A Systematic Approach to Comparing Thermal Activity of the Thoracic Region and Saddle Pressure Distribution beneath the Saddle in a Group of Non-Lame Sports Horses
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Horses
2.2. Rider
2.3. Saddles
2.4. Measuring Systems
2.4.1. Thermal Imaging
2.4.2. Saddle Kinetics
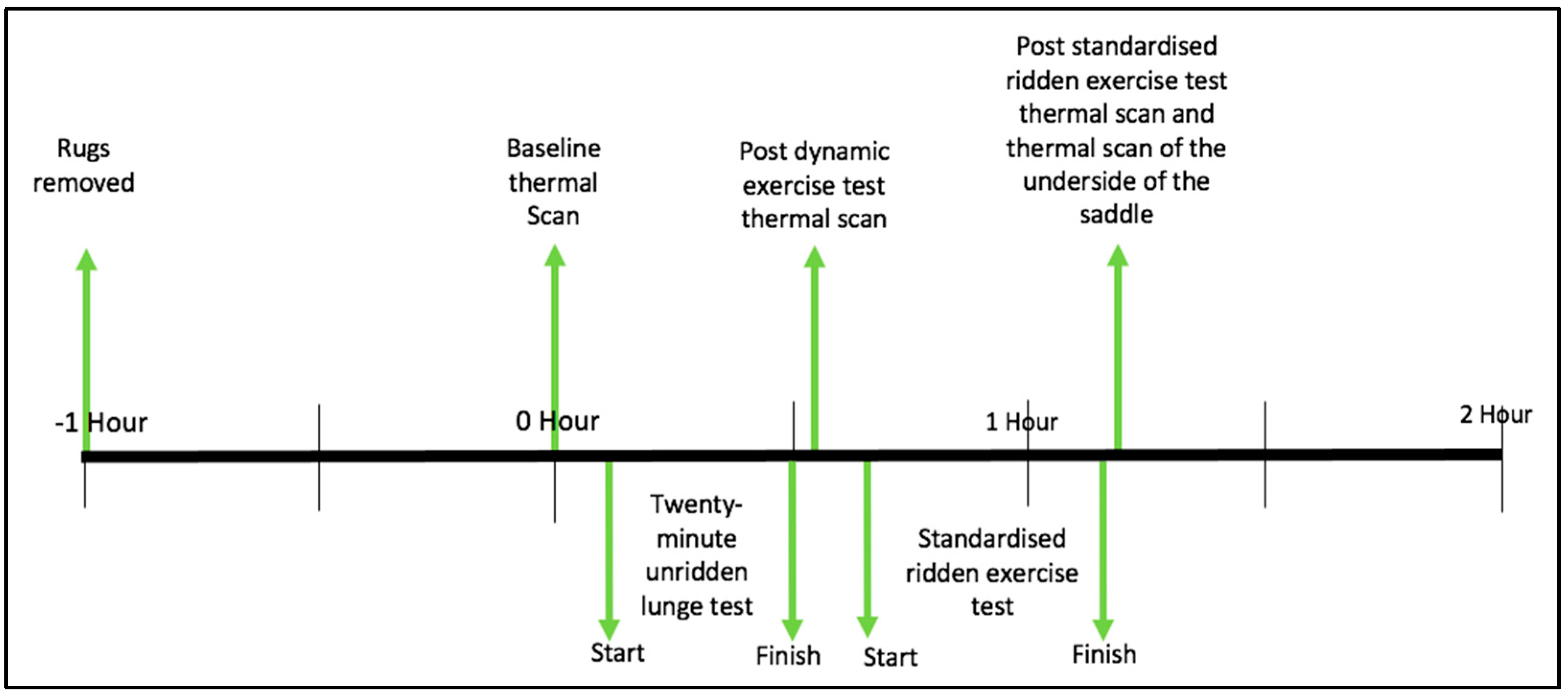
2.5. Study Protocol
2.5.1. Baseline Thermographic Scan
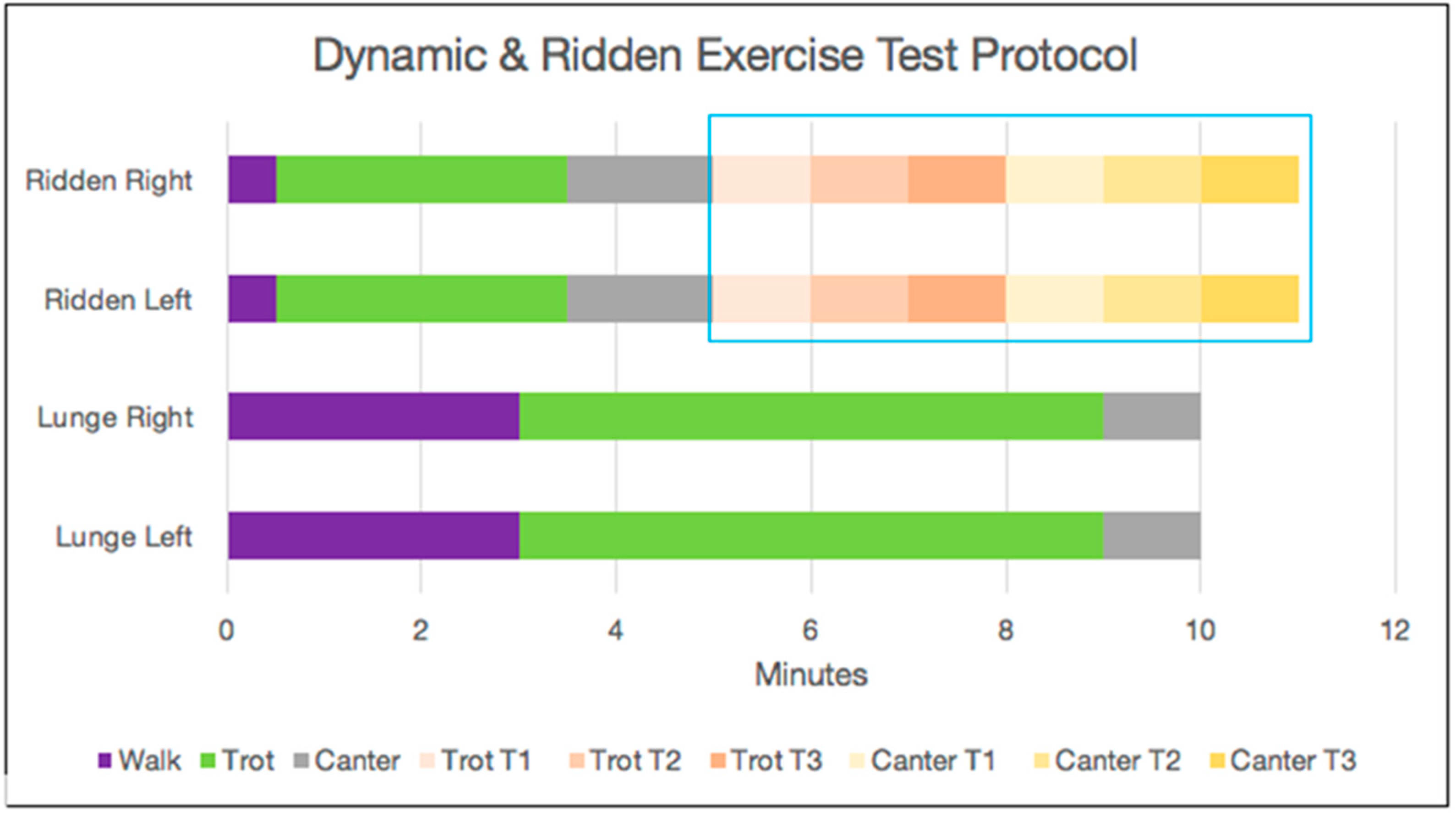
2.5.2. Standardised Unridden Dynamic Exercise (Lunge Test)
2.5.3. Standardised Ridden Exercise Test
2.5.4. Thermal Imaging of the Underside of the Saddle
2.6. Data Collection and Processing
- minimum difference head (HDMin) and pelvis (PDMin): difference between the two minima in vertical (z) displacement observed during the two diagonal stance phases in trot;
- maximum difference head (HDMax) and pelvis (PDMax): difference between the two maxima in vertical (z) displacement observed after the two diagonal stance phases in trot;
- hip hike difference (HHD): difference between vertical upward movement amplitude of left and right tuber coxae during contra-lateral stance.
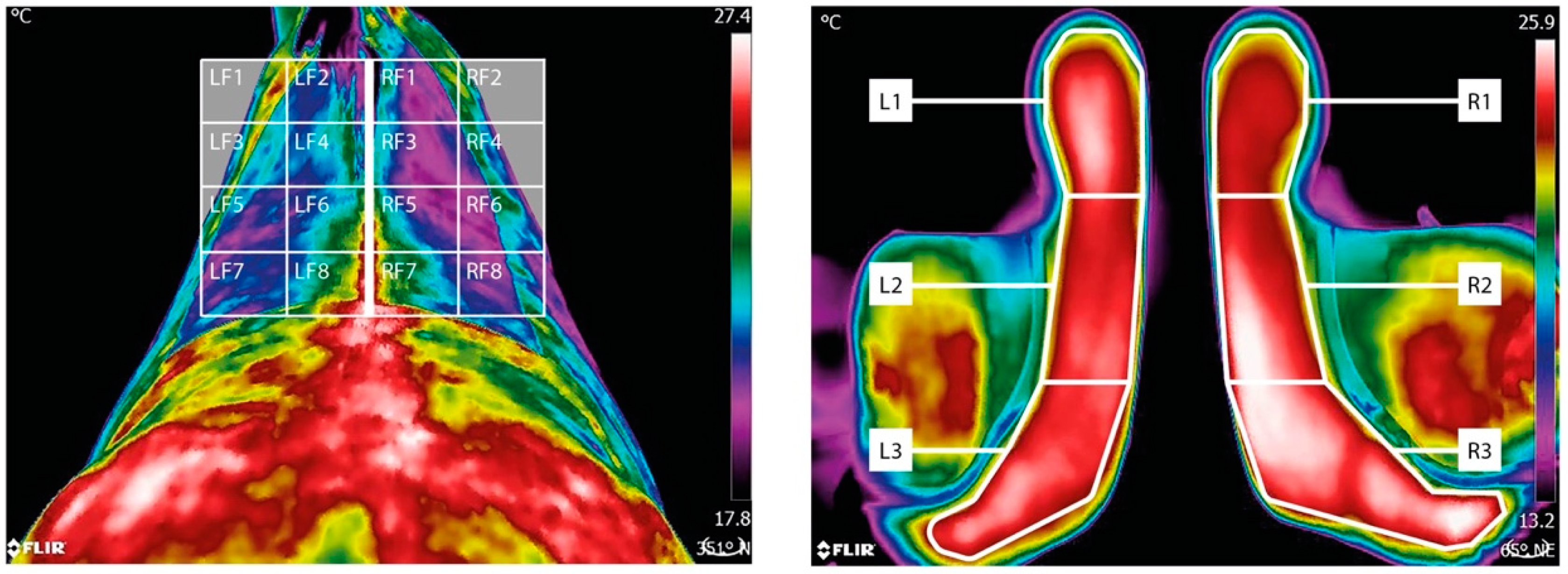
- for the left cranial region defined as LF1, LF2, LF3 and LF4;
- for the right cranial region defined as RF1, RF2, RF3 and RF4;
- for the left caudal region defined as LF5, LF6, LF7 and LF8;
- for the right caudal region defined as RF5, RF6, RF7 and RF8.
- Symmetry values:
- left–right symmetry for minimum, maximum and mean thermal values of the cranial region;
- left–right symmetry for minimum, maximum and mean thermal values of the caudal region;
- front–back symmetry for minimum, maximum and mean thermal values.
- for the cranial region defined as L3 and R3;
- for the mid region defined as L2 and R2;
- for the caudal region defined as L1 and R1.
- Symmetry values:
- left—right symmetry for minimum, maximum and mean thermal values of the cranial region;
- left—right symmetry for minimum, maximum and mean thermal values of the mid region;
- left—right symmetry for minimum, maximum and mean thermal values of the caudal region;
- front—back symmetry for minimum, maximum and mean thermal values.
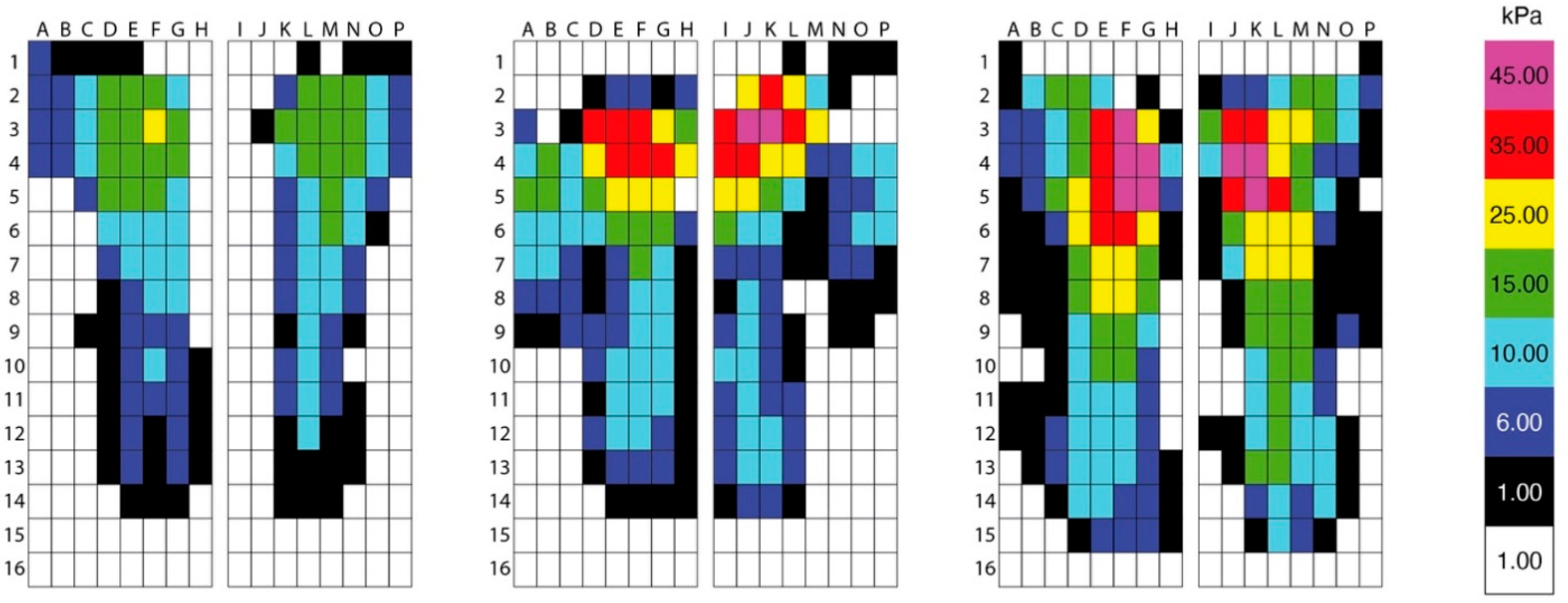
2.7. Saddle Pressure Data
- pressures beneath the cranial aspect of the saddle defined as rows 1–8 and columns A–H (left side) and I–J (right side).
- pressures beneath the caudal aspect of the saddle defined as rows 9–16 and columns A–H (left side) and I–J (right side).
- left-to-right saddle pressure symmetry for the cranial region defined as rows 1–8 and columns A–H (left side) and I–J (right side).
- left-to-right saddle pressure symmetry for the caudal region (left—right) defined as rows 9–16 and columns A–H (left side) and I–J (right side).
- front-to-back pressure differences between cranial and caudal regions (front-back) defined as cranial (rows 1–8, columns A–J)– caudal (rows 9–16, columns A–J).
2.8. Statistical Analysis
3. Results
3.1. Horse Inclusion
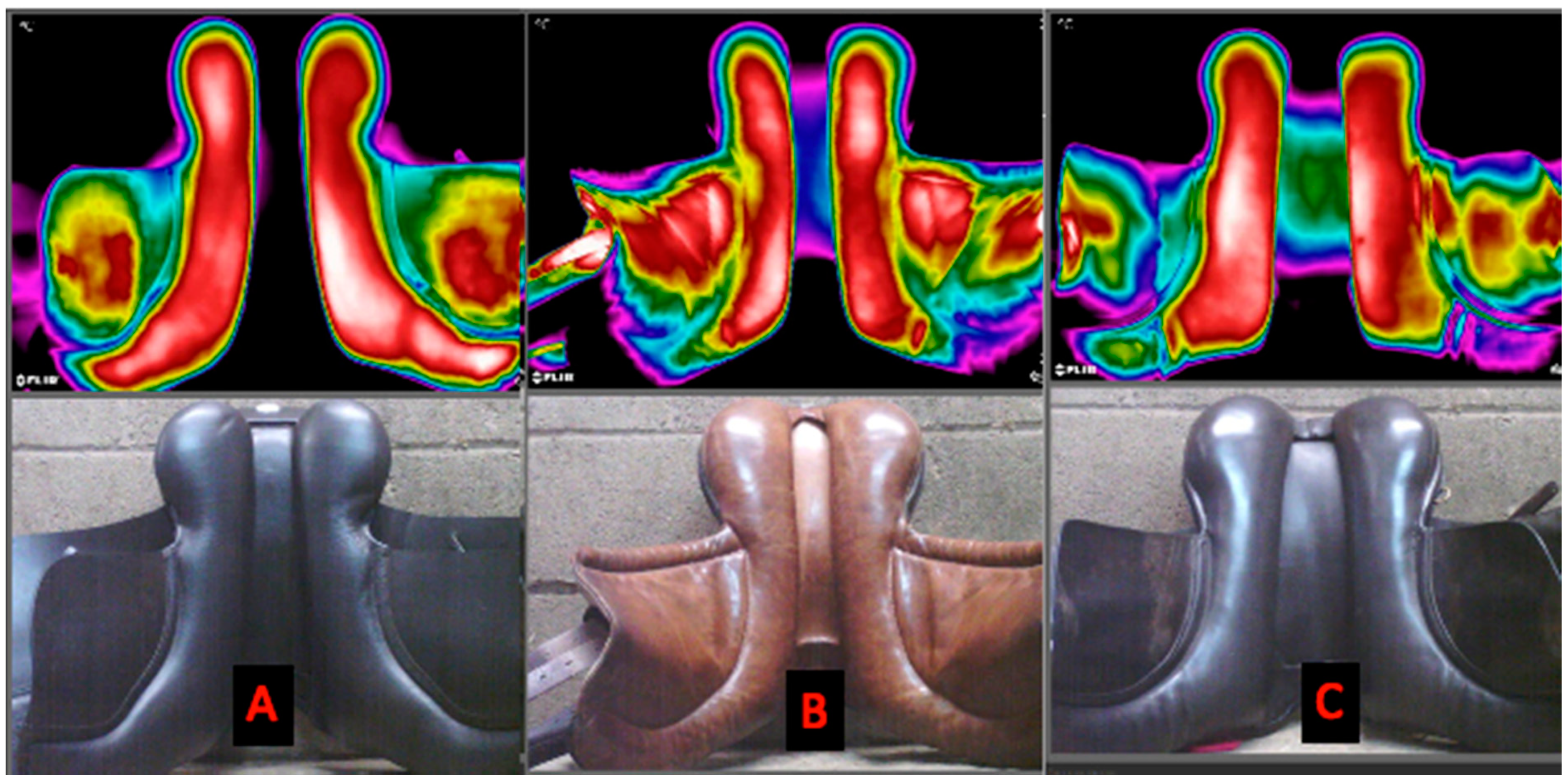
3.2. Saddle Fit
3.3. Thermographic Data of the Thoracic Region
3.3.1. Minimum Temperatures (°C)
3.3.2. Maximum Temperatures (°C)
3.3.3. Mean Temperatures (°C)
3.4. Thermographic Data of the Underside of the Saddle
3.5. Saddle Pressure Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mackechnie-Guire, R.; Mackechnie-Guire, E.; Fisher, M.; Mathie, H.; Bush, R.; Pfau, T.; Weller, R. Relationship Between Saddle and Rider Kinematics, Horse Locomotion, and Thoracolumbar Pressures in Sound Horses. J. Equine Vet. Sci. 2018, 69, 43–52. [Google Scholar] [CrossRef]
- Mackechnie-Guire, R.; Mackechnie-Guire, E.; Fairfax, V.; Fisher, D.; Fisher, M.; Pfau, T. The Effect of Tree Width on Thoracolumbar and Limb Kinematics, Saddle Pressure Distribution, and Thoracolumbar Dimensions in Sports Horses in Trot and Canter. Animals 2019, 9, 842. [Google Scholar] [CrossRef] [PubMed]
- Meschan, E.M.; Peham, C.; Schobesberger, H.; Licka, T.F. The influence of the width of the saddle tree on the forces and the pres-sure distribution under the saddle. Vet. J. 2007, 173, 578–584. [Google Scholar] [CrossRef]
- Clayton, H.M.; Hampson, A.; Fraser, P.; White, A.; Egenvall, A. Comparison of rider stability in a flapless saddle versus a conven-tional saddle. PLoS ONE 2018, 13, e0196960. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Cheze, L.; Pourcelot, P.; Desquilbet, L.; Duray, L.; Chateau, H. Effects of Large Saddle Panels on the Biomechanics of the Equine Back During Rising Trot: Preliminary Results. J. Equine Vet. Sci. 2017, 48, 15–22. [Google Scholar] [CrossRef]
- Murray, R.; Guire, R.; Fisher, M.; Fairfax, V. Reducing Peak Pressures Under the Saddle Panel at the Level of the 10th to 13th Thoracic Vertebrae May Be Associated With Improved Gait Features, Even When Saddles Are Fitted to Published Guidelines. J. Equine Vet. Sci. 2017, 54, 60–69. [Google Scholar] [CrossRef]
- Murray, R.; Mackechnie-Guire, R.; Fisher, M.; Fairfax, V. Reducing peak pressures under the saddle at thoracic vertebrae 10–13 is associated with alteration in jump kinematics. Comp. Exerc. Physiol. 2018, 14, 239–247. [Google Scholar] [CrossRef]
- Murray, R.; Mackechnie-Guire, R.; Fisher, M.; Fairfax, V. Could Pressure Distribution Under Race-Exercise Saddles Affect Limb Kinematics and Lumbosacral Flexion in the Galloping Racehorse? J. Equine Vet. Sci. 2019, 81, 102795. [Google Scholar] [CrossRef]
- Belock, B.; Kaiser, L.J.; Lavagnino, M.; Clayton, H.M. Comparison of pressure distribution under a conventional saddle and a tree-less saddle at sitting trot. Vet. J. 2012, 193, 87–91. [Google Scholar] [CrossRef]
- Clayton, H.M.; O’Connor, K.A.; Kaiser, L.J. Force and pressure distribution beneath a conventional dressage saddle and a treeless dressage saddle with panels. Vet. J. 2014, 199, 44–48. [Google Scholar] [CrossRef]
- Latif, S.N.; Von Peinen, K.; Wiestner, T.; Bitschnau, C.; Renk, B.; Weishaupt, M.A. Saddle pressure patterns of three different training saddles (normal tree, flexible tree, treeless) in Thoroughbred racehorses at trot and gallop. Equine Vet. J. 2010, 42, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Harman, J. Tack and Saddle Fit. Vet. Clin. Am Equine Pract. 1999, 15, 247–261. [Google Scholar] [CrossRef]
- Greve, L.; Dyson, S.; Pfau, T. Alterations in thoracolumbosacral movement when pain causing lameness has been improved by diagnostic analgesia. Vet. J. 2017, 224, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Greve, L.; Dyson, S.J. An investigation of the relationship between hindlimb lameness and saddle slip. Equine Vet. J. 2013, 45, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Byström, A.; Stalfelt, A.; Egenvall, A.; Von Peinen, K.; Morgan, K.; Roepstorff, L. Influence of girth strap placement and panel flocking material on the saddle pressure pattern during riding of horses. Equine Vet. J. 2010, 42, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Peham, C.; Licka, T.; Schobesberger, H.; Meschan, E. Influence of the rider on the variability of the equine gait. Hum. Mov. Sci. 2004, 23, 663–671. [Google Scholar] [CrossRef]
- Dyson, S.; Carson, S.; Fisher, M. Saddle fitting, recognising an ill-fitting saddle and the consequences of an ill-fitting saddle to horse and rider. Equine Vet. Educ. 2015, 27, 533–543. [Google Scholar] [CrossRef]
- de Cocq, P.; Clayton, H.M.; Terada, K.; Muller, M.; van Leeuwen, J.L. Usability of normal force distribution measurements to evalu-ate asymmetrical lo.ading of the back of the horse and different rider positions on a standing horse. Vet. J. 2009, 181, 266–273. [Google Scholar] [CrossRef] [PubMed]
- de Cocq, P.; van Weeren, P.R.; Back, W. Saddle pressure measuring: Validity, reliability and power to discriminate between dif-ferent saddle-fits. Vet. J. 2006, 172, 265–273. [Google Scholar] [CrossRef]
- Fruehwirth, B.; Peham, C.; Scheidl, M.; Schobesberger, H. Evaluation of pressure distribution under an English saddle at walk, trot and canter. Equine Vet. J. 2010, 36, 754–757. [Google Scholar] [CrossRef]
- Kotschwar, A.; Baltacis, A.; Peham, C. The influence of different saddle pads on force and pressure changes beneath saddles with excessively wide trees. Vet. J. 2010, 184, 322–325. [Google Scholar] [CrossRef]
- Kotschwar, A.B.; Baltacis, A.; Peham, C. The effects of different saddle pads on forces and pressure distribution beneath a fitting saddle. Equine Vet. J. 2010, 42, 114–118. [Google Scholar] [CrossRef]
- Alvarez, C.B.G.; Wennerstrand, J.; Bobbert, M.F.; Lamers, L.; Johnston, C.; Back, W.; Van Weeren, P.R. The effect of induced forelimb lameness on thoracolumbar kinematics during treadmill locomotion. Equine Vet. J. 2007, 39, 197–201. [Google Scholar] [CrossRef]
- Buchner, H.H.F.; Schamhardt, H.; Barneveld, A. Head and trunk movement adaptations in horses with experimentally induced fore- or hindlimb lameness. Equine Vet. J. 1996, 28, 71–76. [Google Scholar] [CrossRef]
- Gomez Alvarez, C.B.; Bobbert, M.F.; Lamers, L.; Johnston, C.; Back, W.; van Weeren, P.R. The effect of induced hindlimb lameness on thoracolumbar kinematics during treadmill locomotion. Equine Vet. J. 2008, 40, 147–152. [Google Scholar] [CrossRef]
- Greve, L.; Dyson, S.J. The interrelationship of lameness, saddle slip and back shape in the general sports horse population. Equine Vet. J. 2014, 46, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Landman, M.A.A.M.; de Blaauw, J.A.; Hofland, L.J.; van Weeren, P.R. Field study of the prevalence of lameness in horses with back problems. Vet. Rec. 2004, 155, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Keegan, K.G.; Dent, E.V.; Wilson, D.A.; Janicek, J.; Kramer, J.; Lacarrubba, A.; Walsh, D.M.; Cassells, M.W.; Esther, T.M.; Schiltz, P.; et al. Repeatability of subjective evaluation of lameness in horses. Equine Vet. J. 2010, 42, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Keegan, K.G.; Wilson, D.A.; Wilson, D.J.; Smith, B.; Gaughan, E.M.; Pleasant, R.S.; Lillich, J.D.; Kramer, J.; Howard, R.D.; Bacon-Miller, C.; et al. Evaluation of mild lameness in horses trotting on a treadmill by clinicians and interns or residents and correlation of their assessments with kinematic gait analysis. Am. J. Vet. Res. 1998, 59, 1370–1377. [Google Scholar]
- Hewetson, M.; Christley, R.M.; Hunt, I.D.; Voute, L.C. Investigations of the reliability of observational gait analysis for the assessment of lameness in horses. Vet. Rec. 2006, 158, 852–858. [Google Scholar] [CrossRef]
- Parkes, R.S.V.; Weller, R.; Groth, A.M.; May, S.; Pfau, T. Evidence of the development of ‘domain-restricted’ expertise in the recogni-tion of asymmetric motion characteristics of hindlimb lameness in the horse. Equine Vet. J. 2009, 41, 112–117. [Google Scholar] [CrossRef] [PubMed]
- McCracken, M.J.; Kramer, J.; Keegan, K.G.; Lopes, M.; Wilson, D.A.; Reed, S.K.; Lacarrubba, A.; Rasch, M. Comparison of an inertial sensor system of lameness quantification with subjective lameness evaluation. Equine Vet. J. 2012, 44, 652–656. [Google Scholar] [CrossRef]
- Soroko, M.; Howell, K. Infrared Thermography: Current Applications in Equine Medicine. J. Equine Vet. Sci. 2018, 60, 90–96.e2. [Google Scholar] [CrossRef]
- Schweinitz, D.V. Thermographic diagnosis in equine back pain. Vet. North Clin. N. Am. Equine Pract. 1999, 15, 161–177. [Google Scholar] [CrossRef]
- Arruda, T.Z.; Brass, K.E.; De La Corte, F.D. Thermographic Assessment of Saddles Used on Jumping Horses. J. Equine Vet. Sci. 2011, 31, 625–629. [Google Scholar] [CrossRef]
- Soroko, M.; Cwynar, P.; Howell, K.; Yarnell, K.; Dudek, K.; Zaborski, D. Assessment of Saddle Fit in Racehorses Using Infrared Thermography. J. Equine Vet. Sci. 2018, 63, 30–34. [Google Scholar] [CrossRef]
- Soroko, M.; Zaborski, D.; Dudek, K.; Yarnell, K.; Górniak, W.; Vardasca, R. Evaluation of thermal pattern distributions in racehorse saddles using infrared thermography. PLoS ONE 2019, 14, e0221622. [Google Scholar] [CrossRef]
- Turner, T.A.; Waldsmith, J.K.; Wilson, J.H. How to assess saddle fit in horses. Proc. Am. Assoc. Equine Pract. 2004, 50, 196–201. [Google Scholar]
- Masko, M.; Krajewska, A.; Zdrojkowski, L.; Domino, M.; Gajewski, Z. An application of temperature mapping of horse’s back for leisure horse-rider-matching. Anim. Sci. J. 2019, 90, 1396–1406. [Google Scholar] [CrossRef] [PubMed]
- Soroko, M.; Jodkowska, E.; Zabłocka, M. The Use of Thermography to Evaluate Back Musculoskeletal Responses of Young Racehorses to Training. Thermol. Intern. 2012, 22, 114. [Google Scholar]
- Witkowska-Pilaszewicz, O.; Masko, M.; Domino, M.; Winnicka, A. Infrared Thermography Correlates with Lactate Concentra-tion in Blood during Race Training in Horses. Animals 2020, 10, 2072. [Google Scholar] [CrossRef]
- Pfau, T.; Witte, T.H.; Wilson, A.M. A method for deriving displacement data during cyclical movement using an inertial sensor. J. Exp. Biol. 2005, 208, 2503–2514. [Google Scholar] [CrossRef] [PubMed]
- Guire, R.; Weller, R.; Fisher, M.; Beavis, J. Investigation Looking at the Repeatability of 20 Society of Master Saddlers Qualified Saddle Fitters’ Observations During Static Saddle Fit. J. Equine Vet. Sci. 2017, 56, 1–5. [Google Scholar] [CrossRef]
- Guilds and City. Certificate in Saddle Fitting, in Association with the Society of Master Saddlers; City and Guilds, NPTC: London, UK. Available online: http://tinyurlcom/y82f9at22007:4750-80 (accessed on 30 September 2020).
- Westermann, S.; Buchner, H.; Schramel, P.; Tichy, T.; Stanek, C. Effects of infrared camera angle and distance on meas-urement and reproducibilityof thermographically determined temperatures of the distolateral aspects of the forelimbs in horses. J. Am. Vet. Med. Assoc. 2013, 242, 388–395. [Google Scholar] [CrossRef] [PubMed]
- von Peinen, K.; Wiestner, T.; von Rechenberg, B.; Weishaupt, M.A. Relationship between saddle pressure measurements and clin-ical signs of saddle soreness at the withers. Equine Vet. J Suppl. 2010, 38, 650–653. [Google Scholar] [CrossRef]
- Dantas, F.; Duarte, M.; Marins, J.; Fonseca, B. Thermographic assessment of saddles used in Mangalarga Marchador horses. Arq. Bras. de Med. Vet. e Zootec. 2019, 71, 1165–1170. [Google Scholar] [CrossRef]
- Mara, C.A.; Cribbie, R.A. Paired-Samples Tests of Equivalence. Commun. Stat.-Simul. Comput. 2012, 41, 1928–1943. [Google Scholar] [CrossRef]
- Nyikos, S.; Von Rechenberg, B.; Werner, D.; Müller, J.A.; Buess, C.; Keel, R.; Kalpen, A.; Vontobel, H.-D.; Von Plocki, K.A.; Auer, J.A. Measurements of saddle pressure in conjunction with back problems in horses. Pferdeheilkunde Equine Med. 2005, 21, 187–198. [Google Scholar] [CrossRef]
- Le, K.M.; Madsen, B.L.; Barth, P.W.; Ksander, G.A.; Angell, J.B.; Vistnes, L.M. An in-depth look at pressure sores using monolithic silicon pressure sensors. Plast. Reconstr. Surg. 1984, 74, 745–754. [Google Scholar] [CrossRef]
- Chang, W.L.; Seigreg, A.A. Prediction of ulcer formation on the skin. Med. Hypotheses 1999, 53, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Mackechnie-Guire, R.; Fisher, M.; Pfau, T. Effect of a Half Pad on Pressure Distribution in Sitting Trot and Canter beneath a Saddle Fitted to Industry Guidelines. J. Equine Vet. Sci. 2021, 96, 103307. [Google Scholar] [CrossRef] [PubMed]
- Mackechnie-Guire, R.; Mackechnie-Guire, E.; Bush, R.; Fisher, D.; Fisher, M.; Weller, R. Local Back Pressure Caused by a Training Roller During Lunging With and Without a Pessoa Training Aid. J. Equine Vet. Sci. 2018, 67, 112–117. [Google Scholar] [CrossRef]
- Murray, R.; Guire, R.; Fisher, M.; Fairfax, V. A Bridle Designed to Avoid Peak Pressure Locations under the Headpiece and Noseband Is Associated with More Uniform Pressure and Increased Carpal and Tarsal Flexion, Compared with the Horse’s Usual Bridle. J. Equine Vet. Sci. 2015, 35, 947–955. [Google Scholar] [CrossRef]
| Baseline (BL) (°C) EMM (SE) | Post Lunge (PL) (°C) EMM (SE) | Post Ridden (PR) (°C) EMM (SE) | Exercise Main Effects p-Value | Pairwise Bonferroni Post Hoc p ≤ 0.05 | Correct Saddle Width (°C) EMM (SE) | Narrow Saddle Width (°C) EMM (SE) | Wide Saddle Width (°C) EMM (SE) | Saddle Width Main Effects p Value | Pairwise Bonferroni Post Hoc p ≤ 0.05 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Minimum Temperature (°C) | ||||||||||
| Left Cranial Region | 19.9 (0.7) | 22.3 (0.7) | 20.8 (0.7) | 0.12 | - | 20.3 (0.9) | 20.6 (0.7) | 22.2 (0.7) | 0.28 | - |
| Right Cranial Region | 18.7 (0.8) | 21.7 (0.8) | 20.3 (0.8) | 0.05 | BL < PL, p = 0.05 | 20.4 (1.1) | 19.1 (0.8) | 21.2 (0.8) | 0.29 | - |
| Left Caudal region | 20.6 (0.9) | 23.4 (0.9) | 22.4 (0.9) | 0.04 | BL<PL, p = 0.04 | 21.1 (1.3) | 22.5 (1.1)) | 22.9 (1.1) | 0.59 | - |
| Right Caudal Region | 20.4 (1.0) | 22.9 (1.0) | 21.9 (1.0) | 0.05 | - | 21.1 (1.7) | 21.7 (1.4) | 22.5 (1.4) | 0.81 | - |
| Cranial Region Symmetry (left–right) | 1.2 (0.5) | 0.5 (0.5) | 0.5 (0.5) | 0.58 | - | −0.1 (0.6) | 1.5 (0.5) | 0.9 (0.5) | 0.24 | - |
| Caudal Region Symmetry (left–right) | 0.1 (0.2) | 0.5 (0.2) | 0.5 (0.2) | 0.29 | - | −0.0 (0.4) | 0.7 (0.3) | 0.4 (0.3) | 0.47 | - |
| Difference between cranial and caudal | −1.1 (0.5) | −1.1 (0.5) | −1.5 (0.5) | 0.22 | - | −0.6 (1.0) | −2.2 (0.8) | −0.9 (0.8) | 0.45 | - |
| Maximum Temperatures (°C) | ||||||||||
| Left Cranial Region | 26.7 (0.5) | 30.9 (0.5) | 29.7 (0.5) | <0.0001 | BL < PL, p = <0.0001 BL < PR, p = 0.002 | 28.5 (0.6) | 28.1 (0.5) | 30.6 (0.5) | 0.05 | - |
| Right Cranial Region | 26.9 (0.4) | 29.7 (0.4) | 29.9 (0.4) | <0.0001 | BL < PL, p = 0.001 BL < PR, p = <0.0001 | 28.1 (0.8) | 27.9 (0.6) | 30.0 (0.6) | 0.16 | - |
| Left Caudal region | 26.9 (0.4) | 29.5 (0.4) | 30.1 (0.4) | 0.001 | BL < PL, p = 0.005 BL < PR, p = 0.002 | 28.1 (0.6) | 28.6 (0.5) | 29.6 (0.5) | 0.28 | - |
| Right Caudal Region | 26.9 (0.4) | 29.8 (0.4) | 30.1 (0.4) | 0.001 | BL < PL, p = 0.002 BL < PR, p = 0.001 | 28.5 (0.6) | 28.7 (0.5) | 29.6 (0.5) | 0.40 | - |
| Cranial Region Symmetry (left–right) | 0.7 (0.3) | 0.5 (0.3) | −0.1 (0.3) | 0.12 | - | 0.4 (0.3) | 0.2 (0.3) | 0.6 (0.3) | 0.71 | - |
| Caudal Region Symmetry (left–right) | −0.0 (0.1) | −0.3 (0.1) | −0.1 (0.1) | 0.38 | - | −0.3 (0.2) | −0.7 (1.6) | 0.2 (0.1) | 0.34 | - |
| Difference between cranial and caudal | −0.6 (0.3) | 0.9 (0.3) | −0.2 (0.3) | 0.006 | BL < PL, p = 0.006 PL > PR, p = 0.04 | 0.3 (0.3) | −0.6 (0.2) | 0.6 (0.2) | 0.05 | - |
| Mean Temperatures (°C) | ||||||||||
| Left Cranial Region | 23.1 (0.6) | 26.3 (0.6) | 25.2 (0.6) | 0.003 | BL < PL, p = 0.003 BL < PR, p = 0.03 | 24.4 (0.9) | 24.5 (0.8) | 25.7 (0.8) | 0.53 | - |
| Right Cranial Region | 22.1 (0.6) | 26.1 (0.6) | 24.9 (0.6) | <0.0001 | BL < PL, p ≤ 0.0001 BL < PR, p = 0.006 | 24.6 (0.9) | 23.3 (0.8) | 25.3 (0.8) | 0.29 | - |
| Left Caudal region | 23.3 (0.6) | 26.1 (0.6) | 26.2 (0.6) | 0.003 | BL < PL, p = 0.008 BL < PR, p = 0.006 | 24.2 (1.1) | 25.1 (0.8) | 26.2 (0.8) | 0.40 | - |
| Right Caudal Region | 23.3 (0.8) | 25.8 (0.8) | 26.0 (0.8) | 0.006 | BL < PL, p = 0.01 BL < PR, p = 0.01 | 24.5 (1.4) | 24.7 (1.1) | 26.0 (1.1) | 0.67 | - |
| Cranial Region Symmetry (left–right) | 0.9 (0.3) | 0.1 (0.1) | 0.3 (0.30 | 0.14 | - | −0.1 (0.6) | 1.2 (0.5) | 0.4 (0.5) | 0.29 | - |
| Caudal Region Symmetry (left–right) | −0.0 (0.2) | 0.2 (0.2) | 0.2 (0.2) | 0.32 | - | −0.2 (0.4) | 0.4 (0.3) | 0.2 (0.3) | 0.45 | - |
| Difference between cranial and caudal | 0.7 (0.3) | −0.2 (0.3) | 1.0 (0.3) | 0.006 | BL > PL, p = 0.02 PL < PR, p = 0.008 | −0.1 (0.4) | 0.9 (0.3) | 0.6 (0.3) | 0.22 | - |
| Correct Saddle Width (°C) Mean ± SD | Narrow Saddle Width (°C) Mean ± SD | Wide Saddle Width (°C) Mean ± SD | Saddle Width Main Effects (ANOVA) p Value | Pairwise Tukey Post Hoc p ≤ 0.05 | |
|---|---|---|---|---|---|
| Minimum Temperature (°C) | |||||
| Cranial Region (left and right) | 19.6 ± 2.5 | 17.8 ± 1.6 | 19.2 ± 0.6 | 0.04 | - |
| Mid Region (left and right) | 19.4 ± 0.9 | 17.2 ± 1.5 | 20.1 ± 0.8 | 0.06 | - |
| Caudal Region (left and right) | 20.8 ± 1.4 | 17.2 ± 2.5 | 20.8 ± 1.5 | 0.13 | - |
| Cranial Symmetry (Difference between left and right, cranial region) | 0.6 ± 1.4 | 0.2 ± 0.5 | −0.7 ± 0.2 | 0.19 | - |
| Mid Symmetry (Difference between left and right, mid region) | 2.7 ± 4.1 | 0.6 ± 1.5 | 1.2 ± 1.1 | 0.62 | - |
| Caudal Symmetry (Difference between left and right, caudal region) | 2.2 ± 0.7 | −2.3 ± 4.9 | 0.2 ± 0.4 | 0.33 | - |
| Cranial-caudal symmetry (Differences between front and back) | −1.2 ± 0.3 | −1.0 ± 1.9 | −1.1 ± 1.6 | 0.98 | - |
| Maximum Temperature (°C) | |||||
| Cranial Region (left and right) | 23.6 ± 1.6 | 22.2 ± 0.5 | 23.1 ± 1.2 | 0.32 | - |
| Mid Region (left and right) | 23.6 ± 1.6 | 22.2 ± 0.5 | 23.1 ± 1.2 | 0.46 | - |
| Caudal Region (left and right) | 24.2 ± 1.6 | 22.5 ± 0.3 | 23.4 ± 1.4 | 0.35 | - |
| Cranial Symmetry (Difference between left and right, cranial region) | 24.2 ± 1.5 | 22.9 ± 0.1 | 23.8± 1.7 | 0.53 | - |
| Mid Symmetry (Difference between left and right, mid region) | 0.8 ± 0.7 | −0.4 ± 0.2 | 0.1 ± 0.1 | 0.04 | Correct > narrow, p = 0.03 |
| Caudal Symmetry (Difference between left and right, caudal region) | −0.6 ± 0.8 | −0.1 ± 0.9 | −0.3 ± 0.3 | 0.81 | - |
| Cranial-caudal symmetry (Differences between front and back) | −0.4 ± 0.7 | 0.2 ± 0.7 | −0.2 ± 0.3 | 0.53 | - |
| Mid Region (left and right) | −0.3 ± 0.1 | −0.5 ± 0.3 | −0.5 ± 0.4 | 0.89 | - |
| Mean Temperature (°C) | |||||
| Cranial Region (left and right) | 22.6 ± 2.1 | 20.1 ± 0.8 | 21.8 ± 1.1 | 0.67 | - |
| Mid Region (left and right) | 22.6 ± 2.1 | 20.1 ± 0.8 | 21.8 ± 1.1 | 0.16 | - |
| Caudal Region (left and right) | 22.8 ± 1.9 | 20.5 ± 0.6 | 22.4 ± 1.3 | 0.16 | - |
| Cranial Symmetry (Difference between left and right, cranial region) | 23.7± 1.5 | 21.7 ± 0.2 | 22.8 ± 1.5 | 0.30 | - |
| Mid Symmetry (Difference between left and right, mid region) | 0.7 ± 0.3 | −0.3 ± 0.4 | 0.1 ± 0.3 | 0.08 | - |
| Caudal Symmetry (Difference between left and right, caudal region) | 0.6 ± 0.7 | −0.2 ± 0.4 | 0.3 ± 0.3 | 0.26 | - |
| Cranial-caudal symmetry (Differences between front and back) | −0.2 ± 0.5 | −0.1 ± 0.3 | 0.5 ± 0.4 | 0.21 | - |
| Mid Region (left and right) | −0.1 ± 0.4 | −1.4 ± 0.7 | −0.6 ± 0.7 | 0.43 | - |
| Correct Saddle Width (kPa) Mean ± SD | Narrow Saddle Width (kPa) Mean ± SD | Wide Saddle Width (kPa) Mean ± SD | Saddle Width Main Effects (ANOVA) p Value | Pairwise Tukey Post Hoc p ≤ 0.05 | |
|---|---|---|---|---|---|
| Mean Saddle Pressures (kPa) | |||||
| Left Cranial Region | 17.2 ± 2.4 | 20.2 ± 6.3 | 25.1 ± 7.4 | 0.06 | - |
| Right Cranial Region | 17.5 ± 2.9 | 20.4 ± 8.5 | 23.9 ± 6.1 | 0.19 | - |
| Left Caudal Region | 5.1 ± 1.1 | 5.1 ± 3.5 | 6.8 ± 2.2 | 0.31 | - |
| Right Caudal Region | 7.4 ± 1.6 | 5.5 ± 4.2 | 9.6 ± 2.2 | 0.03 | Narrow < wide, p = 0.05 |
| Cranial Region Symmetry (Left–Right) | −0.3 ± 1.5 | −0.1 ± 2.5 | 1.1 ± 1.8 | 0.29 | - |
| Caudal Region Symmetry (Left–Right) | −2.2 ± 1.0 | −0.4 ± 0.9 | −2.8 ± 0.9 | <0.0001 | Correct > Narrow, p = 0.009 Narrow < wide, p ≤ 0.0001 |
| Difference between Cranial and Caudal regions (front–back) | 11.1 ± 1.3 | 15.0 ± 5.6 | 16.2 ± 5.1 | 0.05 | - |
| Peak Saddle Pressures (kPa) | |||||
| Left Cranial Region | 36.6 ± 2.8 | 51.5 ± 7.8 | 51.1 ± 18.1 | 0.05 | - |
| Right Cranial Region | 38.3 ± 2.9 | 53.1 ± 13.2 | 53.1 ± 13.3 | 0.03 | Correct < narrow, p = 0.04 Correct < wide p = 0.04 |
| Left Caudal Region | 21.7±5.1 | 20.4 ± 6.9 | 23.4 ± 6.1 | 0.62 | - |
| Right Caudal Region | 25.5 ± 3.1 | 21.1 ± 8.8 | 27 ± 7.2 | 0.20 | - |
| Cranial Region Symmetry (Left—Right) | −1.7 ± 1.6 | −1.6 ± 9.2 | −2.1 ± 6.7 | 0.99 | - |
| Caudal Region Symmetry (Left—Right) | −3.8 ± 5.1 | −0.5 ± 2.9 | −3.8 ± 1.9 | 0.09 | - |
| Difference between Cranial and Caudal regions (front–back) | 13.8 ± 4.4 | 31.6 ± 10.8 | 26.7 ± 11.7 | 0.01 | Correct < narrow, p = 0.008 Correct < wide p = 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
MacKechnie-Guire, R.; Fisher, M.; Mathie, H.; Kuczynska, K.; Fairfax, V.; Fisher, D.; Pfau, T. A Systematic Approach to Comparing Thermal Activity of the Thoracic Region and Saddle Pressure Distribution beneath the Saddle in a Group of Non-Lame Sports Horses. Animals 2021, 11, 1105. https://doi.org/10.3390/ani11041105
MacKechnie-Guire R, Fisher M, Mathie H, Kuczynska K, Fairfax V, Fisher D, Pfau T. A Systematic Approach to Comparing Thermal Activity of the Thoracic Region and Saddle Pressure Distribution beneath the Saddle in a Group of Non-Lame Sports Horses. Animals. 2021; 11(4):1105. https://doi.org/10.3390/ani11041105
Chicago/Turabian StyleMacKechnie-Guire, Russell, Mark Fisher, Helen Mathie, Kat Kuczynska, Vanessa Fairfax, Diana Fisher, and Thilo Pfau. 2021. "A Systematic Approach to Comparing Thermal Activity of the Thoracic Region and Saddle Pressure Distribution beneath the Saddle in a Group of Non-Lame Sports Horses" Animals 11, no. 4: 1105. https://doi.org/10.3390/ani11041105
APA StyleMacKechnie-Guire, R., Fisher, M., Mathie, H., Kuczynska, K., Fairfax, V., Fisher, D., & Pfau, T. (2021). A Systematic Approach to Comparing Thermal Activity of the Thoracic Region and Saddle Pressure Distribution beneath the Saddle in a Group of Non-Lame Sports Horses. Animals, 11(4), 1105. https://doi.org/10.3390/ani11041105






