Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress
Abstract
Simple Summary
Abstract
1. Introduction
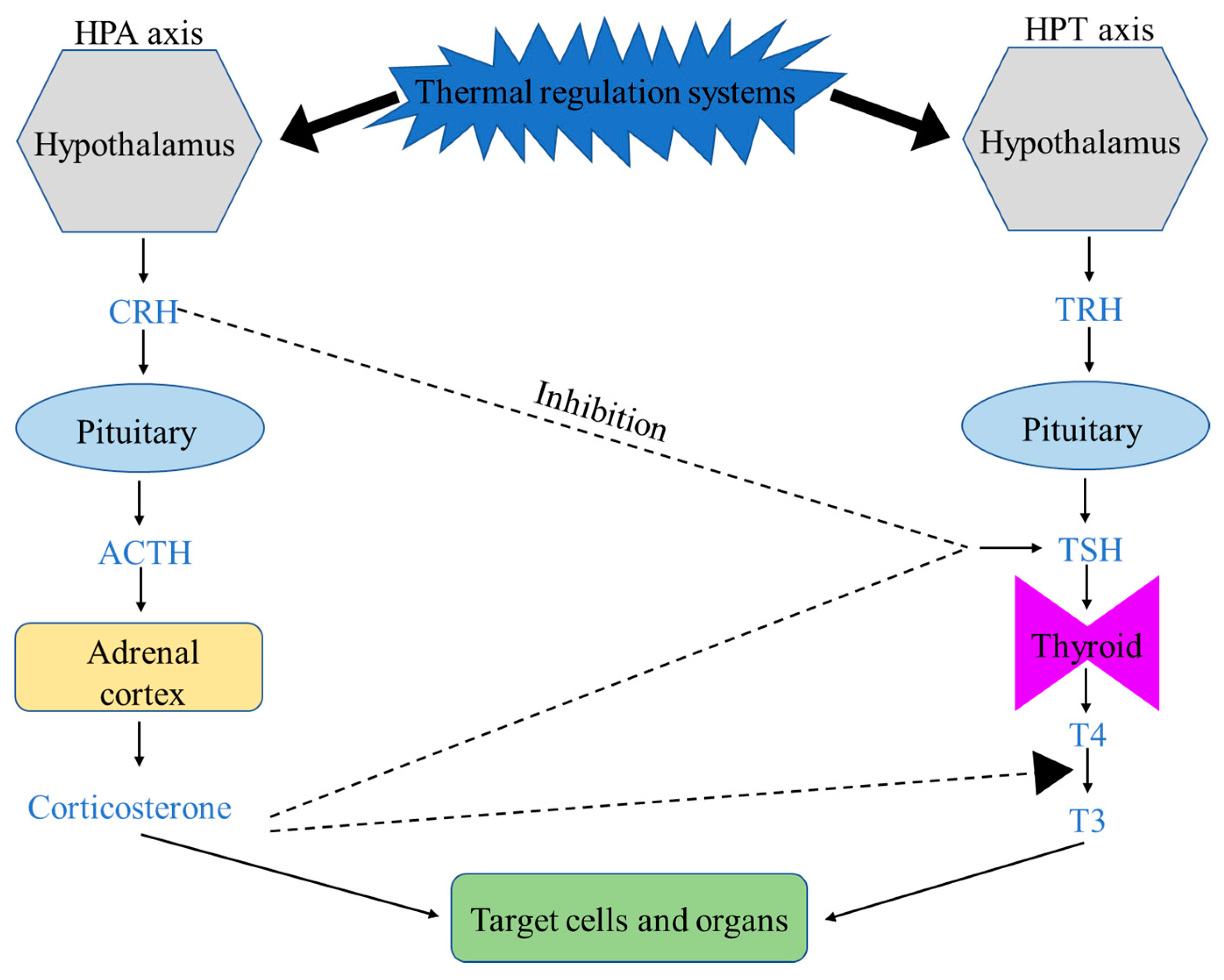
2. Heat Regulation in Broiler Chickens
3. Heat Tolerance and Heat Stress in Commercial Broilers
4. Heat Stress and Skeletal Health of Broilers
5. Heat Stress and Gut Microbiota
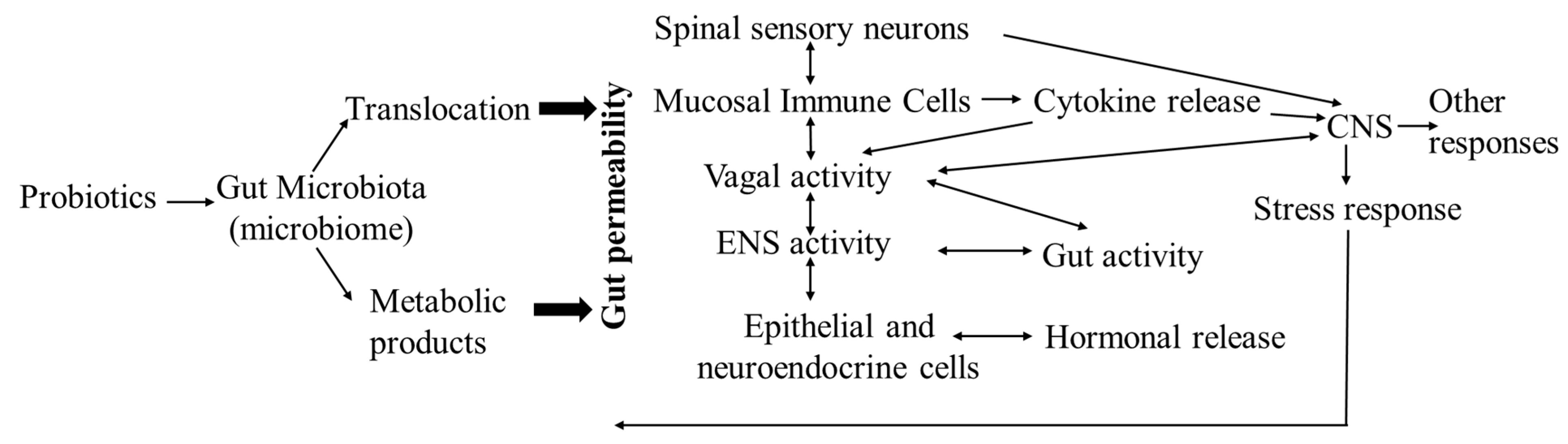
6. Probiotics and the Gut–Microbiota–Brain Axis in Broilers
6.1. The Potential Mechanisms of Probiotic Effects on Gut and Brain Functions
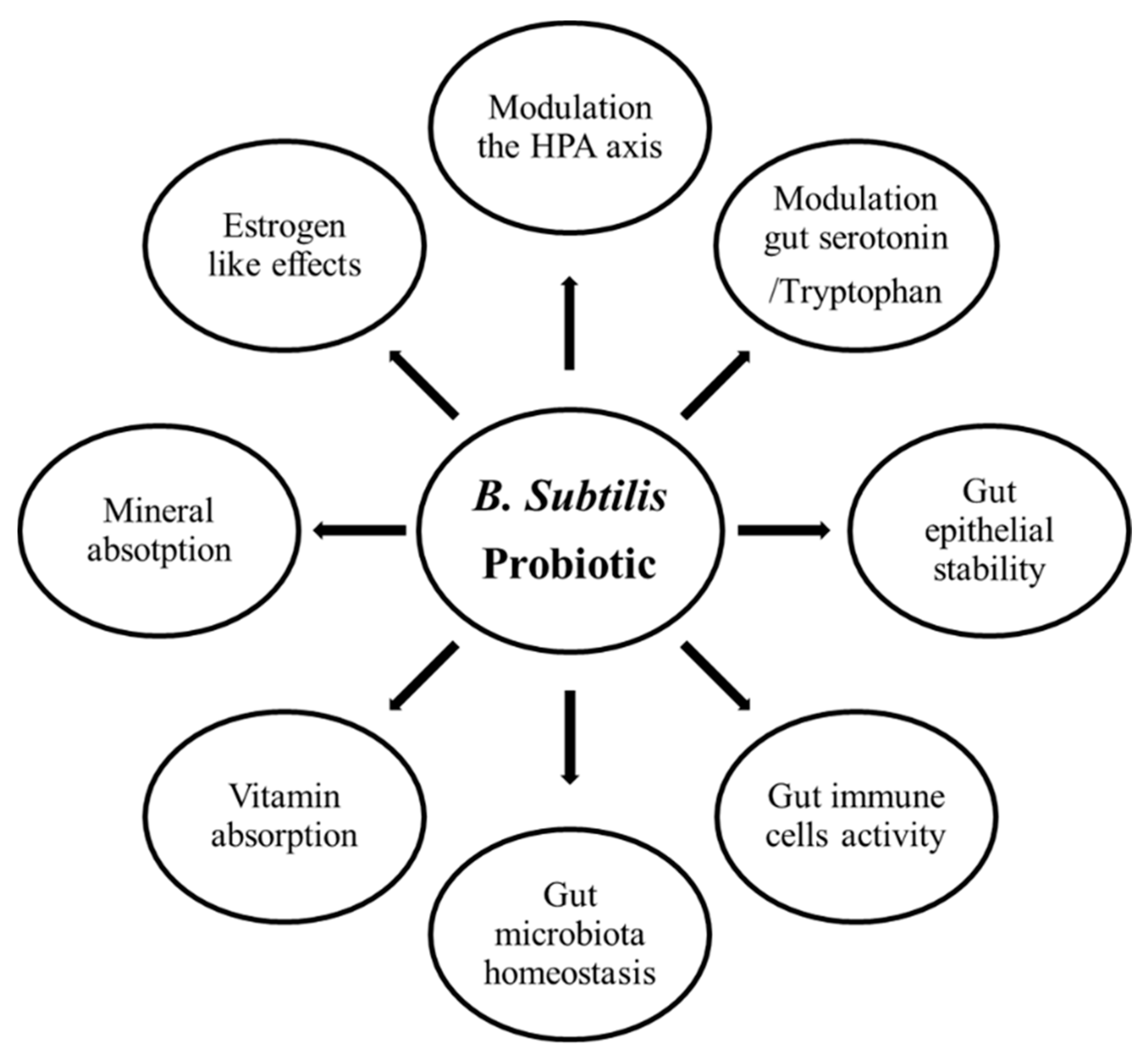
6.2. Bacillus subtilis and Broiler Skeletal Health in Broilers
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guzman-Bautista, E.R.; Suzuki, K.; Asami, S.; Fagarasan, S. Bacteria-immune cells dialog and the homeostasis of the systems. Curr. Opin. Immunol. 2020, 66, 82–89. [Google Scholar] [CrossRef]
- Jensen, E.A.; Young, J.A.; Mathes, S.C.; List, E.O.; Carroll, R.K.; Kuhn, J.; Onusko, M.; Kopchick, J.J.; Murphy, E.R.; Berryman, D.E. Crosstalk between the growth hormone/insulin-like growth factor-1 axis and the gut microbiome: A new frontier for microbial endocrinology. Growth Horm. IGF Res. 2020, 53–54, 101333. [Google Scholar] [CrossRef]
- Yoo, J.Y.; Groer, M.; Dutra, S.V.O.; Sarkar, A.; McSkimming, D.I. Gut Microbiota and Immune System Interactions. Microorganisms 2020, 8, 1587. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, A.; Yang, D.; Vella, M.; Chiu, I.M. The intestinal neuro-immune axis: Crosstalk between neurons, immune cells, and microbes. Mucosal Immunol. 2021, 14, 555–565. [Google Scholar] [CrossRef]
- Tu, Y.; Yang, R.; Xu, X.; Zhou, X. The microbiota-gut-bone axis and bone health. J. Leukoc. Biol. 2021. [Google Scholar] [CrossRef]
- Van der Hee, B.; Wells, J.M. Microbial Regulation of Host Physiology by Short-chain Fatty Acids. Trends Microbiol. 2021. [Google Scholar] [CrossRef]
- Yang, W.; Cong, Y. Gut microbiota-derived metabolites in the regulation of host immune responses and immune-related inflammatory diseases. Cell. Mol. Immunol. 2021, 18, 866–877. [Google Scholar] [CrossRef]
- Antonson, A.M.; Evans, M.V.; Galley, J.D.; Chen, H.J.; Rajasekera, T.A.; Lammers, S.M.; Hale, V.L.; Bailey, M.T.; Gur, T.L. Unique maternal immune and functional microbial profiles during prenatal stress. Sci. Rep. 2020, 10, 20288. [Google Scholar] [CrossRef] [PubMed]
- Bajinka, O.; Tan, Y.; Abdelhalim, K.A.; Özdemir, G.; Qiu, X. Extrinsic factors influencing gut microbes, the immediate consequences and restoring eubiosis. AMB Express 2020, 10, 130. [Google Scholar] [CrossRef]
- Bennett, C.J.; Henry, R.; Snipe, R.M.J.; Costa, R.J.S. Is the gut microbiota bacterial abundance and composition associated with intestinal epithelial injury, systemic inflammatory profile, and gastrointestinal symptoms in response to exertional-heat stress? J. Sci. Med. Sport 2020, 23, 1141–1153. [Google Scholar] [CrossRef]
- Boehme, M.; van de Wouw, M.; Bastiaanssen, T.F.S.; Olavarría-Ramírez, L.; Lyons, K.; Fouhy, F.; Golubeva, A.V.; Moloney, G.M.; Minuto, C.; Sandhu, K.V.; et al. Mid-life microbiota crises: Middle age is associated with pervasive neuroimmune alterations that are reversed by targeting the gut microbiome. Mol. Psychiatry 2020, 25, 2567–2583. [Google Scholar] [CrossRef]
- Redondo-Useros, N.; Nova, E.; González-Zancada, N.; Díaz, L.E.; Gómez-Martínez, S.; Marcos, A. Microbiota and Lifestyle: A Special Focus on Diet. Nutrients 2020, 12, 1776. [Google Scholar] [CrossRef] [PubMed]
- Chi, H.; Cao, W.; Zhang, M.; Su, D.; Yang, H.; Li, Z.; Li, C.; She, X.; Wang, K.; Gao, X.; et al. Environmental noise stress disturbs commensal microbiota homeostasis and induces oxi-inflammmation and AD-like neuropathology through epithelial barrier disruption in the EOAD mouse model. J. Neuroinflamm. 2021, 18, 9. [Google Scholar] [CrossRef] [PubMed]
- González Olmo, B.M.; Butler, M.J.; Barrientos, R.M. Evolution of the human diet and its impact on gut microbiota, immune responses, and brain health. Nutrients 2021, 13, 196. [Google Scholar] [CrossRef] [PubMed]
- Losso, J.N. Food processing, dysbiosis, gastrointestinal inflammatory diseases, and antiangiogenic functional foods or beverages. Annu. Rev. Food Sci. Technol. 2021, 12, 235–258. [Google Scholar] [CrossRef] [PubMed]
- Garber, A.; Hastie, P.; Murray, J.A. Factors influencing equine gut microbiota: Current knowledge. J. Equine Vet. Sci. 2020, 88, 102943. [Google Scholar] [CrossRef]
- Deng, Y.; Zhou, M.; Wang, J.; Yao, J.; Yu, J.; Liu, W.; Wu, L.; Wang, J.; Gao, R. Involvement of the microbiota-gut-brain axis in chronic restraint stress: Disturbances of the kynurenine metabolic pathway in both the gut and brain. Gut Microbes 2021, 13, 1–16. [Google Scholar]
- Hou, Y.; Wei, W.; Guan, X.; Liu, Y.; Bian, G.; He, D.; Fan, Q.; Cai, X.; Zhang, Y.; Wang, G.; et al. A diet-microbial metabolism feedforward loop modulates intestinal stem cell renewal in the stressed gut. Nat. Commun. 2021, 12, 271. [Google Scholar] [CrossRef]
- Vignal, C.; Guilloteau, E.; Gower-Rousseau, C.; Body-Malapel, M. Review article: Epidemiological and animal evidence for the role of air pollution in intestinal diseases. Sci. Total Environ. 2021, 757, 143718. [Google Scholar] [CrossRef]
- Colquhoun, C.; Duncan, M.; Grant, G. Inflammatory bowel diseases: Host-microbial-environmental interactions in dysbiosis. Diseases 2020, 8, 13. [Google Scholar] [CrossRef]
- Gao, W.; Baumgartel, K.L.; Alexander, S.A. The gut microbiome as a component of the gut-brain axis in cognitive health. Biol. Res. Nurs. 2020, 22, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Ogunrinola, G.A.; Oyewale, J.O.; Oshamika, O.O.; Olasehinde, G.I. The human microbiome and its impacts on health. Int. J. Microbiol. 2020, 2020, 8045646. [Google Scholar] [CrossRef] [PubMed]
- García-Montero, C.; Fraile-Martínez, O.; Gómez-Lahoz, A.M.; Pekarek, L.; Castellanos, A.J.; Noguerales-Fraguas, F.; Coca, S.; Guijarro, L.G.; García-Honduvilla, N.; Asúnsolo, A.; et al. Nutritional components in western diet versus mediterranean diet at the gut microbiota-immune system interplay. Implications for health and disease. Nutrients 2021, 13, 699. [Google Scholar] [CrossRef] [PubMed]
- Massier, L.; Blüher, M.; Kovacs, P.; Chakaroun, R.M. Impaired intestinal barrier and tissue bacteria: Pathomechanisms for metabolic diseases. Front. Endocrinol. 2021, 12, 616506. [Google Scholar] [CrossRef]
- Shaik, L.; Kashyap, R.; Thotamgari, S.R.; Singh, R.; Khanna, S. Gut-brain axis and its neuro-psychiatric effects: A narrative review. Cureus 2020, 12, e11131. [Google Scholar] [CrossRef]
- Mars, R.A.T.; Frith, M.; Kashyap, P.C. Functional gastrointestinal disorders and the microbiome-what is the best strategy for moving microbiome-based therapies for functional gastrointestinal disorders into the clinic? Gastroenterology 2021, 160, 538–555. [Google Scholar] [CrossRef] [PubMed]
- Settanni, C.R.; Ianiro, G.; Bibbò, S.; Cammarota, G.; Gasbarrini, A. Gut microbiota alteration and modulation in psychiatric disorders: Current evidence on fecal microbiota transplantation. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 109, 110258. [Google Scholar] [CrossRef]
- Bermúdez-Humarán, L.G.; Salinas, E.; Ortiz, G.G.; Ramirez-Jirano, L.J.; Morales, J.A.; Bitzer-Quintero, O.K. From probiotics to psychobiotics: Live beneficial bacteria which act on the brain-gut axis. Nutrients 2019, 11, 890. [Google Scholar] [CrossRef]
- Del Toro-Barbosa, M.; Hurtado-Romero, A.; Garcia-Amezquita, L.E.; García-Cayuela, T. Psychobiotics: Mechanisms of action, evaluation methods and effectiveness in applications with food products. Nutrients 2020, 12, 3896. [Google Scholar] [CrossRef] [PubMed]
- Kalmar, I.D.; Vanrompay, D.; Janssens, G.P. Broiler ascites syndrome: Collateral damage from efficient feed to meat conversion. Nutrients 2013, 197, 169–174. [Google Scholar] [CrossRef]
- Eren, U.; Kum, S.; Nazligul, A.; Gules, O.; Aka, E.; Zorlu, S.; Yildiz, M. The several elements of intestinal innate immune system at the beginning of the life of broiler chicks. Microsc. Res. Tech. 2016, 79, 604–614. [Google Scholar] [CrossRef] [PubMed]
- Feighner, S.D.; Dashkevicz, M.P. Subtherapeutic levels of antibiotics in poultry feeds and their effects on weight gain, feed efficiency, and bacterial cholyltaurine hydrolase activity. Appl. Environ. Microbiol. 1987, 53, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, H.M. Antibiotic-free poultry production: Is it sustainable? J. Appl. Poult. Res. 2015, 24, 91–97. [Google Scholar] [CrossRef]
- Khalique, A.; Zeng, D.; Shoaib, M.; Wang, H.; Qing, X.; Rajput, D.S.; Pan, K.; Ni, X. Probiotics mitigating subclinical necrotic enteritis (SNE) as potential alternatives to antibiotics in poultry. AMB Express 2020, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Neveling, D.P.; Dicks, L.M.T. Probiotics: An Antibiotic replacement strategy for healthy broilers and productive rearing. Probiotics Antimicrob. Proteins 2021, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tarradas, J.; Tous, N.; Esteve-Garcia, E.; Brufau, A.J. The control of intestinal inflammation: A major objective in the research of probiotic strains as alternatives to antibiotic growth promoters in poultry. Microorganisms 2020, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Joerger, R.D.; Ganguly, A. Current status of the preharvest application of pro- and prebiotics to farm animals to enhance the microbial safety of animal products. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. The role of probiotics, prebiotics and synbiotics in animal nutrition. Gut Pathog. 2018, 10, 21. [Google Scholar] [CrossRef]
- Heak, C.; Sukon, P.; Sornplang, P. Effect of direct-fed microbials on culturable gut microbiotas in broiler chickens: A meta-analysis of controlled trials. Asian Australas. J. Anim. Sci. 2018, 31, 1781–1794. [Google Scholar] [CrossRef]
- Jha, R.; Das, R.; Oak, S.; Mishra, P. Probiotics (direct-fed microbials) in poultry nutrition and their effects on nutrient utilization, growth and laying performance, and gut health: A systematic review. Animals 2020, 10, 1863. [Google Scholar] [CrossRef]
- Khan, S.; Moore, R.J.; Stanley, D.; Chousalkar, K.K. The gut microbiota of laying hens and its manipulation with prebiotics and probiotics to enhance gut health and food safety. Appl. Environ. Microbiol. 2020, 86, e00600-20. [Google Scholar] [CrossRef]
- Elshaghabee, F.M.F.; Rokana, N.; Gulhane, R.D.; Sharma, C.; Panwar, H. Bacillus As Potential Probiotics: Status, Concerns, and Future Perspectives. Front. Microbiol. 2017, 8, 1490. [Google Scholar] [CrossRef] [PubMed]
- Mingmongkolchai, S.; Panbangred, W. Bacillus probiotics: An alternative to antibiotics for livestock production. J. Appl. Microbiol. 2018, 124, 1334–1346. [Google Scholar] [CrossRef] [PubMed]
- Yahav, S.; Berkovich, Z.; Ostrov, I.; Reifen, R.; Shemesh, M. Encapsulation of beneficial probiotic bacteria in extracellular matrix from biofilm-forming Bacillus subtilis. Artif. Cells Nanomed. Biotechnol. 2018, 46, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Elisashvili, V.; Kachlishvili, E.; Chikindas, M.L. Recent advances in the physiology of spore formation for bacillus probiotic production. Probiotics Antimicrob. Proteins 2019, 11, 731–747. [Google Scholar] [CrossRef]
- Lee, N.K.; Kim, W.S.; Paik, H.D. Bacillus strains as human probiotics: Characterization, safety, microbiome, and probiotic carrier. Food Sci. Biotechnol. 2019, 28, 1297–1305. [Google Scholar] [CrossRef]
- Danilova, I.; Sharipova, M. The Practical Potential of Bacilli and Their Enzymes for Industrial Production. Front. Microbiol. 2020, 11, 1782. [Google Scholar] [CrossRef]
- Ugwuodo, C.J.; Nwagu, T.N. Stabilizing enzymes by immobilization on bacterial spores: A review of literature. Int. J. Biol. Macromol. 2021, 166, 238–250. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Zhang, N.; Han, J.C.; Chang, C.W.; Hsiao, F.S.; Yu, Y.H. Optimization of surfactin production from Bacillus subtilis in fermentation and its effects on Clostridium perfringens-induced necrotic enteritis and growth performance in broilers. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1232–1244. [Google Scholar] [CrossRef]
- Grant, A.; Gay, C.G.; Lillehoj, H.S. Bacillus spp. as direct-fed microbial antibiotic alternatives to enhance growth, immunity, and gut health in poultry. Avian Pathol. 2018, 47, 339–351. [Google Scholar] [CrossRef]
- Dela Cruz, P.J.D.; Dagaas, C.T.; Mangubat, K.M.M.; Angeles, A.A.; Abanto, O.D. Dietary effects of commercial probiotics on growth performance, digestibility, and intestinal morphometry of broiler chickens. Trop. Anim. Health Prod. 2019, 51, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Ramlucken, U.; Lalloo, R.; Roets, Y.; Moonsamy, G.; van Rensburg, C.J.; Thantsha, M.S. Advantages of Bacillus-based probiotics in poultry production. Livest. Sci. 2020, 241, 15. [Google Scholar] [CrossRef]
- Zaiss, M.M.; Jones, R.M.; Schett, G.; Pacifici, R. The gut-bone axis: How bacterial metabolites bridge the distance. J. Clin. Investig. 2019, 129, 3018–3028. [Google Scholar] [CrossRef] [PubMed]
- Behera, J.; Ison, J.; Tyagi, S.C.; Tyagi, N. The role of gut microbiota in bone homeostasis. Bone 2020, 135, 115317. [Google Scholar] [CrossRef]
- Li, J.; Ho, W.T.P.; Liu, C.; Chow, S.K.; Ip, M.; Yu, J.; Wong, H.S.; Cheung, W.H.; Sung, J.J.Y.; Wong, R.M.Y. The role of gut microbiota in bone homeostasis. Bone Jt. Res. 2021, 10, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Schepper, J.D.; Irwin, R.; Kang, J.; Dagenais, K.; Lemon, T.; Shinouskis, A.; Parameswaran, N.; McCabe, L.R. Probiotics in gut-bone signaling. Adv. Exp. Med. Biol. 2017, 1033, 225–247. [Google Scholar]
- Kiousi, D.E.; Karapetsas, A.; Karolidou, K.; Panayiotidis, M.I.; Pappa, A.; Galanis, A. Probiotics in extraintestinal diseases: Current trends and new directions. Nutrients 2019, 11, 788. [Google Scholar] [CrossRef] [PubMed]
- Poultry Housing: Guide for Building Suitable House for Poultry. Available online: https://www.roysfarm.com/poultry-housing/ (accessed on 20 December 2020).
- Pereira, D.F.; Nääs, I.A. Estimating the thermoneutral zone for broiler breeders using behavioral analysis. Comput. Electron. Agric. 2008, 62, 2–7. [Google Scholar] [CrossRef]
- Dawson, W.R.; Whittow, G.C. Regulation of body temperature. In Avian Physiology, 2nd ed.; Whittow, G.C., Ed.; Academic Press: San Diego, CA, USA, 2000; pp. 343–390. [Google Scholar]
- Tabarean, I.; Morrison, B.; Marcondes, M.C.; Bartfai, T.; Conti, B. Hypothalamic and dietary control of temperature-mediated longevity. Ageing Res. Rev. 2010, 9, 41–50. [Google Scholar] [CrossRef]
- Chiamolera, M.I.; Wondisford, F.E. Minireview: Thyrotropin-releasing hormone and the thyroid hormone feedback mechanism. Endocrinology 2009, 150, 1091–1096. [Google Scholar] [CrossRef]
- Morrison, S.F.; Nakamura, K. Central neural pathways for thermoregulation. Front. Biosci. 2011, 16, 74–104. [Google Scholar] [CrossRef] [PubMed]
- Ortiga-Carvalho, T.M.; Chiamolera, M.I.; Pazos-Moura, C.C.; Wondisford, F.E. Hypothalamus-pituitary-thyroid axis. Compr. Physiol. 2016, 6, 1387–1428. [Google Scholar] [PubMed]
- Zhang, Z.; Boelen, A.; Kalsbeek, A.; Fliers, E. TRH neurons and thyroid hormone coordinate the hypothalamic response to cold. Eur. Thyroid. J. 2018, 7, 279–288. [Google Scholar] [CrossRef]
- McNabb, F.M. Avian thyroid development and adaptive plasticity. Gen. Comp. Endocrinol. 2006, 147, 93–101. [Google Scholar] [CrossRef]
- Mullur, R.; Liu, Y.Y.; Brent, G.A. Thyroid hormone regulation of metabolism. Physiol. Rev. 2014, 94, 355–382. [Google Scholar] [CrossRef] [PubMed]
- Research, H.J.E. Thyroid hormone and the gut. Endocr. Res. 1988, 14, 203–224. [Google Scholar]
- Knezevic, J.; Starchl, C.; Tmava Berisha, A.; Amrein, K. Thyroid-Gut-Axis: How Does the Microbiota Influence Thyroid Function? Nutrients 2020, 12, 1769. [Google Scholar] [CrossRef]
- Zheng, W.H.; Lin, L.; Liu, J.S.; Xu, X.J.; Li, M. Geographic variation in basal thermogenesis in little buntings: Relationship to cellular thermogenesis and thyroid hormone concentrations. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2013, 164, 483–490. [Google Scholar] [CrossRef]
- Sharp, P.J.; Klandorf, H. Environmental and physiological factors controlling thyroid function in Galliformes. In The Endocrine System and the Environment; Follett, B.K., Ishii, S., Eds.; Japan Scientific Societies Press: Tokyo, Japan, 1985; pp. 175–188. [Google Scholar]
- Wentworth, B.C.; Ringer, R.K. Thyroids. In Avian Physiology; Sturkie, P.D., Ed.; Springer: New York, NY, USA, 1986; pp. 452–465. [Google Scholar]
- Luijten, I.H.N.; Cannon, B.; Nedergaard, J. Glucocorticoids and brown adipose tissue: Do glucocorticoids really inhibit thermogenesis? Mol. Asp. Med. 2019, 68, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.; Armani, A.; Mammi, C.; Fabbri, A.; Caprio, M. Impact of adrenal steroids on regulation of adipose tissue. Compr. Physiol. 2017, 7, 1425–1447. [Google Scholar]
- Facchi, J.C.; Lima, T.A.L.; Oliveira, L.R.; Costermani, H.O.; Miranda, G.D.S.; de Oliveira, J.C. Perinatal programming of metabolic diseases: The role of glucocorticoids. Metabolism 2020, 104, 154047. [Google Scholar] [CrossRef]
- Rahimi, L.; Rajpal, A.; Ismail-Beigi, F. Glucocorticoid-induced fatty liver disease. Diabetes Metab. Syndr. Obes. 2020, 13, 1133–1145. [Google Scholar] [CrossRef]
- Heyma, P.; Larkins, R.G. Glucocorticoids decrease in conversion of thyroxine into 3,5,3′-tri-iodothyronine by isolated rat renal tubules. Clin. Sci. 1982, 62, 215–220. [Google Scholar] [CrossRef]
- Senda, A.; Endo, A.; Tachimori, H.; Fushimi, K.; Otomo, Y. Early administration of glucocorticoid for thyroid storm: Analysis of a national administrative database. Crit. Care 2020, 24, 470. [Google Scholar] [CrossRef]
- Harvey, S.; Carsia, R. Adrenals. In Sturkie’s Avian Physiology, 5th ed.; Whittow, G.C., Ed.; Academic Press: London, UK, 1999; pp. 489–537. [Google Scholar]
- Etches, R.J.; John, T.M.; Verrinder-Gibbins, G.A.M. Behavioural, physiological, neuroendocrine and molecular responses to heat stress. In Poultry Production in Hot Climates, 2nd ed.; CABI: Oxfordshire, UK, 2008; pp. 31–66. [Google Scholar]
- Lara, L.J.; Rostagno, M.H. Impact of Heat Stress on Poultry Production. Animals 2013, 3, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Butcher, G.D.; Miles, R.; Heat Stress Management in Broiler. Nebraska Extension: Community Environment. Available online: http://edis.ifas.ufl.edu (accessed on 20 December 2020).
- Sandercock, D.A.; Hunter, R.R.; Nute, G.R.; Mitchell, M.A.; Hocking, P.M. Acute heat stress-induced alterations in blood acid-base status and skeletal muscle membrane integrity in broiler chickens at two ages: Implications for meat quality. Poult. Sci. 2001, 80, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Comito, R.W.; Reece, W.O.; Trampel, D.W.; Koehler, K.J. Acid-base balance of the domestic turkey during thermal panting. Poult. Sci. 2007, 86, 2649–2652. [Google Scholar] [CrossRef] [PubMed]
- Felver-Gant, J.N.; Mack, L.A.; Dennis, R.L.; Eicher, S.D.; Cheng, H.W. Genetic variations alter physiological responses following heat stress in 2 strains of laying hens. Poult. Sci. 2012, 91, 1542–1551. [Google Scholar] [CrossRef]
- Sahin, K.; Sahin, N.; Kucuk, O.; Hayirli, A.; Prasad, A.S. Role of dietary zinc in heat-stressed poultry: A review. Poult. Sci. 2009, 88, 2176–2183. [Google Scholar] [CrossRef] [PubMed]
- Russo, S.; Sillmann, J.; Sterl, A. Humid heat waves at different warming levels. Sci. Rep. 2017, 7, 7477. [Google Scholar] [CrossRef]
- Hartcher, K.M.; Lum, H.K. Genetic selection of broilers and welfare consequences: A review. World Poult. Sci. J. 2019, 76, 1–14. [Google Scholar] [CrossRef]
- Lu, Z.; He, X.; Ma, B.; Zhang, L.; Li, J.; Jiang, Y.; Zhou, G.; Gao, F. Chronic heat stress impairs the quality of breast-muscle meat in broilers by affecting redox status and energy-substance metabolism. J. Agric. Food Chem. 2017, 65, 11251–11258. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, V.S. Heat stress biomarker amino acids and neuropeptide afford thermotolerance in chicks. J. Poult. Sci. 2019, 56, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.E.; Kim, J.H.; Goo, D.; Han, G.P.; Pitargue, F.M.; Kang, H.K.; Kil, D.Y.J.L.S. Effect of dietary supplementation of betaine on productive performance, egg quality and jejunal tight junction-related gene expression in laying hens raised under hot environmental conditions. Livest. Sci. 2018, 214, 79–82. [Google Scholar] [CrossRef]
- Goo, D.; Kim, J.H.; Park, G.H.; Delos Reyes, J.B.; Kil, D.Y. Effect of heat stress and stocking density ongrowth performance, breast meat quality, and intestinal barrier function in broiler chickens. Animals 2019, 9, 107. [Google Scholar] [CrossRef]
- Saeed, M.; Abbas, G.; Alagawany, M.; Kamboh, A.A.; Abd El-Hack, M.E.; Khafaga, A.F.; Chao, S. Heat stress management in poultry farms: A comprehensive overview. J. Therm. Biol. 2019, 84, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Goel, A. Heat stress management in poultry. J. Anim. Physiol. Anim. Nutr. 2021. [Google Scholar] [CrossRef]
- Moore, P.R.; Evenson, A.; Luckey, T.D.; Mccoy, E.; Hart, E.B. Use of sulfasuxidine, streptothricin, and streptomycin in nutritional studies with the chick. J. Biol. Chem. 1946, 165, 437–441. [Google Scholar] [CrossRef]
- Dibner, J.J.; Richards, J.D. Antibiotic growth promoters in agriculture: History and mode of action. Poult. Sci. 2005, 84, 634–643. [Google Scholar] [CrossRef]
- Mehdi, Y.; Létourneau-Montminy, M.P.; Gaucher, M.L.; Chorfi, Y.; Suresh, G.; Rouissi, T.; Brar, S.K.; Côté, C.; Ramirez, A.A.; Godbout, S. Use of antibiotics in broiler production: Global impacts and alternatives. Anim. Nutr. 2018, 4, 170–178. [Google Scholar] [CrossRef]
- Faruque, S.; Islam, M.S.; Afroz, M.A.; Rahman, M.M. Evaluation of the performance of native chicken and estimation of heritability for body weight. J. Bangladesh Aca. Sci. 2013, 37, 93–101. [Google Scholar] [CrossRef]
- Williams, B.; Solomon, S.; Waddington, D.; Thorp, B.; Farquharson, C. Skeletal development in the meat-type chicken. Br. Poult. Sci. 2000, 41, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Leach, R.M., Jr.; Monsonego-Ornan, E. Tibial dyschondroplasia 40 years later. Poult. Sci. 2007, 86, 2053–2058. [Google Scholar] [CrossRef] [PubMed]
- Ospina-Rojas, I.C.; Murakami, A.E.; Duarte, C.R.A.; Sakamoto, M.I.; Aguihe, P.C.; Pozza, P.C.; Santos, T.C. Tibiotarsus bone characteristics and tibial dyschondroplasia incidence of broilers fed diets supplemented with leucine and valine. J. Anim. Physiol. Anim. Nutr. 2018, 102, e768–e776. [Google Scholar] [CrossRef]
- Crandall, C.J.; Miller-Martinez, D.; Greendale, G.A.; Binkley, N.; Seeman, T.E.; Karlamangla, A.S. Socioeconomic status, race, and bone turnover in the Midlife in the US Study. Osteoporos. Int. 2012, 23, 1503–1512. [Google Scholar] [CrossRef]
- Azuma, K.; Adachi, Y.; Hayashi, H.; Kubo, K.Y. Chronic psychological stress as a risk factor of osteoporosis. J. UOEH 2015, 37, 245–253. [Google Scholar] [CrossRef]
- Brennan-Olsen, S.L.; Page, R.S.; Berk, M.; Riancho, J.A.; Leslie, W.D.; Wilson, S.G.; Saban, K.L.; Janusek, L.; Pasco, J.A.; Hodge, J.M.; et al. DNA methylation and the social gradient of osteoporotic fracture: A conceptual model. Bone 2016, 84, 204–212. [Google Scholar] [CrossRef] [PubMed]
- USDA. Livestock and Poultry: World Markets and Trade. 2014. Available online: https://apps.fas.usda.gov/psdonline/circulars/livestock_poultry.pdf (accessed on 18 January 2021).
- Granquist, E.G.; Vasdal, G.; de Jong, I.C.; Moe, R.O. Lameness and its relationship with health and production measures in broiler chickens. Animal 2019, 13, 2365–2372. [Google Scholar] [CrossRef]
- Wideman, R.F., Jr.; Blankenship, J.; Pevzner, I.Y.; Turner, B.J. Efficacy of 25-OH Vitamin D3 prophylactic administration for reducing lameness in broilers grown on wire flooring. Poult. Sci. 2015, 94, 1821–1827. [Google Scholar] [CrossRef]
- Wijesurendra, D.S.; Chamings, A.N.; Bushell, R.N.; Rourke, D.O.; Stevenson, M.; Marenda, M.S.; Noormohammadi, A.H.; Stent, A. Pathological and microbiological investigations into cases of bacterial chondronecrosis and osteomyelitis in broiler poultry. Avian Pathol. 2017, 46, 683–694. [Google Scholar] [CrossRef]
- Weimer, S.L.; Wideman, R.F.; Scanes, C.G.; Mauromoustakos, A.; Christensen, K.D.; Vizzier-Thaxton, Y. The utility of infrared thermography for evaluating lameness attributable to bacterial chondronecrosis with osteomyelitis. Poult. Sci. 2019, 98, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- Bassler, A.W.; Arnould, C.; Butterworth, A.; Colin, L.; De Jong, I.C.; Ferrante, V.; Ferrari, P.; Haslam, S.; Wemelsfelder, F.; Blokhuis, H.J. Potential risk factors associated with contact dermatitis, lameness, negative emotional state, and fear of humans in broiler chicken flocks. Poult. Sci. 2013, 92, 2811–2826. [Google Scholar] [CrossRef] [PubMed]
- Vasdal, G.; Vas, J.; Newberry, R.C.; Moe, R.O. Effects of environmental enrichment on activity and lameness in commercial broiler production. J. Appl. Anim. Welf. Sci. 2019, 22, 197–205. [Google Scholar] [CrossRef]
- CWF (Compassion in World Farming). Revealed: The Distressing Cause of Those Thin White Stripes Often Found in Raw Chicken Breast. 2017. Available online: https://www.news.com.au/lifestyle/health/diet/revealed-the-distressing-cause-of-those-thin-white-stripes-often-found-in-raw-chicken-breast/news-story/7b93ab41f47eff6aa773791c27dd4a01 (accessed on 18 January 2021).
- Frank, D.N.; St Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef]
- Bosi, A.; Banfi, D.; Bistoletti, M.; Giaroni, C.; Baj, A. Tryptophan metabolites along the microbiota-gut-brain axis: An interkingdom communication system influencing the gut in health and disease. Int. J. Tryptophan. Res. 2020, 13, 1178646920928984. [Google Scholar] [CrossRef] [PubMed]
- Jameson, K.G.; Olson, C.A.; Kazmi, S.A.; Hsiao, E.Y. Toward understanding microbiome-neuronal signaling. Mol. Cell 2020, 78, 577–583. [Google Scholar] [CrossRef]
- Kong, G.; Ellul, S.; Narayana, V.K.; Kanojia, K.; Ha, H.T.T.; Li, S.; Renoir, T.; Cao, K.L.; Hannan, A.J. An integrated metagenomics and metabolomics approach implicates the microbiota-gut-brain axis in the pathogenesis of Huntington’s disease. Neurobiol. Dis. 2021, 148, 105199. [Google Scholar] [CrossRef]
- Konjevod, M.; Nikolac Perkovic, M.; Sáiz, J.; Svob Strac, D.; Barbas, C.; Rojo, D. Metabolomics analysis of microbiota-gut-brain axis in neurodegenerative and psychiatric diseases. J. Pharm. Biomed. Anal. 2021, 194, 113681. [Google Scholar] [CrossRef] [PubMed]
- Sudo, N. Microbiome, HPA axis and production of endocrine hormones in the gut. Adv. Exp. Med. Biol. 2014, 817, 177–194. [Google Scholar] [PubMed]
- Farzi, A.; Fröhlich, E.E.; Holzer, P. Gut Microbiota and the Neuroendocrine System. Neurotherapeutics 2018, 15, 5–22. [Google Scholar] [CrossRef]
- Wang, S.; Harvey, L.; Martin, R.; van der Beek, E.M.; Knol, J.; Cryan, J.F.; Renes, I.B. Targeting the gut microbiota to influence brain development and function in early life. Neurosci. Biobehav. Rev. 2018, 95, 191–201. [Google Scholar] [CrossRef]
- Li, C.; Niu, Z.; Zou, M.; Liu, S.; Wang, M.; Gu, X.; Lu, H.; Tian, H.; Jha, R. Probiotics, prebiotics, and synbiotics regulate the intestinal microbiota differentially and restore the relative abundance of specific gut microorganisms. J. Dairy Sci. 2020, 103, 5816–5829. [Google Scholar] [CrossRef]
- Da Silva, T.F.; Casarotti, S.N.; de Oliveira, G.L.V.; Penna, A.L.B. The impact of probiotics, prebiotics, and synbiotics on the biochemical, clinical, and immunological markers, as well as on the gut microbiota of obese hosts. Crit. Rev. Food Sci. Nutr. 2021, 61, 337–355. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Huang, L.; Yin, P.; Liu, F.; Liu, Y.; Zhang, Z.; Lin, J.; Zou, W.; Li, C. L-Arginine alleviates heat stress-induced intestinal epithelial barrier damage by promoting expression of tight junction proteins via the AMPK pathway. Mol. Biol. Rep. 2019, 46, 6435–6451. [Google Scholar] [CrossRef] [PubMed]
- Rostagno, M.H. Effects of heat stress on the gut health of poultry. J. Anim. Sci. 2020, 98, skaa090. [Google Scholar] [CrossRef]
- Tabler, T.W.; Greene, E.S.; Orlowski, S.K.; Hiltz, J.Z.; Anthony, N.B.; Dridi, S. Intestinal barrier integrity in heat-stressed modern broilers and their ancestor wild jungle fowl. Front. Vet. Sci. 2020, 7, 249. [Google Scholar] [CrossRef]
- Shi, D.; Bai, L.; Qu, Q.; Zhou, S.; Yang, M.; Guo, S.; Li, Q.; Liu, C. Impact of gut microbiota structure in heat-stressed broilers. Poult. Sci. 2019, 98, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Luo, M.; Ma, W.; Ma, S.; Wang, Y.; Zhang, K. Protective effects of 1,8-cineole microcapsules against inflammation and gut microbiota imbalance associated weight loss induced by heat stress in broiler chicken. Front. Pharmacol. 2020, 11, 585945. [Google Scholar] [CrossRef]
- Wang, M.; Lin, X.; Jiao, H.; Uyanga, V.; Zhao, J.; Wang, X.; Li, H.; Zhou, Y.; Sun, S.; Lin, H. Mild heat stress changes the microbiota diversity in the respiratory tract and the cecum of layer-type pullets. Poult. Sci. 2020, 99, 7015–7026. [Google Scholar] [CrossRef]
- Ducray, H.A.G.; Globa, L.; Pustovyy, O.; Morrison, E.; Vodyanoy, V.; Sorokulova, I. Yeast fermentate prebiotic improves intestinal barrier integrity during heat stress by modulation of the gut microbiota in rats. J. Appl. Microbiol. 2019, 127, 1192–1206. [Google Scholar] [CrossRef] [PubMed]
- Alhenaky, A.; Abdelqader, A.; Abuajamieh, M.; Al-Fataftah, A.R. The effect of heat stress on intestinal integrity and Salmonella invasion in broiler birds. J. Therm. Biol. 2017, 70, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Slawinska, A.; Mendes, S.; Dunislawska, A.; Siwek, M.; Zampiga, M.; Sirri, F.; Meluzzi, A.; Tavaniello, S.; Maiorano, G. Avian model to mitigate gut-derived immune response and oxidative stress during heat. Biosystems 2019, 178, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Maldonado Galdeano, C.; Cazorla, S.I.; Lemme Dumit, J.M.; Velez, E.; Perdigon, G. Beneficial effects of probiotic consumption on the immune system. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef]
- Monteagudo-Mera, A.; Rastall, R.A.; Gibson, G.R.; Charalampopoulos, D.; Chatzifragkou, A. Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Appl. Microbiol. Biotechnol. 2019, 103, 6463–6472. [Google Scholar] [CrossRef]
- Peters, V.B.M.; van de Steeg, E.; van Bilsen, J.; Meijerink, M. Mechanisms and immunomodulatory properties of pre- and probiotics. Benef. Microbes 2019, 10, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Qattan, S.Y.A.; Batiha, G.E.; Khafaga, A.F.; Abdel-Moneim, A.E.; Alagawany, M. Probiotics in poultry feed: A comprehensive review. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1835–1850. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, E.Z. Human gut microbiota/microbiome in health and diseases: A review. Antonie Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef]
- Mörkl, S.; Butler, M.I.; Holl, A.; Cryan, J.F.; Dinan, T.G. Probiotics and the microbiota-gut-brain axis: Focus on psychiatry. Curr. Nutr. Rep. 2020, 9, 171–182. [Google Scholar] [CrossRef]
- Rychlik, I. Composition and function of chicken gut microbiota. Animals 2020, 10, 103. [Google Scholar] [CrossRef]
- Carlessi, A.S.; Borba, L.A.; Zugno, A.I.; Quevedo, J.; Réus, G.Z. Gut microbiota-brain axis in depression: The role of neuroinflammation. Eur. J. Neurosci. 2021, 53, 222–235. [Google Scholar] [CrossRef]
- Yarandi, S.S.; Peterson, D.A.; Treisman, G.J.; Moran, T.H.; Pasricha, P.J. Modulatory effects of gut microbiota on the central nervous system: How Gut could play a role in neuropsychiatric health and diseases. J. Neurogastroenterol. Motil. 2016, 22, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Penaloza-Vazquez, A.; Ma, L.M.; Rayas-Duarte, P. Isolation and characterization of Bacillus spp. strains as potential probiotics for poultry. Can. J. Microbiol. 2019, 65, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Si, W.; Barbe, F.; Chevaux, E.; Sienkiewicz, O.; Zhao, X. Effects of novel probiotic strains of Bacillus pumilus and Bacillus subtilis on production, gut health, and immunity of broiler chickens raised under suboptimal conditions. Poult. Sci. 2021, 100. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.L.; Yan, T.; Li, X.Y.; Duan, Y.L.; Yang, X.; Yang, X.J. Effects of Bacillus subtilis and antibiotic growth promoters on the growth performance, intestinal function and gut microbiota of pullets from 0 to 6 weeks. Animal 2020, 14, 1619–1628. [Google Scholar] [CrossRef]
- Oladokun, S.; Koehler, A.; MacIsaac, J.; Ibeagha-Awemu, E.M.; Adewole, D.I. Bacillus subtilis delivery route: Effect on growth performance, intestinal morphology, cecal short-chain fatty acid concentration, and cecal microbiota in broiler chickens. Poult. Sci. 2021, 100. [Google Scholar] [CrossRef]
- Bar Shira, E.; Friedman, A. Innate immune functions of avian intestinal epithelial cells: Response to bacterial stimuli and localization of responding cells in the developing avian digestive tract. PLoS ONE 2018, 13, e0200393. [Google Scholar] [CrossRef]
- Park, I.; Zimmerman, N.P.; Smith, A.H.; Rehberger, T.G.; Lillehoj, E.P.; Lillehoj, H.S. Dietary supplementation with bacillus subtilis direct-fed microbials alters chicken intestinal metabolite levels. Front. Vet. Sci. 2020, 7, 123. [Google Scholar] [CrossRef]
- Jayaraman, S.; Das, P.P.; Saini, P.C.; Roy, B.; Chatterjee, P.N. Use of Bacillus Subtilis PB6 as a potential antibiotic growth promoter replacement in improving performance of broiler birds. Poult. Sci. 2017, 96, 2614–2622. [Google Scholar] [CrossRef]
- Wang, X.; Peebles, E.D.; Kiess, A.S.; Wamsley, K.G.S.; Zhai, W. Effects of coccidial vaccination and dietary antimicrobial alternatives on the growth performance, internal organ development, and intestinal morphology of Eimeria-challenged male broilers. Poult. Sci. 2019, 98, 2054–2065. [Google Scholar] [CrossRef]
- Abudabos, A.M.; Aljumaah, M.R.; Alkhulaifi, M.M.; Alabdullatif, A.; Suliman, G.M.; Al Sulaiman, A.R. Comparative effects of Bacillus subtilis and Bacillus licheniformis on live performance, blood metabolites and intestinal features in broiler inoculated with Salmonella infection during the finisher phase. Microb. Pathog. 2020, 139, 4. [Google Scholar] [CrossRef]
- Ankaiah, D.; Mitra, S.; Srivastava, D.; Sivagnanavelmurugan, M.; Ayyanna, R.; Jha, N.; Venkatesan, A. Probiotic characterization of bacterial strains from fermented South Indian tomato pickle and country chicken intestine having antioxidative and antiproliferative activities. J. Appl. Microbiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Galagarza, O.A.; Smith, S.A.; Drahos, D.J.; Eifert, J.D.; Williams, R.C.; Kuhn, D.D. Modulation of innate immunity in Nile tilapia (Oreochromis niloticus) by dietary supplementation of Bacillus subtilis endospores. Fish Shellfish Immunol. 2018, 83, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Li, M.; Zhang, C.; Zhang, X.; Wu, Y. Dietary Administration of the Bacillus subtilis enhances immune responses and disease resistance in chickens. Front. Microbiol. 2020, 11, 1768. [Google Scholar] [CrossRef] [PubMed]
- Gong, L.; Wang, B.; Mei, X.; Xu, H.; Qin, Y.; Li, W.; Zhou, Y. Effects of three probiotic Bacillus on growth performance, digestive enzyme activities, antioxidative capacity, serum immunity, and biochemical parameters in broilers. Anim. Sci. J. 2018, 89, 1561–1571. [Google Scholar] [CrossRef]
- Lyte, J.M.; Lyte, M. Review: Microbial endocrinology: Intersection of microbiology and neurobiology matters to swine health from infection to behavior. Animal 2019, 13, 2689–2698. [Google Scholar] [CrossRef]
- Obrenovich, M.E.M. Leaky Gut, Leaky Brain? Microorganisms 2018, 6, 107. [Google Scholar] [CrossRef]
- Kraimi, N.; Dawkins, M.; Gebhardt-Henrich, S.G.; Velge, P.; Rychlik, I.; Volf, J.; Creach, P.; Smith, A.; Colles, F.; Leterrier, C. Influence of the microbiota-gut-brain axis on behavior and welfare in farm animals: A review. Physiol. Behav. 2019, 210, 12. [Google Scholar] [CrossRef]
- Soroko, M.; Zaborski, D. Investigation of the effects of probiotic, Bacillus subtilis on stress reactions in laying hens using infrared thermography. PLoS ONE 2020, 15, e0234117. [Google Scholar] [CrossRef]
- D’Amelio, P.; Sassi, F. Gut microbiota, immune system, and bone. Calcif. Tissue Int. 2018, 102, 415–425. [Google Scholar] [CrossRef]
- McGinty, T.; Mallon, P.W.G. Fractures and the gut microbiome. Curr. Opin. HIV AIDS 2018, 13, 28–37. [Google Scholar] [CrossRef]
- Hao, M.L.; Wang, G.Y.; Zuo, X.Q.; Qu, C.J.; Yao, B.C.; Wang, D.L. Gut microbiota: An overlooked factor that plays a significant role in osteoporosis. J. Int. Med. Res. 2019, 47, 4095–4103. [Google Scholar] [CrossRef]
- Bizzoca, D.; Solarino, G.; Vicenti, G.; Moretti, L.; Nappi, V.S.; Belluati, A.; Moretti, B. Novel directions in the study of osteoporosis: Focus on gut microbiota as a potential therapeutic target. J. Biol. Regul. Homeost. Agents 2020, 34, 29–35. [Google Scholar] [PubMed]
- Li, S.; Mao, Y.; Zhou, F.; Yang, H.; Shi, Q.; Meng, B. Gut microbiome and osteoporosis: A review. Bone Jt. Res. 2020, 9, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.L.; Rios-Arce, N.D.; Schepper, J.D.; Parameswaran, N.; McCabe, L.R. The potential of probiotics as a therapy for osteoporosis. Microbiol. Spectr. 2017, 5, 10. [Google Scholar]
- Sgambato, D.; Gimigliano, F.; De Musis, C.; Moretti, A.; Toro, G.; Ferrante, E.; Miranda, A.; De Mauro, D.; Romano, L.; Iolascon, G.; et al. Bone alterations in inflammatory bowel diseases. World J. Clin. Cases 2019, 7, 1908–1925. [Google Scholar] [CrossRef]
- Yatsonsky Ii, D.; Pan, K.; Shendge, V.B.; Liu, J.; Ebraheim, N.A. Linkage of microbiota and osteoporosis: A mini literature review. World J. Orthop. 2019, 10, 123–127. [Google Scholar] [PubMed]
- Xu, Z.; Xie, Z.; Sun, J.; Huang, S.; Chen, Y.; Li, C.; Sun, X.; Xia, B.; Tian, L.; Guo, C.; et al. Gut microbiome reveals specific dysbiosis in primary osteoporosis. Front. Cell. Infect. Microbiol. 2020, 10, 160. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Cao, G.; Yuan, S.; Luo, C.; Yu, J.; Cai, M. Probiotic supplements and bone health in postmenopausal women: A meta-analysis of randomised controlled trials. BMJ Open 2021, 11, e041393. [Google Scholar] [CrossRef]
- Yan, J.; Charles, J.F. Gut Microbiome and Bone: To Build, Destroy, or Both? Curr. Osteoporos. Rep. 2017, 15, 376–384. [Google Scholar] [CrossRef]
- Rios-Arce, N.D.; Schepper, J.D.; Dagenais, A.; Schaefer, L.; Daly-Seiler, C.S.; Gardinier, J.D.; Britton, R.A.; McCabe, L.R.; Parameswaran, N. Post-antibiotic gut dysbiosis-induced trabecular bone loss is dependent on lymphocytes. Bone 2020, 134, 115269. [Google Scholar] [CrossRef]
- Schepper, J.D.; Collins, F.; Rios-Arce, N.D.; Kang, H.J.; Schaefer, L.; Gardinier, J.D.; Raghuvanshi, R.; Quinn, R.A.; Britton, R.; Parameswaran, N.; et al. Involvement of the gut microbiota and barrier function in glucocorticoid-induced osteoporosis. J. Bone Miner. Res. 2020, 35, 801–820. [Google Scholar] [CrossRef]
- Jones, R.M.; Mulle, J.G.; Pacifici, R. Osteomicrobiology: The influence of gut microbiota on bone in health and disease. Bone 2018, 115, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, C.; Sjögren, K. Osteomicrobiology: A new cross-disciplinary research field. Calcif. Tissue Int. 2018, 102, 426–432. [Google Scholar] [CrossRef] [PubMed]
- D’Amelio, P.; Sassi, F. Osteoimmunology: From mice to humans. BoneKEy Rep. 2016, 5, 802. [Google Scholar] [CrossRef] [PubMed]
- Huidrom, S.; Beg, M.A.; Masood, T. Post-menopausal osteoporosis and probiotics. Curr. Drug Targets 2021, 22, 816. [Google Scholar] [CrossRef]
- Yang, L.C.; Lin, S.W.; Li, I.C.; Chen, Y.P.; Tzu, S.Y.; Chou, W.; Chen, C.C.; Lin, W.C.; Chen, Y.L.; Lin, W.H. Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation. Nutrients 2020, 12, 1914. [Google Scholar] [CrossRef]
- Hor, Y.Y.; Ooi, C.H.; Lew, L.C.; Jaafar, M.H.; Lau, A.S.; Lee, B.K.; Azlan, A.; Choi, S.B.; Azzam, G.; Liong, M. The molecular mechanisms of probiotic strains in improving ageing bone and muscle of d-galactose-induced ageing rats. J. Appl. Microbiol. 2021, 130, 1307–1322. [Google Scholar] [CrossRef]
- Ruiz Sella, S.R.B.; Bueno, T.; de Oliveira, A.A.B.; Karp, S.G.; Soccol, C.R. Bacillus subtilis natto as a potential probiotic in animal nutrition. Crit. Rev. Biotechnol. 2021, 41, 355–369. [Google Scholar] [CrossRef]
- Ozdamar, T.H.; Sentürk, B.; Yilmaz, O.D.; Calik, G.; Celik, E.; Calik, P. Expression system for recombinant human growth hormone production from Bacillus subtilis. Biotechnol. Prog. 2009, 25, 75–84. [Google Scholar] [CrossRef]
- Şahin, B.; Öztürk, S.; Çalık, P.; Özdamar, T.H. Feeding strategy design for recombinant human growth hormone production by Bacillus subtilis. Bioprocess Biosyst. Eng. 2015, 38, 1855–1865. [Google Scholar] [CrossRef]
- Karimi, M.; Behzadian, F.; Rouhaninejad, H.; Yari, S. A feasibility study to evaluate Bacillus subtilis as a host for producing recombinant human parathyroid hormone. Avicenna J. Med. Biotechnol. 2018, 10, 147–151. [Google Scholar] [PubMed]
- Shasaltaneh, M.D.; Moosavi-Nejad, Z.; Gharavi, S.; Fooladi, J. Cane molasses as a source of precursors in the bioproduction of tryptophan by Bacillus subtilis. Iran. J. Microbiol. 2013, 5, 285–292. [Google Scholar] [PubMed]
- McAdams, N.M.; Gollnick, P. The Bacillus subtilis TRAP protein can induce transcription termination in the leader region of the tryptophan biosynthetic (trp) operon independent of the trp attenuator RNA. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Bjerre, K.; Cantor, M.D.; Nørgaard, J.V.; Poulsen, H.D.; Blaabjerg, K.; Canibe, N.; Jensen, B.B.; Stuer-Lauridsen, B.; Nielsen, B.; Derkx, P.M. Development of Bacillus subtilis mutants to produce tryptophan in pigs. Biotechnol. Lett. 2017, 39, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Mu, C.L.; Farzi, A.; Zhu, W.Y. Tryptophan Metabolism: A link between the gut microbiota and brain. Adv. Nutr. 2020, 11, 709–723. [Google Scholar] [CrossRef]
- Driessler, F.; Baldock, P.A. Hypothalamic regulation of bone. J. Mol. Endocrinol. 2010, 45, 175. [Google Scholar] [CrossRef]
- Ducy, P. 5-HT and bone biology. Curr. Opin. Pharmacol. 2011, 11, 34–38. [Google Scholar] [CrossRef]
- Lavoie, B.; Lian, J.B.; Mawe, G.M. Regulation of bone metabolism by serotonin. Adv. Exp. Med. Biol. 2017, 1033, 35–46. [Google Scholar]
- Amin, N.; Boccardi, V.; Taghizadeh, M.; Jafarnejad, S. Probiotics and bone disorders: The role of RANKL/RANK/OPG pathway. Aging Clin. Exp. Res. 2020, 32, 363–371. [Google Scholar] [CrossRef]
- Takimoto, T.; Hatanaka, M.; Hoshino, T.; Takara, T.; Tanaka, K.; Shimizu, A.; Morita, H.; Nakamura, T. Effect of Bacillus subtilis C-3102 on bone mineral density in healthy postmenopausal Japanese women: A randomized, placebo-controlled, double-blind clinical trial. Biosci. Microbiota Food Health 2018, 37, 87–96. [Google Scholar] [CrossRef]
- Abdelqader, A.; Abuajamieh, M.; Hayajneh, F.; Al-Fataftah, A.R. Probiotic bacteria maintain normal growth mechanisms of heat stressed broiler chickens. J. Therm. Biol. 2020, 92, 102654. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.A.; Zaki, R.S.; Negm, E.A.; Mahmoud, M.A.; Cheng, H.W. Effects of dietary supplementation of a probiotic (Bacillus subtilis) on bone mass and meat quality of broiler chickens. Poult. Sci. 2020, 100. [Google Scholar] [CrossRef]
- Ciurescu, G.; Dumitru, M.; Gheorghe, A.; Untea, A.E.; Drăghici, R. Effect of Bacillus subtilis on growth performance, bone mineralization, and bacterial population of broilers fed with different protein sources. Poult. Sci. 2020, 99, 5960–5971. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.F.; Wang, W.C.; Cheng, H.W. Bacillus subtilis-based probiotic promotes bone growth by inhibition of inflammation in broilers subjected to cyclic heating episodes. Poult. Sci. 2020, 99, 5252–5260. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Xv, J.; Li, Y.; Bi, Y.; Hou, Y.; Ding, B. Interactive effects of dietary vitamin K(3) and Bacillus subtilis PB6 on the growth performance and tibia quality of broiler chickens with sex separate rearing. Animal 2020, 14, 1610–1618. [Google Scholar] [CrossRef] [PubMed]
- Latorre, J.D.; Hernandez-Velasco, X.; Bielke, L.R.; Vicente, J.L.; Wolfenden, R.; Menconi, A.; Hargis, B.M.; Tellez, G. Evaluation of a Bacillus direct-fed microbial candidate on digesta viscosity, bacterial translocation, microbiota composition and bone mineralisation in broiler chickens fed on a rye-based diet. Br. Poult. Sci. 2015, 56, 723–732. [Google Scholar] [CrossRef]
- Latorre, J.D.; Hernandez-Velasco, X.; Vicente, J.L.; Wolfenden, R.; Hargis, B.M.; Tellez, G. Effects of the inclusion of a Bacillus direct-fed microbial on performance parameters, bone quality, recovered gut microflora, and intestinal morphology in broilers consuming a grower diet containing corn distillers dried grains with solubles. Poult. Sci. 2017, 96, 2728–2735. [Google Scholar] [CrossRef]
- Sadeghi, A.A. Bone Mineralization of broiler chicks challenged with Salmonella enteritidis fed diet containing probiotic (Bacillus subtilis). Probiotics Antimicrob. Proteins 2014, 6, 136–140. [Google Scholar] [CrossRef]
- Mutuş, R.; Kocabagli, N.; Alp, M.; Acar, N.; Eren, M.; Gezen, S.S. The effect of dietary probiotic supplementation on tibial bone characteristics and strength in broilers. Poult. Sci. 2006, 85, 1621–1625. [Google Scholar] [CrossRef]
- Abdelqader, A.; Irshaid, R.; Al-Fataftah, A.R. Effects of dietary probiotic inclusion on performance, eggshell quality, cecal microflora composition, and tibia traits of laying hens in the late phase of production. Trop. Anim. Health Prod. 2013, 45, 1017–1024. [Google Scholar] [CrossRef]
- Alam, S.; Masood, S.; Zaneb, H.; Rabbani, I.; Khan, R.U.; Shah, M.; Ashraf, S.; Alhidary, I.A. Effect of Bacillus cereus and phytase on the expression of musculoskeletal strength and gut health in japanese quail (coturnix japonica). J. Poult. Sci. 2020, 57, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Dar, H.Y.; Pal, S.; Shukla, P.; Mishra, P.K.; Tomar, G.B.; Chattopadhyay, N.; Srivastava, R.K. Bacillus clausii inhibits bone loss by skewing Treg-Th17 cell equilibrium in postmenopausal osteoporotic mice model. Nutrition 2018, 54, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Pazzini, C.A.; Pereira, L.J.; da Silva, T.A.; Montalvany-Antonucci, C.C.; Macari, S.; Marques, L.S.; de Paiva, S.M. Probiotic consumption decreases the number of osteoclasts during orthodontic movement in mice. Arch. Oral Biol. 2017, 79, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Messora, M.R.; Pereira, L.J.; Foureaux, R.; Oliveira, L.F.; Sordi, C.G.; Alves, A.J.; Napimoga, M.H.; Nagata, M.J.; Ervolino, E.; Furlaneto, F.A. Favourable effects of Bacillus subtilis and Bacillus licheniformis on experimental periodontitis in rats. Arch. Oral Biol. 2016, 66, 108–119. [Google Scholar] [CrossRef]
- Messora, M.R.; Oliveira, L.F.; Foureaux, R.C.; Taba, M., Jr.; Zangerônimo, M.G.; Furlaneto, F.A.; Pereira, L.J. Probiotic therapy reduces periodontal tissue destruction and improves the intestinal morphology in rats with ligature-induced periodontitis. J. Periodontol. 2013, 84, 1818–1826. [Google Scholar] [CrossRef]
- Elefteriou, F.; Ahn, J.D.; Takeda, S.; Starbuck, M.; Yang, X.; Liu, X.; Kondo, H.; Richards, W.G.; Bannon, T.W.; Noda, M.; et al. Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature 2005, 434, 514–520. [Google Scholar] [CrossRef]
- Kajimura, D.; Hinoi, E.; Ferron, M.; Kode, A.; Riley, K.J.; Zhou, B.; Guo, X.E.; Karsenty, G. Genetic determination of the cellular basis of the sympathetic regulation of bone mass accrual. J. Exp. Med. 2011, 208, 841–851. [Google Scholar] [CrossRef]
- Fu, L.; Patel, M.S.; Bradley, A.; Wagner, E.F.; Karsenty, G. The molecular clock mediates leptin-regulated bone formation. Cell 2005, 122, 803–815. [Google Scholar] [CrossRef]
- Bonnet, N.; Benhamou, C.L.; Brunet-Imbault, B.; Arlettaz, A.; Horcajada, M.N.; Richard, O.; Vico, L.; Collomp, K.; Courteix, D. Severe bone alterations under beta2 agonist treatments: Bone mass, microarchitecture and strength analyses in female rats. Bone 2005, 37, 622–633. [Google Scholar] [CrossRef]
- Niedermair, T.; Kuhn, V.; Doranehgard, F.; Stange, R.; Wieskötter, B.; Beckmann, J.; Salmen, P.; Springorum, H.R.; Straub, R.H.; Zimmer, A.; et al. Absence of substance P and the sympathetic nervous system impact on bone structure and chondrocyte differentiation in an adult model of endochondral ossification. Matrix Biol. 2014, 38, 22–35. [Google Scholar] [CrossRef]
- Karl, J.P.; Hatch, A.M.; Arcidiacono, S.M.; Pearce, S.C.; Pantoja-Feliciano, I.G.; Doherty, L.A.; Soares, J.W. Effects of psychological, environmental and physical stressors on the gut microbiota. Front. Microbiol. 2018, 9, 2013. [Google Scholar] [CrossRef]
- Laudisi, F.; Stolfi, C.; Monteleone, G. Impact of Food Additives on Gut Homeostasis. Nutrients 2019, 11, 2334. [Google Scholar] [CrossRef] [PubMed]
- Molina-Torres, G.; Rodriguez-Arrastia, M.; Roman, P.; Sanchez-Labraca, N.; Cardona, D. Stress and the gut microbiota-brain axis. Behav. Pharmacol. 2019, 30, 187–200. [Google Scholar] [CrossRef]
- Liu, H.; Gu, R.; Li, W.; Zhou, W.; Cong, Z.; Xue, J.; Liu, Y.; Wei, Q.; Zhou, Y. Lactobacillus rhamnosus GG attenuates tenofovir disoproxil fumarate-induced bone loss in male mice via gut-microbiota-dependent anti-inflammation. Ther. Adv. Chronic Dis. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.C.; Yan, F.F.; Hu, J.Y.; Amen, O.A.; Cheng, H.W. Supplementation of Bacillus subtilis-based probiotic reduces heat stress-related behaviors and inflammatory response in broiler chickens. J. Anim. Sci. 2018, 96, 1654–1666. [Google Scholar] [CrossRef] [PubMed]
- Cramer, T.A.; Kim, H.W.; Chao, Y.; Wang, W.; Cheng, H.W.; Kim, Y.H.B. Effects of probiotic (Bacillus subtilis) supplementation on meat quality characteristics of breast muscle from broilers exposed to chronic heat stress. Poult. Sci. 2018, 97, 3358–3368. [Google Scholar] [CrossRef]
- Park, I.; Lee, Y.; Goo, D.; Zimmerman, N.P.; Smith, A.H.; Rehberger, T.; Lillehoj, H.S. The effects of dietary Bacillus subtilis supplementation, as an alternative to antibiotics, on growth performance, intestinal immunity, and epithelial barrier integrity in broiler chickens infected with Eimeria maxima. Poult. Sci. 2020, 99, 725–733. [Google Scholar] [CrossRef]
- Whelan, R.A.; Doranalli, K.; Rinttilä, T.; Vienola, K.; Jurgens, G.; Apajalahti, J. The impact of Bacillus subtilis DSM 32315 on the pathology, performance, and intestinal microbiome of broiler chickens in a necrotic enteritis challenge. Poult. Sci. 2019, 98, 3450–3463. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhou, F.; Yuan, P.; Dou, G.; Liu, X.; Liu, S.; Wang, X.; Jin, R.; Dong, Y.; Zhou, J. T cell-depleting nanoparticles ameliorate bone loss by reducing activated T cells and regulating the Treg/Th17 balance. Bioact. Mater. 2021, 6, 3150–3163. [Google Scholar] [CrossRef]
- Mansoori, M.N.; Raghuvanshi, A.; Shukla, P.; Awasthi, P.; Trivedi, R.; Goel, A.; Singh, D. Medicarpin prevents arthritis in post-menopausal conditions by arresting the expansion of TH17 cells and pro-inflammatory cytokines. Int. Immunopharmacol. 2020, 82, 106299. [Google Scholar] [CrossRef]
- Sapra, L.; Dar, H.Y.; Bhardwaj, A.; Pandey, A.; Kumari, S.; Azam, Z.; Upmanyu, V.; Anwar, A.; Shukla, P.; Mishra, P.K.; et al. Lactobacillus rhamnosus attenuates bone loss and maintains bone health by skewing Treg-Th17 cell balance in Ovx mice. Sci. Rep. 2021, 11, 1807. [Google Scholar] [CrossRef] [PubMed]
| Trade Name | Bacillus Strain(s) | Manufacturer 1 |
|---|---|---|
| Alterion® | B. subtilis | Adiddeo (Paris, France)-Novozyme(Copenhagen, Denmark). https://www.adisseo.com |
| B-ACT® | B. licheniformis | Agrihealth (Monaghan, Ireland) https://agrihealth.co.nz |
| Calsporin® | B. subtilis (C-3102) | Orffa.(Werkendam, Netherlands) https://orffa.com |
| CorrlinkTM ABS1781 | B. subtilis (B. velezensis NRRL B-67259) | Elanco Animal Health (Fort Dodge, IA, USA) https://efsa.onlinelibrary.wiley.com |
| Clostat | B. subtilis PB6 | Kemin Industries, Inc. (Des Moines, IA USA), https://www.kemin.com |
| Ecobiol® | B. amyloliquefaciens (CECT 5940) | Norel Animal Nutrition (Madrid, Spain) https://animal-nutrition.evonik.com |
| Enviva® PRO 201 C | B. subtilis | Dupont-Danisco (Palo Alto, CA, USA) https://www.chemunique.co.za |
| Enviva® PRO 202 GT | B. amyloliquefaciens (PTA-6507, NRRL B-50013, 50104) | Dupont-Danisco (Palo Alto, CA, USA) https://www.chemunique.co.za |
| FloraFix-BIRDS | B. subtilis | Biogrowcompany (Victoria, Australia) https://biogrowcompany.com/australia |
| Gallipro® MS | B. subtilis (DSM5750), B. licheniformis (DSM5749) | Chr. Hansen, Inc. (Hoersholm, Denmark) https://www.chr-hansen.com |
| Gallipro® Fit | B. subtilis (DSM 32324, 32325) B. licheniformis (DSM 25840) | Chr. Hansen, Inc. (Hoersholm, Denmark). https://www.chr-hansen.com |
| POULTRY-FEED | B. subtilis, B. licheniformis | Bionetix-International, Inc. (Quebec, Canada) www.bionextix-international.com |
| SPORULIN® | B. subtilis | Novus International, Inc. (St. Charles, MI, USA) http://www.novusint.com |
| Toyocerin | B.cereus var toyoi | Asahi Vet S.A. (Tokyo, Japan) https://trademark.trademarkia.com |
| Strain | Species (Model) | Bone Metabolism | Biochemistry/Biological Change | Reference |
|---|---|---|---|---|
| B. subtilis C-3102 | Women | ↑hip BMD, ↓bone resorption | ↓uNTx, TRACP-5b, ↓genus Fusobacterium | [189] |
| B. subtilis | Broiler (HS) | ↑tibia traits 1 | ↑production, absorptive epithelial area, ↑serum GH, IGF-1, and cholesterol, glucose | [190] |
| B. subtilis | Broiler | ↑tibia traits | ↑plasma Ca, P levels | [191] |
| B. subtilis ATCC 6051a | Broiler | ↑Tibia P | ↓cecal Escherichia coli, Staphylococcus spp. | [192] |
| B. subtilis | Broiler (HS) | ↑tibia/femur BMC/traits | ↓serum concentration of CTX, TNF-α | [193] |
| B. subtilis PB6 | Broiler | ↑tibia traits, tibia ash, Ca, P | ↑serum OCN, BALP | [194] |
| B. subtilis B. amyloliquefecieus | Broiler | ↑tibia breaking strength mineral | ↑tibia breaking strength ↓TGNB, ↑TLAB | [195,196] |
| B. subtilis | Broiler (SE) | ↑tibia ash and Ca | - | [197] |
| B. subtilis B. licheniformis | Broiler | ↑tibia lateral and medial wall tibiotarsal index, tibia ash and P | - | [198] |
| B. subtilis | Laying hen | ↑tibia traits | ↑gut microbiota balance, egg traits 2 | [199] |
| B. cereus | Quail | ↑tibia traits | ↑absorptive epithelial area | [200] |
| B. Clausii | Mouse (OVX) | ↑bone mass, BMD, ↑bone microarchitecture | ↑Treg cells↓Th17 cells, ↑anti-inflammatory cytokines, ↓proinflammatory cytokines | [201] |
| B. subtilis | Mouse (OTM) | ↓osteoclasts,↑osteoblasts | - | [202] |
| B. subtilis B. licheniformis | Rat (periodontitis) | ↓bone loss | ↓inflammation | [203] |
| B. subtilis | Rat (periodontitis) | ↓bone loss | ↓inflammation | [204] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, S.; Yan, F.-F.; Hu, J.-Y.; Mohammed, A.; Cheng, H.-W. Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress. Animals 2021, 11, 1494. https://doi.org/10.3390/ani11061494
Jiang S, Yan F-F, Hu J-Y, Mohammed A, Cheng H-W. Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress. Animals. 2021; 11(6):1494. https://doi.org/10.3390/ani11061494
Chicago/Turabian StyleJiang, Sha, Fei-Fei Yan, Jia-Ying Hu, Ahmed Mohammed, and Heng-Wei Cheng. 2021. "Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress" Animals 11, no. 6: 1494. https://doi.org/10.3390/ani11061494
APA StyleJiang, S., Yan, F.-F., Hu, J.-Y., Mohammed, A., & Cheng, H.-W. (2021). Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress. Animals, 11(6), 1494. https://doi.org/10.3390/ani11061494





