Simple Summary
Reproductive biotechnologies in beef farms are key issues, such as artificial insemination, fixed-time artificial insemination (FTAI), embryo transfer, and ovum pick up, among others. Artificial insemination takes the first place with these available tools. Thus, science keeps improving hormonal treatments and procedures to optimize pregnancies after insemination. A synchronization protocol commonly used in beef heifers is the 5d Co-synch protocol, which fits better in terms of ovarian synchronization and resulting conception rate. We propose a modification of this protocol, which consists of delaying by 24 h the removal of the intravaginal progesterone device, to decrease the percentage of heifers showing estrus before the date of FTAI. Moreover, this modification will make easier the workload when inseminating herds with heifers and cows at the same time. Based on our results, the protocol 5d Co-synch with the delayed removal of the intravaginal progesterone device fits as well as the original protocol in terms of synchronization and conception rate. Therefore, it can be implemented in the heifers of beef cattle commercial farms.
Abstract
In beef herds, increasing animal welfare, improving reproductive performance and easing animal management are key goals in farm economics. We explored whether delaying the removal of the intravaginal progesterone device by 24 h in heifers synchronized with a 5d Co-synch 72-h protocol could improve reproductive efficiency of fixed-time artificial insemination (FTAI). In experiment 1, we examined the total synchronization rate (TSR) in cycling Holstein heifers. Heifers (13.4 ± 0.69 mo.) were randomly assigned to the standard 5d Co-synch 56-h protocol (5dCo56; n = 10), 5d Co-synch 72-h (5dCo72; n = 17), or the modified 5d Co-synch 72-h protocol, in which removal of the progesterone device was delayed by 24 h (6dCo48; n = 19). In experiment 2, 309 cycling beef heifers on 18 commercial farms were subjected to the 5d Co-synch 72-h or 6-d Co-synch 48-h protocol and conception rate (CR) studied. In experiment 1, the three protocols led no differences on TSRs of 80.0% (5dCo56), 88.2% (5dCo72), and 89.5% (6dCo48). In experiment 2, the CR from the beef heifers, observed during two consecutive reproductive seasons did not differ: 59.7% for 5dCo72 and 62.0% for 6dCo48 (p = 0.907). Therefore, delaying removal by 24 h provides satisfactory results without reducing reproductive efficiency of heifers.
1. Introduction
The first protocol to synchronize ovulation for fixed-time artificial insemination (FTAI) based on gonadotropin-releasing hormone (GnRH) was reported in 1995 [1]. Since then, several modifications have been developed, including such protocols as G6G [2], double-ovsynch [3], presynch-ovsynch [4], five-day ovsynch [5,6], seven-day Co-synch [7], five-day Co-synch [8], and others [9,10]. Research on fixed-time artificial insemination has traditionally focused on dairy cattle, but some protocols and data are available for beef cattle [10]. In fact, certain FTAI protocols have been developed explicitly for beef cattle, such as the five- and seven-day Co-synch protocols plus an intravaginal progesterone (P4) device (IPD) [10].
In GnRH-based protocols, the initial GnRH is intended to induce LH release and ovulation, with emergence of a new follicular wave approximately 2 d later and luteinization of the follicle present at administration guaranteeing an appropriate level of progesterone during follicle growth. The 5d Co-synch protocol, in which FTAI is performed 72 h after the first prostaglandin administration, shortens the follicular dominance by two days and lengthens the proestrus phase by 16–24 h, ultimately leading to higher mean conception rate (CR) [8,11,12,13,14].
Some protocols administer GnRH on Day 0 to enhance subsequent response to GnRH on Day 8 in beef heifers [15] and dairy heifers [16], increasing CR [17]. Some studies suggest that GnRH on Day 0 provides no reproductive benefits in dairy or beef heifers [5,18] and may even induce pregnancy loss in acyclic heifers [19]. However, an FTAI protocol in which cows received prostaglandin three days before the protocol began and then were artificially inseminated at 66 h after IPD removal led to a higher percentage of cows showing a new follicular wave after the initial GnRH as well as higher conception rate than the classical 5d Co-synch 72-h protocol [20]. Such pre-synchronization has been associated with higher CR in dairy heifers [21], higher fertility rate after FTAI in multiparous beef cattle [20], and greater ovarian synchronization in beef heifers [22]. In beef heifers, however, another study did not observe different CRs between the 5d Co-synch 72-h protocol and a prostaglandin 6d-IPD protocol, consisting of prostaglandin (25 mg) on Day −9, GnRH (100 μg) and IPD insertion on Day −6, PG and IPD removal on Day 0, and FTAI at 66 h after IPD removal [23]. Unlike the prostaglandin 6d-IPD protocol, the modified protocol in the present study did not include pre-synchronization with prostaglandin.
In contrast, early work [24] suggested that the 5d Co-synch 56-h (5dCo56) protocol was more appropriate for beef heifers, probably because following P4 device removal, heifers tend to display estrus earlier than cows. However, another study [25] reported higher conception rate in dairy heifers if they received the final GnRH administration concurrent with Artificial Insemination (AI) at 72 h after PGF than if they received it at 16 h before AI. Still another study found no fertility difference between beef heifers who received FTAI at 66 or 72 h after GnRH [19].
A shortened proestrus phase should not affect heifer synchronization or CR, as long as prostaglandin administration is maintained on Days 5 and 6 of the protocol. In fact, the serum concentration of progesterone induced by an intravaginal device is significantly lower than that induced by a natural CL [26]. Prostaglandin administration on Days 5 and 6 promotes corpus luteum (CL) regression [16] and increases CR in dairy and beef heifers [27,28]. While some studies question the usefulness of the second prostaglandin dose in heifers [18,19,29], being indispensable for FTAI programs in multiparous cows [30,31].
A preliminary study [32] found no difference in CR in dairy heifers with a conventional 5d Co-synch 72-h protocol or a modified protocol (Day 0: GnRH + IPD; Day 5: PGF; Day 6: PGF + IPD removal; Day 8: GnRH + FTAI). They described that delaying IPD removal from dairy heifers by 24 h during the 5d Co-synch protocol avoided the demonstration of estrus earlier than 16 h prior to FTAI. However, that study did not examine whether the modified protocol altered the ovarian dynamics compared to the 72-h protocol, nor did it compare total synchronization rates (TSR). The administration of eCG promoting follicular growth in the proestrus stage is another possibility to enhance results, even in cycling animals, as described by other authors [33]. Thus, the optimal window when ovulation should be induced with GnRH relative to FTAI in heifers subjected to the 5d Co-synch protocol warrants further research. Indeed, modification of existing Co-synch protocols may increase the versatility of FTAI programs on beef farms.
New hormonal procedures could increase synchronization and conception rates, together with other strategies, such as nutritional ones [34], body condition score improvement [35], and avoiding stress while handling [36].
The modified protocol, previously proposed [32], henceforth referred to as “6dCo48”, consisted of IPD removal delayed by 24 h relative to the conventional 5dCo72 protocol. We compared these two protocols in terms of ovarian synchronization and conception rates, with the aim at testing its value in the field, in beef heifers. Therefore, the objective of experiment 1 was to examine whether the 6dCo48 protocol would provide an adequate TSR compared to 5dCo72 and 5dCo56 protocols in dairy heifers, based on analysis of follicular growth, luteolysis, and ovulation. Dairy heifers were chosen (easier to obtain an adequate sample of animals homogeneous in age, size, weight and ovarian stage, under a controlled environment) to check that this modification did not significantly worsen the ovarian response nor synchronization rate. The objective of experiment 2 was to compare CR of beef heifers in commercial beef herds after FTAI using the 6dCo48 or 5dCo72 protocols (the two protocols with the best synchronization rate in experiment 1). Our hypothesis was that delaying by 24 h the removal of the intravaginal progesterone device within a 5dCo72 protocol in heifers would not negatively affect conception rates after FTAI and might improve the synchronization rate of heifers, making it an additional synchronization protocol available to beef farmers.
2. Material and Methods
2.1. Experiment 1: Synchronization Study
Experiment 1 was conducted in a small sample of Holstein heifers to ensure that the new protocol (6dCo48) would not induce a worse synchronization rate than conventional protocols.
Holstein heifers on a single dairy farm in Toledo, in central Spain, were housed in free-stall barns and fed a total mixed ration adapted to their daily requirements [34], with ad libitum access to feed and water. A total of 46 dairy heifers were included, 29 during a first replicate (March 2017) and 17 during a second replicate (April 2017). Two replicates with different heifers were performed due to the few heifers’ availability at once. To be included in the study, animals had to be in adequate health; show a body condition score (defined below) ≥2 and <3.5; lack a history of artificial insemination; and lack anoestrus. Animals needed to present P4 concentrations higher than 1 ng/mL in one or both samples taken seven days apart previous to Day 0 of the study. In addition, initial ovarian cyclicity had to be observed by transrectal ultrasonography on Days −7 and 0 (see Section 2.2.). Of the 51 heifers screened for inclusion, five were excluded because they showed no CL on at least one day, and subsequent P4 assay showed concentrations below 1 ng/mL.
On Day 0 of the protocols, body condition score (BCS) on a scale from 1 (cachectic) to 5 (extremely obese) [35] and stress score (SS) were determined. SS was measured on a scale from 1 to 5 [37] when animals were in the chute, where a score of 1 meant that the animal was calm, without unexpected, sudden movements; 2, the animal was slightly restless; 3, the animal was squirming and occasionally shaking; 4, the animal moved continuously with very vigorous movements that shook the chute; and 5, the animal was rearing, twisting the body, and struggling violently [37].
In the first replicate (March 2017), a total of 29 dairy heifers were assessed for age, BCS, and SS (Table 1) and randomly assigned to undergo the 5dCo72 protocol (n = 9), 5dCo56 protocol (n = 10), or 6dCo48 protocol (n = 10). All protocols involved the insertion of an intravaginal progesterone device. In the second replicate (April 2017), dairy heifers (Table 1) were randomly assigned to undergo our 6dCo48 protocol (n = 9) or 5dCo72 protocol (n = 8), since the 5dCo72 protocol gave as good TSR as the 5dCo56 protocol in the first replicate (see Results).

Table 1.
Characteristics of dairy heifers included in experiment 1, by fixed-time AI experimental protocols.
The protocol 5dCo72 was performed as described [8]. Briefly, it consisted of intramuscular administration of 100 μg GnRH (Cystoreline® CEVA Santé Animale SA, Libourne, France) and insertion of a 1.55-g IPD (PRID-delta®, CEVA) on Day 0. On Day 55, 25 mg dinoprost (Enzaprost T®, CEVA) and 500 I.U. eCG (Syncrostim® 500 I.U., CEVA) were administered intramuscularly, and the progesterone device was removed. The 500IU eCG dosage was decided based on drug management guidance for Syncrostim® (SPC product: F-DVM-01-03). This dosage has been demonstrated to be safe and not associated with increased rate of multiple ovulation [33]. On Day 6, 25 mg prostaglandin was administered intramuscularly. On Day 8, 72 h after removal of the intravaginal progesterone device, FTAI was performed and 100 μg GnRH was administered intramuscularly. The 5dCo56 protocol was identical to 5dCo72, except that FTAI and GnRH treatment were performed at 56 h after removal of the progesterone device. The 6dCo48 protocol was identical to 5dCo72, except that the progesterone device was removed on Day 6, simultaneously with the second prostaglandin administration (Figure 1).
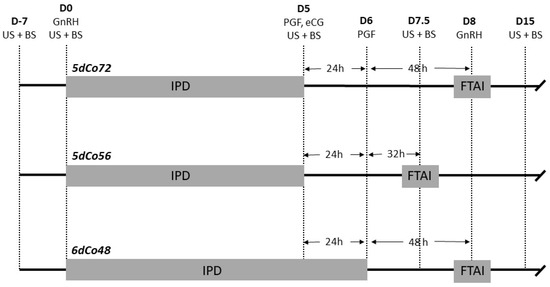
Figure 1.
Illustration of treatments and activities during experiment 1: 5dCo72 = 5d Co-synch 72-h protocol with intravaginal progesterone device (IPD); 5dCo56 = 5d Co-synch 56-h protocol with IPD; 6dCo48 = 6-d Co-synch 48-h with IPD; FTAI = Fixed Time Artificial Insemination; D = Day of the study; US = ultrasonography; BS = blood sampling; GnRH = gonadorelin; IPD = intravaginal progesterone device; PGF = prostaglandin; eCG = equine chorionic gonadotropin.
Unfortunately, CR after FTAI could not be analyzed, because heifers received conventional or sex-sorted semen from different bulls based on farmer assessments. This may have confounded analyses of CR. Nevertheless, we considered the TSR results to be sufficiently reliable to proceed to a field test of the modified protocol in beef heifers in experiment 2 of the study.
2.2. Ultrasonography and Blood Analyses
Transrectal ultrasonography (US) was performed by the same experienced veterinarian on every heifer using a SIUI CTS 800® system (Guangdong, China) with 7.0-MHz transrectal transducer on Days −7, 0, 5, 7.5, and 15. A detailed ovarian analysis was performed in order to link hormonal results to ovarian structure and function. Ovulation was considered to have occurred after the first GnRH if a CL was observed on Day 5 in the ovary where a follicle (diameter > 8 mm) was previously located. Ovulation was considered to have occurred after the second GnRH if on Day 15 there was a CL where the dominant follicle (diameter > 8 mm) was located. Luteolysis was considered to have occurred after the second prostaglandin if on Day 7.5 there was no longer a CL where it had previously been detected. The largest follicle diameter (in mm) was measured on Days 5 and 7.5, and an ovarian map was drawn (from Day 7 to Day 15) in order to study ovarian dynamics.
To explore whether P4 values on D0 influenced synchronization efficacy, we categorized initial P4 concentration on D0 as low (<0.6 ng/mL), intermediate (0.6–7 ng/mL), or high (≥ 7ng/mL). These categories were based on a study in dairy cows [38] that reported higher CR with intermediate P4 concentrations of 0.5–6 ng/mL on Day 0. Attending the metabolic differences between cattle and heifers, the cut-off values were changed in the current study. Moreover, the classical cut-off of <1 or >1 ng/mL P4 was also applied [30] to double check whether P4 values could interfere on the Co-synch modification proposed.
An active CL produces >1 ng/mL P4, and its diameter ranges between 21 and 26 mm [39]. Heifers were considered to be synchronized if they had a CL and P4 > 1 ng/mL on Day 5, underwent luteolysis on Day 7.5, and had a CL and P4 > 1 ng/mL on Day 15. The total synchronization rate (TSR) was calculated based on the number of animals synchronized on Days 5, 7.5, and 15.
Blood was sampled from the coccygeal vein into 4-mL EDTA K2 vacutainer tubes (Fisher Scientific, Pittsburgh, PA, USA), immediately centrifuged at 4500 g for 15 min, and the plasma was transferred to a fresh tube and stored at −80 °C until progesterone determination. Plasma progesterone concentrations were measured in a single analysis using an enzyme immunoassay kit (Demeditec Diagnostics, Kiel-Wellsee, Germany) as described [40]. Assay sensitivity was 0.045 ng/mL and the manufacturer-specified intra-assay variation coefficient was 5%.
2.3. Experiment 2: Field Study
A total of 309 beef heifers from 18 commercial beef farms in central and south Spain were included in the study. The animals grazed on pastures and were fed once daily with a complete fodder adapted to their requirements [41] and with ad libitum access to water. Data were collected during two consecutive reproductive seasons, from 187 animals during autumn–spring (2017–2018) and from 122 animals during autumn–spring (2018–2019). Of all 309 animals, 137 were crossbreds, and the remaining 172 heifers were full-bred, comprising 106 Limousine, 55 Charolaise, and 11 Spanish Black Iberian Avileña.
Inclusion criteria for farms were an adequate farm health program, routine SS assessment at the beginning of the reproductive season, and a nutritional program with supplementation when recommended. Individual inclusion criteria for heifers were no prior insemination, nulliparity, age ≥ 17.5 and < 25 months, BCS ≥ 2.5 and ≤ 3.5, and presence of a CL at the beginning of the FTAI protocol. These parameters were measured on Day 0, as described for experiment 1.
Heifers within each farm were randomly submitted to one of two protocols, 5dCo72 or 6dCo48 (Table 2). First, FTAI was performed by two experienced veterinarians using commercial frozen semen from 41 bulls. Pregnancy was diagnosed by US on Day 30–45 after insemination and the conception rate (CR) was calculated. A subset of 122 heifers (39.50%) was randomly selected for blood sampling and P4 assay (see Section 2.2) at the beginning of the 5d Co-synch. This assay allowed us to analyze whether initial P4 affected rates of synchronization.

Table 2.
Characteristics of beef heifers included in experiment 2, by fixed-time AI experimental protocols.
2.4. Statistical Analyses
All data were analyzed using SPSS® 25 (IBM, Armonk, NY, USA). Probability values less than or equal to 0.05 were considered significant, and those between 0.05 and 0.10 were considered trends. All data were reported as mean (percentage) or as mean ± SD. Inter-group differences were assessed for significance using the chi-squared and Student’s t test when data were normally distributed or using non-parametric analyses (Kruskal–Wallis) when data were skewed. Results from both phases of the study were analyzed using logistic regression that included farm as a fixed factor and several possible confounding factors in a stepwise forward method based on the Wald statistic criterion p > 0.10. In experiment 2 of the study, data were separately analyzed in two regression models, one with data on all 309 animals, and the other with data on 122 animals with the P4 information.
3. Results
3.1. Experiment 1
Replicates were clustered by introducing the factor “replicate” in the regression model. Percentages of heifers synchronized at the different time points in the protocol and totally synchronized are summarized in Table 3. TSR did not vary significantly with any of the categorical factors included in the study, including synchronization protocol.

Table 3.
Summary of synchronization results of dairy heifers in experiment 1 by synchronization days.
The age on D0 was not different between totally synchronized heifers (13.5 ± 0.89 months) and other heifers (13.6 ± 0.65 mo.; p = 0.699). Similarly, the size of the largest follicle on Day 7.5 was not different between totally synchronized heifers (12.8 ± 2.29 mm) and not totally synchronized heifers (12.8 ± 1.16 mm; p = 0.968). The P4 values at the time points D0, D5, and D7.5 did not influence the probability of synchronization on the subsequent day where synchronization was evaluated, nor the probability of being totally synchronized at the end of the protocol. The exception was P4 on Day 7.5: each 1-ng/mL P4 increase on Day 7.5 was associated with significantly lower probability of total synchronization at the end of the protocol (OR 0.02, 95% CI 0.001-0.416; p = 0.012).
3.2. Experiment 2
Pregnant heifers after FTAI (n = 188/305) were 21.7 ± 3.77 months old, and non-pregnant ones (n = 121/305) were 21.7 ± 3.32 months old (p = 0.922). Mean BCS was 3.15 ± 0.478 for pregnant animals, and 3.22 ± 0.503 for non-pregnant ones (p = 0.277). Similarly, SS was 2.03 ± 0.701 in pregnant heifers and 2.13 ± 0.763 in non-pregnant ones (p = 0.235). Conception rates by group, breed, AI technician, and P4 concentrations on Day 0 are summarized in Table 4.

Table 4.
Conception rates of beef heifers in experiment 2 and effect of the different factors analyzed.
Farm and semen did not significantly influence conception rate (p = 0.907 and 0.329, respectively) in the regression model. Similarly, season (p = 0.924), interaction between age and protocol (p = 0.746), and interaction between AI technician and protocol (p = 0.706) did not significantly affect conception rate.
4. Discussion
This study comprises a 5dGnRH-based Co-synch protocol without presynchronization but with GnRH on Day 0 and with prostaglandin administration on Days 5 and 6. The proposed 6dCo48 protocol, a modified version of the conventional 5dCo72 protocol, achieved an ovarian synchronization rate of 89.5% and CR of 62.0%, which were not different from those obtained with the conventional protocol. Thus, the 6dCo48 may be an alternative protocol to optimize the reproductive management of beef heifers subjected to FTAI, enhancing flexibility when considering implementing FTAI strategies in beef cattle farms, without worsening meaningfully reproductive efficiency.
If farmers decide to implement different protocols for cows (5dCo72h) and heifers (5dCo56h), artificial insemination of a single herd must occur in two steps, once in heifers and 16 h later in cows. With the protocol described here, in contrast, beef heifers and multiparous cows can be synchronized and inseminated at the same time. Moreover, as hypothesized, our modification of the conventional 5dCo-synch protocols did not clinically worsen reproductive outcomes in heifers, probably because we maintained prostaglandin administration on Day 5. This mitigated the effect of proestrus shortening on reproductive efficiency. Although the sample size of the study may be a limitation in statistical power, a meaningful difference of 15% CR could have been detected.
A total synchronization rate of 80–90% was observed in the current work with the three hormonal protocols tested in dairy heifers, similarly to previous studies [19,25]. This phase of the study was performed in Holstein heifers, because it was easier to obtain an adequate sample of animals homogeneous in age, size, weight, and ovarian stage that could be maintained in the same controlled environment within a proper handling facility. Moreover, dairy heifers show a comparable ovarian physiology to beef heifers [20,21]. Likewise, a conception rate around 60% was obtained in beef heifers, comparable to that achieved in other studies with the protocol Co-synch 72 and 56 h [9,10,13,14,16,18,24,27,28,29,30,31].
Nevertheless, we cannot exclude that the modified protocol may be associated with higher risk that a heifer has an old oocyte when inseminated [8], resulting in increased pregnancy loss.
In experiment 1, mean age of the 46 dairy heifers in the first part of the study was 13.5 ± 0.86 months, and did not differ between synchronized and other animals. Similarly, the age of beef heifers in the second part of this study did not influence the probability of pregnancy after FTAI. For adequately developed beef heifers, reaching puberty before FTAI may be even more important for fertility than age [23]. Age may not have influenced conception rate in our study because the heifers (21.7 ± 3.54 months old) had reached puberty and had adequate body condition. Since these two factors are the main determinants of fertility in heifers [42,43], they may also explain the lack of differences on TSR between our experimental groups. We also cannot exclude that the lack of a significant difference in TSR is an artifact of the relatively small samples in our study.
Body condition score did not affect the TSR in dairy heifers, which is not surprising given that scores were in the physiological range between 2.5 and 3.5. In contrast, cachectic or extremely obese body condition reduces reproductive performance in cattle [44,45,46]. As in dairy heifers, BCS in our beef heifers also did not affect CR, independently of synchronization protocol. The best reproductive results can be achieved in beef herds [47] with an appropriate nutrition plan [48,49,50] that allows them to achieve a balanced BCS of 2.5–3 at the beginning of the reproductive season. This balanced BCS in heifers is associated with appropriate hormonal regulation [47,50].
Controlling stress reduces cortisol release [51] and ensures cyclical secretion of luteinizing hormone [52,53], improving reproductive efficiency [54,55]. Our dairy heifers showed an SS of 1.71 ± 0.714, consistent with low stress and appropriate management [56]. This may help explain why SS did not influence TSR. SS was similarly low (around 2) in pregnant and non-pregnant beef heifers, which may reflect that beef farms in our study had already implemented stress-reducing measures at the chute. Studies have suggested that SS < 3 indicates good animal temperament [54,55,56,57]. The uniformly low SS in our animals may help explain why conception rate did not differ significantly with beef breed or with the interaction between breed and FTAI protocol. Our results suggest that although CR can depend on temperament in certain breeds [54,55,57], animal temperament in our study was modulated mainly by management.
We found no significant difference in synchronization rates between animals stratified by P4 concentrations on Day 0, using a stratification approach like previous work [38]. That previous work suggested that initial P4 in dairy cows undergoing FTAI should range between 0.5 and 6 ng/mL at the first GnRH administration [38]. When we categorized our animals by initial P4 concentration, we broadened the definition of “intermediate” concentrations to consider that heifers show higher P4 concentrations than dairy cows [58]. We did find a tendency toward higher TSR in dairy heifers with intermediate P4 concentrations on Day 0, but this may not be real, since only one heifer in experiment 1 was in the estrus stage of its cycle on Day 0, and more than 50% of animals showed initial P4 values >7 ng/mL. Our failure to observe a robust dependence of TSR or CR on initial P4 concentration may reflect differences in ovarian physiology between dairy cattle and heifers [59], suggesting that beef heifers can show a strong ovarian response to FTAI protocols independently of initial P4. At the same time, we cannot exclude that the lack of significant variation in TSR or CR reflects the small samples in our study.
Nevertheless, we did find that P4 concentration on Day 7.5, when luteolysis should happen, inversely and significantly affected the rate of synchronization on that day (OR 0.02, 95% CI 0.001–0.416). This may reflect that progesterone inhibits luteinizing hormone release through a negative feedback loop, disrupting ovulation at the end of the synchronization protocol [60].
We found no difference on follicle size around 12.8 ± 2.29 mm before AI across all protocols, also, without differences to the 13.4 ± 0.3 mm reported for beef heifers under the 7-d and 5d Co-synch 72-h protocols [61] and the 11.0 ± 0.5 mm reported for dairy heifers under the 5d Co-synch 72-h protocol with PGF2α presynchronization at two days prior to IPD insertion [21]. Therefore, delaying removal of the intravaginal progesterone device may not meaningfully reduce the TSR.
In conclusion, our modification of the 5d Co-synch protocol for heifers, in which removal of the intravaginal progesterone device is delayed by 24 h, may achieve reproductive results as good as those obtained with 5d Co-synch protocols that stagger insemination of heifers and cows. Therefore, the 6dCo48 protocol seems to be a suitable protocol for beef heifers on commercial farms, which could provide another tool to expand FTAI possibilities. However, due to the limited sample size of this study, additional confirmatory studies are required.
Author Contributions
Conceptualization: S.A. methodology, all authors; software, A.F.-N., S.S.-L., J.L.P.-P., N.P.-V., J.V.G.-M. and S.A.; validation, A.F.-N., N.P.-V. and S.A.; formal analysis, A.F.-N., S.S.-L., J.L.P.-P., N.P.-V., A.H.-M. and S.A.; investigation, A.F.-N. and S.A.; resources, A.F.-N., S.S.-L., N.P.-V. and S.A.; data curation, A.F.-N., S.S.-L., N.P.-V., A.H.-M., J.V.G.-M. and S.A.; writing—original draft preparation, A.F.-N., N.P.-V. and S.A.; writing—review and editing, all authors; visualization, A.F.-N. and S.A.; supervision, A.F.-N., N.P.-V. and S.A.; project administration, S.A.; funding acquisition, A.F.-N., S.S.-L. All authors have read and agreed to the published version of the manuscript.
Funding
CEVA Santé Animale partially funded hormonal treatments.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
We are especially grateful to Antonio Jiménez and Jose Luis Pérez from CEVA Santé Animale for their contribution, support and knowledge. We thank Pedro Cuesta and Iagoba Cano (Department of Research Support, Complutense University of Madrid) for their help with statistical analyses. We thank all the farmers and farm workers for their contributions.
Conflicts of Interest
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Pursley, J.R.; Mee, M.O.; Wiltbank, M.C. Synchronization of ovulation in dairy cows using PGF2α and GnRH. Theriogenology 1995, 44, 915–923. [Google Scholar] [CrossRef]
- Bello, N.M.; Steibel, J.P.; Pursley, J.R. Optimizing ovulation to first GnRH improved outcomes to each hormonal injection of Ovsynch in lactating dairy cows. J. Dairy Sci. 2006, 89, 3413–3424. [Google Scholar] [CrossRef]
- Souza, A.H.; Ayres, H.; Ferreira, R.M.; Wiltbank, M.C. A new presynchronization system (Double-Ovsynch) increases fertility at first postpartum timed AI in lactating dairy cows. Theriogenology 2008, 70, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Moreira, F.; Orlandi, C.; Risco, C.A.; Mattos, R.; Lopes, F.; Thatcher, W.W. Effects of presynchronization and bovine somatotropin on pregnancy rates to a timed artificial insemination protocol in lactating dairy cows. J. Dairy Sci. 2001, 84, 1646–1659. [Google Scholar] [CrossRef]
- Colazo, M.G.; Ambrose, D.J. Effect of initial GnRH and duration of progesterone insert treatment on the fertility of lactating dairy cows. Reprod. Domest. Anim. 2015, 50, 497–504. [Google Scholar] [CrossRef]
- Santos, J.E.P.; Narciso, C.D.; Rivera, F.; Thatcher, W.W.; Chebel, R.C. Effect of reducing the period of follicle dominance in a timed artificial insemination protocol on reproduction of dairy cows. J. Dairy Sci. 2010, 93, 2976–2988. [Google Scholar] [CrossRef] [PubMed]
- El-Zarkouny, S.Z.; Cartmill, J.A.; Hensley, B.A.; Stevenson, J.S. Pregnancy in dairy cows after synchronized ovulation regimens with or without presynchronization and progesterone. J. Dairy Sci. 2004, 87, 1024–1037. [Google Scholar] [CrossRef]
- Bridges, G.A.; Helser, L.A.; Grum, D.E.; Mussard, M.L.; Gasser, C.L.; Day, M.L. Decreasing the interval between GnRH and PGF2 alpha from 7 to 5 days and lengthening proestrus increases timed-AI pregnancy rates in beef cows. Theriogenology 2008, 69, 843–851. [Google Scholar] [CrossRef]
- Sanz, A.; Macmillan, K.; Colazo, M.G. Revisión de los programas de sincronización ovárica basados en el uso de hormona liberadora de gonadotropinas y prostaglandina F2α para novillas de leche y de carne. ITEA 2019, 115, 326–341. [Google Scholar] [CrossRef]
- Patterson, D.J.; Thomas, J.M.; Martin, N.T.; Nash, J.M.; Smith, M.F. Control of estrus and ovulation in beef heifers. Vet. Clin. N. Am. Food. Anim. Pract. 2013, 29, 591–617. [Google Scholar] [CrossRef]
- Thatcher, W.W.; Macmillan, K.L.; Hansen, P.J.; Drost, M. Concepts for regulation of corpus luteum function by the conceptus and ovarian follicles to improve fertility. Theriogenology 1989, 31, 149–164. [Google Scholar] [CrossRef]
- Larson, J.E.; Lamb, G.C.; Stevenson, J.S.; Johnson, S.K.; Day, M.L.; Geary, T.W.; Kesler, D.J.; DeJarnette, J.M.; Schrick, F.N.; DiCostanzo, A.; et al. Synchronization of estrus in suckled beef cows for detected estrus and artificial insemination and timed artificial insemination using gonadotropin-releasing hormone, prostaglandin F2 alpha, and progesterone. J. Anim. Sci. 2006, 84, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Lucy, M.C.; Billings, H.J.; Butler, W.R.; Ehnis, L.R.; Fields, M.J.; Kesler, D.J.; Kinder, J.E.; Mattos, R.C.; Short, R.E.; Thatcher, W.W.; et al. Efficacy of an intravaginal progesterone insert and an injection of PGF2 alpha for synchronizing estrus and shortening the interval to pregnancy in postpartum beef cows, peripubertal beef heifers, and dairy heifers. J. Anim. Sci. 2001, 79, 982–995. [Google Scholar] [CrossRef]
- Ahmadzadeh, A.; Gunn, D.; Hall, J.B.; Glaze, J.B. Evaluation of treatment with a 5-day versus 7-day controlled internal drug-release insert on reproductive outcomes of beef heifers using a modified timed–artificial insemination protocol. Prof. Anim. Sci. 2015, 31, 270–277. [Google Scholar] [CrossRef]
- Atkins, J.A.; Busch, D.C.; Bader, J.F.; Keisler, D.H.; Patterson, D.J.; Lucy, M.C.; Smith, M.F. Gonadotropin-releasing hormone-induced ovulation and luteinizing hormone release in beef heifers:Effect of day of the cycle. J. Anim. Sci. 2008, 86, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Lima, F.S.; Ribeiro, E.S.; Bisinotto, R.S.; Greco, L.F.; Martinez, N.; Amstalden, M.; Thatcher, W.W.; Santos, J.E.P. Hormonal manipulations in the 5-day timed artificial insemination protocol to optimize estrous cycle synchrony and fertility in dairy heifers. J. Dairy Sci. 2013, 96, 7054–7065. [Google Scholar] [CrossRef]
- Kasimanickam, R.K.; Firth, P.; Schuenemann, G.M.; Whitlock, B.K.; Gay, J.M.; Moore, D.A.; Hall, J.B.; Whittier, W.D. Effect of the first GnRH and two doses of PGF2α in a 5-day progesterone-based CO-Synch protocol on heifer pregnancy. Theriogenology 2014, 81, 797–804. [Google Scholar] [CrossRef]
- Cruppe, L.H.; Day, M.L.; Abreu, F.M.; Kruse, S.; Lake, S.L.; Biehl, M.V.; Cipriano, R.S.; Mussard, M.L.; Bridges, G.A. The requirement of GnRH at the beginning of the five-day CO-Synch + controlled internal drug release protocol in beef heifers. J. Anim. Sci. 2014, 92, 4198–4203. [Google Scholar] [CrossRef]
- Helguera, I.L.; Whittaker, P.; Behrouzi, A.; Mapletoft, R.J.; Colazo, M.G. Effect of initial GnRH and time of insemination on reproductive performance in cyclic and acyclic beef heifers subjected to a 5-d Co-Synch plus progesterone protocol. Theriogenology 2018, 106, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Perry, G.A.; Perry, B.L.; Krantz, J.H.; Rodgers, J. Influence of inducing luteal regression before a modified fixed-time artificial insemination protocol in postpartum beef cows on pregnancy success. J. Anim. Sci. 2012, 90, 489–494. [Google Scholar] [CrossRef]
- Karakaya-Bilen, E.; Ribeiro, E.S.; Bisinotto, R.S.; Gümen, A.; Santos, J.E.P. Effect of presynchronization with prostaglandin F2α before the 5-d timed AI protocol on ovarian responses and pregnancy in dairy heifers. Theriogenology 2019, 132, 138–143. [Google Scholar] [CrossRef]
- Grant, J.K.; Abreu, F.M.; Hojer, N.L.; Fields, S.D.; Perry, B.L.; Perry, G.A. Influence of inducing luteal regression before a modified controlled internal drug-releasing device treatment on control of follicular development. J. Anim. Sci. 2011, 89, 3531–3541. [Google Scholar] [CrossRef]
- Bridges, G.A.; Lake, S.L.; Kruse, S.G.; Bird, S.L.; Funnell, B.J.; Arias, R.; Walker, J.A.; Grant, J.K.; Perry, G.A. Comparison of three CIDR-based fixed-time AI protocols in beef heifers. J. Anim. Sci. 2014, 92, 3127–3133. [Google Scholar] [CrossRef]
- Kasimanickam, R.; Asay, M.; Firth, P.; Whittier, W.D.; Hall, J.B. Artificial insemination at 56 h after intravaginal progesterone device removal improved AI pregnancy rate in beef heifers synchronized with five-day CO-Synch + controlled internal drug release (CIDR) protocol. Theriogenology 2012, 77, 1624–1631. [Google Scholar] [CrossRef] [PubMed]
- Lima, F.S.; Ayres, H.; Favoreto, M.G.; Bisinotto, R.S.; Greco, L.F.; Ribeiro, E.S.; Baruselli, P.S.; Risco, C.A.; Thatcher, W.W.; Santos, J.E.P. Effects of gonadotropin-releasing hormone at initiation of the 5-d timed artificial insemination (AI) program and timing of induction of ovulation relative to AI on ovarian dynamics and fertility of dairy heifers. J. Dairy Sci. 2011, 94, 4997–5004. [Google Scholar] [CrossRef] [PubMed]
- Bisinotto, R.S.; Castro, L.O.; Pansani, M.B.; Narciso, C.D.; Martinez, N.; Sinedino, L.D.P.; Pinto, T.L.C.; Van de Burgwal, N.S.; Bosman, H.M.; Surjus, R.S.; et al. Progesterone supplementation to lactating dairy cows without a corpus luteum at initiation of the Ovsynch protocol. J. Dairy Sci. 2015, 98, 2515–2528. [Google Scholar] [CrossRef] [PubMed]
- White, S.S.; Kasimanickam, R.K.; Kasimanickam, V.R. Fertility after two doses of PGF2α concurrently or at 6-hour interval on the day of CIDR removal in 5-day CO-Synch progesterone-based synchronization protocols in beef heifers. Theriogenology 2016, 86, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Say, E.; Çoban, S.; Nak, Y.; Nak, D.; Kara, U.; White, S.; Kasimanickam, V.; Kasimanickam, R. Fertility of Holstein heifers after two doses of PGF2α in 5-day CO-Synch progesterone-based synchronization protocol. Theriogenology 2016, 86, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Colazo, M.G.; Ambrose, D.J. Neither duration of progesterone insert nor initial GnRH treatment affected pregnancy per timed-insemination in dairy heifers subjected to a Co-Synch protocol. Theriogenology 2011, 76, 578–588. [Google Scholar] [CrossRef]
- Kasimanickam, R.; Day, M.L.; Rudolph, J.S.; Hall, J.B.; Whittier, W.D. Two doses of prostaglandin improve pregnancy rates to timed-AI in a 5-day progesterone-based synchronization protocol in beef cows. Theriogenology 2009, 71, 762–767. [Google Scholar] [CrossRef]
- Whittier, W.D.; Kasimanickam, R.K.; Currin, J.F.; Schramm, H.H.; Vlcek, M. Effect of timing of second prostaglandin F 2 alpha administration in a 5-day, progesterone-based CO-Synch protocol on AI pregnancy rates in beef cows. Theriogenology 2010, 74, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Fernandez, P.; Santos, V.; Carvalho, P.; Maia, C.; Carneiro, M.; Valenza, A.; Fricke, P. Delaying PRID Delta removal by 24 h during a 5-day PRID-Synch protocol decreased expression of estrus before timed artificial insemination without affecting fertility in Holstein heifers. In Proceedings of the 5th European Buiatrics Forum (EBF 2017), Bilbao, Spain, 4–6 October 2017; p. 104. [Google Scholar]
- Martinez, M.F.; Tutt, D.; Quirke, L.D.; Tattersfield, G.; Juengel, J.L. Development of a GnRH-PGF2α-progesterone-based synchronization protocol with ECG for inducing single and double ovulations in beef cattle. J. Anim. Sci. 2014, 92, 4935–4948. [Google Scholar] [CrossRef] [PubMed]
- National Research Council; Board on Agriculture and Natural Resources; Subcommittee on Dairy Cattle Nutrition; Committee on Animal Nutrition. Nutrient Requirements of Dairy Cattle, 7th revised ed.; National Academy Press: Washington, DC, USA, 2001. [Google Scholar] [CrossRef]
- Edmonson, A.J.; Lean, I.J.; Weaver, L.D.; Farver, T.; Webster, G. A body condition scoring chart for Holstein dairy cows. J. Dairy Sci. 1989, 72, 68–78. [Google Scholar] [CrossRef]
- Fernandez-Novo, A.; Pérez-Garnelo, S.S.; Villagrá, A.; Pérez-Villalobos, N.; Astiz, S. The effect of stress on reproduction and reproductive technologies in beef cattle-a review. Animals 2020, 10, 2096. [Google Scholar] [CrossRef]
- Grandin, T. Behavioral agitation during handling of cattle is persistent over time. Appl. Anim. Behav. Sci. 1993, 36, 1–9. [Google Scholar] [CrossRef]
- Carvalho, P.D.; Santos, V.G.; Giordano, J.O.; Wiltbank, M.C.; Fricke, P.M. Development of fertility programs to achieve high 21-day pregnancy rates in high-producing dairy cows. Theriogenology 2018, 114, 165–172. [Google Scholar] [CrossRef]
- Lüttgenau, J.; Bollwein, H. Evaluation of bovine luteal blood flow by using color Doppler ultrasonography. Reprod. Biol. 2014, 14, 103–109. [Google Scholar] [CrossRef]
- Ueshiba, H.; Zerah, M.; New, M.I. Enzyme-linked immunosorbent assay (ELISA) method for screening of non-classical steroid 21-hydroxylase deficiency. Horm. Metab. Res. 1994, 26, 43–45. [Google Scholar] [CrossRef] [PubMed]
- National Research Council; Board on Agriculture; Subcommittee on Beef Cattle Nutrition; Committee on Animal Nutrition. Nutrient Requirements of Beef Cattle, 7th revised ed.; The National Academies Press: Washington, DC, USA, 2000. [Google Scholar] [CrossRef]
- Chelikani, P.K.; Ambrose, J.D.; Kennelly, J.J. Effect of dietary energy and protein density on body composition, attainment of puberty, and ovarian follicular dynamics in dairy heifers. Theriogenology 2003, 60, 707–725. [Google Scholar] [CrossRef]
- Le Cozler, Y.; Lollivier, V.; Lacasse, P.; Disenhaus, C. Rearing strategy and optimizing first-calving targets in dairy heifers:A review. Animal 2008, 2, 1393–1404. [Google Scholar] [CrossRef]
- Clempson, A.M.; Pollott, G.E.; Brickell, J.S.; Wathes, D.C. Associations between bovine IGFBP2 polymorphisms with fertility, milk production, and metabolic status in UK dairy cows. Anim. Biotechnol. 2012, 23, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, M.J.; Romero-Ruiz, A.; Tena-Sempere, M. Roles of leptin in reproduction, pregnancy and polycystic ovary syndrome:Consensus knowledge and recent developments. Metabolism 2015, 64, 79–91. [Google Scholar] [CrossRef] [PubMed]
- De Haas, Y.; Janss, L.L.G.; Kadarmideen, H.N. Genetic correlations between body condition scores and fertility in dairy cattle using bivariate random regression models. J. Anim. Breed. Genet. 2007, 124, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, S.E.; Elmore, M.F.; Kriese-Anderson, L.; Elmore, J.B.; Walker, B.N.; Dyce, P.W.; Rodning, S.P.; Biase, F.H. Evaluation of age, weaning weight, body condition score, and reproductive tract score in pre-selected beef heifers relative to reproductive potential. J Anim. Sci. Biotechnol. 2019, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Cassady, J.M.; Maddock, T.D.; DiCostanzo, A.; Lamb, G.C. Initial body condition score affects hormone and metabolite response to nutritional restriction and repletion in yearling postpubertal beef heifers. J. Anim. Sci. 2009, 87, 2262–2273. [Google Scholar] [CrossRef]
- D’Occhio, M.J.; Baruselli, P.S.; Campanile, G. Influence of nutrition, body condition, and metabolic status on reproduction in female beef cattle:A review. Theriogenology 2019, 125, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Baruselli, P.S.; Ferreira, R.M.; Colli, M.H.A.; Elliff, F.M.; Sá_Filho, M.F.; Vieira, L.; Freitas, B.G. Timed artificial insemination:Current challenges and recent advances in reproductive efficiency in beef and dairy herds in Brazil. Anim. Reprod. 2017, 14, 558–571. [Google Scholar] [CrossRef]
- Curley, K.O.; Neuendorff, D.A.; Lewis, A.W.; Cleere, J.J.; Welsh, T.H.; Randel, R.D. Functional characteristics of the bovine hypothalamic-pituitary-adrenal axis vary with temperament. Horm. Behav. 2008, 53, 20–27. [Google Scholar] [CrossRef]
- Li, P.S.; Wagner, W.C. In vivo and in vitro studies on the effect of adrenocorticotropic hormone or cortisol on the pituitary response to gonadotropin releasing hormone. Biol. Reprod. 1983, 29, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Dobson, H.; Tebble, J.E.; Smith, R.F.; Ward, W.R. Is stress really all that important? Theriogenology 2001, 55, 65–73. [Google Scholar] [CrossRef]
- Kasimanickam, R.; Schroeder, S.; Assay, M.; Kasimanickam, V.; Moore, D.; Gay, J.; Whittier, W. Influence of temperament score and handling facility on stress, reproductive hormone concentrations, and fixed time AI pregnancy rates in beef heifers. Reprod. Dom. Anim. 2014, 49, 775–782. [Google Scholar] [CrossRef]
- Kasimanickam, R.; Asay, M.; Schroeder, S.; Kasimanickam, V.; Gay, J.; Kastelic, J.; Hall, J.; Whittier, W. Calm temperament improves reproductive performance of beef cows. Reprod. Dom. Anim. 2014, 49, 1063–1067. [Google Scholar] [CrossRef]
- Cooke, R.F.; Bill, E. Kunkle Interdisciplinary Beef Symposium: Temperament and acclimation to human handling influence growth, health, and reproductive responses in Bos taurus and Bos indicus cattle. J. Anim. Sci. 2014, 92, 5325–5333. [Google Scholar] [CrossRef]
- Cooke, R.F.; Bohnert, D.W.; Cappellozza, B.I.; Mueller, C.J.; Delcurto, T. Effects of temperament and acclimation to handling on reproductive performance of Bos taurus beef females. J. Anim. Sci. 2012, 90, 3547–3555. [Google Scholar] [CrossRef] [PubMed]
- Sangsritavong, S.; Combs, D.K.; Sartori, R.; Armentano, L.E.; Wiltbank, M.C. High feed intake increases liver blood flow and metabolism of progesterone and estradiol-17beta in dairy cattle. J. Anim. Sci. 2002, 85, 2831–2842. [Google Scholar] [CrossRef]
- Endo, N.; Nagai, K.; Tanaka, T.; Kamomae, H. Comparison between lactating and non-lactating dairy cows on follicular growth and corpus luteum development, and endocrine patterns of ovarian steroids and luteinizing hormone in the estrous cycles. Anim. Reprod. Sci. 2012, 134, 112–118. [Google Scholar] [CrossRef]
- Stevenson, J.S.; Pulley, S.L. Feedback effects of estradiol and progesterone on ovulation and fertility of dairy cows after gonadotropin-releasing hormone-induced release of luteinizing hormone. J. Dairy Sci. 2016, 99, 3003–3015. [Google Scholar] [CrossRef] [PubMed]
- Bridges, G.A.; Mussard, M.L.; Helser, L.A.; Day, M.L. Comparison of follicular dynamics and hormone concentrations between the 7-day and 5-day CO-Synch + CIDR program in primiparous beef cows. Theriogenology 2014, 81, 632–638. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).