Molecular Survey of Babesia spp. and Anaplasma phagocytophilum in Roe Deer from a Wildlife Rescue Center in Italy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Collection
2.2. Molecular Analyses
2.3. Phylogenetic and Statistical Analyses
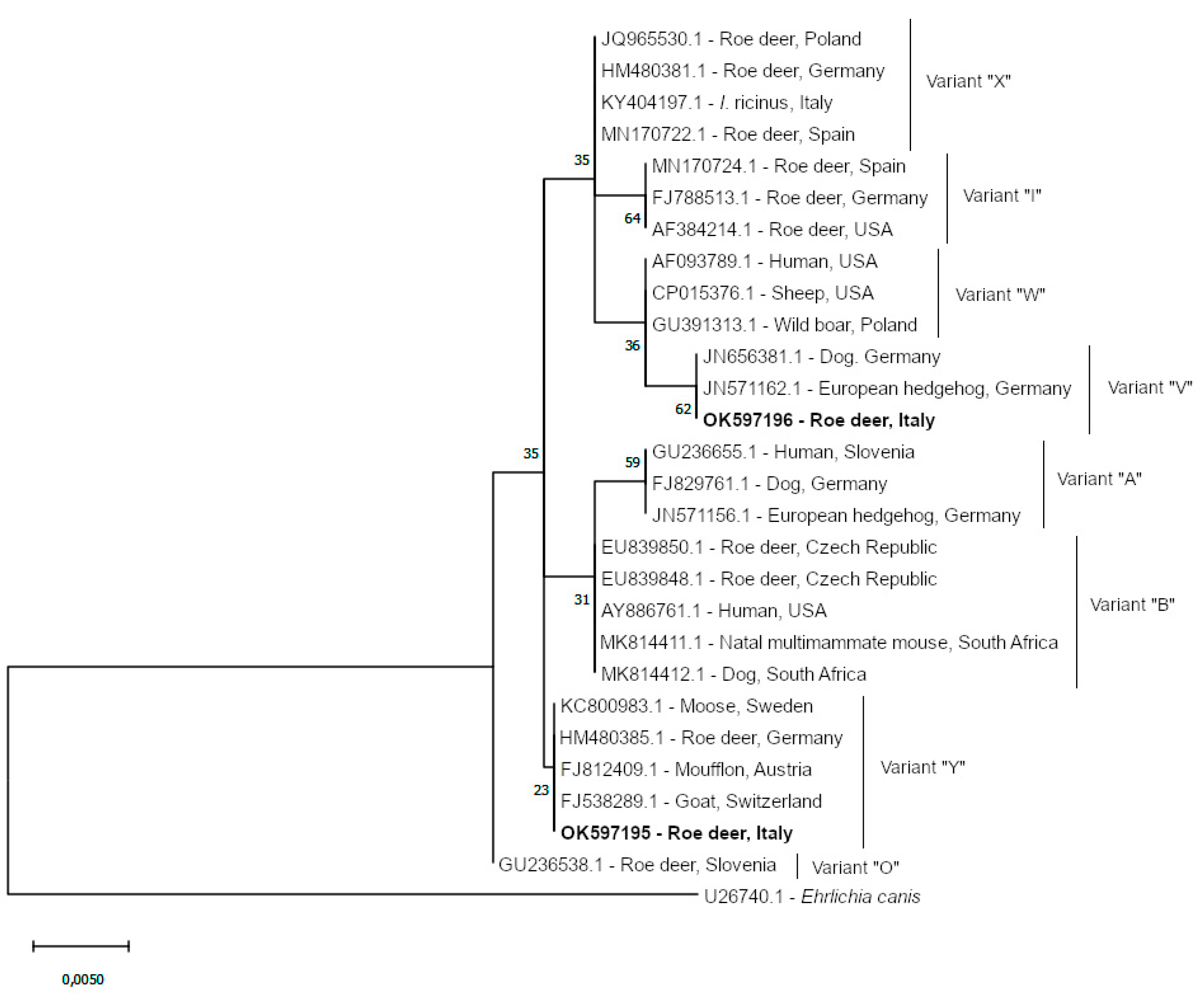
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vu Hai, V.; Almeras, L.; Socolovschi, C.; Raoult, D.; Parola, P.; Pagès, F. Monitoring human tick-borne disease risk and tick bite exposure in Europe: Available tools and promising future methods. Ticks Tick-Borne Dis. 2014, 5, 607–619. [Google Scholar] [CrossRef]
- Medlock, J.M.; Hansford, K.M.; Bormane, A.; Derdakova, M.; Estrada-Peña, A.; George, J.-C.; Golovljova, I.; Jaenson, T.G.T.; Jensen, J.-K.; Jensen, P.M.; et al. Driving Forces for changes in geographical distribution of Ixodes ricinus ticks in Europe. Parasit. Vectors 2013, 6, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogden, N. Changing Geographic ranges of ticks and tick-borne pathogens: Drivers, mechanisms and consequences for pathogen diversity. Front. Cell. Infect. Microbiol. 2013, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-Fons, F.; Fernández-de-Mera, I.G.; Acevedo, P.; Gortázar, C.; de la Fuente, J. Factors driving the abundance of Ixodes ricinus ticks and the prevalence of zoonotic I. ricinus-borne pathogens in natural foci. Appl. Environ. Microbiol. 2012, 78, 2669–2676. [Google Scholar] [CrossRef] [Green Version]
- Carpio, A.J.; Apollonio, M.; Acevedo, P. Wild ungulate overabundance in Europe: Contexts, causes, monitoring and management recommendations. Mammal Rev. 2021, 51, 95–108. [Google Scholar] [CrossRef]
- Apollonio, M.; Andersen, R.; Putman, R. European Ungulates and Their Management in the 21st Century; Cambridge University Press: Cambridge, UK, 2010; ISBN 978-0-521-76061-4. [Google Scholar]
- De Pasquale, D.; Dondina, O.; Scancarello, E.; Meriggi, A. Long-term viability of a reintroduced population of roe deer Capreolus capreolus, in a lowland area of northern Italy. Folia Zool. 2019, 68, 9–20. [Google Scholar] [CrossRef]
- Randi, E. Management of wild ungulate populations in Italy: Captive-breeding, hybridisation and genetic consequences of translocations. Vet. Res. Commun. 2005, 29, 71–75. [Google Scholar] [CrossRef]
- Randi, E.; Alves, P.C.; Carranza, J.; Milošević-Zlatanović, S.; Sfougaris, A.; Mucci, N. Phylogeography of roe deer (Capreolus capreolus) populations: The effects of historical genetic subdivisions and recent nonequilibrium dynamics. Mol. Ecol. 2004, 13, 3071–3083. [Google Scholar] [CrossRef]
- Stuen, S. Anaplasma phagocytophilum-the most widespread tick-borne infection in animals in Europe. Vet. Res. Commun. 2007, 31, 79–84. [Google Scholar] [CrossRef]
- Woldehiwet, Z. Immune evasion and immunosuppression by Anaplasma phagocytophilum, the causative agent of tick-borne fever of ruminants and human granulocytic anaplasmosis. Vet. J. 2008, 175, 37–44. [Google Scholar] [CrossRef]
- Ravnik, U.; Tozon, N.; Strasek, K.; Zupanc, T.A. Clinical and haematological features in Anaplasma phagocytophilum seropositive dogs. Clin. Microbiol. Infect. 2009, 15, 39–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stuen, S.; Olsson Engvall, E.; Artursson, K. Persistence of Ehrlichia phagocytophila infection in lambs in relation to clinical parameters and antibody responses. Vet. Rec. 1998, 143, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Kogler, S.; Gotthalmseder, E.; Shahi-Barogh, B.; Harl, J.; Fuehrer, H.-P. Babesia spp. and Anaplasma phagocytophilum in free-ranging wild ungulates in central Austria. Ticks Tick-Borne Dis. 2021, 12, 101719. [Google Scholar] [CrossRef] [PubMed]
- Overzier, E.; Pfister, K.; Herb, I.; Mahling, M.; Böck, G.; Silaghi, C. Detection of tick-borne pathogens in roe deer (Capreolus capreolus), in questing ticks (Ixodes ricinus), and in ticks infesting roe deer in southern Germany. Ticks Tick-Borne Dis. 2013, 4, 320–328. [Google Scholar] [CrossRef]
- Sun, Y.; Li, S.-G.; Jiang, J.-F.; Wang, X.; Zhang, Y.; Wang, H.; Cao, W.-C. Babesia venatorum infection in child, China. Emerg. Infect. Dis. 2014, 20, 896–897. [Google Scholar] [CrossRef]
- Malandrin, L.; Jouglin, M.; Sun, Y.; Brisseau, N.; Chauvin, A. Redescription of Babesia capreoli (Enigk and Friedhoff, 1962) from roe deer (Capreolus capreolus): Isolation, cultivation, host specificity, molecular characterisation and differentiation from Babesia divergens. Int. J. Parasitol. 2010, 40, 277–284. [Google Scholar] [CrossRef]
- Häselbarth, K.; Tenter, A.M.; Brade, V.; Krieger, G.; Hunfeld, K.-P. First case of human babesiosis in Germany–Clinical presentation and molecular characterisation of the pathogen. Int. J. Med. Microbiol. 2007, 297, 197–204. [Google Scholar] [CrossRef]
- Fonseca, M.S.; Bahiense, T.C.; Silva, A.A.B.; Onofrio, V.C.; Barral, T.D.; Souza, B.M.P.; Lira-da-Silva, R.M.; Biondi, I.; Meyer, R.; Portela, R.W. Ticks and associated pathogens from rescued wild animals in rainforest fragments of northeastern Brazil. Front. Vet. Sci. 2020, 7, 177. [Google Scholar] [CrossRef]
- Szekeres, S.; van Leeuwen, A.D.; Tóth, E.; Majoros, G.; Sprong, H.; Földvári, G. Road-killed mammals provide insight into tick-borne bacterial pathogen communities within urban habitats. Transbound. Emerg. Dis. 2019, 66, 277–286. [Google Scholar] [CrossRef] [Green Version]
- Chianucci, F.; Mattioli, L.; Amorini, E.; Giannini, T.; Marcon, A.; Chirichella, R.; Apollonio, M.; Cutini, A. Early and long-term impacts of browsing by roe deer in oak coppiced woods along a gradient of population density. Ann. Silvic. Res. 2015, 39. [Google Scholar] [CrossRef]
- Dondina, O.; Orioli, V.; Chiatante, G.; Meriggi, A.; Bani, L. Species specialization limits movement ability and shapes ecological networks: The case study of 2 forest mammals. Curr. Zool. 2019, 65, 237–249. [Google Scholar] [CrossRef]
- Putzu, N.; Bonetto, D.; Civallero, V.; Fenoglio, S.; Meneguz, P.G.; Preacco, N.; Tizzani, P. Temporal patterns of ungulate-vehicle collisions in a subalpine Italian region. Ital. J. Zool. 2014, 81, 463–470. [Google Scholar] [CrossRef]
- Bonnot, N.; Morellet, N.; Verheyden, H.; Cargnelutti, B.; Lourtet, B.; Klein, F.; Hewison, A.J.M. Habitat use under predation risk: Hunting, roads and human dwellings influence the spatial behaviour of roe deer. Eur. J. Wildl. Res. 2013, 59, 185–193. [Google Scholar] [CrossRef]
- Riga, F.; Genghini, M.; Cascone, C.; Di Luzio, P. Impatto Degli Ungulati Sulle Culture Agricole a Forestali Proposta per Linee Guida Nazionali; Manuali e Linee Guida ISPRA; ISPRA Publ.: Rome, Italy, 2011; Volume 68. [Google Scholar]
- Di Lorenzo, E.; Rossi, R.; Ferrari, F.; Martini, V.; Comazzi, S. Blood L-lactate concentration as an indicator of outcome in roe deer (Capreolus capreolus) admitted to a wildlife rescue center. Animals 2020, 10, 1066. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, V.; González, I.; Martín, I.; Rojas, M.; Hernández, P.E.; García, T.; Martín, R. Real-time PCR for detection and quantification of red deer (Cervus elaphus), fallow deer (Dama dama), and roe deer (Capreolus capreolus) in meat mixtures. Meat Sci. 2008, 79, 289–298. [Google Scholar] [CrossRef]
- Casati, S.; Sager, H.; Gern, L.; Piffaretti, J.-C. Presence of potentially pathogenic Babesia sp. for human in Ixodes ricinus in Switzerland. Ann. Agric. Environ. Med. AAEM 2006, 13, 65–70. [Google Scholar]
- Aktas, M.; Altay, K.; Dumanli, N.; Kalkan, A. Molecular detection and identification of Ehrlichia and Anaplasma species in ixodid ticks. Parasitol. Res. 2009, 104, 1243. [Google Scholar] [CrossRef] [PubMed]
- Gouy, M.; Guindon, S.; Gascuel, O. SeaView Version 4: A Multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol. Biol. Evol. 2010, 27, 221–224. [Google Scholar] [CrossRef] [Green Version]
- Cafiso, A.; Olivieri, E.; Floriano, A.M.; Chiappa, G.; Serra, V.; Sassera, D.; Bazzocchi, C. Investigation of tick-borne pathogens in Ixodes ricinus in a peri-urban park in Lombardy (Italy) reveals the presence of emerging pathogens. Pathogens 2021, 10, 732. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Social Science Statistics. Available online: https://www.socscistatistics.com/ (accessed on 26 October 2021).
- Remesar, S.; Díaz, P.; Prieto, A.; García-Dios, D.; Fernández, G.; López, C.M.; Panadero, R.; Díez-Baños, P.; Morrondo, P. Prevalence and molecular characterization of Anaplasma phagocytophilum in roe deer (Capreolus capreolus) from Spain. Ticks Tick-Borne Dis. 2020, 11, 101351. [Google Scholar] [CrossRef] [PubMed]
- Tampieri, M.P.; Galuppi, R.; Bonoli, C.; Cancrini, G.; Moretti, A.; Pietrobelli, M. Wild ungulates as Babesia hosts in northern and central Italy. Vector-Borne Zoonotic Dis. 2008, 8, 667–674. [Google Scholar] [CrossRef]
- Duh, D.; Petrovec, M.; Avsic-Zupanc, T. Diversity of Babesia infecting european sheep ticks (Ixodes ricinus). J. Clin. Microbiol. 2001, 39, 3395–3397. [Google Scholar] [CrossRef] [Green Version]
- Hoby, S.; Mathis, A.; Doherr, M.G.; Robert, N.; Ryser-Degiorgis, M.-P. Babesia capreoli infections in alpine chamois (Rupicapra r. rupicapra), roe deer (Capreolus c. capreolus) and red deer (Cervus elaphus) from Switzerland. J. Wildl. Dis. 2009, 45, 748–753. [Google Scholar] [CrossRef] [Green Version]
- Kauffmann, M.; Rehbein, S.; Hamel, D.; Lutz, W.; Heddergott, M.; Pfister, K.; Silaghi, C. Anaplasma phagocytophilum and Babesia spp. in roe deer (Capreolus capreolus), fallow deer (Dama dama) and mouflon (Ovis musimon) in Germany. Mol. Cell. Probes 2017, 31, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Silaghi, C.; Hamel, D.; Pfister, K.; Rehbein, S. Babesia species and co-infection with Anaplasma phagocytophilum in free-ranging ungulates from Tyrol. Tierärztliche Mschr. Vet. Med. Austria 2011, 98, 268–274. [Google Scholar]
- Andersson, M.O.; Bergvall, U.A.; Chirico, J.; Christensson, M.; Lindgren, P.-E.; Nordström, J.; Kjellander, P. Molecular detection of Babesia capreoli and Babesia venatorum in wild Swedish roe deer, Capreolus capreolus. Parasit. Vectors 2016, 9, 221. [Google Scholar] [CrossRef] [Green Version]
- Gray, A.; Capewell, P.; Loney, C.; Katzer, F.; Shiels, B.R.; Weir, W. Sheep as host species for zoonotic Babesia venatorum, United Kingdom. Emerg. Infect. Dis. 2019, 25, 2257–2260. [Google Scholar] [CrossRef] [Green Version]
- Zanet, S.; Trisciuoglio, A.; Bottero, E.; de Mera, I.G.F.; Gortazar, C.; Carpignano, M.G.; Ferroglio, E. Piroplasmosis in wildlife: Babesia and Theileria affecting free-ranging ungulates and carnivores in the Italian Alps. Parasit. Vectors 2014, 7, 70. [Google Scholar] [CrossRef] [Green Version]
- Hoby, S.; Robert, N.; Mathis, A.; Schmid, N.; Meli, M.L.; Hofmann-Lehmann, R.; Lutz, H.; Deplazes, P.; Ryser-Degiorgis, M.-P. Babesiosis in free-ranging chamois (Rupicapra r. rupicapra) from Switzerland. Vet. Parasitol. 2007, 148, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Penzhorn, B.L. Babesiosis of wild carnivores and ungulates. Vet. Parasitol. 2006, 138, 11–21. [Google Scholar] [CrossRef] [Green Version]
- Fanelli, A. A historical review of Babesia spp. associated with deer in Europe: Babesia divergens/Babesia divergens-like, Babesia capreoli, Babesia venatorum, Babesia cf. odocoilei. Vet. Parasitol. 2021, 294, 109433. [Google Scholar] [CrossRef] [PubMed]
- Aureli, S.; Galuppi, R.; Ostanello, F.; Foley, J.E.; Bonoli, C.; Rejmanek, D.; Rocchi, G.; Orlandi, E.; Tampieri, M.P. Abundance of questing ticks and molecular evidence for pathogens in ticks in three parks of Emilia-Romagna region of northern Italy. Ann. Agric. Environ. Med. 2015, 22, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Herwaldt, B.L.; Cacciò, S.; Gherlinzoni, F.; Aspöck, H.; Slemenda, S.B.; Piccaluga, P.; Martinelli, G.; Edelhofer, R.; Hollenstein, U.; Poletti, G.; et al. Molecular Characterization of a non–Babesia divergens organism causing zoonotic babesiosis in Europe. Emerg. Infect. Dis. 2003, 9, 943–948. [Google Scholar] [CrossRef]
- Zhao, L.; Jiang, R.; Jia, N.; Ning, N.; Zheng, Y.; Huo, Q.; Sun, Y.; Yuan, T.; Jiang, B.; Li, T.; et al. Human case infected with Babesia venatorum: A 5-year follow-up study. Open Forum Infect. Dis. 2020, 7. [Google Scholar] [CrossRef] [Green Version]
- Michel, A.O.; Mathis, A.; Ryser-Degiorgis, M.-P. Babesia spp. in European wild ruminant species: Parasite diversity and risk factors for infection. Vet. Res. 2014, 45, 65. [Google Scholar] [CrossRef] [Green Version]
- Razanske, I.; Rosef, O.; Radzijevskaja, J.; Bratchikov, M.; Griciuviene, L.; Paulauskas, A. Prevalence and co-infection with tick-borne Anaplasma phagocytophilum and Babesia spp. in red deer (Cervus elaphus) and roe deer (Capreolus capreolus) in southern Norway. Int. J. Parasitol. Parasites Wildl. 2019, 8, 127–134. [Google Scholar] [CrossRef]
- Di Domenico, M.; Pascucci, I.; Curini, V.; Cocco, A.; Dall’Acqua, F.; Pompilii, C.; Cammà, C. Detection of Anaplasma phagocytophilum genotypes that are potentially virulent for human in wild ruminants and Ixodes ricinus in central Italy. Ticks Tick-Borne Dis. 2016, 7, 782–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebani, V.V.; Cerri, D.; Fratini, F.; Ampola, M.; Andreani, E. Anaplasma phagocytophilum infection in a fallow deer (Dama dama) population in a preserve of central Italy. New Microbiol. 2007, 30, 161–165. [Google Scholar]
- Grassi, L.; Franzo, G.; Martini, M.; Mondin, A.; Cassini, R.; Drigo, M.; Pasotto, D.; Vidorin, E.; Menandro, M.L. Ecotyping of Anaplasma phagocytophilum from wild ungulates and ticks shows circulation of zoonotic strains in northeastern Italy. Animals 2021, 11, 310. [Google Scholar] [CrossRef]
- Carpi, G.; Bertolotti, L.; Pecchioli, E.; Cagnacci, F.; Rizzoli, A. Anaplasma phagocytophilum groEL gene heterogeneity in Ixodes ricinus larvae feeding on roe deer in northeastern Italy. Vector-Borne Zoonotic Dis. 2008, 9, 179–184. [Google Scholar] [CrossRef] [Green Version]
- Hornok, S.; Sugár, L.; Fernández de Mera, I.G.; de la Fuente, J.; Horváth, G.; Kovács, T.; Micsutka, A.; Gönczi, E.; Flaisz, B.; Takács, N.; et al. Tick- and fly-borne bacteria in ungulates: The prevalence of Anaplasma phagocytophilum, haemoplasmas and rickettsiae in water buffalo and deer species in central Europe, Hungary. BMC Vet. Res. 2018, 14, 98. [Google Scholar] [CrossRef]
- Jahfari, S.; Coipan, E.C.; Fonville, M.; van Leeuwen, A.D.; Hengeveld, P.; Heylen, D.; Heyman, P.; van Maanen, C.; Butler, C.M.; Földvári, G.; et al. Circulation of four Anaplasma phagocytophilum ecotypes in Europe. Parasit. Vectors 2014, 7, 365. [Google Scholar] [CrossRef] [Green Version]
- Huhn, C.; Winter, C.; Wolfsperger, T.; Wüppenhorst, N.; Smrdel, K.S.; Skuballa, J.; Pfäffle, M.; Petney, T.; Silaghi, C.; Dyachenko, V.; et al. Analysis of the population structure of Anaplasma phagocytophilum using multilocus sequence typing. PLoS ONE 2014, 9, e93725. [Google Scholar] [CrossRef] [Green Version]
- Dugat, T.; Lagrée, A.-C.; Maillard, R.; Boulouis, H.-J.; Haddad, N. Opening the black box of Anaplasma phagocytophilum diversity: Current situation and future perspectives. Front. Cell. Infect. Microbiol. 2015, 5, 61. [Google Scholar] [CrossRef]
- Silaghi, C.; Fröhlich, J.; Reindl, H.; Hamel, D.; Rehbein, S. Anaplasma phagocytophilum and Babesia species of sympatric roe deer (Capreolus capreolus), fallow deer (Dama dama), sika deer (Cervus nippon) and red deer (Cervus elaphus) in Germany. Pathogens 2020, 9, 968. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.C.; Barbet, A.F. Persistent infections and immunity in ruminants to arthropod-borne bacteria in the family Anaplasmataceae. Annu. Rev. Anim. Biosci. 2016, 4, 177–197. [Google Scholar] [CrossRef] [PubMed]
- Bastian, S.; Jouglin, M.; Brisseau, N.; Malandrin, L.; Klegou, G.; L’Hostis, M.; Chauvin, A. Antibody prevalence and molecular identification of Babesia spp. in roe deer in France. J. Wildl. Dis. 2012, 48, 416–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cafiso, A.; Bazzocchi, C.; Cavagna, M.; Di Lorenzo, E.; Serra, V.; Rossi, R.; Comazzi, S. Molecular Survey of Babesia spp. and Anaplasma phagocytophilum in Roe Deer from a Wildlife Rescue Center in Italy. Animals 2021, 11, 3335. https://doi.org/10.3390/ani11113335
Cafiso A, Bazzocchi C, Cavagna M, Di Lorenzo E, Serra V, Rossi R, Comazzi S. Molecular Survey of Babesia spp. and Anaplasma phagocytophilum in Roe Deer from a Wildlife Rescue Center in Italy. Animals. 2021; 11(11):3335. https://doi.org/10.3390/ani11113335
Chicago/Turabian StyleCafiso, Alessandra, Chiara Bazzocchi, Martina Cavagna, Elena Di Lorenzo, Valentina Serra, Riccardo Rossi, and Stefano Comazzi. 2021. "Molecular Survey of Babesia spp. and Anaplasma phagocytophilum in Roe Deer from a Wildlife Rescue Center in Italy" Animals 11, no. 11: 3335. https://doi.org/10.3390/ani11113335
APA StyleCafiso, A., Bazzocchi, C., Cavagna, M., Di Lorenzo, E., Serra, V., Rossi, R., & Comazzi, S. (2021). Molecular Survey of Babesia spp. and Anaplasma phagocytophilum in Roe Deer from a Wildlife Rescue Center in Italy. Animals, 11(11), 3335. https://doi.org/10.3390/ani11113335






