Clinical and Hematological Follow-Up of Long-Term Oral Therapy with Type-I Interferon in Cats Naturally Infected with Feline Leukemia Virus or Feline Immunodeficiency Virus
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Treatment
2.2. Clinical Evaluation and Blood Sampling
2.3. Biopathological Analyses and Determination of Anti-IFN-α Antibodies
2.4. Evaluation of the CD4+/CD8+ Ratio
2.5. Statistical Analysis
3. Results
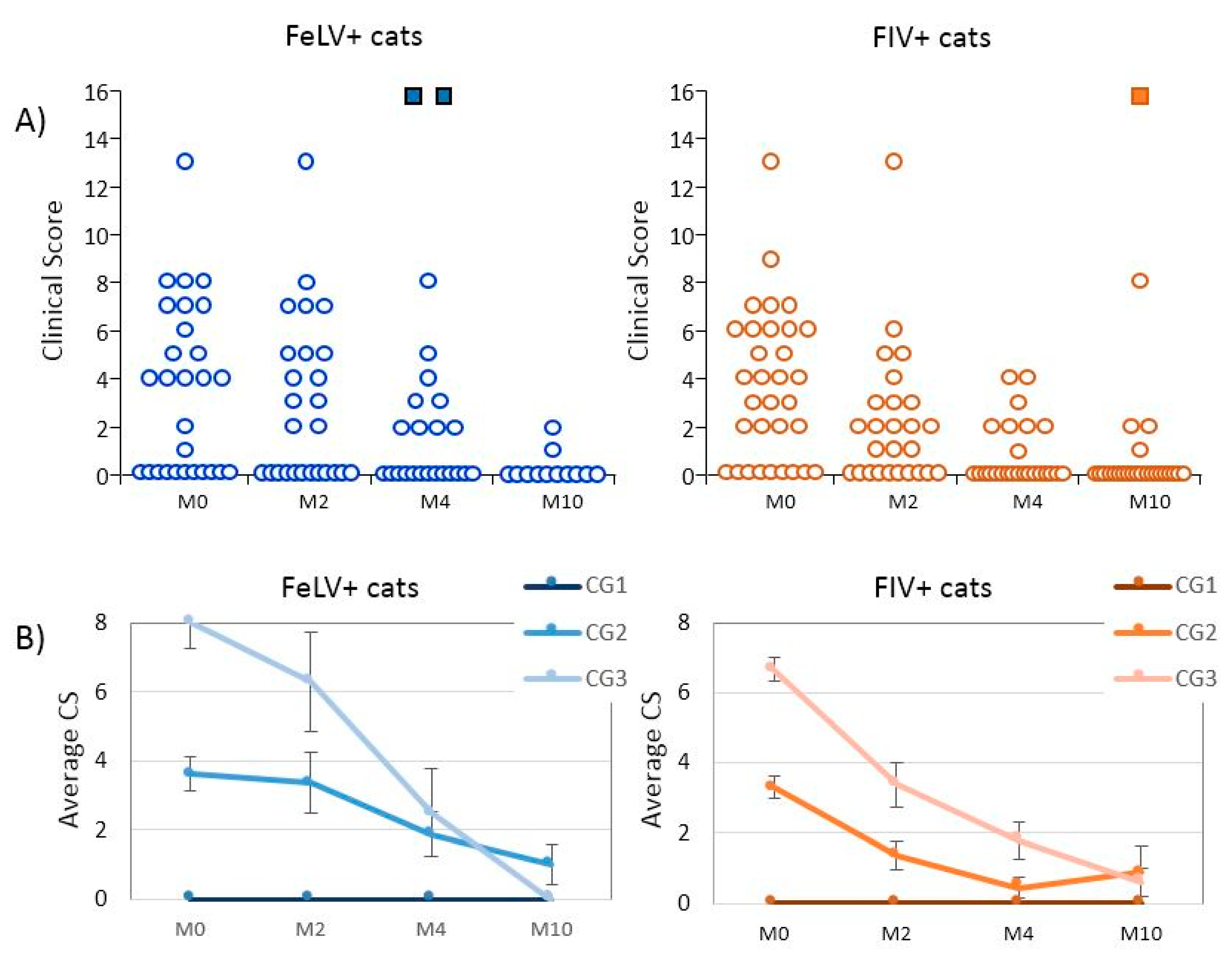
3.1. Epidemiological Data and Clinical Evolution of Cats Treated with rHuIFN-α
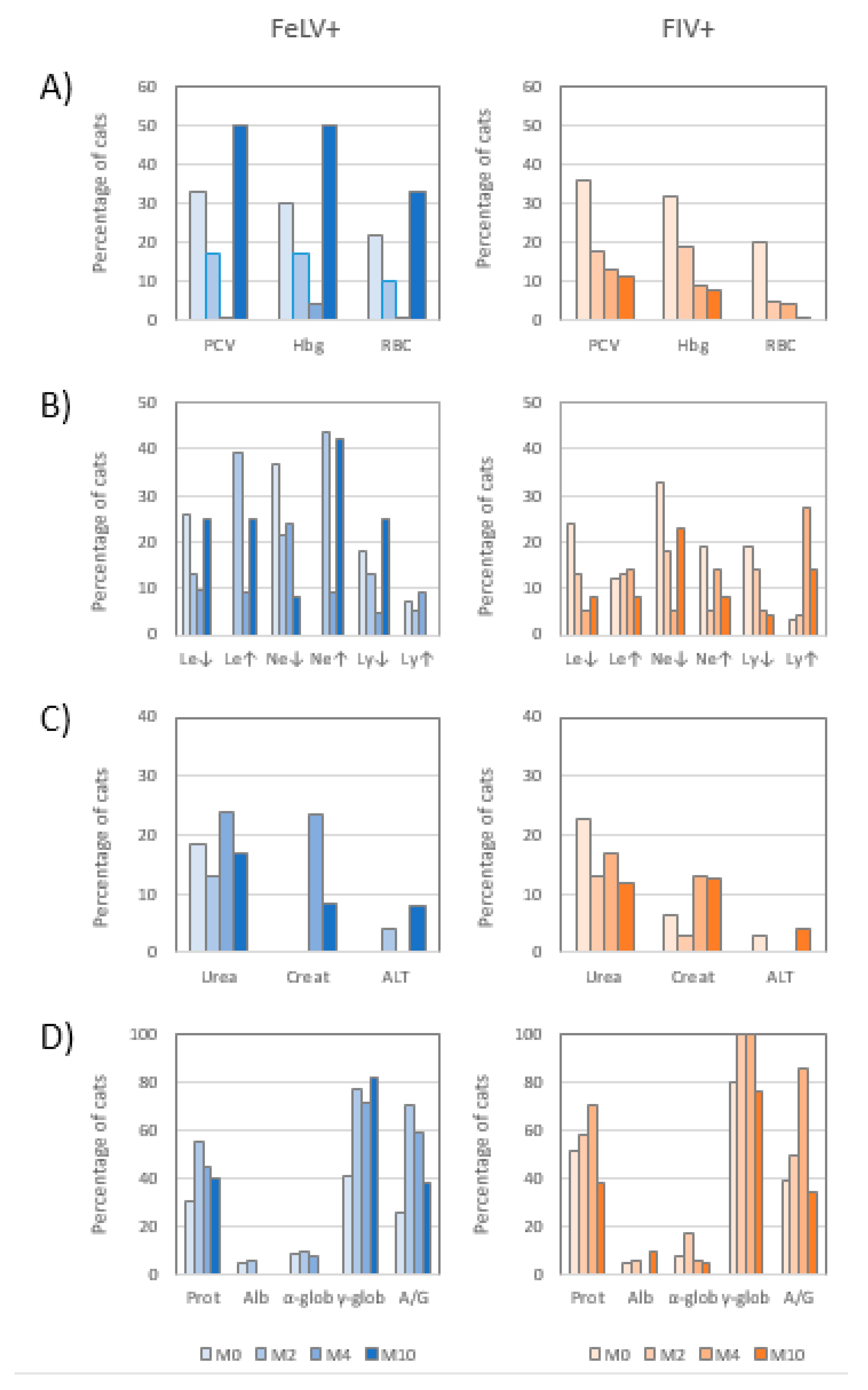
3.2. Hemogram
3.3. Leukogram
3.4. Biochemical Profile
3.5. Electrophoretogram
3.6. Evolution of the CD4+/CD8+ Ratio
3.7. Detection of Antibodies Anti-Interferon α by ELISA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hartmann, K. Clinical aspects of feline retroviruses: A review. Viruses 2012, 4, 2684–2710. [Google Scholar] [CrossRef] [PubMed]
- Hofmann-Lehmann, R.; Cattori, V.; Tandon, R.; Boretti, F.S.; Meli, M.L.; Riond, B.; Pepin, A.C.; Willi, B.; Ossent, P.; Lutz, H. Vaccination against the feline leukaemia virus: Outcome and response categories and long-term follow-up. Vaccine 2007, 25, 5531–5539. [Google Scholar] [CrossRef] [PubMed]
- Little, S.; Levy, J.; Hartmann, K.; Hofmann-Lehmann, R.; Hosie, M.; Olah, G.; Denis, K.S. 2020 AAFP Feline Retrovirus Testing and Management Guidelines. J. Feline Med. Surg. 2020, 22, 5–30. [Google Scholar] [CrossRef] [PubMed]
- Collado, V.M.; Domenech, A.; Miró, G.; Martin, S.; Escolar, E.; Gomez-Lucia, E. Epidemiological aspects and clinicopathological findings in cats naturally infected with feline leukemia virus (FeLV) and/or feline immunodeficiency virus (FIV). Open J. Vet. Med. 2012, 2, 13–20. [Google Scholar] [CrossRef][Green Version]
- Dunham, S.P.; Graham, E. Retroviral infections of small animals. Vet. Clin. North Am. Small Anim. Pract. 2008, 38, 879–901. [Google Scholar] [CrossRef]
- Bęczkowski, P.M.; Litster, A.; Lin, T.L.; Mellor, D.J.; Willett, B.J.; Hosie, M.J. Contrasting clinical outcomes in two cohorts of cats naturally infected with feline immunodeficiency virus (FIV). Vet. Microbiol. 2015, 176, 50–60. [Google Scholar] [CrossRef]
- Collado, V.M.; Gómez-Lucía, E.; Tejerizo, G.; Miró, G.; Escolar, E.; Martín, S.; Doménech, A. Effect of type I interferons on the expression of feline leukaemia virus. Vet. Microbiol. 2007, 123, 180–186. [Google Scholar] [CrossRef]
- Gómez-Lucía, E.; Collado, V.M.; Miró, G.; Doménech, A. Effect of type-I interferon on retroviruses. Viruses 2009, 1, 545–573. [Google Scholar] [CrossRef]
- Hartmann, K. Efficacy of antiviral chemotherapy for retrovirus-infected cats: What does the current literature tell us? J. Feline Med. Surg. 2015, 17, 925–939. [Google Scholar] [CrossRef]
- de Mari, K.; Maynard, L.; Sanquer, A.; Lebreux, B.; Eun, H.-M. Therapeutic effects of recombinant feline interferon-omega on feline leukemia virus (FeLV)-infected and FeLV/feline immunodeficiency virus (FIV)-coinfected symptomatic cats. J. Vet. Intern. Med. 2004, 18, 477–482. [Google Scholar] [CrossRef]
- Paltrinieri, S.; Crippa, A.; Comerio, T.; Angioletti, A.; Roccabianca, P. Evaluation of inflammation and immunity in cats with spontaneous parvovirus infection: Consequences of recombinant feline interferon-omega administration. Vet. Immunol. Immunopathol. 2007, 118, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Shibanai, A.; Tanaka, S.; Uchida, K.; Mochizuki, M. Use of recombinant feline interferon and glucocorticoid in the treatment of feline infectious peritonitis. J. Feline Med. Surg. 2004, 6, 107–109. [Google Scholar] [CrossRef] [PubMed]
- Doménech, A.; Gómez, N.; Gomez-Lucia, E. Capítulo 44. Inmunodeficiencia felina. In Enfermedades Infecciosas de los Caninos y los Felinos; Editorial Intermédica: Buenos Aires, Argentina, 2011; pp. 353–365. ISBN 978-950-555-360-0300. [Google Scholar]
- Doménech, A.; Gómez, N.; Gomez-Lucia, E. Capítulo 45. Leucemia felina. In Enfermedades Infecciosas de los Caninos y los Felinos; Editorial Intermédica: Buenos Aires, Argentina, 2011; pp. 367–378. ISBN 978-950-555-360-0300. [Google Scholar]
- Burling, A.N.; Levy, J.K.; Scott, H.M.; Crandall, M.M.; Tucker, S.J.; Wood, E.G.; Foster, J.D. Seroprevalences of feline leukemia virus and feline immunodeficiency virus infection in cats in the United States and Canada and risk factors for seropositivity. J. Am. Vet. Med. Assoc. 2017, 251, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Marenzoni, M.L.; Lauzi, S.; Miglio, A.; Coletti, M.; Arbia, A.; Paltrinieri, S.; Antognoni, M.T. Comparison of three blood transfusion guidelines applied to 31 feline donors to minimise the risk of transfusion-transmissible infections. J. Feline Med. Surg. 2017. [Google Scholar] [CrossRef]
- Cummins, J.M.; Tompkins, M.B.; Olsen, R.G.; Tompkins, W.A.; Lewis, M.G. Oral use of human alpha interferon in cats. J. Biol. Res. Mod. 1988, 7, 513–523. [Google Scholar]
- Zeidner, N.S.; Rose, L.M.; Mathiason-DuBard, C.K.; Myles, M.H.; Hill, D.L.; Mullins, J.I.; Hoover, E.A. Zidovudine in combination with alpha interferon and interleukin-2 as prophylactic therapy for FeLV-induced immunodeficiency syndrome (FeLV-FAIDS). J. Acquir. Immune Defic. Syndr. 1990, 3, 787–796. [Google Scholar]
- Li, S.-F.; Zhao, F.-R.; Shao, J.-J.; Xie, Y.-L.; Chang, H.-Y.; Zhang, Y.-G. Interferon-omega: Current status in clinical applications. Int. Immunopharmacol. 2017, 52, 253–260. [Google Scholar] [CrossRef]
- Gomez-Lucia, E.; Collado, V.M.; Miró, G.; Martín, S.; Benítez, L.; Doménech, A. Follow-uuuuuup of viral parameters in FeLV- or FIV-naturally infected cats treated orally with low doses of human interferon alpha. Viruses 2019, 11. [Google Scholar] [CrossRef]
- Pedretti, E.; Passeri, B.; Amadori, M.; Isola, P.; Di Pede, P.; Telera, A.; Vescovini, R.; Quintavalla, F.; Pistello, M. Low-dose interferon-alpha treatment for feline immunodeficiency virus infection. Vet. Immunol. Immunopathol. 2006, 109, 245–254. [Google Scholar] [CrossRef]
- Arjona, A.; Escolar, E.; Soto, I.; Barquero, N.; Martin, D.; Gomez-Lucia, E. Seroepidemiological survey of infection by feline leukemia virus and immunodeficiency virus in Madrid and correlation with some clinical aspects. J. Clin. Microbiol. 2000, 38, 3448–3449. [Google Scholar] [CrossRef]
- Miró, G.; Doménech, A.; Escolar, E.; Collado, V.M.; Tejerizo, G.; De Las Heras, A.; Gómez-Lucía, E. Plasma electrophoretogram in feline immunodeficiency virus (FIV) and/or feline leukaemia virus (FeLV) infections. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2007, 54, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Vogt, M.; Hirsch, M.S. Prospects for the prevention and therapy of infections with the Human Immunodeficiency Virus. Clin. Infect. Dis. 1986, 8, 991–1000. [Google Scholar] [CrossRef]
- Gil, S.; Leal, R.O.; Duarte, A.; McGahie, D.; Sepúlveda, N.; Siborro, I.; Cravo, J.; Cartaxeiro, C.; Tavares, L.M. Relevance of feline interferon omega for clinical improvement and reduction of concurrent viral excretion in retrovirus infected cats from a rescue shelter. Res. Vet. Sci. 2013, 94, 753–763. [Google Scholar] [CrossRef]
- Leal, R.O.; Gil, S.; Sepúlveda, N.; McGahie, D.; Duarte, A.; Niza, M.M.R.E.; Tavares, L. Monitoring acute phase proteins in retrovirus infected cats undergoing feline interferon-ω therapy. J. Small Anim. Pract. 2014, 55, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Leal, R.O.; Gil, S.; Duarte, A.; McGahie, D.; Sepúlveda, N.; Niza, M.M.R.E.; Tavares, L. Evaluation of viremia, proviral load and cytokine profile in naturally feline immunodeficiency virus infected cats treated with two different protocols of recombinant feline interferon omega. Res. Vet. Sci. 2015, 99, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Doménech, A.; Miró, G.; Collado, V.M.; Ballesteros, N.; Sanjosé, L.; Escolar, E.; Martin, S.; Gomez-Lucia, E. Use of recombinant interferon omega in feline retrovirosis: From theory to practice. Vet. Immunol. Immunopathol. 2011, 143, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Gil, S.; Leal, R.O.; McGahie, D.; Sepúlveda, N.; Duarte, A.; Niza, M.M.R.E.; Tavares, L. Oral Recombinant Feline Interferon-Omega as an alternative immune modulation therapy in FIV positive cats: Clinical and laboratory evaluation. Res. Vet. Sci. 2014, 96, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Spada, E.; Perego, R.; Sgamma, E.A.; Proverbio, D. Survival time and effect of selected predictor variables on survival in owned pet cats seropositive for feline immunodeficiency and leukemia virus attending a referral clinic in northern Italy. Prev. Vet. Med. 2018, 150, 38–46. [Google Scholar] [CrossRef]
- Kociba, G.J.; Garg, R.C.; Khan, K.N.M.; Reiter, J.A.; Chatfield, R.C. Effects of orally administered interferon-α on the pathogenesis of feline leukaemia virus-induced erythroid aplasia. Comp. Haematol. Int. 1995, 5, 79–83. [Google Scholar] [CrossRef]
- Kölbl, S.; Skolek, R.; Hirt, R.; Seiser, M.; Thalhammer, J. Effects of long term low dose interferon α in cats persistently infected with FeLV. Kleintierpraxis 2000, 45, 497–510. [Google Scholar]
- McCaw, D.L.; Boon, G.D.; Jergens, A.E.; Kern, M.R.; Bowles, M.H.; Johnson, J.C. Immunomodulation therapy for feline leukemia virus infection. J. Am. Anim. Hosp. Assoc. 2001, 37, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Hofmann-Lehmann, R.; Holznagel, E.; Ossent, P.; Lutz, H. Parameters of disease progression in long-term experimental feline retrovirus (feline immunodeficiency virus and feline leukemia virus) infections: Hematology, clinical chemistry, and lymphocyte subsets. Clin. Diagn. Lab. Immunol. 1997, 4, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Flynn, J.N.; Cannon, C.A.; Lawrence, C.E.; Jarrett, O. Polyclonal B-cell activation in cats infected with feline immunodeficiency virus. Immunology 1994, 81, 626–630. [Google Scholar] [PubMed]
- Gleich, S.; Hartmann, K. Hematology and serum biochemistry of feline immunodeficiency virus-infected and feline leukemia virus-infected cats. J. Vet. Intern. Med. 2009, 23, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Riondato, F.; Gianella, P.; Guglielmino, R.; Cagnasso, A.; Bo, S. Effects of interferon alpha (INF-alpha) therapy on peripheral blood lymphocyte subsets from FIV and FeLV naturally infected cats. Vet. Res. Commun. 2003, 27 (Suppl. 1), 429–432. [Google Scholar] [CrossRef]
- Thomasy, S.M.; Maggs, D.J. A review of antiviral drugs and other compounds with activity against feline herpesvirus type 1. Vet. Ophthalmol. 2016, 19 (Suppl. 1), 119–130. [Google Scholar] [CrossRef]
| FeLV+ (n = 27) | FIV+ (n = 31) | |
|---|---|---|
| Age in years: mean (range) | 1.9 (0.3–6) | 4.8 (0.6–14) |
| Sex: males/females | 11/16 | 20/11 |
| Neutered: yes/no | 18/9 | 21/10 |
| Breed: mixed breed/(breed) | 25/2 (Persian) | 26/5 (2 Persian, 2 Siamese, 1 Bombay) |
| CG1 (CS = 0; asymptomatic) | 11 (40.7%) | 9 (29.0%) |
| CG2 (CS: 1–5; mild disease) | 8 (29.6%) | 13 (41.9%) |
| CG3 (CS: ≥6; severe disease) | 8 (29.6%) | 9 (29.0%) |
| Infection | Evolution | M2 n (%) | M4 n (%) | M10 n (%) |
|---|---|---|---|---|
| FeLV+ | favorable | 7 (43.8%) | 13 (81.2%) | 5 (100%) |
| stable | 8 (50.0%) | 0 | 0 | |
| unfavorable | 1 (6.2%) | 3 a (18.8%) | 0 | |
| FIV+ | favorable | 17 (89.4%) | 18 (100%) | 15 (88.2%) |
| stable | 1 (5.3%) | 0 | 1 b (5.9%) | |
| unfavorable | 1 (5.3%) | 0 | 1 a (5.9%) |
| FeLV | FIV | |||||||
|---|---|---|---|---|---|---|---|---|
| M0 (n = 27) | M2 (n = 26) | M4 (n = 22) | M10 (n = 12) | M0 (n = 31) | M2 (n = 25) | M4 (n = 25) | M10 (n = 24) | |
| Loss of appetite | 33.3 | 32.0 | 13.6 | 0.0 | 54.8 | 25.0 | 11.5 | 4.2 |
| Asthenia | 33.3 | 28.0 | 9.1 | 0.0 | 41.9 | 29.2 | 15.4 | 4.2 |
| Body condition | 18.5 | 4.0 | 4.5 | 0.0 | 19.4 | 8.3 | 0.0 | 4.2 |
| Lymphadenomegaly | 18.5 | 16.0 | 13.6 | 8.3 | 19.4 | 12.5 | 7.7 | 0.0 |
| Altered mucosae | 25.9 | 12.0 | 4.5 | 0.0 | 29.0 | 4.2 | 0.0 | 4.2 |
| Conjunctivitis | 25.9 | 24.0 | 13.6 | 0.0 | 16.1 | 12.5 | 7.7 | 4.2 |
| Oral lesions | 29.6 | 28.0 | 22.7 | 0.0 | 32.3 | 20.8 | 3.8 | 4.2 |
| Digestive disorders | 11.1 | 4.0 | 0.0 | 8.3 | 9.7 | 8.3 | 0.0 | 4.2 |
| Skin lesions | 11.1 | 8.0 | 0.0 | 0.0 | 12.9 | 4.2 | 3.8 | 0.0 |
| Respiratory disease | 29.6 | 24.0 | 9.1 | 0.0 | 22.6 | 4.2 | 7.7 | 8.3 |
| Infection | M0 | M2 | M4 | M10 | |
|---|---|---|---|---|---|
| Average CD4+/CD8+ ratio | FeLV+ | 1.66 | 1.79 | 2.11 | 2.38 |
| FIV+ | 0.80 | 1.17 | 1.40 | 1.29 | |
| Percentage of cats with altered CD4+/CD8+ ratio (<0.9) | FeLV+ | 26.1% | 10.0% | 5.3% | 0% |
| FIV+ | 69.2% | 30.0% | 20.0% | 35.0% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomez-Lucia, E.; Collado, V.M.; Miró, G.; Martín, S.; Benítez, L.; Doménech, A. Clinical and Hematological Follow-Up of Long-Term Oral Therapy with Type-I Interferon in Cats Naturally Infected with Feline Leukemia Virus or Feline Immunodeficiency Virus. Animals 2020, 10, 1464. https://doi.org/10.3390/ani10091464
Gomez-Lucia E, Collado VM, Miró G, Martín S, Benítez L, Doménech A. Clinical and Hematological Follow-Up of Long-Term Oral Therapy with Type-I Interferon in Cats Naturally Infected with Feline Leukemia Virus or Feline Immunodeficiency Virus. Animals. 2020; 10(9):1464. https://doi.org/10.3390/ani10091464
Chicago/Turabian StyleGomez-Lucia, Esperanza, Victorio M. Collado, Guadalupe Miró, Sonsoles Martín, Laura Benítez, and Ana Doménech. 2020. "Clinical and Hematological Follow-Up of Long-Term Oral Therapy with Type-I Interferon in Cats Naturally Infected with Feline Leukemia Virus or Feline Immunodeficiency Virus" Animals 10, no. 9: 1464. https://doi.org/10.3390/ani10091464
APA StyleGomez-Lucia, E., Collado, V. M., Miró, G., Martín, S., Benítez, L., & Doménech, A. (2020). Clinical and Hematological Follow-Up of Long-Term Oral Therapy with Type-I Interferon in Cats Naturally Infected with Feline Leukemia Virus or Feline Immunodeficiency Virus. Animals, 10(9), 1464. https://doi.org/10.3390/ani10091464





