Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues
Abstract
Simple Summary
Abstract
1. Introduction
2. Issues on the Regulation of Drug Residues in Food of Animal Origin
2.1. How Veterinary Drug Residues Are Regulated
2.2. Acute Reference Dose Instead of Acceptable Daily Intake
2.3. Metabolite to Parent Drug Ratio
2.4. Lack of Global Harmonization: MRLs/Tolerances and Withdrawal Periods
2.5. Extra-Label Use: When Can It Be Allowed?
3. Negative Impact of the Presence of Residues on Edible Animal Tissues
3.1. Economic Impact
3.2. Human Health Impact
3.3. Environmental Health Impact
4. Relevance of Monitoring Drug Residue Programmes
4.1. Official Monitoring Drug Residue Programmes
4.2. Unofficial Drug Residue Studies: From Residue Quantification and Exposure Assessment to Detailed Risk Characterization
4.3. Residues in Food above the Acceptable Levels: Possible Causes
5. Factors Affecting the Drug Tissue Residue Profiles
5.1. Drug Treatment-Related Factors
5.2. Animal Host-Related Factors
5.3. Impact of Cooking on the Drug Residue Levels
6. Antimicrobial Use in Food-Producing Animals: Risk of Residues in Food
6.1. Trends in the Use of Antibacterial Agents in Livestock
| Molecule | Matrix | Processing Methods | Mean drug Reductions (−)/Increments (+) Obtained after Cooking | Stability | References |
|---|---|---|---|---|---|
| Oxytetracycline (OTC) | Chicken thigh, chest, liver, and meat | Boiling (5 m–100 °C) Microwaving (3 m–900 W) Roasting (30 m–200 °C) Grilling (2.5 m–8 kW) | −56% −70% −63% −25% | OTC residues can be significantly reduced by heat treatments | [166] |
| Chicken muscle | Boling (45 m–80 °C) | −47% | Cooking does not guarantee full breakdown of OTC | [162] | |
| Chicken muscle | Boiling Grilling Frying | −85% −97% −94% | Cooking methods have positive effects on OTC residues in total and partial degradation | [172] | |
| Bird muscle and liver | Boiling (30 m–100 °C) Microwaving (3 m–full power) Roasting (30 m–200 °C) | −61 −80 −71 | Cooking methods can generally reduce OTC con centration in meat | [185] | |
| Doxicycline (DOC) | Chicken muscle, liver and gizzard | Boiling (9/24/85 m–100 °C) Roasting (25/40/60 m–200 °C) Microwaving (3 m–full power) | −79% 75% −88% | Cooking processes do not guarantee full breakdown of these drugs | [163] |
| Chicken thigh and breast | Boiling (20 m/30 m/40 m–100 °C) Microwaving (10 m/15 m/20 m–full power) Roasting (40/60/80 m–180 °C) | −28% −33% −28% | DOC is an unstable drug that will be degraded during cooking | [186] | |
| Pig muscle | Boiling (3/6/9 m–100 °C) Deep-frying (3/6/9 m–170 °C) Microwaving (0.5/0.75/1 m-full power) | −36% −36% −28% | DOC residues are significantly affected by cooking | [157] | |
| Egg | Boiling (0.5/2/4/6/8 m–100 °C) Frying (0.5/2/4/6 m–180 °C) Microwaving (0.5/1/2/4 m–full power) | −21% −31% −38% | Ordinary cooking does not eliminate all DOC residues present in eggs | [164] | |
| Tetracycline (TC) Chlortetracycline (CTC) | Chicken breast and thigh | Boiling (20 m/30 m/40 m–100 °C) Microwaving (10 m/15 m/20 m– full power) Roasting (40/60/80 m–180 °C) | TC: −52% CTC: −46% TC: −61% CTC: −60% TC: −69% CTC: −62% | TC and CTC are unstable drugs that will be degraded during cooking | [186] |
| Pig muscle | Boiling (3/6/9 m–100 °C) Deep-frying (3/6/9 m–170 °C) Microwaving (0.5/0.75/1 m–full power) | TC: −44% CTC: −55% TC: −43% CTC: −55% TC: −32% CTC: −40% | TC and CTC residues are significantly affected by cooking | [157] | |
| Eggs | Boiling (5/10/15 m–100 °C) Frying (1/3/5 m–160 °C) | CTC: −41% CTC: −80% | CTC residues were highly sensitive to boiling or frying | [187] | |
| Sulfamethazine | Chicken muscle | Deep frying (3/6/9 m–170/180/190 °C) | −27% | Deep-frying ensures safety of sulfamethazine residues consumption in food | [160] |
| Piglet muscle | Boiling (5/10/15 m–100 °C) Microwaving (0.5/1/1.5 m–full power) Autoclaving (10/15/20 m–121 °C) | −16% −19% −30% | Heat-treatments do not guarantee full removal of sulfamethazine residues | [165] | |
| Sulfadiazine (SDZ) Sulfamethoxazole (SMX) Sulfaquinoxaline (SQ) | Chicken muscle | Deep frying (3/6/9 m–170/180/190 °C) | SDZ: −37% SMX: −40% SQ: −27% | Deep frying ensures the safety of SDZ, SMX, and SQ residue consumption in food | [160] |
| Chicken muscle | Roasting (3/6/9/12 m-170 °C) Microwaving (0.25/0.5/0.75/1 m-full power) Boiling (3/6/9/12 m-100 °C) | SDZ: −3% SMX: −21% SQ: −24% SDZ: −28% SMX: −27% SQ: −34% SDZ: −53% SMX: −44% SQ: −39% | Cooking methods reduce SDZ, SMX, and SQ residues in chicken muscle effectively | [188] | |
| Sulfanilamide | Eggs | Boiling (5/10/15 m-100 °C) Frying (1/3/5 m–160 °C) | −66% −78% | Sulfanilamide residues were highly sensitive to boiling or frying | [187] |
| Tylosin | Chicken muscle | Boiling (10/20/30 m-100 °C) Microwaving (1/1.5/2 m-full power) | −75% −20% | Exposure to tylosin residues may be reduced with a suitable cooking method | [171] |
| Timicosin | Chicken muscle | Boiling (30 m-100 °C) Microwaving (15 m-900 W) Frying (10 m–200 °C) | −36% −74% −46% | Sufficient heating temperature and time can reduce nearly 50% of tilmicosin residues | [189] |
| Enrofloxacin (EFX) | Chicken muscle | Microwaving (3.5 m–full power) Roasting (10 m–200 °C) Boiling (10 m–100 °C) Grilling (10m) Frying (10 m) | −58% +92% −52% +59% −41% | Cooking procedures did not affect quinolone residual levels | [167] |
| Chicken thigh, chest, liver, and muscle | Boiling (5 m–100 °C) Microwaving (3 m–900 W) Roasting (30 m–200 °C) Grilling (2.5 m–8 kW) | −60% −52% −64% −34% | EFX residues can be significantly reduced by the application of heat treatments | [166] | |
| Ciprofloxacin (CFX) | Chicken meat | Boiling (5/10 m–100 °C) Deep frying (3/6 m–170 °C) Microwaving (1/2 m–full power) | −29% −34% −52% | Cooking processes can cause a significant decrease in the level of CFX in meat | [170] |
| Chicken thigh, chest, liver, and muscle | Boiling (5 m–100 °C) Microwaving (3 m–900 W) Roasting (30 m–200 °C) Grilling (2.5 m–8 kW) | −60% −52% −62% −25% | CFX residues can be significantly reduced by the application of heat treatments | [166] | |
| Gentamicin | Chicken muscle | Boiling (30 m–100 °C) Microwaving (15 m–900 W) Frying (10 m–200 °C) | −36% −50% −56% | Sufficient heating temperature and time can reduce nearly 50% of gentamicin residues | [189] |
| Egg | Boiling (1.5/5 m–100 °C) Frying | 0% −10% | Gentamicin residue levels in eggs were not reduced by different cooking procedures | [190] | |
| Ampicillin | Chicken muscle | BoilingGrilling Frying | −81%, −94% −90% | Application of different cooking methods has a positive effect on degrading ampicillin residues | [172] |
| Amoxicillin | Egg | Boiling (5/30/45 m–100 °C) Microwaving (0.5/1/1.5 m–full power) Omelette making (1/2/3 m–130 °C) | −49% −61% −75% | AMX reduction was observed during the cooking procedures | [168] |
| Nitrofurans: 3-amino-2-oxazolidinone (AOZ) 3-amino-5-morpholinomethyl-2-oxazolidone (AMOZ) 1-aminohydantoin (AHD) Semicarbazide (SEM) | Pig muscle and liver | Frying (5/6 min–medium heat) Grilling (8 m–medium heat) Roasting (20 m–170 °C) Microwaving (2.5 m–800 W) | AOZ: −21% AMOZ: −11% AHD: −17% SEM: −6% AOZ: −10% AMOZ: −7% AHD: +4% SEM: −14% AOZ: −22% AMOZ: −20% AHD: −11% SEM: −17% AOZ: −15% AMOZ: −14% AHD: +1% SEM: −13% | The various stability data presented here demonstrate that AOZ, AMOZ, AHD, and SEM show remarkable chemical stability. They are resistant to conventional domestic cooking procedures | [191] |
| Ivermectin | Beef muscle | Boiling (9 m–100 °C) Frying (10/13/16 m-177/192 °C) | −45% −48% | Ordinary cooking procedures appear to give an additional safety margin in the exposure to ivermectin residues | [175] |
| Pig muscle and liver Cattle muscle | Boiling (20 m–100 °C) Microwaving (full power) Frying | +10% −5% −2% | Ivermectin was found to be stable to the effects of cooking | [173] | |
| Cattle muscle and liver | Roasting (40 m–190 °C) Frying (12/15 m-high/medium heat) | 0% −18% | Ivermectin residues in food are resistant to degradation under conventional cooking | [174] | |
| Levamisole | Beef and pork muscle | Boiling (40 m–100 °C) Roasting (45 m–180 °C) Grilling (9 m-medium heat) Frying (23.5 m) Microwaving (4.5 m–full power) | −6% 0% −11% +13% −7% | Levamisole was stable under normal cooking conditions | [192] |
| Cattle muscle and liver | Roasting (40 m–90 °C) Frying (12/15 m–high/medium heat) | +1% −26% | Levamisole residues in food are resistant to degradation under conventional cooking | [174] | |
| Oxfendazole (OFX) Fenbendazole (FEN) | Cattle muscle and liver | Frying Braising (18 m–low heat) Microwaving (full power) | OFX: −77% FEN: +112% OFX: −42% FEN: +10% OFX: −36% FEN: −54% | Oxfendazole concentrations in raw tissue may not be directly applicable for use in consumer exposure | [176] |
| Cattle muscle and liver | Roasting (40 m–190 °C) Frying (12/15 m–high/medium heat) | FEN: −5% FEN: −4% | FEN residues are resistant to degradation under conventional cooking | [174] |
6.2. Residues in Food and Antimicrobial Resistance (AMR)
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Falowo, A.B.; Akimoladun, O.F. Veterinary drug residues in meat and meat products: Occurrence, detection and implications. In Veterinary Medicine and Pharmaceuticals; Oppong Bekoe, S., Saravanan, M., Adosraku, R.K., Ramkumar, P.K., Eds.; IntechOpen: London, UK, 2020; Volume 3, pp. 194, 154–196. ISBN 9781789854404. [Google Scholar]
- Henley, P. COVID-19 and One Health: Shifting the paradigm in how we think about health. JBI Evid. Synth. 2020, 18, 1154–1155. [Google Scholar] [CrossRef]
- Deplazes, P.; Eckert, J.; Mathis, A.; Von Samson-Himmelstjerna, G.; Zahner, H. Parasitology in Veterinary Medicine; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; p. 650. ISBN 9789086862740. [Google Scholar]
- Mehlhorn, H. Encyclopedia of Parasitology, 3rd ed.; Springer Nature: New York, NY, USA, 2008; Volume 2, ISBN 978-3-540-48994-8. [Google Scholar]
- Selzer, P.M.; Epe, C. Antiparasitics in Animal Health: Quo Vadis? Trends Parasitol. 2021, 37, 77–87. [Google Scholar] [CrossRef]
- Daeseleire, E.; Van Pamel, E.; Van Poucke, C.; Croubels, S. Veterinary drug residues in foods. Chem. Contam. Residues Food 2017, 117–153. [Google Scholar] [CrossRef]
- Patel, T.; Marmulak, T.; Gehring, R.; Pitesky, M.; Clapham, M.O.; Tell, L.A. Drug residues in poultry meat: A literature review of commonly used veterinary antibacterials and anthelmintics used in poultry. J. Vet. Pharmacol. Ther. 2018, 41, 761–789. [Google Scholar] [CrossRef] [PubMed]
- Moyo, B.; Tavengwa, N.T. Modern extraction and cleanup methods of veterinary drug residues in food samples of animal Origin. In Recent Advances in Analytical Chemistry, 1st ed.; Ince, M., Ince, O.K., Eds.; IntechOpen: London, UK, 2019; pp. 23–44. ISBN 978-1-78985-810-5. [Google Scholar]
- Flynn, K.; Pérez Villarreal, B.; Barranco, A.; Belca, N.; Björnsdóttira, B.; Fusco, V.; Rainieri, S.; Smaradóttir, S.E.; Smeu, I.; Teixeira, P.; et al. An introduction to current food safety needs. Trends Food Sci. Technol. 2019, 84, 1–3. [Google Scholar] [CrossRef]
- European Commission. Farm to Fork Strategy. Available online: https://ec.europa.eu/food/system/files/2020-05/f2f_action-plan_2020_strategy-info_en.pdf (accessed on 5 June 2021).
- Joint FAO/WHO Expert Committee on Food Additives. Guidance Document for the Establishment of Acute Reference Dose (ARfD) for Veterinary Drug Residues in Food. Available online: https://www.who.int/foodsafety/chem/jecfa/Guidance-document-ARfD-2017.pdf?ua=1 (accessed on 5 June 2021).
- Codex Alimentarius. Section I. Basic Texts and Definitions. 2021. Available online: http://www.fao.org/fao-who-codexalimentarius/codex-texts/procedural-manual/sections/section1/section1-4/en/ (accessed on 15 June 2021).
- Pulce, C.; Lamaison, D.; Keck, G.; Bostvironnois, C.; Nicolas, J.; Descotes, J. Collective human food poisonings by clenbuterol residues in veal liver. Vet. Hum. Toxicol. 1991, 33, 480–481. [Google Scholar]
- Salleras, L.; Dominguez, A.; Mata, E.; Taberner, J.L.; Moro, I.; Sala, P. Epidemiologic study of an outbreak of an outbreak of clenbuterol poisoning in Catalonia, Spain. Public Health Rep. 1995, 110, 338–342. [Google Scholar] [PubMed]
- Sporano, V.; Grasso, L.; Esposito, M.; Oliviero, G.; Brambilla, G.; Loizzo, A. Clenbuterol residues in non-liver containing meat as a cause of collective food poisoning. Vet. Hum. Toxicol. 1998, 40, 141–143. [Google Scholar]
- Barbosa, J.; Cruz, C.; Martins, J.; Silva, J.M.; Neves, C.; Alves, C.; Ramos, F.; Noronha Da Silveira, M.I. Food poisoning by clenbuterol in Portugal. Food Addit. Contam. 2005, 22, 563–566. [Google Scholar] [CrossRef]
- Reeves, P.T. Residues of veterinary drugs at injection sites. J. Vet. Pharmacol. Ther. 2007, 30, 1–17. [Google Scholar] [CrossRef]
- Sanquer, A.; Wackowiez, G.; Havrileck, B. Qualitative assessment of human exposure to consumption of injection site residues. J. Vet. Pharmacol. Ther. 2006, 29, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Sanquer, A.; Wackowiez, G.; Havrileck, B. Critical review on the withdrawal period calculation for injection site residues. J. Vet. Pharmacol. Ther. 2006, 29, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Solecki, R.; Davies, L.; Dellarco, V.; Dewhurst, I.; Van Raaij, M.; Tritscher, A. Guidance on setting of acute reference dose (ARfD) for pesticides. Food Chem. Toxicol. 2005, 43, 1569–1593. [Google Scholar] [CrossRef]
- Joint Meeting of the FAO Panel of Experts on Pesticide Residues in Food and the Environment and the WHO Core Assessment Group on Pesticide Residues. Pesticide Residues in Food. 2004. Available online: http://www.fao.org/3/y5764e/y5764e00.htm (accessed on 15 June 2021).
- European Commission. Guidance for the Setting of an Acute Reference Dose (ARfD). 2001. Available online: https://ec.europa.eu/food/system/files/2016-10/pesticides_ppp_app-proc_guide_tox_acute-ref-dose.pdf (accessed on 5 June 2021).
- Organisation for Economic Co-operation and Development (OECD). Inter-Organization Programme for the Sound Management of Chemicals. Available online: https://www.oecd.org/chemicalsafety/inter-organization-programme-management-of-chemicals.htm (accessed on 25 May 2021).
- European Medicines Agency. VICH GL54: Studies to Evaluate the Safety of Residues of Veterinary Drugs in Human Food: General Approach to Establish an Acute Reference Dose (ARfD). Available online: https://www.ema.europa.eu/en/vich-gl54-studies-evaluate-safety-residues-veterinary-drugs-human-food-general-approach-establish (accessed on 5 June 2021).
- Joint FAO/WHO Expert Committee on Food Additives. Evaluation of Certain Veterinary Drug Residues in Food. Available online: https://apps.who.int/iris/bitstream/handle/10665/259895/9789241210171-eng.pdf?sequence=1&isAllowed=y (accessed on 5 June 2021).
- Kissell, L.W.; Leavens, T.L.; Baynes, R.E.; Riviere, J.E.; Smith, G.W. Comparison of Pharmacokinetics and Milk Elimination of Flunixin in Healthy Cows and Cows with Mastitis. J. Am. Vet. Med. Assoc. 2015, 246, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Shelver, W.L.; Smith, D.J.; Tell, L.A.; Baynes, R.E.; Schroeder, J.W.; Riviere, J.E. Screening and confirmatory analyses of flunixin in tissues and bodily fluids after intravenous or intramuscular administration to cull dairy cows with or without lipopolysaccharide challenge. J. Agric. Food Chem. 2016, 64, 336–345. [Google Scholar] [CrossRef]
- Lin, Z.; Vahl, C.I.; Riviere, J.E. Human food safety implications of variation in food animal drug metabolism. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, A. Veterinary Drug MRLs—A Global Perspective. Bryant Christie Inc. 2017. Available online: https://www.bryantchristie.com/Portals/0/Veterinary%20Drug%20MRLs%20White%20Paper_Oct2017.pdf (accessed on 25 May 2021).
- Gehring, R.; Baynes, R.E.; Wang, J.; Craigmill, A.L.; Riviere, J.E. A web-based decision support system to estimate extended withdrawal intervals. Comput. Electron. Agric. 2004, 44, 145–151. [Google Scholar] [CrossRef]
- Martı́n-Jiménez, T.; Baynes, R.E.; Craigmill, A.; Riviere, J.E. Extrapolated withdrawal-interval estimator (EWE) algorithm: A quantitative approach to establishing extralabel withdrawal times. Regul. Toxicol. Pharmacol. 2002, 36, 131–137. [Google Scholar] [CrossRef]
- Concordet, D.; Toutain, P.L. The withdrawal time estimation of veterinary drugs: A non-parametric approach. J. Vet. Pharmacol. Ther. 1997, 20, 374–379. [Google Scholar] [CrossRef]
- Buur, J.L.; Baynes, R.E.; Smith, G.; Riviere, J.E. Use of probabilistic modeling within a physiologic based pharmacokinetic model to predict sulfamethazine residue withdrawal times in edible tissues in swine. Antimicrob. Agents Chemother. 2006, 50, 2344–2351. [Google Scholar] [CrossRef]
- Gehring, R.; Baynes, R.E.; Craigmill, A.L.; Riviere, J.E. Feasibility of using half-life multipliers to estimate extended withdrawal intervals following the extralabel use of drugs in food-producing animals. J. Food Prot. 2004, 67, 555–560. [Google Scholar] [CrossRef]
- Mason, S.E.; Wu, H.; Yeatts, J.E.; Baynes, R.E. Tissue concentrations of sulfamethazine and tetracycline hydrochloride of swine (Sus scrofa domestica) as it relates to withdrawal methods for international export. Regul. Toxicol. Pharmacol. 2015, 71, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Baynes, R.E.; Leavens, T.; Tell, L.A.; Riviere, J.E. Use of population pharmacokinetic modeling and Monte Carlo simulation to capture individual animal variability in the prediction of flunixin withdrawal times in cattle. J. Vet. Pharmacol. Ther. 2013, 36, 248–257. [Google Scholar] [CrossRef]
- Concordet, D.; Toutain, P.L. The withdrawal time estimation of veterinary drugs revisited. J. Vet. Pharmacol. Ther. 1997, 20, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Udiani, O.; Mason, S.; Smith, G.; Mzyk, D.; Gehring, R.; Tell, L.; Riviere, J.E.; Baynes, R.E. Automation and applications of the tolerance limit method in estimating meat withdrawal periods for veterinary drugs. Comput. Electron. Agric. 2018, 146, 125–135. [Google Scholar] [CrossRef]
- Damte, D.; Jeong, H.J.; Lee, S.J.; Cho, B.H.; Kim, J.C.; Park, S.C. Evaluation of linear regression statistical approaches for withdrawal time estimation of veterinary drugs. Food Chem. Toxicol. 2012, 50, 773–778. [Google Scholar] [CrossRef]
- Letendre, L.; Gerhart, J. International methods for consumer risk assessment of injectable veterinary medicines lack of harmonization can impact international trade. J. Vet. Pharmacol. Ther. 2015, 38, 18–19. [Google Scholar]
- Diario Oficial de las Comunidades Europeas. Parlamento Europeo/Consejo de la Unión Europea. Directiva 2001/82/CE del Parlamento Europeo y del Consejo. 2001, L311, 0001–0066. Available online: https://www.boe.es/doue/2001/311/L00001-00066.pdf (accessed on 2 June 2021).
- European Union Law. Commission Directive 2004/28/EC of the European Parliament and of the Council of 31 March 2004 amending Directive 2001/82/EC. Available online: https://eur-lex.europa.eu/legal-content/ES/ALL/?uri=CELEX%3A32004L0028 (accessed on 2 June 2021).
- Comyn, G. Extra-label drug use in veterinary medicine. Food Drug Adm. Vet. Newsl. 2003, 28, 6–9. [Google Scholar]
- Government of Canada. Extra-Label Drug Use (ELDU) in Animals. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/veterinary-drugs/extra-label-drug-use.html (accessed on 5 June 2021).
- Food and Drug Administration. Code of Federal Regulations Title 21. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=530&showFR=1 (accessed on 5 June 2021).
- Spenser, E.L. Compounding, extralabel drug use, and other pharmaceutical quagmires in avian and exotics practice. Semin. Avian Exotic Pet. Med. 2004, 13, 16–24. [Google Scholar] [CrossRef]
- AMDUCA FDA. (U.S. Food and Drug Administration). Animal Medicinal Drug Use Clarification Act of 1994 (AMDUCA). Available online: https://www.fda.gov/animal-veterinary/guidance-regulations/animal-medicinal-drug-use-clarification-act-1994-amduca (accessed on 8 June 2021).
- Food and Drug Administration. General Principles for Evaluating the Human Food Safety of New Animal Drugs Used in Food-Producing Animals. Available online: https://www.fda.gov/media/70028/download (accessed on 25 June 2021).
- Food and Drug Administration. Extralabel Use and Antimicrobials. Extralabel Use Background. 2021. Available online: https://www.fda.gov/animal-veterinary/antimicrobial-resistance/extralabel-use-and-antimicrobials (accessed on 25 June 2021).
- Bader, C.; Starling, D.E.; Jones, D.E.; Brewer, M.T. Use of praziquantel to control platyhelminth parasites of fish. J. Vet. Pharmacol. Ther. 2019, 42, 139–153. [Google Scholar] [CrossRef]
- Smith, G.W.; Davis, J.L.; Tell, L.A.; Webb, A.I.; Riviere, J.E. Extralabel use of nonsteroidal anti-inflammatory drugs in cattle. J. Am. Vet. Med. Assoc. 2008, 232, 697–701. [Google Scholar] [CrossRef]
- Angulo, F.J.; Mølbak, K. Human health consequences of antimicrobial drug—Resistant Salmonella and other foodborne pathogens. Clin. Infect. Dis. 2005, 41, 1613–1620. [Google Scholar] [CrossRef]
- Aidara-Kane, A.; Angulo, F.J.; Conly, J.M.; Minato, Y.; Silbergeld, E.K.; McEwen, S.A.; Collignon, P.J. World Health Organization (WHO) guidelines on use of medically important antimicrobials in food-producing animals. Antimicrob. Resist. Infect. Control. 2018, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ajslev, T.A.; Andersen, C.S.; Gamborg, M.; Sørensen, T.I.A.; Jess, T. Childhood overweight after establishment of the gut microbiota: The role of delivery mode, pre-pregnancy weight and early administration of antibiotics. Int. J. Obes. 2011, 35, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Blustein, J.; Attina, T.; Liu, M.; Ryan, A.M.; Cox, L.M.; Blaser, M.J.; Trasande, L. Association of caesarean delivery with child adiposity from age 6 weeks to 15 years. Int. J. Obes. 2013, 37, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Trasande, L.; Blustein, J.; Liu, M.; Corwin, E.; Cox, L.M.; Blaser, M.J. Infant antibiotic exposures and early-life body mass. Int. J. Obes. 2013, 37, 16–23. [Google Scholar] [CrossRef]
- Cox, L.M.; Blaser, M.J. Antibiotics in early life and obesity. Nat. Rev. Endocrinol. 2015, 11, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Gomes, E.R.; Demoly, P. Epidemiology of hypersensitivity drug reactions. Curr. Opin. Allergy Clin. Immunol. 2005, 5, 309–316. [Google Scholar] [CrossRef]
- Paturkar, A.M.; Waskar, V.S.; Mokal, K.V.; Zende, R.Z. Antimicrobial drug residues in meat and their public health significance-a review. Ind. J. Anim. Sci. 2005, 75, 1103–1111. [Google Scholar]
- Fri Briefings. Food Research Institute—University of Wisconsin, Madison. Veterinary Drug Residues in Processed Meats—Potential Health Risk. A Review of the Scientific Literature. 2006. Available online: http://fri.wisc.edu/docs/pdf/FRIBrief_VetDrgRes.pdf (accessed on 20 June 2021).
- Raison-Peyron, N.; Messaad, D.; Bousquet, J.; Demoly, P. Anaphylaxis to beef in penicillin-allergic patient. Allergy 2001, 56, 796–797. [Google Scholar] [CrossRef]
- Botsoglou, N.A.; Fletouris, D.J. Drug Residues in Foods, Pharmacology, Food Safety, and Analysis; Marcel Dekker: New York, NY, USA, 2000; ISBN 0-8247-8959-8. [Google Scholar]
- CNN. China: 70 Ill from Tainted Pig Organs. CNN. 2009. Available online: http://www.cnn.com/2009/WORLD/asiapcf/02/22/china.poisonings/index.html (accessed on 28 June 2021).
- Mohsina, Z.; Yang, S.; Muhammad Tarique, T.; Qiu, J. Use of banned Veterinary Drugs in Feed: Food Safety Challenges and Strategies in China: A review. Eur. Acad. Res. 2015, 3, 2871–2892. [Google Scholar]
- Food Safety News. Suspected Clenbuterol poisonings in Mexico under Investigation. Available online: https://www.foodsafetynews.com/2020/08/suspected-clenbuterol-poisonings-in-mexico-under-investigation/ (accessed on 15 June 2021).
- Mateo, R.; Sánchez-Barbudo, I.S.; Camarero, P.R.; Martínez, J.M. Risk assessment of bearded vulture (Gypaetus barbatus) exposure to topical antiparasitics used in livestock within an ecotoxicovigilance framework. Sci. Total Environ. 2015, 536, 704–712. [Google Scholar] [CrossRef]
- Gómez-Ramírez, P.; Jiménez-Montalbán, P.J.; Delgado, D.; Martínez-López, E.; María-Mojica, P.; Godino, A.; García-Fernández, A.J. Development of a QuEChERS method for simultaneous analysis of antibiotics in carcasses for supplementary feeding of endangered vultures. Sci. Total Environ. 2018, 626, 319–327. [Google Scholar] [CrossRef]
- Blanco, G.; Junza, A.; Segarra, D.; Barbosa, J.; Barrón, D. Wildlife contamination with fluoroquinolones from livestock: Widespread occurrence of enrofloxacin and marbofloxacin in vultures. Chemosphere 2016, 144, 1536–1543. [Google Scholar] [CrossRef]
- Blanco, G.; Junza, A.; Barrón, D. Occurrence of veterinary pharmaceuticals in golden eagle nestlings: Unnoticed scavenging on livestock carcasses and other potential exposure routes. Sci. Total Environ. 2017, 586, 355–361. [Google Scholar] [CrossRef]
- Blanco, G.; Junza, A.; Barrón, D. Food safety in scavenger conservation: Diet-associated exposure to livestock pharmaceuticals and opportunist mycoses in threatened Cinereous and Egyptian vultures. Ecotoxicol. Environ. Saf. 2017, 135, 292–301. [Google Scholar] [CrossRef]
- Pitarch, A.; Gil, C.; Blanco, G. Oral mycoses in avian scavengers exposed to antibiotics from livestock farming. Sci. Total Environ. 2017, 605, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Oaks, J.L.; Gilbert, M.; Virani, M.Z.; Watson, R.T.; Meteyer, C.U.; Rideout, B.A.; Shivaprasad, H.L.; Ahmed, S.; Chaudhry, M.J.I.; Arshad, M.; et al. Diclofenac residues as the cause of vulture population decline in Pakistan. Nature 2004, 427, 630–633. [Google Scholar] [CrossRef]
- Green, R.E.; Newton, I.A.N.; Shultz, S.; Cunningham, A.A.; Gilbert, M.; Pain, D.J.; Prakash, V. Diclofenac poisoning as a cause of vulture population declines across the Indian subcontinent. J. Appl. Ecol. 2004, 41, 793–800. [Google Scholar] [CrossRef]
- Shultz, S.; Baral, H.S.; Charman, S.; Cunningham, A.A.; Das, D.; Ghalsasi, G.R.; Goudar, M.S.; Green, R.E.; Jones, A.; Nighot, P.; et al. Diclofenac poisoning is widespread in declining vulture populations across the Indian subcontinent. Proc. R. Soc. Lond. B. 2004, 27, S458–S460. [Google Scholar] [CrossRef]
- Green, R.E.; Taggart, M.A.; Senacha, K.R.; Raghavan, B.; Pain, D.J.; Jhala, Y.; Cuthbert, R. Rate of decline of the oriental white-backed vulture population in India estimated from a survey of diclofenac residues in carcasses of ungulates. PLoS ONE 2007, 2, e686. [Google Scholar] [CrossRef]
- Swan, G.E.; Cuthbert, R.; Quevedo, M.; Green, R.E.; Pain, D.J.; Bartels, P.; Cunningham, A.A.; Duncan, N.; Meharg, A.; Oaks, J.L.; et al. Toxicity of diclofenac to Gyps vultures. Biol. Lett. 2006, 2, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, V.; Wolter, K.; Cuthbert, R.; Duncan, N. Veterinary diclofenac threatens Africa’s endangered vulture species. Regul. Toxicol. Pharmacol. 2009, 53, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Cuthbert, R.; Taggart, M.A.; Prakash, V.; Saini, M.; Swarup, D.; Upreti, S.; Mateo, R.; Chakraborty, S.S.; Deori, P.; Green, R.E. Effectiveness of action in India to reduce exposure of Gyps vultures to the toxic veterinary drug diclofenac. PLoS ONE 2011, 6, e19069. [Google Scholar] [CrossRef] [PubMed]
- Herrero-Villar, M.; Delepoulle, E.; Suarez-Regalado, L.; Solano-Manrique, C.; Juan-Salles, C.; Iglesias-Lebrija, J.J.; Camarero, P.R.; Gonzalez, F.; Alvarez, E.; Mateo, R. First diclofenac intoxication in a wild avian scavenger in Europe. Sci. Total Environ. 2021, 782, 146890. [Google Scholar] [CrossRef]
- Halley, B.A.; Jacob, T.A.; Lu, A.Y. The environmental impact of the use of ivermectin: Environmental effects and fate. Chemosphere 1989, 18, 1543–1563. [Google Scholar] [CrossRef]
- Chiu, S.H.L.; Green, M.L.; Baylis, F.P.; Eline, D.; Rosegay, A.; Meriwether, H.; Jacob, T.A. Absorption, tissue distribution, and excretion of tritium-labeled ivermectin in cattle, sheep, and rat. J. Agric. Food Chem. 1990, 38, 2072–2078. [Google Scholar] [CrossRef]
- Canga, A.G.; Prieto, A.M.S.; Liébana, M.J.D.; Martínez, N.F.; Vega, M.S.; Vieitez, J.J.G. The pharmacokinetics and metabolism of ivermectin in domestic animal species. Vet. J. 2009, 179, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Boxall, A.B.; Fogg, L.A.; Blackwell, P.A.; Kay, P.; Pemberton, E.J.; Croxford, A. Veterinary medicines in the environment. Rev. Environ. Contam Toxicol. 2004, 180, 1–91. [Google Scholar] [CrossRef] [PubMed]
- Wardhaugh, K.G.; Holter, P.; Whitby, W.A.; Shelley, K. Effects of drug residues in the faeces of cattle treated with injectable formulations of ivermectin and moxidectin on larvae of the bush fly, Musca vetustissima and the house fly, Musca domestica. Aust. Vet. J. 1996, 74, 370–374. [Google Scholar] [CrossRef]
- Rombke, J.; Krogh, K.A.; Moser, T.; Scheffczyk, A.; Liebig, M. Effects of the veterinary pharmaceutical ivermectin on soil invertebrates in laboratory tests. Arch. Environ. Contam. Toxicol. 2010, 58, 332–340. [Google Scholar] [CrossRef]
- Forster, B.; Boxall, A.; Coors, A.; Jensen, J.; Liebig, M.; Pope, L.; Moser, T.; Rombke, J. Fate and effects of ivermectin on soil invertebrates in terrestrial model ecosystems. Ecotoxicology 2011, 20, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Verdu, J.R.; Lobo, J.M.; Sánchez-Piñero, F.; Gallego, B.; Numa, C.; Lumaret, J.P.; Cortez, V.; Ortiz, A.J.; Tonelli, M.; Garcia-Teba, J.P.; et al. Ivermectin residues disrupt dung beetle diversity, soil properties and ecosystem functioning: An interdisciplinary field study. Sci. Total Environ. 2018, 618, 219–228. [Google Scholar] [CrossRef]
- Lumaret, J.P.; Errouissi, F.; Floate, K.; Rombke, J.; Wardhaugh, K. A review on the toxicity and non-target effects of macrocyclic lactones in terrestrial and aquatic environments. Curr. Pharm. Biotechnol. 2012, 13, 1004–1060. [Google Scholar] [CrossRef] [PubMed]
- Eichberg, C.; Wohde, M.; Mueller, K.; Rausch, A.; Scherrmann, C.; Scheuren, T.; During, R.A.; Donath, T.W. The anthelmintic ingredient moxidectin negatively affects seed germination of three temperate grassland species. PLoS ONE 2016, 11, e0166366. [Google Scholar] [CrossRef]
- Ivan, V.; Šadibolová, M.; Podlipná, R.; Lamka, J.; Prchal, L.; Sobotová, D.; Lokvencová, K.; Szotáková, B.; Skálová, L. Ivermectin environmental impact: Excretion profile in sheep and phytotoxic effect in Sinapis alba. Ecotoxicol. Environ. Saf. 2019, 169, 944–949. [Google Scholar] [CrossRef]
- Ortelli, D.; Spörri, A.S.; Edder, P. Veterinary drug residue in food of animal origin in Switzerland: A health concern? Chimia 2018, 72, 713–717. [Google Scholar] [CrossRef]
- European Food Safety Authority. Report for 2019 on the results from the monitoring of veterinary medicinal product residues and other substances in live animals and animal products. EFSA J. 2021, 18, 1–82. [Google Scholar] [CrossRef]
- Nonga, H.E.; Sungura, K.H.; Ngowi, H.A. Assessment of veterinary drug use and determination of antimicrobial residues in broiler chicken meat in Urban district, Zanzibar, Tanzania. Tanzan. Vet. J. 2013, 28. [Google Scholar]
- Donkor, E.S.; Newman, M.J.; Tay, S.C.; Dayie, N.T.; Bannerman, E.; Olu-Taiwo, M. Investigation into the risk of exposure to antibiotic residues contaminating meat and egg in Ghana. Food Control 2011, 22, 869–873. [Google Scholar] [CrossRef]
- Tuyet-Hanh, T.T.; Sinh, D.X.; Phuc, P.D.; Ngan, T.T.; Van Tuat, C.; Grace, D.; Unger, F.; Nguyen-Viet, H. Exposure assessment of chemical hazards in pork meat, liver, and kidney, and health impact implication in Hung Yen and Nghe An provinces, Vietnam. Int. J. Public Health 2017, 62, 75–82. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Joint FAO/WHO Expert Committee on Food Additives. Annex 3–Pilot of New Approaches to Estimate Dietary Exposure to Veterinary Drug Residues. Available online: https://www.who.int/foodsafety/chem/Annex1_Pilot_of_Dietary_Exposure_Assessment.pdf (accessed on 20 May 2021).
- World Health Organization. Dietary exposure assessment for chemicals in food. In Environmental Health Criteria 240: Principles and Methods for the Risk Assessment of Chemicals in Food, 2nd ed.; Sheffer, M., Ed.; WHO Press: Geneva, Switzerland, 2020; pp. 1–175. ISBN 978 92 4 157240 8. [Google Scholar]
- Darko, G.; Mensah, J.K.; Dapaah, S.S.; Odei, J. Estimated dietary exposure to veterinary residues in chicken and eggs. Food Contam. 2015, 2, 1–8. [Google Scholar] [CrossRef]
- He, X.; Deng, M.; Wang, Q.; Yang, Y.; Yang, Y.; Nie, X. Residues and health risk assessment of quinolones and sulfonamides in cultured fish from Pearl River Delta, China. Aquaculture 2016, 458, 38–46. [Google Scholar] [CrossRef]
- Liu, X.; Lu, S.; Meng, W.; Zheng, B. Residues and health risk assessment of typical antibiotics in aquatic products from the Dongting Lake, China—“Did you eat “Antibiotics” today?”. Environ. Sci. Pollut. Res. 2018, 25, 3913–3921. [Google Scholar] [CrossRef]
- Tsai, M.Y.; Lin, C.F.; Yang, W.C.; Lin, C.T.; Hung, K.H.; Chang, G.R. Health risk assessment of banned veterinary drugs and quinolone residues in shrimp through liquid chromatography–tandem mass spectrometry. Appl. Sci. 2019, 9, 2463. [Google Scholar] [CrossRef]
- Ji, X.; Xu, Y.; Wang, J.; Lyu, W.; Li, R.; Tan, S.; Xiao, Y.; Yang, H.; Qian, M. Multiresidue determination of antibiotics in ready-to-eat duck eggs marketed through e-commerce stores in China and subsequent assessment of dietary risks to consumers. J. Food Sci. 2021, 86, 2145–2162. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.M.; Whelan, M.; Kennedy, D.G.; Trigueros, G.; Cannavan, A.; Boon, P.E.; Wapperom, D.; Danaher, M. Anthelmintic drug residues in beef: UPLC-MS/MS method validation, European retail beef survey, and associated exposure and risk assessments. Food Addit. Contam. Part A 2012, 29, 746–760. [Google Scholar] [CrossRef]
- Mingle, C.L.; Darko, G.; Borquaye, L.S.; Asare-Donkor, N.K.; Woode, E.; Koranteng, F. Veterinary Drug Residues in Beef, Chicken, and Egg from Ghana. Chem. Afr. 2021, 4, 339–348. [Google Scholar] [CrossRef]
- Vougat Ngom, R.R.; Foyet, H.S.; Garabed, R.; Zoli, A.P. Human Health Risks Related to Penicillin G and Oxytetracycline Residues Intake Through Beef Consumption and Consumer Knowledge About Drug Residues in Maroua, Far North of Cameroon. Front Vet. Sci. 2020, 7, 478. [Google Scholar] [CrossRef]
- Wang, H.; Ren, L.; Yu, X.; Hu, J.; Chen, Y.; He, G.; Jiang, Q. Antibiotic residues in meat, milk and aquatic products in Shanghai and human exposure assessment. Food Control 2017, 80, 217–225. [Google Scholar] [CrossRef]
- Canton, L.; Signorini, M.; Canton, C.; Dominguez, P.; Farias, C.; Alvarez, L.; Lanusse, C.; Moreno, L. Quantitative Exposure Assessment and Risk Characterization for Fipronil Residues in Laying Hen Eggs. In Proceedings of the 28th Conference of the World Association for the Advancement of Veterinary Parasitology WAAVP 2021, Dublin, Ireland, 19–22 July 2021; p. 461. [Google Scholar]
- Hu, Y.; Cheng, H.; Tao, S. Environmental and human health challenges of industrial livestock and poultry farming in China and their mitigation. Environ. Int. 2017, 107, 111–130. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Lee, S.B.; Shin, D.; Jeong, J.; Hong, J.H.; Rhee, G.S. Occurrence of veterinary drug residues in farmed fishery products in South Korea. Food Control 2018, 85, 57–65. [Google Scholar] [CrossRef]
- Baazize-Ammi, D.; Dechicha, A.S.; Tassist, A.; Gharbi, I.; Hezil, N.; Kebbal, S.; Morsli, W.; Beldjoudi, S.; Saadaoui, M.R.; Guetarni, D. Screening and quantification of antibiotic residues in broiler chicken meat and milk in the central region of Algeria. Rev. Sci. Tech. 2019, 38, 863–877. [Google Scholar] [CrossRef]
- Cayci, M.; Celik, A.S.; Oruc, H.H.; Sariyev, R. Screening of veterinary growth-promoting agent and antibacterial residues in beef cattle and broiler meats consumed in Bursa, Turkey. J. Res. Vet. Med. 2019, 38, 52–58. [Google Scholar] [CrossRef][Green Version]
- Hassan, M.A.; Amin, R.; Abo Elroos, N. Enrofloxacin residues in chicken meat and giblete. Benha Vet. Med. J. 2019, 36, 175–183. [Google Scholar] [CrossRef]
- Doğan, Y.N.; Pamuk, Ş.; Gürler, Z. Chloramphenicol and sulfonamide residues in sea bream (Sparus aurata) and sea bass (Dicentrarchus labrax) fish from aquaculture farm. Environ. Sci. Pollut. Res. 2020, 27, 41248–41252. [Google Scholar] [CrossRef]
- Mokh, S.; El Hawari, K.; Rahim, H.A.; Al Iskandarani, M.; Jaber, F. Antimicrobial residues survey by LC-MS in food-producing animals in Lebanon. Food Addit. Contam. Part B 2020, 13, 121–129. [Google Scholar] [CrossRef]
- KuKanich, B.; Gehring, R.; Webb, A.I.; Craigmill, A.L.; Riviere, J.E. Effect of formulation and route of administration on tissue residues and withdrawal times. J. Am. Vet. Med. Assoc. 2005, 227, 1574–1577. [Google Scholar] [CrossRef]
- Delahaut, P.; Pierret, G.; Ralet, N.; Dubois, M.; Gillard, N. Multi-residue method for detecting coccidiostats at carry-over level in feed by HPLC–MS/MS. Food Addit. Contam. Part A 2010, 27, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Borràs, S.; Companyó, R.; Granados, M.; Guiteras, J.; Pérez-Vendrell, A.M.; Brufau, J.; Medina, M.; Bosch, J. Analysis of antimicrobial agents in animal feed. Trends Analyt. Chem. 2011, 30, 1042–1064. [Google Scholar] [CrossRef]
- Stolker, A.A.M.; Manti, V.; Zuidema, T.; Van Egmond, H.; Deckers, E.R.; Herbes, R.; Hooglugt, J.; Heuvel, E.O.; De Jong, J. Carry-over of veterinary drugs from medicated to non-medicated feeds in commercial feed manufacturing plants. Food Addit. Contam. Part A 2013, 30, 1100–1107. [Google Scholar] [CrossRef]
- Barbosa Da Silva, A.; Back, M.; Daguer, H.; Palmeira, M.; Antunes De Sá Ploêncio, L.; Molognoni, L.; Peripolli, V.; Bianchi, I. Carry-over and contamination of veterinary drugs in feed production lines for poultry and pigs. Food Addit. Contam. Part A 2019, 36, 740–751. [Google Scholar] [CrossRef]
- De Souza Borges, P.A.R. Métodos de Descontaminação de Productos Veterinários Utilizados na Produção de Alimentos Para Animais. Ph.D. Thesis, Universidade Federal de Santa Catarina, Florianópolis, Brazil, October 2012. [Google Scholar]
- European Commission. RASFF Annual Report 2018. Rapid Alert System for Food and Feed. 2018. Available online: https://op.europa.eu/en/publication-detail/-/publication/c3318331-d9c4-11e9-9c4e-01aa75ed71a1/language-en/format-PDF/source-211856390 (accessed on 5 June 2021).
- European Union Law. Commission Directive 2009/8/EC of 10 February 2009 Amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as regards Maximum Levels of Unavoidable Carry-over of Coccidiostats or Histomonostats in Non-Target Feed. Available online: eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32009L0008 (accessed on 20 June 2021).
- European Food Safety Authority. Cross-contamination of the Non-Target Feeding Stuffs by Narasin Authorised for Use as a Feed Additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2007, 552, 1–35. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding stuffs by lasalocid authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2007, 553, 1–46. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding stuffs by salinomycin authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 591, 1–38. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding stuffs by monensina authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 592, 1–40. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by semduramycin authorised for use as a feed additive. Scientific Opinion on the Panel of Contaminants in the Food Chain. EFSA J. 2008, 593, 1–27. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by maduramycin authorised for use as a feed additive. Scientific Opinion on the Panel of Contaminants in the Food Chain. EFSA J. 2008, 594, 1–30. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by halofuginone authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 657, 1–31. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by nicarbazin authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 553, 1–34. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by diclazuril authorised for use as a feed additive. Scientific Opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 716, 1–31. [Google Scholar] [CrossRef]
- European Food Safety Authority. Cross-contamination of the non-target feeding-stuffs by robenidine authorised for use as a feed additive. Scientific Opinion on the Panel of Contaminants in the Food Chain. EFSA J. 2008, 655, 1–29. [Google Scholar] [CrossRef]
- Laffont, C.M.; Alvinerie, M.; Bousquet-Mélou, A.; Toutain, P.L. Licking behaviour and environmental contamination arising from pour-on ivermectin for cattle. Int. J. Parasitol. 2001, 31, 1687–1692. [Google Scholar] [CrossRef]
- Gokbulut, C.; Karademir, U.; Boyacioglu, M. Comparison of plasma pharmacokinetic profile of ivermectin following administration of subcutaneous injection (Baymec®) and oral tablet (Efektin®) in goats. J. Vet. Pharmacol. Therap. 2007, 30, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Lespine, A.; Alvinerie, M.; Sutra, J.F.; Pors, I.; Chartier, C. Influence of the route of administration on efficacy and tissue distribution of ivermectin in goat. Vet. Parasitol. 2005, 128, 251–260. [Google Scholar] [CrossRef]
- Google Patents. Administration of an Injectable Antibiotic in the Ear of An Animal. Available online: https://patents.google.com/patent/US6074657A/en (accessed on 15 June 2021).
- Pyörälä, S.; Laurila, T.; Lehtonen, S.; Leppä, S.; Kaartinen, L. Local tissue damage in cows after intramuscular administration of preparations containing phenylbutazone, flunixin, ketoprofen and metamizole. Acta Vet. Scand. 1999, 40, 145–150. [Google Scholar] [CrossRef]
- Rasmussen, F. Tissue damage at the injection site after intramuscular injection of drugs. Vet. Res. Commun. 1978, 2, 173–182. [Google Scholar] [CrossRef]
- Lifschitz, A.; Sallovitz, J.; Imperiale, F.; Pis, A.; Jauregui Lorda, J.; Lanusse, C. Pharmacokinetic evaluation of four ivermectin generic formulations in calves. Vet. Parasitol. 2004, 119, 247–257. [Google Scholar] [CrossRef]
- Lespine, A.; Ménez, C.; Bourguinat, C.; Prichard, R.K. P-glycoproteins and other multidrug resistance transporters in the pharmacology of anthelmintics:Prospects for reversing transport-dependent anthelmintic resistance. Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 58–75. [Google Scholar] [CrossRef]
- Lespine, A.; Alvinerie, M.; Vercruysse, J.; Prichard, R.K.; Geldhof, P. ABC transporter modulation: A strategy to enhance the activity of macrocyclic lactone anthelmintics. Trends Parasitol. 2008, 24, 293–298. [Google Scholar] [CrossRef]
- Lifschitz, A.; Ballent, M.; Lanusse, C. Macrocyclic lactones and cellular transport-related drug interactions: A perspective from in vitro assays to nematode control in the field. Curr. Pharm. Biotechnol. 2012, 13, 912–923. [Google Scholar] [CrossRef] [PubMed]
- Medlicott, N.J.; Waldron, N.A.; Foster, T.P. Sustained release veterinary parenteral products. Adv. Drug Deliv. Rev. 2004, 56, 1345–1365. [Google Scholar] [CrossRef] [PubMed]
- Brunt, L.M.; Rast, L.; Hernandez-Jover, M.; Brockwell, Y.M.; Woodgate, R.G. A producer survey of knowledge and practises on gastrointestinal nematode control within the Australian goat industry. Vet. Parasitol. Reg. Stud. Rep. 2019, 18, 100325. [Google Scholar] [CrossRef] [PubMed]
- Sallovitz, J.; Lifschitz, A.; Imperiale, F.; Pis, A.; Virkel, G.; Lanusse, C. Breed differences on the plasma availability of moxidectin administered pour-on to calves. Vet. J. 2002, 164, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Vercruysse, J.; Deprez, P.; Everaert, D.; Bassissi, F.; Alvinerie, M. Breed differences in the pharmacokinetics of ivermectin administered subcutaneously to Holstein and Belgian Blue calves. Vet. Parasitol. 2008, 152, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Gokbulut, C.; Bilgili, A.; Hanedan, B.; Aksit, D.; Aksoy, A.M.; Turgut, C. Breed-related plasma disposition of ivermectin following subcutaneous administration in Kilis and Damascus goats. Res. Vet. Sci. 2009, 87, 445–448. [Google Scholar] [CrossRef]
- Gokbulut, C.; Bilgili, A.; Hanedan, B.; Aksit, D.; Aksoy, A.M.; Turgut, C. Sex-related plasma disposition of ivermectin following pour-on administration in goats. Vet. Parasitol. 2009, 162, 342–345. [Google Scholar] [CrossRef]
- Toutain, P.L.; Upson, D.W.; Terhune, T.N.; McKenzie, M.E. Comparative pharmacokinetics of doramectin and ivermectin in cattle. Vet. Parasitol. 1997, 72, 3–8. [Google Scholar] [CrossRef]
- Craven, J.; Bjørn, H.; Hennessy, D.R.; Friis, C. The effects of body composition on the pharmacokinetics of subcutaneously injected ivermectin and moxidectin in pigs. J. Vet. Pharmacol. Ther. 2002, 25, 227–232. [Google Scholar] [CrossRef]
- Moreno, L.; Ceballos, L.; Lifschitz, A.; Bistoletti, M.; Álvarez, L.; Lanusse, C. Combined subcutaneous administration of ivermectina and nitroxynil in lambs: Body weight related changes to kinetic dispositionof both compounds. Res. Vet. Sci. 2010, 88, 315–320. [Google Scholar] [CrossRef]
- Alvarez, L.; Sanchez, S.; Lanusse, C. Modified plasma and abomasal disposition of albendazole in nematode-infected sheep. Vet. Parasitol. 1997, 69, 241–253. [Google Scholar] [CrossRef]
- Pérez, R.; Palma, C.; Nuñez, M.J.; Cabezas, I. Patterns of doramectin tissue residue depletion in parasitized vs nonparasitized lambs. Parasitol. Res. 2008, 102, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Herrera, I.; Schneider, M.J.; Cole, K.; Farnell, M.B.; Blore, P.J.; Donoghue, D.J. Concentrations of antibiotic residues vary between different edible muscle tissues in poultry. J. Food Prot. 2005, 68, 2217–2219. [Google Scholar] [CrossRef]
- Moreno, L.; Alvarez, L.; Ceballos, L.; Sánchez Bruni, S.; Lanusse, C. Pattern of ivermectin (sheep) and doramectin (cattle) residues in muscular tissue from different anatomical locations. Food Addit. Contam. Part A 2008, 25, 406–412. [Google Scholar] [CrossRef]
- World Health Organization. Dietary Exposure Assessment of Chemicals in Food. Available online: https://apps.who.int/iris/bitstream/handle/10665/44027/9789241597470_eng.pdf (accessed on 15 June 2021).
- Nguyen, V.; Li, M.; Khan, M.A.; Li, C.; Zhou, G. Effect of cooking methods on tetracycline residues in pig meat. Afr. J. Pharm. Pharmacol. 2013, 7, 1448–1454. [Google Scholar] [CrossRef]
- Heshmati, A.; Kamkar, A.; Salaramoli, J.; Hassan, J.; Jahed, G.H. Effect of deep-frying processing on tylosin residue in meat. J. Food Sci. Technol. 2014, 12, 42–52. [Google Scholar]
- Shaltout, F.A.E.; Shatter, M.A.E.; Sayed, N.F. Impacts of Different Types of Cooking and Freezing on Antibiotic Residues in Chicken Meat. J. Food Sci. Nutr. 2019, 5, 45. [Google Scholar]
- Ismail-Fitry, M.R.; Jinap, S.; Jamilah, B.; Saleha, A.A. Effect of deep-frying at different temperature and time on sulfonamide residues in chicken meat-balls. J. Food Drug Anal. 2008, 16, 81–86. [Google Scholar] [CrossRef]
- Ibrahim, A.; Moats, W.A. Effect of cooking procedures on oxytetracycline residues in lamb muscle. J. Agric. Food Chem. 1994, 42, 2561–2563. [Google Scholar] [CrossRef]
- Ziani, K.; Pérez-López, M.; Mansouri, A.; Khaled, M.B.; Rodriguez, A.S.; Slimani, M. Assessment of oxytetracycline residue in cooked and raw meat of chicken broilers before and after the end of official withdrawal period. Food Anal. Methods. 2018, 11, 2528–2537. [Google Scholar] [CrossRef]
- Javadi, A. Effect of roasting, boiling and microwaving cooking method on doxycline residues in edible tissues of poultry by microbial method. Afr. J. Pharm. Pharmacol. 2011, 5, 1034–1037. [Google Scholar] [CrossRef]
- Gajda, A.; Bladek, T.; Gbylik-Sikorska, M.; Posyniak, A. The influence of cooking procedures on doxycycline concentration in contaminated eggs. Food Chem. 2017, 221, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- Papapanagiotou, E.P.; Fletouris, D.J.; Psomas, E.I. Effect of various heat treatments and cold storage on sulphamethazine residues stability in incurred piglet muscle and cow milk samples. Anal. Chim. Acta 2005, 529, 305–309. [Google Scholar] [CrossRef]
- Khan, A.A.; Randhawa, M.A.; Butt, M.S.; Nawaz, H. Impact of various processing techniques on dissipation behavior of antibiotic residues in poultry meat. J. Food Process. Preserv. 2016, 40, 76–82. [Google Scholar] [CrossRef]
- Lolo, M.; Pedreira, S.; Miranda, J.M.; Vázquez, B.I.; Franco, C.M.; Cepeda, A.; Fente, C. Effect of cooking on enrofloxacin residues in chicken tissue. Food Addit. Contam. 2006, 23, 988–993. [Google Scholar] [CrossRef]
- Canton, L.; Alvarez, L.; Canton, C.; Ceballos, L.; Farias, C.; Lanusse, C.; Moreno, L. Effect of cooking on the stability of veterinary drug residues in chicken eggs. Food Addit. Contam. Part A 2019, 36, 1055–1067. [Google Scholar] [CrossRef]
- Fahim, H.M. Evaluate antibiotic residues in beef and effect of cooking and freezing on it. Benha Vet. Med J. 2019, 36, 109–116. [Google Scholar] [CrossRef]
- Gogoi, R.; Roy, D.C. Effect of cooking on ciprofloxacin level in chicken meat. Pharma. Innov. J. 2019, 8, 208–210. [Google Scholar]
- Salaramoli, J.; Heshmati, A.; Kamkar, A.; Hassan, J. Effect of cooking procedures on tylosin residues in chicken meatball. J. Verbr. Lebensm. 2016, 11, 53–60. [Google Scholar] [CrossRef]
- Elbagory, A.M.; Yasin, N.A.; Algazar, E.A. Effect of Various Cooking Methods on Some Antibacterial Residues in Imported and Local Frozen Dressed Broilers and their Giblets in Egypt. Nutr. Food Technol. 2016, 2, 127. [Google Scholar] [CrossRef]
- Rose, M.D.; Farrington, W.H.H.; Shearer, G. The effect of cooking on veterinary drug residues in food: 7. Ivermectin. Food Addit. Contam. 1998, 15, 157–161. [Google Scholar] [CrossRef]
- Cooper, K.M.; Whelan, M.; Danaher, M.; Kennedy, D.G. Stability during cooking of anthelmintic veterinary drug residues in beef. Food Addit. Contam. Part A 2011, 28, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Slanina, P.; Kuivinen, J.; Ohlsén, C.; Ekström, L.G. Ivermectin residues in the edible tissues of swine and cattle: Effect of cooking and toxicological evaluation. Food Addit. Contam. 1989, 6, 475–481. [Google Scholar] [CrossRef]
- Rose, M.D.; Shearer, G.; Farrington, W.H.H. The effect of cooking on veterinary drug residues in food; 5. Oxfendazole. Food Addit. Contam. 1997, 14, 15–26. [Google Scholar] [CrossRef]
- Muaz, K.; Riaz, M.; Akhtar, S.; Park, S.; Ismail, A. Antibiotic residues in chicken meat: Global prevalence, threats, and decontamination strategies: A review. J. Food Prot. 2018, 81, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Vishnuraj, M.R.; Kandeepan, G.; Rao, K.H.; Chand, S.; Kumbhar, V. Occurrence, public health hazards and detection methods of antibiotic residues in foods of animal origin: A comprehensive review. Cogent Food Agric. 2016, 2, 1235458. [Google Scholar] [CrossRef]
- Boeckel, T.P.V.; Brower, C.; Gilbert, M.; Grenfel, B.T.l.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2018. Available online: https://www.ema.europa.eu/en/documents/report/sales-veterinary-antimicrobial-agents-31-european-countries-2018-trends-2010-2018-tenth-esvac-report_en.pdf (accessed on 5 June 2021).
- Schar, D.; Sommanustweechai, A.; Laxminarayan, R.; Tangcharoensathien, V. Surveillance of antimicrobial consumption in animal production sectors of low-and middle-income countries: Optimizing use and addressing antimicrobial resistance. PLoS Med. 2018, 15, 1–9. [Google Scholar] [CrossRef]
- Bacanlı, M.; Başaran, N. Importance of antibiotic residues in animal food. Food Chem. Toxicol. 2019, 125, 462–466. [Google Scholar] [CrossRef]
- Toutain, P.L.; Ferran, A.A.; Bousquet-Melou, A.; Pelligand, L.; Lees, P. Veterinary medicine needs new green antimicrobial drugs. Front. Microbiol. 2016, 7, 1196. [Google Scholar] [CrossRef] [PubMed]
- Andersson, D.I.; Hughes, D. Antibiotic resistance and its cost: Is it possible to reverse resistance? Nat. Rev. Microbiol. 2010, 8, 260–271. [Google Scholar] [CrossRef]
- Vivienne, E.E.; Josephine, O.K.O.; Anaelom, N.J. Effect of temperature (cooking and freezing) on the concentration of oxytetracycline residue in experimentally induced birds. Vet. World 2018, 11, 167. [Google Scholar] [CrossRef] [PubMed]
- Abou-Raya, S.H.; Shalaby, A.R.; Salama, N.A.; Emam, W.H.; Mehaya, F.M. Effect of ordinary cooking procedures on tetracycline residues in chicken meat. J. Food Drug Anal. 2013, 21, 80–86. [Google Scholar]
- Alaboudi, A.; Basha, E.A.; Musallam, I. Chlortetracycline and sulfanilamide residues in table eggs: Prevalence, distribution between yolk and white and effect of refrigeration and heat treatment. Food Cont. 2013, 33, 281–286. [Google Scholar] [CrossRef]
- Furusawa, N.; Hanabusa, R. Cooking effects on sulfonamide residues in chicken thigh muscle. Food Res. Int. 2002, 35, 37–42. [Google Scholar] [CrossRef]
- Hussein, M.A.; Ahmed, M.M.; Morshedy, A.M. Effect of cooking methods on some antibiotic residues in chicken meat. Jpn. J. Vet. Res. 2016, 64, S225–S231. [Google Scholar]
- Sireli, U.T.; Ilazi, A.; Cadirci, O. Effect of cooking and storage times on gentamicin residues in eggs. Ital. J. Food Sci. 2006, 18, 441–446. [Google Scholar]
- Cooper, K.M.; Kennedy, D.G. Stability studies of the metabolites of nitrofuran antibiotics during storage and cooking. Food Addit. Contam. 2007, 24, 935–942. [Google Scholar] [CrossRef]
- Rose, M.D.; Argent, L.C.; Shearer, G.; Farrington, W.H. The effect of cooking on veterinary drug residues in food: 2. levamisole. Food Addit. Contam. 1995, 12, 185–194. [Google Scholar] [CrossRef]
- World Health Organization. Worldwide Country Situation Analysis: Response to Antimicrobial Resistance. Available online: http://apps.who.int/iris/bitstream/10665/163468/1/9789241564946_eng.pdf (accessed on 5 June 2021).
- Food Water & Watch. Antibiotic Resistance 101. How Antibiotic Misuse on Factory Farms Can Make You Sick. 2012. Available online: https://www.yumpu.com/en/document/read/24346542/antibiotic-resistance-101-food-water-watch (accessed on 6 August 2021).
- European Union. Antimicrobials in Agriculture and the Environment: Reducing Unnecessary Use and Waste. Available online: https://ec.europa.eu/health/sites/default/files/antimicrobial_resistance/docs/amr_studies_2015_am-in-agri-and-env.pdf (accessed on 25 May 2021).
- Mole, B. MRSA: Farming up trouble. Nature 2013, 499, 398. [Google Scholar] [CrossRef] [PubMed]
- Reardon, S. Resistance to last-ditch antibiotic has spread farther than anticipated. Nat. News 2017, 10, 1–3. [Google Scholar] [CrossRef]
- Moodley, A.; Guardabassi, L. Transmission of IncN plasmids carrying bla CTX-M-1 between commensal Escherichia coli in pigs and farm workers. Antimicrob. Agents Chemother. 2009, 53, 1709–1711. [Google Scholar] [CrossRef]
- Marshall, B.M.; Levy, S.B. Food animals and antimicrobials: Impacts on human health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Abdi, R.D.; Mengstie, F.; Beyi, A.F.; Beyene, T.; Waktole, H.; Mammo, B.; Ayana, D.; Abunna, F. Determination of the sources and antimicrobial resistance patterns of Salmonella isolated from the poultry industry in Southern Ethiopia. BMC Infect Dis. 2017, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Categorisation of Antibiotics in the European Union. Available online: https://www.ema.europa.eu/en/documents/report/categorisation-antibiotics-european-union-answer-request-european-commission-updating-cientific_en.pdf (accessed on 5 June 2021).
- Pijpers, A.; Schoevers, E.J.; Van Gogh, H.; van Leengoed, L.A.M.G.; Visser, I.J.R.; van Miert, A.S.J.P.A.M.; Verheijden, J.H.M. The influence of disease on feed and water consumption and on pharmacokinetics of orally administered oxytetracycline in pigs. J. Anim. Sci. 1991, 69, 2947–2954. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.; Gyrd-Hansen, N. Bioavailability of oxytetracycline, tetracycline and chlortetracycline after oral administration to fed and fasted pigs. J. Vet. Pharmacol. Ther. 1996, 19, 305–311. [Google Scholar] [CrossRef]
- Pollet, R.A.; Glatz, C.E.; Dyer, D.C.; Barnes, H.J. Pharmacokinetics of chlortetracycline potentiation with citric acid in the chicken. Am. J. Vet. Res. 1983, 44, 1718–1721. [Google Scholar]
- Hansen, L.H.; Aarestrup, F.; Sørensen, S.J. Quantification of bioavailable chlortetracycline in pig feces using a bacterial whole-cell biosensor. Vet. Microbiol. 2002, 87, 51–57. [Google Scholar] [CrossRef]
- Herrick, J.B.; Haynes, R.; Heringa, S.; Brooks, J.M.; Sobota, L.T. Coselection for resistance to multiple late-generation human therapeutic antibiotics encoded on tetracycline resistance plasmids captured from uncultivated stream and soil bacteria. J. Appl. Microbiol. 2014, 117, 380–389. [Google Scholar] [CrossRef]
- Bibbal, D.; Dupouy, V.; Ferré, J.P.; Toutain, P.L.; Fayet, O.; Prère, M.F.; Bousquet-Mélou, A. Impact of three ampicillin dosage regimens on selection of ampicillin resistance in Enterobacteriaceae and excretion of bla TEM genes in swine feces. Appl. Environ. Microbiol. 2007, 73, 4785–4790. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Price volatility continues to affect farmed salmon sector as new records are breached once again. Available online: http://www.fao.org/in-action/globefish/marketreports/resource-detail/es/c/1156020/ (accessed on 5 June 2021).
- Barani, A.; Fallah, A.A. Occurrence of tetracyclines, sulfonamides, fluoroquinolones and florfenicol in farmed rainbow trout in Iran. Food Agric. Immun. 2015, 26, 420–429. [Google Scholar] [CrossRef]
- Cabello, F.C.; Godfrey, H.P.; Buschmann, A.H.; Dölz, H.J. Aquaculture as yet another environmental gateway to the development and globalisation of antimicrobial resistance. Lancet Infect. Dis. 2016, 16, e127–e133. [Google Scholar] [CrossRef]
- Madec, J.Y.; Haenni, M. Antimicrobial resistance plasmid reservoir in food and food-producing animals. Plasmid 2018, 99, 72–81. [Google Scholar] [CrossRef]
- Oloso, N.O.; Fagbo, S.; Garbati, M.; Olonitola, S.O.; Awosanya, E.J.; Aworh, M.K.; Adamu, H.; Odetokun, I.A.; Fasina, F.O. Antimicrobial resistance in food animals and the environment in Nigeria: A review. Int. J. Environ. Res. Public Health 2018, 15, 1284. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Zhou, Y.; Li, J.; Yin, W.; Wang, S.; Zhang, S.; Shen, J.; Shen, Z.; Wang, Y. Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae. Emerg. Microbes. Infect. 2018, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Xavier, B.B.; Lammens, C.; Ruhal, R.; Kumar-Singh, S.; Butaye, P.; Goossens, H.; Malhotra-Kumar, S. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, Belgium, June 2016. Euro Surveill. 2016, 21, 30280. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.Q.; Li, Y.X.; Lei, C.W.; Zhang, A.Y.; Wang, H.N. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 1791–1795. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Walsh, T.R.; Liu, D.; Shen, Z.; Zhang, R.; Yin, W.; Yao, H.; Li, J.; Shen, J. Plasmid-mediated novel bla NDM-17 gene encoding a carbapenemase with enhanced activity in a sequence type 48 Escherichia coli strain. Antimicrob. Agents Chemother. 2017, 61, e02233-16. [Google Scholar] [CrossRef]
- Pulss, S.; Semmler, T.; Prenger-Berninghoff, E.; Bauerfeind, R.; Ewers, C. First report of an Escherichia coli strain from swine carrying an OXA-181 carbapenemase and the colistin resistance determinant MCR-1. Int. J. Antimicrob. Agents 2017, 50, 232–236. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Wang, Y.; Walsh, T.R.; Yi, L.X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Skov, R.L.; Monnet, D.L. Plasmid-mediated colistin resistance (mcr-1 gene): Three months later, the story unfolds. Euro Surveill. 2016, 21, 30155. [Google Scholar] [CrossRef]
- Tendencia, E.A.; de la Peña, L.D. Antibiotic resistance of bacteria from shrimp ponds. Aquaculture 2001, 195, 193–204. [Google Scholar] [CrossRef]
- Teophilo, G.; dos Fernandes Vieira, R.; dos Prazeres Rodrigues, D.; Menezes, F.R. Escherichia coli isolated from seafood: Toxicity and plasmid profiles. Int. Microbiol. 2002, 5, 11–14. [Google Scholar] [CrossRef]
- Petersen, A.; Dalsgaard, A. Species composition and antimicrobial resistance genes of Enterococcus spp., isolated from integrated and traditional fish farms in Thailand. Environ. Microbiol. 2003, 5, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N.; Singh, A.S.; Lekshmi, M.; Nayak, B.B.; Kumar, S. Characterization of bla NDM-harboring, multidrug-resistant Enterobacteriaceae isolated from seafood. Environ. Sci. Pollut. Res. Int. 2019, 26, 2455–2463. [Google Scholar] [CrossRef]
- Beshiru, A.; Igbinosa, I.H.; Igbinosa, E.O. Prevalence of antimicrobial resistance and virulence gene elements of Salmonella serovars from ready-to-eat (RTE) shrimps. Front. Microbiol. 2019, 10, 1613. [Google Scholar] [CrossRef]
- Wegener, H.C. Antibiotic resistance—Linking human and animal health. In Improving Food Safety through a One Health Approach: Workshop Summary; Choffnes, E.R., Relman, D.A., Olsen, L., Hutton, R., Mack, A., Eds.; The National Academies Press: Washington, DC, USA, 2012; p. 331. ISBN 978-0-309-25933-0. [Google Scholar]
- Andersson, D.I.; Hughes, D. Microbiological effects of sublethal levels of antibiotics. Nat. Rev. Microbiol. 2014, 12, 465–478. [Google Scholar] [CrossRef] [PubMed]
- Subirats, J.; Domingues, A.; Topp, E. Does dietary consumption of antibiotics by humans promote antibiotic resistance in the gut microbiome? J. Food. Prot. 2019, 82, 1636–1642. [Google Scholar] [CrossRef] [PubMed]
- Adesokan, H.K.; Akanbi, I.M.; Akanbi, I.O.; Obaweda, R.A. Pattern of antimicrobial usage in livestock animals in south-western Nigeria: The need for alternative plans. Onderstepoort. J. Vet. Res. 2015, 82, 1–6. [Google Scholar] [CrossRef]
- Adetunji, V.O.; Olaoye, O.O. Detection of β-Lactam antibiotics (Penicillin and Amoxicillin) residues in Goat milk. Nat. Sci. 2012, 10, 60–64. [Google Scholar]
- FAO (Food and Agriculture Organization of the United Nations). Codex Alimentarius. International Food Standars. Available online: http://www.fao.org/fao-who-codexalimentarius/roster/detail/en/c/455337/ (accessed on 15 June 2021).
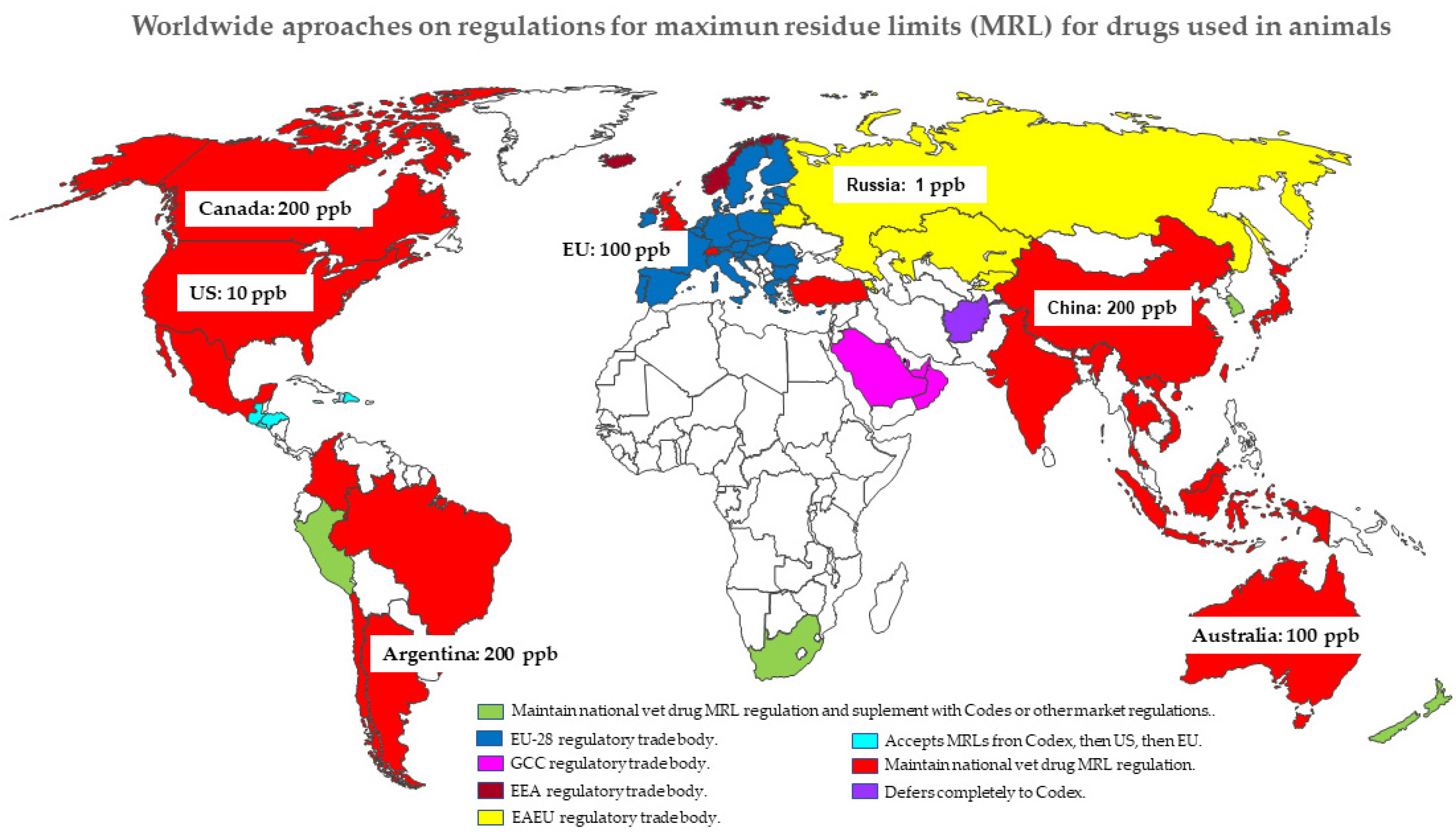
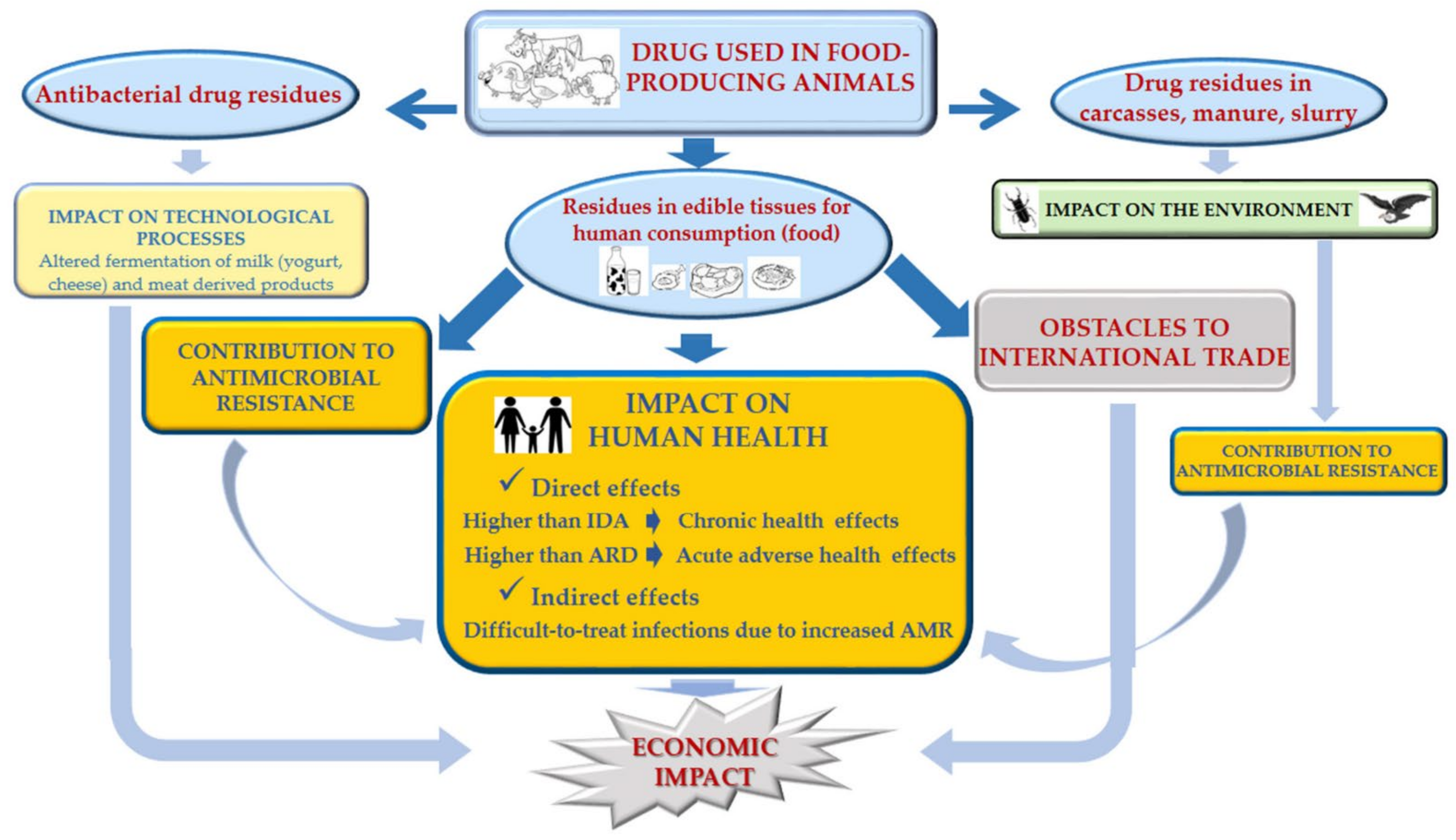
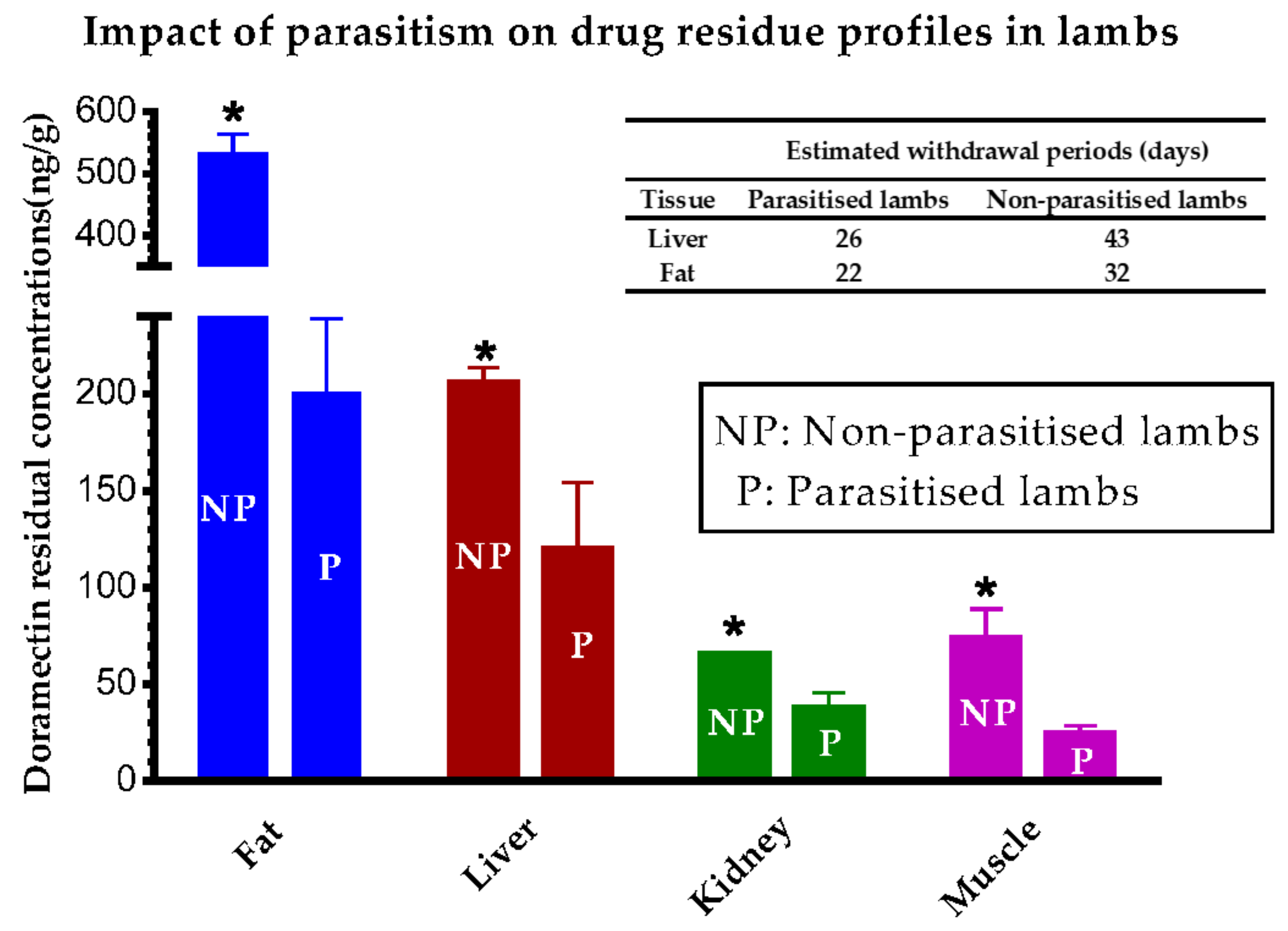
| Official Agencies/Bodies | Area of Application | Links |
|---|---|---|
| Codex Alimentarius The Codex Alimentarius Commission (CAC) is the body responsible for all matters regarding the implementation of the Joint FAO/WHO Food Standards Programme. | International |
|
| European Medicines Agency (EMA) EMA is a decentralized agency of the European Union (EU) responsible for the scientific evaluation, supervision, and safety monitoring of medicines. | EU | • http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/landing/vet_mrl_search.jsp&mid=WC0b01ac058006488e (accessed on 30 September 2021) |
| The Food and Drug Administration (FDA) FDA is an agency within the U.S. Department of Health and Human Services. It consists of the Office of the Commissioner and four directorates overseeing the core functions of the agency: Medical Products and Tobacco, Foods and Veterinary Medicine, Global Regulatory Operations and Policy, and Operations. | USA | • https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=556&showFR=1 (accessed on 30 September 2021) |
| Government of Canada Health Canada through the Veterinary Drugs Directorate (VDD) evaluates and monitors the safety, quality and effectiveness, sets standards, and promotes the prudent use of veterinary drugs administered to food-producing and companion animals. | Canada |
|
| Australian Government Department of Agriculture, Water and the Environment. Federal Register of Legislation | Australia and New Zealand |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canton, L.; Lanusse, C.; Moreno, L. Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues. Animals 2021, 11, 2878. https://doi.org/10.3390/ani11102878
Canton L, Lanusse C, Moreno L. Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues. Animals. 2021; 11(10):2878. https://doi.org/10.3390/ani11102878
Chicago/Turabian StyleCanton, Lucila, Carlos Lanusse, and Laura Moreno. 2021. "Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues" Animals 11, no. 10: 2878. https://doi.org/10.3390/ani11102878
APA StyleCanton, L., Lanusse, C., & Moreno, L. (2021). Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues. Animals, 11(10), 2878. https://doi.org/10.3390/ani11102878






