Impact of Marker Pruning Strategies Based on Different Measurements of Marker Distance on Genomic Prediction in Dairy Cattle
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. Measurements of Marker Distance
2.3. Genomic BLUP Model
2.4. Model Assessment
2.5. Measurements of Marker Density
3. Results
3.1. Measurements of Marker Density for Different Panels
3.2. Accuracies of Genomic Prediction
3.3. Variance Component Estimates
3.4. Relationships between Measurements of Marker Density and GEBV Accuracy
4. Discussion
4.1. Relationship between Measurement of “Marker Density” and GP Performance
4.2. Development of Cost-Effective Panels
4.3. Pruning Strategies of High-Density SNP Data in Genome prediction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Bolormaa, S.; Gore, K.; van der Werf, J.H.; Hayes, B.J.; Daetwyler, H.D. Design of a low-density SNP chip for the main Australian sheep breeds and its effect on imputation and genomic prediction accuracy. Anim. Genet. 2015, 46, 544–556. [Google Scholar] [CrossRef]
- Meuwissen, T.; Hayes, B.; Goddard, M. Genomic selection: A paradigm shift in animal breeding. Anim. Front. 2016, 6, 6–14. [Google Scholar] [CrossRef] [Green Version]
- Ballesta, P.; Bush, D.; Silva, F.F.; Mora, F. Genomic predictions using low-density SNP markers, pedigree and GWAS information: A case study with the non-model species Eucalyptus cladocalyx. Plants 2020, 9, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Werner, C.R.; Voss-Fels, K.P.; Miller, C.N.; Qian, W.; Hua, W.; Guan, C.Y.; Snowdon, R.J.; Qian, L. Effective genomic selection in a narrow-genepool crop with low-density markers: Asian rapeseed as an example. Plant Genome 2018, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kriaridou, C.; Tsairidou, S.; Houston, R.D.; Robledo, D. Genomic prediction using low density marker panels in aquaculture: Performance across species, traits, and genotyping platforms. Front. Genet. 2020, 11, 124. [Google Scholar] [CrossRef] [PubMed]
- Tsairidou, S.; Hamilton, A.; Robledo, D.; Bron, J.E.; Houston, R.D. Optimizing low-cost genotyping and imputation strategies for genomic selection in Atlantic salmon. G3 2020, 10, 581–590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daetwyler, H.D.; Capitan, A.; Pausch, H.; Stothard, P.; van Binsbergen, R.; Brondum, R.F.; Liao, X.; Djari, A.; Rodriguez, S.C.; Grohs, C.; et al. Whole-genome sequencing of 234 bulls facilitates mapping of monogenic and complex traits in cattle. Nat. Genet. 2014, 46, 858–865. [Google Scholar] [CrossRef]
- Ober, U.; Ayroles, J.F.; Stone, E.A.; Richards, S.; Zhu, D.; Gibbs, R.A.; Stricker, C.; Gianola, D.; Schlather, M.; Mackay, T.F.; et al. Using whole-genome sequence data to predict quantitative trait phenotypes in Drosophila melanogaster. PLoS Genet. 2012, 8, e1002685. [Google Scholar] [CrossRef] [Green Version]
- Speed, D.; Holmes, J.; Balding, D.J. Evaluating and improving heritability models using summary statistics. Nat. Genet. 2020, 52, 458–462. [Google Scholar] [CrossRef]
- Liu, X.; Huang, M.; Fan, B.; Buckler, E.S.; Zhang, Z. Iterative Usage of Fixed and Random Effect Models for Powerful and Efficient Genome-Wide Association Studies. PLoS Genet. 2016, 12, e1005767. [Google Scholar] [CrossRef]
- Habier, D.; Fernando, R.L.; Dekkers, J.C. Genomic selection using low-density marker panels. Genetics 2009, 182, 343–353. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Habier, D.; Peiris, B.L.; Wolc, A.; Kranis, A.; Watson, K.A.; Avendano, S.; Garrick, D.J.; Fernando, R.L.; Lamont, S.J.; et al. Accuracy of genomic prediction using an evenly spaced, low-density single nucleotide polymorphism panel in broiler chickens. Poult. Sci. 2013, 92, 1712–1723. [Google Scholar] [CrossRef]
- Brondum, R.F.; Su, G.; Janss, L.; Sahana, G.; Guldbrandtsen, B.; Boichard, D.; Lund, M.S. Quantitative trait loci markers derived from whole genome sequence data increases the reliability of genomic prediction. J. Dairy Sci. 2015, 98, 4107–4116. [Google Scholar] [CrossRef] [Green Version]
- Lopes, F.B.; Wu, X.L.; Li, H.; Xu, J.; Perkins, T.; Genho, J.; Ferretti, R.; Tait, R.G., Jr.; Bauck, S.; Rosa, G.J.M. Improving accuracy of genomic prediction in Brangus cattle by adding animals with imputed low-density SNP genotypes. J. Anim. Breed. Genet. 2018, 135, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Sargolzaei, M.; Chesnais, J.P.; Schenkel1, F.S. A new approach for efficient genotype imputation using information from relatives. BMC Genom. 2014, 15, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Browning, S.R.; Browning, B.L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 2007, 81, 1084–1097. [Google Scholar] [CrossRef] [Green Version]
- Hickey, J.M.; Kinghorn, B.P.; Tier, B.; van der Werf, J.H.; Cleveland, M.A. A phasing and imputation method for pedigreed populations that results in a single-stage genomic evaluation. Genet. Sel. Evol. 2012, 44, 9. [Google Scholar] [CrossRef] [Green Version]
- Aliloo, H.; Mrode, R.; Okeyo, A.M.; Ni, G.; Goddard, M.E.; Gibson, J.P. The feasibility of using low-density marker panels for genotype imputation and genomic prediction of crossbred dairy cattle of East Africa. J. Dairy Sci. 2018, 101, 9108–9127. [Google Scholar] [CrossRef]
- Wu, X.L.; Xu, J.; Feng, G.; Wiggans, G.R.; Taylor, J.F.; He, J.; Qian, C.; Qiu, J.; Simpson, B.; Walker, J.; et al. Optimal design of low-density SNP arrays for genomic prediction: Algorithm and applications. PLoS ONE 2016, 11, e0161719. [Google Scholar] [CrossRef]
- Berry, D.P.; Kearney, J.F. Imputation of genotypes from low- to high-density genotyping platforms and implications for genomic selection. Animal 2011, 5, 1162–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Ding, X.; Liu, J.; Zhang, Q.; de Koning, D.J. Accuracy of genomic prediction using low-density marker panels. J. Dairy Sci. 2011, 94, 3642–3650. [Google Scholar] [CrossRef] [Green Version]
- Grossi, D.A.; Brito, L.F.; Jafarikia, M.; Schenkel, F.S.; Feng, Z. Genotype imputation from various low-density SNP panels and its impact on accuracy of genomic breeding values in pigs. Animal 2018, 12, 2235–2245. [Google Scholar] [CrossRef]
- Teng, J.; Huang, S.; Chen, Z.; Gao, N.; Ye, S.; Diao, S.; Ding, X.; Yuan, X.; Zhang, H.; Li, J.; et al. Optimizing genomic prediction model given causal genes in a dairy cattle population. J. Dairy Sci. 2020, 103, 10299–10310. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Gao, N.; Zheng, R.; Chen, Z.; Teng, J.; Yuan, X.; Zhang, H.; Chen, Z.; Zhang, X.; Li, J.; et al. Strategies for obtaining and pruning imputed whole-genome sequence data for genomic prediction. Front. Genet. 2019, 10, 673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Yin, L.; Wang, M.; Yuan, X.; Liu, X. Factors affecting the accuracy of genomic selection for agricultural economic traits in maize, cattle, and pig populations. Front. Genet. 2019, 10, 189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brøndum, R.F.; Guldbrandtsen, B.; Sahana, G. Strategies for imputation to whole genome sequence using a single or multi-breed reference population in cattle. BMC Genom. 2014, 15, 728. [Google Scholar] [CrossRef] [Green Version]
- Druet, T.; Macleod, I.; Hayes, B. Toward genomic prediction from whole-genome sequence data: Impact of sequencing design on genotype imputation and accuracy of predictions. Heredity 2014, 112, 39–47. [Google Scholar] [CrossRef] [Green Version]
- van den Berg, I.; Boichard, D.; Lund, M.S. Sequence variants selected from a multi-breed GWAS can improve the reliability of genomic predictions in dairy cattle. Genet. Sel. Evol. 2016, 48, 83. [Google Scholar] [CrossRef] [Green Version]
- Perez-Enciso, M.; Forneris, N.; de Los Campos, G.; Legarra, A. Evaluating sequence-based genomic prediction with an efficient new simulator. Genetics 2017, 205, 939–953. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Yu, Y.; Yuan, J.; Zhang, X.; Huang, H.; Li, F.; Xiang, J. Effects of marker density and population structure on the genomic prediction accuracy for growth trait in Pacific white shrimp Litopenaeus vannamei. BMC Genet. 2017, 18, 45. [Google Scholar] [CrossRef]
- Herry, F.; Hérault, F.; Druet, D.P.; Varenne, A.; Allais, S. Design of a low density SNP chip for genotype imputation in layer chickens. BMC Genet. 2018, 19, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Ober, U.; Erbe, M.; Zhang, H.; Gao, N.; He, J.; Li, J.; Simianer, H. Improving the accuracy of whole genome prediction for complex traits using the results of genome wide association studies. PLoS ONE 2014, 9, e93017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matukumalli, L.K.; Lawley, C.T.; Schnabel, R.D.; Taylor, J.F.; Allan, M.F.; Heaton, M.P.; O’Connell, J.; Moore, S.S.; Smith, T.P.; Sonstegard, T.S.; et al. Development and characterization of a high density SNP genotyping assay for cattle. PLoS ONE 2009, 4, e5350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.; Daly, M.J.; et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misztal, I.; Tsuruta, S.; Strabel, T.; Auvray, B.; Druet, T.; Lee, D.H. BLUPF90 and related programs (BGF90). In Proceedings of the 7th World Congress on Genetics Applied to Livestock Production, Montpellier, France, 19–23 August 2002; p. 2. [Google Scholar]
- VanRaden, P.M. Efficient methods to compute genomic predictions. J. Dairy Sci. 2008, 91, 4414–4423. [Google Scholar] [CrossRef] [Green Version]
- Grisart, B.; Coppieters, W.; Farnir, F.; Karim, L.; Ford, C.; Berzi, P.; Cambisano, N.; Mni, M.; Reid, S.; Simon, P.; et al. Positional candidate cloning of a QTL in dairy cattle: Identification of a missense mutation in the bovine DGAT1 gene with major effect on milk yield and composition. Genome Res. 2002, 12, 222–231. [Google Scholar] [CrossRef] [Green Version]
- Gutierrez, A.P.; Matika, O.; Bean, T.P.; Houston, R.D. Genomic selection for growth traits in pacific oyster (crassostrea gigas): Potential of low-density marker panels for breeding value prediction. Front. Genet. 2018, 9, 391. [Google Scholar] [CrossRef] [Green Version]
- Meuwissen, T.H.; Hayes, B.J.; Goddard, M.E. Prediction of total genetic value using genome-wide dense marker maps. Genetics 2001, 157, 1819–1829. [Google Scholar] [CrossRef]
- Calus, M.P.L.; Veerkamp, R.F. Accuracy of breeding values when using and ignoring the polygenic effect in genomic breeding value estimation with a marker density of one SNP per cM. J. Anim. Breed. Genet. 2007, 124, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Liang, W.; Xu, G.; Huang, B.; Zhang, X.; Hu, C.Y.; Wang, C. Accuracy of genomic prediction using mixed low-density marker panels. Anim. Prod. Sci. 2020, 60, 999–1007. [Google Scholar] [CrossRef]
- Speed, D.; Hemani, G.; Johnson, M.R.; Balding, D.J. Improved Heritability Estimation from Genome-wide SNPs. Am. J. Hum. Genet. 2012, 91, 1011–1021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulder, H.A.; Crump, R.E.; Calus, M.P.L.; Veerkamp, R.F. Unraveling the genetic architecture of environmental variance of somatic cell score using high-density single nucleotide polymorphism and cow data from experimental farms. J. Dairy Sci. 2013, 96, 7306–7317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Erbe, M.; He, J.; Ober, U.; Gao, N.; Zhang, H.; Simianer, H.; Li, J. Accuracy of whole-genome prediction using a genetic architecture-enhanced variance-covariance matrix. G3 2015, 5, 615–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, D.; An, L.; Li, B.; Qiao, L.; Liu, W. Efficient weighting methods for genomic best linear-unbiased prediction (BLUP) adapted to the genetic architectures of quantitative traits. Heredity 2021, 126, 320–334. [Google Scholar] [CrossRef] [PubMed]

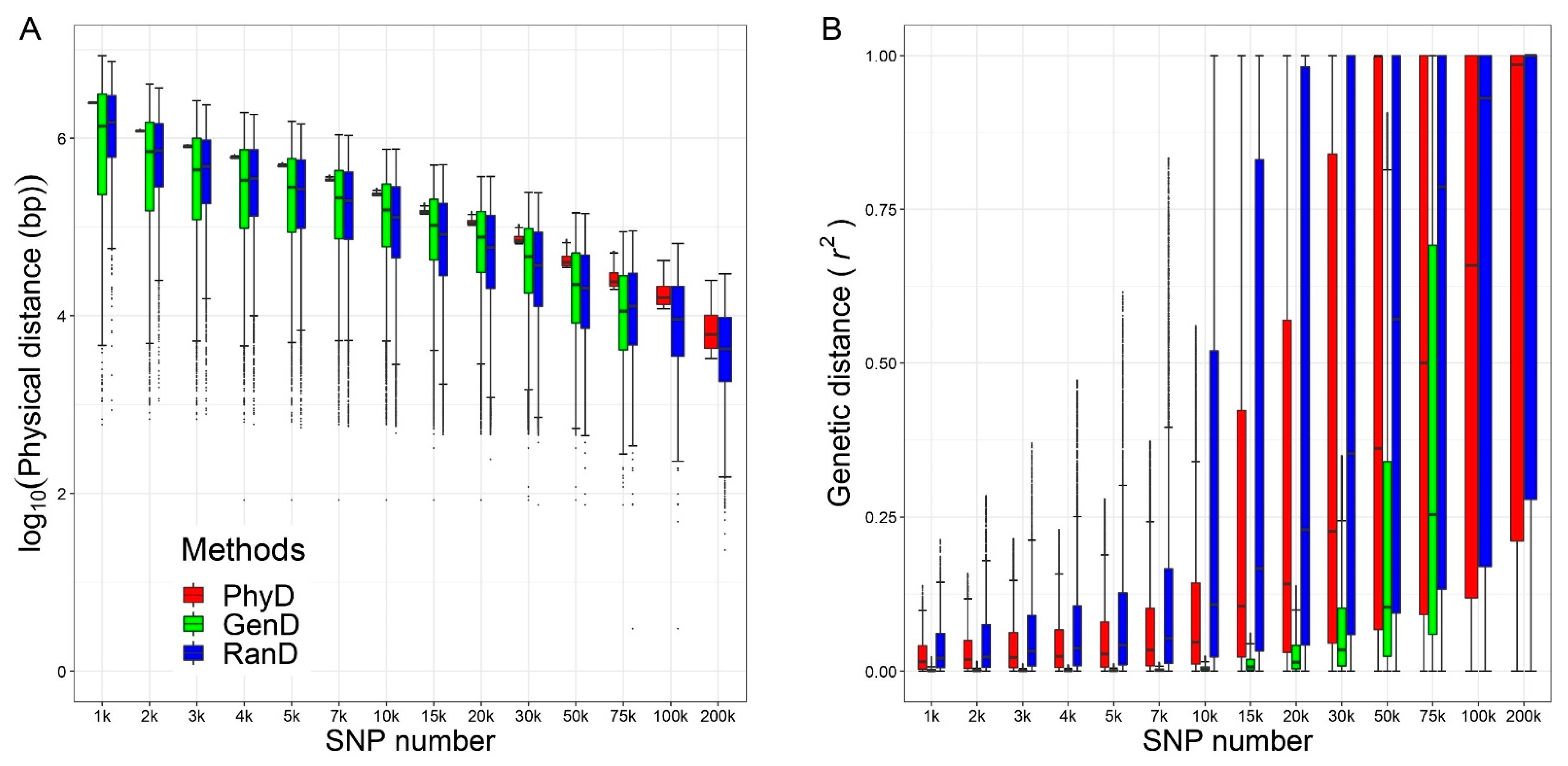
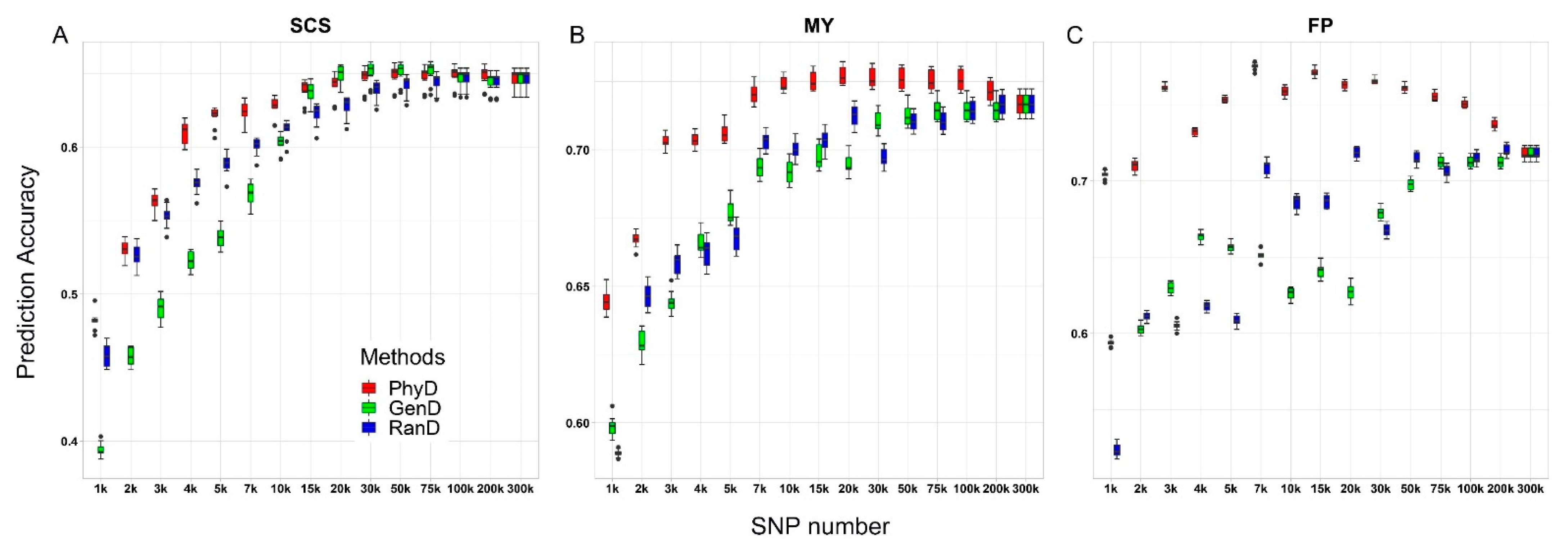
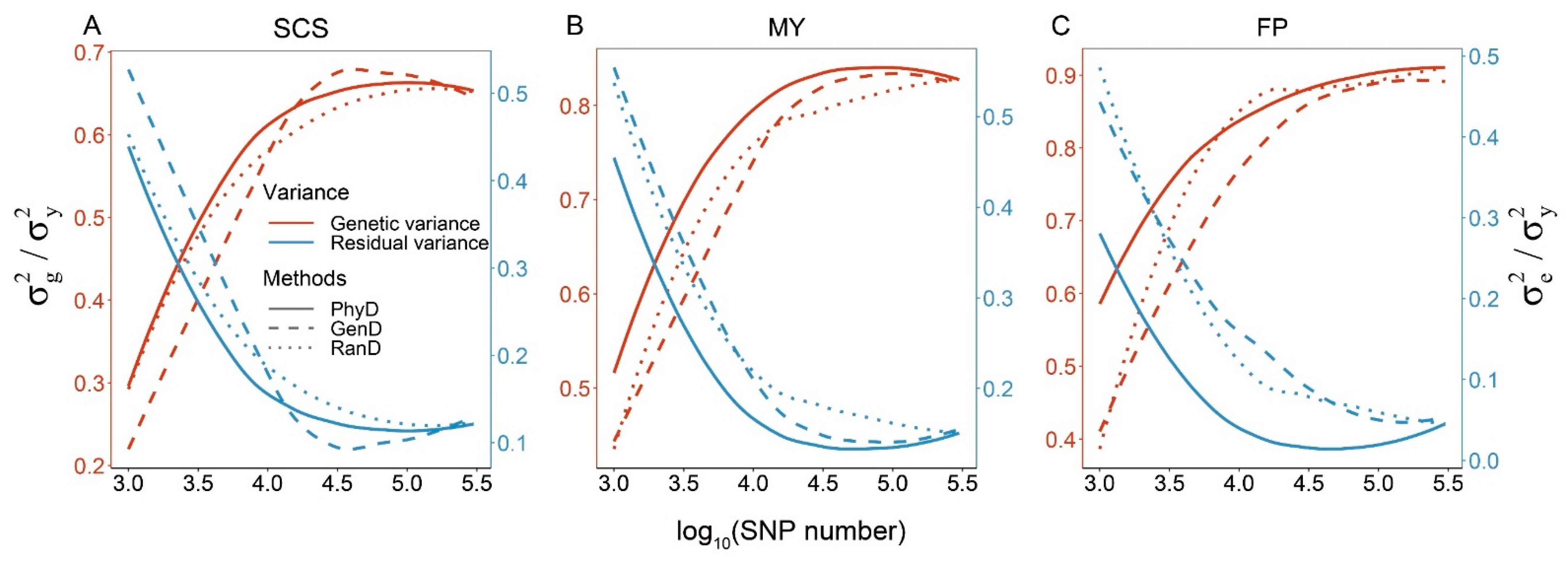

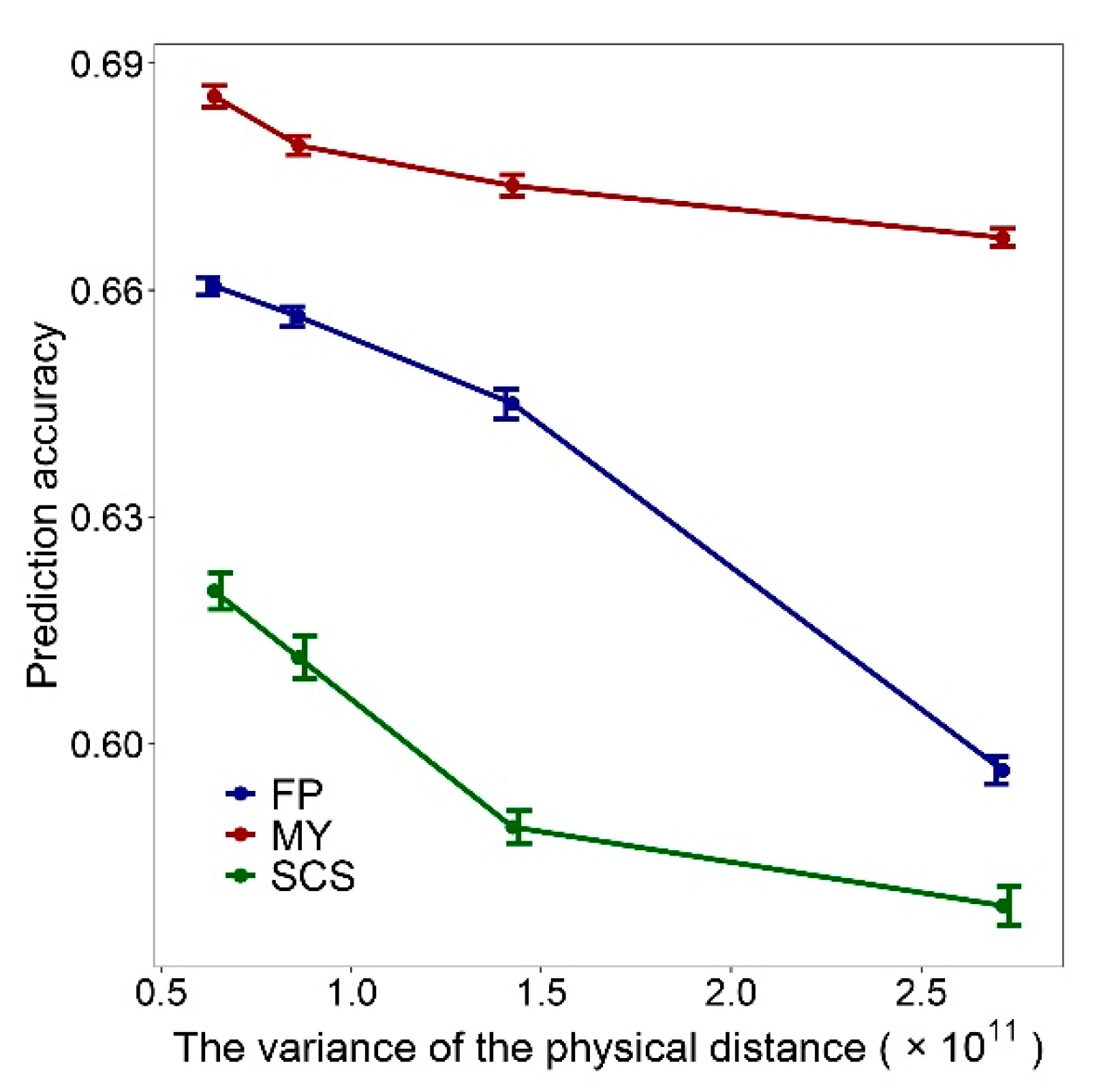
| SNP Number Levels | Methods | Measurements of Marker Density | GEBV Accuracy | ||||
|---|---|---|---|---|---|---|---|
(1010) | FP | MY | SCS | ||||
| 1 k | PhyD | 0.245 | 0.047 | 0.005 | 0.704 | 0.645 | 0.482 |
| GenD | 867.650 | 0.027 | 0.011 | 0.594 | 0.599 | 0.394 | |
| RanD | 639.753 | 0.097 | 0.038 | 0.523 | 0.589 | 0.459 | |
| 2 k | PhyD | 0.216 | 0.061 | 0.012 | 0.710 | 0.667 | 0.531 |
| GenD | 226.149 | 0.030 | 0.014 | 0.603 | 0.630 | 0.458 | |
| RanD | 158.043 | 0.125 | 0.053 | 0.611 | 0.647 | 0.527 | |
| 3 k | PhyD | 0.240 | 0.082 | 0.021 | 0.761 | 0.703 | 0.563 |
| GenD | 97.622 | 0.029 | 0.014 | 0.630 | 0.645 | 0.491 | |
| RanD | 72.558 | 0.164 | 0.075 | 0.605 | 0.659 | 0.553 | |
| 4 k | PhyD | 0.268 | 0.088 | 0.023 | 0.732 | 0.704 | 0.610 |
| GenD | 52.830 | 0.030 | 0.015 | 0.663 | 0.666 | 0.523 | |
| RanD | 43.857 | 0.193 | 0.091 | 0.617 | 0.662 | 0.576 | |
| 5 k | PhyD | 0.242 | 0.108 | 0.034 | 0.753 | 0.706 | 0.621 |
| GenD | 32.881 | 0.030 | 0.015 | 0.656 | 0.677 | 0.539 | |
| RanD | 28.646 | 0.219 | 0.103 | 0.608 | 0.668 | 0.590 | |
| 7 k | PhyD | 0.218 | 0.138 | 0.049 | 0.775 | 0.720 | 0.624 |
| GenD | 16.006 | 0.029 | 0.014 | 0.652 | 0.694 | 0.569 | |
| RanD | 15.380 | 0.253 | 0.117 | 0.709 | 0.703 | 0.601 | |
| 10 k | PhyD | 0.212 | 0.191 | 0.077 | 0.759 | 0.724 | 0.628 |
| GenD | 6.754 | 0.029 | 0.013 | 0.626 | 0.692 | 0.604 | |
| RanD | 8.399 | 0.306 | 0.138 | 0.686 | 0.700 | 0.612 | |
| 15 k | PhyD | 0.170 | 0.275 | 0.114 | 0.771 | 0.725 | 0.640 |
| GenD | 2.976 | 0.031 | 0.010 | 0.641 | 0.697 | 0.638 | |
| RanD | 4.266 | 0.373 | 0.157 | 0.686 | 0.703 | 0.623 | |
| 20 k | PhyD | 0.157 | 0.323 | 0.131 | 0.763 | 0.727 | 0.643 |
| GenD | 1.798 | 0.050 | 0.009 | 0.627 | 0.695 | 0.650 | |
| RanD | 2.627 | 0.420 | 0.167 | 0.718 | 0.712 | 0.628 | |
| 30 k | PhyD | 0.128 | 0.398 | 0.152 | 0.765 | 0.726 | 0.648 |
| GenD | 0.974 | 0.110 | 0.024 | 0.679 | 0.710 | 0.652 | |
| RanD | 1.314 | 0.479 | 0.174 | 0.668 | 0.697 | 0.640 | |
| 50 k | PhyD | 0.098 | 0.476 | 0.167 | 0.761 | 0.726 | 0.650 |
| GenD | 0.485 | 0.238 | 0.075 | 0.698 | 0.712 | 0.652 | |
| RanD | 0.551 | 0.553 | 0.176 | 0.715 | 0.710 | 0.643 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, D.; Teng, J.; Diao, S.; Lin, Q.; Li, J.; Zhang, Z. Impact of Marker Pruning Strategies Based on Different Measurements of Marker Distance on Genomic Prediction in Dairy Cattle. Animals 2021, 11, 1992. https://doi.org/10.3390/ani11071992
Ren D, Teng J, Diao S, Lin Q, Li J, Zhang Z. Impact of Marker Pruning Strategies Based on Different Measurements of Marker Distance on Genomic Prediction in Dairy Cattle. Animals. 2021; 11(7):1992. https://doi.org/10.3390/ani11071992
Chicago/Turabian StyleRen, Duanyang, Jinyan Teng, Shuqi Diao, Qing Lin, Jiaqi Li, and Zhe Zhang. 2021. "Impact of Marker Pruning Strategies Based on Different Measurements of Marker Distance on Genomic Prediction in Dairy Cattle" Animals 11, no. 7: 1992. https://doi.org/10.3390/ani11071992
APA StyleRen, D., Teng, J., Diao, S., Lin, Q., Li, J., & Zhang, Z. (2021). Impact of Marker Pruning Strategies Based on Different Measurements of Marker Distance on Genomic Prediction in Dairy Cattle. Animals, 11(7), 1992. https://doi.org/10.3390/ani11071992






