [CrossRef]
- Murphy, B.G.; Dickinson, P.; Marcellin-Little, D.J.; Batcher, K.; Raverty, S.; Bannasch, D. Pathologic Features of the Intervertebral Disc in Young NovaScotia Duck Tolling Retrievers Confirms Chondrodystrophy Degenerative Phenotype Associated with Genotype. Vet. Pathol. 2019, 566, 895–902. [Google Scholar] [CrossRef]
- Fenn, J.; Olby, N.J.; Canine Spinal Cord Injury Consortium (CANSORT-SCI). Classification of Intervertebral Disc Disease. Front. Vet. Sci. 2020, 7, 579025. [Google Scholar] [CrossRef]
- Aikawa, T.; Fujita, H.; Shibata, M.; Takahashi, T. Recurrent Thoracolumbar Intervertebral Disc Extrusion after Hemilaminectomy and Concomitant Prophylactic Fenestration in 662 Chondrodystrophic Dogs. Vet. Surg. 2012, 41, 381–390. [Google Scholar] [CrossRef]
- Gallucci, A.; Dragone, L.; Menchetti, M.; Gagliardo, T.; Pietra, M.; Cardinali, M.; Gandini, G. Acquisition of Involuntary Spinal Locomotion (Spinal Walking) in Dogs with Irreversible Thoracolumbar Spinal Cord Lesion: 81 Dogs. J. Vet. Intern. Med. 2017, 31, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Zidan, N.; Fenn, J.; Griffith, E.; Early, P.J.; Mariani, C.L.; Muñana, K.R.; Guevar, J.; Olby, N.J. The Effect of Electromagnetic Fields on Post-Operative Pain and Locomotor Recovery in Dogs with Acute, Severe Thoracolumbar Intervertebral Disc Extrusion: A Randomized Placebo-Controlled, Prospective Clinical Trial. J. Neurotrauma 2018, 35, 1726–1736. [Google Scholar] [CrossRef]
- Granger, N.; Blamires, H.; Franklin, R.J.; Jeffery, N.D. Autologous olfactory mucosal cell transplants in clinical spinal cord injury: A randomized double-blinded trial in a canine translational model. Brain 2012, 135, 3227–3237. [Google Scholar] [CrossRef] [PubMed]
- Granger, N.; Chew, D.; Fairhurst, P.; Fawcett, J.W.; Lacour, S.P.; Craggs, M.; Mosse, C.A.; Donaldson, N.; Jeffery, N.D. Use of an implanted sacral nerve stimulator to restore urine voiding in chronically paraplegic dogs. J. Vet. Intern. Med. 2013, 27, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Nishida, H.; Nakayama, M.; Tanaka, H.; Kitamura, M.; Hatoya, S.; Sugiura, K.; Suzuki, Y.; Ide, C.; Inaba, T. Evaluation of transplantation of autologous bone marrow stromal cells into the cerebrospinal fluid for treatment of chronic spinal cord injury in dogs. Am. J. Vet. Res. 2011, 72, 1118–1123. [Google Scholar] [CrossRef]
- Cizkova, D.; Murgoci, A.N.; Cubinkova, V.; Humenik, F.; Mojzisova, Z.; Maloveska, M.; Cizek, M.; Fournier, I.; Salzet, M. Spinal Cord Injury: Animal Models, Imaging Tools and the Treatment Strategies. Neurochem. Res. 2020, 45, 134–143. [Google Scholar] [CrossRef]
- Jeffery, N.; Smith, P.M.; Lakatos, A.; Ibanez, C.; Ito, D.; Franklin, R. Clinical canine spinal cord injury provides an opportunity to examine the issues in translating laboratory techniques into practical therapy. Spinal Cord 2006, 44, 584–593. [Google Scholar] [CrossRef]
- Thomas, W.B.; Olby, N.; Sharon, L. Neurologic Conditions and Physical Rehabilitation of the Neurologic Patient. In Canine Rehabilitation and Physical Therapy, 2nd ed.; Millis, D.L., Levine, D., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2014; pp. 609–627. [Google Scholar]
- Mann, F.; Wagner-Mann, C.; Dunphy, E.; Ruben, D.; Rochat, M.; Bartels, K. Recurrence rate of presumed thoracolumbar intervertebral disc disease in ambulatory dogs with spinal hyperpathia treated with anti-inflammatory drugs: 78 cases (1997–2000). J. Vet. Emerg. Crit. Care 2007, 17, 53–60. [Google Scholar] [CrossRef]
- English, A.W.; Wilhelm, J.C.; Ward, P.J. Exercise, neurotrophins, and axon regeneration in the PNS. Physiology 2014, 29, 437–445. [Google Scholar] [CrossRef] [PubMed]
- López-Álvarez, V.M.; Modol, L.; Navarro, X.; Cobianchi, S. Early increasing-intensity treadmill exercise reduces neuropathic pain by preventing nociceptor collateral sprouting and disruption of chloride cotransporters homeostasis after peripheral nerve injury. Pain 2015, 156, 1812–1825. [Google Scholar] [CrossRef] [PubMed]
- Knikou, M.; Mummidisetty, C.K. Locomotor training improves premotoneuronal control after chronic spinal cord injury. J. Neurophysiol. 2014, 111, 2264–2275. [Google Scholar] [CrossRef]
- Rossignol, S.; Frigon, A. Recovery of locomotion after spinal cord injury: Some facts and mechanisms. Annu. Rev. Neurosci. 2011, 34, 413–440. [Google Scholar] [CrossRef] [PubMed]
- MacKay-Lyons, M. Central pattern generation of locomotion: A review of the evidence. Phys. Ther. 2002, 82, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.R.; Harkema, S.J.; Edgerton, V.R. Basic concepts of activity-based interventions for improved recovery of motor function after spinal cord injury. Arch. Phys. Med. Rehabil. 2012, 93, 1487–1497. [Google Scholar] [CrossRef]
- Chiquoine, J.; McCauley, L.; Van Dyke, J.B. Aquatic Therapy. In Canine Sports Medicine and Rehabilitation, 1st ed.; Zink, M.C., Van Dyke, J.B., Eds.; Wiley-Blackwell: Ames, IA, USA, 2013; pp. 158–175. [Google Scholar]
- Dragone, L. Idroterapia. In Fisioterapia Riabilitativa del Cane e del Gatto, 1st ed.; Mutignani, A., Leschiera, P., Eds.; Elsevier Srl: Milano, Italy, 2011; pp. 57–75. [Google Scholar]
- Hernández, M.P. Hidrocinesioterapia. In Manual de Fisioterapia en Pequeños Animales, 1st ed.; Martin, F.M., Ed.; Multimédica Ediciones Veterinarias: Barcelona, España, 2014; pp. 394–424. [Google Scholar]
- Diniz, R. Hidroterapia. In Fisiatria em Pequenos Animais, 1st ed.; Lopes, R.S., Diniz, R., Eds.; Editora Inteligente: São Paulo, Brasil, 2018; pp. 156–162. [Google Scholar]
- Languth, H.W.; Teasdall, R.D.; Magladery, J.W. Electrophysiological studies of reflex activity in patients with lesions of the nervous system. III. Motoneuron excitability following afferent nerve volleys in patients with rostrally adjacent spinal cord damage. Bull. Johns Hopkins Hosp. 1952, 9, 1257–1266. [Google Scholar]
- Magladery, J.W.; Teadsdall, R.D. Stretch reflexes in patients with spinal cord lesions. Bull. Johns Hopkins Hosp. 1958, 103, 236–241. [Google Scholar]
- Blauch, B. Spinal reflex walking in the dog. Vet. Med. Small Anim. Clin. 1977, 72, 169–173. [Google Scholar]
- Mcbride, W.J.; Hall, P.V.; Chernet, E.; Patrick, J.T.; Shapiro, S. Alterations of Amino Acid Transmitter Systems in Spinal Cords of Chronic Paraplegic Dogs. J. Neurochem. 1984, 42, 1625–1631. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Maeta, N. Efficacy of autologous bone marrow mononuclear cell transplantation in dogs with chronic spinal cord injury. Open Vet. J. 2020, 10, 206–215. [Google Scholar] [CrossRef]
- Hu, R.; Zhou, J.; Luo, C.; Lin, J.; Wang, X.; Li, X.; Bian, X.; Li, Y.; Wan, Q.; Yu, Y.; et al. Glial scar and neuroregeneration: Histological, functional, and magnetic resonance imaging analysis in chronic spinal cord injury. J. Neurosurg. Spine 2010, 13, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.A.; Zidan, N.; Spitzbarth, I.; Nout-Lomas, Y.S.; Granger, N.; da Costa, R.C.; Levine, J.M.; Jeffery, N.D.; Stein, V.M.; Tipold, A.; et al. Development of an International Canine Spinal Cord Injury observational registry: A collaborative data-sharing network to optimize translational studies of SCI. Spinal Cord 2018, 56, 656–665. [Google Scholar] [CrossRef]
- Hu, H.Z.; Jeffery, N.D.; Granger, N. Somatosensory and motor evoked potentials in dogs with chronic severe thoracolumbar spinal cord injury. Vet. J. 2018, 237, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijevic, M.R.; Hsu, C.Y.; McKay, W.B. Neurophysiological assessment of spinal cord and head injury. J. Neurotrauma 1992, 9, S293–S300. [Google Scholar] [PubMed]
- Courtine, G.; Song, B.; Roy, R.R.; Zhong, H.; Herrmann, J.E.; Ao, Y.; Qi, J.; Edgerton, V.R.; Sofroniew, M.V. Recovery of supraspinal control of stepping via indirect propriospinal relay connections after spinal cord injury. Nat. Med. 2008, 14, 69–74. [Google Scholar] [CrossRef]
- Van Den Brand, R.; Heutschi, J.; Barraud, Q.; DiGiovanna, J.; Bartholdi, K.; Huerlimann, M.; Friedli, L.; Vollenweider, I.; Moraud, E.M.; Duis, S. Restoring voluntary control of locomotion after paralyzing spinal cord injury. Science 2012, 336, 1182–1185. [Google Scholar] [CrossRef]
- Filli, L.; Engmann, A.K.; Zorner, B.; Weinmann, O.; Moraitis, T.; Gullo, M.; Kasper, H.; Schenider, R.; Schwab, M.E. Bridging the gap: A reticulo-propriospinal detour bypassing an in- complete spinal cord injury. J. Neurosci. 2014, 34, 13399–13410. [Google Scholar] [CrossRef]
- Hollis, E.R.; Ishiko, N.; Pessian, M.; Tolentino, K.; Lee-Kubli, C.A.; Calcutt, N.A.; Zou, Y. Remodelling of spared proprioceptive circuit involving a small number of neurons supports functional recovery. Nat. Commun. 2015, 6, 6076–6079. [Google Scholar] [CrossRef]
- Lovely, R.G.; Gregor, R.J.; Roy, R.R.; Edgerton, V.R. Effects of training on the recovery of full-weight-bearing stepping in the adult spinal cat. Exp. Neurol. 1986, 92, 421–435. [Google Scholar] [CrossRef]
- Barbeau, H.; Rossignol, S. Recovery of locomotion after chronic spinalization in the adult cat. Brain Res. 1987, 412, 84–95. [Google Scholar] [CrossRef]
- Lavrov, I.; Gerasimenko, Y.P.; Ichiyama, R.M.; Courtine, G.; Zhong, H.; Roy, R.R.; Edgerton, V.R. Plasticity of spinal cord reflexes after a complete transection in adult rats: Relationship to stepping ability. J. Neurophysiol. 2006, 96, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Gerasimenko, Y.; Gorodnichev, R.; Machueva, E.; Pivovarova, E.; Semyenov, D.; Savochin, A.; Roy, R.R.; Edgerton, V.R. Novel and direct access to the human locomotor spinal circuitry. J. Neurosci. 2010, 30, 3700–3708. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S.; Dubuc, R.; Gossard, J.P. Dynamic sensorimotor interactions in locomotion. Physiol. Rev. 2006, 86, 89–154. [Google Scholar] [CrossRef] [PubMed]
- Escalhão, C.C.M.; Ramos, I.P.R.; Hochman-Mendez, C.; Kasai-Brunswick, T.H.; Souza, S.A.L.; Gutfilen, B.; Goldenberg, R.C.D.S.; Coelho-Sampaio, T. Safety of Allogeneic Canine Adipose Tissue-Derived Mesenchymal Stem Cell Intraspinal Transplantation in Dogs with Chronic Spinal Cord Injury. Stem Cells Int. 2017, 2017, 3053759. [Google Scholar] [CrossRef]
- Martin, D.; Robe, P.; Franzen, R.; Delree, P.; Schoenen, J.; Stevenaert, A.; Moonen, G. Effects of Schwann cell transplantation in a contusion model of ratspinal cord injury. J. Neurosci. Res. 1996, 45, 588–597. [Google Scholar] [CrossRef]
- McDonald, J.W.; Liu, X.Z.; Qu, Y.; Liu, S.; Mickey, S.K.; Turetsky, D.; Gottlieb, D.I.; Choi, D.W. Transplanted embryonic stem cells survive, differentiate and promote recovery in injured rat spinal cord. Nat. Med. 1999, 5, 1410–1412. [Google Scholar] [CrossRef]
- Knoller, N.; Auerbach, G.; Fulga, V.; Zelig, G.; Attias, J.; Bakimer, R.; Marder, J.B.; Yoles, E.; Belkin, M.; Schwartz, M.; et al. Clinical experience using incubated autologous macrophages as a treatment for complete spinal cord injury: Phase I study results. Clin. Trial 2005, 3, 173–181. [Google Scholar] [CrossRef]
- Ogawa, M.; Uchida, K.; Yamato, O.; Mizukami, K.; Chambers, J.K.; Nakayama, H. Expression of Autophagy-Related Proteins in the Spinal Cord of Pembroke Welsh Corgi Dogs With Canine Degenerative Myelopathy. Vet. Pathol. 2015, 52, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, O.; Miura, K.; Okada, Y.; Fujiyoshi, K.; Mukaino, M.; Nagoshi, N.; Kitamura, K.; Kumagai, G.; Nishino, M.; Tomisato, S.; et al. Therapeutic potential of appropriately evaluated safe-induced pluripotent stem cells for spinal cord injury. Proc. Natl. Acad. Sci. USA 2010, 107, 12704–12709. [Google Scholar] [CrossRef]
- Dissanayaka, W.L.; Zhu, X.; Zhang, C.; Jin, L. Characterization of dental pulp stem cells isolated from canine premolars. J. Endod. 2011, 37, 1074–1080. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Yan, Q.; Ruan, J.W.; Zhang, Y.Q.; Li, W.J.; Zhang, Y.J.; Li, Y.; Dong, H.; Zeng, Y.S. Electro-acupuncture promotes survival, differentiation of the bone marrow mesenchymal stem cells as well as functional recovery in the spinal cord-transected rats. BMC Neurosci. 2009, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Ruan, J.W.; Ding, Y.; Li, W.J.; Li, Y.; Zeng, Y.S. Electro-acupuncture promotes differentiation of mesenchymal stem cells, regeneration of nerve fibers and partial functional recovery after spinal cord injury. Exp. Toxicol. Pathol. 2011, 63, 151–156. [Google Scholar] [CrossRef]
- Prado, C.; Fratini, P.; Matias, G.S.; Bocabello, R.Z.; Monteiro, J.; Santos, C.J.; Joaquim, J.G.; Giglio, R.F.; Possebon, F.S.; Sakata, S.H.; et al. Combination of stem cells from deciduous teeth and electroacupunture for therapy in dogs with chronic spinal cord injury: A pilot study. Res. Vet. Sci. 2019, 123, 247–251. [Google Scholar] [CrossRef]
- Côté, M.P.; Ménard, A.; Gossard, J.P. Spinal cats on the treadmill: Changes in load pathways. J. Neurosci. 2003, 23, 2789–2796. [Google Scholar] [CrossRef] [PubMed]
- Côté, M.P.; Gossard, J.P. Step training-dependent plasticity in spinal cutaneous pathways. J. Neurosci. 2004, 24, 11317–11327. [Google Scholar] [CrossRef]
- Martinez, M.; Brezun, J.M.; Zennou-Azogui, Y.; Baril, N.; Xerri, C. Sensorimotor training promotes functional recovery and somatosensory cortical map reactivation following cervical spinal cord injury. Eur. J. Neurosci. 2009, 30, 2356–2367. [Google Scholar] [CrossRef] [PubMed]
- Bregman, B.S. Regeneration in the spinal cord. Curr. Opin. Neurobiol. 1998, 8, 800–807. [Google Scholar] [CrossRef]
- Thomas, S.L.; Gorassini, M.A. Increases in corticospinal tract function by treadmill training after incomplete spinal cord injury. J. Neurophysiol. 2005, 94, 2844–2855. [Google Scholar] [CrossRef]
- Olby, N.J.; da Costa, R.C.; Levine, J.M.; Stein, V.M.; The Canine Spinal Cord Injury Consortium (CANSORT SCI). Prognostic Factors in Canine Acute Intervertebral Disc Disease. Front. Vet. Sci. 2020, 7, 596059. [Google Scholar] [CrossRef]
- Duval, J.; Dewey, C.; Roberts, R.; Aron, D. Spinal cord swelling as a myelographic indicator of prognosis: A retrospective study in dogs with intervertebral disc disease and loss of deep pain perception. Vet. Surg. 1996, 25, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.W. Hemilaminectomy for the treatment of thoracolumbar disc disease in the dog: A follow-up study of 40 cases. J. Small Anim. Pract. 1997, 38, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.W.; McKee, W.M. Laminectomy for 34 dogs with thoracolumbar intervertebral disc disease and loss of deep pain perception. J. Small Anim. Pract. 1999, 40, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; Matsunaga, S.; Jeffery, N.D.; Sasaki, N.; Nishimura, R.; Mochizuki, M.; Kasahara, M.; Fujiwara, R.; Ogawa, H. Prognostic value of magnetic resonance imaging in dogs with paraplegia caused by thoracolumbar intervertebral disk extrusion: 77 cases (2000–2003). J. Am. Vet. Med. Assoc. 2005, 227, 1454–1460. [Google Scholar] [CrossRef]
- Tamura, K.; Harada, Y.; Nagashima, N.; Itoi, T.; Ishino, H.; Yogo, T.; Nezu, Y.; Hara, Y.; Suzuki, Y.; Ide, C.; et al. Autotransplanting of Bone Marrow-Derived Mononuclear Cells for Complete Cases of Canine Paraplegia and Loss of Pain Perception, Secondary to Intervertebral Disc Herniation. Exp. Clin. Transpl. 2012, 10, 263–272. [Google Scholar] [CrossRef] [PubMed][Green Version]
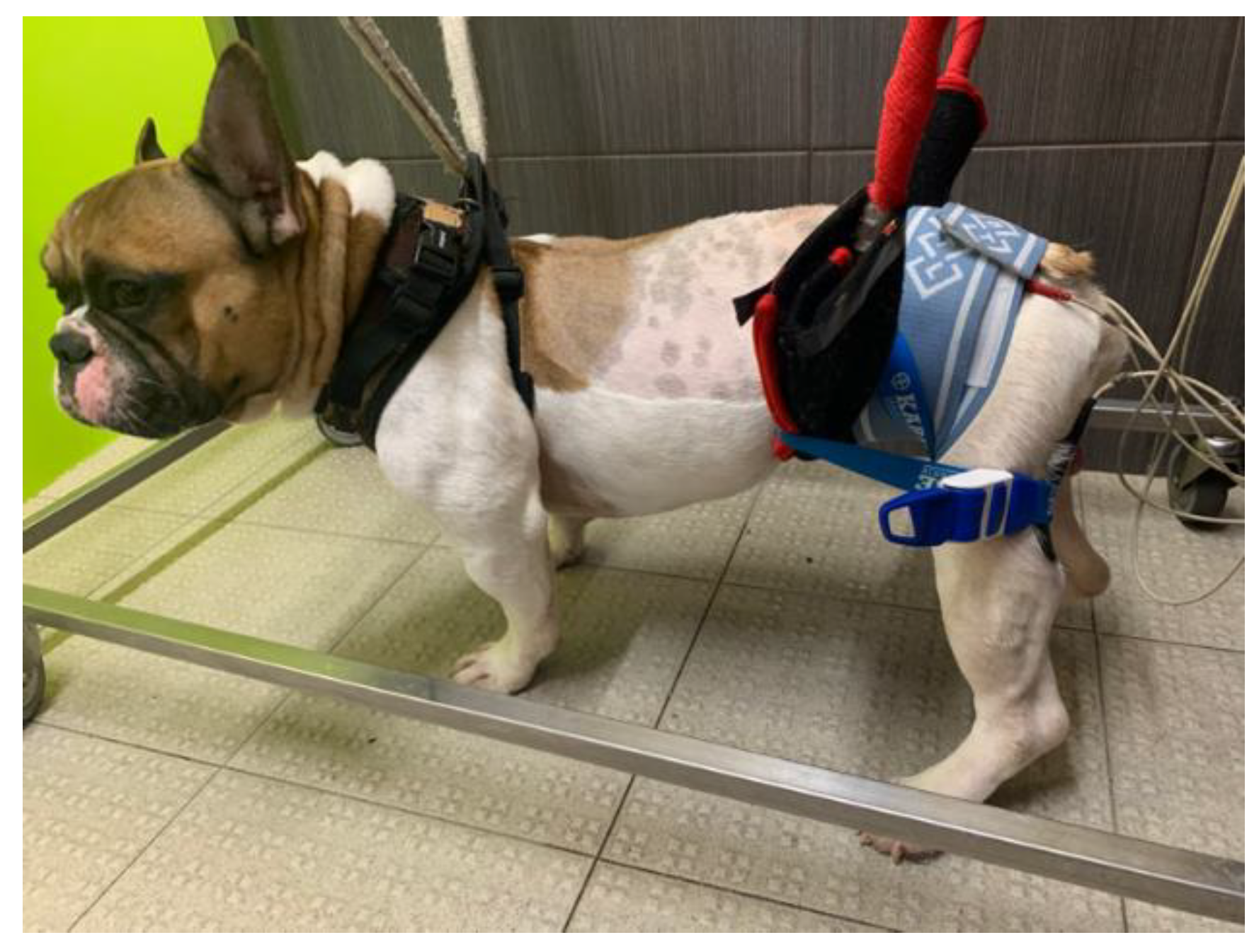

| Protocol | Description | Frequency |
|---|---|---|
| A | 30 min land treadmill | 3 days/week |
| B | 1 h leash ground walk | 4 days/week |
| C | 30 min underwater treadmill | 1–2 days/week |
| Week | Time Duration | Walking Speed | Frequency | Slope |
|---|---|---|---|---|
| 1st–2nd | Up to 5–10 min | 0.8–1.9 km/h | 4–6 times/day 6 days/week | - |
| 3rd–4th | Up to 20 min | 2 km/h | 2–4 times/day 6 days/week | - |
| 5th–6th | Up to 30 min | 2.2 km/h | 2–3 times/day 6 days/week | - |
| 7th–8th | Up to 40 min | 2.5 km/h | 2 times/day 6 days/week | 5° |
| 9th–10th | Up to 40 min | 2.5 km/h | 2 times/day 5 days/week | 10° |
| 11th–12th | Up to 40 min | 2.5 km/h | 1 time/day 5 days/week | 25° |
| Week | Time Duration | Walking Speed | Slope |
|---|---|---|---|
| 1st–2nd | Up to 5–10 min | 1–1.2 km/h | - |
| 3rd–4th | Up to 10–20 min | 1.8–2 km/h | - |
| 5th–6th | Up to 30 min | 2–2.5 km/h | 5° |
| 7th–8th | Up to 40 min | 2.8–3 km/h | 5° |
| 9th–10th | Up to 40 min | 3–3.5 km/h | 5° |
| 11th–12th | Up to 60 min | 3.5 km/h | 10° |
| Week | Exercise | Time Duration | Frequency | |
|---|---|---|---|---|
| 1st–4th | Postural standing | Until 10 min | 2–4 times/day 6 days/week | |
| Flexor movements | 10–20 repetitions | 2–3 times/day 5 days/week | ||
| Bicycle movements | 5–10 repetitions | |||
| 5th–8th | Dog with active postural standing | Different floors gait stimulation | 3–5 min | 2–3 times/day 5 days/week |
| Dog without active postural standing | Postural standing | 10 min | ||
| Different floors gait stimulation | 3–5 min | |||
| 9th–12th | Different floors gait stimulation | 5–10 min | 2 times/day 5 days/week | |
| Balance board | 5 min | |||
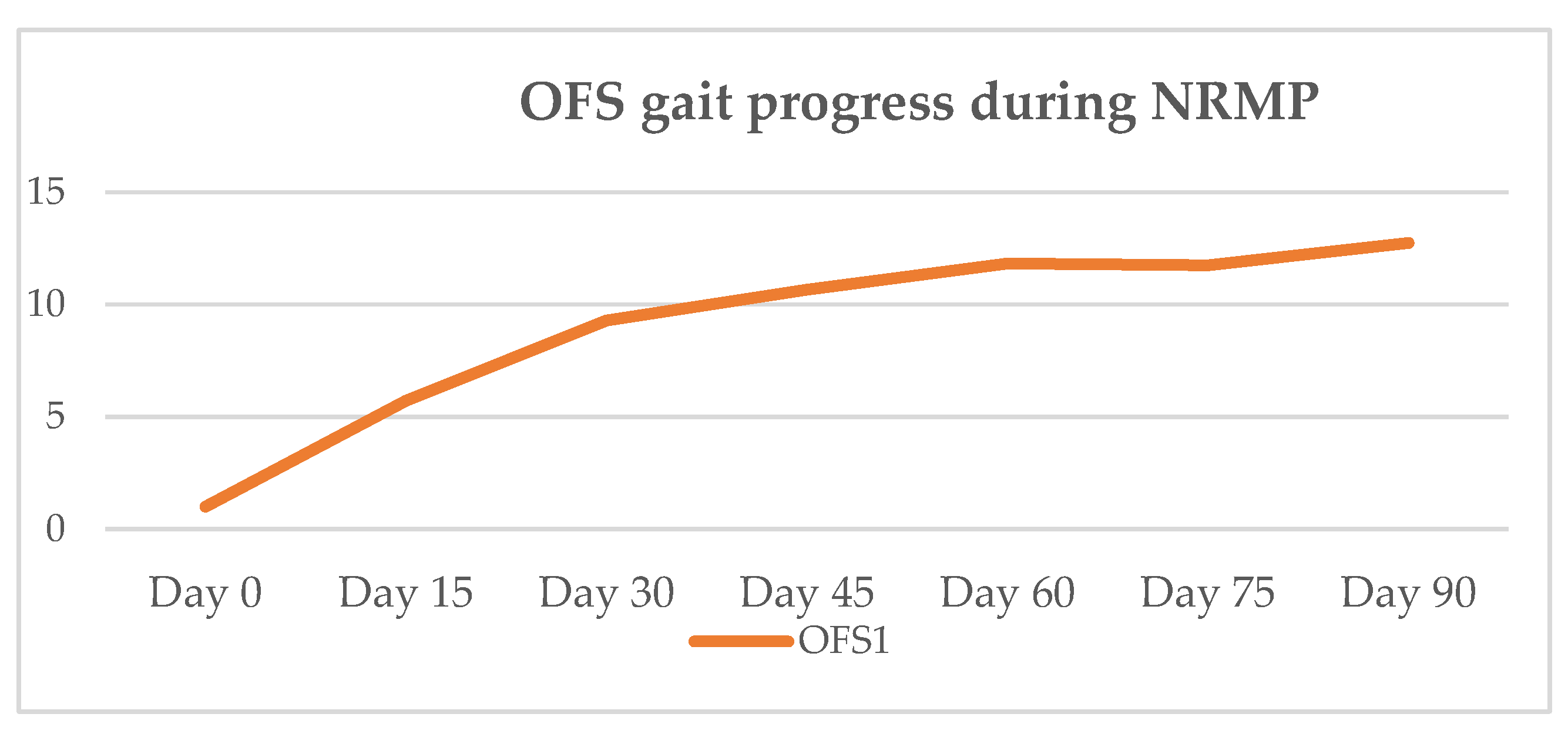
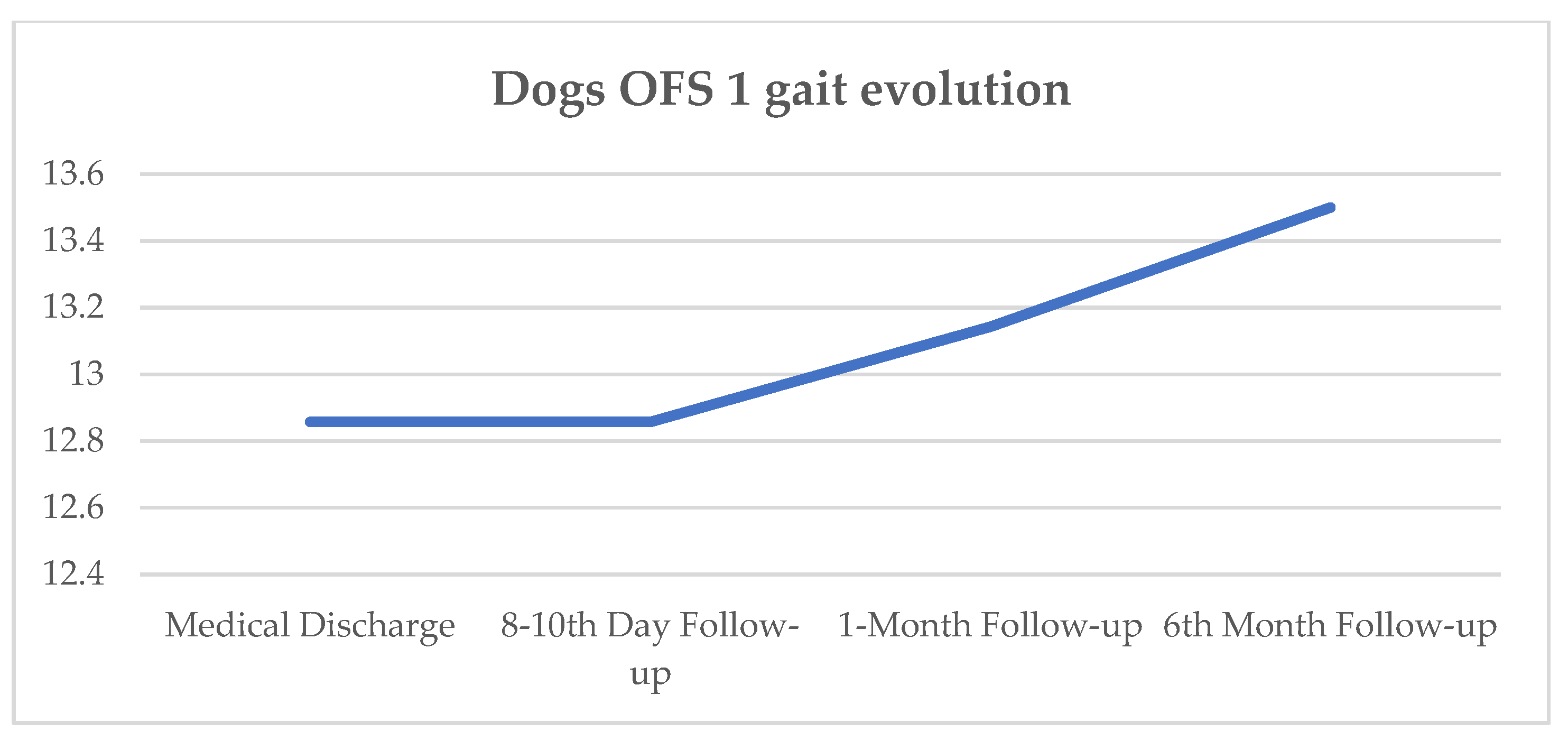
| DPP+ Dogs ID | Day 0 | Day 15 | Day 30 | Day 45 | Day 60 | Day 75 | Day 90 | OFS Medical Discharge | 8–10-Day Follow-Up | 1-Month Follow-Up | 6-Month Follow-Up |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 3 | 7 | 10 | 11 |  | 13 | 13 | 13 | 13 | 14 |
| 2 | 1 | 4 | 9 |  | 13 | 13 | 13 | 14 | 14 | ||
| 3 | 1 | 3 | 7 | 10 | 11 |  | 13 | 13 | 13 | 13 | 14 |
| 4 | 1 | 7 |  | 13 | 13 | 13 | 13 | ||||
| 5 | 1 | 7 | 9 | 11 | 12 |  | 13 | 13 | 13 | 13 | 13 |
| 6 | 1 | 9 | 11 |  | 13 | 13 | 13 | 14 | |||
| 7 | 1 | 7 | 9 | 10 | 11 |  | 12 | 12 | 12 | 12 | 13 |
| OFS Mean | 1 | 5.7 | 9.3 | 10.7 | 11.8 | 11.8 | 12.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, Â.; Gouveia, D.; Cardoso, A.; Carvalho, C.; Silva, C.; Coelho, T.; Gamboa, Ó.; Ferreira, A. Functional Neurorehabilitation in Dogs with an Incomplete Recovery 3 Months following Intervertebral Disc Surgery: A Case Series. Animals 2021, 11, 2442. https://doi.org/10.3390/ani11082442
Martins Â, Gouveia D, Cardoso A, Carvalho C, Silva C, Coelho T, Gamboa Ó, Ferreira A. Functional Neurorehabilitation in Dogs with an Incomplete Recovery 3 Months following Intervertebral Disc Surgery: A Case Series. Animals. 2021; 11(8):2442. https://doi.org/10.3390/ani11082442
Chicago/Turabian StyleMartins, Ângela, Débora Gouveia, Ana Cardoso, Carla Carvalho, Cátia Silva, Tiago Coelho, Óscar Gamboa, and António Ferreira. 2021. "Functional Neurorehabilitation in Dogs with an Incomplete Recovery 3 Months following Intervertebral Disc Surgery: A Case Series" Animals 11, no. 8: 2442. https://doi.org/10.3390/ani11082442
APA StyleMartins, Â., Gouveia, D., Cardoso, A., Carvalho, C., Silva, C., Coelho, T., Gamboa, Ó., & Ferreira, A. (2021). Functional Neurorehabilitation in Dogs with an Incomplete Recovery 3 Months following Intervertebral Disc Surgery: A Case Series. Animals, 11(8), 2442. https://doi.org/10.3390/ani11082442





