Stakeholder Perceptions of Disease Management for Dairy Calves: “It’s Just Little Things That Make Such a Big Difference”
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection—Interviews and Participants
2.2. Data Analysis—Thematic Analysis
3. Results
3.1. Disease Occurrence and Treatments
3.1.1. Pneumonia, Diarrhoea, and Mortality
“Scours we don’t get so much of a problem with. We could count the number of cases on one hand that we get like in 6 months, or even a year.”F9, farm manager.
“I would say scour is by far the most common, but a lot of farmers don’t perceive it as a problem. They’ll know the ones that die because of it, but they’ll often massively underestimate how much of a problem it is.”V11, youngstock veterinarian.
“The change in the Red Tractor standards [assurance scheme] that came in 3–4 years ago, when it’s one of the requirements to actually track your calf mortality, and I think a lot of people maybe didn’t even know that. I think it’s quite scary.”DR1, pharmaceutical company veterinary advisor.
“Mortality rate’s really, really low. Might have about one a month or something, I don’t even know that it would be that. I think we were below double figures last year and the year before.”F11, farm administrator (herd size approximately 400, all-year-round calving).
“You could persevere with some—we put them to sleep. They’re just gonna be poor doers and I don’t think it’s worth wasting several thousand pounds on rearing them when they’re just probably gonna give us poor lactations and just be problems … I’d rather get rid of them after a few weeks.”F24, farm manager.
“There’re a few cows … they’ve had pneumonia as calves and it’s just coming out now, maybe second lactation, and it really, really hurt them.”F22, herd manager.
3.1.2. Understanding of Disease Processes and Treatments
“What we’ve found works best [is] the Metacam [meloxicam, Boehringer Ingelheim Animal Health UK Ltd., Bracknell, UK] anti-inflammatory painkiller. Just gets the calf up on its feet. You get it up, you get it eating, you get it drinking again … feeling better in itself, regardless [of] whether the infection’s gone, you’ve got a lot more chance of him coming right.”F17, farm worker on calf rearing unit.
3.1.3. Antimicrobial Stewardship
“I do the computer work for another farm down the road, and they just feed their calves blanket antibiotics in their milk because they have lots of problems with them and … that’s the easiest, quickest fix.”F2, calf rearer.
“A very big calf rearer … he said … ‘I buy 2000 calves a year and we don’t worry about BVD … because we feed antibiotic milk powder for five days when they arrive on farm’ … We cannot be doing that as an industry; that is not acceptable practice … that kind of stuff really frustrates me.”DR1, pharmaceutical company veterinary advisor. Concerns were not limited to certain individuals. One veterinarian (V4) criticised the inattention to antibiotic usage stemming from treatments against Mycoplasma bovis within the dairy industry.
“Mycoplasma bovis in the national dairy herd is a huge, huge problem. No one talks about it … It’s probably the single biggest cause of antibiotic usage in dairy replacements.”V4, farm veterinarian.
“Particularly the older generation, ‘Why do I want to give them electrolytes when I could give them a pill that works?’ And you’re going … ‘There’s no reason to give antibiotics to something with rotavirus.’ It’s really difficult. Sometimes you just have to let them carry on giving the pills, provided they do the other things that you want them to do as well … [they think] it’s only the pills that have worked and nothing else…”V7, farm veterinarian.
“If we get any scouring calves, we’ll take a [faecal] sample and give it to the vets and they’ll test for what’s actually wrong with it, and then we’ll be injecting them with the right drug … the medicines are expensive, so we don’t want to be injecting them and not utilising the medicine, and we don’t want to use more antibiotics than we need to.”F1, calf rearer.
“Nine times out of ten you know it’s gonna be [coccidiosis] so I just dose [the calf] anyway and they seem to clear up. Trouble is that … you take [faecal] samples to the vets and [they say] ‘Oh yeah, but it’s at a very low level, just see how it goes on’ … It goes on, then a week later it’s got even worse, and you think ‘Well, I could’ve treated that a week ago and it wouldn’t have been knocked back.’”F14, calf rearer.
3.1.4. The Importance of Early Treatment Interventions
“With a calf, you have to be on it. They’re babies. If they’re not very well, the next day they’re nearly dying.”F18, calf rearer.
“If you [detect illnesses] quickly, then [calves] respond much better to treatment than if you leave them until they’re really sick.”F2, calf rearer.
“Say a calf looks a little bit sick, maybe it’s got its ear down, or you just know them, they just look a little bit off colour. Someone else would walk in the shed and say ‘What are you worried about? That calf looks fine!’ But you know it’s not. Take its temperature!”F18, calf rearer.
“There’s a couple of farms I can think of, where if the stockman who rears the calves goes away for the weekend and the farmer rears the calves, there’ll be two or three dead calves come Monday because he’s not fed them right, or he’s not spotted the signs early enough.”V3, youngstock veterinarian.
“I’ve looked at these tags that flash a light at you if [the calf is] hot … I might do a trial on that; do a couple of pens, and you’re not allowed to treat them for antibiotic unless you see there’s a temperature for pneumonia. That could cut our antibiotic usage … They’re quite expensive … I wouldn’t do it unless it saves us money.”F20, farm manager.
3.1.5. Vaccines
“There’s a couple of people who have horrible pneumonia problems and the shed’s not designed for [calves]; they’re overstocked and they’ve mixed different age groups—so they’re doing everything wrong and yet they still won’t vaccinate and it’s just madness!”V8, farm veterinarian.
“We haven’t recovered properly from the [milk] price slump yet, and so to start a new vaccination policy and everything like that, I just don’t think it would be looked upon well.”F22, herd manager.
“Some years, we were treating virtually all the youngstock [for pneumonia], whereas now we get one, two. So [the vaccine has] made a great difference.”F15, calf rearer and farm worker.
“It’s very frustrating because we’ve started vaccinating everything for pneumonia but yet we still have to treat a lot of [calves] with pneumonia.”F24, farm manager.
“We regularly take blood samples of calves that contract pneumonia, even though they’ve been vaccinated, to try to identify the strains and make sure that it’s being covered by the vaccine, or if it wasn’t administered correctly, or if the vaccine simply didn’t work.”F9, farm manager.
“We’ve got any number of vaccines available, and yet they still don’t cover all the infectious causes of pneumonia. And we keep getting hammered by drug companies that we don’t sell enough pneumonia vaccine! Well actually, if we got the colostrum right, and we got the feeding right, and we got the environment right, we wouldn’t need any!”V8, farm veterinarian.
“Part of the expectation of the new calf unit was that we would not need to vaccinate any more … We did have an element of pneumonia in the new calf unit last year, which was disappointing.”F26, farm manager.
3.2. Management of Calf Environment
3.2.1. Calf Group Management
“I’m highly satisfied with all disease levels. Diseases are pretty low in their hutches.”F7, calf rearer and farm manager.
“I wanted to make sure everything went through a single hutch and then went on to group hutches … you end up cleaning out a group hutch, put some calves in, then you have to clean out [the single hutches] as well. Before I came, they didn’t really bother doing that. They just cleaned out the group hutch and then just put the new [calves] in there instead.”F3, calf rearer and farm worker.
“I do like putting them into the big hutches and seeing them run around and seeing them mix and interact, but it is just interesting that we’ve had real variance in growth rates from the group hutches [prior to weaning compared to individually housed calves in a trial].”F1, calf rearer.
“A lot of the guys will pen individually and feed individually and I think that massively reduces scours, but then probably increases pneumonia further along.”V4, farm veterinarian.
“It’s important to keep them in very small groups of a similar age, and don’t mix them.”F10, farm manager.
“Instead of trying to limit it to number, limit it to age range. If we think of how the diseases spread, it’s normally a 10 day to 2 week cycle. So if you open a pen and only fill it for 2 weeks and then shut that pen, independent of whether it’s got three calves in it or 10 calves, which is really the maximum I like to go with, then that’ll stop your disease spread.”V11, youngstock veterinarian.
“One thing that we are quite careful with is that we use the loader that hasn’t been in all the muck where all the Johne’s is. A lot of farms, they’ll do everything for Johne’s, but then they’ll just drive in and scrape up with the tractor that scraped up all the cow muck.”F1, calf rearer.
“We do go from adult cows to young calves. We don’t go in the pens with the calves without washing our wellies [boots].”F9, farm manager.
“Overstocking is probably the biggest issue … we had so many I had to double up all the single hutches.”F3, calf rearer and farm worker.
“One of the sheds that we were moving calves into had older cattle in, because we were a bit tight for space. The vet said that ’older cattle are resistant to a lot of the pneumonias, but of course they’re still breathing out the virus, so then if you’re putting youngstock in with older ones, then you’re at high risk of passing on.’ So we’ve tried to break that link [by putting up some new sheds as calf housing].”F5, farm manager.
3.2.2. Thermal Comfort and Hygiene within Calf Accommodation
“We did think about putting a cover over the back, like they do with pigs, with some straw on top to keep them warm. The vet suggested it, actually. We might do it, but it hasn’t been too bad this year; we’ve had a reasonably kind winter.”F20, farm manager.
“We’ll put a calf jacket on anything that has had the scours, really, and just looks generally not very thrifty.”F20, farm manager.
“I don’t know when you would take a calf jacket off. Some people say once it goes on, it stays on until you’ve weaned them … But if you put a jacket on like for a week, and then take it off, does a calf go back [lose condition] then? I don’t know.”F14, calf manager.
“I don’t want the calves to ever look dirty … If they look dirty, I’m a day late with the straw going in … I get moaned at by dad because he thinks I use too much straw, but it saves me [using] antibiotics.”F19, farm manager.
“I’ve had a lot of coccidiosis in that one particular pen, but you can’t just go and clean that one pen out … you have to clean the whole shed out. Well, then you sort of think ‘it can wait another week’. Things like that don’t get done as quick as what they should because it’s quite a job to push everything out, take all the pens down, then clean out, then put it all back up again.”F14, calf rearer.
“We don’t have enough space, so we can’t have [the pens] resting. It’s a day’s rest. They’re cleaned out, they’re sprayed with peracetic acid, washed down with it, and then left ‘til they dry, but it’s not that long. A nice drying day like today helps. A wet, drizzly day and they don’t really ever dry out.”F19, farm manager.
“Leaving a shed clean, dry and empty for a few months massively reduces the pathogen challenge … You see a lot less disease, especially at the start of the block. It might build up towards the end of the block, but compared to these guys who are constantly housed, it definitely helps.”V3, youngstock veterinarian.
3.2.3. Investing in Calf Accommodation
“Access to clean out the shed is very difficult … And the floors, they should be on more of an angle … but they’re reasonably flat, so drainage into all the drains isn’t particularly great. It’s not the best calf shed, really, considering we’ve got this nice dairy.”F24, herd manager.
“Most buildings in the UK are old buildings that you use for calves. You’ll spend money on your buildings for your cows, but you won’t spend it on the calves. Calves go in some poorly ventilated, or cold, damp area.”V2, youngstock veterinarian.
“It all comes down to money at the end of the day; so, it’s a shed here empty, so we use it and you’ve gotta make the most of it and just get on with it … We’ve got a couple of those Igloo things [group hutches], I hate them … I’ve never seen so many ill calves … [The farm manager] said we could put a concrete slab there and use those Igloos and I said, ‘Nah—I’d rather use this [shed that’s not ideal]!’”F23, farm worker and calf rearer.
“Some vets have this similar sort of mindset: in an ideal world you could do [with] a new space, well it’s not an ideal world, so what are we gonna do? Some don’t have that, they come out with theory … we all read the same books, but how do we get different results [on our farm]?”F25, farm manager.
“Eventually we came to the conclusion that we had to spend some money, this [the new calf accommodation] was desperately needed [to improve calf health].”F26, farm manager.
“The farmer may know that the shed he rears the calves in is just awful … but he also knows he hasn’t got x thousand pounds to put up a new one … He’d need to be very convinced that if he goes out and borrows x amount to put up a calf house that there is gonna be a return that will pay for his borrowings, and that can be a challenge.”V10, formerly practising veterinarian, now feed consultant.
“Sometimes you turn up to what feel like slightly helpless situations, where they’re going, ‘I know this shed is awful … I can’t deal with it now [because of financial pressures].’ … It can reach a brick wall where people are much happier to go, ‘Well it’s broken, we’ll just use the drugs’, than to really start investing their time and energy in patching together that shed.”V6, youngstock veterinarian.
3.2.4. Designing Replacement Calf Accommodation
“I have seen plenty of big, shiny units … that don’t necessarily perform as well as they were hoped by the person who designed them … People get advice from different sources, and often the animal health side of things only actually comes in once you’ve got animals in the shed and maybe they’re not performing.”DR1, pharmaceutical company veterinary advisor.
“If they could involve the vet more in building planning … I think we could save them thousands and thousands of pounds, but it’s often one of the last people that a farmer will consider speaking to is their vet when they’re putting up a new shed … Shed design is probably not something they think that we know an awful lot about.”V4, farm veterinarian.
“[A farm client] building this new shed … that had a 1 in 20 slope … When they were building it, he called me out because the builders were going, ‘We will do it, not a problem, but [1 in 20 is a very steep incline] on your head—are you sure?’ … If we commit the cardinal sin that has led to these sheds in the past of looking at it and going, ‘Phwar, bit steep, maybe a little less?’ then … it’ll still be 30,000 GBP [42,323 USD], it just won’t work as well as it might.”V6, youngstock veterinarian.
“The whole sheds are designed with multipurpose in mind. As time’s gone on, they’ve become more angled towards calves, but if things changed tomorrow and the money dropped out of calves, it’d probably take us a week to convert this shed into a pig shed.”F17, farm worker on calf rearing unit.
“Cost 7000 GBP [9876 USD] to build; that’s everything, metalwork, concrete panels. We can fit 42 calves in here … A lot of farmers would need to [get input from the veterinarian]. I went online and looked it up; it’s all on the internet … We wanted to make physical barriers so then we could … take this pen out, steam clean it, and that pen can still be there! … I know of two farmers that have copied it since we’ve done it.”F20, farm manager.
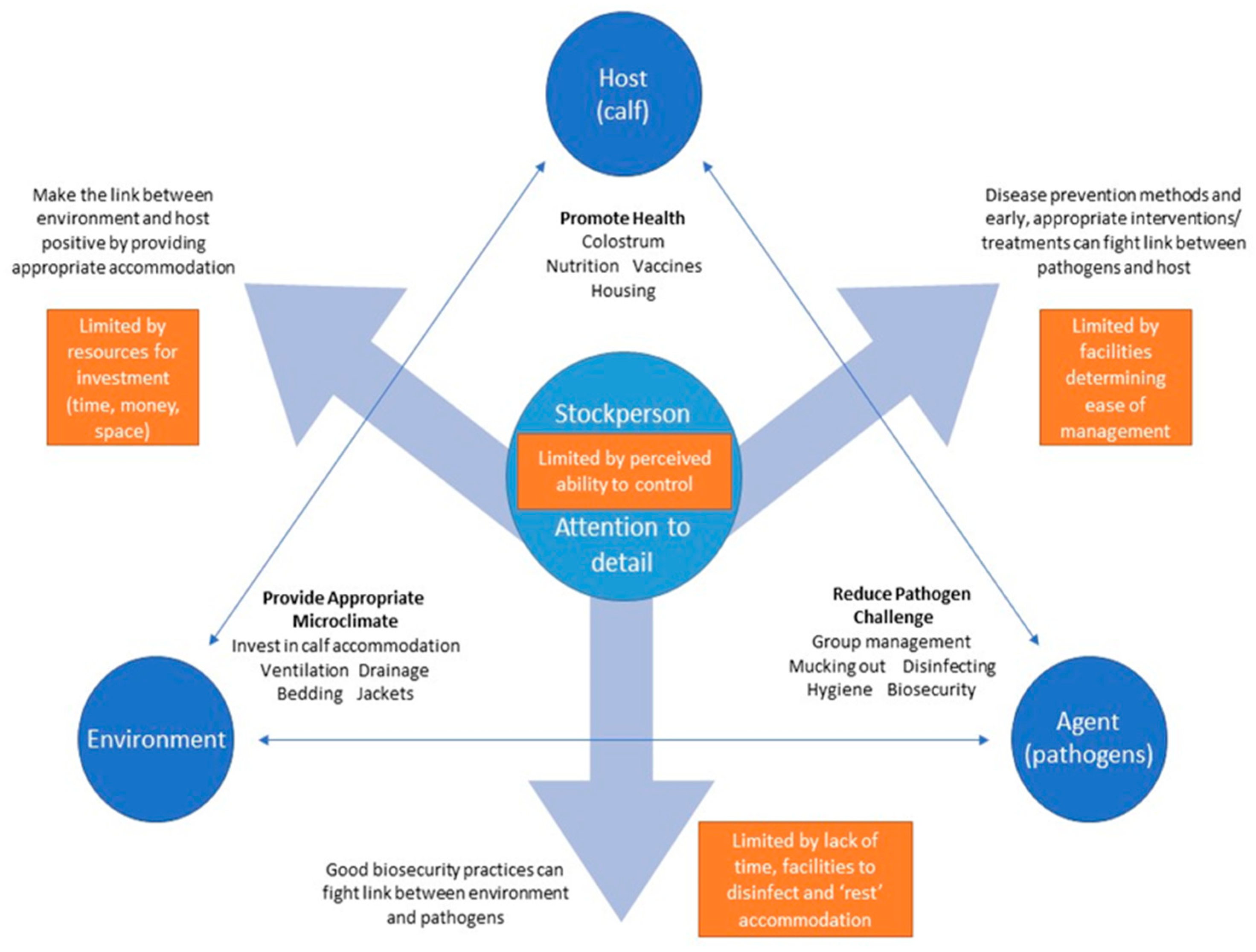
3.3. The Role of Stockmanship and Perceived Control
“It’s just little things that make such a big difference to calf rearing … if you’ve got a problem, deal with it straight away, and if you can move them to a fresh place, a fresh, clean place, that makes a huge difference.”F2, calf rearer.
“I’ve walked into some sheds that I have thought ‘[swears], this is an awful place to see calves’, and actually, when you look at the calves, they are growing really well—you can’t put a value on good husbandry.”DR1, pharmaceutical company veterinary advisor.
“You could have the most amazing shed in the world, but if you don’t have attention to detail of like the stupid little things … you’re never gonna get it right.”F18, calf rearer.
Perceived Control over Disease Processes
“If you’ve got unhealthy calves, it doesn’t matter what you do, you’re on a backwards spiral all the time. They’re not very well, then they don’t drink [milk], so then they don’t gain weight, and because they’re not getting that adequate nutrition, you get more health issues.”F2, calf rearer.
“We get pneumonia every single month of the year—even in the middle of summer … We vaccinate for it [pneumonia], we’re looking out for it all the time, we never lose any with it, but we do jab [inject] a lot of calves for pneumonia. There’s no sort of pattern to it … they’re bedded up well, but we still get it.”F8, farm manager of dairy bull calf rearing unit.
“Dad had two [calves] the other day that didn’t do very well. I don’t know what happened there; they looked like calves that missed their colostrum.”F19, farm manager.
“It’s rare that you get one die, I mean, you always get the odd one”(F14, calf rearer).
“I think it’s a waste of time rearing heifer calves if you have got BVD … We don’t have BVD so we’ve not got that sort of threat on them being pushed towards them getting pneumonia and scours and all that business.”F10, farm manager.
“You see some farms where they keep their calves in appalling conditions and never have any problems because … there’s no BVD.”V8, veterinarian.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boulton, A.C.; Rushton, J.; Wathes, D.C. An empirical analysis of the cost of rearing dairy heifers from birth to first calving and the time taken to repay these costs. Animal 2017, 11, 1372–1380. [Google Scholar] [CrossRef] [Green Version]
- Closs, G.; Dechow, C. The effect of calf-hood pneumonia on heifer survival and subsequent performance. Livest. Sci. 2017, 205, 5–9. [Google Scholar] [CrossRef]
- Al Mawly, J.; Grinberg, A.; Prattley, D.; Moffat, J.; Marshall, J.; French, N. Risk factors for neonatal calf diarrhoea and enteropathogen shedding in New Zealand dairy farms. Vet. J. 2015, 203, 155–160. [Google Scholar] [CrossRef]
- Johnson, K.F.; Burn, C.; Wathes, D.C. Rates and risk factors for contagious disease and mortality in young dairy heifers. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2011, 6, 59. [Google Scholar] [CrossRef]
- Brickell, J.S.; McGowan, M.M.; Pfeiffer, D.U.; Wathes, D.C. Mortality in Holstein-Friesian calves and replacement heifers, in relation to body weight and IGF-I concentration, on 19 farms in England. Animal 2009, 3, 1175–1182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baxter-Smith, K.; Simpson, R. Insights into UK farmers’ attitudes towards cattle youngstock rearing and disease. Livestock 2020, 25, 274–281. [Google Scholar] [CrossRef]
- Johnson, K.F.; Chancellor, N.; Burn, C.C.; Wathes, D.C. Prospective cohort study to assess rates of contagious disease in pre-weaned UK dairy heifers: Management practices, passive transfer of immunity and associated calf health. Vet. Rec. Open 2017, 4, e000226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinrichs, A.J.; Heinrichs, B.S. A prospective study of calf factors affecting first-lactation and lifetime milk production and age of cows when removed from the herd. J. Dairy Sci. 2011, 94, 336–341. [Google Scholar] [CrossRef]
- Bartram, D.; Hogan, C.; Penny, C. Estimating the lifetime total economic costs of respiratory disease in beef and dairy calves in the UK. Value Health 2017, 20, A643. [Google Scholar] [CrossRef]
- Brickell, J.S.S.; Wathes, D.C.C. A descriptive study of the survival of Holstein-Friesian heifers through to third calving on English dairy farms. J. Dairy Sci. 2011, 94, 1831–1838. [Google Scholar] [CrossRef]
- Gulliksen, S.M.; Jor, E.; Lie, K.I.; Hamnes, I.S.; Løken, T.; Åkerstedt, J.; Østerås, O. Enteropathogens and risk factors for diarrhea in Norwegian dairy calves. J. Dairy Sci. 2009, 92, 5057–5066. [Google Scholar] [CrossRef]
- Stott, A.W.; Humphry, R.W.; Gunn, G.J.; Higgins, I.; Hennessy, T.; O’Flaherty, J.; Graham, D.A. Predicted costs and benefits of eradicating BVDV from Ireland. Ir. Vet. J. 2012, 65, 12. [Google Scholar] [CrossRef]
- Evans, C.A.; Pinior, B.; Larska, M.; Graham, D.; Schweizer, M.; Guidarini, C.; Decaro, N.; Ridpath, J.; Gates, M.C. Global knowledge gaps in the prevention and control of bovine viral diarrhoea (BVD) virus. Transbound. Emerg. Dis. 2019, 66, 640–652. [Google Scholar] [CrossRef]
- Peterhans, E.; Jungi, T.W.; Schweizer, M. BVDV and innate immunity. Biologicals 2003, 31, 107–112. [Google Scholar] [CrossRef]
- BVDFree England. The Scheme. Available online: https://bvdfree.org.uk/the-scheme (accessed on 11 July 2021).
- Reardon, F.; Graham, D.A.; Clegg, T.A.; Tratalos, J.A.; O’Sullivan, P.; More, S.J. Quantifying the role of Trojan dams in the between-herd spread of bovine viral diarrhoea virus (BVDv) in Ireland. Prev. Vet. Med. 2018, 152, 65–73. [Google Scholar] [CrossRef]
- CHAWG. Cattle Health and Welfare Group Antimicrobial Usage Subgroup (CHAWG AMU) Recommendations for Measuring and Comparing the Use of Antibiotics on UK Beef Farms; CHAWG: Kenilworth, UK, 2020. [Google Scholar]
- Lorenz, I.; Fagan, J.; More, S.J. Calf health from birth to weaning. II. Management of diarrhoea in pre-weaned calves. Ir. Vet. J. 2011, 64, 9. [Google Scholar] [CrossRef] [Green Version]
- Constable, P.D. Treatment of Calf Diarrhea: Antimicrobial and Ancillary Treatments. Vet. Clin. N. Am. Food Anim. Pract. 2009, 25, 101–120. [Google Scholar] [CrossRef]
- Philipp, H.; Schmidt, H.; Düring, F.; Salamon, E. Efficacy of Meloxicam (Metacam®) as Adjunct to a Basic Therapy for the Treatment of Diarrhoea in Calves. Acta Vet. Scand. 2003, 44, P95. [Google Scholar] [CrossRef] [Green Version]
- Todd, C.G.; Millman, S.T.; McKnight, D.R.; Duffield, T.F.; Leslie, K.E. Nonsteroidal anti-inflammatory drug therapy for neonatal calf diarrhea complex: Effects on calf performance1. J. Anim. Sci. 2010, 88, 2019–2028. [Google Scholar] [CrossRef] [PubMed]
- Laven, R. An introduction to NSAIDs for cattle: Recommendations for the veterinary surgeon. Livestock 2020, 25, 174–177. [Google Scholar] [CrossRef]
- Lorenz, I.; Earley, B.; Gilmore, J.; Hogan, I.; Kennedy, E.; More, S.J. Calf health from birth to weaning. III. housing and management of calf pneumonia. Ir. Vet. J. 2011, 64, 14. [Google Scholar] [CrossRef] [Green Version]
- Mahendran, S.A.; Booth, R.; Burge, M.; Bell, N.J. Randomised positive control trial of NSAID and antimicrobial treatment for calf fever caused by pneumonia. Vet. Rec. 2017, 181, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenz, I.; Mee, J.F.; Earley, B.; More, S.J. Calf health from birth to weaning. I. General aspects of disease prevention. Ir. Vet. J. 2011, 64, 10. [Google Scholar] [CrossRef] [PubMed]
- Responsible Use of Medicines in Agriculture (RUMA) Alliance. Targets Task Force Report 2020: Responsible Use of Antibiotics in UK Farming—Progress against 2020 Targets, New Targets 2021–2024. RUMA. Available online: SO-469-RUMA-REPORT-021220.pdf (accessed on 4 September 2021).
- Pfeiffer, D.U. Veterinary Epidemiology—An Introduction; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Johnson, K.F.; Chancellor, N.; Wathes, D.C. A cohort study risk factor analysis for endemic disease in pre-weaned dairy heifer calves. Animals 2021, 11, 378. [Google Scholar] [CrossRef]
- Gorden, P.J.; Plummer, P. Control, Management, and Prevention of Bovine Respiratory Disease in Dairy Calves and Cows. Vet. Clin. N. Am. Food Anim. Pract. 2010, 26, 243–259. [Google Scholar] [CrossRef]
- Nordlund, K.V.; Halbach, C.E. Calf Barn Design to Optimize Health and Ease of Management. Vet. Clin. N. Am. Food Anim. Pract. 2019, 35, 29–45. [Google Scholar] [CrossRef]
- Costa, J.H.C.; von Keyserlingk, M.A.G.; Weary, D.M. Invited review: Effects of group housing of dairy calves on behavior, cognition, performance, and health. J. Dairy Sci. 2016, 99, 2453–2467. [Google Scholar] [CrossRef]
- Curtis, G.C.; Argo, C.M.; Jones, D.; Grove-White, D.H. Impact of feeding and housing systems on disease incidence in dairy calves. Vet. Rec. 2016, 179, 512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haskell, M.J. What to do with surplus dairy calves? Welfare, economic, and ethical considerations. Landbauforsch. J. Sustain. Org. Agric. Syst. 2020, 70, 45–48. [Google Scholar]
- Butler, A.; Lobley, M.; Winter, M. Economic Impact Assessment of Bovine Tuberculosis in the South West of England; University of Exeter: Exeter, UK, 2010. [Google Scholar]
- Brennan, M.L.; Christley, R.M. Biosecurity on Cattle Farms: A Study in North-West England. PLoS ONE 2012, 7, e28139. [Google Scholar] [CrossRef] [Green Version]
- Brennan, M.; Wright, N.; Wapenaar, W.; Jarratt, S.; Hobson-West, P.; Richens, I.; Kaler, J.; Buchanan, H.; Huxley, J.; O’Connor, H. Exploring Attitudes and Beliefs towards Implementing Cattle Disease Prevention and Control Measures: A Qualitative Study with Dairy Farmers in Great Britain. Animals 2016, 6, 61. [Google Scholar] [CrossRef] [Green Version]
- Sumner, C.L.; von Keyserlingk, M.A.G.; Weary, D.M. Perspectives of farmers and veterinarians concerning dairy cattle welfare. Anim. Front. 2018, 8, 8–13. [Google Scholar] [CrossRef]
- Hansen, B.G.; Greve, A. Dairy farmers’ values and how their values affect their decision making. Agric. Food Sci. 2014, 23, 278–290. [Google Scholar] [CrossRef] [Green Version]
- Hansson, H.; Lagerkvist, C.J. Dairy farmers’ use and non-use values in animal welfare: Determining the empirical content and structure with anchored best-worst scaling. J. Dairy Sci. 2016, 99, 579–592. [Google Scholar] [CrossRef] [Green Version]
- Santman-Berends, I.M.G.A.; Buddiger, M.; Smolenaars, A.J.G.; Steuten, C.D.M.; Roos, C.A.J.; Van Erp, A.J.M.; Van Schaik, G. A multidisciplinary approach to determine factors associated with calf rearing practices and calf mortality in dairy herds. Prev. Vet. Med. 2014, 117, 375–387. [Google Scholar] [CrossRef]
- Vaarst, M.; Sørensen, J.T. Danish dairy farmers’ perceptions and attitudes related to calf-management in situations of high versus no calf mortality. Prev. Vet. Med. 2009, 89, 128–133. [Google Scholar] [CrossRef]
- Leach, K.A.; Whay, H.R.; Maggs, C.M.; Barker, Z.E.; Paul, E.S.; Bell, A.K.; Main, D.C.J.J. Working towards a reduction in cattle lameness: 2. Understanding dairy farmers’ motivations. Res. Vet. Sci. 2010, 89, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Palczynski, L.J.; Bleach, E.C.L.; Brennan, M.L.; Robinson, P.A. Giving calves “the best start”: Perceptions of colostrum management on dairy farms in England. Anim. Welf. 2020, 29, 45–58. [Google Scholar] [CrossRef]
- Palczynski, L.J.; Bleach, E.C.L.; Brennan, M.L.; Robinson, P.A. Appropriate Dairy Calf Feeding from Birth to Weaning: “It’s an Investment for the Future”. Animals 2020, 10, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sumner, C.L.; von Keyserlingk, M.A.G.; Weary, D.M. How benchmarking motivates farmers to improve dairy calf management. J. Dairy Sci. 2018, 101, 3323–3333. [Google Scholar] [CrossRef] [PubMed]
- Vetouli, T.; Lund, V.; Kaufmann, B. Farmers’ Attitude Towards Animal Welfare Aspects and Their Practice in Organic Dairy Calf Rearing: A Case Study in Selected Nordic Farms. J. Agric. Environ. Ethics 2012, 25, 349–364. [Google Scholar] [CrossRef]
- Garforth, C. Livestock keepers’ reasons for doing and not doing things which governments, vets and scientists would like them to do. Zoonoses Public Health 2015, 62, 29–38. [Google Scholar] [CrossRef]
- May, C. Discovering new areas of veterinary science through qualitative research interviews: Introductory concepts for veterinarians. Aust. Vet. J. 2018, 96, 278–284. [Google Scholar] [CrossRef]
- Robinson, P.A. “They’ve got to be testing and doing something about it”: Farmer and veterinarian views on drivers for Johne’s disease control in dairy herds in England. Prev. Vet. Med. 2020, 182, 105094. [Google Scholar] [CrossRef]
- Redfern, E.A.; Sinclair, L.A.; Robinson, P.A. Dairy cow health and management in the transition period: The need to understand the human dimension. Res. Vet. Sci. 2021, 137, 94–101. [Google Scholar] [CrossRef]
- Maxwell, J.A. A Realist Approach to Qualitative Research; Sage Publications: Thousand Oaks, CA, USA, 2012; ISBN 10-0761929231. [Google Scholar]
- Palczynski, L.J.; Bleach, E.C.L.; Brennan, M.L.; Robinson, P.A. Calf Management as “the Key for Everything”? Perceived Value of Youngstock and the Role of Advice and Calf Performance Monitoring on Dairy Farms, Unpublished paper in preparation.
- Tong, A.; Sainsbury, P.; Craig, J. Consolidated criteria for reporting qualitative research (COREQ): A 32-item checklist for interviews and focus groups. Int. J. Qual. Health Care 2007, 19, 349–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, L.; Manion, L.; Morrison, K. Research Methods in Education, 6th ed.; Routledge: London, UK, 2007; ISBN 0-203-02905-4. [Google Scholar]
- Turner, D.W. Qualitative Interview Design: A Practical Guide for Novice Investigators. Qual. Rep. 2010, 15, 754–760. [Google Scholar]
- Miles, M.B.; Huberman, A.M.; Saldana, J. Qualitative Data Analysis: A Methods Sourcebook, 3rd ed.; Sage Publications: Thousand Oaks, CA, USA, 2014. [Google Scholar]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef] [Green Version]
- Burton, R.J.F.; Peoples, S.; Cooper, M.H. Building ‘cowshed cultures’: A cultural perspective on the promotion of stockmanship and animal welfare on dairy farms. J. Rural Stud. 2012, 28, 174–187. [Google Scholar] [CrossRef] [Green Version]
- Vasseur, E.; Pellerin, D.; de Passillé, A.; Winckler, C.; Lensink, B.; Knierim, U.; Rushen, J. Assessing the welfare of dairy calves: Outcome-based measures of calf health versus input-based measures of the use of risky management practices. Anim. Welf. 2012, 21, 77–86. [Google Scholar] [CrossRef] [Green Version]
- Torsein, M.; Lindberg, A.; Sandgren, C.H.; Waller, K.P.; Törnquist, M.; Svensson, C. Risk factors for calf mortality in large Swedish dairy herds. Prev. Vet. Med. 2011, 99, 136–147. [Google Scholar] [CrossRef]
- Holstege, M.M.C.; de Bont-Smolenaars, A.J.G.; Santman-Berends, I.M.G.A.; van der Linde-Witteveen, G.M.; van Schaik, G.; Velthuis, A.G.J.; Lam, T.J.G.M. Factors associated with high antimicrobial use in young calves on Dutch dairy farms: A case-control study. J. Dairy Sci. 2018, 101, 9259–9265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sumner, C.L.; von Keyserlingk, M.A.G.; Weary, D.M. How benchmarking promotes farmer and veterinarian cooperation to improve calf welfare. J. Dairy Sci. 2020, 103, 702–713. [Google Scholar] [CrossRef] [Green Version]
- Morgans, L.; Reyher, K.K.; Barrett, D.C.; Turner, A.; Bellini, J.; Elkins, P.; Clarke, T. Changing farmer and veterinarian behaviour around antimicrobial use. Livestock 2019, 24, 75–80. [Google Scholar] [CrossRef]
- Morgans, L.C.; Bolt, S.; Bruno-McClung, E.; van Dijk, L.; Escobar, M.P.; Buller, H.J.; Main, D.C.J.; Reyher, K.K. A participatory, farmer-led approach to changing practices around antimicrobial use on UK farms. J. Dairy Sci. 2021, 104, 2212–2230. [Google Scholar] [CrossRef] [PubMed]
- Overton, M.W.; Dhuyvetter, K.C. Symposium review: An abundance of replacement heifers: What is the economic impact of raising more than are needed? J. Dairy Sci. 2020, 103, 3828–3837. [Google Scholar] [CrossRef]
- McGuirk, S.M. Disease Management of Dairy Calves and Heifers. Vet. Clin. N. Am. Food Anim. Pract. 2008, 24, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Klerkx, L.; Jakku, E.; Labarthe, P. A review of social science on digital agriculture, smart farming and agriculture 4.0: New contributions and a future research agenda. NJAS—Wagening. J. Life Sci. 2019, 90, 100315. [Google Scholar] [CrossRef]
- Van Erp-van der Kooij, E. Using precision farming to improve animal welfare. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2020, 15, 1–10. [Google Scholar]
- Bach, A.; Ahedo, J. Record Keeping and Economics of Dairy Heifers. Vet. Clin. N. Am. Food Anim. Pract. 2008, 24, 117–138. [Google Scholar] [CrossRef]
- The Veterinary Medicines Regulations. 2013. Available online: https://www.legislation.gov.uk/uksi/2013/2033/contents/2019-03-26 (accessed on 12 July 2021).
- Shaw, H.J.; Innes, E.A.; Morrison, L.J.; Katzer, F.; Wells, B. Long-term production effects of clinical cryptosporidiosis in neonatal calves. Int. J. Parasitol. 2020, 50, 371–376. [Google Scholar] [CrossRef]
- Williams, P.D.; Paixão, G. On-farm storage of livestock vaccines may be a risk to vaccine efficacy: A study of the performance of on-farm refrigerators to maintain the correct storage temperature. BMC Vet. Res. 2018, 14, 136. [Google Scholar] [CrossRef] [PubMed]
- Cresswell, E.; Brennan, M.L.; Barkema, H.W.; Wapenaar, W. A questionnaire-based survey on the uptake and use of cattle vaccines in the UK. Vet. Rec. Open 2013, 1, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Constable, P.D. Antimicrobial Use in the Treatment of Calf Diarrhea. J. Vet. Intern. Med. 2004, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Thickett, B.; Mitchell, D.; Hallows, B. Calf health. In Calf Rearing; Farming Press Ltd.: Ipswich, UK, 1986; pp. 122–161. [Google Scholar]
- Bard, A.M.; Main, D.; Roe, E.; Haase, A.; Whay, H.R.; Reyher, K.K. To change or not to change? Veterinarian and farmer perceptions of relational factors influencing the enactment of veterinary advice on dairy farms in the United Kingdom. J. Dairy Sci. 2019, 102, 10379–10394. [Google Scholar] [CrossRef] [PubMed]
- Marcé, C.; Guatteo, R.; Bareille, N.; Fourichon, C. Dairy calf housing systems across Europe and risk for calf infectious diseases. Animal 2010, 4, 1588–1596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krawczel, P.D. Group Housing for Dairy Calves—A viable Option with Potential Benefits; UT Extension Publications: Knoxville, TN, USA, 2016; Volume W382, pp. 1–3. [Google Scholar]
- Chamberlain, T. Environmental aspects of pneumonia control in calf units on British dairy farms. Livestock 2015, 20, 306–314. [Google Scholar] [CrossRef]
- Maunsell, F.; Donovan, G.A. Biosecurity and Risk Management for Dairy Replacements. Vet. Clin. N. Am. Food Anim. Pract. 2008, 24, 155–190. [Google Scholar] [CrossRef]
- Garforth, C.J.; Bailey, A.P.; Tranter, R.B. Farmers’ attitudes to disease risk management in England: A comparative analysis of sheep and pig farmers. Prev. Vet. Med. 2013, 110, 456–466. [Google Scholar] [CrossRef]
- Pothmann, H.; Nechanitzky, K.; Sturmlechner, F.; Drillich, M. Consultancy to dairy farmers relating to animal health and herd health management on small- and medium-sized farms. J. Dairy Sci. 2014, 97, 851–860. [Google Scholar] [CrossRef]
- Innovative Farmers Field Labs. Available online: https://www.innovativefarmers.org/about/what-is-a-field-lab (accessed on 12 July 2021).
- Medrano-Galarza, C.; LeBlanc, S.J.; DeVries, T.J.; Jones-Bitton, A.; Rushen, J.; Marie de Passillé, A.; Haley, D.B. A survey of dairy calf management practices among farms using manual and automated milk feeding systems in Canada. J. Dairy Sci. 2017, 100, 6872–6884. [Google Scholar] [CrossRef] [PubMed]
- Vasseur, E.; Rushen, J.; de Passillé, A.M.; Lefebvre, D.; Pellerin, D. An advisory tool to improve management practices affecting calf and heifer welfare on dairy farms. J. Dairy Sci. 2010, 93, 4414–4426. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.; Lewis, M.; Crane, R.; Robertson, P.; Bonner, J.; Davenport, R.; Riley, M. FarmLevel Performance: Identifying Common Factors Determining Levels of Performance; Rural Business Research Report; RBR: Ottawa, ON, Canada, 2012. [Google Scholar]
- Delves, J. How to Succeed in Dairy Farming: Attitude Determines Altitude; A Nuffield Farming Scholarships Trust Report. Available online: https://www.nuffieldscholar.org/sites/default/files/reports/2012_UK_Joe-Delves_How-To-Succeed-In-Dairy-Farming-Attitude-Determines-Altitude.pdf (accessed on 4 September 2021).
- Agriculture and Horticulture Development Board (AHDB). Preparing for Change: The Characteristics of Top Performing Farms; Market Intelligence September 2018; AHDB: Kenilworth, UK, 2018; Available online: https://ahdb.org.uk/knowledge-library/preparing-for-change-the-characteristics-of-top-performing-farms (accessed on 4 September 2021).
- Mee, J.F. The role of the veterinarian in bovine fertility management on modern dairy farms. Theriogenology 2007, 68, S257–S265. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.C.; Keating, C.; Morris, C. Understand How to Influence Farmers’ Decision-Making Behaviour: A Social Science Literature Review; AHDB: Kenilworth, UK, 2018; Available online: https://ahdb.org.uk/knowledge-library/understand-how-to-influence-farmers-decision-making-behaviour (accessed on 4 September 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palczynski, L.J.; Bleach, E.C.L.; Brennan, M.L.; Robinson, P.A. Stakeholder Perceptions of Disease Management for Dairy Calves: “It’s Just Little Things That Make Such a Big Difference”. Animals 2021, 11, 2829. https://doi.org/10.3390/ani11102829
Palczynski LJ, Bleach ECL, Brennan ML, Robinson PA. Stakeholder Perceptions of Disease Management for Dairy Calves: “It’s Just Little Things That Make Such a Big Difference”. Animals. 2021; 11(10):2829. https://doi.org/10.3390/ani11102829
Chicago/Turabian StylePalczynski, Laura J., Emma C. L. Bleach, Marnie L. Brennan, and Philip A. Robinson. 2021. "Stakeholder Perceptions of Disease Management for Dairy Calves: “It’s Just Little Things That Make Such a Big Difference”" Animals 11, no. 10: 2829. https://doi.org/10.3390/ani11102829
APA StylePalczynski, L. J., Bleach, E. C. L., Brennan, M. L., & Robinson, P. A. (2021). Stakeholder Perceptions of Disease Management for Dairy Calves: “It’s Just Little Things That Make Such a Big Difference”. Animals, 11(10), 2829. https://doi.org/10.3390/ani11102829





