Genetic Parameters of Linear Type Traits Scored at 30 Months in Italian Heavy Draught Horse
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of Data
2.2. Estimates of (Co)Variance Components, Correlations and Genetic Trend
3. Results
3.1. Description of Data
3.2. Estimates of (Co)Variance Components
3.3. Genetic and Phenotypic Correlations
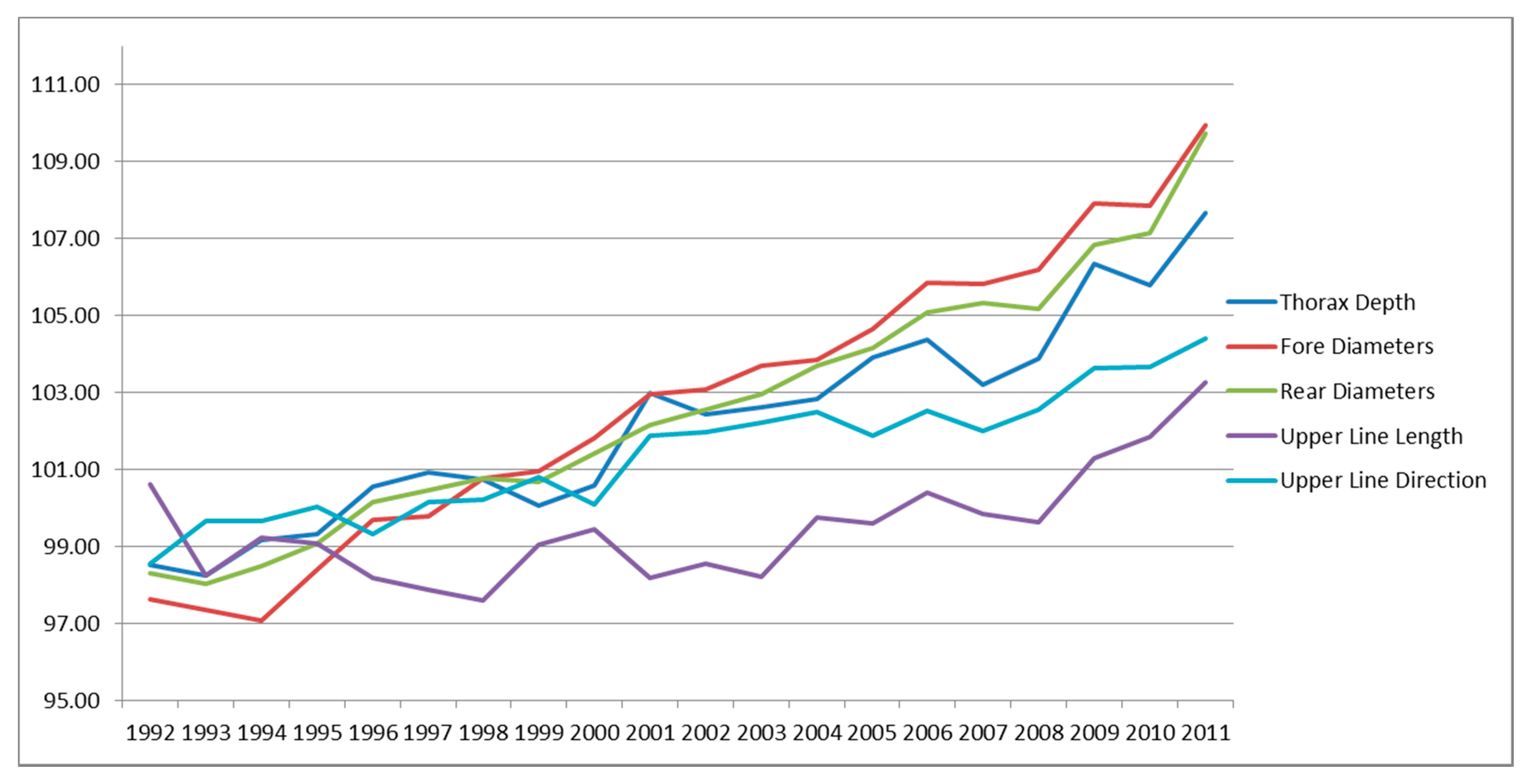
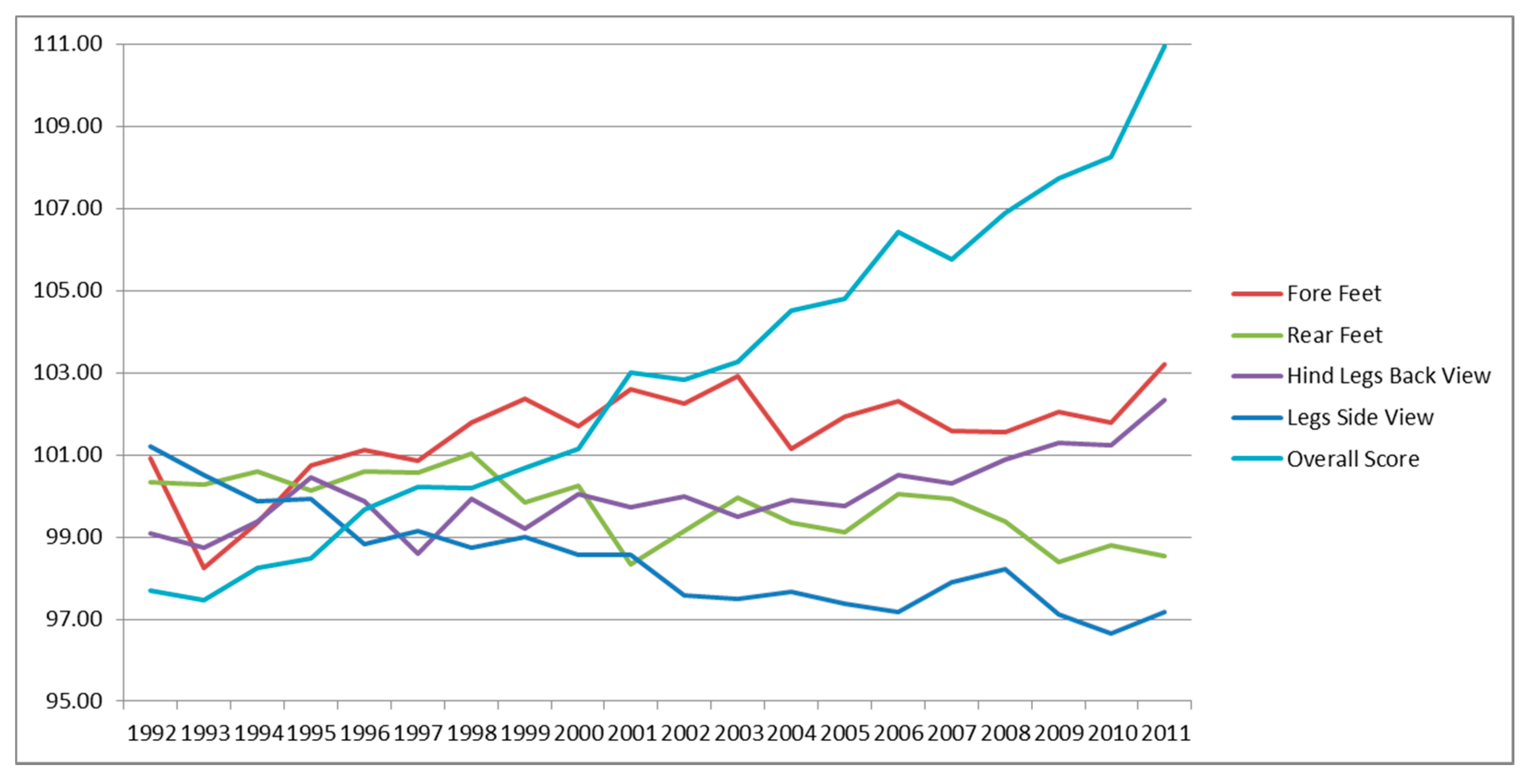
3.4. Genetic Trends of Traits
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Duensing, J.; Stock, K.F.; Krieter, J. Implementation and Prospects of Linear Pro fi ling in the Warmblood Horse. J. Equine Vet. Sci. 2014, 34, 360–368. [Google Scholar] [CrossRef]
- Saastamoinen, M.T.; Barrey, E. Genetics of conformation, locomotion and physiological traits. In The Genetics of the Horse; CABI Publishing: Wallingford, Oxon, UK, 2009. [Google Scholar]
- Thompson, J.R.; Freeman, A.E.; Wilson, D.J.; Chapin, C.A.; Berger, P.J.; Kuck, A. Evaluation of a Linear Type Program in Holsteins. J. Dairy Sci. 1981. [Google Scholar] [CrossRef]
- Koenen, E.P.C.; van Veldhuizen, A.E.; Brascamp, E.W. Genetic parameters of linear scored conformation traits and their relation to dressage and show-jumping performance in the Dutch Warmblood Riding Horse population. Livest. Prod. Sci. 1995, 43, 85–94. [Google Scholar] [CrossRef]
- Samoré, A.B.; Pagnacco, G.; Miglior, F. Genetic parameters and breeding values for linear type traits in the Haflinger horse. Livest. Prod. Sci. 1997, 52, 105–111. [Google Scholar] [CrossRef]
- Druml, T.; Baumung, R.; Sölkner, J. Morphological analysis and effect of selection for conformation in the Noriker draught horse population. Livest. Sci. 2008, 115, 118–128. [Google Scholar] [CrossRef]
- Fioretti, M.; Catalano, A.L.; Rosati, A.; Martuzzi, F. Bardigiano horse selection: A genetic global index for linear type traits. Publ. Assoc. Anim. Prod. 2005, 116, 147. [Google Scholar]
- Gómez, M.D.; Valera, M.; Cervantes, I.; Vinuesa, M.; Peña, F.; Molina, A. Development of a linear type trait system for Spanish Purebred horses (preliminary analysis). In Proceedings of the 57th Annual Meeting of the European Association for Animal Production, Antalya, Turkey, 17–20 September 2006. [Google Scholar]
- Folla, F.; Sartori, C.; Guzzo, N.; Pigozzi, G.; Mantovani, R. Genetics of linear type traits scored on young foals belonging to the Italian Heavy Draught Horse breed. Livest. Sci. 2019, 219, 91–96. [Google Scholar] [CrossRef]
- Folla, F.; Mantovani, R. Source of variation of linear type traits evaluated on Italian Heavy Draught Horse breed. Agric. Conspec. Sci. 2013, 78, 177–181. [Google Scholar]
- Ducro, B.J.; Koenen, E.P.C.; van Tartwijk, J.M.F.M.; Bovenhuis, H. Genetic relations of movement and free-jumping traits with dressage and show-jumping performance in competition of Dutch Warmblood horses. Livest. Sci. 2007, 107, 227–234. [Google Scholar] [CrossRef]
- Vostry, L.; Vostrà-Vydrovà, H.; Hofmanovà, B.; Veselà, Z.; Schmidovà, J.; Majzlik, I. Genetic Parameters for Linear Type Traits in Three Czech Draught Horse Breeds. Agric. Conspec. Sci. 2017, 82, 111–115. [Google Scholar]
- Simčič, M.; Mesarič, M.; Potočnik, K. Analysis of conformation traits of the Posavje horse in Slovenia. Slov. Vet. Res. 2012, 49, 141–148. [Google Scholar]
- Molina, A.; Valera, M.; dos Santos, R.; Rodero, A. Genetic parameters of morphofunctional traits in Andalusian horse. Livest. Prod. Sci. 1999, 60, 295–303. [Google Scholar] [CrossRef]
- Kashiwamura, F.; Avgaandorj, A.; Furumura, K. Relationships among body size, conformation, and racing performance in banei draft racehorses. J. Equine Sci. 2001, 12, 1–7. [Google Scholar] [CrossRef]
- Sabbioni, A.; Beretti, V.; Zanon, A.; Pagani, G.P.; Filippini, S.; Superchi, P.; Catalano, A.L. Morphological evolution of Bardigiano horse. Ital. J. Anim. Sci. 2005, 4, 412–414. [Google Scholar] [CrossRef]
- Ablondi, M.; Summer, A.; Vasini, M.; Simoni, M.; Sabbioni, A. Genetic parameters estimation in an Italian horse native breed to support the conversion from agricultural uses to riding purposes. J. Anim. Breed. Genet. 2020, 137, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Rustin, M.; Janssens, S.; Buys, N.; Gengler, N. Multi-trait animal model estimation of genetic parameters for linear type and gait traits in the Belgian warmblood horse. J. Anim. Breed. Genet. 2009, 126, 378–386. [Google Scholar] [CrossRef]
- De Oliveira, F.; Perez, C.; Vieira, R.; Fonseca, F.; Gabriela, M.; Diniz, C.; Guimarães, R.; Chicaroni, E.; Bento, J.; Ferraz, S.; et al. Genetic analysis of morphological and functional traits in Campolina horses using Bayesian multi-trait model. Livest. Sci. 2018, 216, 119–129. [Google Scholar] [CrossRef]
- Novotná, A.; Svitáková, A.; Veselá, Z.; Vostrý, L. Estimation of genetic parameters for linear type traits in the population of sport horses in the Czech Republic. Livest. Sci. 2017, 202, 1–6. [Google Scholar] [CrossRef]
- Vostrý, L.; Capková, Z.; Andrejsová, L.; Mach, K.; Majzlík, I. Linear type trait analysis in the Coldblood breeds: Czech-Moravian Belgian horse and Silesian Noriker. Slovak J. Anim. Sci. 2009, 42, 99–106. [Google Scholar]
- Gmel, A.I.; Druml, T.; Portele, K.; von Niederha, R.; Neuditschko, M. Repeatability, reproducibility and consistency of horse shape data and its association with linearly described conformation traits in Franches-Montagnes stallions. PLoS ONE 2018, 13, e0202931. [Google Scholar] [CrossRef]
- Schröder, W.; Stock, K.F.; Distl, O. Genetic evaluation of Hanoverian warmblood horses for conformation traits considering the proportion of genes of foreign breeds. Arch. Anim. Breed. 2010, 53, 377–387. [Google Scholar] [CrossRef]
- Kristjansson, T.; Bjornsdottir, S.; Albertsdóttir, E.; Sigurdsson, A.; Pourcelot, P.; Crevier-denoix, N.; Arnason, T. Association of conformation and riding ability in Icelandic horses. Livest. Sci. 2016, 189, 91–101. [Google Scholar] [CrossRef]
- Bakhtiari, J.; Heshmat, G. Estimation of genetic parameters of conformation traits in Iranian Thoroughbred horses. Livest. Sci. 2009, 123, 116–120. [Google Scholar] [CrossRef]
- Zechner, P.; Zohman, F.; Sölkner, J.; Bodo, I.; Habe, F.; Marti, E.; Brem, G. Morphological description of the Lipizzan horse population. Livest. Prod. Sci. 2001. [Google Scholar] [CrossRef]
- Druml, T.; Dobretsberger, M.; Brem, G. The use of novel phenotyping methods for validation of equine conformation scoring results. Animal 2015, 928–937. [Google Scholar] [CrossRef]
- Vicente, A.A.; Carolino, N.; Ralão-Duarte, J.; Gama, L.T. Selection for morphology, gaits and functional traits in Lusitano horses: I. Genetic parameter estimates. Livest. Sci. 2014, 164, 1–12. [Google Scholar] [CrossRef]
- Sole, M.; Cervantes, I.; Gutierrez, J.P.; Gomez, M.D.; Valera, M. Estimation of genetic parameters for morphological and functional traits in a Menorca horse population. Span. J. Agric. Res. 2014, 12, 125–132. [Google Scholar] [CrossRef]
- Bramante, G.; Maria, A.; Ciani, E.; Pieragostini, E. Multi-trait animal model estimation of genetic parameters for morphometric measurements in the Murgese horse breed. Livest. Sci. 2016, 191, 139–142. [Google Scholar] [CrossRef]
- Jakubec, V.; Rejfkovà, M.; Volenec, J.; Majzlìk, I.; Vostry, L. Analysis of linear description of type traits in the varieties and studs of the Old Kladrub horse. Czech J. Anim. Sci. 2007, 52, 299–307. [Google Scholar] [CrossRef]
- Novotná, A.; Svitáková, A.; Schmidová, J.; Přibyl, J. Variance components, heritability estimates, and breeding values for performance test traits in Old Kladruber horses. Czech J. Anim. Sci. 2016, 2016, 369–376. [Google Scholar] [CrossRef]
- Miserani, M.G.; McManus, C.; Santos, S.A.; Silva, J.A.; Mariante, A.S.; Abreu, U.G.P.; Mazza, M.C.; Sereno, J.R.B. Heritability estimates for biometric measures of the pantaneiro horse. Archivos De Zootecnia 2002, 51, 107–112. [Google Scholar]
- Sánchez, M.J.; Gómez, M.D.; Molina, A.; Valera, M. Genetic analyses for linear conformation traits in Pura Raza Español horses. Livest. Sci. 2013, 157, 57–64. [Google Scholar] [CrossRef]
- Sánchez-Guerrero, M.J.; Cervantes, I.; Molina, A.; Gutiérrez, J.P.; Valera, M. Designing an early selection morphological linear traits index for dressage in the Pura Raza Español horse. Animal 2017, 11, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Giontella, A.; Sarti, F.M.; Cardinali, I.; Cherchi, R. Genetic variability and population structure in Sardinian Anglo-Arab horse. Animals 2020, 10, 1018. [Google Scholar] [CrossRef] [PubMed]
- Van Bergen, H.M.; van Arendonk, J.A. Genetic parameters for linear type traits in Shetland Ponies. Livest. Prod. Sci. 1993, 36, 273–284. [Google Scholar] [CrossRef]
- Cervantes, I.; Baumung, R.; Molina, A.; Druml, T.; Gutiérrez, J.P.; Sölkner, J.; Valera, M. Size and shape analysis of morphofunctional traits in the Spanish Arab horse. Livest. Sci. 2009, 125, 43–49. [Google Scholar] [CrossRef]
- Gómez, M.D.; Azor, P.J.; Alonso, M.E.; Jordana, J.; Valera, M. Morphological and genetic characterization of Spanish heavy horse breeds: Implications for their conservation. Livest. Sci. 2012, 144, 57–66. [Google Scholar] [CrossRef]
- Viklund, Å.; Thorén Hellsten, E.; Näsholm, A.; Strandberg, E.; Philipsson, J. Genetic parameters for traits evaluated at field tests of 3- and 4-year-old Swedish Warmblood horses. Animal 2008, 2, 1832–1841. [Google Scholar] [CrossRef]
- Jönsson, L.; Näsholm, A.; Roepstorff, L.; Egenvall, A.; Dalin, G.; Philipsson, J. Conformation traits and their genetic and phenotypic associations with health status in young Swedish warmblood riding horses. Livest. Sci. 2014, 163, 12–25. [Google Scholar] [CrossRef]
- Kaproń, M.; Czerniak, E.; Łukaszewicz, M.; Danielewicz, A. Genetic parameters of body conformation and performance traits of Wielkopolski horses registered in the successive volumes of the Herdbook. Arch. Anim. Breed. 2013, 56, 127–136. [Google Scholar] [CrossRef]
- Mantovani, R.; Pigozzi, G.; Bittante, G. The Italian Heavy Draught Horse breed: Origin, breeding program, efficiency of the selection scheme and inbreeding. Publ. Assoc. Anim. Prod. 2005, 116, 155. [Google Scholar]
- Mantovani, R.; Guzzo, N.; Sartori, C.; Bailoni, L. In vivo performance of Italian Heavy Draft Horse weanlings fed two protein levels and slaughtered at two ages 1. J. Anim. Sci. 2014, 92, 4998–5008. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, R.; Sartori, C.; Pigozzi, G. Retrospective and statistical analysis of breeding management on the Italian Heavy Draught Horse breed. Anim. Int. J. Anim. Biosci. 2013, 1053. [Google Scholar] [CrossRef] [PubMed]
- Martin-rosset, W. Equine Nutrition: INRA Nutrient Requirements, Recommended Allowances and Feed Tables; Wageningen Academic Publishers: Wageningen, The Netherlands, 2015. [Google Scholar]
- Mantovani, R.; Sartori, C.; Pigozzi, G. Genetics of Temperament and Productive Traits in the Italian Heavy Draught Horse breed. In Proceedings of the 9th World Congress on Genetics Applied to Livestock Production, Leipzig, Germany, 1–6 August 2010; pp. 1–6. [Google Scholar]
- Johnson, D.L.; Thompson, R. Restricted maximum likelihood estimation of variance components for univariate animal models using sparse matrix techniques and average information. J. Dairy Sci. 1995, 78, 449–456. [Google Scholar] [CrossRef]
- Misztal, I.; Tsuruta, S.; Lourenco, D.; Aguilar, I.; Legarra, A.; Vitezica, Z. Manual for BLUPF90 Family of Programs; University of Georgia: Athens, GA, USA, 2018. [Google Scholar]
- Akaike, H. A New Look at the Statistical Model Identification. IEEE Trans. Automat. Contr. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Lynch, M.; Walsh, B. ; Genetics and Analysis of Quantitative Traits; Sinauer: Sunderland, MA, USA, 1998; Volume 1. [Google Scholar]
- Kohn, L.A.P.; Atchley, W.R. How Similar are Genetic Correlation Structures Data from Mice and Rats. Evolution 1988. [Google Scholar] [CrossRef]
- Koenen, E.P.C.; Aldridge, L.I.; Philipsson, J. An overview of breeding objectives for warmblood sport horses. Livest. Prod. Sci. 2004, 88, 77–84. [Google Scholar] [CrossRef]
- Pallottino, F.; Steri, R.; Menesatti, P.; Antonucci, F.; Costa, C.; Figorilli, S.; Catillo, G. Comparison between manual and stereovision body traits measurements of Lipizzan horses. Comput. Electron. Agric. 2015, 118, 408–413. [Google Scholar] [CrossRef]
- Druml, T.; Horna, M.; Grilz-Seger, G.; Dobretsberger, M.; Brem, G. Association of body shape with amount of Arabian genetic contribution in the Lipizzan horse. Arch. Anim. Breed. 2018, 61, 79–85. [Google Scholar] [CrossRef]
- Preisinger, R.; Wilkens, J.; Kalm, E. Estimation of genetic parameters and breeding values for conformation traits for foals and mares in the Trakehner populations and their practical implications. Livest. Prod. Sci. 1991, 29, 77–86. [Google Scholar] [CrossRef]
- Dall’Olio, S.; Wang, Y.; Sartori, C.; Fontanesi, L.; Mantovani, R. Association of myostatin (MSTN) gene polymorphisms with morphological traits in the italian heavy draft horse breed. Livest. Sci. 2014, 160, 29–36. [Google Scholar] [CrossRef]
- Albera, A.; Mantovani, R.; Bittante, G.; Groen, A.F.; Carnier, P. Genetic parameters for daily live-weight gain, live fleshiness and bone thinness in station-tested Piemontese young bulls. Anim. Sci. Glasg. 2001, 72, 449–456. [Google Scholar] [CrossRef]
- Mantovani, R.; Cassandro, M.; Contiero, B.; Albera, A.; Bittante, G. Genetic evaluation of type traits in hypertrophic Piemontese cows. J. Anim. Sci. 2010, 88, 3504–3512. [Google Scholar] [CrossRef] [PubMed]
- Hanset, R.; Farnir, F.; Leroy, P. Correlations among linear type traits in the Belgian Blue breed. Stočarstvo Časopis Za Unapređenje Stočarstva 1998, 52, 35–42. [Google Scholar]
- Gutiérrez, J.P.; Goyache, F. Estimation of genetic parameters of type traits in Asturiana de los Valles beef cattle breed. J. Anim. Breed. Genet. 2002, 119, 93–100. [Google Scholar] [CrossRef]
- Veselá, Z.; Pribyl, J.; Safus, P.; Vostry, L.; Seba, K.; Stolc, L. Breeding value for type traits in beef cattle in the Czech Republic. Czech J. Anim. Sci. (Czech Repub.) 2005, 50, 385–393. [Google Scholar] [CrossRef]

| Breed | Purpose | Evaluation 1 | Age | Ref. |
|---|---|---|---|---|
| Banei | Draught | BM | ≥2 years | [15] |
| Bardigiano horse | Draught/Sport | LS/SJ | ≥3 years | [16,17] |
| Belgian Warmblood horse | Sport | LS | 3–4 years | [18] |
| Campolina horse | Sport | BM | 22–52 months | [19] |
| Czech Warmblood | Sport | LS | 3–4 years | [20] |
| Czech-Moravian Belgian | Draught | LS/BM | ≥3 years | [12,21] |
| Dutch Warmblood horse | Sport | LS; SJ | 3–7 years | [4,11] |
| Franches-Montagnes horse | Sport | DI | 3 years | [22] |
| Haflinger | Draught/Sport | LS | 30 months | [5] |
| Hanoverian Warmblood horse | Sport | SJ/BM | ≥3 years | [23] |
| Icelandic horse | Sport/Leisure | BM/DI | 4–18 years | [24] |
| Iranian Thoroughbred horse | Sport | BM | 2–4 years | [25] |
| Italian Heavy Draught Horse | Draught/Meat | LS | 6 months | [9,10] |
| Lipizzan horse | Sport | BM; LS/DI | ≥4 years | [26,27] |
| Lusitano horse | Sport | SJ/BM | ≥3 years | [28] |
| Menorca horse | Sport | BM/DI | ≥3 years | [29] |
| Murgese horse | Draught | BM | 30 months | [30] |
| Noriker | Draught | BM; LS | ≥3 years | [6,12] |
| Old Kladruber | Sport | SJ/BM | ≥4 years | [31,32] |
| Pantaneiro horse | Draught/Meat | LS | [33] | |
| Posavje | Draught | LS/BM | 30–60 months | [13] |
| Pura Raza Español horse (Andalusian) | Sport | BM; LS | ≥3 years | [14,34,35] |
| Sardinian Anglo Arab horse | Sport | BM | [36] | |
| Shetland pony | Sport/Leisure | LS | 3 years | [37] |
| Silesian Noriker | Draught | LS | ≥3 years | [12] |
| Spanish Arab horse | Sport | DI | ≥3 years | [38] |
| Spanish heavy horse breeds 2 | Draught/Meat | BM | ≥4 years | [39] |
| Swedish Warmblood horse | Sport | LS | 3–4 years | [40,41] |
| Wielkopolski | Sport | BM | [42] |
| Trait | Mean ± SD | Skewness | Kurtosis | Minimum | Maximum |
|---|---|---|---|---|---|
| Head size (HS) | 3.04 ± 0.64 | 0.05 | −0.14 | Heavy | Light |
| Temperament/movement (Te/m) | 3.29 ± 0.54 | 0.17 | 0.45 | Lymphatic | Nevrile |
| Frame size (FS) | 3.20 ± 0.71 | 0.07 | −0.25 | Little | Large |
| Fleshiness (Fl) | 3.28 ± 0.54 | −0.03 | 0.09 | Poor | Excellent |
| Bone incidence (BI) | 2.88 ± 0.39 | −0.76 | 2.53 | Fine-boned | Heavy-boned |
| Thorax depth (TD) | 3.55 ± 0.53 | −0.19 | −0.38 | Little | Large |
| Fore diameter (FD) | 2.93 ± 0.65 | 0.17 | −0.25 | Narrow | Wide |
| Rear diameter (RD) | 3.37 ± 0.56 | 0.01 | −0.28 | Narrow | Wide |
| Upper line length (UL) | 3.28 ± 0.45 | 0.65 | −0.37 | Short | Long |
| Upper line direction (UD) | 2.85 ± 0.36 | −1.80 | 1.97 | Kyphotic | Curved |
| Legs side view (LS) | 2.58 ± 0.47 | −0.28 | −1.06 | Sickle | Straight |
| Fore feet (FF) | 3.22 ± 0.51 | 0.32 | 0.52 | Diverging | Converging |
| Rear feet (RF) | 3.01 ± 0.42 | 0.03 | 2.88 | Diverging | Converging |
| Hind legs back view (HL) | 2.90 ± 0.33 | −1.73 | 4.40 | Diverging | Converging |
| Overall score (OS) | 2.05 ± 0.79 | 0.28 | −0.51 | Fair | Excellent |
| Trait | σa2 (SE) | σe2 (SE) | σp2 (SE) | h2 (SE) |
|---|---|---|---|---|
| Head size (HS) | 10.09 (1.15) | 22.20 (0.92) | 32.29 (0.72) | 0.31 (0.032) |
| Temperament/movement (Te/m) | 4.75 (0.71) | 18.01 (0.64) | 22.77 (0.49) | 0.21 (0.029) |
| Frame size (FS) | 13.98 (1.35) | 20.92 (1.00) | 34.9 (0.81) | 0.40 (0.033) |
| Fleshiness (Fl) | 5.47 (0.71) | 16.21 (0.61) | 21.68 (0.47) | 0.25 (0.030) |
| Bone incidence (BI) | 1.68 (0.37) | 11.31 (0.37) | 12.99 (0.27 | 0.13 (0.028) |
| Thorax depth (TD) | 4.31 (0.64) | 16.00 (0.58) | 20.31 (0.43) | 0.21 (0.030) |
| Fore diameter (FD) | 8.95 (1.00) | 19.57 (0.81) | 28.52 (0.63) | 0.31 (0.031) |
| Rear diameter (RD) | 6.26 (0.79) | 17.16 (0.67) | 23.42 (0.51) | 0.27 (0.031) |
| Upper line length (UL) | 1.58 (0.40) | 14.79 (0.44) | 16.37 (0.34) | 0.10 (0.024) |
| Upper line direction (UD) | 0.28 (0.17) | 8.41 (0.22) | 8.69 (0.17) | 0.03 (0.019) |
| Legs side view (LS) | 1.94 (0.43) | 13.81 (0.44) | 15.75 (0.33) | 0.12 (0.026) |
| Fore feet (FF) | 1.85 (0.53) | 21.3 (0.61) | 23.15 (0.47) | 0.08 (0.023) |
| Rear feet (RF) | 0.40 (0.24) | 13.98 (0.35) | 14.37 (0.29) | 0.03 (0.017) |
| Hind legs back view (HL) | 0.54 (0.19) | 7.98 (0.22) | 8.53 (0.17) | 0.06 (0.022) |
| Overall score (OS) | 13.47 (1.47) | 30.22 (1.20) | 43.69 (0.96) | 0.31 (0.030) |
| Trait1 | HS | Te | FS | F | BI | TD | FD | RD | UL | UD | LS | FF | RF | HL | OS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HS | 0.67 | 0.13 | 0.12 | −0.64 | 0.14 | 0.18 | 0.1 | −0.01 | −0.1 | 0.12 | −0.15 | −0.23 | −0.21 | 0.45 | |
| Te | 0.3 | 0.24 | 0.09 | −0.74 | 0.15 | 0.13 | 0.09 | 0.18 | 0.05 | 0.25 | −0.19 | −0.15 | −0.12 | 0.47 | |
| FS | 0.08 | 0.13 | 0.45 | 0.22 | 0.71 | 0.52 | 0.73 | 0.41 | 0.38 | −0.03 | 0.13 | 0.03 | −0.1 | 0.85 | |
| Fl | 0.11 | 0.09 | 0.31 | 0.07 | 0.55 | 0.74 | 0.91 | 0.09 | −0.18 | −0.33 | 0.33 | 0.09 | 0.04 | 0.61 | |
| BI | −0.29 | −0.18 | 0.05 | −0.08 | 0.13 | 0.03 | 0.15 | 0.01 | 0.22 | −0.23 | 0.36 | 0.4 | 0.13 | −0.13 | |
| TD | 0.09 | 0.05 | 0.39 | 0.3 | 0.01 | 0.56 | 0.74 | 0.01 | −0.09 | −0.29 | 0.14 | 0.08 | 0.13 | 0.72 | |
| FD | 0.14 | 0.11 | 0.36 | 0.47 | −0.04 | 0.36 | 0.76 | 0.23 | 0.05 | −0.27 | 0.35 | 0.26 | 0.18 | 0.7 | |
| RD | 0 | 0.07 | 0.44 | 0.51 | −0.01 | 0.4 | 0.5 | 0.34 | −0.01 | −0.25 | 0.16 | 0.1 | −0.25 | 0.77 | |
| UL | −0.04 | 0.03 | 0.13 | −0.05 | 0.05 | −0.03 | 0.04 | 0.03 | 0.14 | 0.04 | 0.15 | −0.17 | 0.16 | 0.19 | |
| UD | 0.01 | 0.03 | 0.06 | −0.03 | 0.05 | −0.02 | −0.04 | −0.01 | −0.06 | 0.1 | −0.12 | −0.99 | −0.21 | 0.23 | |
| LS | 0.1 | 0.11 | −0.03 | −0.04 | −0.1 | −0.04 | −0.01 | −0.06 | −0.07 | 0.02 | −0.13 | −0.24 | 0.39 | 0.05 | |
| FF | −0.01 | −0.03 | 0.04 | 0.08 | 0.01 | 0.06 | 0.12 | 0.11 | 0.02 | −0.01 | −0.03 | 0.56 | −0.06 | 0.1 | |
| RF | −0.02 | −0.01 | 0.01 | 0.02 | 0.02 | 0.02 | 0.03 | 0.01 | 0.01 | 0.01 | −0.02 | 0.09 | 0.49 | −0.11 | |
| HL | 0.03 | 0.01 | −0.01 | 0.02 | 0.01 | 0.01 | 0.04 | −0.01 | −0.01 | −0.01 | 0.08 | 0.02 | 0.1 | −0.04 | |
| OS | 0.31 | 0.3 | 0.58 | 0.46 | −0.09 | 0.41 | 0.52 | 0.54 | 0.01 | 0.04 | 0.06 | 0.06 | 0.01 | 0.04 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Folla, F.; Sartori, C.; Mancin, E.; Pigozzi, G.; Mantovani, R. Genetic Parameters of Linear Type Traits Scored at 30 Months in Italian Heavy Draught Horse. Animals 2020, 10, 1099. https://doi.org/10.3390/ani10061099
Folla F, Sartori C, Mancin E, Pigozzi G, Mantovani R. Genetic Parameters of Linear Type Traits Scored at 30 Months in Italian Heavy Draught Horse. Animals. 2020; 10(6):1099. https://doi.org/10.3390/ani10061099
Chicago/Turabian StyleFolla, Fabio, Cristina Sartori, Enrico Mancin, Giuseppe Pigozzi, and Roberto Mantovani. 2020. "Genetic Parameters of Linear Type Traits Scored at 30 Months in Italian Heavy Draught Horse" Animals 10, no. 6: 1099. https://doi.org/10.3390/ani10061099
APA StyleFolla, F., Sartori, C., Mancin, E., Pigozzi, G., & Mantovani, R. (2020). Genetic Parameters of Linear Type Traits Scored at 30 Months in Italian Heavy Draught Horse. Animals, 10(6), 1099. https://doi.org/10.3390/ani10061099






