Optimization of UV-C Processing of Donkey Milk: An Alternative to Pasteurization?
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Milk Samples
2.2. Bacterial Strains
2.3. Growth of Bacterial Strains and Milk Inoculation
2.4. UV-C Processing of Artificially Contaminated Raw Donkey Milk
2.5. Donkey Milk Pasteurization (Control)
2.6. Microbiological Analysis
2.7. Lethality Calculations
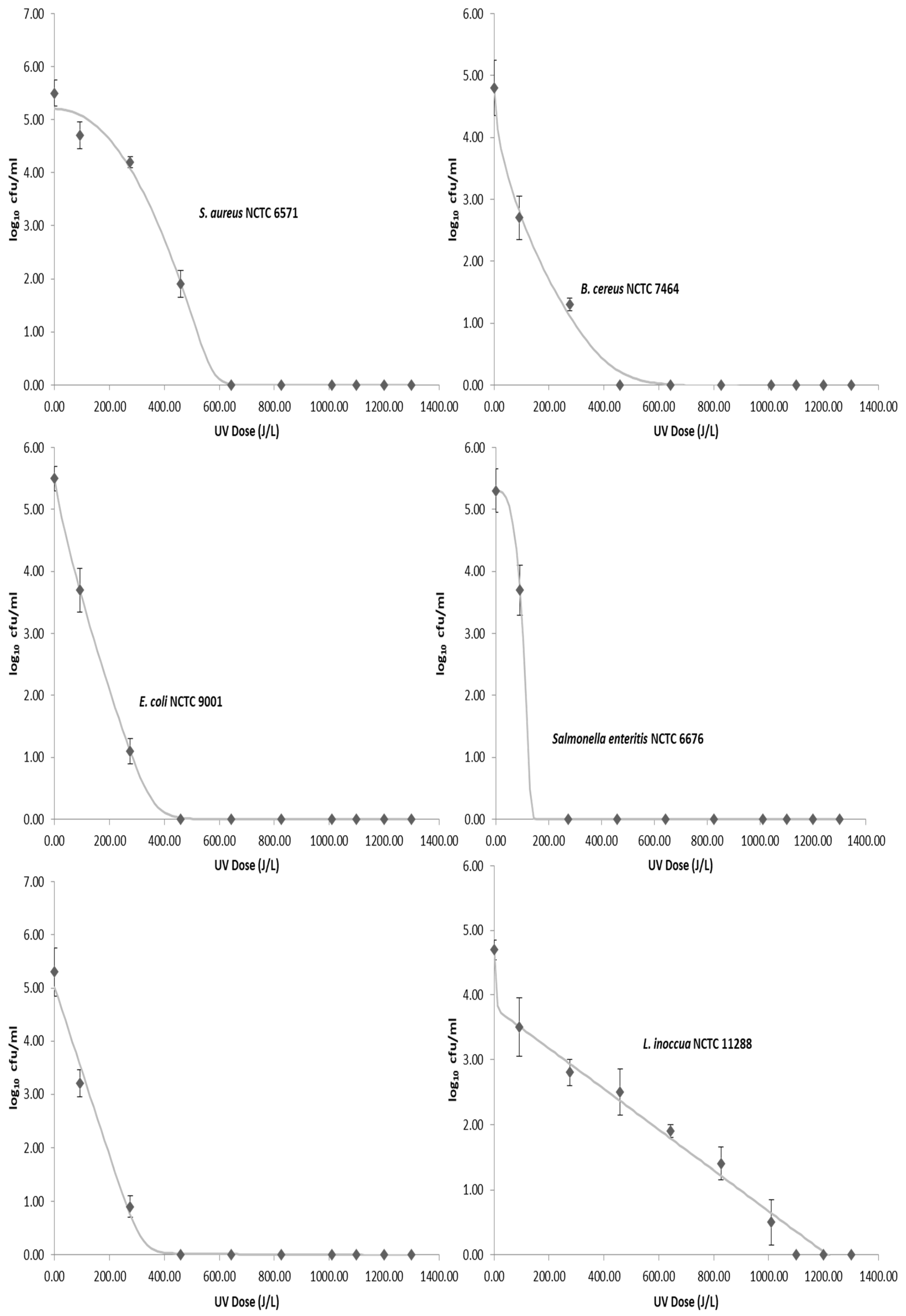
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Souroullas, K.; Aspri, M.; Papademas, P. Donkey milk as a supplement in infant formula: Benefits and technological challenges. Food Res. Int. 2018, 109, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Martini, M.; Salari, F.; Altomonte, I.; Ragona, G.; Piazza, A.; Gori, R.; Casati, D.; Brajon, G. Effects of pasteurization and storage conditions on donkey milk nutritional and hygienic characteristics. J. Dairy Res. 2018, 85, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Conte, F.; Panebianco, A. Potential hazards associated with raw donkey milk consumption: A review. Int. J. Food Sci. 2019. Available online: https://doi.org/10.1155/2019/5782974 (accessed on 24 December 2020).
- Mottola, A.; Alberghini, L.; Giaccone, V.; Marchetti, P.; Tantillo, G.; Di Pinto, A. Microbiological safety and quality of Italian donkey milk. J. Food Saf. 2018, 38, e12444. [Google Scholar] [CrossRef]
- Faccia, M.; D’Alessandro, A.G.; Summer, A.; Hailu, Y. Milk products from minor dairy species: A review. Animals 2020, 10, 1260. [Google Scholar] [CrossRef]
- Bintsis, T.; Litopoulou-Tzanetaki, E.; Robinson, R.K. Existing and potential applications of ultraviolet light in the food industry–a critical review. J. Sci. Food Agric. 2000, 80, 637–645. [Google Scholar] [CrossRef]
- Matak, K.; Churey, J.; Worobo, R.; Sumner, S.; Hovingh, E.; Hackney, C.; Pierson, M. Efficacy of UV light for the reduction of Listeria monocytogenes in goat’s milk. J. Food Prot. 2005, 68, 2212–2216. [Google Scholar] [CrossRef]
- Rossitto, P.; Cullor, J.S.; Crook, J.; Parko, J.; Sechi, P.; Cenci-Goga, B. Effects of UV irradiation in a continuous turbulent flow UV reactor on microbiological and sensory characteristics of cow’s milk. J. Food Prot. 2012, 75, 2197–2207. [Google Scholar] [CrossRef]
- Cappozzo, J.C.; Koutchma, T.; Barnes, G. Chemical characterization of milk after treatment with thermal (HTST and UHT) and nonthermal (turbulent flow ultraviolet) processing technologies. J. Dairy Sci. 2015, 98, 5068–5079. [Google Scholar] [CrossRef]
- Crook, J.A.; Rossitto, P.V.; Parko, J.; Koutchma, T.; Cullor, J.S. Efficacy of ultraviolet (UV-C) light in a thin-film turbulent flow for the reduction of milkborne pathogens. Foodborne Pathog. Dis. 2015, 12, 506–513. [Google Scholar] [CrossRef]
- Krishnamurthy, K.; Demirci, A.; Irudayaraj, J. Inactivation of Staphylococcus aureus in milk using flow-through pulsed UV-light treatment system. J. Food Sci. 2007, 72, M233–M239. [Google Scholar] [CrossRef]
- Christen, L.; Lai, C.T.; Hartmann, B.; Hartmann, P.E.; Geddes, D.T. The effect of UV-C pasteurization on bacteriostatic properties and immunological proteins of donor human milk. PLoS ONE 2013, 8, e85867. [Google Scholar] [CrossRef] [PubMed]
- Bandla, S.; Choudhary, R.; Watson, D.G.; Haddock, J. Impact of UV-C processing of raw cow milk treated in a continuous flow coiled tube ultraviolet reactor. Agric. Eng. Int. CIGR J. 2012, 14, 86–93. [Google Scholar]
- Gunter-Ward, D.M.; Patras, A.S.; Bhullar, M.; Kilonzo-Nthenge, A.; Pokharel, B.; Sasges, M. Efficacy of ultraviolet (UV-C) light in reducing foodborne pathogens and model viruses in skim milk. J. Food Process. Preserv. 2018, 42, e13485. [Google Scholar] [CrossRef]
- Alberini, F.; Simmons, M.J.; Parker, D.; Koutchma, T. Validation of hydrodynamic and microbial inactivation models for UV-C treatment of milk in a swirl-tube ‘SurePure Turbulator™. J. Food Eng. 2015, 162, 63–69. [Google Scholar] [CrossRef]
- Koutchma, T.; Bissonnette, S.; Popovic, V. An update on research, development and implementation of UV and pulsed light technologies for nonthermal preservation of milk and dairy products. Ref. Module Food Sci. 2019, 1–21. [Google Scholar]
- Ansari, J.A.; Ismail, M.; Farid, M. Investigate the efficacy of UV pretreatment on thermal inactivation of Bacillus subtilis spores in different types of milk. Innov. Food Sci. Emerg. Technol. 2019, 52, 387–393. [Google Scholar] [CrossRef]
- Martysiak-Żurowska, D.; Puta, M.; Kotarska, J.; Cybula, K.; Malinowska-Pańczyk, E.; Kołodziejska, I. The effect of UV-C irradiation on lipids and selected biologically active compounds in human milk. Int. Dairy J. 2017, 66, 42–48. [Google Scholar] [CrossRef]
- Gayán, E.; Álvarez, I.; Condón, S. Inactivation of bacterial spores by UV-C light. Innov. Food Sci. Emerg. Technol. 2013, 19, 140–145. [Google Scholar] [CrossRef]
- Peila, C.; Emmerik, N.E.; Giribaldi, M.; Stahl, B.; Ruitenberg, J.E.; Van Elburg, R.M.; Moro, G.E.; Bertino, E.; Coscia, A.; Cavallarin, L. Human milk processing: A systematic review of innovative techniques to ensure the safety and quality of donor milk. J. Pediatric Gastroenterol. Nutr. 2017, 64, 353–361. [Google Scholar] [CrossRef]
- Weaver, G.; Bertino, E.; Gebauer, C.; Grovslien, A.; Mileusnic-Milenovic, R.; Arslanoglu, S.; Barnett, D.; Boquien, C.-Y.; Buffin, R.; Gaya, A. Recommendations for the establishment and operation of human milk banks in Europe: A consensus statement from the European Milk Bank Association (EMBA). Front. Pediatr. 2019, 7, 53. [Google Scholar] [CrossRef]
- Geeraerd, A.; Valdramidis, V.; Van Impe, J. GInaFiT, a freeware tool to assess non-log-linear microbial survivor curves. Int. J. Food Microbiol. 2005, 102, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Bigelow, W.; Esty, J. The thermal death point in relation to time of typical thermophilic organisms. J. Infect. Dis. 1920, 27, 602–617. [Google Scholar] [CrossRef]
- Mafart, P.; Couvert, O.; Gaillard, S.; Leguérinel, I. On calculating sterility in thermal preservation methods: Application of the Weibull frequency distribution model. Int. J. Food Microbiol. 2002, 72, 107–113. [Google Scholar] [CrossRef]
- Coroller, L.; Leguérinel, I.; Mettler, E.; Savy, N.; Mafart, P. General model, based on two mixed Weibull distributions of bacterial resistance, for describing various shapes of inactivation curves. Appl. Environ. Microbiol. 2006, 72, 6493–6502. [Google Scholar] [CrossRef]
- Cerf, O. A review tailing of survival curves of bacterial spores. J. Appl. Bacteriol. 1977, 42, 1–19. [Google Scholar] [CrossRef]
- Gabriel, A.A.; Marquez, G.G.F. Inactivation behaviors of selected bacteria in ultraviolet-C-treated human breast milk. Innov. Food Sci. Emerg. Technol. 2017, 41, 216–223. [Google Scholar] [CrossRef]
- Keyser, M.; Műller, I.A.; Cilliers, F.P.; Nel, W.; Gouws, P.A. Ultraviolet radiation as a non-thermal treatment for the inactivation of microorganisms in fruit juice. Innov. Food Sci. Emerg. Technol. 2008, 9, 348–354. [Google Scholar] [CrossRef]
- Fredericks, I.N.; Du Toit, M.; Krügel, M. Efficacy of ultraviolet radiation as an alternative technology to inactivate microorganisms in grape juices and wines. Food Microbiol. 2011, 28, 510–517. [Google Scholar] [CrossRef]
- Lu, G.; Li, C.; Liu, P. UV inactivation of milk-related microorganisms with a novel electrodeless lamp apparatus. Eur. Food Res. Technol. 2011, 233, 79–87. [Google Scholar] [CrossRef]
- Beauchamp, S.; Lacroix, M. Resistance of the genome of Escherichia coli and Listeria monocytogenes to irradiation evaluated by the induction of cyclobutane pyrimidine dimers and 6-4 photoproducts using gamma and UV-C radiations. Radiat. Phys. Chem. 2012, 81, 1193–1197. [Google Scholar] [CrossRef]
- Cheigh, C.-I.; Park, M.-H.; Chung, M.-S.; Shin, J.-K.; Park, Y.-S. Comparison of intense pulsed light-and ultraviolet (UVC)-induced cell damage in Listeria monocytogenes and Escherichia coli O157: H7. Food Control 2012, 25, 654–659. [Google Scholar] [CrossRef]
- Baysal, A.H. Short-wave ultraviolet light inactivation of pathogens in fruit juices. In Fruit Juices; Elsevier: Amsterdam, The Netherlands, 2018; pp. 463–510. [Google Scholar]
- Possas, A.; Valero, A.; García-Gimeno, R.M.; Pérez-Rodríguez, F.; de Souza, P.M. Influence of temperature on the inactivation kinetics of Salmonella Enteritidis by the application of UV-C technology in soymilk. Food Control 2018, 94, 132–139. [Google Scholar] [CrossRef]
- Keklik, N.M.; Demirci, A.; Puri, V.M.; Heinemann, P.H. Modeling the inactivation of Salmonella Typhimurium, Listeria monocytogenes, and Salmonella Enteritidis on poultry products exposed to pulsed UV light. J. Food Prot. 2012, 75, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, E.; Rodríguez, C.; Ródenas, J.; Navarro, N.; Riquelme, C.; Rosell, R.; Campbell, J.; Crenshaw, J.; Segalés, J.; Pujols, J. Evaluation of the effectiveness of the SurePure Turbulator ultraviolet-C irradiation equipment on inactivation of different enveloped and non-enveloped viruses inoculated in commercially collected liquid animal plasma. PLoS ONE 2019, 14, e0212332. [Google Scholar]

| Microbiological Parameter | Microbiological Media | Incubation Conditions | Reference |
|---|---|---|---|
| S. aureus NCTC6571 | Baird Parker | 37 °C/48 h | ISO 6888-1:1999 |
| L. inoccua NCTC 11288 | ALOA | 37 °C/48 h | ISO 11290-2:2017 |
| B. cereus NCTC 7464 | MYP | 30 °C/48 h | ISO 7932:2004 |
| E. coli NCTC 9001 | TBX | 44 °C/24 h | ISO 16649-2:2001 |
| Salmonella enteritis NCTC 6676 | Chromogenic Salmonella | 37 °C/48 h | ISO 6579-1:2017 |
| Cronobacter sakazakii NCTC 11467 | Chromogenic | 44 °C/24 h | ISO 22964:2017 |
| Inactivation Model | Equation | Reference |
|---|---|---|
| Log Linear Model | [23] | |
| Weibull Model | [24] | |
| Double Weibull Model | [25] | |
| Biphasic Model | [26] |
| Microbiological Parameters (log cfu/mL) | Min | Max | Mean | SD |
|---|---|---|---|---|
| TVC | 2.90 | 5.10 | 3.80 | 0.02 |
| Enterobacteriaceae | <1.00 | 3.40 | 1.90 | 0.04 |
| Staphylococcus | <1.00 | 4.70 | 3.10 | 0.06 |
| E. coli | <1.00 | <1.00 | <1.00 | <1.00 |
| Bacillus cereus | <1.00 | <1.00 | <1.00 | <1.00 |
| Listeria monocytogenes | ND | ND | ND | ND |
| Chemical Parameters (g/100 mL) | ||||
| Fat | 0.30 | 1.40 | 0.84 | 0.07 |
| Protein | 1.30 | 1.96 | 1.62 | 0.05 |
| Total Solids | 7.29 | 10.59 | 9.23 | 0.69 |
| Inactivation Model | Log Linear | Weibull | Double Weibull | Biphasic | ||||
|---|---|---|---|---|---|---|---|---|
| Bacteria | RMSE | R2 | RMSE | R2 | RMSE | R2 | RMSE | R2 |
| S. aureus | 1.00 | 0.83 | 0.94 | 0.87 | 0.21 | 0.99 | 0.42 | 0.98 |
| L. inoccua | 0.32 | 0.97 | 0.27 | 0.98 | 0.29 | 0.98 | 0.23 | 0.99 |
| B. cereus | 1.25 | 0.50 | 0.83 | 0.80 | 0.12 | 0.99 | 0.30 | 0.98 |
| E. coli | 1.30 | 0.61 | 0.85 | 0.85 | 0.0096 | 1.00 | 0.11 | 0.99 |
| Salmonella enteritis | 1.43 | 0.52 | 1.27 | 0.67 | 0.0008 | 1.00 | 0.12 | 0.99 |
| Cronobacter sakazakii | 1.25 | 0.59 | 0.96 | 0.78 | 0.64 | 0.92 | 0.20 | 0.99 |
| Pathogen | Inactivation Model | 4D Value (J/L) |
|---|---|---|
| S. aureus | Double Weibull | 507 |
| L. inoccua | Biphasic | 1001 |
| B. cereus | Double Weibull | 338 |
| E. coli | Double Weibull | 247 |
| Salmonella enteritis | Double Weibull | 130 |
| Cronobacter sakazakii | Biphasic | 260 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papademas, P.; Mousikos, P.; Aspri, M. Optimization of UV-C Processing of Donkey Milk: An Alternative to Pasteurization? Animals 2021, 11, 42. https://doi.org/10.3390/ani11010042
Papademas P, Mousikos P, Aspri M. Optimization of UV-C Processing of Donkey Milk: An Alternative to Pasteurization? Animals. 2021; 11(1):42. https://doi.org/10.3390/ani11010042
Chicago/Turabian StylePapademas, Photis, Panagiotis Mousikos, and Maria Aspri. 2021. "Optimization of UV-C Processing of Donkey Milk: An Alternative to Pasteurization?" Animals 11, no. 1: 42. https://doi.org/10.3390/ani11010042
APA StylePapademas, P., Mousikos, P., & Aspri, M. (2021). Optimization of UV-C Processing of Donkey Milk: An Alternative to Pasteurization? Animals, 11(1), 42. https://doi.org/10.3390/ani11010042






