Performance of Co-Housed Neon Tetras (Paracheirodon innesi) and Glowlight Rasboras (Trigonostigma hengeli) Fed Commercial Flakes and Lyophilized Natural Food
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Fish and Experimental Setup Preparation
2.2. Feeding Experiment
- Group T was only given the Tetra flakes;
- Group O was only given the Omega flakes;
- Group TO was alternately given the Tetra and Omega flakes;
- Group TOL was alternately given the Tetra and Omega flakes, but also the lyophilizate mix (only twice a week, instead of one of the flake meals).
2.3. Sampling and Basic Body Parameters
2.4. Histological Analysis
2.5. Whole-Mount Skeletal Analysis
2.6. Statistical Analysis
3. Results
3.1. Body Weight, Survival and Other Observations
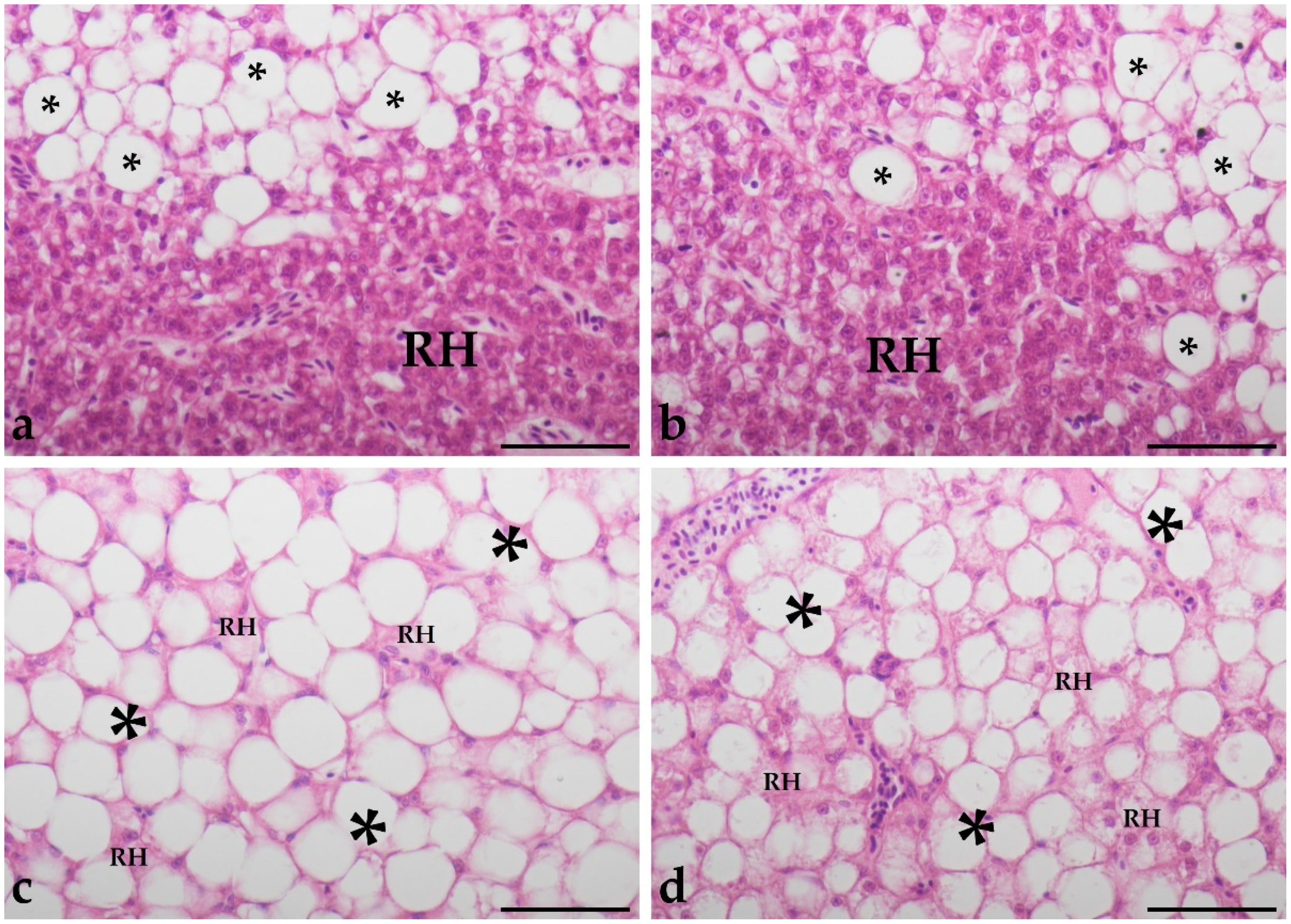
3.2. Histological Analysis
3.3. Whole-Mount Skeletal Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- King, T.A. Wild caught ornamental fish: A perspective from the UK ornamental aquatic industry on the sustainability of aquatic organisms and livelihoods. J. Fish Biol. 2019, 94, 925–936. [Google Scholar] [CrossRef]
- Evers, H.G.; Pinnegar, J.K.; Taylor, M.I. Where are they all from? - sources and sustainability in the ornamental freshwater fish trade. J. Fish Biol. 2019, 94, 909–916. [Google Scholar] [CrossRef] [PubMed]
- FAO. The State of World Fisheries and Aquaculture-Sustainability in Action; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Earle, K.E. The nutritional requirements of ornamental fish. Vet. Q. 1995, 17, 53–55. [Google Scholar] [CrossRef]
- Siddik, M.A.B.; Howieson, J.; Fotedar, R.; Partridge, G.J. Enzymatic fish protein hydrolysates in finfish aquaculture: A review. Rev. Aquac. 2021, 13, 406–430. [Google Scholar] [CrossRef]
- Galkanda-Arachchige, H.S.C.; Wilson, A.E.; Davis, D.A. Success of fishmeal replacement through poultry by-product meal in aquaculture feed formulations: A meta-analysis. Rev. Aquac. 2020, 12, 1624–1636. [Google Scholar] [CrossRef]
- Zhang, F.; Man, Y.B.; Mo, W.Y.; Wong, M.H. Application of Spirulina in aquaculture: A review on wastewater treatment and fish growth. Rev. Aquac. 2020, 12, 582–599. [Google Scholar] [CrossRef]
- Agboola, J.O.; Øverland, M.; Skrede, A.; Hansen, J.Ø. Yeast as major protein-rich ingredient in aquafeeds: A review of the implications for aquaculture production. Rev. Aquac. 2021, 13, 949–970. [Google Scholar] [CrossRef]
- Alhazzaa, R.; Nichols, P.D.; Carter, C.G. Sustainable alternatives to dietary fish oil in tropical fish aquaculture. Rev. Aquac. 2019, 11, 1195–1218. [Google Scholar] [CrossRef]
- Sprague, M.; Betancor, M.B.; Tocher, D.R. Microbial and genetically engineered oils as replacements for fish oil in aquaculture feeds. Biotechnol. Lett. 2017, 39, 1599–1609. [Google Scholar] [CrossRef]
- Hossain, M.S.; Koshio, S.; Kestemont, P. Recent advances of nucleotide nutrition research in aquaculture: A review. Rev. Aquac. 2020, 12, 1028–1053. [Google Scholar] [CrossRef]
- Ringø, E.; Olsen, R.E.; Gifstad, T.; Dalmo, R.A.; Amlund, H.; Hemre, G.I.; Bakke, A.M. Prebiotics in aquaculture: A review. Aquac. Nutr. 2010, 16, 117–136. [Google Scholar] [CrossRef]
- Pandiyan, P.; Balaraman, D.; Thirunavukkarasu, R.; George, E.G.J.; Subaramaniyan, K.; Manikkam, S.; Sadayappan, B. Probiotics in aquaculture. Drug Invent. Today 2013, 5, 55–59. [Google Scholar] [CrossRef]
- Kandathil Radhakrishnan, D.; AkbarAli, I.; Schmidt, B.V.; John, E.M.; Sivanpillai, S.; Thazhakot Vasunambesan, S. Improvement of nutritional quality of live feed for aquaculture: An overview. Aquac. Res. 2020, 51, 1–17. [Google Scholar] [CrossRef]
- Lim, L.C.; Cho, Y.L.; Dhert, P.; Wong, C.C.; Nelis, H.; Sorgeloos, P. Use of decapsulated Artemia cysts in ornamental fish culture. Aquac. Res. 2002, 33, 575–589. [Google Scholar] [CrossRef]
- Lim, L.C.; Dhert, P.; Sorgeloos, P. Recent developments in the application of live feeds in the freshwater ornamental fish culture. Aquaculture 2003, 227, 319–331. [Google Scholar] [CrossRef]
- Lipscomb, T.N.; Patterson, J.T.; Wood, A.L.; Watson, C.A.; DiMaggio, M.A. Larval growth, survival, and partial budget analysis related to replacing Artemia in larval culture of six freshwater ornamental fishes. J. World Aquac. Soc. 2020, 51, 1132–1144. [Google Scholar] [CrossRef]
- Best, J.; Adatto, I.; Cockington, J.; James, A.; Lawrence, C. A novel method for rearing first-feeding larval zebrafish: Polyculture with type L saltwater rotifers (Brachionus plicatilis). Zebrafish 2010, 7, 289–295. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Araújo, L.; Santos, M.M. Rearing zebrafish (Danio rerio) larvae without live food: Evaluation of a commercial, a practical and a purified starter diet on larval performance. Aquac. Res. 2006, 37, 1107–1111. [Google Scholar] [CrossRef]
- Hernandez, R.E.; Galitan, L.; Cameron, J.; Goodwin, N.; Ramakrishnan, L. Delay of Initial Feeding of Zebrafish Larvae until 8 Days Postfertilization Has No Impact on Survival or Growth Through the Juvenile Stage. Zebrafish 2018, 15, 515–518. [Google Scholar] [CrossRef]
- Clift, D.; Richendrfer, H.; Thorn, R.J.; Colwill, R.M.; Creton, R. High-throughput analysis of behavior in zebrafish larvae: Effects of feeding. Zebrafish 2014, 11, 455–461. [Google Scholar] [CrossRef]
- Lucore, E.C.; Connaughton, V.P. Observational learning and irreversible starvation in first-feeding zebrafish larvae: Is it okay to copy from your friends? Zoology 2021, 145, 125896. [Google Scholar] [CrossRef]
- Hedrera, M.I.; Galdames, J.A.; Jimenez-Reyes, M.F.; Reyes, A.E.; Avendaño-Herrera, R.; Romero, J.; Feijóo, C.G. Soybean Meal Induces Intestinal Inflammation in Zebrafish Larvae. PLoS ONE 2013, 8, 69983. [Google Scholar] [CrossRef]
- Hensley, M.R.; Leung, Y.F. A convenient dry feed for raising zebrafish larvae. Zebrafish 2010, 7, 219–231. [Google Scholar] [CrossRef]
- Sales, J.; Janssens, G.P.J. Nutrient requirements of ornamental fish. Aquat. Living Resour. 2003, 16, 533–540. [Google Scholar] [CrossRef]
- Boonyaratpalin, M.; Lovell, R.T. Diet preparation for aquarium fishes. Aquaculture 1977, 12, 53–62. [Google Scholar] [CrossRef]
- James, R.; Sampath, K. Effect of feed type on growth and fertility in ornamental fish, Xiphophorus Helleri. Isr. J. Aquac.-Bamidgeh 2004, 56, 264–273. [Google Scholar] [CrossRef]
- Harpaz, S.; Slosman, T.; Segev, R. Effect of feeding guppy fish fry (Poecilia reticulata) diets in the form of powder versus flakes. Aquac. Res. 2005, 36, 996–1000. [Google Scholar] [CrossRef]
- Pannevis, M.C.; Earle, K.E. Maintenance energy requirement of five popular species of ornamental fish. J. Nutr. 1994, 124, 2616S–2618S. [Google Scholar] [CrossRef] [PubMed]
- Sealey, W.M.; Barrows, F.T.; Casten, M.; Hardy, R.W. Dietary Protein Source and Level Affects Growth in Neon Tetras. N. Am. J. Aquac. 2009, 71, 320–324. [Google Scholar] [CrossRef]
- Luna-Figueroa, J.; Figueroa Torres, J.; Soriano Salazar, M. Efecto de diferentes niveles de proteína de la dieta sobre el crecimiento de juveniles del pez neón Paracheirodon innesi (pisces:characidae). Uniciencia 2001, 18, 15–20. [Google Scholar]
- Dernekbasi, S.; Unal, H.; Karayucel, I.; Aral, O. Effect of dietary supplementation of different rates of spirulina (spirulina platensis) on growth and feed conversion in guppy (Poecilia reticulata Peters, 1860). J. Anim. Vet. Adv. 2010, 9, 1395–1399. [Google Scholar] [CrossRef]
- James, R.; Sampath, K.; Thangarathinam, R.; Vasudevan, I. Effect of dietary Spirulina level on growth, fertility, coloration and leucocyte count in red swordtail, Xiphophorus helleri. Isr. J. Aquac.-Bamidgeh 2006, 58, 97–104. [Google Scholar]
- Nurhidayat, N.; Wardin, L.; Sitorus, E. The survival and growth performance of juvenile cardinal tetra (Paracheirodon axelrodi) with application of tropical almond (Terminalia catappa) leaves. Nusant. Biosci. 2016, 8, 1–4. [Google Scholar] [CrossRef]
- Uribe, E.A.; Archundia, M.P.F.; Luna-Figueroa, J. The Effect of Live Food on the Coloration and Growth in Guppy Fish, Poecilia reticulata. Agric. Sci. 2018, 9, 171–179. [Google Scholar] [CrossRef]
- James, R.; Sampath, K. Effect of feeding frequency on growth and fecundity in an ornamental fish, Betta splendens (Regan). Isr. J. Aquac.-Bamidgeh 2004, 56, 138–147. [Google Scholar]
- Puello-Cruz, A.C.; Velasco-Blanco, G.; Martínez-Rodríguez, I.E.; Felix-Ramos, E.; Voltolina, D. Growth and survival of siamese fighting fish, Betta splendens: Larvae at low salinity and with different diets. J. World Aquac. Soc. 2010, 41, 823–828. [Google Scholar] [CrossRef]
- Khatoon, H.; Leng, M.Y.; Rahman, M.R.; Sarker, J.; Yoke Leng, M.; Minhaz, T.M.; Sultana, R.; Nayma, Z.; Mukta, F.A. Efficiency of Chlorella vulgaris beads in improving water quality and growth of juvenile siamese fighting fish (Betta splendens). Bangladesh J. Vet. Anim. Sci. 2021, 9, 74–86. [Google Scholar]
- Mejia-Mejia, M.; Arce, E.; García-Rodríguez, J.; Burciaga, L.M. Effect of feeding mosquito larvae on the coloration of siamese fighting fish (Betta splendens) during grow-out. Int. Aquat. Res. 2021, 13, 71–79. [Google Scholar] [CrossRef]
- Zuanon, J.A.S.; Hisano, H.; Falcon, D.R.; Sampaio, F.G.; Barros, M.M.; Pezzato, L.E. Digestibility of protein feedstuffs and energetic feedstuffs for female beta. Rev. Bras. Zootec. 2007, 36, 987–991. [Google Scholar] [CrossRef]
- Thongprajukaew, K.; Kovitvadhi, U.; Kovitvadhi, S.; Somsueb, P.; Rungruangsak-Torrissen, K. Effects of different modified diets on growth, digestive enzyme activities and muscle compositions in juvenile Siamese fighting fish (Betta splendens Regan, 1910). Aquaculture 2011, 322–323, 1–9. [Google Scholar] [CrossRef]
- Thongprajukaew, K.; Kovitvadhi, U.; Kovitvadhi, S.; Engkagul, A.; Rungruangsak-Torrissen, K. Evaluation of growth performance and nutritional quality of diets using digestive enzyme markers and in vitro digestibility in Siamese fighting fish (Betta splendens Regan, 1910). Afr. J. Biotechnol. 2013, 12, 1689–1702. [Google Scholar] [CrossRef]
- Thongprajukaew, K.; Pettawee, S.; Muangthong, S.; Saekhow, S.; Phromkunthong, W. Freeze-dried forms of mosquito larvae for feeding of Siamese fighting fish (Betta splendens Regan, 1910). Aquac. Res. 2019, 50, 296–303. [Google Scholar] [CrossRef]
- Mandal, S.C.; Kohli, M.P.S.; Das, P.; Singh, S.K.; Munilkumar, S.; Sarma, K.; Baruah, K. Effect of substituting live feed with formulated feed on the reproductive performance and fry survival of Siamese fighting fish, Betta splendens (Regan, 1910). Fish Physiol. Biochem. 2012, 38, 573–584. [Google Scholar] [CrossRef]
- Dabrowski, K.; Miller, M. Contested paradigm in raising zebrafish (Danio rerio). Zebrafish 2018, 15, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Nowosad, J.; Kucharczyk, D.; Targońska, K. Enrichment of Zebrafish Danio rerio (Hamilton, 1822) Diet with Polyunsaturated Fatty Acids Improves Fecundity and Larvae Quality. Zebrafish 2017, 14, 364–370. [Google Scholar] [CrossRef]
- Martins, G.; Diogo, P.; Santos, T.; Cabrita, E.; Pinto, W.; Dias, J.; Gavaia, P.J. Microdiet Formulation with Phospholipid Modulate Zebrafish Skeletal Development and Reproduction. Zebrafish 2020, 17, 27–37. [Google Scholar] [CrossRef]
- Martins, G.; Diogo, P.; Pinto, W.; Gavaia, P.J. Early Transition to Microdiets Improves Growth, Reproductive Performance and Reduces Skeletal Anomalies in Zebrafish (Danio rerio). Zebrafish 2019, 16, 300–307. [Google Scholar] [CrossRef]
- Frederickson, S.C.; Steinmiller, M.D.; Blaylock, T.R.; Wisnieski, M.E.; Malley, J.D.; Pandolfo, L.M.; Castranova, D. Comparison of Juvenile Feed Protocols on Growth and Spawning in Zebrafish. J. Am. Assoc. Lab. Anim. Sci. 2021, 60, 298–305. [Google Scholar] [CrossRef]
- O’Brine, T.M.; Vrtělová, J.; Snellgrove, D.L.; Davies, S.J.; Sloman, K.A. Growth, oxygen consumption, and behavioral responses of Danio rerio to variation in dietary protein and lipid levels. Zebrafish 2015, 12, 296–304. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Randazzo, B.; Gioacchini, G.; Truzzi, C.; Giorgini, E.; Riolo, P.; Gioia, G.; Bertolucci, C.; Osimani, A.; Cardinaletti, G.; et al. Zebrafish (Danio rerio) physiological and behavioural responses to insect-based diets: A multidisciplinary approach. Sci. Rep. 2020, 10, 10648. [Google Scholar] [CrossRef]
- Kaur, V.I.; Mohanta, K.N. Feeds and feeding management for freshwater ornamental fish production. In Best Management Practices for Freshwater Ornamental Fish Production; Jain, A.K., Saini, V.P., Kaur, V.I., Eds.; National Fisheries Development Board (Ministry of Agriculture and Farmer’s Welfare, GOI): Hyderabad, India, 2016; pp. 76–92. [Google Scholar]
- Nelson, J.S.; Grande, T.C.; Wilson, M.V.H. Fishes of the World: Fifth Edition; Wiley Blackwell: Hoboken, NJ, USA, 2016; ISBN 9781119174844. [Google Scholar]
- Jandzik, D.; Stock, D.W. Differences in developmental potential predict the contrasting patterns of dental diversification in characiform and cypriniform fishes. Proc. R. Soc. B Biol. Sci. 2021, 288, 20202205. [Google Scholar] [CrossRef] [PubMed]
- Stock, D.W.; Jackman, W.R.; Trapani, J. Developmental genetic mechanisms of evolutionary tooth loss in cypriniform fishes. Development 2006, 133, 3127–3137. [Google Scholar] [CrossRef]
- Wilson, J.M.; Castro, L.F.C. Morphological diversity of the gastrointestinal tract in fishes. Fish Physiol. 2010, 30, 1–55. [Google Scholar] [CrossRef]
- Dey, V.K. The Global Trade in Ornamental Fish. Infofish Int. 2016, 4, 52–55. [Google Scholar]
- Kucharczyk, D.; Targońska, K.; Zarski, D.; Krejszeff, S.; Kupren, K.; Łuczyński, M.J.; Szczerbowski, A. The reproduction of neon tetra, Paracheirodon innesi (Myers, 1936), under controlled conditions. Pol. J. Nat. Sci. 2010, 25, 81–92. [Google Scholar] [CrossRef]
- Chapman, F.A.; Colle, D.E.; Rottmann, R.W.; Shireman, J.V. Controlled Spawning of the Neon Tetra. Progress. Fish-Cult. 1998, 60, 32–37. [Google Scholar] [CrossRef]
- Pathirana, P.R.A.; Jayamanne, S.C. Development of a Low Cost Method for Breeding Paracheirodon innesi (Neon Tetra) in Captivity. In Proceedings of the Empowering Regional Development through Science and Technology, First Annual Science Research Session (2012); Faculty of Applied Science, South Eastern University of Sri Lanka: Oluvil, Sri Lanka, 2012; p. 21. [Google Scholar]
- Budiardi, T.; Solehudin, M.A.; Wahjuningrum, D. Production of M-Size Neon Tetra Fish Paracheriodon innesi in Recirculation System with Density of 25, 50, 75 and 100 litre-1. J. Akuakultur Indones. 2008, 7, 19. [Google Scholar] [CrossRef][Green Version]
- Sanaye, S.V.; Singh, H.; Tibile, R.M. Growth and survival of neon tetra, Paracheirodon innesi (Myers, 1936) fry fed mixed zooplankton, formulated feed and combination thereof. Ann. Biol. Res. 2012, 3, 5665–5668. [Google Scholar]
- Sanaye, S.V.; Dhaker, H.S.; Tibile, R.M.; Mhatre, V.D. Effect of Green Water and Mixed Zooplankton on Growth and Survival in Neon Tetra, Paracheirodon innesi (Myers, 1936) during Larval and Early Fry Rearing. Int. J. Biol. Biomol. Agric. Food Biotechnol. Eng. 2014, 8, 159–163. [Google Scholar]
- Darve, S.I.; Singh, H. Dietary influence of amino acids on growth of neon tetra Paracheirodon innesi. Anim. Nutr. Feed Technol. 2014, 14, 385–391. [Google Scholar] [CrossRef]
- Darve, S.I.; Wani, G.B.; Singh, H. Dietary Lipid Requirements for Neon tetra (Paracheirodon innesi): A Valuable Ornamental fish. SKUAST J. Res. 2016, 18, 146–153. [Google Scholar]
- Davis, E.; Arts, K.; Raja, S.; Arts, K. Comparative analysis of animal based feed preferences in selected Aquarium fishes. Int. J. Fish. Aquat. Stud. 2019, 7, 42–45. [Google Scholar]
- Ardi, I.; Zamroni, M.; Setiadi, E. Utility of almond leaf (Terminalia cattapa) for improving growth and color quality of neon tetra fish (Paracheirodon innesi). In Proceedings of the IOP Conference Series: Earth and Environmental Science, Beijing, China, 23–26 October 2020; IOP Publishing: Bristol, UK, 2020; Volume 441, p. 012136. [Google Scholar]
- Tan, H.H.; Kottelat, M. The fishes of the Batang Hari drainage, Sumatra, with description of six new species. Ichthyol. Explor. Freshw. 2009, 20, 13–69. [Google Scholar]
- Teletchea, F. Domestication level of the most popular aquarium fish species: Is the aquarium trade dependent on wild populations? Cybium 2016, 40, 21–29. [Google Scholar]
- Rašković, B.; Stanković, M.; Marković, Z.; Poleksić, V. Histological methods in the assessment of different feed effects on liver and intestine of fish. J. Agric. Sci. Belgrade 2011, 56, 87–100. [Google Scholar] [CrossRef]
- Ostaszewska, T.; Dabrowski, K.; Palacios, M.E.; Olejniczak, M.; Wieczorek, M. Growth and morphological changes in the digestive tract of rainbow trout (Oncorhynchus mykiss) and pacu (Piaractus mesopotamicus) due to casein replacement with soybean proteins. Aquaculture 2005, 245, 273–286. [Google Scholar] [CrossRef]
- Glencross, B.D.; Booth, M.; Allan, G.L. A feed is only as good as its ingredients – A review of ingredient evaluation strategies for aquaculture feeds. Aquac. Nutr. 2007, 13, 17–34. [Google Scholar] [CrossRef]
- Boglione, C.; Gavaia, P.J.; Koumoundouros, G.; Gisbert, E.; Moren, M.; Fontagné, S.; Witten, P.E. Skeletal anomalies in reared European fish larvae and juveniles. Part 1: Normal and anomalous skeletogenic processes. Rev. Aquac. 2013, 5, 99–120. [Google Scholar] [CrossRef]
- Boglione, C.; Gisbert, E.; Gavaia, P.J.; Witten, P.E.; Moren, M.; Fontagné, S.; Koumoundouros, G. Skeletal anomalies in reared European fish larvae and juveniles. Part 2: Main typologies, occurrences and causative factors. Rev. Aquac. 2013, 5, 121–167. [Google Scholar] [CrossRef]
- Shim, K.F.; Ho, C.S. Calcium and Phosphorus Requirements of Guppy Poecilia reticulata. Bull. Jpn. Soc. Sci. Fish. 1989, 55, 1947–1953. [Google Scholar] [CrossRef]
- Fracalossi, D.M.; Allen, M.E.; Nichols, D.K.; Oftedal, O.T. Oscars, Astronotus Ocellatus, have a dietary requirement for vitamin C. J. Nutr. 1998, 128, 1745–1751. [Google Scholar] [CrossRef]
- Martini, A.; Huysseune, A.; Witten, P.E.; Boglione, C. Plasticity of the skeleton and skeletal deformities in zebrafish (Danio rerio) linked to rearing density. J. Fish Biol. 2021, 98, 971–986. [Google Scholar] [CrossRef]
- Lawrence, C.; Best, J.; James, A.; Maloney, K. The effects of feeding frequency on growth and reproduction in zebrafish (Danio rerio). Aquaculture 2012, 368–369, 103–108. [Google Scholar] [CrossRef]
- Latimer, G.W.; Horwitz, W. (Eds.) Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Rockville, MD, USA, 2007. [Google Scholar]
- Rašković, B.; Cruzeiro, C.; Poleksić, V.; Rocha, E. Estimating volumes from common carp hepatocytes using design-based stereology and examining correlations with profile areas: Revisiting a nutritional assay and unveiling guidelines to microscopists. Microsc. Res. Tech. 2019, 82, 861–871. [Google Scholar] [CrossRef]
- Baeverfjord, G.; Krogdahl, Å. Development and regression of soybean meal induced enteritis in Atlantic salmon, Salmo salar L., distal intestine: A comparison with the intestines of fasted fish. J. Fish Dis. 1996, 19, 375–387. [Google Scholar] [CrossRef]
- Kasprzak, R.; Ostaszewska, T.; Kamaszewski, M. Effects of feeding commercial diets on the development of juvenile crucian carp Carassius carassius: Digestive tract abnormalities. Aquat. Biol. 2019, 28, 159–173. [Google Scholar] [CrossRef]
- Fernández, I.; Toledo-Solís, F.J.; Tomás-Almenar, C.; Larrán, A.M.; Cárdaba, P.; Laguna, L.M.; Galán, M.S.; Mateo, J.A. Skeletal Development and Deformities in Tench (Tinca tinca): From Basic knowledge to Regular Monitoring Procedure. Animals 2021, 11, 621. [Google Scholar] [CrossRef] [PubMed]
- Fernández, I.; Hontoria, F.; Ortiz-Delgado, J.B.; Kotzamanis, Y.; Estévez, A.; Zambonino-Infante, J.L.; Gisbert, E. Larval performance and skeletal deformities in farmed gilthead sea bream (Sparus aurata) fed with graded levels of Vitamin A enriched rotifers (Brachionus plicatilis). Aquaculture 2008, 283, 102–115. [Google Scholar] [CrossRef]
- Saxby, A.; Adams, L.; Snellgrove, D.; Wilson, R.W.; Sloman, K.A. The effect of group size on the behaviour and welfare of four fish species commonly kept in home aquaria. Appl. Anim. Behav. Sci. 2010, 125, 195–205. [Google Scholar] [CrossRef]
- Andersson, M.; Kettunen, P. Effects of Holding Density on the Welfare of Zebrafish: A Systematic Review. Zebrafish 2021, 18, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Brahman, L.K.; Chandra, R. Effect of environmental attributes and mosquito larvae on growth parameter of larvicidal fish, Rasbora daniconius. Int. J. Mosq. Res. 2016, 3, 53–57. [Google Scholar]
- Putri, M.W.D.; Prayogo; Budi, D.S. Effect of Spirulina platensis supplementation in the diet to sperm performance of silver rasbora (Rasbora argyrotaenia). IOP Conf. Ser. Earth Environ. Sci. 2020, 441, 2–6. [Google Scholar] [CrossRef]
- Tan, M.; Armbruster, J.W. Phylogenetic classification of extant genera of fishes of the order Cypriniformes (Teleostei: Ostariophysi). Zootaxa 2018, 4476, 6–39. [Google Scholar] [CrossRef]
- Kasprzak, R.; Ostaszewska, T.; Wagner, B. The effect of feeding commercial diets on the development of juvenile crucian carp (Carassius carassius, L.). Part 1: Skeletal deformations. Aquac. Nutr. 2019, 25, 78–87. [Google Scholar] [CrossRef]
- Caballero, M.J.; López-Calero, G.; Socorro, J.; Roo, F.J.; Izquierdo, M.S.; Férnandez, A.J. Combined effect of lipid level and fish meal quality on liver histology of gilthead seabream (Sparus aurata). Aquaculture 1999, 179, 277–290. [Google Scholar] [CrossRef]
- Rašković, B.; Čičovački, S.; Ćirić, M.; Marković, Z.; Poleksić, V. Integrative approach of histopathology and histomorphometry of common carp (Cyprinus carpio L.) organs as a marker of general fish health state in pond culture. Aquac. Res. 2016, 47, 3455–3463. [Google Scholar] [CrossRef]
- Castro, C.; Couto, A.; Pérez-Jiménez, A.; Serra, C.R.; Díaz-Rosales, P.; Fernandes, R.; Corraze, G.; Panserat, S.; Oliva-Teles, A. Effects of fish oil replacement by vegetable oil blend on digestive enzymes and tissue histomorphology of European sea bass (Dicentrarchus labrax) juveniles. Fish Physiol. Biochem. 2016, 42, 203–217. [Google Scholar] [CrossRef]
- Rašković, B.; Ćirić, M.; Koko, V.; Stanković, M.; Živić, I.; Marković, Z.; Poleksić, V. Effect of supplemental feeds on liver and intestine of common carp (Cyprinus carpio) in semi-intensive rearing system: Histological implications. Biologia 2016, 71, 212–219. [Google Scholar] [CrossRef]
- Spisni, E.; Tugnoli, M.; Ponticelli, A.; Mordenti, T.; Tomasi, V. Hepatic steatosis in artificially fed marine teleosts. J. Fish Dis. 1998, 21, 177–184. [Google Scholar] [CrossRef]
- Król, J.; Hliwa, P.; Woźniak, M.; Stabińska-Ułas, A.; Krejszeff, S.; Stabiński, R.; Czesny, S.J. Effect of various commercial diets on rearing performance, body chemical and fatty acid composition, liver histology and vertebral column anomalies in Eurasian perch (Perca fluviatilis L.) post-larvae. Aquac. Rep. 2021, 20, 100690. [Google Scholar] [CrossRef]
- Hardy, R.W. The Nutritional Pathology of Teleosts. In Fish Pathology; Roberts, R.J., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 402–424. [Google Scholar]
- Ostaszewska, T.; Dabrowski, K.; Hliwa, P.; Gomółka, P.; Kwasek, K. Nutritional regulation of intestine morphology in larval cyprinid fish, silver bream (Vimba vimba). Aquac. Res. 2008, 39, 1268–1278. [Google Scholar] [CrossRef]
- Ostaszewska, T.; Dabrowski, K.; Czumińska, K.; Olech, W.; Olejniczak, M. Rearing of pike-perch larvae using formulated diets-First success with starter feeds. Aquac. Res. 2005, 36, 1167–1176. [Google Scholar] [CrossRef]
- Benedito-Palos, L.; Navarro, J.C.; Sitjà-Bobadilla, A.; Gordon Bell, J.; Kaushik, S.; Pérez-Sánchez, J. High levels of vegetable oils in plant protein-rich diets fed to gilthead sea bream (Sparus aurata L.): Growth performance, muscle fatty acid profiles and histological alterations of target tissues. Br. J. Nutr. 2008, 100, 992–1003. [Google Scholar] [CrossRef]
- Lu, K.; Xu, W.; Li, J.; Li, X.; Huang, G.; Liu, W. Alterations of liver histology and blood biochemistry in blunt snout bream Megalobrama amblycephala fed high-fat diets. Fish. Sci. 2013, 79, 661–671. [Google Scholar] [CrossRef]
- de Borba, M.R.; Fracalossi, D.M.; Pezzato, L.E.; Menoyo, D.; Bautista, J.M. Growth, lipogenesis and body composition of piracanjuba (Brycon orbignyanus) fingerlings fed different dietary protein and lipid concentrations. Aquat. Living Resour. 2003, 16, 362–369. [Google Scholar] [CrossRef]
- Rios, F.S.; Donatti, L.; Fernandes, M.N.; Kalinin, A.L.; Rantin, F.T. Liver histopathology and accumulation of melano-macrophage centres in Hoplias malabaricus after long-term food deprivation and re-feeding. J. Fish Biol. 2007, 71, 1393–1406. [Google Scholar] [CrossRef]
- Park, I.-S. Effect of Starvation on the Weight and Structure in Some Tissues of Cyprinid Loach, Misgurnus anguillicaudatus. J. Fish. Mar. Sci. Educ. 2018, 30, 1170–1181. [Google Scholar] [CrossRef]
- Božić, G.; Rašković, B.; Stanković, M.; Poleksić, V.; Marković, Z. Effects of different feeds on growth performance parameters, histology of liver, distal intestine, and erythrocytes morphology of common carp (Cyprinus carpio L.). Biologia 2021, 114. [Google Scholar] [CrossRef]
- Ostaszewska, T.; Korwin-Kossakowski, M.; Wolnicki, J. Morphological changes of digestive structures in starved tench Tinca tinca (L.) juveniles. Aquac. Int. 2006, 14, 113–126. [Google Scholar] [CrossRef]
- Segner, H.; Rösch, R.; Schmidt, H.; von Poeppinghausen, K.J. Studies on the suitability of commercial dry diets for rearing of larval Coregonus lavaretus from Lake Constance. Aquat. Living Resour. 1988, 1, 231–238. [Google Scholar] [CrossRef]
- Strüssmann, C.A.; Takashima, F. Hepatocyte nuclear size and nutritional condition of larval pejerrey, Odontesthes bonariensis (Cuvier et Valenciennes). J. Fish Biol. 1990, 36, 59–65. [Google Scholar] [CrossRef]
- Wold, P.A.; Hoehne-Reitan, K.; Cahu, C.L.; Infante, J.Z.; Rainuzzo, J.; Kjørsvik, E. Comparison of dietary phospholipids and neutral lipids: Effects on gut, liver and pancreas histology in Atlantic cod (Gadus morha L.) larvae. Aquac. Nutr. 2009, 15, 73–84. [Google Scholar] [CrossRef]
- Bakke-McKellep, A.M.; Penn, M.H.; Salas, P.M.; Refstie, S.; Sperstad, S.; Landsverk, T.; Ringø, E.; Krogdahl, Å. Effects of dietary soyabean meal, inulin and oxytetracycline on intestinal microbiota and epithelial cell stress, apoptosis and proliferation in the teleost Atlantic salmon (Salmo salar L.). Br. J. Nutr. 2007, 97, 699–713. [Google Scholar] [CrossRef]
- Lall, S.P.; Lewis-McCrea, L.M. Role of nutrients in skeletal metabolism and pathology in fish — An overview. Aquaculture 2007, 267, 3–19. [Google Scholar] [CrossRef]
- Lall, S.P. The Minerals. In Fish Nutrition; Hardy, R.W., Ed.; Academic Press Inc.: Cambridge, MA, USA, 2002; pp. 259–308. [Google Scholar]
- Witten, P.E.; Owen, M.A.G.; Fontanillas, R.; Soenens, M.; McGurk, C.; Obach, A. A primary phosphorus-deficient skeletal phenotype in juvenile Atlantic salmon Salmo salar: The uncoupling of bone formation and mineralization. J. Fish Biol. 2016, 88, 690–708. [Google Scholar] [CrossRef]
- Kacem, A.; Meunier, F.J. Halastatic demineralization in the vertebrae of Atlantic salmon, during their spawning migration. J. Fish Biol. 2003, 63, 1122–1130. [Google Scholar] [CrossRef]
- Witten, P.E.; Gil-Martens, L.; Huysseune, A.; Takle, H.; Hjelde, K. Towards a classification and an understanding of developmental relationships of vertebral body malformations in Atlantic salmon (Salmo salar L.). Aquaculture 2009, 295, 6–14. [Google Scholar] [CrossRef]
- Kamiński, R.; Sikorska, J.; Polak-Juszczak, L.; Wolnicki, J. Effects of temperature on body chemical composition and incidence of deformities in juvenile tench, Tinca tinca (Actinopterygii: Cypriniformes: Cyprinidae), fed a commercial dry diet and/or natural food. Acta Ichthyol. Piscat. 2017, 47, 63–71. [Google Scholar] [CrossRef]
- Kamiński, R.; Sikorska, J.; Wolnicki, J. Diet and water temperature affect growth and body deformities in juvenile tench Tinca tinca (L.) reared under controlled conditions. Aquac. Res. 2017, 48, 1327–1337. [Google Scholar] [CrossRef]
- García, V.; Celada, J.D.; González, R.; Carral, J.M.; Sáez-Royuela, M.; González, Á. Response of juvenile tench (Tinca tinca L.) fed practical diets with different protein contents and substitution levels of fish meal by soybean meal. Aquac. Res. 2015, 46, 28–38. [Google Scholar] [CrossRef]
- Myszkowski, L.; Kamiński, R.; Quiros, M.; Stanny, L.A.; Wolnicki, J. Dry diet-influenced growth, size variability, condition and body deformities in juvenile crucian carp Carassius carassius L. reared under controlled conditions. Arch. Pol. Fish. 2012, 20, 157–163. [Google Scholar] [CrossRef]
- Myszkowski, L.; Kamler, E.; Kwiatkowski, S. Weak compensatory growth makes short-term starvation an unsuitable technique to mitigate body deformities of Tinca tinca juveniles in intensive culture. Rev. Fish Biol. Fish. 2010, 20, 381–388. [Google Scholar] [CrossRef]
- Kamler, E.; Wolnicki, J.; Kamiński, R.; Sikorska, J. Fatty acid composition, growth and morphological deformities in juvenile cyprinid, Scardinius erythrophthalmus fed formulated diet supplemented with natural food. Aquaculture 2008, 278, 69–76. [Google Scholar] [CrossRef]
- Kamler, E.; Myszkowski, L.; Kamiński, R.; Korwin-Kossakowski, M.; Wolnicki, J. Does overfeeding affect tench Tinca tinca (L.) juveniles? Aquac. Int. 2006, 14, 99–111. [Google Scholar] [CrossRef]
- Wolnicki, J.; Myszkowski, L.; Korwin-Kossakowski, M.; Kamiński, R.; Stanny, L.A. Effects of different diets on juvenile tench, Tinca tinca (L.) reared under controlled conditions. Aquac. Int. 2006, 14, 89–98. [Google Scholar] [CrossRef]
- Kamiński, R.; Korwin-Kossakowski, M.; Kusznierz, J.; Myszkowski, L.; Stanny, L.A.; Wolnicki, J. Response of a juvenile cyprinid, lake minnow Eupallasella perenurus (Pallas), to different diets. Aquac. Int. 2005, 13, 479–486. [Google Scholar] [CrossRef]
- Meyers, J.R. Zebrafish: Development of a Vertebrate Model Organism. Curr. Protoc. Essent. Lab. Tech. 2018, 16, e19. [Google Scholar] [CrossRef]
- Kaushik, S.; Georga, I.; Koumoundouros, G. Growth and body composition of zebrafish (Danio rerio) larvae fed a compound feed from first feeding onward: Toward implications on nutrient requirements. Zebrafish 2011, 8, 87–95. [Google Scholar] [CrossRef]
- Lawrence, C.; James, A.; Mobley, S. Successful Replacement of Artemia salina nauplii with Marine Rotifers (Brachionus plicatilis) in the Diet of Preadult Zebrafish (Danio rerio). Zebrafish 2015, 12, 366–371. [Google Scholar] [CrossRef]
- Farias, M.; Certal, A.C. Different Feeds and Feeding Regimens have an Impact on Zebrafish Larval Rearing and Breeding Performance. Int. J. Mar. Biol. Res. 2016, 1, 1–8. [Google Scholar] [CrossRef]
- Monteiro, J.F.; Martins, S.; Farias, M.; Costa, T.; Certal, A.C. The impact of two different cold-extruded feeds and feeding regimens on zebrafish survival, growth and reproductive performance. J. Dev. Biol. 2018, 6, 15. [Google Scholar] [CrossRef]
- Gonzales, J.M.; Law, S.H.W. Feed and feeding regime affect growth rate and gonadosomatic index of adult zebrafish (Danio rerio). Zebrafish 2013, 10, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Fowler, L.A.; Williams, M.B.; Dennis-Cornelius, L.N.; Farmer, S.; Barry, R.J.; Powell, M.L.; Watts, S.A. Influence of Commercial and Laboratory Diets on Growth, Body Composition, and Reproduction in the Zebrafish Danio rerio. Zebrafish 2019, 16, 508–521. [Google Scholar] [CrossRef] [PubMed]
- Siccardi, A.J.; Garris, H.W.; Jones, W.T.; Moseley, D.B.; D’Abramo, L.R.; Watts, S.A. Growth and survival of Zebrafish (Danio rerio) fed different commercial and laboratory diets. Zebrafish 2009, 6, 275–280. [Google Scholar] [CrossRef] [PubMed]





| TetraMin Flakes | Omega One Freshwater Flakes | Lyophilizate Mix | |
|---|---|---|---|
| Crude protein * (%) | 47.8 | 43.3 | 36.4 |
| Crude fat * (%) | 10.4 | 11.6 | 7.7 |
| Crude fiber * (%) | 2.7 | 0.9 | 5.4 |
| Ash * (%) | 6.3 | 8.0 | 4.9 |
| Moisture (%) | 5.7 | 6.6 | 5.2 |
| Main ingredients | Fish and fish derivatives, Cereals, Yeasts, Vegetable protein extracts, Mollusks and crustaceans, Oils and fats, Various sugars (Oligofructose 1%), Algae, Minerals | Salmon (20%), Halibut (16%), Pollock (14%), Herring (13%), Shrimps (11%), Krill (10%), Wheat Flour (9%), Wheat Gluten (4%), Kelp (2%) | Lyophilized: krill (20%), tubifex (20%), brine shrimp (20%), bloodworms (20%), cyclops (20%) |
| Additives | Vit. A (37,680 IU/kg), Vit. D3 (1990 IU/kg) | Vit. A (15,400 IU/kg), Vit. C (0.88 g/kg), Vit. D3 (2200 IU/kg), Vit. E (750 IU/kg), Vit. B2 (22 IU/kg), Vit. B3 (0.11 g/kg), Vit. B5 (0.015 g/kg), Vit. B7 (0.055 g/kg), Vit. B9 (0.08 g/kg), Vit. B12 (0.055 IU/kg), Inositol (0.11 g/kg), Astaxanthin (0.6 g/kg), Lecithin (0.5 g/kg), Tocopherol (0.1 g/kg), Ethoxyquin (1 g/kg) | N/A |
| Monday | Tuesday | Wednesday | Thursday | Friday | Saturday | Sunday | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Meal Time | 9:00 | 16:00 | 9:00 | 16:00 | 9:00 | 16:00 | 9:00 | 16:00 | 9:00 | 16:00 | 9:00 | 16:00 | 9:00 | 16:00 |
| Group T | T | T | T | T | T | T | T | T | T | T | T | T | No meals | |
| Group O | O | O | O | O | O | O | O | O | O | O | O | O | ||
| Group TO | T | O | T | O | T | O | O | T | O | T | O | T | ||
| Group TOL | T | O | T | O | T | L | O | T | O | T | O | L | ||
| T | O | TO | TOL | ||
|---|---|---|---|---|---|
| Neon tetra | IBW (mg) | 275 ± 44 | 269 ± 48 | 272 ± 50 | 274 ± 58 |
| FBW (mg) | 384 * ± 55 | 344 ± 72 | 371 ± 118 | 343 ± 47 | |
| SGR (% day−1) | 0.398 | 0.289 | 0.371 | 0.269 | |
| Survival | 12/16 (75%) | 9/16 (56%) | 11/16 (69%) | 13/16 (81%) | |
| Glowlight rasbora | IBW (mg) | 225 ± 64 | 229 ± 61 | 226 ± 56 | 228 ± 89 |
| FBW (mg) | 352 * ± 86 | 348 * ± 125 | 359 * ± 81 | 380 * ± 159 | |
| SGR (% day−1) | 0.534 | 0.497 | 0.548 | 0.609 | |
| Survival | 16/16 (100%) | 15/16 (94%) | 16/16 (100%) | 12/16 (75%) |
| “Initial” | T | O | TO | TOL | ||
|---|---|---|---|---|---|---|
| Neon tetra | NA (µm2) | 20.76 C ± 2.81 | 21.23 C ± 3.27 | 22.08 B ± 4.22 | 20.91 C ± 2.96 | 23.01 A ± 5.12 |
| CA (µm2) | 79.74 A ± 28.21 | 55.87 C ± 23.97 | 62.32 B ± 58.42 | 47.66 D ± 17.39 | 45.61 D ± 20.15 | |
| NCI (%) | 28.78 D ± 9.25 | 45.32 C ± 20.50 | 44.94 C ± 18.52 | 49.15 B ± 17.59 | 59.01 A ± 25.02 | |
| Glowlight rasbora | NA (µm2) | 17.23 BC ± 2.51 | 17.18 BC ± 3.22 | 17.46 B ± 2.81 | 16.88 C ± 2.73 | 18.13 A ± 3.59 |
| CA (µm2) | 164.98 B ± 34.77 | 139.52 C ± 65.13 | 113.35 D ± 31.34 | 98.95 E ± 34.41 | 204.39 A ± 147.00 | |
| NCI (%) | 10.89 E ± 2.73 | 14.03 C ± 4.80 | 16.41 B ± 4.68 | 18.66 A ± 5.72 | 12.38 D ± 6.15 |
| “Initial” | T | O | TO | TOL | |
|---|---|---|---|---|---|
| Neon tetra FL | 157.76 C ± 56.04 | 259.03 A ± 80.83 | 231.00 B ± 94.17 | 171.77 C ± 71.18 | 215.55 B ± 91.66 |
| Glowlight rasbora FL | 158.91 B ± 69.26 | 157.55 B ± 77.40 | 160.77 B ± 68.53 | 179.18 A ± 63.32 | 159.01 B ± 53.63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kasprzak, R.; Grzeszkiewicz, A.B.; Górecka, A. Performance of Co-Housed Neon Tetras (Paracheirodon innesi) and Glowlight Rasboras (Trigonostigma hengeli) Fed Commercial Flakes and Lyophilized Natural Food. Animals 2021, 11, 3520. https://doi.org/10.3390/ani11123520
Kasprzak R, Grzeszkiewicz AB, Górecka A. Performance of Co-Housed Neon Tetras (Paracheirodon innesi) and Glowlight Rasboras (Trigonostigma hengeli) Fed Commercial Flakes and Lyophilized Natural Food. Animals. 2021; 11(12):3520. https://doi.org/10.3390/ani11123520
Chicago/Turabian StyleKasprzak, Robert, Anna Beata Grzeszkiewicz, and Aleksandra Górecka. 2021. "Performance of Co-Housed Neon Tetras (Paracheirodon innesi) and Glowlight Rasboras (Trigonostigma hengeli) Fed Commercial Flakes and Lyophilized Natural Food" Animals 11, no. 12: 3520. https://doi.org/10.3390/ani11123520
APA StyleKasprzak, R., Grzeszkiewicz, A. B., & Górecka, A. (2021). Performance of Co-Housed Neon Tetras (Paracheirodon innesi) and Glowlight Rasboras (Trigonostigma hengeli) Fed Commercial Flakes and Lyophilized Natural Food. Animals, 11(12), 3520. https://doi.org/10.3390/ani11123520






