Alterations of the Muscular Fatty Acid Composition and Serum Metabolome in Bama Xiang Mini-Pigs Exposed to Dietary Beta-Hydroxy Beta-Methyl Butyrate
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Sample Collection
2.3. Measurements of Serum Amino Acids and Lipid-Related Substances
2.4. Intramuscular Fat and Fatty Acid Composition
2.5. Quantitative Real-Time PCR Analysis
2.6. Liquid Chromatography—Tandem Mass Spectrometry (LC-MS/MS) Analysis
2.7. Statistical Analysis
3. Results
3.1. Growth Performance and IMF
3.2. Serum Amino Acid Profile and Lipid-Related Substances
3.3. Fatty Acid Composition of Skeletal Muscle Tissue
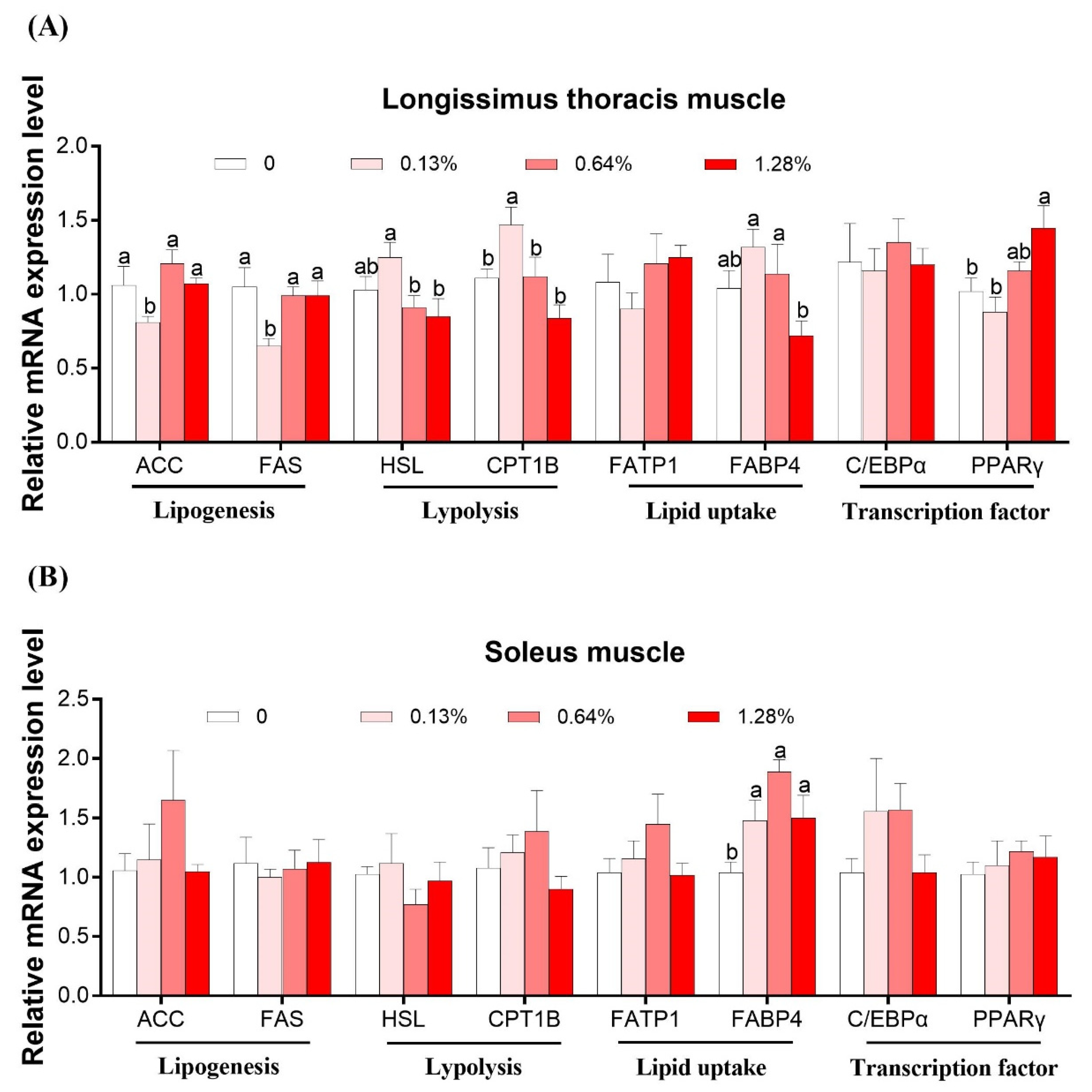
3.4. mRNA Expression of Lipid Metabolism-Related Genes in Skeletal Muscles
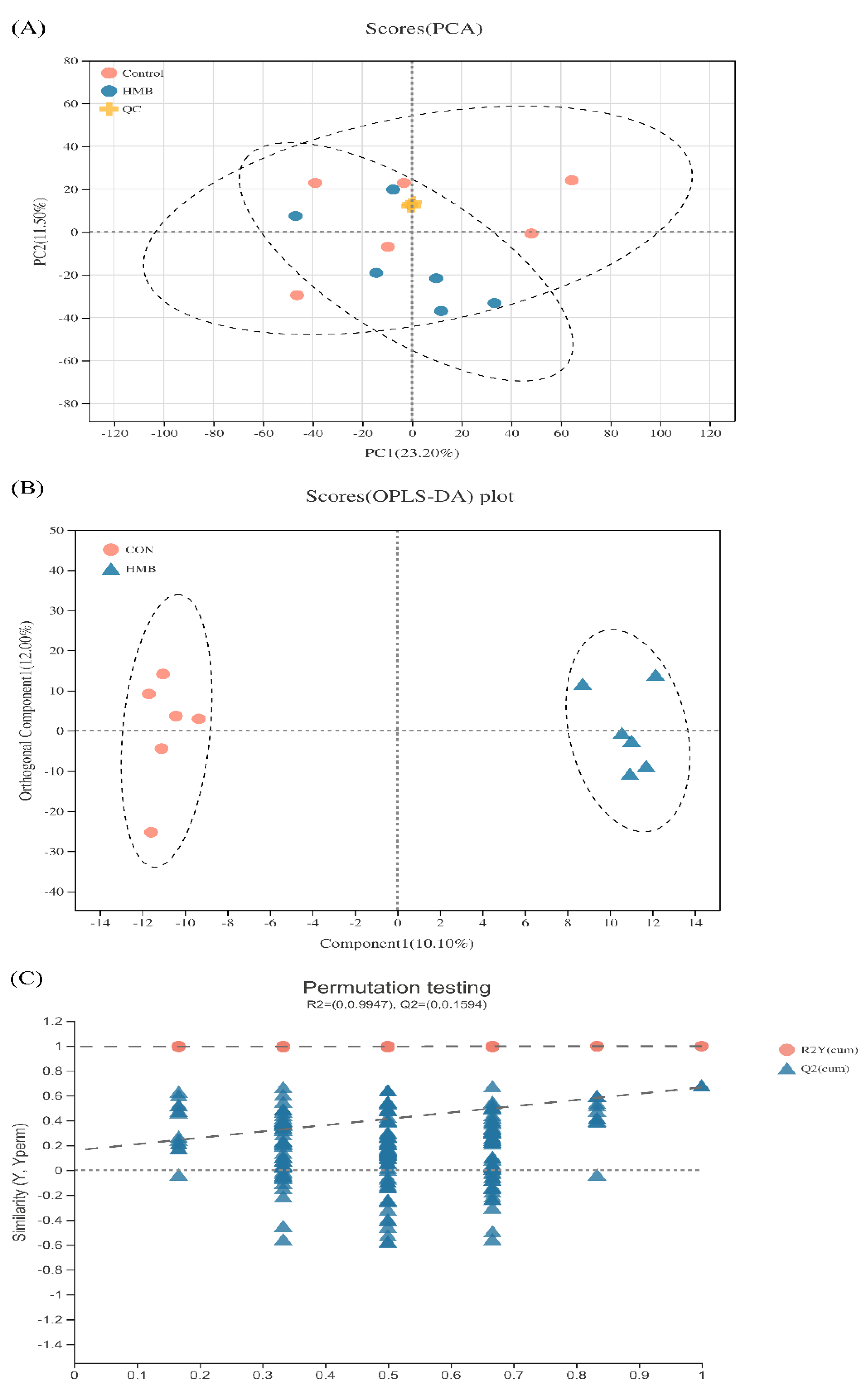
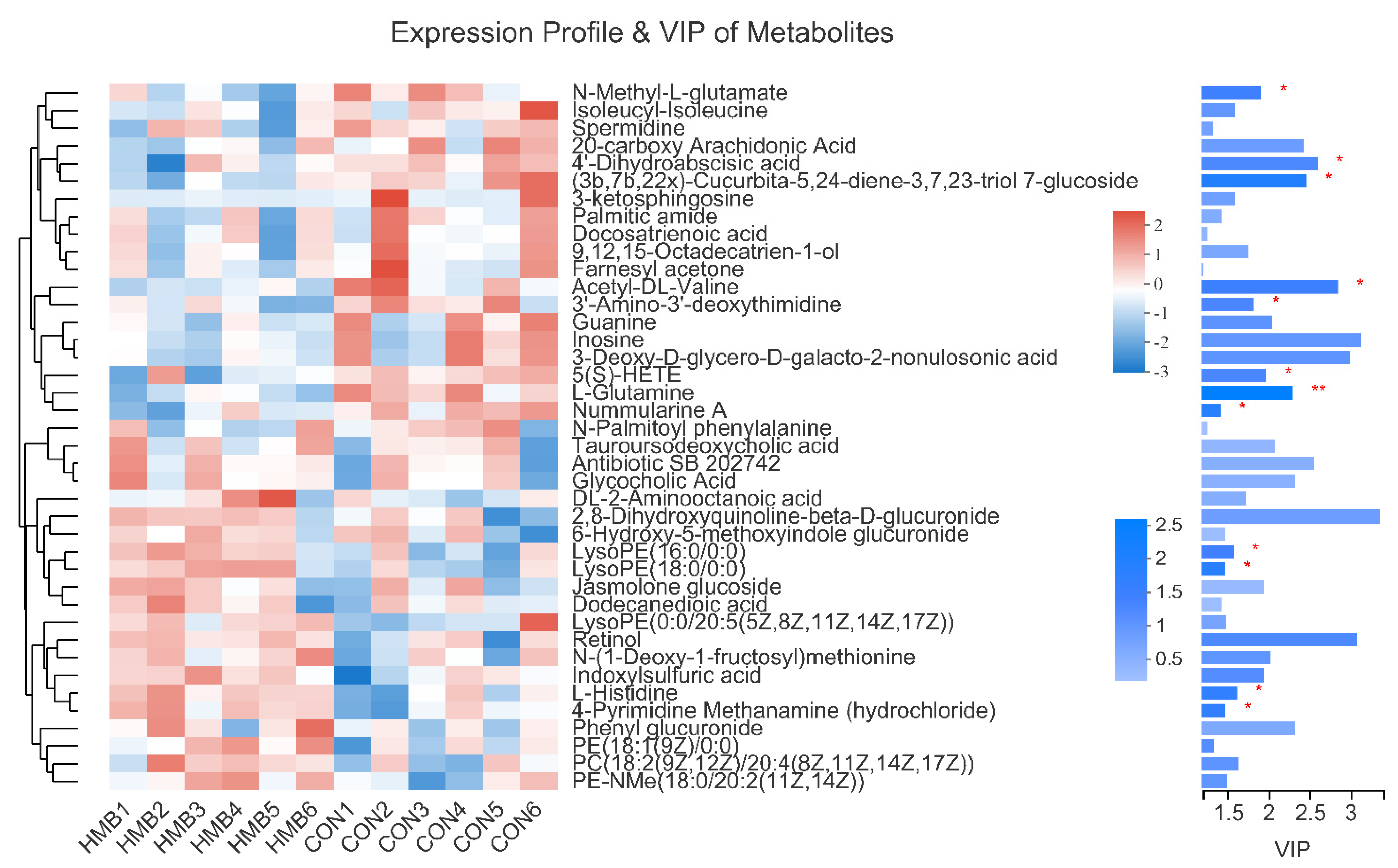
3.5. Metabolomics Analysis
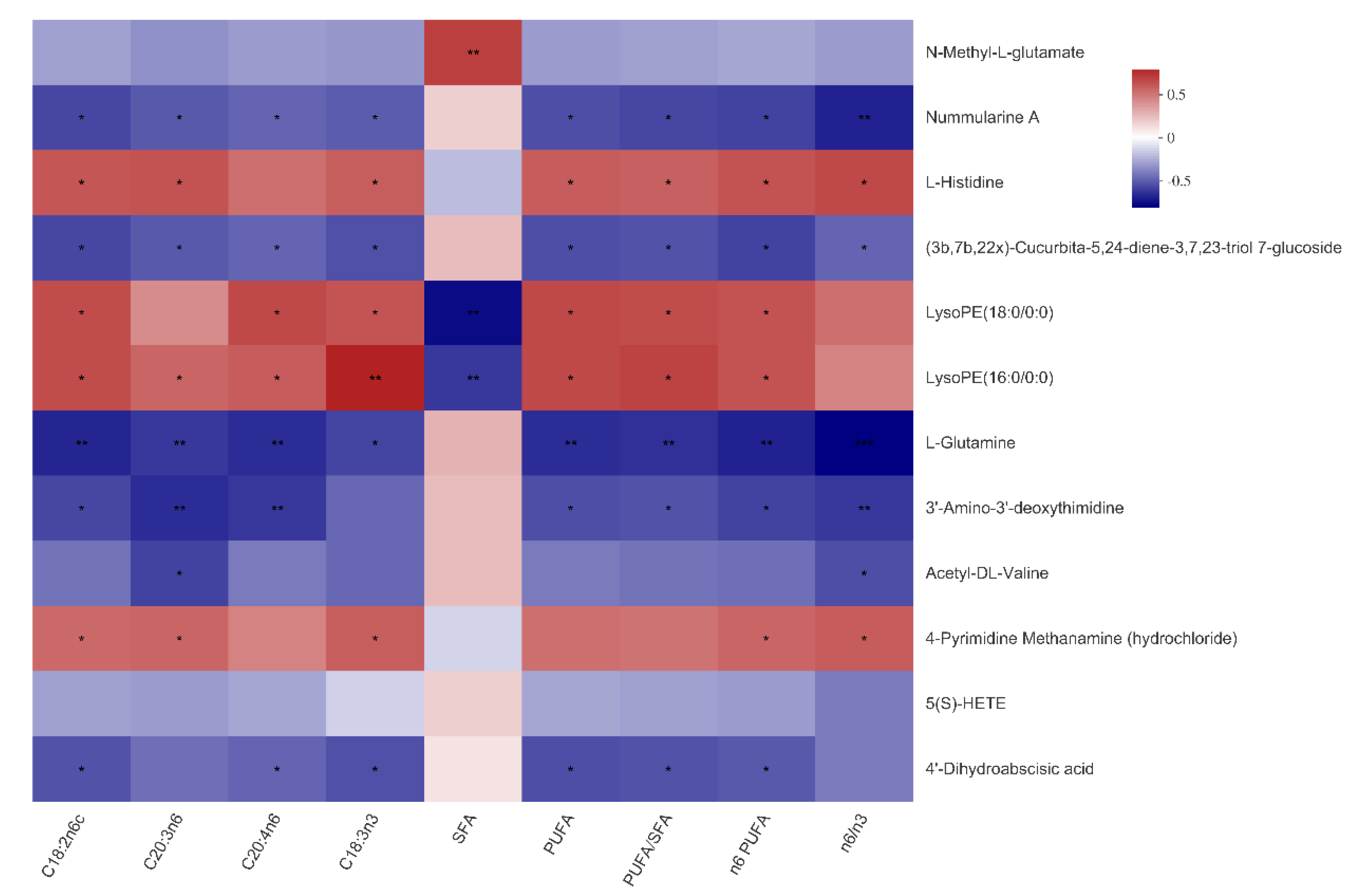
3.6. The Relationship between Serum Metabolites and Fatty Acid Composition of Muscles
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Matarneh, S.K.; Silva, S.L.; Gerrard, D.E. New insights in muscle biology that alter meat quality. Annu. Rev. Anim. Biosci. 2021, 9, 355–377. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.H.; Duan, Y.M.; Li, F.N.; Li, Y.H.; Guo, Q.P.; Ji, Y.J.; Tan, B.E.; Li, T.J.; Yin, Y.L. Effects of supplementation with branched-chain amino acids to low-protein diets on expression of genes related to lipid metabolism in skeletal muscle of growing pigs. Amino Acids 2016, 48, 2131–2144. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, H.; Gao, H.; Kang, B.; Chen, F.; Li, Y.; Fu, C.; Yao, K. Effects of dietary supplementation of alpha-ketoglutarate in a low-protein diet on fatty acid composition and lipid metabolism related gene expression in muscles of growing pigs. Animals 2019, 9, 838. [Google Scholar] [CrossRef] [PubMed]
- Dannenberger, D.; Nuernberg, G.; Nuernberg, K.; Katja, W.; Nicolas, S.; Marion, S. Effects of diets supplemented with n –3 or n –6 PUFA on pig muscle lipid metabolites measured by non-targeted LC–MS lipidomic profiling. J. Food Compos. Anal. 2017, 56, 47–54. [Google Scholar] [CrossRef]
- Cameron, N.D.; Enser, M.; Nute, G.R.; Whittington, F.M.; Penman, J.C.; Perry, A.M.; Wood, J.D. Genotype with nutrition interaction on fatty acid composition of intramuscular fat and the relationship with flavour of pig meat. Meat Sci. 2000, 55, 187–195. [Google Scholar] [CrossRef]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Sheard, P.R.; Richardson, R.I.; Hughes, S.I.; Whittington, F.M. Fat deposition, fatty acid composition and meat quality: A review. Meat Sci. 2008, 78, 343–358. [Google Scholar] [CrossRef] [PubMed]
- Okrouhlá, M.; Stupka, R.; Čítek, J.; Šprysl, M.; Trnka, M.; Kluzáková, E. Effect of lean meat proportion on the chemical composition of pork. Czech J. Food Sci. 2008, 26, 464–469. [Google Scholar] [CrossRef]
- Devol, D.L.; Mckeith, F.K.; Bechtel, P.J.; Novakofski, J.; Shanks, R.D.; Carr, T.R. Variation in composition and palatability traits and relationships between muscle characteristics and palatability in a random sample of pork carcasses. J. Anim. Sci. 1988, 66, 385–395. [Google Scholar] [CrossRef]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef]
- Hu, F.; Stampfer, M.J.; Manson, J.E.; Ascherio, A.; Colditz, G.A.; Speizer, F.E.; Hennekens, C.H.; Willett, W.C. Dietary saturated fats and their food sources in relation to the risk of coronary heart disease in women. Am. J. Clin. Nutr. 1999, 70, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Zeng, X.; Zhu, J.; Liu, Y.; Ye, Q.; Qiao, S.; Zeng, X. Dietary N-carbamylglutamate supplementation in a reduced protein diet affects carcass traits and the profile of muscle amino acids and fatty acids in finishing pigs. J. Agric. Food. Chem. 2017, 65, 5751–5758. [Google Scholar] [CrossRef] [PubMed]
- Holecek, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopeni 2017, 8, 529–541. [Google Scholar] [CrossRef]
- Duan, Y.H.; Zeng, L.M.; Li, F.N.; Kong, X.F.; Xu, K.; Guo, Q.P.; Wang, W.L.; Zhang, L.Y. Beta-hydroxy-beta-methyl butyrate promotes leucine metabolism and improves muscle fibre composition in growing pigs. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1328–1339. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.H.; Li, F.N.; Song, B.; Zheng, C.B.; Zhong, Y.Z.; Xu, K.; Kong, X.F.; Yin, Y.L.; Wang, W.C.; Shu, G. β-hydroxy-β-methyl butyrate, but not α-ketoisocaproate and excess leucine, stimulates skeletal muscle protein metabolism in growing pigs fed low-protein diets. J. Funct. Foods 2019, 52, 34–42. [Google Scholar] [CrossRef]
- Duan, Y.H.; Zhang, L.Y.; Li, F.N.; Guo, Q.P.; Yin, Y.L.; Kong, X.F.; Peng, M.J.; Wang, W.C. Beta-Hydroxy-beta-methylbutyrate modulates lipid metabolism in adipose tissues of growing pigs. Food Funct. 2018, 9, 4836–4846. [Google Scholar] [CrossRef]
- Duan, Y.H.; Zhong, Y.Z.; Xiao, H.; Zheng, C.B.; Song, B.; Wang, W.L.; Guo, Q.P.; Li, Y.Y.; Han, H.; Gao, J.; et al. Gut microbiota mediates the protective effects of dietary beta-hydroxy-beta-methylbutyrate (HMB) against obesity induced by high-fat diets. FASEB J. 2019, 33, 10019–10033. [Google Scholar] [CrossRef]
- Panton, L.B.; Rathmacher, J.A.; Baier, S.; Nissen, D. Nutritional supplementation of the leucine metabolite b-Hydroxy-b-Methylbutyrate (HMB) during resistance training. Nutrition 2000, 16, 734–739. [Google Scholar] [CrossRef]
- Yao, K.; Duan, Y.H.; Li, F.N.; Tan, B.E.; Hou, Y.Q.; Wu, G.Y.; Yin, Y.L. Leucine in obesity: Therapeutic prospects. Trends Pharmacol. Sci. 2016, 37, 714–727. [Google Scholar] [CrossRef]
- Duan, Y.H.; Li, F.N.; Li, Y.H.; Tang, Y.L.; Kong, X.F.; Feng, Z.M.; Anthony, T.G.; Walford, M.; Hou, Y.Q.; Wu, G.Y.; et al. The role of leucine and its metabolites in protein and energy metabolism. Amino Acids 2016, 48, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Baira, E.; Dagla, I.; Siapi, E.; Zoumpoulakis, P.; Simitzis, P.; Goliomytis, M.; Deligeorgis, S.G.; Skaltsounis, A.L.; Gikas, E. UHPLC-HRMS-based tissue untargeted metabolomics study of naringin and hesperidin after dietary supplementation in chickens. Food Chem. 2018, 269, 276–285. [Google Scholar] [CrossRef]
- Zhu, X.; Zhan, Q.; Wei, Y.; Yan, A.; Feng, Q.; Liu, L.; Lu, S.; Tang, D. CRISPR/Cas9-mediated MSTN disruption accelerates the growth of Chinese Bama pigs. Reprod. Domest. Anim. 2020, 55, 1314–1327. [Google Scholar] [CrossRef] [PubMed]
- Xiang, G.; Ren, J.; Hai, T.; Fu, R.; Yu, D.; Wang, J.; Li, W.; Wang, H.; Zhou, Q. Editing porcine IGF2 regulatory element improved meat production in Chinese Bama pigs. Cell. Mol. Life Sci. 2018, 75, 4619–4628. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Liu, Y. Research advances on nutrient requirements of laboratory minipigs. Chin. J. Comp. Med. 2009, 19, 74–78. [Google Scholar]
- Liu, Y.; Kong, X.; Li, F.; Tan, B.; Li, Y.; Duan, Y.; Yin, Y.; He, J.; Hu, C.; Blachier, F.; et al. Co-dependence of genotype and dietary protein intake to affect expression on amino acid/peptide transporters in porcine skeletal muscle. Amino Acids 2016, 48, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Li, F.N.; Duan, Y.H.; Li, Y.H.; Tang, Y.L.; Geng, M.M.; Oladele, O.A.; Kim, S.W.; Yin, Y.L. Effects of dietary n-6:n-3 PUFA ratio on fatty acid composition, free amino acid profile and gene expression of transporters in finishing pigs. Br. J. Nutr. 2015, 113, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.H.; Li, F.N.; Tan, B.E.; Lin, B.B.; Kong, X.F.; Li, Y.H.; Yin, Y.L. Myokine interleukin-15 expression profile is different in suckling and weaning piglets. Anim. Nutr. 2015, 1, 30–35. [Google Scholar] [CrossRef]
- Wang, W.; Han, Z.; Guo, D.; Xiang, Y. UHPLC-QTOFMS-based metabolomic analysis of serum and urine in rats treated with musalais containing varying ethyl carbamate content. Anal. Bioanal. Chem. 2020, 412, 7627–7637. [Google Scholar] [CrossRef] [PubMed]
- Wheatley, S.M.; El-Kadi, S.W.; Suryawan, A.; Boutry, C.; Orellana, R.A.; Nguyen, H.V.; Davis, S.R.; Davis, A. Protein synthesis in skeletal muscle of neonatal pigs is enhanced by administration of beta-hydroxy-beta-methylbutyrate. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E91–E99. [Google Scholar] [CrossRef]
- Duan, Y.H.; Zheng, C.B.; Zhong, Y.Z.; Song, B.; Yan, Z.M.; Kong, X.F.; Deng, J.P.; Li, F.N.; Yin, Y.L. Beta-hydroxy beta-methyl butyrate decreases muscle protein degradation via increased Akt/FoxO3a signaling and mitochondrial biogenesis in weanling piglets after lipopolysaccharide challenge. Food Funct. 2019, 10, 5152–5165. [Google Scholar] [CrossRef]
- Duan, Y.H.; Li, F.N.; Tan, B.E.; Yao, K.; Yin, Y.L. Metabolic control of myofibers: Promising therapeutic target for obesity and type 2 diabetes. Obes. Rev. 2017, 18, 647–659. [Google Scholar] [CrossRef] [PubMed]
- Rhee, K.S.; Waldron, D.F.; Ziprin, Y.A.; Rhee, K.C. Fatty acid composition of goat diets vs intramuscular fat. Meat Sci. 2000, 54, 313–318. [Google Scholar] [CrossRef]
- Perry, D.; Nicholls, P.J.; Thompson, J.M. The effect of sire breed on the melting point and fatty acid composition of subcutaneous fat in steers. J. Anim. Sci. 1998, 76, 87–95. [Google Scholar] [CrossRef]
- Zhong, Y.; Song, B.; Zheng, C.; Li, F.; Kong, X.; Duan, Y.; Deng, J. α-Ketoisocaproate and β-hydroxy-β-methyl butyrate regulate fatty acid composition and lipid metabolism in skeletal muscle of growing pigs. J. Anim. Physiol. Anim. Nutr. 2019, 103, 846–857. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Liu, H.B. Nutritional indices for assessing fatty acids: A mini-review. Int. J. Mol. Sci. 2020, 21, 5695. [Google Scholar] [CrossRef] [PubMed]
- Semt, S.S.; Raes, K.; Demeyer, D. Meat fatty acid composition as affected by fatness and genetic factors: A review. Anim. Res. 2004, 53, 81–98. [Google Scholar]
- Enser, M.; Richardson, R.I.; Wood, J.D.; Gill, B.P.; Sheard, P.R. Feeding linseed to increase the n-3 PUFA of pork: Fatty acid composition of muscle, adipose tissue, liver and sausages. Meat Sci. 2000, 55, 201–212. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: Nutritional implications for chronic diseases. Biomed. Pharmacother. 2006, 60, 502–507. [Google Scholar] [CrossRef]
- Russo, G.L. Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochem. Pharmacol. 2009, 77, 937–946. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary aspects of the dietary omega-6:omega-3 fatty acid ratio: Medical implications. World Rev. Nutr. Diet 2009, 100, 1–21. [Google Scholar]
- Daniel, C.R.; McCullough, M.L.; Patel, R.C.; Jacobs, E.J.; Flanders, W.D.; Thun, M.J. Dietary intake of omega-6 and omega-3 fatty acids and risk of colorectal cancer in a prospective cohort of U.S. men and women. Cancer Epidemiol. Biomark. Prev. 2009, 18, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, F.; He, L.; Tan, B.; Deng, J.; Kong, X.; Li, Y.; Geng, M.; Yin, Y.; Wu, G. Dietary protein intake affects expression of genes for lipid metabolism in porcine skeletal muscle in a genotype-dependent manner. Br. J. Nutr. 2015, 113, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Koonen, D.P.Y.; Glatz, J.F.C.; Bonen, A.; Luikem, J.J.F.P. Long-chain fatty acid uptake and FAT/CD36 translocation in heart and skeletal muscle. BBA-Mol. Cell Biol. Lipids 2005, 1736, 163–180. [Google Scholar] [CrossRef]
- Schwenk, R.W.; Holloway, G.P.; Luiken, J.J.F.P.; Bonen, A.; Glatz, J.F.C. Fatty acid transport across the cell membrane: Regulation by fatty acid transporters. Prostag. Leuko. Ess. 2010, 82, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, R.; Strauss, J.G.; Haemmerle, G.; Schoiswohl, G.; Birner-Gruenberger, R.; Riderer, M.; Lass, A.; Neuberger, G.; Eisenhaber, F.; Hermetter, A.; et al. Fat mobilization in adipose tissue is promoted by adipose triglyceride lipase. Science 2004, 306, 1383–1386. [Google Scholar] [CrossRef] [PubMed]
- Farmer, S.R. Transcriptional control of adipocyte formation. Cell Metab. 2006, 4, 263–273. [Google Scholar] [CrossRef]

| Item | Dietary Levels of HMB, % | |||
|---|---|---|---|---|
| 0 | 0.13 | 0.64 | 1.28 | |
| Ingredients, % | ||||
| Corn | 61.00 | 62.00 | 61.21 | 60.45 |
| Soybean meal | 22.00 | 22.05 | 22.15 | 22.60 |
| Wheat bran | 14.00 | 12.82 | 13.00 | 12.72 |
| HMB-Ca | 0.00 | 0.13 | 0.64 | 1.28 |
| L-Lysine HCl | 0.10 | 0.10 | 0.10 | 0.10 |
| Dicalcium phosphate | 0.70 | 0.70 | 0.70 | 0.70 |
| Limestone | 0.90 | 0.90 | 0.90 | 0.85 |
| Salt | 0.30 | 0.30 | 0.30 | 0.30 |
| Premix b | 1.00 | 1.00 | 1.00 | 1.00 |
| Total | 100.00 | 100.00 | 100.00 | 100.00 |
| Chemical composition, % c | ||||
| Digestibe energy, MJ/kg | 13.60 | 13.63 | 13.55 | 13.49 |
| Metabolizable energy, MJ/kg | 12.15 | 12.18 | 12.10 | 12.03 |
| Dry matter | 87.22 | 87.30 | 87.35 | 87.40 |
| Crude protein | 15.98 | 15.94 | 15.95 | 16.03 |
| Crude fiber | 3.18 | 3.13 | 3.16 | 3.15 |
| Ether extract | 3.15 | 3.13 | 3.14 | 3.10 |
| Crude ash | 5.96 | 6.47 | 6.62 | 7.06 |
| Calcium | 0.61 | 0.62 | 0.69 | 0.76 |
| Total phosphorus | 0.54 | 0.54 | 0.54 | 0.54 |
| Amino Acid (AA, nmol/mL) 1 | Dietary Levels of HMB, % | p-Value | |||
|---|---|---|---|---|---|
| 0 | 0.13 | 0.64 | 1.28 | ||
| Nutritionally EAA | |||||
| L-leucine | 30.41 ± 2.38 | 33.33 ± 2.04 | 30.52 ± 1.79 | 28.05 ± 1.10 | 0.299 |
| L-isoleucine | 16.73 ± 1.52 | 20.73 ± 2.45 | 18.29 ± 2.06 | 16.03 ± 0.72 | 0.294 |
| L-valine | 41.98 ± 3.15 | 47.09 ± 3.28 | 43.37 ± 3.21 | 41.13 ± 2.23 | 0.525 |
| L-histidine | 9.60 ± 0.44 b | 11.65 ± 0.62 a,b | 12.37 ± 0.61 a | 11.60 ± 0.70 a,b | 0.023 |
| L-lysine | 19.29 ± 1.47 | 23.66 ± 2.17 | 19.51 ± 0.25 | 19.82 ± 1.53 | 0.170 |
| L-methionine | 2.16 ± 0.11 ab | 2.54 ± 0.16 a | 2.02 ± 0.06 b | 1.98 ± 0.11 b | 0.009 |
| L-phenylalanine | 18.07 ± 1.22 | 18.39 ± 0.69 | 18.99 ± 0.58 | 17.61 ± 0.94 | 0.740 |
| L-threonine | 12.52 ± 0.84 ab | 14.61 ± 1.22 a | 9.82 ± 0.70 b | 12.84 ± 1.85 a,b | 0.084 |
| Total EAA | 150.76 ± 9.06 | 171.99 ± 9.23 | 154.89 ± 8.05 | 149.07 ± 4.79 | 0.193 |
| Nutritionally NEAA | |||||
| L-alanine | 24.88 ± 0.72 b | 41.71 ± 3.30 a | 35.28 ± 4.21 a,b | 38.34 ± 4.04 a | 0.013 |
| L-arginine | 30.21 ± 1.20 b | 33.46 ± 0.31 a,b | 30.65 ± 1.31 a,b | 34.58 ± 0.87 a | 0.014 |
| L-aspartic acid | 1.75 ± 0.28 a | 0.97 ± 0.09 b | 1.43 ± 0.23 a,b | 1.80 ± 0.16 a | 0.034 |
| L-glutamic acid | 51.57 ± 3.56 | 59.49 ± 3.63 | 55.40 ± 2.15 | 57.34 ± 6.35 | 0.599 |
| L-glycine | 39.28 ± 3.42 | 38.86 ± 0.86 | 30.40 ± 2.94 | 39.37 ± 3.46 | 0.105 |
| L-serine | 10.68 ± 0.92 | 12.15 ± 0.57 | 10.63 ± 0.71 | 11.36 ± 0.22 | 0.348 |
| L-tyrosine | 10.10 ± 0.67 a,b | 11.52 ± 0.33 a | 8.63 ± 0.53 b | 9.78 ± 0.74 a,b | 0.020 |
| L-proline | 16.80 ± 0.53 | 17.63 ± 1.13 | 19.05 ± 1.97 | 17.46 ± 1.59 | 0.721 |
| Total NEAA | 185.27 ± 9.12 | 215.79 ± 6.59 | 191.47 ± 11.34 | 210.03 ± 9.04 | 0.087 |
| Other AA | |||||
| O-phosphoserine | 1.04 ± 0.10 b | 1.31 ± 0.06 a | 0.84 ± 0.04 b,c | 0.65 ± 0.04 c | <0.001 |
| Taurine | 30.43 ± 1.86 | 31.28 ± 2.09 | 33.55 ± 2.09 | 29.37 ± 2.77 | 0.602 |
| L-α-amino-adipic acid | 10.67 ± 1.44 | 11.55 ± 1.11 | 8.83 ± 0.47 | 8.68 ± 0.24 | 0.119 |
| L-citrulline | 4.82 ± 0.46 | 4.58 ± 0.20 | 4.22 ± 0.48 | 4.28 ± 0.51 | 0.742 |
| L-α-amino-n-butyric acid | 0.74 ± 0.12 | 0.90 ± 0.16 | 0.90 ± 0.14 | 1.25 ± 0.15 | 0.113 |
| L-cystathionine | 1.93 ± 0.27 | 1.40 ± 0.05 | 2.03 ± 0.24 | 2.18 ± 0.41 | 0.247 |
| β-alanine | 0.75 ± 0.01 a | 0.32 ± 0.07 b | 0.88 ± 0.12 a | 0.26 ± 0.04 b | <0.001 |
| L-ornithine | 6.25 ± 0.46 b | 8.74 ± 0.61 a | 6.17 ± 0.43 b | 7.70 ± 0.86 a,b | 0.022 |
| 3-methyl-l-histidine | 2.35 ± 0.21 | 2.32 ± 0.20 | 2.74 ± 0.36 | 2.72 ± 0.14 | 0.458 |
| Items 1 | Dietary Levels of HMB, % | p-Values | |||
|---|---|---|---|---|---|
| 0 | 0.13 | 0.64 | 1.28 | ||
| G6PD, ng/mL | 9.28 ± 0.22 a | 5.76 ± 0.30 b | 9.53 ± 0.18 a | 6.71 ± 0.33 b | <0.001 |
| ATGL, ng/mL | 388.12 ± 24.41 a,b | 428.76 ± 21.07 a | 251.42 ± 19.16 c | 315.70 ± 12.07 b,c | <0.001 |
| LPL, ng/mL | 5.21 ± 0.44 b | 2.68 ± 0.45 c | 7.13 ± 0.39 a | 3.52 ± 0.41 c | <0.001 |
| HMGR, ng/mL | 58.77 ± 2.98 b | 64.81 ± 4.57 b | 89.54 ± 6.06 a | 76.18 ± 7.39 a,b | 0.004 |
| Items | Dietary Levels of HMB, % | p-Value | |||
|---|---|---|---|---|---|
| 0 | 0.13 | 0.64 | 1.28 | ||
| C14:0 | 2.07 ± 0.12 a | 1.43 ± 0.05 b | 1.88 ± 0.15 a | 1.83 ± 0.13 a | 0.008 |
| C16:0 | 31.86 ± 0.44 a | 28.53 ± 0.37 b | 30.06 ± 0.53 a,b | 30.42 ± 0.79 a,b | 0.004 |
| C16:1 | 2.77 ± 0.14 | 2.05 ± 0.20 | 2.67 ± 0.22 | 2.64 ± 0.21 | 0.068 |
| C17:0 | 0.21 ± 0.01 | 0.20 ± 0.01 | 0.18 ± 0.01 | 0.18 ± 0.01 | 0.025 |
| C18:0 | 17.86 ± 0.53 | 18.50 ± 0.52 | 17.34 ± 0.27 | 17.83 ± 0.16 | 0.267 |
| C18:1 n9t | 0.19 ± 0.01 | 0.18 ± 0.03 | 0.14 ± 0.01 | 0.17 ± 0.01 | 0.255 |
| C18:1 n9c | 37.63 ± 0.42 | 32.89 ± 2.41 | 35.44 ± 0.60 | 35.21 ± 1.13 | 0.150 |
| C18:2 n6c | 4.22 ± 0.51 b | 10.14 ± 1.19 a | 7.50 ± 0.57 a,b | 7.18 ± 1.08 a,b | 0.002 |
| C20:0 | 0.25 ± 0.01 | 0.25 ± 0.02 | 0.28 ± 0.01 | 0.23 ± 0.01 | 0.176 |
| C20:1 | 1.53 ± 0.11 | 1.19 ± 0.09 | 1.32 ± 0.08 | 1.21 ± 0.05 | 0.044 |
| C18:3 n3 | 0.20 ± 0.02 b | 0.26 ± 0.01 a | 0.25 ± 0.01 a,b | 0.23 ± 0.02 a,b | 0.033 |
| C20:2 | 0.19 ± 0.03 b | 0.37 ± 0.05 a | 0.32 ± 0.01 a,b | 0.29 ± 0.04 a,b | 0.010 |
| C20:3 n6 | 0.14 ± 0.01 b | 0.40 ± 0.06 a | 0.25 ± 0.03 a,b | 0.24 ± 0.05 a,b | 0.005 |
| C20:4 n6 | 0.66 ± 0.09 b | 3.26 ± 0.74 a | 1.97 ± 0.27 a,b | 1.80 ± 0.46 a,b | 0.007 |
| SFA 1 | 52.25 ± 0.57 a | 48.92 ± 0.80 b | 49.73 ± 0.60 a,b | 50.48 ± 0.80 a,b | 0.020 |
| PUFA 2 | 5.41 ± 0.64 b | 14.43 ± 1.86 a | 10.29 ± 0.85 a,b | 9.73 ± 1.64 a,b | 0.002 |
| ∑PUFA:SFA | 0.10 ± 0.01 b | 0.29 ± 0.03 a | 0.21 ± 0.02 a,b | 0.19 ± 0.03 a,b | 0.001 |
| ∑n3 PUFA 3 | 0.20 ± 0.02 b | 0.26 ± 0.01 a | 0.25 ± 0.01 a,b | 0.23 ± 0.02 a,b | 0.033 |
| ∑n6 PUFA 4 | 5.02 ± 0.60 b | 13.80 ± 1.83 a | 9.73 ± 0.85 a,b | 9.22 ± 1.58 a,b | 0.002 |
| ∑n6:n3 PUFA | 24.88 ± 2.10 b | 52.83 ± 6.95 a | 39.68 ± 3.49 a,b | 38.82 ± 4.21 a,b | 0.004 |
| h/H 5 | 1.27 ± 0.03 b | 1.58 ± 0.05 a | 1.43 ± 0.05 a,b | 1.40 ± 0.07 a,b | 0.005 |
| AI 6 | 0.84 ± 0.02 a | 0.67 ± 0.02 b | 0.75 ± 0.03 a,b | 0.77 ± 0.04 a,b | 0.004 |
| TI 7 | 2.13 ± 0.05 a | 1.87 ± 0.06 b | 1.93 ± 0.05 a,b | 1.99 ± 0.07 a,b | 0.019 |
| Items | Dietary Levels of HMB, % | p-Value | |||
|---|---|---|---|---|---|
| 0 | 0.13 | 0.64 | 1.28 | ||
| C14:0 | 1.35 ± 0.12 | 1.28 ± 0.15 | 1.48 ± 0.12 | 1.37 ± 0.09 | 0.711 |
| C16:0 | 28.16 ± 0.56 | 27.92 ± 0.62 | 29.07 ± 0.49 | 29.32 ± 0.38 | 0.197 |
| C16:1 | 1.48 ± 0.21 | 1.98 ± 0.29 | 1.80 ± 0.20 | 1.74 ± 0.12 | 0.418 |
| C17:0 | 0.27 ± 0.03 | 0.24 ± 0.02 | 0.21 ± 0.01 | 0.20 ± 0.01 | 0.095 |
| C18:0 | 20.08 ± 0.88 | 17.93 ± 0.56 | 19.73 ± 0.62 | 20.28 ± 0.39 | 0.062 |
| C18:1 n9t | 0.28 ± 0.05 | 0.19 ± 0.02 | 0.20 ± 0.02 | 0.21 ± 0.02 | 0.224 |
| C18:1 n9c | 25.09 ± 2.08 | 31.66 ± 3.45 | 29.77 ± 2.07 | 28.81 ± 1.90 | 0.313 |
| C18:2 n6c | 15.21 ± 1.51 a | 11.04 ± 1.05 b | 11.28 ± 1.30 b | 11.45 ± 0.83 b | 0.072 |
| C20:0 | 0.24 ± 0.02 a,b | 0.22 ± 0.01 b | 0.27 ± 0.01 a | 0.24 ± 0.01 a,b | 0.058 |
| C20:1 | 0.94 ± 0.12 | 0.86 ± 0.07 | 1.08 ± 0.12 | 0.94 ± 0.06 | 0.450 |
| C18:3 n3 | 0.27 ± 0.00 a,b | 0.29 ± 0.01 a | 0.26 ± 0.00 a,b | 0.25 ± 0.01 b | 0.014 |
| C20:2 | 0.40 ± 0.02 | 0.34 ± 0.02 | 0.37 ± 0.02 | 0.37 ± 0.02 | 0.243 |
| C20:3 n6 | 0.54 ± 0.06 | 0.41 ± 0.10 | 0.42 ± 0.07 | 0.43 ± 0.06 | 0.571 |
| C20:4 n6 | 5.11 ± 0.50 | 3.62 ± 0.95 | 3.60 ± 0.55 | 3.90 ± 0.66 | 0.380 |
| SFA 1 | 50.09 ± 0.66 a | 47.58 ± 0.70 b | 50.75 ± 0.56 a | 51.41 ± 0.65 a | 0.003 |
| PUFA 2 | 21.53 ± 2.06 | 17.37 ± 3.46 | 15.92 ± 1.92 | 16.40 ± 1.48 | 0.343 |
| ∑PUFA:SFA | 0.43 ± 0.04 | 0.36 ± 0.07 | 0.31 ± 0.04 | 0.32 ± 0.03 | 0.292 |
| ∑n3 PUFA 3 | 0.27 ± 0.00 a,b | 0.29 ± 0.01 a | 0.26 ± 0.00 ab | 0.25 ± 0.01 b | 0.014 |
| ∑n6 PUFA 4 | 20.86 ± 2.04 | 15.07 ± 2.03 | 15.30 ± 1.92 | 15.78 ± 1.48 | 0.014 |
| ∑n6:n3 PUFA | 78.09 ± 7.10 a | 52.33 ± 6.54 b | 59.26 ± 7.41 a,b | 63.81 ± 7.06 a,b | 0.099 |
| h/H 5 | 1.58 ± 0.05 | 1.62 ± 0.07 | 1.49 ± 0.04 | 1.47 ± 0.04 | 0.173 |
| AI 6 | 0.67 ± 0.02 | 0.63 ± 0.03 | 0.71 ± 0.02 | 0.72 ± 0.02 | 0.064 |
| TI 7 | 1.95 ± 0.05 a | 1.76 ± 0.05 b | 2.01 ± 0.04 a | 2.06 ± 0.05 a | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, C.; Song, B.; Guo, Q.; Zheng, J.; Li, F.; Duan, Y.; Peng, C. Alterations of the Muscular Fatty Acid Composition and Serum Metabolome in Bama Xiang Mini-Pigs Exposed to Dietary Beta-Hydroxy Beta-Methyl Butyrate. Animals 2021, 11, 1190. https://doi.org/10.3390/ani11051190
Zheng C, Song B, Guo Q, Zheng J, Li F, Duan Y, Peng C. Alterations of the Muscular Fatty Acid Composition and Serum Metabolome in Bama Xiang Mini-Pigs Exposed to Dietary Beta-Hydroxy Beta-Methyl Butyrate. Animals. 2021; 11(5):1190. https://doi.org/10.3390/ani11051190
Chicago/Turabian StyleZheng, Changbing, Bo Song, Qiuping Guo, Jie Zheng, Fengna Li, Yehui Duan, and Can Peng. 2021. "Alterations of the Muscular Fatty Acid Composition and Serum Metabolome in Bama Xiang Mini-Pigs Exposed to Dietary Beta-Hydroxy Beta-Methyl Butyrate" Animals 11, no. 5: 1190. https://doi.org/10.3390/ani11051190
APA StyleZheng, C., Song, B., Guo, Q., Zheng, J., Li, F., Duan, Y., & Peng, C. (2021). Alterations of the Muscular Fatty Acid Composition and Serum Metabolome in Bama Xiang Mini-Pigs Exposed to Dietary Beta-Hydroxy Beta-Methyl Butyrate. Animals, 11(5), 1190. https://doi.org/10.3390/ani11051190







