Efficacy of a Synergistic Blend of Organic Acids and ß-1,4 Mannobiose on Cecal Salmonella Counts and Growth Performance in Salmonella Challenged Broiler Chickens: A Meta-Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Studies
2.2. Study Design
2.3. Parameters
2.4. Statistics
3. Results
3.1. Selection of Studies
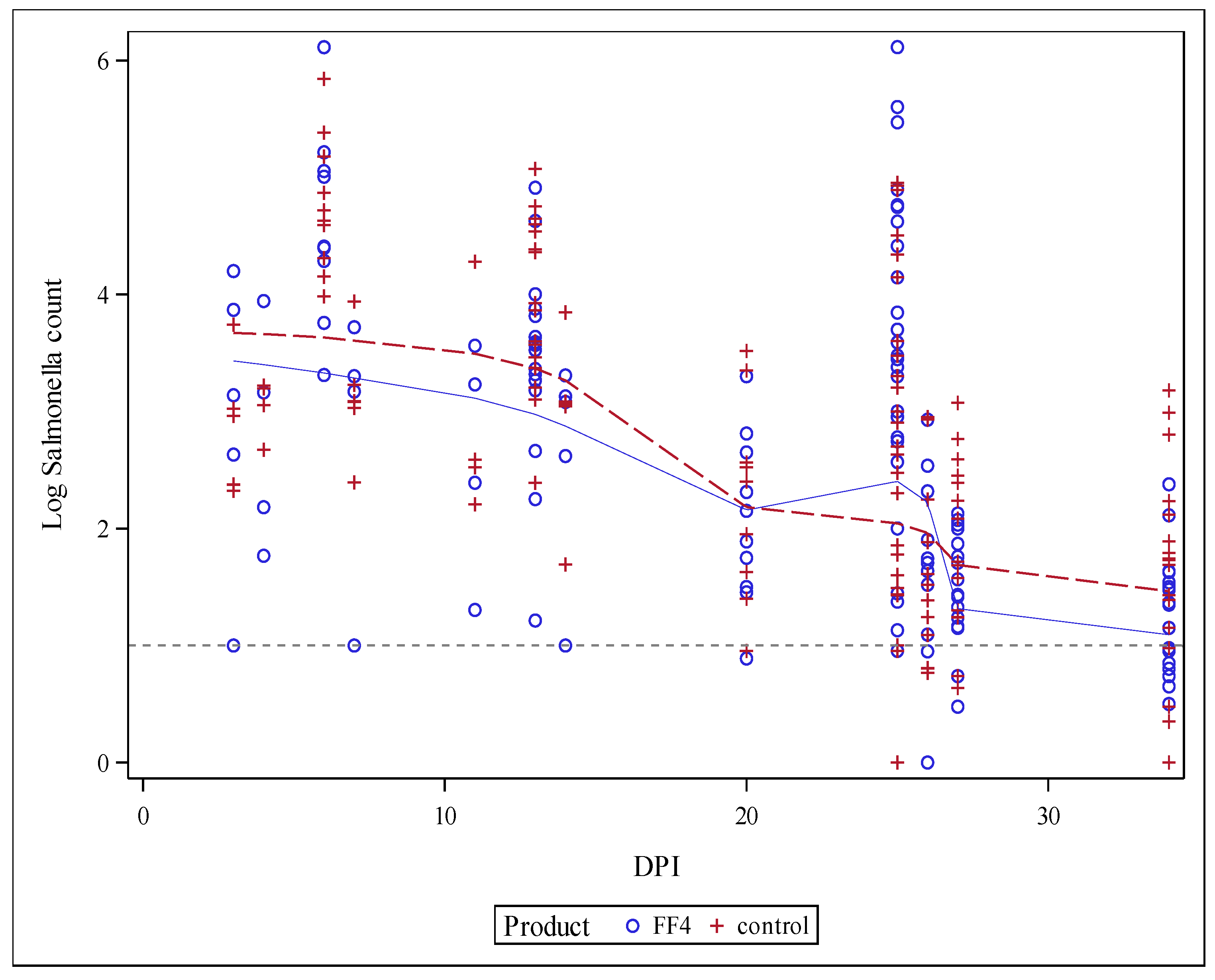
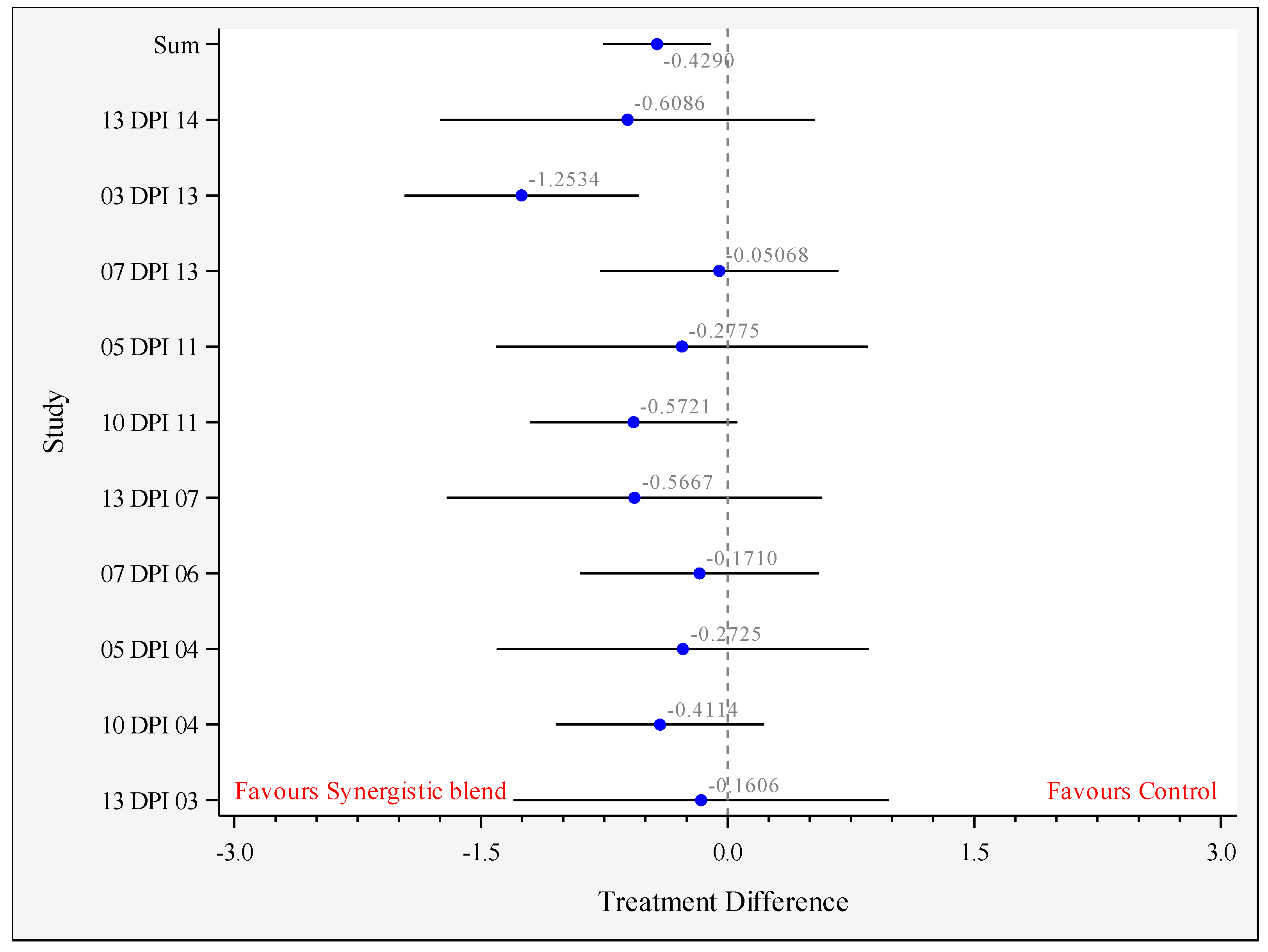
3.2. Cecal Salmonella Counts
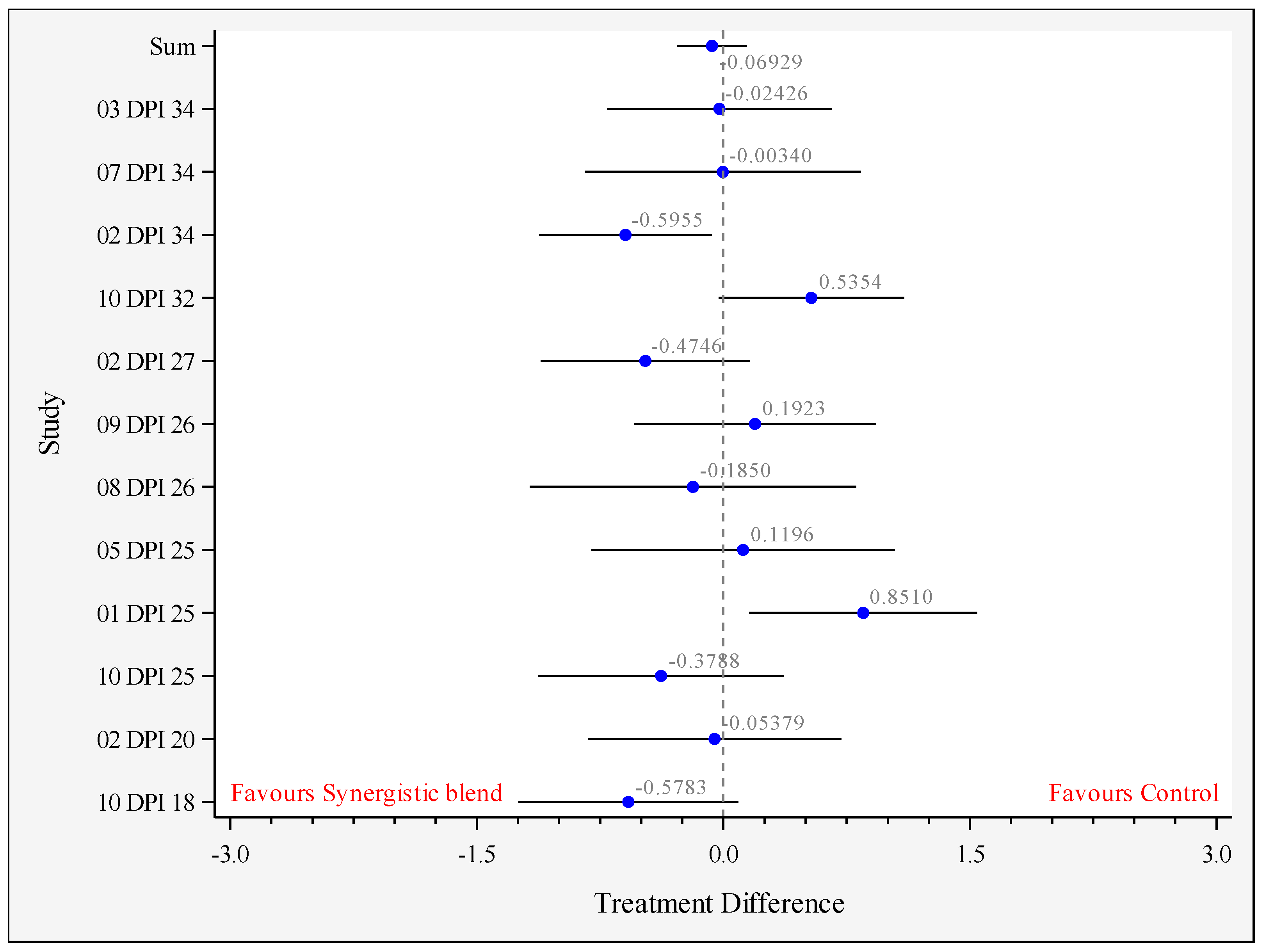
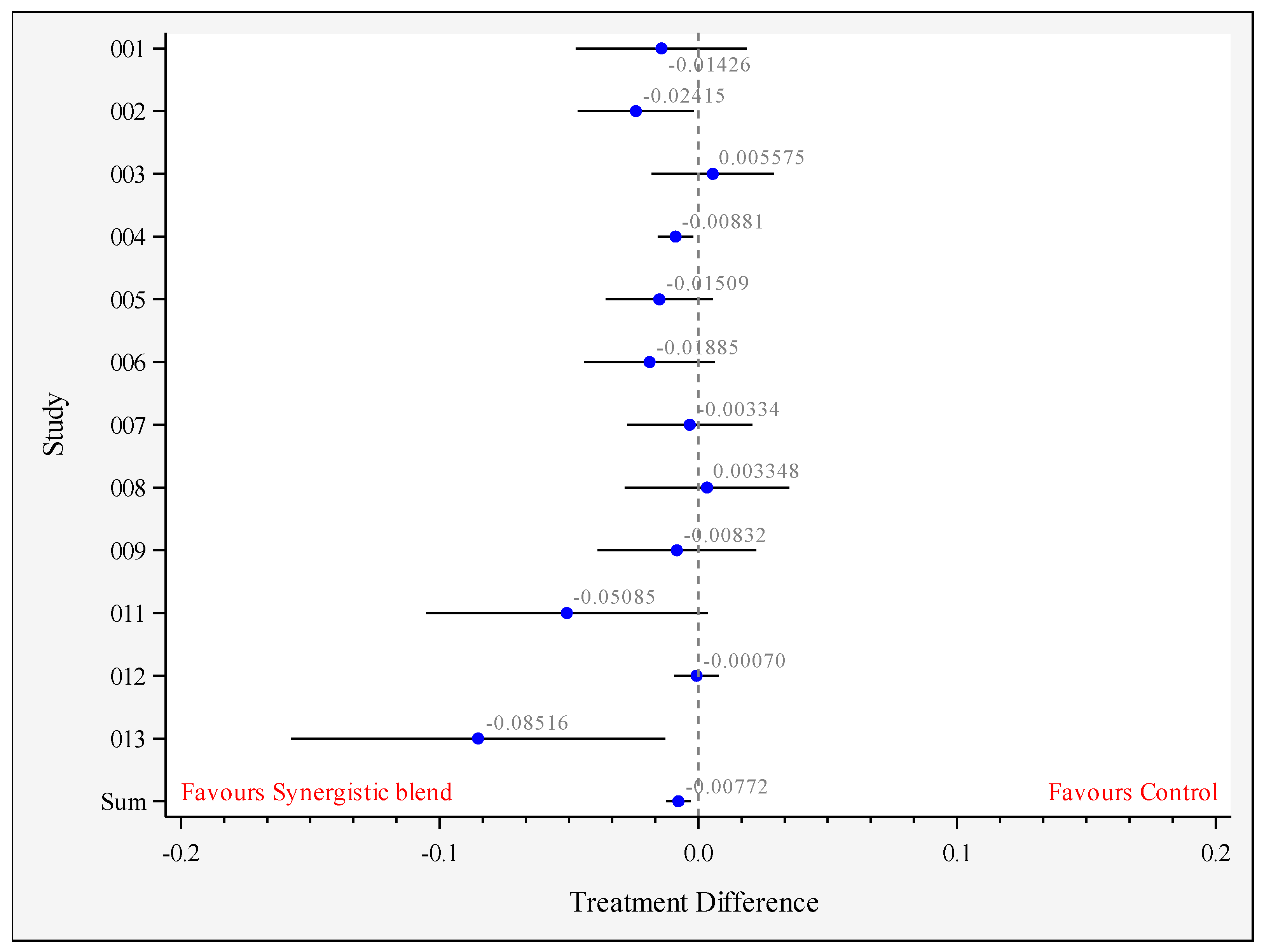
3.3. Growth Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Castro-Vargas, R.E.; Herrera-Sánchez, M.P.; Rodríguez-Hernández, R.; Rondón-Barragán, I.S. Antibiotic resistance in Salmonella spp. Isolated from poultry: A global overview. Vet. World 2020, 13, 2070–2084. [Google Scholar] [CrossRef]
- Eng, S.-K.; Pusparajah, P.; Ab Mutalib, N.-S.; Ser, H.L.; Chan, K.-G.; Lee, L.-H. Salmonella: A review on pathogenesis, epidemiology and antibiotic resistance. Front. Life Sci. 2015, 8, 284–293. [Google Scholar] [CrossRef] [Green Version]
- Hoffman, S.; Devleesschauwer, B.; Aspinall, W.; Cooke, R.; Corrigan, T.; Havelaar, A.; Angulo, F.; Gibb, H.; Kirk, M.; Lake, R.; et al. Attribution of global foodborne disease to specific foods: Findings from a World Health Organisation structured expert elicitation. PLoS ONE 2017, 12, e0183641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, F.T.; Richardson, K.E. Salmonella in commercially manufactured feeds. Poult. Sci. 2004, 83, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Van Immerseel, F.; De Zutter, L.; Houf, K.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R. Strategies to control Salmonella in the broiler production chain. World Poult. Sci. J. 2009, 65, 367–392. [Google Scholar] [CrossRef] [Green Version]
- Chlebicz, A.; Slizewska, K. Campylobacteriosis, salmonellosis, yersiniosis, and listeriosis as zoonotic foodborne diseases: A review. Int. J. Environ. Res. Public Health 2018, 15, 863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nair, D.V.T.; Venkitanarayanan, K.; Johny, A.K. Antibiotic-resistant Salmonella in the food supply and the potential role of antibiotic alternatives for control. Foods 2018, 7, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pulido-Landinez, M. Food Safety—Salmonella update in broilers. Anim. Feed Sci. Technol. 2019, 250, 53–58. [Google Scholar] [CrossRef]
- Van Immerseel, F.; Cauwerts, K.; Devriese, L.A.; Haesebrouck, F.; Ducatelle, R. Feed additives to control Salmonella in poultry. World Poult. Sci. J. 2002, 58, 501–513. [Google Scholar] [CrossRef]
- Li, L. The strategic use of organic acids to improve gut health in poultry. In Proceedings of the 21st Annual Australian Poultry Science Symposium, Sydney, NSW, Australia, 1–3 February 2010; pp. 115–117. [Google Scholar]
- Wales, A.D.; Allen, V.M.; Davies, R.H. Chemical treatment of animal feed and water for the control of Salmonella. Foodborne Pathog. Dis. 2010, 7, 3–15. [Google Scholar] [CrossRef] [Green Version]
- Van Immerseel, F.; Boyen, F.; Gantois, I.; Timbermont, L.; Bohez, L.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R. Supplementation of coated butyric acid in the feed reduces colonization and shedding of Salmonella in poultry. Poult. Sci. 2005, 84, 1851–1856. [Google Scholar] [CrossRef]
- Elnesr, S.S.; Alagawany, M.; Elwan, H.A.M.; Fathi, M.A.; Farag, M.R. Effect of sodium butyrate on intestinal health of poultry—Review. Ann. Anim. Sci. 2020, 20, 29–41. [Google Scholar] [CrossRef] [Green Version]
- Panda, A.K.; Rama Rao, S.V.; Raju, M.V.L.N.; Shyam Sunder, G. Effect of butyric acid on performance, gastrointestinal tract health and carcass characteristics in broiler chickens. Asian-Australas. J. Anim. Sci. 2009, 22, 1026–1031. [Google Scholar] [CrossRef]
- Ibuki, M.; Kovacs-Nolan, J.; Fukui, K.; Kanatani, H.; Mine, Y. ß 1-4 mannobiose enhances Salmonella-killing activity and activates innate immune responses in chicken macrophages. Vet. Immunol. Immunopathol. 2011, 139, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Agunos, A.; Ibuki, M.; Yokomizo, F. Effect of dietary ß 1-4 mannobiose in the prevention of Salmonella enteritidis infection in broilers. Br. Poult. Sci. 2007, 48, 331–341. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Nutrient Requirements of Poultry, 9th ed.; The National Academies Press: Washington, DC, USA, 1994. [Google Scholar]
- Andino, A.; Hanning, I. Salmonella enterica: Survival, colonization, and virulence differences among serovars. Sci. World J. 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stern, N.J. Salmonella species and Campylobacter jejuni cecal colonization model in broilers. Poult. Sci. 2008, 87, 2399–2403. [Google Scholar] [CrossRef]
- EFSA BIOHAZ Panel (EFSA Panel on Biological Hazards); Kousoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Chemaly, M.; De Cesare, A.; Herman, L.; Hilbert, F.; et al. Scientific opinion on the Salmonella control in poultry flocks and its public health impact. EFSA J. 2019, 17, 5596. [Google Scholar]
- EFSA and ECDC (European Food Safety Authority and European Centre for Disease Prevention and Control). The European Union One Health 2018 Zoonosis report. EFSA J. 2019, 17, 5926. [Google Scholar]
- Wang, X.; Biswas, S.; Paudyal, N.; Pan, H.; Li, X.; Fang, W.; Yue, M. Antibiotic resistance in Salmonella typhimurium isolates recovered from the food chain through national antimicrobial resistance monitoring system between 1996 and 2016. Front. Microbiol. 2019, 10, 985. [Google Scholar] [CrossRef] [Green Version]
- Van Immerseel, F.; Russell, J.B.; Flythe, M.D.; Gantois, I.; Timbermont, L.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R. The use of organic acids to combat Salmonella in poultry: A mechanistic explanation of the efficacy. Avian Pathol. 2006, 35, 182–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polycarpo, G.V.; Andretta, I.; Kipper, M.; Cruz-Polycarpo, V.C.; Dadalt, J.C.; Rodrigues, P.H.M.; Albuquerque, R. Meta-analytic study of organic acids as an alternative performance-enhancing feed additive to antibiotics for broiler chickens. Poult. Sci. 2017, 96, 3645–3653. [Google Scholar] [CrossRef] [PubMed]
- Ibuki, M.; Kovacs-Nolan, J.; Fukui, K.; Kanatani, H.; Mine, Y. Analysis of gut immune-modulating activity of ß-1,4-mannobiose using microarray and real-time reverse transcription polymerase chain reaction. Poult. Sci. 2010, 89, 1894–1904. [Google Scholar] [CrossRef] [PubMed]
- Ibuki, M.; Fukui, K.; Yamauchi, K. Effect of dietary mannanase-hydrolysed copra meal on growth performance and intestinal histology in broiler chickens. J. Anim. Physiol. Anim. Nutr. 2014, 98, 636–642. [Google Scholar] [CrossRef] [PubMed]
- Ibuki, M.; Yoshimoto, Y.; Inui, M.; Fukui, K.; Yonemoto, H.; Saneyasu, T.; Honda, K.; Kamisoyama, H. Dietary mannanase-hydrolyzed copra meal improves growth and increases muscle weights in growing broiler chickens. Anim. Sci. J. 2014, 85, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Van Immerseel, F.; Fievez, V.; de Buck, J.; Pasmans, F.; Martel, A.; Haesebrouck, F.; Ductatelle, R. Microencapsulated short-chain fatty acids in feed modify colonization and invasion early after infection with Salmonella enteritidis in young chickens. Poult. Sci. 2004, 83, 69–74. [Google Scholar] [CrossRef]
- Fernández-Rubio, C.; Ordóñez, C.; Abad-González, J.; Garcia-Gallego, A.; Pilar Honrubia, M.; Jose Mallo, J.; Balaña-Fouce, R. Butyric acid-based feed additives help protect broiler chickens from Salmonella Enteritidis infection. Poult. Sci. 2009, 88, 943–948. [Google Scholar] [CrossRef]
- Aljumaah, M.R.; Alkhulaifi, M.M.; Abudabos, A.M.; Alabdullatifb, A.; El-Mubarak, A.H.; Al Suliman, A.R.; Stanley, D. Organic acid blend supplementation increases butyrate and acetate production in Salmonella enterica serovar Typhimurium challenged broilers. PLoS ONE 2020, 15, e0232831. [Google Scholar] [CrossRef]
- Abudabos, A.M.; Al-Mufarrey, S.I.; Alyemni, A.H.; Yehia, H.M.; Garelnabi, A.R.; Alotybi, M.N. Effect of using organic acids to substitute antimicrobial growth promoters on broiler chickens performance. J. Food Agric. Environ. 2014, 12, 447–451. [Google Scholar]
- Drenth, P.J.D. The European code of conduct for research integrity. In Promoting Research Integrity in a Global Environment; World Scientific: Singapore, 2012. [Google Scholar]
| Study Number | Facility | Country | Study Duration (Days) | Model | Salmonella Serovar | Day of Inoculation | Included Parameters 1 |
|---|---|---|---|---|---|---|---|
| 01 | Trouw Nutrition Poultry Research Centre | Spain | 32 | Infection | Enteritidis | 7 | Growth performance, 15–34 DPI |
| 02 | Trouw Nutrition Poultry Research Centre | Spain | 42 | Infection | Typhimurium | 8 | Growth performance, 15–34 DPI |
| 03 | Trouw Nutrition Poultry Research Centre | Spain | 35 | Infection | Enteritidis | 8 | Growth performance, 0–14 DPI, 15–34 DPI |
| 04 | Trouw Nutrition Poultry Research Centre | Spain | 42 | Seeder | Enteritidis | 8 | Growth performance |
| 05 | Trouw Nutrition Poultry Research Centre | Spain | 32 | Infection | Enteritidis | 7 | Growth performance, 0–14 DPI, 15–34 DPI |
| 06 | Trouw Nutrition Poultry Research Centre | Spain | 39 | Seeder | Enteritidis | 7 | Growth performance |
| 07 | Trouw Nutrition Poultry Research Centre | Spain | 42 | Infection | Enteritidis | 8 | Growth performance, 0–14 DPI, 15–34 DPI |
| 08 | Trouw Nutrition Poultry Research Centre | Spain | 33 | Infection | Enteritidis | 7 | Growth performance, 15–34 DPI |
| 09 | Trouw Nutrition Poultry Research Centre | Spain | 33 | Infection | Typhimurium | 7 | Growth performance, 15–34 DPI |
| 10 | Mercolab | Brazil | 35 | Seeder | Heidelberg | 3 | 0–14 DPI, 15–34 DPI |
| 11 | USP | Brazil | 42 | Infection | Heidelberg | 10 | Growth performance |
| 12 | IRTA | Spain | 35 | Seeder | Enteritidis | 1 | Growth performance |
| 13 | Chulalongkorn | Thailand | 35 | Infection | Typhimurium | 7 | Growth performance, 0–14 DPI |
| Outcome | Salmonella 0–14 DPI 1 (Log CFU/g) | Salmonella 15–34 DPI 2 (Log CFU/g) |
|---|---|---|
| Mean-Control | 3.097 a | 1.605 |
| Mean-Synergistic blend | 2.669 b | 1.536 |
| Difference of the means | −0.429 | −0.069 |
| Standard error of the difference | 0.164 | 0.107 |
| Lower 95% confidence limit | −0.757 | −0.281 |
| Upper 95% confidence limit | −0.101 | 0.142 |
| p-value | 0.011 | 0.519 |
| Outcome | Final BW 1 (g) | ADG 2 (g/Bird/Day) | ADFI 3 (g/Bird/Day) | FCR (g:g) 4 |
|---|---|---|---|---|
| Mean-Control | 2304.5 | 62.31 | 92.36 | 1.482 a |
| Mean-Synergistic blend | 2304.8 | 62.50 | 91.97 | 1.474 b |
| Difference between means | 0.28 | 0.188 | −0.386 | −0.0077 |
| Standard error of the difference | 18.64 | 0.291 | 0.396 | 0.0023 |
| Lower 95% confidence limit | −36.45 | −0.388 | −1.179 | −0.0124 |
| Upper 95% confidence limit | 37.01 | 0.764 | 0.407 | −0.0030 |
| p-value | 0.988 | 0.518 | 0.334 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Kuijk, S.J.A.; Han, Y. Efficacy of a Synergistic Blend of Organic Acids and ß-1,4 Mannobiose on Cecal Salmonella Counts and Growth Performance in Salmonella Challenged Broiler Chickens: A Meta-Analysis. Animals 2021, 11, 2988. https://doi.org/10.3390/ani11102988
van Kuijk SJA, Han Y. Efficacy of a Synergistic Blend of Organic Acids and ß-1,4 Mannobiose on Cecal Salmonella Counts and Growth Performance in Salmonella Challenged Broiler Chickens: A Meta-Analysis. Animals. 2021; 11(10):2988. https://doi.org/10.3390/ani11102988
Chicago/Turabian Stylevan Kuijk, Sandra J. A., and Yanming Han. 2021. "Efficacy of a Synergistic Blend of Organic Acids and ß-1,4 Mannobiose on Cecal Salmonella Counts and Growth Performance in Salmonella Challenged Broiler Chickens: A Meta-Analysis" Animals 11, no. 10: 2988. https://doi.org/10.3390/ani11102988
APA Stylevan Kuijk, S. J. A., & Han, Y. (2021). Efficacy of a Synergistic Blend of Organic Acids and ß-1,4 Mannobiose on Cecal Salmonella Counts and Growth Performance in Salmonella Challenged Broiler Chickens: A Meta-Analysis. Animals, 11(10), 2988. https://doi.org/10.3390/ani11102988






