Beyond Glucocorticoids: Integrating Dehydroepiandrosterone (DHEA) into Animal Welfare Research
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
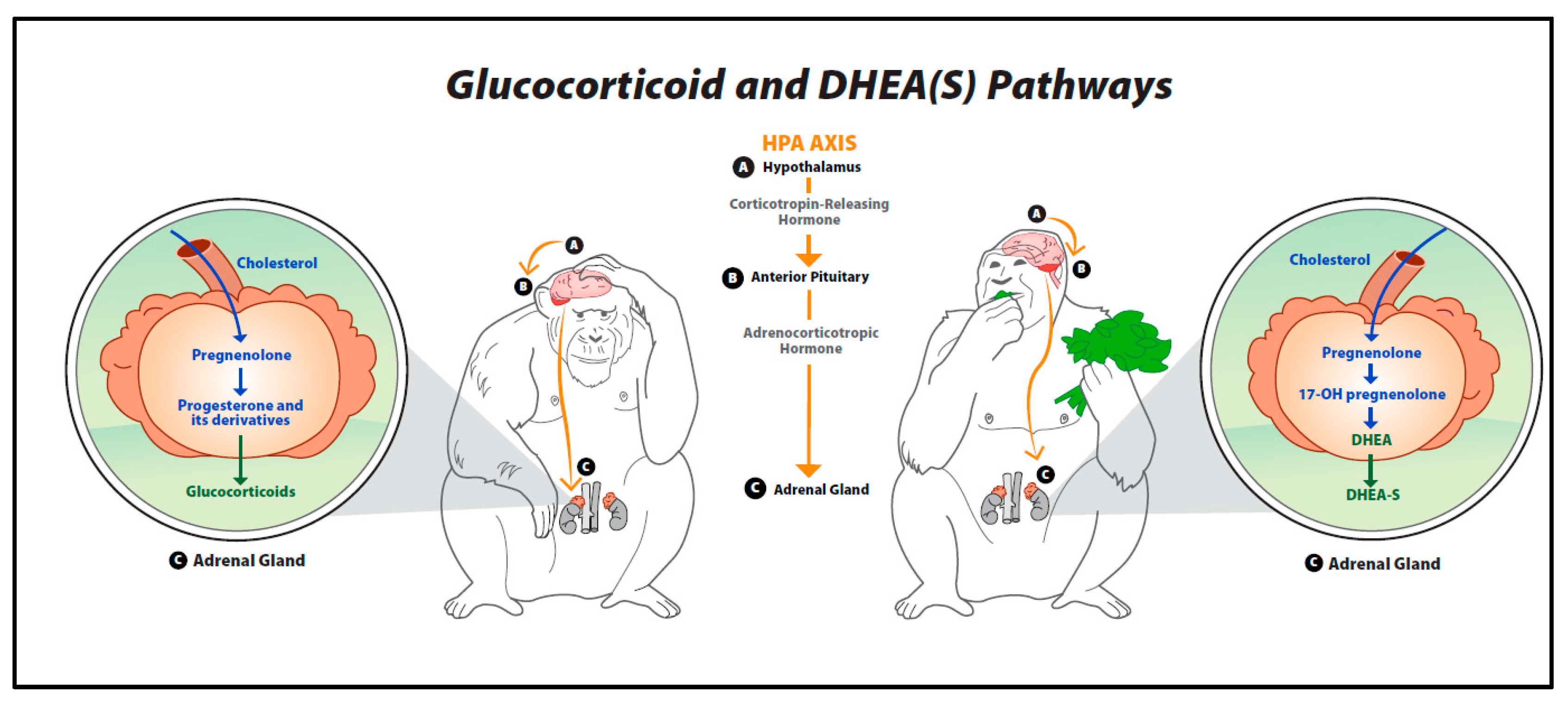
3. Overview of the Hypothalamic–Pituitary–Adrenal (HPA) Axis
4. Glucocorticoids
5. Dehydroepiandrosterone (DHEA)
6. Glucocorticoid:DHEA Ratio
7. General Methodological Considerations
7.1. Diurnal Rhythms of Hormone Secretion
7.2. Seasonal Variation in Hormone Secretion
7.3. Subject-Related Factors Influencing Hormone Secretion
7.4. Sample Types for Measuring Glucocorticoids and Dehydroepiandrosterone
7.4.1. Blood Samples
7.4.2. Saliva Samples
7.4.3. Urine Samples
7.4.4. Fecal Samples
7.4.5. Other Sample Types
8. Quantifying Hormones and Hormone Metabolites
8.1. Radio-Immunoassays and Enzyme-Immunoassays
8.2. Assay Validation
8.3. Additional Physiological Validation for Dehydroepiandrosterone
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- AZA (Association of Zoos and Aquariums) Animal Welfare Committee. Available online: https://www.aza:animal_welfare_committee (accessed on 15 June 2020).
- Butterworth, A.; Mench, J.A.; Wielebnowski, N. Practical Strategies to Assess (and Improve) Welfare. In Animal Welfare, 2nd ed.; Appleby, M.C., Mench, J.A., Olsson, I.A.S., Hughes, B.O., Eds.; CABI Publishing: Cambridge, UK, 2011; pp. 200–214. [Google Scholar]
- Whitham, J.C.; Wielebnowski, N. New directions for zoo animal welfare science. Appl. Anim. Behav. Sci. 2013, 147, 247–260. [Google Scholar] [CrossRef]
- Staley, M.; Conners, M.G.; Hall, K.; Miller, L.J. Linking stress and immunity: Immunoglobulin A as a non-invasive physiological biomarker in animal welfare studies. Horm. Behav. 2018, 102, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Hänsel, A.; Hong, S.; Cámara, R.J.A.; Von Kaenel, R. Inflammation as a psychophysiological biomarker in chronic psychosocial stress. Neurosci. Biobehav. Rev. 2010, 35, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrino 2009, 34, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Pressman, S.D.; Cohen, S. Does positive affect influence health? Psychol. Bull. 2005, 131, 925–971. [Google Scholar] [CrossRef] [PubMed]
- Dickens, M.J.; Romero, L.M. A consensus endocrine profile for chronically stressed wild animals does not exist. Gen. Comp. Endocr. 2013, 191, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Touma, C.; Palme, R. Measuring fecal glucocorticoid metabolites in mammals and birds: The importance of validation. Ann. N. Y. Acad. Sci. 2005, 1046, 54–74. [Google Scholar] [CrossRef]
- Wielebnowski, N.; Watters, J. Applying fecal endocrine monitoring to conservation and behavior studies of wild mammals: Important considerations and preliminary tests. Isr. J. Ecol. Evol. 2007, 53, 439–460. [Google Scholar] [CrossRef]
- Baxter-Gilbert, J.H.; Riley, J.L.; Mastromonaco, G.F.; Litzgus, J.D.; Lesbarrères, D. A novel technique to measure chronic levels of corticosterone in turtles living around a major roadway. Conserv. Physiol. 2014, 2, cou036. [Google Scholar] [CrossRef]
- Dantzer, B.; Fletcher, Q.E.; Boonstra, R.; Sheriff, M.J. Measures of physiological stress: A transparent or opaque window into the status, management and conservation of species? Conserv. Physiol. 2014, 2, cou023. [Google Scholar] [CrossRef]
- Koren, L.; Nakagawa, S.; Burke, T.; Soma, K.K.; Wynne-Edwards, K.E.; Geffen, E. Non-breeding feather concentrations of testosterone, corticosterone and cortisol are associated with subsequent survival in wild house sparrows. Proc. Roy. Soc. B-Biol. Sci. 2011, 279, 1560–1566. [Google Scholar] [CrossRef] [PubMed]
- Santymire, R.M.; Manjerovic, M.B.; Sacerdote-Velat, A. A novel method for the measurement of glucocorticoids in dermal secretions of amphibians. Conserv. Physiol. 2018, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, C.; Montillo, M.; Prandi, A.; Mkupasi, E.M.; Ngowi, H.A.; Johansen, M.V. Hair cortisol and dehydroepiandrosterone concentrations in naturally Taenia solium infected pigs in Tanzania. Gen. Comp. Endocr. 2017, 246, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Wielebnowski, N.C.; Fletchall, N.; Carlstead, K.; Busso, J.M.; Brown, J.L. Noninvasive assessment of adrenal activity associated with husbandry and behavioral factors in the North American clouded leopard population. Zoo Biol. 2002, 21, 77–98. [Google Scholar] [CrossRef]
- Miller, G.; Chen, E.; Zhou, E. If it goes up, must it come down? Chronic stress and the hypothalamic-pituitary-adrenocortical axis in humans. Psychol. Bull. 2007, 133, 25–45. [Google Scholar] [CrossRef]
- Wielebnowski, N. Stress and distress: Evaluating their impact for the well-being of zoo animals. J. Am. Vet. Med. Assoc. 2003, 223, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, T.G.; Edwards, L. Chronic stress and the HPA axis: Clinical assessment and therapeutic considerations. Standard 2010, 9, 1–12. [Google Scholar]
- Almeida, P.E.; Weber, P.S.D.; Burton, J.L.; Zanella, A.J. Depressed DHEA and increased sickness response behaviors in lame dairy cows with inflammatory foot lesions. Domest. Anim. Endocrinol. 2008, 34, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Buckham Sporer, K.R.; Weber, P.S.D.; Burton, J.L.; Earley, B.; Crowe, M.A. Transportation of young beef bulls alters circulating physiological parameters that may be effective biomarkers of stress. J. Anim. Sci. 2008, 86, 1325–1334. [Google Scholar] [CrossRef]
- Fels, M.; Rauterberg, S.; Schwennen, C.; Ligges, U.; Herbrandt, S.; Kemper, N.; Schmicke, M. Cortisol/dehydroepiandrosterone ratio in saliva: Endocrine biomarker for chronic stress in pigs? Livest. Sci. 2019, 222, 21–24. [Google Scholar] [CrossRef]
- Goncharova, N.D.; Marenin, V.Y.; Oganyan, T.E. Aging of the hypothalamic-pituitary-adrenal axis in nonhuman primates with depression-like and aggressive behavior. Aging 2010, 2, 854–866. [Google Scholar] [CrossRef] [PubMed]
- Gundlach, N.H.; Schmicke, M.; Ludes-Wehrmeister, E.; Ulrich, S.A.; Araujo, M.G.; Siebert, U. New approach to stress research in phocids—Potential of dehydroepiandrosterone and cortisol/dehydroepiandrosterone ratio as markers for stress in harbor seals (Phoca vitulina) and gray seals (Halichoerus grypus). J. Zoo Wildl. Med. 2018, 49, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Kamin, H.S.; Kertes, D.A. Cortisol and DHEA in development and psychopathology. Horm. Behav. 2017, 89, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Peric, T.; Corazzin, M.; Romanzin, A.; Bovolenta, S.; Prandi, A.; Montillo, M.; Comin, A. Cortisol and DHEA concentrations in the hair of dairy cows managed indoor or on pasture. Livest. Sci. 2017, 202, 39–43. [Google Scholar] [CrossRef]
- Placci, M.; Marliani, G.; Sabioni, S.; Gabai, G.; Mondo, E.; Borghetti, P.; De Angelis, E.; Accorsi, P.A. Natural horse boarding vs traditional stable: A comparison of hormonal, hematological and immunological parameters. J. Appl. Anim. Welf. Sci. 2020, 23, 366–377. [Google Scholar] [CrossRef] [PubMed]
- De Kloet, E.R.; Joëls, M.; Holsboer, F. Stress and the brain: From adaptation to disease. Nat. Rev. Neurosci. 2005, 6, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Leowattana, W. DHEAS as a new diagnostic tool. Clin. Chim. Acta 2004, 341, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Raison, C.; Miller, A. When not enough is too much: The role of insufficient glucocorticoid signaling in the pathophysiology of stress-related disorders. Am. J. Psychiat. 2003, 160, 1554–1565. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr. Rev. 2000, 21, 55–89. [Google Scholar] [CrossRef]
- Fries, E.; Hesse, J.; Hellhammer, J.; Hellhammer, D.H. A new view on hypocortisolism. Psychoneuroendocrinology 2005, 30, 1010–1016. [Google Scholar] [CrossRef]
- Heim, C.; Ehlert, U.; Hellhammer, D.H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 2000, 25, 1–35. [Google Scholar] [CrossRef]
- Lupien, S.J.; de Leon, M.; De Santi, S.; Convit, A.; Tarshish, C.; Nair, N.P.V.; Thakur, M.; McEwen, B.S.; Hauger, R.L.; Meaney, M.J. Cortisol levels during human aging predict hippocampal atrophy and memory deficits. Nat. Neurosci. 1998, 1, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.E. Chronic stress and immunosenescence: A review. Neuroimmunomodulation 2008, 15, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Tsigos, C.; Chrousos, G.P. Hypothalamic–pituitary–adrenal axis, neuroendocrine factors and stress. J. Psychosom. Res. 2002, 53, 865–871. [Google Scholar] [CrossRef]
- Campbell, K.; Peebles, R. Eating disorders in children and adolescents: State of the art review. Pediatrics 2014, 134, 582–592. [Google Scholar] [CrossRef]
- Stetler, C.; Miller, G.E. Depression and hypothalamic-pituitary-adrenal activation: A quantitative summary of four decades of research. Psychosom. Med. 2011, 73, 114–126. [Google Scholar] [CrossRef]
- De Jong, I.C.; Ekkel, E.D.; van de Burgwal, J.A.; Lambooij, E.; Korte, S.M.; Ruis, M.A.; Koolhaas, J.M.; Blokhuis, H.J. Effects of strawbedding on physiological responses to stressors and behavior in growing pigs. Physiol. Behav. 1998, 64, 303–310. [Google Scholar] [CrossRef]
- Geverink, N.A.; Schouten, W.G.P.; Gort, G.; Wiegant, V.M. Individual differences in behaviour, physiology and pathology in breeding gilts housed in groups or stalls. Appl. Anim. Behav. Sci. 2003, 81, 29–41. [Google Scholar] [CrossRef]
- Romero, L.M. Seasonal changes in plasma glucocorticoid concentrations in free-living vertebrates. Gen. Comp. Endocrinol. 2002, 128, 1–24. [Google Scholar] [CrossRef]
- Chrousos, G.P.; Kino, T.; Charmandari, E. Evaluation of the hypothalamic-pituitary-adrenal axis function in childhood and adolescence. Neuroimmunomodulat 2009, 16, 272–283. [Google Scholar] [CrossRef]
- Romero, L.M.; Remage-Healey, L. Daily and seasonal variation in response to stress in captive starlings (Sturnus vulgaris): Corticosterone. Gen. Comp. Endocrinol. 2000, 119, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M.; Krey, L.C.; McEwen, B.S. The neuroendocrinology of stress and aging: The glucocorticoid cascade hypothesis. Endocr. Rev. 1986, 7, 284–301. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Dornelles, F.; Preissler, T.; Collaziol, D.; da Cruz, I.M.; Bauer, M.E. Impact of psychological and endocrine factors on cytokine production of healthy elderly people. Mech. Ageing Dev. 2003, 124, 887–895. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Altmann, J. Incidence of hypercortisolism and dexamethasone resistance increases with age among wild baboons. Biol. Psychiat. 1991, 30, 1008–1016. [Google Scholar] [CrossRef]
- O’Brien, J.K.; Steinman, K.J.; Fetter, G.A.; Robeck, T.R. Androgen and glucocorticoid production in the male killer whale (Orcinus orca): Influence of age, maturity, and environmental factors. Andrology 2017, 5, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Muehlenbein, M.P.; Campbell, B.C.; Richards, R.J.; Svec, F.; Phillippi-Falkenstein, K.M.; Murchison, M.A.; Myers, L. Dehydroepiandrosterone-sulfate as a biomarker of senescence in male non-human primates. Exp. Gerontol. 2003, 38, 1077–1085. [Google Scholar] [CrossRef]
- Pieper, D.R.; Lobocki, C.A. Characterization of serum dehydroepiandrosterone secretion in golden hamsters. Proc. Soc. Exp. Biol. Med. 2000, 224, 278–284. [Google Scholar] [CrossRef]
- Rosado, B.; García-Belenguer, S.; Leon, M.; Chacón, G.; Villegas, A.; Palacio, J. Blood concentrations of serotonin, cortisol and dehydroepiandrosterone in aggressive dogs. Appl. Anim. Behav. Sci. 2010, 123, 124–130. [Google Scholar] [CrossRef]
- Soma, K.K.; Scotti, M.A.L.; Newman, A.E.; Charlier, T.D.; Demas, G.E. Novel mechanisms for neuroendocrine regulation of aggression. Front. Neuroendocr. 2008, 29, 476–489. [Google Scholar] [CrossRef]
- Van Weerden, W.M.; Bierings, H.G.; Vansteenbrugge, G.J.; de Jong, F.H.; Schröder, F.H. Adrenal glands of mouse and rat do not synthesize androgens. Life Sci. 1992, 50, 857–861. [Google Scholar] [CrossRef]
- Maninger, N.; Wolkowitz, O.M.; Reus, V.I.; Epel, E.S.; Mellon, S.H. Neurobiological and neuropsychiatric effects of dehydroepiandrosterone (DHEA) and DHEA sulfate (DHEAS). Front. Neuroendocr. 2009, 30, 65–91. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Zheng, P. Dehydroepiandrosterone sulphate: Action and mechanism in the brain. J. Neuroendocr. 2012, 24, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Kimonides, V.G.; Spillantini, M.G.; Sofroniew, M.V.; Fawcett, J.W.; Herbert, J. Dehydroepiandrosterone antagonizes the neurotoxic effects of corticosterone and translocation of stress-activated protein kinase 3 in hippocampal primary cultures. Neuroscience 1999, 89, 429–436. [Google Scholar] [CrossRef]
- Alhaj, H.A.; Massey, A.E.; McAllister-Williams, R.H. Effects of DHEA administration on episodic memory, cortisol and mood in healthy young men: A double-blind, placebo-controlled study. Psychopharmacology 2006, 188, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Flood, J.F.; Roberts, E. Dehydroepiandrosterone sulfate improves memory in aging mice. Brain Res. 1988, 448, 178–181. [Google Scholar] [CrossRef]
- Frye, C.A.; Lacey, E.H. The neurosteroids DHEA and DHEAS may influence cognitive performance by altering affective state. Physiol. Behav. 1999, 66, 85–92. [Google Scholar] [CrossRef]
- Alves, V.B.F.; Basso, P.J.; Nardini, V.; Silva, A.; Chica, J.E.L.; de Barros Cardoso, C.R. Dehydroepiandrosterone (DHEA) restrains intestinal inflammation by rendering leukocytes hyporesponsive and balancing colitogenic inflammatory responses. Immunobiology 2016, 221, 934–943. [Google Scholar] [CrossRef]
- Cao, J.; Zhang, H.; Yang, Z.; Zhao, J.; Ma, H. Effect of dehydroepiandrosterone on the immune response and gut microbiota in dextran sulfate sodium-induced colitis mice. Mol. Immunol. 2020, 118, 60–72. [Google Scholar] [CrossRef]
- Hazeldine, J.; Arlt, W.; Lord, J. Dehydroepiandrosterone as a regulator of immune cell function. J. Steroid Biochem. Mol. Biol. 2010, 120, 127–136. [Google Scholar] [CrossRef]
- O’Driscoll, K.; McCabe, M.; Earley, B. Differences in leukocyte profile, gene expression, and metabolite status of dairy cows with or without sole ulcers. J. Dairy Sci. 2015, 98, 1685–1695. [Google Scholar] [CrossRef]
- Buvat, J. Androgen therapy with dehydroepiandrosterone. World J. Urol. 2003, 21, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.; Carroll, D.; Gale, C.; Lord, J. Cortisol, DHEA sulphate, their ratio, and all-cause and cause-specific mortality in the Vietnam Experience Study. Eur. J. Endocrinol. 2010, 163, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Angele, M.K.; Catania, R.A.; Ayala, A.; Cioffi, W.G.; Bland, K.I.; Chaudry, I.H. Dehydroepiandrosterone (DHEA): An inexpensive steroid hormone which decreases the mortality from sepsis. Arch. Surg. 1998, 133, 1281–1288. [Google Scholar] [CrossRef] [PubMed]
- Jarrar, D.; Wang, P.; Cioffi, W.G.; Bland, K.I.; Chaudry, I.H. Mechanisms of the salutary effects of dehydroepiandrosterone after trauma-hemorrhage: Direct or indirect effects on cardiac and hepatocellular functions? Arch. Surg. 2000, 135, 416–423. [Google Scholar] [CrossRef]
- Knöferl, M.W.; Angele, M.K.; Catania, R.A.; Diodato, M.D.; Bland, K.I.; Chaudry, I.H. Immunomodulatory effects of dehydroepiandrosterone in proestrus female mice after trauma-hemorrhage. J. Appl. Physiol. 2003, 95, 529–535. [Google Scholar] [CrossRef]
- Boonstra, R.; Lane, J.E.; Boutin, S.; Bradley, A.; Desantis, L.; Newman, A.E.; Soma, K.K. Plasma DHEA levels in wild, territorial red squirrels: Seasonal variation and effect of ACTH. Gen. Comp. Endocr. 2008, 158, 61–67. [Google Scholar] [CrossRef]
- Fustini, M.; Galeati, G.; Gabai, G.; Mammi, L.E.; Bucci, D.; Baratta, M.; Accorsi, P.A.; Formigoni, A. Overstocking dairy cows during the dry period affects dehydroepiandrosterone and cortisol secretion. J. Dairy Sci. 2017, 100, 620–628. [Google Scholar] [CrossRef]
- Lennartsson, A.K.; Kushnir, M.M.; Bergquist, J.; Jonsdottir, I.H. DHEA and DHEA-S response to acute psychosocial stress in healthy men and women. Biol. Psychol. 2012, 90, 143–149. [Google Scholar] [CrossRef]
- Skarlandtová, H.; Bičíková, M.; NeuŽil, P.; Mlček, M.; Hrachovina, V.; Svoboda, T.; Medová, E.; Kudlička, J.; Dohnalová, A.; Havránek, Š.; et al. Are there any differences between stress hormone levels in non-stress conditions and in potentional stress overload (heart catheterisation) in sows? Physiol. Res. 2014, 63, 733–741. [Google Scholar]
- Edes, A.N.; Edwards, K.L.; Wolfe, B.A.; Brown, J.L.; Crews, D.E. Allostatic load indices with cholesterol and triglycerides predict disease and mortality risk in zoo-housed western lowland gorillas (Gorilla gorilla gorilla). Biomark. Insights 2020, 15, 1–13. [Google Scholar] [CrossRef]
- Hucklebridge, F.; Hussain, T.; Evans, P.; Clow, A. The diurnal patterns of the adrenal steroids cortisol and dehydroepiandrosterone (DHEA) in relation to awakening. Psychoneuroendocrinology 2005, 30, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Collaziol, D.; Preissler, T.; da Cruz, I.M.; Glock, L.; Bauer, M.E. Healthy aging is associated with unaltered production of immunoreactive growth hormone but impaired neuroimmunomodulation. Neuroimmunomodulatin 2006, 13, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Copeland, K.C.; Eichberg, J.W.; Parker, C.R.; Bartke, A., Jr. Puberty in the chimpanzee: Samatomedin-C and its relationship to somatic growth and steroid hormone concentrations. J. Clin. Endocr. Metab. 1985, 60, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Kemnitz, J.W.; Roecker, E.B.; Haffa, A.L.M.; Pinheiro, J.; Kurzman, I.; Ramsey, J.J.; MacEwen, E.G. Serum dehydroepiandrosterone sulfate concentrations across the life span of laboratory-housed rhesus monkeys. J. Med. Primatol. 2000, 29, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M.; Vodelman, J.H.; Orentreich, N.; Altmann, J. Senescent decline in serum dehydroepiandrosterone sulfate concentrations in a population of wild baboons. J. Gerontol. 1993, 48, B196–B200. [Google Scholar] [CrossRef] [PubMed]
- Sorwell, K.G.; Kohama, S.G.; Urbanski, H.F. Perimenopausal regulation of steroidogenesis in the nonhuman primate. Neurobiol. Aging 2012, 33, 1487.e1–1487.e13. [Google Scholar] [CrossRef]
- De Almeida, A.M.; Zachut, M.; Hernández-Castellano, L.E.; Šperanda, M.; Gabai, G.; Mobasheri, A. Biomarkers of fitness and welfare in dairy animals: Healthy living. J. Dairy Res. 2019, 86, 379–387. [Google Scholar] [CrossRef]
- Chen, F.; Knecht, K.; Birzin, E.; Fisher, J.; Wilkinson, H.; Mojena, M.; Moreno, C.T.; Schmidt, A.; Harada, S.; Freedman, L.P.; et al. Direct agonist/antagonist functions of dehydroepiandrosterone. Endocrinology 2005, 146, 4568–4576. [Google Scholar] [CrossRef]
- Sollberger, S.; Ehlert, U. How to use and interpret hormone ratios. Psychoneuroendocrinology 2016, 63, 385–397. [Google Scholar] [CrossRef]
- Ferrari, E.; Cravello, L.; Muzzoni, B.; Casarotti, D.; Paltro, M.; Solerte, S.B.; Fioravanti, M.; Cuzzoni, G.; Pontiggia, B.; Magri, F. Age-related changes of the hypothalamic-pituitary-adrenal axis: Pathophysiological correlates. Eur. J. Endocrinol. 2001, 144, 319–329. [Google Scholar] [CrossRef]
- Goodyer, I.M.; Herbert, J.; Altham, P.M.E. Adrenal steroid secretion and major depression in 8- to 16-year-olds, III. Influence of cortisol/DHEA ratio at presentation on subsequent rates of disappointing life events and persistent major depression. Psychol. Med. 1998, 28, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J.; Huang, Y.S.; Hsiao, C.C.; Chiang, Y.L.; Wu, C.C.; Shang, Z.Y.; Chen, C.K. Salivary dehydroepiandrosterone, but not cortisol, is associated with attention deficit hyperactivity disorder. World J. Biol. Psychiatry 2011, 12, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Jeckel, C.M.M.; Lopes, R.P.; Berleze, M.C.; Luz, C.; Feix, L.; de Lima Argimon, I.I.; Stein, L.M.; Bauer, M.E. Neuroendocrine and immunological correlates of chronic stress in “strictly healthy” populations. Neuroimmunomodulation 2010, 17, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Beishuizen, A.; Thijs, L.G.; Vermes, I. Decreased levels of dehydroepiandrosterone sulphate in severe critical illness: A sign of exhausted adrenal reserve? Crit. Care 2002, 6, 434–438. [Google Scholar] [CrossRef]
- Butcher, S.K.; Killampalli, V.; Lascelles, D.; Wang, K.; Alpar, E.K.; Lord, J.M. Raised cortisol: DHEAS ratios in the elderly after injury: Potential impact upon neutrophil function and immunity. Aging Cell 2005, 4, 319–324. [Google Scholar] [CrossRef]
- Buford, T.W.; Willoughby, D.S. Impact of DHEA(S) and cortisol on immune function in aging: A brief review. Appl. Physiol. Nutr. Metab. 2008, 33, 429–433. [Google Scholar] [CrossRef]
- Kalmijn, S.; Launer, L.J.; Stolk, R.P.; de Jong, F.H.; Pols, H.A.P.; Hofman, A.; Breteler, M.M.B.; Lamberts, S.W.J. A prospective study on cortisol, dehydroepiandrosterone sulfate, and cognitive function in the elderly. J. Clin. Endocr. Metab. 1998, 83, 3487–3492. [Google Scholar] [CrossRef]
- Heaney, J.L.J.; Carroll, D.; Phillips, A.C. Physical activity, life events stress, cortisol, and DHEA: Preliminary findings that physical activity may buffer against the negative effects of stress. J. Aging Phys. Act. 2014, 22, 465–473. [Google Scholar] [CrossRef]
- Markopoulou, K.; Papadopoulos, A.; Juruena, M.F.; Poon, L.; Pariante, C.M.; Cleare, A.J. The ratio of cortisol/DHEA in treatment resistant depression. Psychoneuroendocrinology 2009, 34, 19–26. [Google Scholar] [CrossRef]
- Morgan, C.; Rasmusson, A.; Pietrzak, R. Relationships among plasma dehydroepiandrosterone and dehydroepiandrosterone sulfate, cortisol, symptoms of dissociation, and objective performance in humans. Biol. Psychiat. 2009, 66, 334–340. [Google Scholar] [CrossRef]
- Rasmusson, A.; Vasek, J.; Lipschitz, D.S.; Vojvoda, D.; Mustone, M.E.; Shi, Q.; Gudmundsen, G.; Morgan, C.A.; Wolfe, J.; Charney, D.S. An increased capacity for adrenal DHEA release is associated with decreased avoidance and negative mood symptoms in women with PTSD. Neuropsychopharmacology 2004, 29, 1546–1557. [Google Scholar] [CrossRef] [PubMed]
- Ritsner, M.; Maayan, R.; Gibel, A.; Strous, R.D.; Modai, I.; Weizman, A. Elevation of the cortisol/dehydroepiandrosterone ratio in schizophrenia patients. Eur. Neuropsychopharmacol. 2004, 14, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Van Niekerk, J.K.; Huppert, F.A.; Herbert, J. Salivary cortisol and DHEA: Association with measures of cognition and well-being in normal older men, and effects of three months of DHEA supplementation. Psychoneuroendocrinology 2001, 26, 591–612. [Google Scholar] [CrossRef]
- Goodyer, I.M.; Park, R.J.; Netherton, C.M.; Herbert, J. Possible role of cortisol and dehydroepiandrosterone in human development and psychopathology. Br. J. Psychiatry 2001, 179, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Pajer, K.; Tabbah, R.; Gardner, W.; Rubin, R.T.; Czambel, R.K.; Wang, Y. Adrenal androgen and gonadal hormone levels in adolescent girls with conduct disorder. Psychoneuroendocrinology 2006, 31, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Bergamin, C.; Comin, A.; Corazzin, M.; Faustini, M.; Peric, T.; Scollo, A.; Gottardo, F.; Montillo, M.; Prandi, A. Cortisol, DHEA, and sexual steroid concentrations in fattening pigs’ hair. Animals 2019, 9, 345. [Google Scholar] [CrossRef]
- Goncharova, N.D.; Lapin, B.A. Changes of hormonal function of the adrenal and gonadal glands in baboons of different age groups. J. Med. Primatol. 2000, 29, 26–35. [Google Scholar] [CrossRef]
- Goncharova, N.D.; Lapin, B.A. Age-related endocrine dysfunction in nonhuman primates. Ann. N. Y. Acad. Sci. 2004, 1019, 321–325. [Google Scholar] [CrossRef]
- Carlström, K.; Karlsson, R.; Schoultz, B.V. Diurnal rhythm and effects of oral contraceptives on serum dehydroepiandrosterone sulfate (DHEAS) are related to alterations in serum albumin rather than to changes in adrenocortical steroid secretion. Scand. J. Clin. Lab. Investig. 2002, 62, 361–368. [Google Scholar] [CrossRef]
- Ghiciuc, C.M.; Cozma-Dima, C.L.; Pasquali, V.; Renzi, P.; Simeoni, S.; Lupusoru, C.E.; Patacchioli, F.R. Awakening responses and diurnal fluctuations of salivary cortisol, DHEA-S and alpha-amylase in healthy male subjects. Neuroendocrinol. Lett. 2011, 32, 475–480. [Google Scholar]
- King, J.A.; Rosal, M.C.; Ma, Y.; Reed, G.; Kelly, T.A.; Ockene, I.S. Sequence and seasonal effects of salivary cortisol. Behav. Med. 2000, 26, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Nunes, S.; Pelz, K.M.; Muecke, E.M.; Holekamp, K.E.; Zucker, I. Plasma glucocorticoid concentrations and body mass in ground squirrels: Seasonal variation and circannual organization. Gen. Comp. Endocr. 2006, 146, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Huber, S.; Palme, R.; Arnold, W. Effects of season, sex, and sample collection on concentrations of fecal cortisol metabolites in red deer (Cervus elaphus). Gen. Comp. Endocr. 2003, 130, 48–54. [Google Scholar] [CrossRef]
- Lynch, J.W.; Ziegler, T.E.; Strier, K.B. Individual and seasonal variation in fecal testosterone and cortisol levels of wild male tufted capuchin monkeys, Cebus paella nigritus. Horm. Behav. 2002, 41, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Wiebe, R.H.; Williams, L.E.; Abee, C.R.; Yeoman, R.R.; Diamond, E.J. Seasonal changes in serum dehydroepiandrosterone, androstenedione, and testosterone levels in the squirrel monkey (Saimiri boliviensis boliviensis). Am. J. Primatol. 1988, 14, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Goncharova, N.D.; Lapin, B.A. Effects of aging on hypothalamic–pituitary–adrenal system function in non-human primates. Mech. Ageing Dev. 2002, 123, 1191–1201. [Google Scholar] [CrossRef]
- Hinson, J.P.; Raven, P.W. DHEA deficiency syndrome: A new term for old age? J. Endocrinol. 1999, 163, 1–5. [Google Scholar] [CrossRef]
- Möstl, E.; Palme, R. Hormones as indicators of stress. Domest. Anim. Endocrin. 2002, 23, 67–74. [Google Scholar] [CrossRef]
- Creel, S.; Dantzer, B.; Goymann, W.; Rubenstein, D.R. The ecology of stress: Effects of the social environment. Funct. Ecol. 2013, 27, 66–80. [Google Scholar] [CrossRef]
- Creel, S. Social dominance and stress hormones. Trends Ecol. Evol. 2001, 16, 491–497. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Alberts, S.C.; Altmann, J. Hypercortisolism associated with social subordinance or social-isolation among wild baboons. Arch. Gen. Psychiat. 1997, 54, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Steyer, R.; Schmitt, M.; Eid, M. Latent state–trait theory and research in personality and individual differences. Eur. J. Personal. 1999, 13, 389–408. [Google Scholar] [CrossRef]
- Kudielka, B.M.; Kirschbaum, C. Sex differences in HPA axis responses to stress: A review. Biol. Psychol. 2005, 69, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Palme, R. Non-invasive measurement of glucocorticoids: Advances and problems. Physiol. Behav. 2019, 199, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Kersey, D.C.; Dehnhard, M. The use of noninvasive and minimally invasive methods in endocrinology for threatened mammalian species conservation. Gen. Comp. Endocr. 2014, 203, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Sheriff, M.J.; Dantzer, B.; Delehanty, B.; Palme, R.; Boonstra, R. Measuring stress in wildlife: Techniques for quantifying glucocorticoids. Oecologia 2011, 166, 869–887. [Google Scholar] [CrossRef]
- Bardi, M.; Hampton, J.E.; Lambert, K.G. Fecal dehydroepiandrosterone (DHEA) immunoreactivity as a noninvasive index of circulating DHEA activity in young male laboratory rats. Comp. Med. 2010, 60, 455–460. [Google Scholar]
- Henning, S.J. Plasma concentrations of total and free corticosterone during development in the rat. Am. J. Physiol.-Endoc. Metab. 1978, 235, E451–E456. [Google Scholar] [CrossRef]
- Barsano, C.P.; Baumann, G. Simple algebraic and graphic methods for the apportionment of hormone (and receptor) into bound and free fractions in binding equilibria; or how to calculate bound and free hormone? Endocrinology 1989, 124, 1101–1106. [Google Scholar] [CrossRef]
- Teruhisa, U.; Ryoji, H.; Taisuke, I.; Tatsuya, S.; Fumihiro, M.; Tatsuo, S. Use of saliva for monitoring unbound free cortisol levels in serum. Clin. Chim. Acta 1981, 110, 245–253. [Google Scholar] [CrossRef]
- Lutz, C.K.; Tiefenbacher, S.; Jorgensen, M.J.; Meyer, J.S.; Novak, M.A. Techniques for collecting saliva from awake, unrestrained, adult monkeys for cortisol assay. Am. J. Primatol. 2000, 52, 93–99. [Google Scholar] [CrossRef]
- Kobelt, A.J.; Hemsworth, P.H.; Barnett, J.L.; Butler, K.L. Sources of sampling variation in saliva cortisol in dogs. Res. Vet. Sci. 2003, 75, 157–161. [Google Scholar] [CrossRef]
- Gomez, A.; Jewell, E.; Walker, S.L.; Brown, J.L. Use of salivary steroid analyses to assess ovarian cycles in an Indian rhinoceros at the National Zoological Park. Zoo Biol. 2004, 23, 501–512. [Google Scholar] [CrossRef]
- Menargues, A.; Urios, V.; Mauri, M. Welfare assessment of captive Asian elephants (Elephas maximus) and Indian rhinoceros (Rhinoceros unicornis) using salivary cortisol measurement. Anim. Welf. 2008, 17, 305–312. [Google Scholar]
- Behringer, V.; Borchers, C.; Deschner, T.; Möstl, E.; Selzer, D.; Hohmann, G. Measurements of salivary alpha amylase and salivary cortisol in hominoid primates reveal within-species consistency and between-species differences. PLoS ONE 2013, 8, e60773. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, P.; Leitch, M.M.; Massey, A.E.; McAllister-Williams, R.H.; Young, A.H. Assessing cortisol and dehydroepiandrosterone (DHEA) in saliva: Effects of collection method. J. Psychopharmacol. 2006, 20, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Shirtcliff, E.A.; Granger, D.A.; Schwartz, E.; Curran, M.J. Use of salivary biomarkers in biobehavioral research: Cotton-based sample collection methods can interfere with salivary immunoassay results. Psychoneuroendocrinology 2001, 26, 165–173. [Google Scholar] [CrossRef]
- Granger, D.A.; Johnson, S.B.; Szanton, S.L.; Out, D.; Schumann, L.L. Incorporating salivary biomarkers into nursing research: An overview and review of best practices. Biol. Res. Nurs. 2012, 14, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.A.; Cicchetti, D.; Rogosch, F.A.; Hibel, L.C.; Teisl, M.; Flores, E. Blood contamination in children’s saliva: Prevalence, stability, and impact on the measurement of salivary cortisol, testosterone, and dehydroepiandrosterone. Psychoneuroendocrinology 2007, 32, 724–733. [Google Scholar] [CrossRef]
- Bahr, N.I.; Palme, R.; Möhle, U.; Hodges, J.K.; Heistermann, M. Comparative aspects of the metabolism and excretion of cortisol in three individual nonhuman primates. Gen. Comp. Endocr. 2000, 117, 427–438. [Google Scholar] [CrossRef]
- Behringer, V.; Hohmann, G.; Stevens, J.M.; Weltring, A.; Deschner, T. Adrenarche in bonobos (Pan paniscus): Evidence from ontogenetic changes in urinary dehydroepiandrosterone-sulfate levels. J. Endocrinol. 2012, 214, 55. [Google Scholar] [CrossRef] [PubMed]
- Teskey-Gerstl, A.; Bamberg, E.; Steineck, T.; Palme, R. Excretion of corticosteroids in urine and faeces of hares (Lepus europaeus). J. Comp. Physiol. B 2000, 170, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Price, E.; Coleman, R.; Ahsmann, J.; Glendewar, G.; Hunt, J.; Smith, T.; Wormell, D. Individual, social, and environmental factors affecting salivary and fecal cortisol levels in captive pied tamarins (Saguinus bicolor). Am. J. Primatol. 2019, 81. [Google Scholar] [CrossRef] [PubMed]
- Pauling, C.D.; Lankford, S.E.; Jackson, V.L. Fecal cortisol levels in scimitar-horned oryx, Oryx dammah, reveals differences between captive environments. J. Zoo Wildl. Med. 2017, 48, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Heistermann, M.; Palme, R.; Ganswindt, A. Comparison of different enzymeimmunoassays for assessment of adrenocortical activity in primates based on fecal analysis. Am. J. Primatol. 2006, 68, 257–273. [Google Scholar] [CrossRef]
- Millspaugh, J.J.; Washburn, B.E. Use of fecal glucocorticoid metabolite measures in conservation biology research: Considerations for application and interpretation. Gen. Comp. Endocr. 2004, 138, 189–199. [Google Scholar] [CrossRef]
- Harlow, H.J.; Beck, T.D.I.; Walters, L.M.; Greenhouse, S.S. Seasonal serum glucose, progesterone, and cortisol levels of black bears (Ursus americanus). Can. J. Zool. 1990, 68, 183–187. [Google Scholar] [CrossRef]
- Gregory, L.F.; Schmid, J.R. Stress responses and sexing of wild Kemps Ridley Sea Turtles (Lepidochelys kempii) in the Northeastern Gulf of Mexico. Gen. Comp. Endocr. 2001, 124, 66–74. [Google Scholar] [CrossRef]
- Hood, L.C.; Boersma, P.D.; Wingfield, J.C. The adrenocortical response to stress in incubating magellanic penguins (Spheniscus magellanicus). Auk 1998, 115, 76–84. [Google Scholar] [CrossRef]
- Mathies, T.; Felix, T.A.; Lance, V.A. Effects of trapping and subsequent short-term confinement stress on plasma corticosterone in the brown treesnake (Boiga irregularis) on Guam. Gen. Comp. Endocr. 2001, 124, 106–114. [Google Scholar] [CrossRef]
- Padgett, D.A.; Marucha, P.T.; Sheridan, J.F. Restraint stress slows cutaneous wound healing in mice. Brain Behav. Immun. 1998, 12, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Chrousos, G.P.; Renquist, D.; Brandon, D.; Eil, C.; Pugeat, M.; Vigersky, R.; Cutler, G.B.; Loriaux, D.L.; Lipsett, M.B. Glucocorticoid hormone resistance during primate evolution: Receptor-mediated mechanisms. Proc. Natl. Acad. Sci. USA 1982, 79, 2036–2040. [Google Scholar] [CrossRef] [PubMed]
- LeMaho, Y.; Karmann, H.; Briot, D.; Handrich, Y. Stress in birds due to routine handling and a technique to avoid it. Am. J. Physiol. 1992, 263, R775–R781. [Google Scholar] [CrossRef]
- Ortiz, R.M.; Worthy, G.A.; MacKenzie, D.S. Osmoregulation in wild and captive West Indian manatees (Trichechus manatus). Physiol. Zool. 1998, 71, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Naidenko, S.V.; Antonevich, A.L.; Erofeeva, M.N. Hormonal response to a synthetic analog of adrenocorticotropic hormone in lynx pups (Lynx lynx L.). Dokl. Biol. Sci. 2007, 416, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Bergamasco, L.; Osella, M.C.; Savarino, P.; Larosa, G.; Ozella, L.; Manassero, M.; Badino, P.; Odore, R.; Barbero, R.; Re, G. Heart rate variability and saliva cortisol assessment in shelter dog: Human–animal interaction effects. Appl. Anim. Behav. Sci. 2010, 125, 56–68. [Google Scholar] [CrossRef]
- Anestis, S.F.; Bribiescas, R.G. Rapid changes in chimpanzee (Pan troglodytes) urinary cortisol excretion. Horm. Behav. 2004, 45, 209–213. [Google Scholar] [CrossRef]
- Quinkler, M.; Stewart, P.M. Hypertension and the cortisol-cortisone shuttle. J. Clin. Endocr. Metab. 2003, 88, 2384–2392. [Google Scholar] [CrossRef]
- Hammer, F.; Stewart, P.M. Cortisol metabolism in hypertension. Best Pract. Res. Clin. Endocrinol. Metab. 2006, 20, 337–353. [Google Scholar] [CrossRef]
- Gatti, R.; Antonelli, G.; Prearo, M.; Spinella, P.; Cappellin, E.; Elio, F. Cortisol assays and diagnostic laboratory procedures in human biological fluids. Clin. Biochem. 2009, 42, 1205–1217. [Google Scholar] [CrossRef]
- Castillo, V.A.; Gomez, N.V.; Lalia, J.C.; Blatter, M.C.; Garcia, J.D. Cushing’s disease in dogs: Cabergoline treatment. Res. Vet. Sci. 2008, 85, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Czekala, N.M.; Lance, V.A.; Sutherland-Smith, M. Diurnal urinary corticoid excretion in the human and gorilla. Am. J. Primatol. 1994, 34, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Goossens, M.M.C.; Meyer, H.P.; Voorhout, G.; Sprang, E.P.M. Urinary excretion of glucocorticoids in the diagnosis of hyperadrenocorticism in cats. Domest. Anim. Endocrin. 1995, 12, 355–362. [Google Scholar] [CrossRef]
- Narayan, E.; Molinia, F.; Christi, K.; Morley, C.; Cockrem, J. Urinary corticosterone metabolite responses to capture, and annual patterns of urinary corticosterone in wild and captive endangered Fijian ground frogs (Platymantis vitiana). Aust. J. Zool. 2010, 58, 189–197. [Google Scholar] [CrossRef]
- Brown, J.L.; Wemmer, C.M.; Lehnhardt, J. Urinary cortisol analysis for monitoring adrenal activity in elephants. Zoo Biol. 1995, 14, 533–542. [Google Scholar] [CrossRef]
- McCobb, E.C.; Patronek, G.J.; Marder, A.; Dinnage, J.D.; Stone, M.S. Assessment of stress levels among cats in four animal shelters. JAVMA-J. Am. Vet. Med. A. 2005, 226, 548–555. [Google Scholar] [CrossRef]
- Beerda, B.; Schilder, M.B.; Janssen, N.S.; Mol, J.A. The use of saliva cortisol, urinary cortisol, and catecholamine measurements for a noninvasive assessment of stress responses in dogs. Horm. Behav. 1996, 30, 272–279. [Google Scholar] [CrossRef]
- Bennett, C.; Fripp, D.; Othen, L.; Jarsky, T.; French, J.; Loskutoff, N. Urinary corticosteroid excretion patterns in the okapi (Okapia johnstoni). Zoo Biol. 2008, 27, 381–393. [Google Scholar] [CrossRef]
- Robbins, M.M.; Czekala, N.M. A preliminary investigation of urinary testosterone and cortisol levels in wild male mountain gorillas. Am. J. Primatol. 1997, 43, 51–64. [Google Scholar] [CrossRef]
- Kintz, P.; Cirimele, V.; Ludes, B. Physiological concentrations of DHEA in human hair. J. Anal. Toxicol. 1999, 23, 424–428. [Google Scholar] [CrossRef]
- Siiteri, P.K. The continuing saga of dehydroepiandrosterone (DHEA). J. Clin. Endocrinol. Metab. 2005, 90, 3795–3796. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Campbell, B. Adrenarche and the evolution of human life history. Am. J. Hum. Biol. 2006, 18, 569–589. [Google Scholar] [CrossRef] [PubMed]
- Legagneux, P.; Gauthier, G.; Chastel, O.; Picard, G.; Bêty, J. Do glucocorticoids in droppings reflect baseline level in birds captured in the wild? A case study in snow geese. Gen. Comp. Endocr. 2011, 172, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Palme, R.; Rettenbacher, S.; Touma, C.; El-Bahr, S.M.; Möstl, E. Stress hormones in mammals and birds: Comparative aspects regarding metabolism, excretion, and noninvasive measurement in fecal samples. Ann. N. Y. Acad. Sci. 2005, 1040, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Hunt, K.E.; Rolland, R.M.; Kraus, S.D.; Wasser, S.K. Analysis of fecal glucocorticoids in the North Atlantic right whale (Eubalaena glacialis). Gen. Comp. Endocr. 2006, 148, 260–272. [Google Scholar] [CrossRef]
- Watson, R.; Munro, C.; Edwards, K.L.; Norton, V.; Brown, J.L.; Walker, S.L. Development of a versatile enzyme immunoassay for non-invasive assessment of glucocorticoid metabolites in a diversity of taxonomic species. Gen. Comp. Endocr. 2013, 186, 16–24. [Google Scholar] [CrossRef]
- Seraphin, S.B.; Whitten, P.L.; Reynolds, V. The influence of age on fecal steroid hormone levels in male Budongo Forest chimpanzees (Pan troglodytes schweinfurthii). Am. J. Primatol. 2008, 70, 661–669. [Google Scholar] [CrossRef]
- Takeshita, R.S.; Bercovitch, F.B.; Huffman, M.A.; Mouri, K.; Garcia, C.; Rigaill, L.; Shimizu, K. Environmental, biological, and social factors influencing fecal adrenal steroid concentrations in female Japanese macaques (Macaca fuscata). Am. J. Primatol. 2014, 76, 1084–1093. [Google Scholar] [CrossRef]
- Wennig, R. Potential problems with the interpretation of hair analysis results. Forensic Sci. Int. 2000, 107, 5–12. [Google Scholar] [CrossRef]
- Kirschbaum, C.; Tietze, A.; Skoluda, N.; Dettenborn, L. Hair as a retrospective calendar of cortisol production—Increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology 2009, 34, 32–37. [Google Scholar] [CrossRef]
- Fourie, N.H.; Bernstein, R.M. Hair cortisol levels track phylogenetic and age related differences in hypothalamic–pituitary–adrenal (HPA) axis activity in non-human primates. Gen. Comp. Endocr. 2011, 174, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Qiao, S.; Li, X.; Zilioli, S.; Chen, Z.; Deng, H.; Pan, J.; Guo, W. Hair measurements of cortisol, DHEA, and DHEA to cortisol ratio as biomarkers of chronic stress among people living with HIV in China: Known-group validation. PLoS ONE 2017, 12, e0169827. [Google Scholar] [CrossRef] [PubMed]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef] [PubMed]
- De Berker, D.A.R.; André, J.; Baran, R. Nail biology and nail science. Int. J. Cosmet. Sci. 2007, 29, 241–275. [Google Scholar] [CrossRef] [PubMed]
- Warnock, F.; McElwee, K.; Seo, R.J.; McIsaac, S.; Seim, D.; Ramirez-Aponte, T.; Macritchie, K.A.N.; Young, A.H. Measuring cortisol and DHEA in fingernails: A pilot study. Neuropsych. Dis. Treat. 2010, 6, 1–7. [Google Scholar]
- Lendvai, Á.Z.; Giraudeau, M.; Németh, J.; Bakó, V.; McGraw, K.J. Carotenoid-based plumage coloration reflects feather corticosterone levels in male house finches (Haemorhous mexicanus). Behav. Ecol. Sociobiol. 2013, 67, 1817–1824. [Google Scholar] [CrossRef]
- Bortolotti, G.R.; Marchant, T.A.; Blas, J.; German, T. Corticosterone in feathers is a long-term, integrated measure of avian stress physiology. Funct. Ecol. 2008, 22, 494–500. [Google Scholar] [CrossRef]
- Berkvens, C.N.; Hyatt, C.; Gilman, C.; Pearl, D.L.; Barker, I.K.; Mastromonaco, G.F. Validation of a shed skin corticosterone enzyme immunoassay in the African House Snake (Lamprophis fuliginosus) and its evaluation in the Eastern Massasauga Rattlesnake (Sistrurus catenatus catenatus). Gen. Comp. Endocr. 2013, 194, 1–9. [Google Scholar] [CrossRef]
- Scott, A.P.; Hirschenhauser, K.; Bender, N.; Oliveira, R.; Earley, R.L.; Sebire, M.; Ellis, T.; Pavlidis, M.; Hubbard, P.C.; Huertas, M.; et al. Non-invasive measurement of steroids in fish-holding water: Important considerations when applying the procedure to behaviour studies. Behaviour 2008, 145, 1307–1328. [Google Scholar]
- Möstl, E.; Rettenbacher, S.; Palme, R. Measurement of corticosterone metabolites in birds’ droppings: An analytical approach. Ann. N. Y. Acad. Sci. 2005, 1046, 17–34. [Google Scholar] [CrossRef]
- Palme, R. Measuring fecal steroids: Guidelines for practical application. Ann. N. Y. Acad. Sci. 2005, 1046, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Munro, C.; Stabenfeldt, G. Development of a cortisol enzyme-immunoassay in plasma. Clin. Chem. 1985, 31, e956. [Google Scholar]
- Wingfield, J.C.; Vleck, C.M.; Moore, M.C. Seasonal changes of the adrenocortical response to stress in birds of the Sonoran Desert. J. Exp. Zool. 1992, 264, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Dorgan, J.F.; Stanczyk, F.Z.; Longcope, C.; Stephenson, H.E.; Chang, L.; Miller, R.; Franz, C.; Falk, R.T.; Kahle, L. Relationship of serum dehydroepiandrosterone (DHEA), DHEA sulfate, and 5-androstene-3 beta, 17 beta-diol to risk of breast cancer in postmenopausal women. Cancer Epidem. Biomar. 1997, 6, 177–181. [Google Scholar]
- Buchanan, K.L.; Goldsmith, A.R. Noninvasive endocrine data for behavioural studies: The importance of validation. Anim. Behav. 2004, 67, 183–185. [Google Scholar] [CrossRef]
- Knott, S.A.; Cummins, L.J.; Dunshea, F.R.; Leury, B.J. Rams with poor feed efficiency are highly responsive to an exogenous adrenocorticotropin hormone (ACTH) challenge. Domest. Anim. Endocrin. 2008, 34, 261–268. [Google Scholar] [CrossRef]
- Juliana, J.R.S.; Bryant, J.L.; Wielebnowski, N.; Kotler, B.P. Physiological validation of a non-invasive method to evaluate adrenocortical activity and the time course for the excretion of stress hormones in the feces of three species of desert gerbils. Isr. J. Ecol. Evol. 2019, 65, 21–27. [Google Scholar] [CrossRef]
- Zimprich, A.; Garrett, L.; Deussing, J.M.; Wotjak, C.T.; Fuchs, H.; Gailus-Durner, V.; de Angelis, M.H.; Wurst, W.; Hölter, S.M. A robust and reliable non-invasive test for stress responsivity in mice. Front. Behav. Neurosci. 2014, 8, e125. [Google Scholar] [CrossRef]
- Oberbeck, R.; Benschop, R.J.; Jacobs, R.; Hosch, W.; Jetschmann, J.U.; Schürmeyer, T.H.; Schmidt, R.E.; Schedlowski, M. Endocrine mechanisms of stress-induced DHEA-secretion. J. Endocrinol. Invest. 1998, 21, 148–153. [Google Scholar] [CrossRef]
- Jensen, M.A.; Mortier, L.; Koh, E.; Keevil, B.; Hyttinen, S.; Hansen, Å.M. An interlaboratory comparison between similar methods for determination of melatonin, cortisol and testosterone in saliva. Scand. J. Clin. Lab. Inv. 2014, 74, 454–461. [Google Scholar] [CrossRef]
- Moran, F.M.; Chen, J.; Gee, N.A.; Lohstroh, P.; Lasley, B.L. Dehydroepiandrosterone sulfate (DHEAS) levels reflect endogenous LH production and response to human chorionic gonadotropin (hCG) challenge in the older female macaque (Macaca fascicularis). Menopause 2013, 20, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Robertson, T.; Benzeval, M.; Whitley, E.; Popham, F. The role of material, psychosocial and behavioral factors in mediating the association between socioeconomic position and allostatic load (measured by cardiovascular, metabolic and inflammatory markers). Brain Behav. Immun. 2015, 45, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Mason, G.J. Species differences in responses to captivity: Stress, welfare and the comparative method. Trends Ecol. Evol. 2010, 25, 713–721. [Google Scholar] [CrossRef] [PubMed]
| Sample Type | Description | Main Advantages | Main Disadvantages |
|---|---|---|---|
| Blood | Offers point-in-time hormone value, providing insight into the subject’s current state [118]. | Directly measures the hormone [118]. | Invasive—may cause capture-induced stress [117,118,119]. May have to distinguish between bound and unbound, free hormone [120,121]. |
| Saliva | Offers point-in-time hormone value similar to blood hormone concentrations [122]. | Directly measures unbound, free concentration of the hormone [122]. Non-to-minimally invasive. Little disruption to the subject’s routine [22,123,124]. | Subject must be trained and willing to cooperate [22,117,125,126,127]. Collection methods and sampling materials may impact measurement [128,129]. Must examine salivary flow rate [130]. Must avoid contamination from food [131]. |
| Urine | It may be possible to detect the native hormone [132,133]. Metabolites offer a cumulative, pooled concentration [116,132]. | Non-to-minimally invasive. Little disruption to the subject’s routine [116,118]. | Requires either training the subject OR designing/modifying holding areas for sample collection [118]. Must determine excretion rates [134]. |
| Feces | It may be possible to detect the native hormone [135,136]. Metabolites offer a cumulative, pooled concentration [119,137]. | Non-invasive. No disruption to the subject’s routine [9,138]. | May need to add a marker (e.g., food coloring) to distinguish the samples of socially-housed animals [116]. Must determine excretion rates [116]. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whitham, J.C.; Bryant, J.L.; Miller, L.J. Beyond Glucocorticoids: Integrating Dehydroepiandrosterone (DHEA) into Animal Welfare Research. Animals 2020, 10, 1381. https://doi.org/10.3390/ani10081381
Whitham JC, Bryant JL, Miller LJ. Beyond Glucocorticoids: Integrating Dehydroepiandrosterone (DHEA) into Animal Welfare Research. Animals. 2020; 10(8):1381. https://doi.org/10.3390/ani10081381
Chicago/Turabian StyleWhitham, Jessica C., Jocelyn L. Bryant, and Lance J. Miller. 2020. "Beyond Glucocorticoids: Integrating Dehydroepiandrosterone (DHEA) into Animal Welfare Research" Animals 10, no. 8: 1381. https://doi.org/10.3390/ani10081381
APA StyleWhitham, J. C., Bryant, J. L., & Miller, L. J. (2020). Beyond Glucocorticoids: Integrating Dehydroepiandrosterone (DHEA) into Animal Welfare Research. Animals, 10(8), 1381. https://doi.org/10.3390/ani10081381





