Current Advances in Assessment of Dog’s Emotions, Facial Expressions, and Their Use for Clinical Recognition of Pain
Abstract
Simple Summary
Abstract
1. Introduction
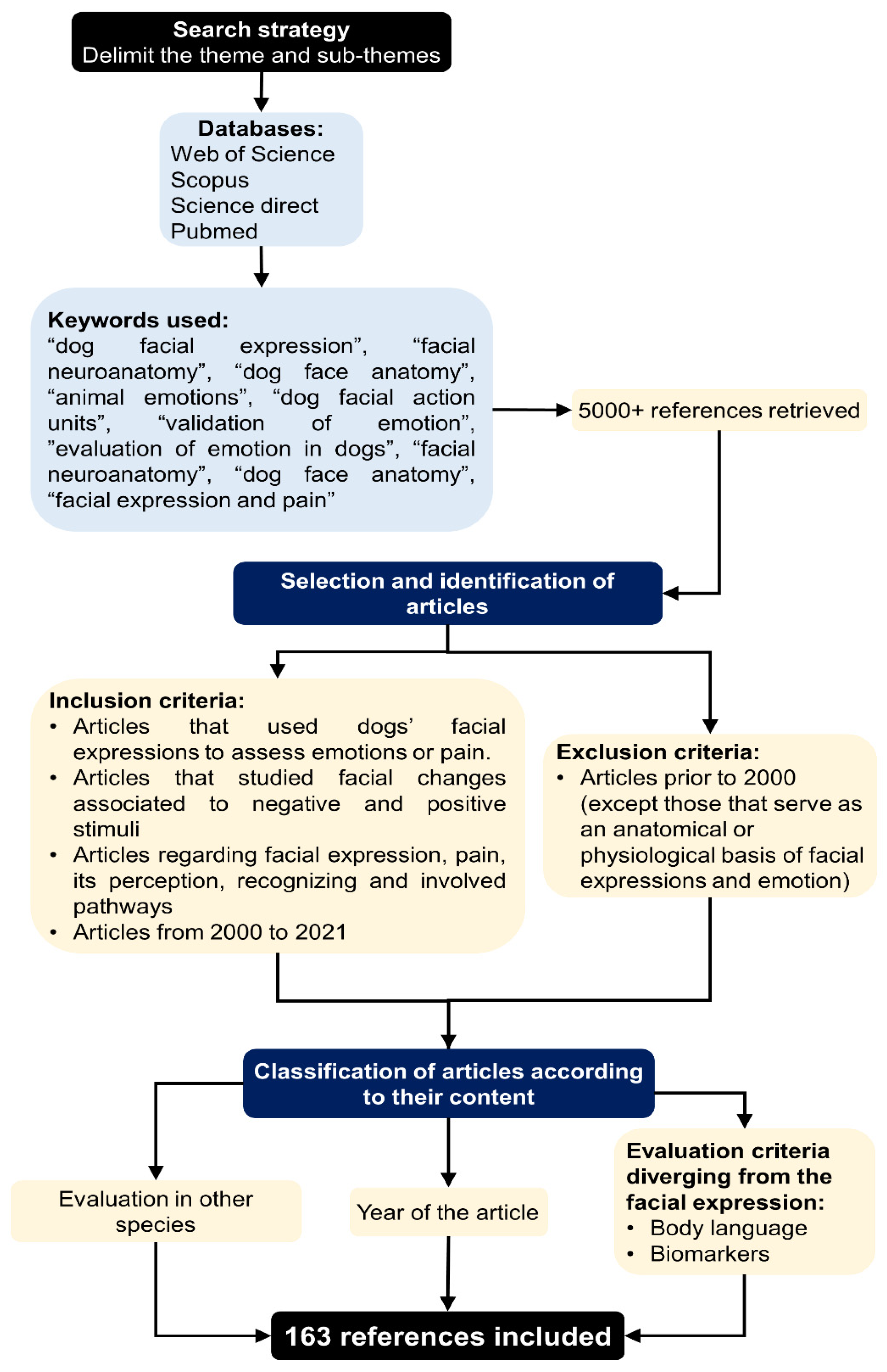
2. Methodology
3. Are Facial Expressions Involuntary and Emotional or Do They Have a Real Communication Purpose?
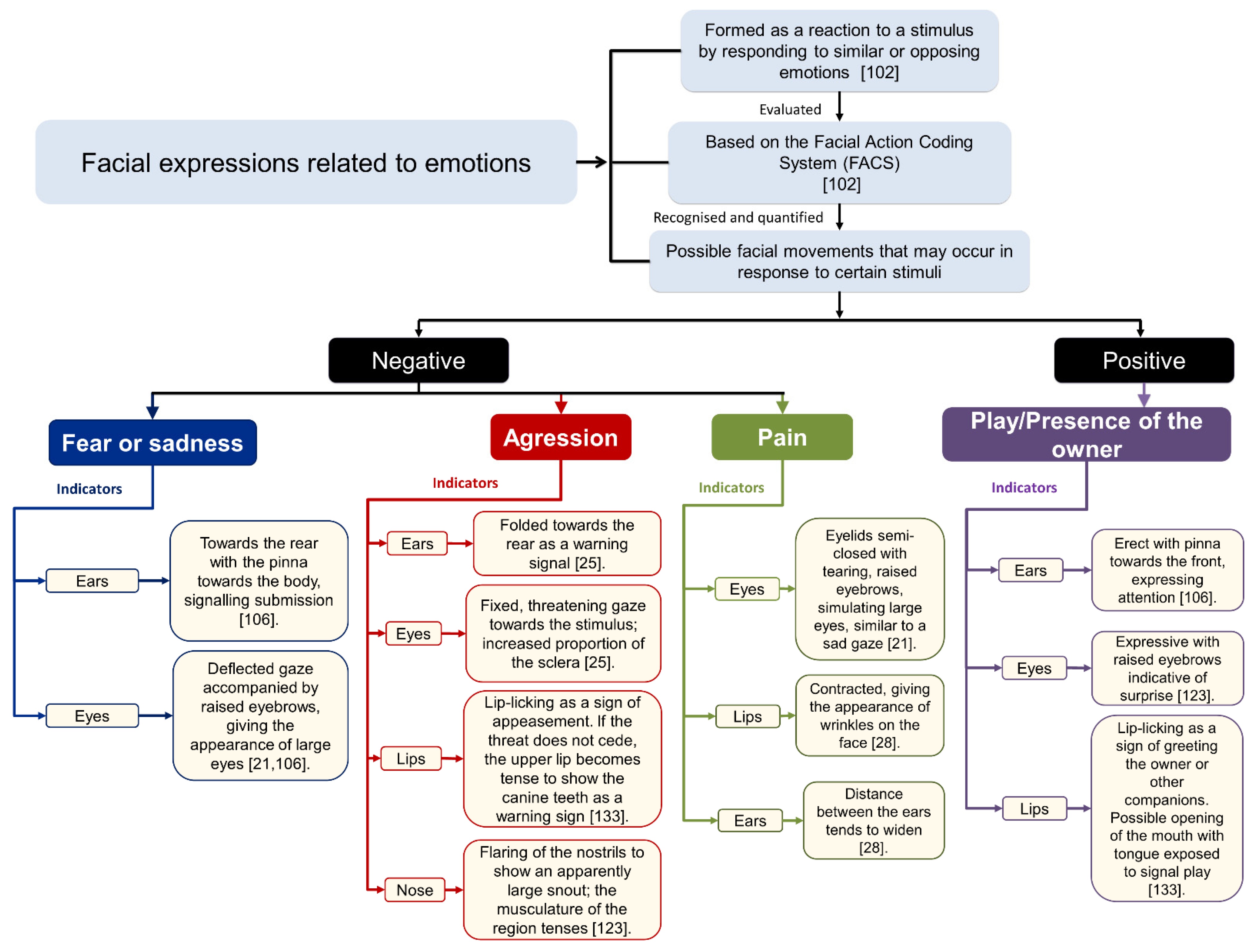
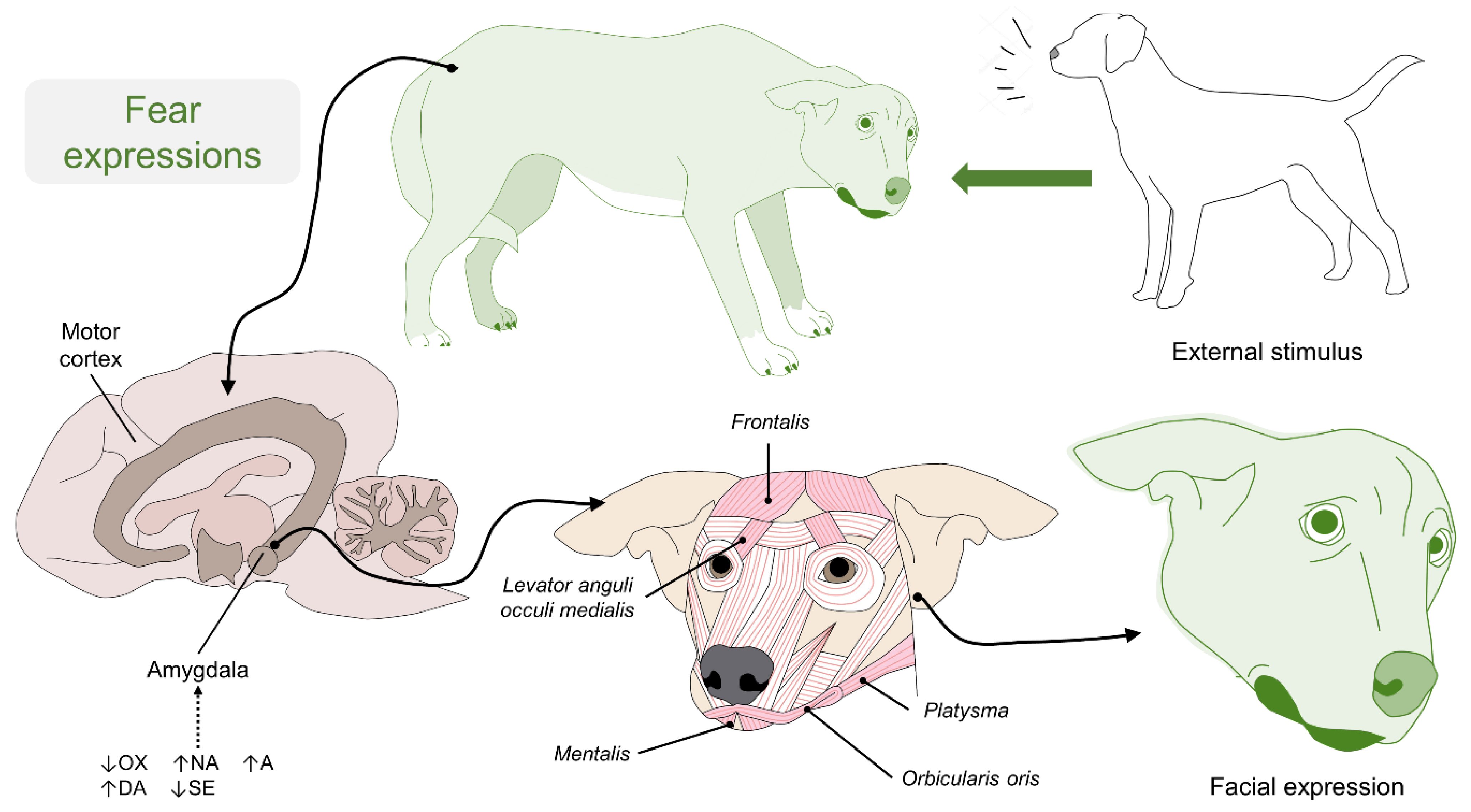
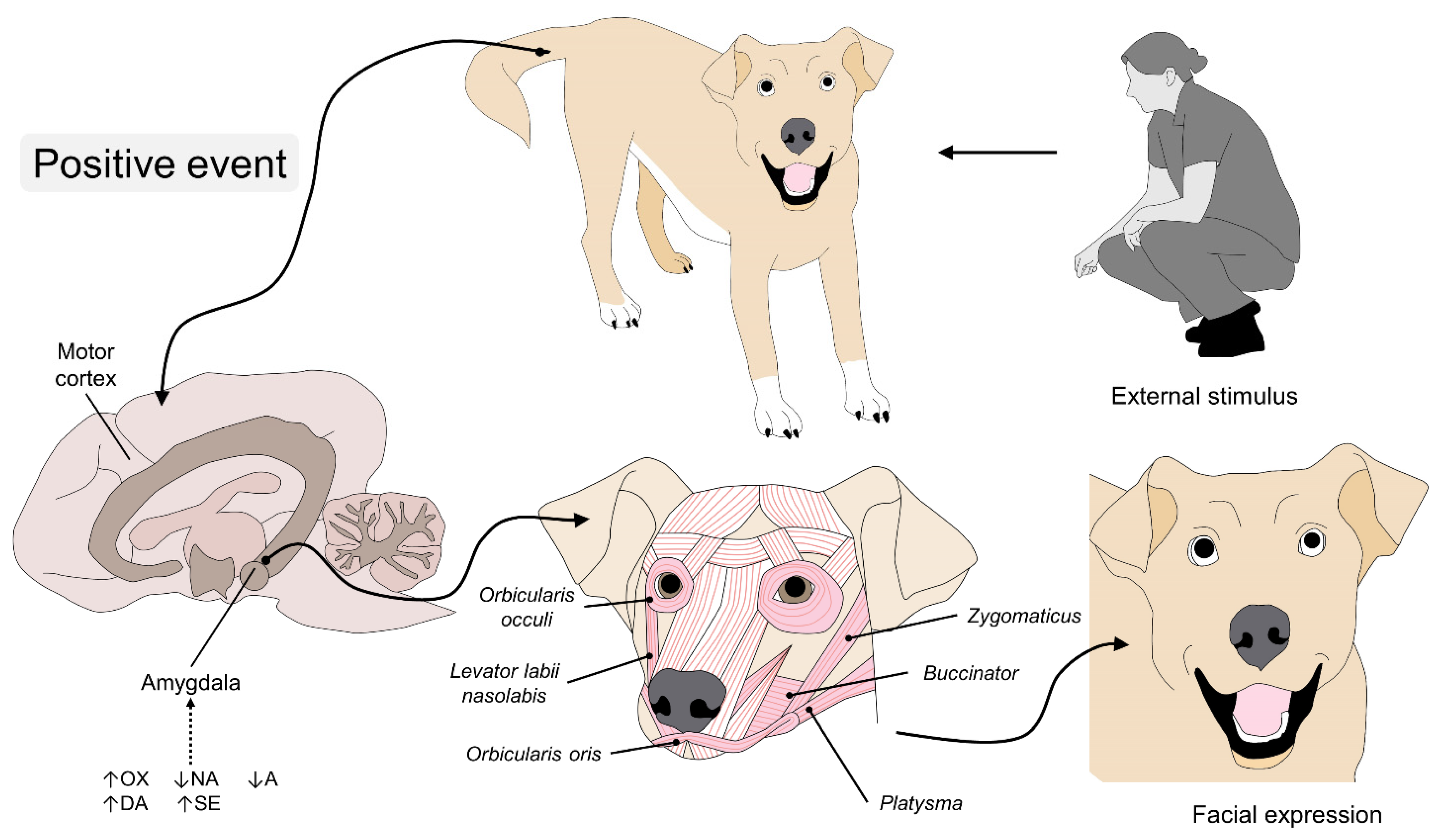
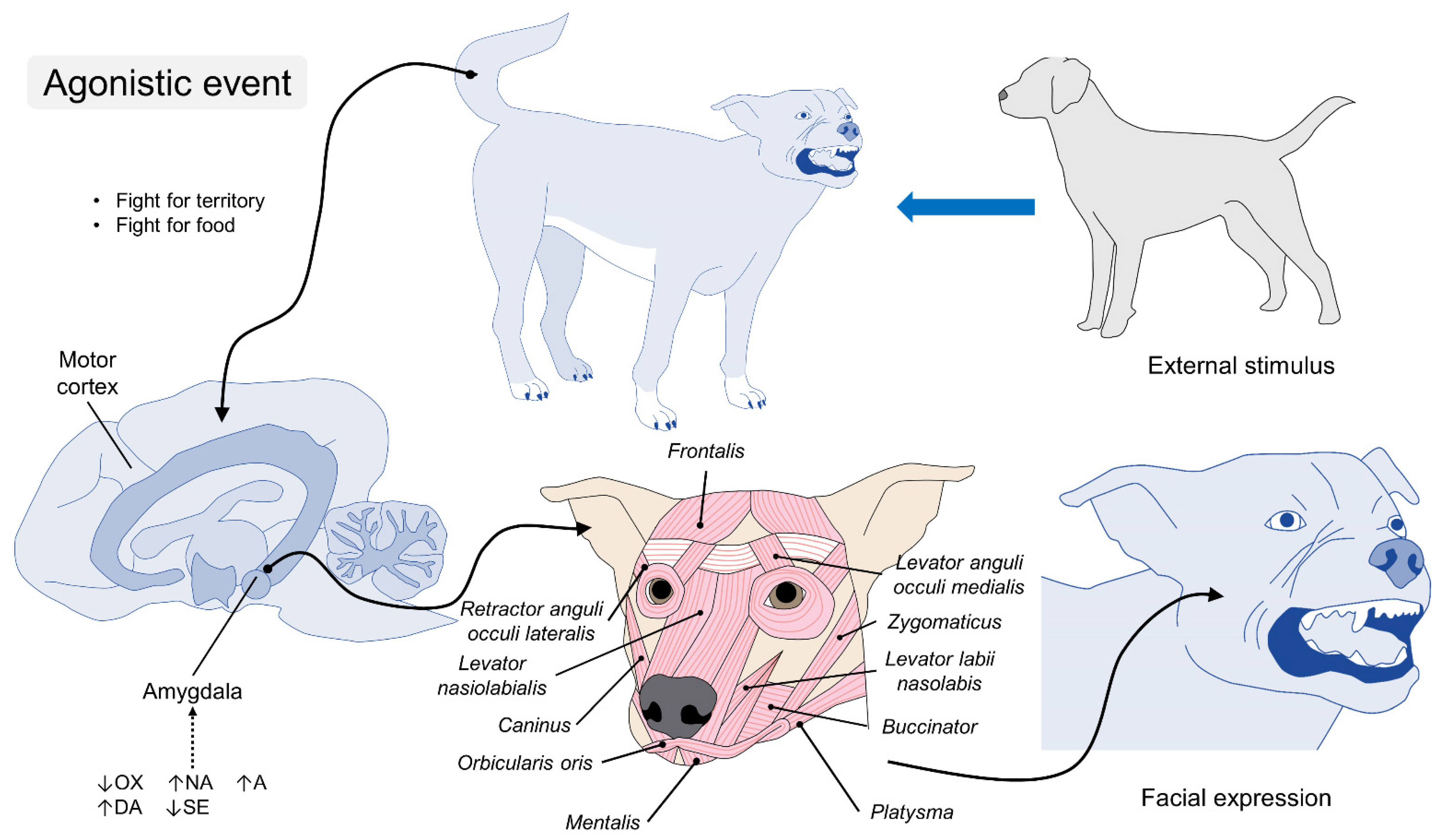
4. The Anatomy of Facial Expressions in Dogs
5. The Physiology of Facial Expressions
6. Emotions in the Dog
7. Interpretation of Perception and Expression Interspecies
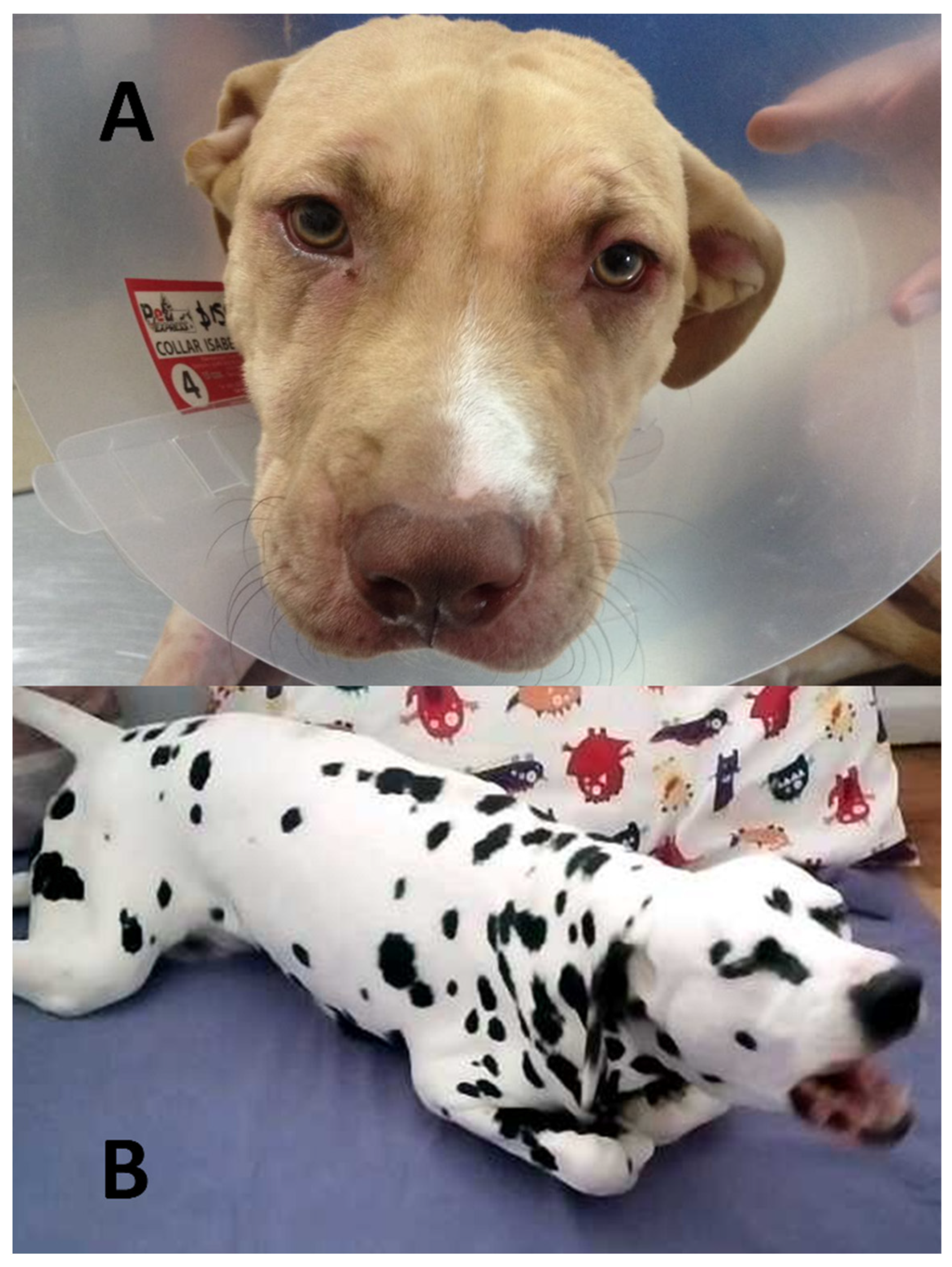
8. Changes in Facial Expressions Related to Pain
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Crivelli, C.; Fridlund, A.J. Facial displays are tools for social influence. Trends Cogn. Sci. 2018, 22, 388–399. [Google Scholar] [CrossRef]
- Mota-Rojas, D.; Olmos-Hernández, A.; Verduzco-Mendoza, A.; Hernández, E.; Martínez-Burnes, J.; Whittaker, A. The utility of grimace escales for practical pain assessment in laboratory animals. Animals 2020, 10, 1838. [Google Scholar] [CrossRef]
- Darwin, C. The Expressions of the Emotions in Man and Animals; William Clowes and Sons: London, UK, 1872; pp. 1–399. [Google Scholar]
- Boissy, A.; Manteuffel, G.; Jensen, M.B.; Moe, R.O.; Spruijt, B.; Keeling, L.J.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef]
- Konok, V.; Nagy, K.; Miklósi, Á. How do humans represent the emotions of dogs? The resemblance between the human representation of the canine and the human affective space. Appl. Anim. Behav. Sci. 2015, 162, 37–46. [Google Scholar] [CrossRef]
- Leliveld, L.M.C.; Langbein, J.; Puppe, B. The emergence of emotional lateralization: Evidence in non-human vertebrates and implications for farm animals. Appl. Anim. Behav. Sci. 2013, 145, 1–14. [Google Scholar] [CrossRef]
- Bennett, V.; Gourkow, N.; Mills, D.S. Facial correlates of emotional behaviour in the domestic cat (Felis catus). Behav. Proces. 2017, 141, 342–350. [Google Scholar] [CrossRef]
- Wathan, J.; Proops, L.; Grounds, K.; McComb, K. Horses discriminate between facial expressions of conspecifics. Sci. Rep. 2016, 6, 38322. [Google Scholar] [CrossRef]
- Wathan, J.; Burrows, A.M.; Waller, B.M.; McComb, K. EquiFACS: The equine facial action coding system. PLoS ONE 2015, 10, e0131738. [Google Scholar] [CrossRef]
- McLennan, K.M.; Rebelo, C.J.B.; Corke, M.J.; Holmes, M.A.; Leach, M.C.; Constantino-Casas, F. Development of a facial expression scale using footrot and mastitis as models of pain in sheep. Appl. Anim. Behav. Sci. 2016, 176, 19–26. [Google Scholar] [CrossRef]
- Häger, C.; Biernot, S.; Buettner, M.; Glage, S.; Keubler, L.M.; Held, N.; Bleich, E.M.; Otto, K.; Müller, C.W.; Decker, S.; et al. The sheep grimace scale as an indicator of post-operative distress and pain in laboratory sheep. PLoS ONE 2017, 12, e0175839. [Google Scholar] [CrossRef] [PubMed]
- Proctor, H.S.; Carder, G. Can ear postures reliably measure the positive emotional state of cows? Appl. Anim. Behav. Sci. 2014, 161, 20–27. [Google Scholar] [CrossRef]
- Di Giminiani, P.; Brierley, V.L.M.H.; Scollo, A.; Gottardo, F.; Malcolm, E.M.; Edwards, S.A.; Leach, M.C. The assessment of facial expressions in piglets undergoing tail docking and castration: Toward the development of the piglet grimace scale. Front. Vet. Sci. 2016, 3, 100. [Google Scholar] [CrossRef] [PubMed]
- Viscardi, A.V.; Hunniford, M.; Lawlis, P.; Leach, M.; Turner, P.V. Development of a piglet grimace scale to evaluate piglet pain using facial expressions following castration and tail docking: A pilot study. Front. Vet. Sci. 2017, 4, 51. [Google Scholar] [CrossRef]
- Mota-Rojas, D.; Orihuela, A.; Martínez-Burnes, J.; Gómez, J.; Mora-Medina, P.; Alavez, B.; Ramírez, L.; González-Lozano, M. Neurological modulation of facial expressions in pigs and implications for production. J. Anim. Behav. Biometeorol. 2020, 8, 232–243. [Google Scholar] [CrossRef]
- Descovich, K. Facial expression: An under-utilised tool for the assessment of welfare in mammals. ALTEX 2017, 34, 409–429. [Google Scholar] [CrossRef]
- Waller, B.M.; Micheletta, J. Facial expression in nonhuman animals. Emot. Rev. 2013, 5, 54–59. [Google Scholar] [CrossRef]
- Waller, B.M.; Peirce, K.; Caeiro, C.C.; Scheider, L.; Burrows, A.M.; McCune, S.; Kaminski, J. Paedomorphic facial expressions give dogs a selective advantage. PLoS ONE 2013, 8, e82686. [Google Scholar] [CrossRef] [PubMed]
- Mariti, C.; Ricci, E.; Zilocchi, M.; Gazzano, A. Owners as a secure base for their dogs. Behaviour 2013, 150, 1275–1294. [Google Scholar] [CrossRef]
- Hare, B.; Tomasello, M. Human-like social skills in dogs? Trends Cogn. Sci. 2005, 9, 439–444. [Google Scholar] [CrossRef]
- Ford, G.; Guo, K.; Mills, D. Human facial expression affects a dog’s response to conflicting directional gestural cues. Behav. Process. 2019, 159, 80–85. [Google Scholar] [CrossRef]
- Katayama, M.; Kubo, T.; Yamakawa, T.; Fujiwara, K.; Nomoto, K.; Ikeda, K.; Mogi, K.; Nagasawa, M.; Kikusui, T. Emotional contagion from humans to dogs is facilitated by duration of ownership. Front. Psychol. 2019, 10, 1678. [Google Scholar] [CrossRef] [PubMed]
- de Waal, F.B.M. What is an animal emotion? Ann. N. Y. Acad. Sci. 2011, 1224, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Ekman, P. Body position, facial expression, and verbal behavior during interviews. J. Abnorm. Soc. Psych. 1964, 68, 295–301. [Google Scholar] [CrossRef]
- Bloom, T.; Friedman, H. Classifying dogs’ (Canis Familiaris) facial expressions from photographs. Behav. Proc. 2013, 96, 1–10. [Google Scholar] [CrossRef]
- Reid, J.; Nolan, A.M.; Scott, E.M. Measuring pain in dogs and cats using structured behavioural observation. Vet. J. 2018, 236, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Corke, M.J. Indicators of pain. In Encyclopedia of Animal Behavior, 2nd ed.; Choe, C.J., Ed.; Academic Press: Oxford, UK, 2019; Volume 1, pp. 147–152. [Google Scholar]
- Lexis, H.; Weary, D.M. Facial Expressions in humans as a measure of empathy towards farm animals in pain. PLoS ONE 2021, 16, e0247808. [Google Scholar] [CrossRef]
- McLennan, K.M.; Miller, A.L.; Dalla Costa, E.; Stucke, D.; Corke, M.J.; Broom, D.M.; Leach, M.C. Conceptual and methological issues relating to pain assessment in mammals: The development and ustilisation of pain facial expressions scales. Appl. Anim. Behav. Sci. 2019, 217, 1–15. [Google Scholar] [CrossRef]
- Holden, E.; Calvo, G.; Collins, M.; Bell, A.; Reid, J.; Scott, E.M.; Nolan, A.M. Evaluation of facial expression in acute pain in cats. J. Small Anim. Pract. 2014, 55, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, M.C.; Watanabe, R.; Leung, V.S.Y.; Monteiro, B.P.; O’Toole, E.; Pang, D.S.J.; Steagall, P.V. Facial Expressions of pain in cats: The development and validation of a feline grimace scale. Sci. Rep. 2019, 9, 19128. [Google Scholar] [CrossRef]
- Langford, D.J.; Bailey, A.L.; Chanda, M.L.; Clarke, S.E.; Drummond, T.E.; Echols, S.; Glick, S.; Ingrao, J.; Klassen-Ross, T.; LaCroix-Fralish, M.L.; et al. Coding of facial expressions of pain in the laboratory mouse. Nat. Methods 2010, 7, 447–449. [Google Scholar] [CrossRef]
- Sotocinal, S.G.; Sorge, R.E.; Zaloum, A.; Tuttle, A.H.; Martin, L.J.; Wieskopf, J.S.; Mapplebeck, J.C.; Wei, P.; Zhan, S.; Zhang, S.; et al. The rat grimace scale: A partially automated method for quantifying pain in the laboratory rat via facial expressions. Mol. Pain 2011, 7, 1744–8069. [Google Scholar] [CrossRef]
- Lezama-García, K.; Orihuela, A.; Olmos-Hernández, A.; Reyes-Long, S.; Mota-Rojas, D. Facial expressions and emotions in domestic animals. CAB. Rev. 2019, 14, 1–12. [Google Scholar] [CrossRef]
- Ekman, P. Are there basic emotions? Psychol. Rev. 1992, 99, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.; Alvarado, N. Facial expressions, smile types, and self-report during humour, tickle, and pain. Cogn. Emot. 2005, 19, 655–669. [Google Scholar] [CrossRef]
- Camerlink, I.; Coulange, E.; Farish, M.; Baxter, E.M.; Turner, S.P. Facial expression as a potential measure of both intent and emotion. Sci. Rep. 2018, 8, 17602. [Google Scholar] [CrossRef]
- Gibson, J.E.; Pick, D.A. An Ecological Approach to Perceptual Learning and Development; Oxford University Press: New York, NY, USA, 2000; pp. 1–248. [Google Scholar]
- Gaynor, J.S.; Muir, W.W. Handbook of Veterinary Pain Management, 3rd ed.; Elsevier: Missouri, MO, USA, 2015; pp. 1–627. [Google Scholar]
- Siniscalchi, M.; D’Ingeo, S.; Minunno, M.; Quaranta, A. Communication in dogs. Animals 2018, 8, 131. [Google Scholar] [CrossRef]
- Handelman, B. Canine Behavior: A Photo Illustrated Handbook; Dogwise Publishing: Wenatchee, WA, USA, 2012. [Google Scholar]
- Hecht, J.; Horowitz, A. Introduction to dog behaviour. In The Ethology of Domestic Animals: An Introductory Text, 3rd ed.; Jensen, P., Ed.; CAB International: Oxford, UK, 2017; pp. 228–238. [Google Scholar]
- Kaminski, J.; Hynds, J.; Morris, P.; Waller, B.M. Human attention affects facial expressions in domestic dogs. Sci. Rep. 2017, 7, 12914. [Google Scholar] [CrossRef] [PubMed]
- Kunz, M.; Faltermeier, N.; Lautenbacher, S. Impact of visual learning on facial expressions of physical distress: A study on voluntary and evoked expressions of pain in congenitally blind and sighted individuals. Biol. Psychol. 2012, 89, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Bremhorst, A.; Mills, D.S.; Stolzlechner, L.; Würbel, H.; Riemer, S. ´Puppy dog eyes´ are associated with eye movements, not communication. Front. Psychol. 2021, 12, 568935. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, V.; Papaleo, F. Understanding others: Emotion recognition in humans and other animals. Genes Brain Behav. 2019, 18, e12544. [Google Scholar] [CrossRef]
- Bremhorst, A.; Sutter, N.A.; Würbel, H.; Mills, D.S.; Riemer, S. Differences in facial expressions during positive anticipation and frustration in dogs awaiting a reward. Sci. Rep. 2019, 9, 19312. [Google Scholar] [CrossRef] [PubMed]
- Panksepp, J. The basic emotional circuits of mammalian brains: Do animals have affective lives? Neurosci. Biobehav. Rev. 2011, 35, 1791–1804. [Google Scholar] [CrossRef] [PubMed]
- Cano-Vindel, A. Orientaciones en el estudio de la emoción. In Manual de Motivación y Emoción, 1st ed.; Fernández, E., Ed.; Editorial Universitaria Ramón Areces: Madrid, Spain, 1995; pp. 337–383. [Google Scholar]
- Domínguez-Oliva, A.; Mota-Rojas, D.; Ruiz-García, A.G.; Miranda-Cortés, Á.E.; Hernández-Avalos, I. Clinical recognition of stress in dog and cats. AMMVEPE 2021, 32, 24–35. [Google Scholar]
- Dawson, L.C.; Cheal, J.; Niel, L.; Mason, G. Humans can identify cats’ affective states from subtle facial expressions. Anim. Welf. 2019, 28, 519–531. [Google Scholar] [CrossRef]
- Andersen, P.H.; Broomé, S.; Rashid, M.; Lundblad, J.; Ask, K.; Li, Z.; Hernlund, E.; Rhodin, M.; Kjellström, H. Towards machine recognition of facial expressions of pain in horses. Animals 2021, 11, 1643. [Google Scholar] [CrossRef]
- Urrego, D.; Múnera, A.; Troncoso, J. Peripheral facial nerve lesion induced long-term dendritic retraction in pyramidal cortico-facial neurons. Biomed. Rev. Inst. Nac. Salud 2011, 31, 560–569. [Google Scholar] [CrossRef]
- Gil, V.A. Anatomía del Sistema Nervioso en el Perro y en el Gato; Servei de Publicacions Universitat Autónoma de Barcelona: Barcelona, Spain, 2008; pp. 1–263. [Google Scholar]
- Castillo, G.D.; de Jorge, J.L. Anatomía y Fisiología del Sistema Nervioso Central, 1st ed.; Fundación Universitaria San Pablo CEU: Madrid, Spain, 2015; pp. 9–699. [Google Scholar]
- Holmberg, J. The secretory nerves of the parotid gland of the dog. J. Physiol. 1971, 219, 463–476. [Google Scholar] [CrossRef]
- Waller, B.M.; Caeiro, C.; Peirce, K.; Burrows, A.M.; Kaminski, J. DogFACS: The Dog Facial Action Coding System Manual; University of Portsmouth: Cambridge, UK, 2013; pp. 1–62. [Google Scholar]
- Popesko, P. Atlas de Anatomía Topográfica en los Animales Domésticos, 2nd ed.; Masson, S.A., Ed.; En Stock: Barcelona, Spain, 1998; Volumen II, pp. 1–190. [Google Scholar]
- López, P.C.; Mayor, A.P.; Labeaga, J.R.; López, B.M.; Pereira, T.H.D.S.; Monteiro, F.O.B. Atlas de Los Músculos del Perro; Edufra: Terra Firme, Brazil, 2018; pp. 1–128. [Google Scholar]
- Burrows, A.M.; Kaminski, J.; Waller, B.M.; Omstead, K.M.; Rogers-Vizena, C.; Mendelson, B. Dog faces exhibit anatomical differences in comparison to other domestic animals. Anat. Rec. 2021, 304, 231–241. [Google Scholar] [CrossRef]
- Vick, S.-J.; Waller, B.M.; Parr, L.A.; Smith Pasqualini, M.C.; Bard, K.A. A Cross-species comparison of facial morphology and movement in humans and chimpanzees using the Facial Action Coding System (FACS). J. Nonverbal. Behav. 2007, 31, 1–20. [Google Scholar] [CrossRef]
- Caeiro, C.C.; Burrows, A.; Waller, B.M. Development and application of CatFACS: Are human cat adopters influenced by cat facial expressions? Appl. Anim. Behav. Sci. 2017, 189, 66–78. [Google Scholar] [CrossRef]
- Kaminski, J.; Waller, B.M.; Diogo, R.; Hartstone-Rose, A.; Burrows, A.M. Evolution of facial muscle anatomy in dogs. Proc. Natl. Acad. Sci. USA 2019, 116, 14677–14681. [Google Scholar] [CrossRef] [PubMed]
- Sternglanz, S.H.; Gray, J.L.; Murakami, M. Adult preferences for infantile facial features: An ethological approach. Anim. Behav. 1977, 25, 108–115. [Google Scholar] [CrossRef]
- Archer, J.; Monton, S. Preferences for infant facial features in pet dogs and cats. Ethology 2011, 118, 217–226. [Google Scholar] [CrossRef]
- Little, A.C. Manipulation of infant-like traits affects perceived cuteness of infant, adult and cat faces. Ethology 2012, 118, 775–782. [Google Scholar] [CrossRef]
- Correira-Caeiro, C.; Guo, K.; Mills, D.S. Perception of dynamic facial expressions of emotion between dogs and humans. Anim. Cogn. 2020, 23, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Erkoç, T.; Ağdoğan, D.; Eskil, M.T. An observation based muscle model for simulation of facial expressions. Signal Process. Image Commun. 2018, 64, 11–20. [Google Scholar] [CrossRef]
- Fenton, B.W.; Shih, E.; Zolton, J. The neurobiology of pain perception in normal and persistent pain. Pain Manag. 2015, 5, 297–317. [Google Scholar] [CrossRef]
- Mota-Rojas, D.; Mariti, C.; Zdeinert, A.; Riggio, G.; Mora-Medina, P.; del Mar Reyes, A.; Gazzano, A.; Domínguez-Oliva, A.; Lezama-García, K.; José-Pérez, N.; et al. Anthropomorphism and Its Adverse Effects on the Distress and Welfare of Companion Animals. Animals 2021, 11, 3263. [Google Scholar] [CrossRef]
- Phillips, M.L. Understanding the neurobiology of emotion perception: Implications for psychiatry. Br. J. Psychiatry 2003, 182, 190–192. [Google Scholar] [CrossRef]
- Calder, A.J.; Lawrence, A.D.; Young, A.W. Neuropsychology of fear and loathing. Nat. Rev. Neurosci. 2001, 2, 352–363. [Google Scholar] [CrossRef]
- Pertovaara, A. The noradrenergic pain regulation system: A potential target for pain therapy. Eur. J. Pharmacol. 2013, 716, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Reid, K.; Rogers, C.W.; Gronqvist, G.; Gee, E.K.; Bolwell, C.F. Anxiety and pain in horses measured by heart rate variability and behavior. J. Vet. Behav. 2017, 22, 1–6. [Google Scholar] [CrossRef]
- Hu, J.; Qi, S.; Becker, B.; Luo, L.; Gao, S.; Gong, Q.; Hurlemann, R.; Kendrick, K.M. Oxytocin selectively facilitates learning with social feedback and increases activity and functional connectivity in emotional memory and reward processing regions. Hum. Brain Mapp. 2015, 36, 2132–2146. [Google Scholar] [CrossRef] [PubMed]
- Valenchon, M.; Lévy, F.; Moussu, C.; Lansade, L. Stress Affects instrumental learning based on positive or negative reinforcement in interaction with personality in domestic horses. PLoS ONE 2017, 12, e0170783. [Google Scholar] [CrossRef] [PubMed]
- Powell, L.; Guastella, A.J.; McGreevy, P.; Bauman, A.; Edwards, K.M.; Stamatakis, E. The physiological function of oxytocin in humans and its acute response to human-dog interactions: A review of the literature. J. Vet. Behav. 2019, 30, 25–32. [Google Scholar] [CrossRef]
- Alexander, R.; Aragón, O.R.; Bookwala, J.; Cherbuin, N.; Gatt, J.M.; Kahrilas, I.J.; Kästner, N.; Lawrence, A.; Lowe, L.; Morrison, R.G.; et al. The neuroscience of positive emotions and affect: Implications for cultivating happiness and wellbeing. Neurosci. Biobehav. Rev. 2021, 121, 220–249. [Google Scholar] [CrossRef]
- Osella, M.; Odore, R.; Badino, P.; Cuniberti, B.; Bergamasco, L. Plasma dopamine neurophysiological correlates in anxious dogs. In Current Issues and Research in Veterinary Behavioral Medicine; Purdue University Press: West Lafayette, IN, USA, 2005; pp. 274–276. [Google Scholar]
- Karpiński, M.; Ognik, K.; Garbiec, A.; Czyżowski, P.; Krauze, M. Effect of stroking on serotonin, noradrenaline, and cortisol levels in the blood of right- and left-pawed dogs. Animals 2021, 11, 331. [Google Scholar] [CrossRef] [PubMed]
- Siracusa, C. IAggression–Dogs. In Small Animal Veterinary Psychiatry; Denenberg, S., Ed.; CAB International: Oxfordshire, UK, 2021; pp. 191–193. [Google Scholar]
- Bethlehem, R.A.I.; van Honk, J.; Auyeung, B.; Baron-Cohen, S. Oxytocin, brain physiology, and functional connectivity: A review of intranasal oxytocin FMRI studies. Psychoneuroendocrinology 2013, 38, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Domes, G.; Heinrichs, M.; Gläscher, J.; Büchel, C.; Braus, D.F.; Herpertz, S.C. Oxytocin attenuates amygdala responses to emotional faces regardless of valence. Biol. Psychiatry 2007, 62, 1187–1190. [Google Scholar] [CrossRef]
- Petersson, M.; Uvnäs-Moberg, K.; Nilsson, A.; Gustafson, L.L.; Hydbring-Sandberg, E.; Handlin, L. Oxytocin and cortisol levels in dog owners and their dogs are associated with behavioral patterns: An exploratory study. Front. Psychol. 2017, 8, 1976. [Google Scholar] [CrossRef]
- Mitsui, S.; Yamamoto, M.; Nagasawa, M.; Mogi, K.; Kikusui, T.; Ohtani, N.; Ohta, M. Urinary oxytocin as a noninvasive biomarker of positive emotion in dogs. Horm. Behav. 2011, 60, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Lansade, L.; Nowak, R.; Lainé, A.-L.; Leterrier, C.; Bonneau, C.; Parias, C.; Bertin, A. facial expression and oxytocin as possible markers of positive emotions in horses. Sci. Rep. 2018, 8, 14680. [Google Scholar] [CrossRef]
- Ogi, A.; Mariti, C.; Baragli, P.; Sergi, V.; Gazzano, A. Effects of stroking on salivary oxytocin and cortisol in guide dogs: Preliminary results. Animals 2020, 10, 708. [Google Scholar] [CrossRef] [PubMed]
- MacLean, E.L.; Gesquiere, L.R.; Gee, N.R.; Levy, K.; Martin, W.L.; Carter, C.S. Effects of affiliative human-animal interaction on dog salivary and plasma oxytocin and vasopressin. Front. Psychol. 2017, 8, 1606. [Google Scholar] [CrossRef]
- Somppi, S.; Törnqvist, H.; Topál, J.; Koskela, A.; Hänninen, L.; Krause, C.M.; Vainio, O. Nasal oxytocin treatment biases dogs’ visual attention and emotional response toward positive human facial expressions. Front. Psychol. 2017, 8, 1854. [Google Scholar] [CrossRef]
- Mobbs, D. What Can the social emotions of dogs teach us about human emotions? Anim. Sentience 2018, 3, 5. [Google Scholar] [CrossRef]
- Anderson, D.J.; Adolphs, R. A Framework for studying emotions across species. Cell 2014, 157, 187–200. [Google Scholar] [CrossRef]
- Lindsley, D.B. Emotion. In Handbook of Experimental Psychology; Stevens, S.S., Ed.; Wiley: Oxford, UK, 2018; pp. 473–516. [Google Scholar]
- Nummenmaa, L.; Saarimäki, H. Emotions as discrete patterns of systemic activity. Neurosci. Lett. 2019, 693, 3–8. [Google Scholar] [CrossRef]
- Hess, U.; Hareli, S. The role of social context for the interpretation of emotional facial expressions. In Understanding Facial Expressions in Communication; Springer: New Delhi, India, 2015; pp. 119–141. [Google Scholar]
- Kraaijenvanger, E.J.; Hofman, D.; Bos, P.A. A neuroendocrine account of facial mimicry and its dynamic modulation. Neurosci. Biobehav. Rev. 2017, 77, 98–106. [Google Scholar] [CrossRef]
- Brudzynski, S.M. Communication of emotions in animals. In Encyclopedia of Behavioral Neuroscience; Elsevier Science: Amsterdam, The Netherlands, 2010; pp. 302–307. [Google Scholar]
- Nagasawa, M.; Murai, K.; Mogi, K.; Kikusui, T. Dogs can discriminate human smiling faces from blank expressions. Anim. Cogn. 2011, 14, 525–533. [Google Scholar] [CrossRef]
- Albuquerque, N.; Guo, K.; Wilkinson, A.; Resende, B.; Mills, D.S. Mouth-licking by dogs as a response to emotional stimuli. Behav. Proc. 2018, 146, 42–45. [Google Scholar] [CrossRef]
- Karl, S.; Sladky, R.; Lamm, C.; Huber, L. Neural responses of pet dogs witnessing their caregiver´s positive interactions with a conspecific: An fMRI study. Cereb. Cortex Commun. 2021, 2, tgab047. [Google Scholar] [CrossRef]
- Kujala, M.; Somppi, S.; Jokela, M.; Vainio, O.; Parkkonen, L. Human empathy, personality and experience affect the emotion ratings of dog and human facial expressions. PLoS ONE 2017, 12, e0170730. [Google Scholar] [CrossRef]
- Beerda, B.; Schilder, M.B.H.; van Hooff, J.A.R.A.M.; de Vries, H.W. Manifestations of chronic and acute stress in dogs. Appl. Anim. Behav. Sci. 1997, 52, 307–319. [Google Scholar] [CrossRef]
- Caeiro, C.; Guo, K.; Mills, D. Dogs and humans respond to emotionally competent stimuli by producing different facial actions. Sci. Rep. 2017, 7, 15525. [Google Scholar] [CrossRef] [PubMed]
- Meints, K.; Racca, A.; Hickey, N. How to prevent dog bite injuries? Children misinterpret dogs, facial expressions. In Proceedings of the 10th World Conference on Injury Prevention and Safety Promotion, London, UK, 21–24 September 2010; BMJ Press: London, UK, 2010. [Google Scholar]
- Bloom, T.; Trevathan-Minnis, M.; Atlas, N.; MacDonald, D.A.; Friedman, H.L. Identifying facial expressions in dogs: A replication and extension study. Behav. Proc. 2021, 186, 104371. [Google Scholar] [CrossRef]
- Boissy, A.; Dwyer, C.M.; Jones, R.B. Fear and other negative emotions. In Animal Welfare; CAB International: Wallingford, UK, 2011; pp. 92–113. [Google Scholar]
- Flint, H.E.; Coe, J.B.; Pearl, D.L.; Serpell, J.A.; Niel, L. Effect of training for dog fear identification on dog owner ratings of fear in familiar and unfamiliar dogs. Appl. Anim. Behav. Sci. 2018, 208, 66–74. [Google Scholar] [CrossRef]
- Mariti, C.; Gazzano, A.; Moore, J.L.; Baragli, P.; Chelli, L.; Sighieri, C. Perception of dogs´stress their owners. J. Vet. Behav. 2012, 7, 213–219. [Google Scholar] [CrossRef]
- Racca, A.; Amadei, E.; Ligout, S.; Guo, K.; Meints, K.; Mills, D. Discrimination of human and dog faces and inversion responses in domestic dogs (Canis familiaris). Anim. Cogn. 2010, 13, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Racca, A.; Guo, K.; Meints, K.; Mills, D.S. Reading faces: Differential Lateral gaze bias in processing canine and human facial expressions in dogs and 4-year-old children. PLoS ONE 2012, 7, e36076. [Google Scholar] [CrossRef]
- Stetina, B.U.; Turner, K.; Burger, E.; Glenk, L.M.; McElheney, J.C.; Handlos, U.; Kothgassner, O.D. Learning emotion recognition from canines? Two for the road. J. Vet. Behav. 2011, 6, 108–114. [Google Scholar] [CrossRef]
- Ogura, T.; Maki, M.; Nagata, S.; Nakamura, S. Dogs (Canis Familiaris) gaze at our hands: A preliminary eye-tracker experiment on selective attention in dogs. Animals 2020, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Travain, T.; Valsecchi, P. Infrared thermography in the study of animals´emotional responses: A critical review. Animals 2021, 11, 2510. [Google Scholar] [CrossRef] [PubMed]
- Langford, D.J. Social modulation of pain as evidence for empathy in mice. Science 2006, 312, 1967–1970. [Google Scholar] [CrossRef]
- Jeon, D.; Kim, S.; Chetana, M.; Jo, D.; Ruley, H.E.; Lin, S.-Y.; Rabah, D.; Kinet, J.-P.; Shin, H.-S. Observational fear learning involves affective pain system and Cav1.2 Ca2+ channels in ACC. Nat. Neurosci. 2010, 13, 482–488. [Google Scholar] [CrossRef]
- Hess, U.; Fischer, A. Emotional mimicry as social regulation. Personal. Soc. Psychol. Rev. 2013, 17, 142–157. [Google Scholar] [CrossRef] [PubMed]
- Ha, J.C.; Campion, T.L. The emotional animal: Using the science of emotions to interpret behavior. In Dog Behavior; Academic Press: London, UK, 2019; pp. 93–108. [Google Scholar]
- Kurachi, T.; Irimajiri, M.; Mizuta, Y.; Satoh, T. Dogs predisposed to anxiety disorders and related factors in japan. Appl. Anim. Behav. Sci. 2017, 196, 69–75. [Google Scholar] [CrossRef]
- Van Bourg, J.; Patterson, J.E.; Wynne, C.D.L. Pet dogs (Canis Lupus Familiaris) release their trapped and distressed owners: Individual variation and evidence of emotional contagion. PLoS ONE 2020, 15, e0231742. [Google Scholar] [CrossRef] [PubMed]
- Marcet-Rius, M.; Pageat, P.; Bienboire-Frosini, C.; Teruel, E.; Monneret, P.; Leclercq, J.; Lafont-Lecuelle, C.; Cozzi, A. Tail and ear movements as possible indicators of emotions in pigs. Appl. Anim. Behav. Sci. 2018, 205, 14–18. [Google Scholar] [CrossRef]
- Reefmann, N.; Bütikofer Kaszàs, F.; Wechsler, B.; Gygax, L. Ear and tail postures as indicators of emotional valence in sheep. Appl. Anim. Behav. Sci. 2009, 118, 199–207. [Google Scholar] [CrossRef]
- Briefer Freymond, S.; Briefer, E.F.; Zollinger, A.; Gindrat-von Allmen, Y.; Wyss, C.; Bachmann, I. Behaviour of horses in a judgment bias test associated with positive or negative reinforcement. Appl. Anim. Behav. Sci. 2014, 158, 34–45. [Google Scholar] [CrossRef]
- Arena, L.; Wemelsfelder, F.; Messori, S.; Ferri, N.; Barnard, S. Application of free choice profiling to assess the emotional state of dogs housed in shelter environments. Appl. Anim. Behav. Sci. 2017, 195, 72–79. [Google Scholar] [CrossRef]
- Tami, G.; Gallagher, A. Description of the behaviour of domestic dog (Canis Familiaris) by experienced and inexperienced people. Appl. Anim. Behav. Sci. 2009, 120, 159–169. [Google Scholar] [CrossRef]
- Diesel, G.; Brodbelt, D.; Pfeiffer, U.D. Reliability of assessment of dogs´behavioural responses by staff working at a welfare charuty in the UK. Appl. Anim. Behav. Sci. 2008, 115, 171–181. [Google Scholar] [CrossRef]
- Lakestani, N.N.; Donaldson, M.L.; Waran, N. Interpretation of dog behavior by children and young adults. Anthrozoös 2014, 27, 65–80. [Google Scholar] [CrossRef]
- Merola, I.; Prato-Previde, E.; Marshall-Pescini, S. Dogs’ social referencing towards owners and strangers. PLoS ONE 2012, 7, e47653. [Google Scholar] [CrossRef]
- Horowitz, A. Disambiguating the “Guilty Look”: Salient prompts to a familiar dog behaviour. Behav. Proc. 2009, 81, 447–452. [Google Scholar] [CrossRef]
- Kujala, M. Canine Emotions as seen through human social cognition. Anim. Sentience 2017, 2, 1. [Google Scholar] [CrossRef]
- Correia-Caeiro, C.; Guo, K.; Mills, D. Bodily emotional expressions are a primary source of information for dogs, but not for humans. Anim. Cogn. 2021, 24, 267–279. [Google Scholar] [CrossRef]
- Csoltova, E.; Mehinagic, E. Where do we stand in the domestic dog (Canis familiaris) positive-emotion assessment: A state-of-the-art review and future directions. Front. Psychol. 2020, 11, 2131. [Google Scholar] [CrossRef]
- Bremhorst, A.; Bütler, S.; Würbel, H.; Riemer, S. Incentive motivation in pet dogs–Preference for constant vs varied food rewards. Sci. Rep. 2018, 8, 9756. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; d’Ingeo, S.; Quaranta, A. Orienting asymmetries and physiological reactivity in dogs’ response to human emotional faces. Learn. Behav. 2018, 46, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Firnkes, A.; Bartels, A.; Bidoli, E.; Erhard, M. Appeasement signals used by dogs during dog–human communication. J. Vet. Behav. 2017, 19, 35–44. [Google Scholar] [CrossRef]
- Nagasawa, M.; Kawai, E.; Mogi, K.; Kikusui, T. Dogs show left facial lateralization upon reunion with their owners. Behav. Proc. 2013, 98, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; D’Ingeo, S.; Fornelli, S.; Quaranta, A. Are dogs red–green colour blind? R. Soc. Open Sci. 2017, 4, 170869. [Google Scholar] [CrossRef]
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Sluka, K.A.; et al. The revised international association for the study of pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Mota-Rojas, D.; Orihuela, A.; Strappini-Asteggiano, A.; Nelly Cajiao-Pachón, M.; Agüera-Buendía, E.; Mora-Medina, P.; Ghezzi, M.; Alonso-Spilsbury, M. Teaching Animal Welfare in Veterinary Schools in Latin America. Int. J. Vet. Sci. Med. 2018, 6, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Avalos, I.; Mota-Rojas, D.; Mora-Medina, P.; Martínez-Burnes, J.; Casas-Alvarado, A.; Verduzco-Mendoza, A.; Lezama-García, K.; Olmos-Hernandez, A. Review of different methods used for clinical recognition and assessment of pain in dogs and cats. Int. J. Vet. Sci. Med. 2019, 7, 43–54. [Google Scholar] [CrossRef]
- de Grauw, J.C.; van Loon, J.P.A.M. Systematic pain assessment in horses. Vet. J. 2016, 209, 14–22. [Google Scholar] [CrossRef]
- Gleerup, K.B.; Forkman, B.; Lindegaard, C.; Andersen, P.H. An equine pain face. Vet. Anaesth. Anal. 2015, 42, 103–114. [Google Scholar] [CrossRef]
- Hernández-Avalos, I.; Mota Rojas, D.; Mendoza-Flores, J.E.; Casas-Alvarado, A.; Flores-Padilla, K.; Miranda-Cortes, A.E.; Torres-Bernal, F.; Gómez-Prado, J.; Mora-Medina, P. Nociceptive pain and anxiety in equines: Physiological and behavioral alterations. Vet. World 2021, in press. [Google Scholar]
- Dalla Costa, E.; Stucke, D.; Dai, F.; Minero, M.; Leach, M.; Lebelt, D. Using the horse grimace scale (HGS) to assess pain associated with acute laminitis in horses (Equus caballus). Animals 2016, 6, 47. [Google Scholar] [CrossRef]
- Parr, L.A.; Waller, B.M.; Burrows, A.M.; Gothard, K.M.; Vick, S.J. Brief communication: MaqFACS: A muscle-based facial movement coding system for the rhesus macaque. Am. J. Phys. Anthropol. 2010, 143, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Keating, S.C.J.; Thomas, A.A.; Flecknell, P.A.; Leach, M.C. Evaluation of EMLA cream for preventing pain during tattooing of rabbits: Changes in physiological, behavioural and facial expression responses. PLoS ONE 2012, 7, e44437. [Google Scholar] [CrossRef]
- Reijgwart, M.L.; Schoemaker, N.J.; Pascuzzo, R.; Leach, M.C.; Stodel, M.; de Nies, L.; Hendriksen, C.F.M.; van der Meer, M.; Vinke, C.M.; van Zeeland, Y.R.A. The composition and initial evaluation of a grimace scale in ferrets after surgical implantation of a telemetry probe. PLoS ONE 2017, 12, e0187986. [Google Scholar] [CrossRef] [PubMed]
- Camps, T.; Amat, M.; Mariotti, V.M.; Le Brech, S.; Manteca, X. Pain-related aggression in dogs: 12 clinical cases. J. Vet. Behav. 2012, 7, 99–102. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Minero, M.; Lebelt, D.; Stucke, D.; Canali, E.; Leach, M.C. Development of the horse grimace scale (HGS) as a pain assessment tool in horses undergoing routine castration. PLoS ONE 2014, 9, e92281. [Google Scholar] [CrossRef]
- Niella, R.V.; Sena, A.S.; Corrêa, J.M.X.; Soares, P.C.L.R.; Pinto, T.M.; Silva Junior, A.C.; Costa, B.A.; de Oliveira, J.N.S.; da Silva, E.B.; de Lavor, M.S.L. Preemptive effect of amantadine as adjuvant in postoperative analgesia of ovaryhisterectomy in dogs. Res. Soc. Dev. 2020, 9, e68091110128. [Google Scholar] [CrossRef]
- Barletta, M.; Young, C.N.; Quandt, J.E.; Hofmeister, E.H. Agreement between veterinary students and anesthesiologists regarding postoperative pain assessment in dogs. Vet. Anaesth. Analg. 2016, 43, 91–98. [Google Scholar] [CrossRef]
- Kinjavdekar, S.P.; Aithal, A.H.P.; Pawde, A.M.; Malik, V. Comparison of analgesic effects of meloxicam and ketoprofen using university of melbourne pain scale in clinical canine orthopaedic patients. J. Appl. Anim. Res. 2010, 38, 261–264. [Google Scholar] [CrossRef][Green Version]
- Reader, R.C.; McCarthy, R.J.; Schultz, K.L.; Volturo, A.R.; Barton, B.A.; O’Hara, M.J.; Abelson, A.L. Comparison of liposomal bupivacaine and 0.5% bupivacaine hydrochloride for control of postoperative pain in dogs undergoing tibial plateau leveling osteotomy. J. Am. Vet. Med. Assoc. 2020, 256, 1011–1019. [Google Scholar] [CrossRef]
- Brondani, J.T.; Mama, K.R.; Luna, S.P.L.; Wright, B.D.; Niyom, S.; Ambrosio, J.; Vogel, P.R.; Padovani, C.R. Validation of the English Version of the UNESP-Botucatu Multidimensional Composite Pain Scale for Assessing Postoperative Pain in Cats. BMC Vet. Res. 2013, 9, 143. [Google Scholar] [CrossRef]
- Belli, M.; de Oliveira, A.R.; de Lima, M.T.; Trindade, P.H.E.; Steagall, P.V.; Luna, S.P.L. Clinical validation of the Short and Long UNESP-Botucatu Scales for Feline Pain Assessment. PeerJ 2021, 9, e11225. [Google Scholar] [CrossRef] [PubMed]
- Meunier, N.V.; Panti, A.; Mazeri, S.; Fernandes, K.A.; Handel, I.G.; Bronsvoort, B.M.d.C.; Gamble, L.; Mellanby, R.J. Randomised trial of perioperative tramadol for canine sterilisation pain management. Vet. Rec. 2019, 185, 406. [Google Scholar] [CrossRef] [PubMed]
- Reid, J.; Scott, E.M.; Calvo, G.; Nolan, A.M. Definitive Glasgow acute pain scale for cats: Validation and intervention level. Vet. Rec. 2017, 180, 449. [Google Scholar] [CrossRef]
- Steagall, P.V.; Beatriz, M.P. Acute pain in cats recent advances in clinical assessment. J. Feline Med. Surg. 2019, 21, 25–34. [Google Scholar] [CrossRef]
- Mich, P.M.; Hellyer, P.W. Objective, categoric methods for assessing pain and analgesia. In Handbook of Veterinary Pain Management, 2nd ed.; Gaynor, J.S., Muir, W.W., Eds.; Mosby: St. Louis, MO, USA, 2008; pp. 78–109. [Google Scholar]
- Lu, Y.; Mahmoud, M.; Robinson, P. Estimating sheep pain level using facial action unit detection. In Proceedings of the 12th IEEE International Conference on Automatic Face and Gesture Recognition, Cambridge, UK, 30 May–3 June 2017; pp. 394–399. [Google Scholar] [CrossRef]
- Neethirajan, S.; Reimert, I.; Kemp, B. Measuring farm animal emotions—Sensor-based approaches. Sensors 2021, 21, 553. [Google Scholar] [CrossRef]
- Casas-Alvarado, A.; Mota-Rojas, D.; Hernández-Avalos, I.; Mora-Medina, P.; Olmos-Hernández, A.; Verduzco-Mendoza, A.; Reyes-Sotelo, B.; Martínez-Burnes, J. Advances in infrared thermography: Surgical aspects, vascular changes, and pain monitoring in veterinary medicine. J. Therm. Biol. 2020, 92, 102664. [Google Scholar] [CrossRef] [PubMed]
- Mota-Rojas, D.; Pereira, A.; Wang, D.; Martínez-Burnes, J.; Ghezzi, M.; Lendez, P.; Bertoni, A.; Geraldo, A.M. Clinical applications and factors involved in validating thermal windows used in infrared thermography to assess health and productivity. Animals 2021, 11, 2247. [Google Scholar] [CrossRef]
- Reyes-Sotelo, B.; Mota-Rojas, D.; Martínez-Burnes, J.; Gómez, J.; Lezama, K.; González-Lozano, M.; Hernández-Ávalos, I.; Casas, A.; Herrera, Y.; Mora-Medina, P. Tail docking in dogs: Behavioural, physiological and ethical aspects. CAB Rev. 2020, 15, 1–13. [Google Scholar]
- Mota-Rojas, D.; Broom, D.M.; Orihuela, A.; Velarde, A.; Napolitano, F.; Alonso-Spilsbury, M. Effects of human-animal relationship on animal welfare. J. Anim. Behav. Biometeorol. 2020, 8, 196–205. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mota-Rojas, D.; Marcet-Rius, M.; Ogi, A.; Hernández-Ávalos, I.; Mariti, C.; Martínez-Burnes, J.; Mora-Medina, P.; Casas, A.; Domínguez, A.; Reyes, B.; et al. Current Advances in Assessment of Dog’s Emotions, Facial Expressions, and Their Use for Clinical Recognition of Pain. Animals 2021, 11, 3334. https://doi.org/10.3390/ani11113334
Mota-Rojas D, Marcet-Rius M, Ogi A, Hernández-Ávalos I, Mariti C, Martínez-Burnes J, Mora-Medina P, Casas A, Domínguez A, Reyes B, et al. Current Advances in Assessment of Dog’s Emotions, Facial Expressions, and Their Use for Clinical Recognition of Pain. Animals. 2021; 11(11):3334. https://doi.org/10.3390/ani11113334
Chicago/Turabian StyleMota-Rojas, Daniel, Míriam Marcet-Rius, Asahi Ogi, Ismael Hernández-Ávalos, Chiara Mariti, Julio Martínez-Burnes, Patricia Mora-Medina, Alejandro Casas, Adriana Domínguez, Brenda Reyes, and et al. 2021. "Current Advances in Assessment of Dog’s Emotions, Facial Expressions, and Their Use for Clinical Recognition of Pain" Animals 11, no. 11: 3334. https://doi.org/10.3390/ani11113334
APA StyleMota-Rojas, D., Marcet-Rius, M., Ogi, A., Hernández-Ávalos, I., Mariti, C., Martínez-Burnes, J., Mora-Medina, P., Casas, A., Domínguez, A., Reyes, B., & Gazzano, A. (2021). Current Advances in Assessment of Dog’s Emotions, Facial Expressions, and Their Use for Clinical Recognition of Pain. Animals, 11(11), 3334. https://doi.org/10.3390/ani11113334