Roles of Nitrocompounds in Inhibition of Foodborne Bacteria, Parasites, and Methane Production in Economic Animals
Simple Summary
Abstract
1. Introduction
2. Inhibition of Pathogenic Bacteria, Ammonia and Methane Production
3. Inhibition of Eimeria spp.
4. Toxicity of Nitrocompounds
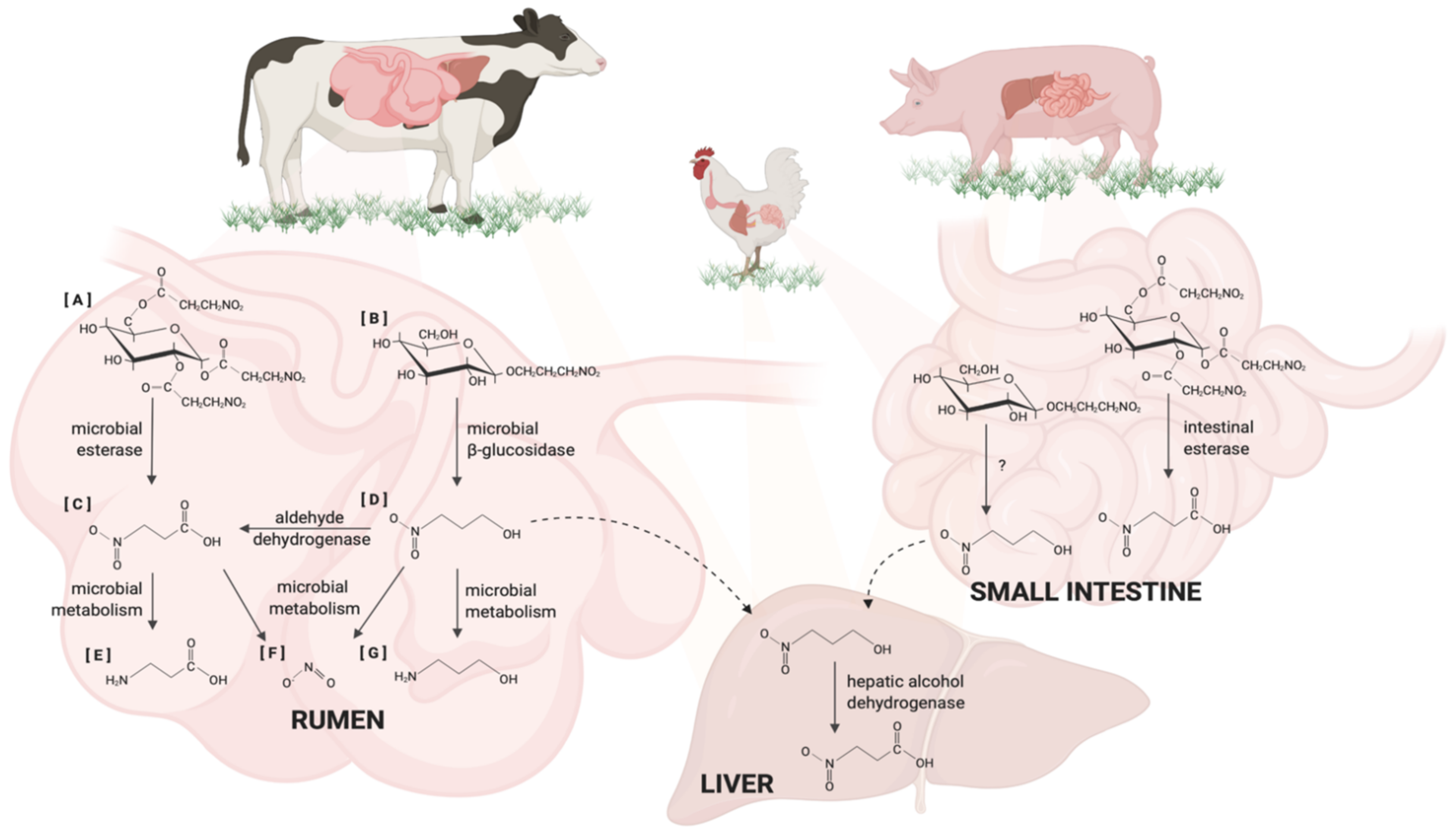
5. Metabolism of Nitrocompounds
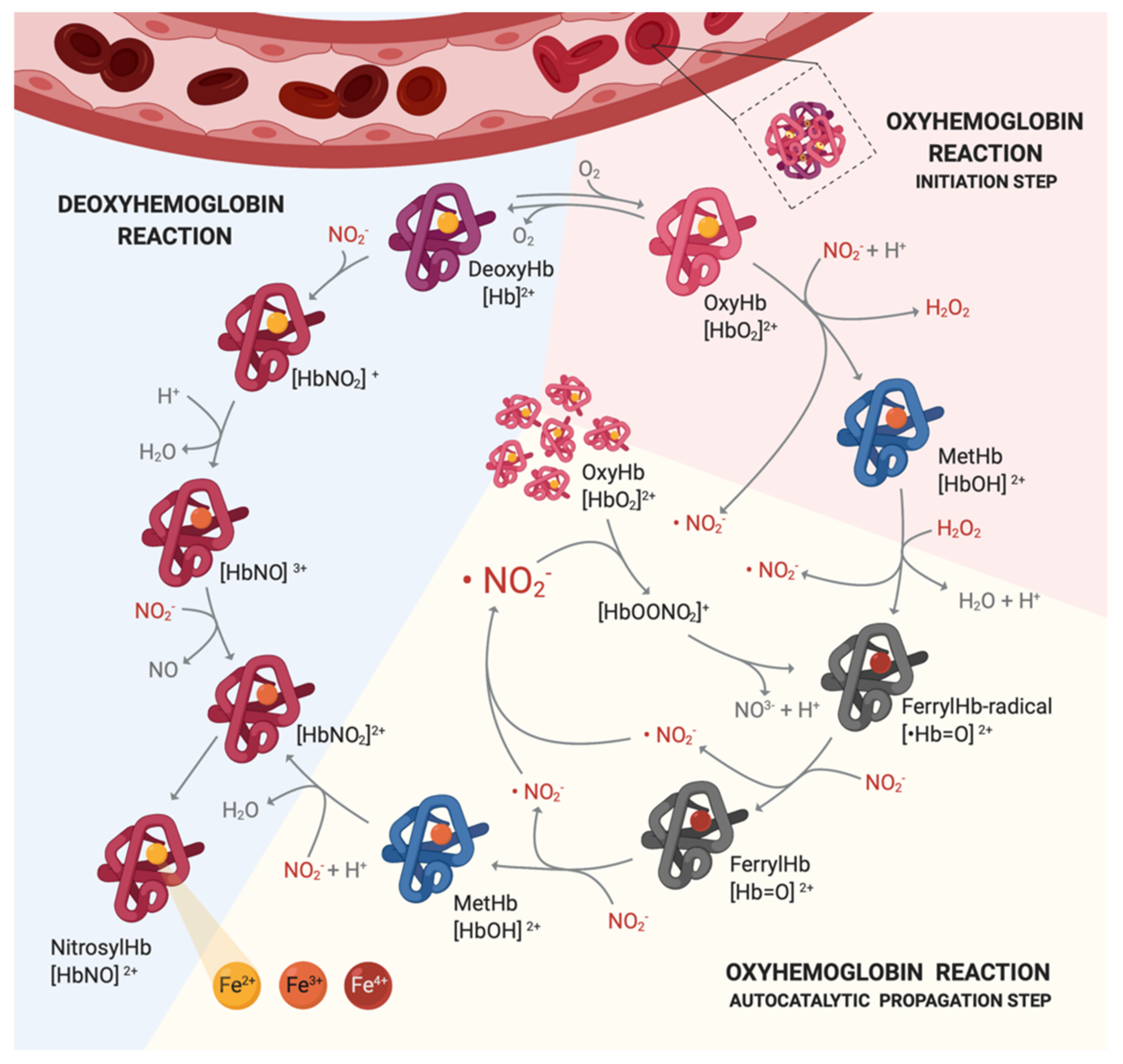
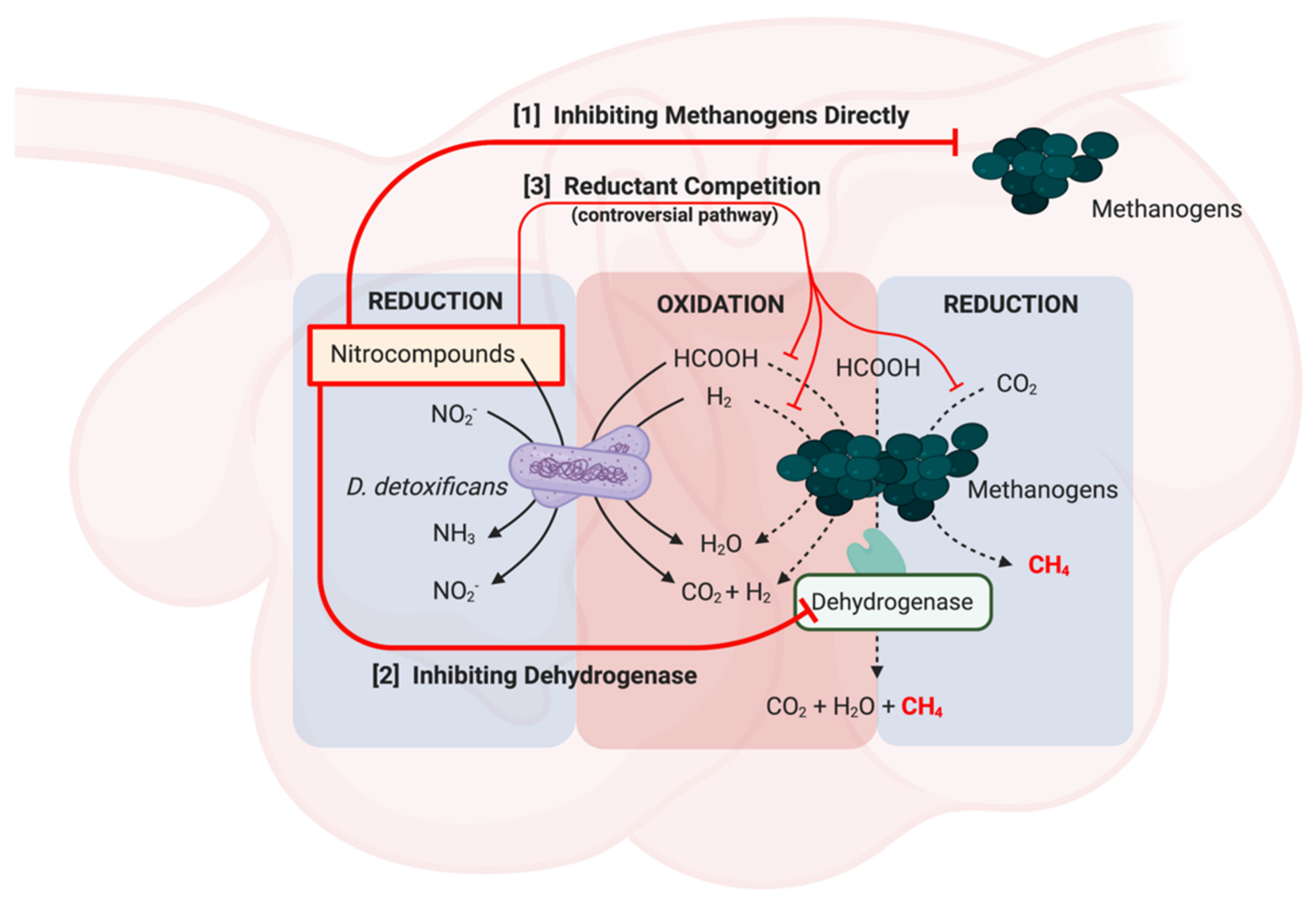
6. Mechanisms of Actions of Nitrocompounds
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Z.-W.; Cao, Z.-J.; Wang, Y.-L.; Wang, Y.-J.; Yang, H.-J.; Li, S.-L. Nitrocompounds as potential methanogenic inhibitors in ruminant animals: A review. Anim. Feed Sci. Tech. 2018, 236, 107–114. [Google Scholar] [CrossRef]
- Shvekhgeimer, M.-G.A. Aliphatic nitro alcohols. Synthesis, chemical transformations and applications. Russ. Chem. Rev. 1998, 67, 35–68. [Google Scholar] [CrossRef]
- Gustine, D.L. Aliphatic Nitro Compounds in Crownvetch: A Review. Crop. Sci. 1979, 19, 197–203. [Google Scholar] [CrossRef]
- Anderson, R.C.; Majak, W.; Rassmussen, M.A.; Callaway, T.R.; Beier, R.C.; Nisbet, D.J.; Allison, M.J. Toxicity and metabolism of the conjugates of 3-nitropropanol and 3-nitropropionic acid in forages poisonous to livestock. J. Agric. Food Chem. 2005, 53, 2344–2350. [Google Scholar] [CrossRef]
- Stermitz, F.R.; Norris, F.A.; Williams, M.C. Miserotoxin, new naturally occurring nitro compound. J. Am. Chem. Soc. 1969, 91, 4599–4600. [Google Scholar] [CrossRef]
- Burdock, G.A.; Carabin, I.G.; Soni, M.G. Safety assessment of β-nitropropionic acid: A monograph in support of an acceptable daily intake in humans. Food Chem. 2001, 75, 1–27. [Google Scholar] [CrossRef]
- Adhikari, P.; Cosby, D.E.; Cox, N.A.; Kim, W.K. Effect of dietary supplementation of nitrocompounds on Salmonella colonization and ileal immune gene expression in laying hens challenged with Salmonella enteritidis. Poult. Sci. 2017, 96, 4280–4286. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Anderson, R.C.; Edrington, T.S.; Genovese, K.J.; Byrd, J.A.; Callaway, T.R.; Nisbet, D.J. Experimental use of 2-nitropropanol for reduction of Salmonella typhimurium in the Ceca of broiler chicks. J. Food Prot. 2004, 67, 1945–1947. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Carstens, G.E.; Miller, R.K.; Callaway, T.R.; Schultz, C.L.; Edrington, T.S.; Harvey, R.B.; Nisbet, D.J. Effect of oral nitroethane and 2-nitropropanol administration on methane-producing activity and volatile fatty acid production in the ovine rumen. Bioresour. Technol. 2006, 97, 2421–2426. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.K.; Weeks, L.J.; Anderson, R.C.; Nisbet, D.J.; Dunkley, K.; Ricke, S.C. Effects of nitrocompounds on uric acid-utilizing microorganisms, nitrogen retention, and microbial community in laying hen manure. J. Environ. Sci. Health B 2009, 44, 403–406. [Google Scholar] [CrossRef]
- Morente, E.O.; Abriouel, H.; Lopez, R.L.; Ben Omar, N.; Galvez, A. Antibacterial activity of carvacrol and 2-nitro-1-propanol against single and mixed populations of foodborne pathogenic bacteria in corn flour dough. Food Microbiol. 2010, 27, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Anderson, R.C.; Callaway, T.R.; Edrington, T.S.; Genovese, K.J.; Harvey, R.B.; Poole, T.L.; Nisbet, D.J. Inhibitory activity of 2-nitropropanol against select food-borne pathogens in vitro. Lett. Appl. Microbiol. 2004, 39, 471–476. [Google Scholar] [CrossRef]
- Dimitrijevic, M.; Anderson, R.C.; Callaway, T.R.; Jung, Y.S.; Harvey, R.B.; Ricke, S.C.; Nisbet, D.J. Inhibitory Effect of Select Nitrocompounds on growth and survivability of Listeria monocytogenes in vitro. J. Food Prot. 2006, 69, 1061–1065. [Google Scholar] [CrossRef] [PubMed]
- Horrocks, S.M.; Jung, Y.S.; Huwe, J.K.; Harvey, R.B.; Ricke, S.C.; Carstens, G.E.; Callaway, T.R.; Anderson, R.C.; Ramlachan, N.; Nisbet, D.J. Effects of short-chain nitrocompounds against Campylobacter jejuni and Campylobacter coli in vitro. J. Food Sci. 2007, 72, M50–M55. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.K.; Anderson, R.C.; Ratliff, A.L.; Nisbet, D.J.; Ricke, S.C. Growth inhibition by nitrocompounds of selected uric acid-utilizing microorganisms isolated from poultry manure. J. Environ. Sci. Health B 2006, 41, 97–107. [Google Scholar] [CrossRef]
- Antonio, C.M.; Abriouel, H.; Lopez, R.L.; Omar, N.B.; Valdivia, E.; Galvez, A. Enhanced bactericidal activity of enterocin AS-48 in combination with essential oils, natural bioactive compounds and chemical preservatives against Listeria monocytogenes in ready-to-eat salad. Food Chem. Toxicol. 2009, 47, 2216–2223. [Google Scholar]
- Cobo Molinos, A.; Lucas Lopez, R.; Abriouel, H.; Ben Omar, N.; Valdivia, E.; Galvez, A. Inhibition of Salmonella enterica Cells in deli-type salad by enterocin AS-48 in combination with other antimicrobials. Probiotics Antimicrob. Proteins 2009, 1, 85–90. [Google Scholar] [CrossRef]
- Anderson, R.C.; Jung, Y.S.; Oliver, C.E.; Horrocks, S.M.; Genovese, K.J.; Harvey, R.B.; Callaway, T.R.; Edrington, T.S.; Nisbet, D.J. Effects of nitrate or nitro supplementation, with or without added chlorate, on Salmonella enterica serovar Typhimurium and Escherichia coli in swine feces. J. Food Prot. 2007, 70, 308–315. [Google Scholar] [CrossRef]
- Ruiz-Barrera, O.; Anderson, R.C.; Hume, M.E.; Corrales-Millan, J.; Castillo-Castillo, Y.; Corral-Luna, A.; Guevara-Valdez, J.L.; Salinas-Chavira, J.; Rodriguez-Muela, C.; Arzola-Alvarez, C. Short chain nitrocompounds as a treatment of layer hen manure and litter; effects on in vitro survivability of Salmonella, generic E. coli and nitrogen metabolism. J. Environ. Sci. Health B 2017, 52, 23–29. [Google Scholar] [CrossRef]
- Mowrer, J.E.; Sedlacek, P.; Kim, J.; Ritz, C.; Kim, W.K. Supplementation of nitrocompounds in broiler diets: Effects on bird performance, ammonia volatilization and nitrogen retention in broiler manure. J. Environ. Sci. Health B 2016, 51, 126–131. [Google Scholar] [CrossRef]
- Beier, R.C.; Anderson, R.C.; Krueger, N.A.; Edrington, T.S.; Callaway, T.R.; Nisbet, D.J. Effect of nitroethane and nitroethanol on the production of indole and 3-methylindole (skatole) from bacteria in swine feces by gas chromatography. J. Environ. Sci. Health B 2009, 44, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Haisan, J.; Sun, Y.; Guan, L.L.; Beauchemin, K.A.; Iwaasa, A.; Duval, S.; Barreda, D.R.; Oba, M. The effects of feeding 3-nitrooxypropanol on methane emissions and productivity of Holstein cows in mid lactation. J. Dairy Sci. 2014, 97, 3110–3119. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Callaway, T.R.; Van Kessel, J.A.; Jung, Y.S.; Edrington, T.S.; Nisbet, D.J. Effect of select nitrocompounds on ruminal fermentation; an initial look at their potential to reduce economic and environmental costs associated with ruminal methanogenesis. Bioresour. Technol. 2003, 90, 59–63. [Google Scholar] [CrossRef]
- Saengkerdsub, S.; Kim, W.K.; Anderson, R.C.; Nisbet, D.J.; Ricke, S.C. Effects of nitro compounds and feedstuffs on in vitro methane production in chicken cecal contents and rumen fluid. Anaerobe 2006, 12, 85–92. [Google Scholar] [CrossRef]
- Gutierrez-Banuelos, H.; Anderson, R.C.; Carstens, G.E.; Slay, L.J.; Ramlachan, N.; Horrocks, S.M.; Callaway, T.R.; Edrington, T.S.; Nisbet, D.J. Zoonotic bacterial populations, gut fermentation characteristics and methane production in feedlot steers during oral nitroethane treatment and after the feeding of an experimental chlorate product. Anaerobe 2007, 13, 21–31. [Google Scholar] [CrossRef]
- Gutierrez-Banuelos, H.; Anderson, R.C.; Carstens, G.E.; Tedeschi, L.O.; Pinchak, W.E.; Cabrera-Diaz, E.; Krueger, N.A.; Callaway, T.R.; Nisbet, D.J. Effects of nitroethane and monensin on ruminal fluid fermentation characteristics and nitrocompound-metabolizing bacterial populations. J. Agric. Food Chem. 2008, 56, 4650–4658. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fernandez, G.; Abecia, L.; Arco, A.; Cantalapiedra-Hijar, G.; Martin-Garcia, A.I.; Molina-Alcaide, E.; Kindermann, M.; Duval, S.; Yanez-Ruiz, D.R. Effects of ethyl-3-nitrooxy propionate and 3-nitrooxypropanol on ruminal fermentation, microbial abundance, and methane emissions in sheep. J. Dairy Sci. 2014, 97, 3790–3799. [Google Scholar] [CrossRef]
- Reynolds, C.K.; Humphries, D.J.; Kirton, P.; Kindermann, M.; Duval, S.; Steinberg, W. Effects of 3-nitrooxypropanol on methane emission, digestion, and energy and nitrogen balance of lactating dairy cows. J. Dairy Sci. 2014, 97, 3777–3789. [Google Scholar] [CrossRef] [PubMed]
- Hristova, A.N.; Oha, J.; Giallongoa, F.; Fredericka, T.W.; Harpera, M.T.; Weeksa, H.L.; Brancob, A.F.; Moatec, P.J.; Deightonc, M.H.; Williamsc, S.R.O.; et al. An inhibitor persistently decreased enteric methane emission from dairy cows with no negative effect on milk production. Proc. Natl. Acad. Sci. USA 2015, 112, 10663–10668. [Google Scholar] [CrossRef]
- Chapman, H.D. Milestones in avian coccidiosis research: A review. Poult. Sci. 2014, 93, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Chapman, H.D.; Jeffers, T.K.; Williams, R.B. Forty years of monensin for the control of coccidiosis in poultry. Poult. Sci. 2010, 89, 1788–18001. [Google Scholar] [CrossRef] [PubMed]
- Teng, P.Y.; Fuller, A.L.; Kim, W.K. Evaluation of nitro compounds as feed additives in diets of Eimeria-challenged broilers in vitro and in vivo. Poult. Sci. 2020, 99, 1320–1325. [Google Scholar] [CrossRef]
- Zhang, Z.W.; Wang, Y.L.; Chen, Y.Y.; Wang, W.K.; Zhang, L.T.; Luo, H.L.; Yang, H.J. Nitroethanol in comparison with monensin exhibits greater feed efficiency through inhibiting rumen methanogenesis more efficiently and persistently in feedlotting lambs. Animals 2019, 9, 784. [Google Scholar] [CrossRef] [PubMed]
- Bangoura, B.; Bardsley, K.D. Ruminant Coccidiosis. Vet. Clin. North. Am. Food Anim. Pract. 2020, 36, 187–203. [Google Scholar] [CrossRef]
- Teng, P.Y.; Yadav, S.; Dos Santos, T.S.; Fuller, A.L.; Kim, W.K. 2-Nitro-1-propanol improved nutrient digestibility and oocyst shedding but not growth performance of Eimeria-challenged broilers. Poult. Sci. 2020, 99, 4314–4322. [Google Scholar] [CrossRef]
- Paik, D.C.; Wen, Q.; Braunstein, R.E.; Trokel, S.L. Short chain aliphatic beta-nitro alcohols for corneoscleral cross-linking: Corneal endothelial toxicity studies. J. Refract. Surg. 2008, 24, S741–S747. [Google Scholar] [CrossRef]
- Kim, M.; Takaoka, A.; Hoang, Q.V.; Trokel, S.L.; Paik, D.C. Pharmacologic alternatives to riboflavin photochemical corneal cross-linking: A comparison study of cell toxicity thresholds. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3247–3257. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.C.; James, L.F. Toxicity of nitro-containing Astragalus to sheep and chicks. J. Range Manag. 1975, 28, 260–263. [Google Scholar] [CrossRef]
- Smith, D.J.; Anderson, R.C. Toxicity and metabolism of nitroalkanes and substituted nitroalkanes. J. Agric. Food Chem. 2013, 61, 763–779. [Google Scholar] [CrossRef]
- Majak, W.; Wikeem, B.M. Miserotoxin levels in fertilized Astragalus miser var. serotinus. J. Range Manag. 1986, 39, 130–131. [Google Scholar] [CrossRef][Green Version]
- Majak, W.; Pass, M.A.; Madryga, F.J. Toxicity of miserotoxin and its aglycone. Toxicol. Lett. 1983, 19, 171–178. [Google Scholar] [CrossRef]
- Griffin, T.B.; Stein, A.A.; Coulston, F. Chronic inhalation exposure of rats to vapors of nitroethane. Ecotoxicol. Environ. Saf. 1988, 16, 11–24. [Google Scholar] [CrossRef]
- Majak, W. Further enhancement of 3-nitropropanol detoxification by ruminal bactdria in cattle. Can. J. Anim. Sci. 1992, 72, 863–870. [Google Scholar] [CrossRef]
- Majak, W.; Clark, L.J. Metabolism of aliphatic nitro compounds in bovine rumen fluid. Can. J. Anim. Sci. 1980, 60, 319–325. [Google Scholar] [CrossRef]
- Muir, A.D.; Majak, W.; Pass, M.A.; Yost, G.S. Conversion of 3-nitropropanol (miserotoxin aglycone) to 3-nitropropionic acid in cattle and sheep. Toxicol. Lett. 1984, 20, 137–141. [Google Scholar] [CrossRef]
- Anderson, R.C.; Rasmussen, M.A.; Allison, M.J. Metabolism of the plant toxins nitropropionic acid and nitropropanol by ruminal microorganisms. Appl. Environ. Microbiol. 1993, 59, 3056–3061. [Google Scholar] [CrossRef] [PubMed]
- Majak, W.; Cheng, K.J.; Hall, J.W. Enhanced degradation of 3-nitropropanol by ruminal microorganisms. J. Anim. Sci. 1986, 62, 1072–1080. [Google Scholar] [CrossRef][Green Version]
- Zhang, Z.W.; Wang, Y.L.; Wang, W.K.; Chen, Y.Y.; Si, X.M.; Wang, Y.J.; Wang, W.; Cao, Z.J.; Li, S.L.; Yang, H.J. The Antimethanogenic nitrocompounds can be cleaved into nitrite by rumen microorganisms: A comparison of Nitroethane, 2-Nitroethanol, and 2-Nitro-1-propanol. Metabolites 2020, 10, 15. [Google Scholar] [CrossRef]
- Majak, W.; Cheng, K.J. Identification of rumen bacteria that anaerobically degrade aliphatic nitrotoxins. Can. J. Microbiol. 1981, 27, 646–650. [Google Scholar] [CrossRef]
- Majak, W.; Pass, M.A.; Muir, A.D.; Rode, L.M. Absorption of 3-nitropropanol (miserotoxin aglycone) from the compound stomach of cattle. Toxicol. Lett. 1984, 23, 9–15. [Google Scholar] [CrossRef]
- Pass, M.A.; Majak, W.; Muir, A.D.; Yost, G.S. Absorption of 3-nitropropanol and 3-nitropropionic acid from the digestive system of sheep. Toxicol. Lett. 1984, 23, 1–7. [Google Scholar] [CrossRef]
- Benn, M.; McDiarmid, R.; Majak, W. In-vitro biotransformation of 3-nitropropanol (miserotoxin aglycone) by horse liver alcohol dehydrogenase. Toxicol. Lett. 1989, 47, 165–172. [Google Scholar] [CrossRef]
- Pass, M.A.; Muir, A.D.; Majak, W.; Yost, G.S. Effect of alcohol and aldehyde dehydrogenase inhibitors on the toxicity of 3-nitropropanol in rats. Toxicol. Appl. Pharmacol. 1985, 78, 310–315. [Google Scholar] [CrossRef]
- Little, H.N. Oxidation of nitroethane by extracts from Neurospora. J. Biol. Chem. 1951, 193, 347–358. [Google Scholar] [CrossRef]
- Anderson, R.C.; Rasmussen, M.A.; Jensen, N.S.; Allison, M.J. Denitrobacterium detoxificans gen. nov., sp. nov., a ruminal bacterium that respires on nitrocompounds. Int. J. Syst. Evol. Microbiol. 2000, 50(Pt. 2), 633–638. [Google Scholar] [CrossRef] [PubMed]
- Angermaier, L.; Simon, H. On the reduction of aliphatic and aromatic nitro compounds by Clostridia, the role of ferredoxin and its stabilization. Hoppe Seylers Z Physiol. Chem. 1983, 364, 961–975. [Google Scholar] [CrossRef] [PubMed]
- Shiono, H.; Yagi, Y.; Thongnoon, P.; Kurabayashi, N.; Chikayama, Y.; Miyazaki, S.; Nakamura, I. Acquired methemoglobinemia in anemic cattle infected with Theileria sergenti. Vet. Parasitol. 2001, 102, 45–51. [Google Scholar] [CrossRef]
- Alston, T.A.; Mela, L.; Bright, H.J. 3-Nitropropionate, the toxic substance of Indigofera, is a suicide inactivator of succinate dehydrogenase. Proc. Natl. Acad. Sci. USA 1977, 74, 3767–3771. [Google Scholar] [CrossRef]
- Coles, C.J.; Edmondson, D.E.; Singer, T.P. Inactivation of succinate dehydrogenase by 3-nitropropionate. J. Biol. Chem. 1979, 254, 5161–5167. [Google Scholar] [CrossRef]
- Anderson, R.C.; Krueger, N.A.; Stanton, T.B.; Callaway, T.R.; Edrington, T.S.; Harvey, R.B.; Jung, Y.S.; Nisbet, D.J. Effects of select nitrocompounds on in vitro ruminal fermentation during conditions of limiting or excess added reductant. Bioresour. Technol. 2008, 99, 8655–8661. [Google Scholar] [CrossRef]
- Keszler, A.; Piknova, B.; Schechter, A.N.; Hogg, N. The reaction between nitrite and oxyhemoglobin: A mechanistic study. J. Biol. Chem. 2008, 283, 9615–9622. [Google Scholar] [CrossRef]
- Hathazi, D.; Mahut, S.D.; Scurtu, F.V.; Bischin, C.; Stanciu, C.; Attia, A.A.; Damian, G.; Silaghi-Dumitrescu, R. Involvement of ferryl in the reaction between nitrite and the oxy forms of globins. J. Biol. Inorg. Chem. 2014, 19, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, M.; Tarjan, G. Succinate Dehydrogenase Complex: An Updated Review. Arch. Pathol. Lab. Med. 2018, 142, 1564–1570. [Google Scholar] [CrossRef]
- Hylin, J.W.; Matsumoto, H. Inhibition of succinic dehydrogenase by 3-nitropropanoate. Toxicol. Appl. Pharmacol. 1964, 6, 168–171. [Google Scholar] [CrossRef]
- Harada, S.; Inaoka, D.K.; Ohmori, J.; Kita, K. Diversity of parasite complex II. Biochim. Biophys. Acta 2013, 1827, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Herrera, J.; Garcia, L.G. Regulation of succinate dehydrogenase in Escherichia coli. J. Gen. Microbiol. 1972, 72, 29–35. [Google Scholar] [CrossRef][Green Version]
- Pecsi, I.; Hards, K.; Ekanayaka, N.; Berney, M.; Hartman, T.; Jacobs, W.R., Jr.; Cook, G.M. Essentiality of succinate dehydrogenase in Mycobacterium smegmatis and its role in the generation of the membrane potential under hypoxia. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Huwe, J.K.; Smith, D.J.; Stanton, T.B.; Krueger, N.A.; Callaway, T.R.; Edrington, T.S.; Harvey, R.B.; Nisbet, D.J. Effect of nitroethane, dimethyl-2-nitroglutarate and 2-nitro-methyl-propionate on ruminal methane production and hydrogen balance in vitro. Bioresour. Technol. 2010, 101, 5345–5349. [Google Scholar] [CrossRef]
- Anderson, R.C.; Rasmussen, M.A. Use of a novel nitrotoxin-metabolizing bacterium to reduce ruminal methane production. Bioresour. Technol. 1998, 64, 89–95. [Google Scholar] [CrossRef]
- Tapio, I.; Snelling, T.J.; Strozzi, F.; Wallace, R.J. The ruminal microbiome associated with methane emissions from ruminant livestock. J. Anim. Sci. Biotechnol. 2017, 8, 7. [Google Scholar] [CrossRef]
- Asanuma, N.; Iwamoto, M.; Hino, T. Formate metabolism by ruminal microorganisms in relation to methanogenesis. Anim. Sci. Technol. 1998, 69, 576–584. [Google Scholar]
- Hungate, R.E.; Smith, W.; Bauchop, T.; Yu, I.; Rabinowitz, J.C. Formate as an intermediate in the bovine rumen fermentation. J. Bacteriol. 1970, 102, 389–397. [Google Scholar] [CrossRef] [PubMed]

| Nitrocompound | Dosage | Unit | Pathogens Inhibition | Reference |
|---|---|---|---|---|
| In vitro | ||||
| 2NEOH | 10, 20 | mM | Campylobacter coli | [14] |
| 10, 20 | mM | Campylobacter jejuni | [14] | |
| 8 | mM | Clostridium perfringens | Unpublished data | |
| 15 | mM | Listeria monocytogenes strain 18 | [13] | |
| 50 | mM | uric acid-utilizing microorganisms | [15] | |
| 2NMP | 10, 20 | mM | Campylobacter jejuni | [14] |
| 2NPOH | 5 | % | Bacillus cereus1 | [11] |
| 10, 20 | mM | Campylobacter coli | [14] | |
| 10, 20 | mM | Campylobacter jejuni | [14] | |
| 4, 8 | mM | Clostridium perfringens | Unpublished data | |
| 10 | mM | Enterococcus faecalis | [12] | |
| 2.5, 5, 10 | mM | Escherichia coli | [12] | |
| 0.5, 2, 5 | % | Escherichia coli1 | [11] | |
| 10, 15 | mM | Listeria monocytogenes strain 18 | [13] | |
| 50 | mM | Listeria monocytogenes2 | [17] | |
| 0.5, 2, 5 | % | Salmonella enterica serovar Enteritidis 1 | [11] | |
| 2.5, 5, 10 | mM | Salmonella Typhimurium | [12] | |
| 0.5, 2, 5 | % | Staphylococcus aureus1 | [11] | |
| 50 | mM | uric acid-utilizing microorganisms | [15] | |
| 3NPA | 50 | mM | uric acid-utilizing microorganisms | [15] |
| NE | 10, 20 | mM | Campylobacter coli | [14] |
| 10, 20 | mM | Campylobacter jejuni | [14] | |
| 15 | mM | Listeria monocytogenes strain 18 | [13] | |
| In vivo/feces incubation | ||||
| 2NEOH | 20 | mM | Escherichia coli | [18] |
| 20 | mM | Escherichia coli | [18] | |
| 44 | mM | Salmonella Typhimurium | [19] | |
| 13, 65, 130 | mg/bird | Salmonella Typhimurium2 | [8] | |
| 44 | mM | Salmonella Typhimurium | [19] | |
| 20 | mM | Salmonella Typhimurium | [18] | |
| 100 | mM | uric acid-utilizing microorganisms | [10] | |
| 3NPA | 100 | mM | uric acid-utilizing microorganisms | [10] |
| Ethyl-nitroacetate | 44 | mM | Salmonella Typhimurium | [19] |
| NE | 12 | mM | Escherichia coli | [19] |
| 44 | mM | Salmonella Typhimurium | [19] | |
| 20 | mM | Salmonella Typhimurium | [18] | |
| 12 | mM | Total Coliforms | [19] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, P.-Y.; Kim, W.K. Roles of Nitrocompounds in Inhibition of Foodborne Bacteria, Parasites, and Methane Production in Economic Animals. Animals 2021, 11, 923. https://doi.org/10.3390/ani11040923
Teng P-Y, Kim WK. Roles of Nitrocompounds in Inhibition of Foodborne Bacteria, Parasites, and Methane Production in Economic Animals. Animals. 2021; 11(4):923. https://doi.org/10.3390/ani11040923
Chicago/Turabian StyleTeng, Po-Yun, and Woo Kyun Kim. 2021. "Roles of Nitrocompounds in Inhibition of Foodborne Bacteria, Parasites, and Methane Production in Economic Animals" Animals 11, no. 4: 923. https://doi.org/10.3390/ani11040923
APA StyleTeng, P.-Y., & Kim, W. K. (2021). Roles of Nitrocompounds in Inhibition of Foodborne Bacteria, Parasites, and Methane Production in Economic Animals. Animals, 11(4), 923. https://doi.org/10.3390/ani11040923






