Effects of Exogenous Glucoamylase Enzymes Alone or in Combination with a Neutral Protease on Apparent Total Tract Digestibility and Feces D-Lactate in Crossbred Angus Bulls Fed a Ration Rich in Rolled Corn
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Housing, Experimental Design and Diets
2.2. Measurements and Sample Collection
2.2.1. Feed Consumption
2.2.2. Apparent Total Tract Digestibility and D-lactate Concentration in Feces
2.2.3. Chemical Analyses
2.3. Calculations and Statistical Analyses
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Owens, F.N.; Secrist, D.S.; Hill, W.J.; Gill, D.R. The Effect of Grain Source and Grain Processing Performance of Feedlot Cattle: A Review. J. Anim. Sci. 1997, 75, 868–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drouillard, J.S. Current situation and future trends for beef production in the United States of America—A review. Asian Australas. J. Anim. Sci. 2018, 31, 1007–1016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huntington, G.B. Starch utilization by ruminants: From basics to the bunk. J. Anim. Sci. 1997, 75, 852–867. [Google Scholar] [CrossRef] [PubMed]
- Owens, F.N.; Zinn, R.A. Corn grain for cattle: Influence of processing on site and extent of digestion. Southwest Nutr. Conf. Univ. Arizona. 2005, 78–85. Available online: http://animal.cals.arizona.edu/swnmc/2005/index.htm (accessed on 21 June 2020).
- Stewart, R.D.; Auffret, M.D.; Warr, A.; Wiser, A.H.; Press, M.O.; Langford, K.W.; Liachko, I.; Snelling, T.J.; Dewhurst, R.J.; Walker, A.W.; et al. Assembly of 913 microbial genomes from metagenomic sequencing of the cow rumen. Nat. Commun. 2018, 9, 870. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Zhang, G.; Xu, H.; Xin, H.; Zhang, Y. Metagenomic analyses of microbial and carbohydrate-active enzymes in the rumen of Holstein cows fed different forage-to-concentrate ratios. Front. Microbiol. 2019, 10, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Yu, S.; Kragh, K.M.; Li, W. Use of low pH active alpha-1,4/1,6-glycoside hydrolases as a feed additive for ruminants to enhance starch digestion. WO 2018/057420 A1. 2018. Available online: https://worldwide.espacenet.com/publicationDetails/biblio?DB=EPODOC&II=4&ND=4&adjacent=true&locale=en_EP&FT=D&date=20190829&CC=WO&NR=2018057420A1&KC=A (accessed on 21 June 2020).
- Shaw, A.; Wallace, L.; Estell DA Hommes RW, J.; Lee, S.; Oh, H.; Sadlowski, E.S. Thermostable Neutral Metalloproteases. U.S. Patent US 8574884 B2, 5 November 2013. [Google Scholar]
- McAllister, T.A.; Ribeiro, G. Microbial strategies in the ruminal digestion of starch. In Proceedings of the 50th Annual Meeting Brazilian Society Animal Science, Campinas, Brazil, 23 July 2013; pp. 140–151. [Google Scholar]
- DiLorenzo, N.; Smith, D.R.; Quinn, M.J.; May, M.L.; Ponce, C.H.; Steinberg, W.; Engstrom, M.A.; Galyean, M.L. Effects of grain processing and supplementation with exogenous amylase on nutrient digestibility in feedlot diets. Livest. Sci. 2011, 137, 178–184. [Google Scholar] [CrossRef]
- Vera, J.M.; Smith, H.; ZoBell, D.R.; Young, A.J.; Eun, J.S. Effects of an exogenous proteolytic enzyme on growth performance of beef steers and in vitro ruminal fermentation in continuous cultures. Prof. Anim. Sci. 2012, 28, 452–463. [Google Scholar] [CrossRef]
- Christopher, M.M.; Broussard, J.D.; Fallin, C.W.; Drost, N.J.; Peterson, M.E. Increased serum D-lactate associated with diabetic ketoacidosis. Metabolism 1995, 44, 287–290. [Google Scholar] [CrossRef]
- Del Estado, B.O. Royal Decree 53/2013 of February 1st on the protection of animals used for experimentation or other scientific purposes. Boletín Oficial del Estado 2013, 34, 11370–11421. [Google Scholar]
- NRC. Nutrient Requirements of Beef Cattle, 7th ed.; National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Grant, R.J.; Dann, H.M.; Woolpert, M.E. Time required for adaptation of behavior, feed intake and dietary digestibility in cattle. J. Anim. Sci. 2015, 93 (Suppl. 3), 312. [Google Scholar]
- Machado, M.; Detmann, G.E.; Mantovani, H.C.; Valadares Filho, S.C.; Bento CB, P.; Marcondes, M.I.; Assunçao, A.S. Evaluation of the length of adaptation period for changeover and crossover nutritional experiments with cattle fed tropical forage-based diets. Anim. Feed Sci. Technol. 2016, 222, 132–148. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 16th ed.; Official Methods of Analysis: Arlington, VA, USA, 1995. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. Available online: http://linkinghub.elsevier.com/retrieve/pii/S0022030291785512 (accessed on 21 June 2020). [CrossRef]
- Le Du, Y.L.P.; Penning, P.D. Animal based techniques for estimating herbage intake. In Herbage Intake Handbook; Leaver, J.D., Ed.; Grassland Res. Inst.: Hurley, UK, 1982; pp. 37–75. [Google Scholar]
- Larsen, T. Fluorometric determination of D-Lactate in biological fluids. Anal. Biochem. 2017, 539, 152–157. [Google Scholar]
- Zinn, R.A.; Adam, C.F.; Tamayo, M.S. Interaction of feed intake level on comparative ruminal and total tract digestion of dry-rolled and steam-flaked corn. J. Anim. Sci. 1995, 73, 1239–1245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owens, C.E.; Zinn, R.A.; Hassen, A.; Owens, N. Mathematical linkage of total tract digestion of starch and neutral detergent fiber to their fecal concentrations and the effect of site of starch digestion on extent of digestion and energetic efficiency of cattle. Profess. Anim. Sci. 2016, 32, 531–549. [Google Scholar] [CrossRef] [Green Version]
- Rojo, R.; Mendoza, G.D.; González, S.; Landois, L.; Bárcena, R.; Crosby, M.M. Effects of exogenous amylases from Bacillus licheniformis and Aspergillus niger on ruminal starch digestion and lamb performance. Anim. Feed Sci. Technol. 2005, 123, 655–665. [Google Scholar] [CrossRef]
- Mota, N.; Mendoza, G.D.; Plata, F.X.; Martínez, J.A.; Lee, H.; Rojo, R.; Crosby, M.M. Effect of exogenous glucoamylase enzymes and reduction of grain level on lamb performance. J. Appl. Anim. Res. 2011, 39, 129–131. [Google Scholar] [CrossRef]
- Meale, S.J.; Beauchemin, K.A.; Hristov, A.N.; Chaves, A.V.; McAllister, T.A. Opportunities and challenges in using exogenous enzymes to improve ruminant production. J. Anim. Sci. 2014, 92, 427–442. [Google Scholar] [CrossRef] [Green Version]
- McAllister, T.A.; Cheng, K.J. Microbial strategies in the ruminal digestion of cereal grains. Anim. Feed Sci. Technol. 1996, 62, 29–36. [Google Scholar] [CrossRef]
- Grifé, L.; Arís, A.; Bach, A.; García-Fruitós, E. Trends in recombinant protein use in animal production. Microbial Cell Factories 2017, 16, 40. [Google Scholar]
- Amaro, F.X.; Arriola, K.G.; Kim, D.; Fernandes, T.; Agarussi, M.C.N.; Silva, V.P.; Cervantes, A.P.; Jiang, Y.; Ferraretto, L.F.; Vyas, D.; et al. The effects of adding exogenous amylases and proteases on ruminal in vitro dry matter and starch digestibility of dent corn grain. J. Dairy Sci. 2018, 101 (Suppl. 2), 356. [Google Scholar]
- Eun, J.S.; Beauchemin, K.A. Effects of proteolytic feed enzyme on intake, digestion, ruminal fermentation, and milk production. J. Dairy Sci. 2005, 88, 2140–2153. [Google Scholar] [CrossRef] [Green Version]
- Hristov, A.N.; McAllister, T.A.; Cheng, K.J. Intraruminal supplementation with increasing levels of exogenous polysaccharide-degrading enzymes: Effects on nutrient digestion in cattle fed a barley grain diet. J. Anim. Sci. 2000, 78, 477–487. [Google Scholar] [CrossRef] [Green Version]
| Item | |
|---|---|
| Ingredients, % | |
| Rolled corn | 70.0 |
| Corn DDG | 15.0 |
| Alfalfa hay chopped at 3–5 cm | 10.0 |
| Soybean meal 44% CP | 2.40 |
| Calcium carbonate | 1.00 |
| Bicalcium phosphate | 0.40 |
| Sodium bicarbonate | 0.40 |
| Urea | 0.30 |
| Salt | 0.30 |
| Vitamin-mineral premix 1 | 0.20 |
| Nutrients, % DM basis | |
| ME, Mcal/kg | 3.19 |
| CP, % | 16.7 |
| Ether extract, % | 6.2 |
| NDF. % | 21.6 |
| Starch, % | 48.0 |
| Ash | 5.1 |
| Calculated non-structural carbohydrate | 50.4 |
| Treatment 1 | ||||||
|---|---|---|---|---|---|---|
| CTR | TrGA | AfuGA | AfuGA + BamPro | SEM | p-Value 2 | |
| Intake, kg/d | ||||||
| DM | 7.8 | 7.9 | 8.0 | 7.6 | 0.12 | 0.19 |
| OM | 7.4 | 7.5 | 7.6 | 7.3 | 0.12 | 0.18 |
| Starch | 3.7 | 3.8 | 3.8 | 3.7 | 0.07 | 0.29 |
| CP | 1.3 | 1.3 | 1.3 | 1.2 | 0.03 | 0.11 |
| Ether extract | 0.49 | 0.49 | 0.49 | 0.46 | 0.009 | 0.10 |
| NDF | 1.7 | 1.7 | 1.7 | 1.7 | 0.03 | 0.33 |
| Fecal output, kg/d | ||||||
| DM | 2.5 | 2.3 | 2.0 | 2.1 | 0.16 | 0.15 |
| OM | 2.4 | 2.1 | 1,9 | 1.9 | 0.16 | 0.15 |
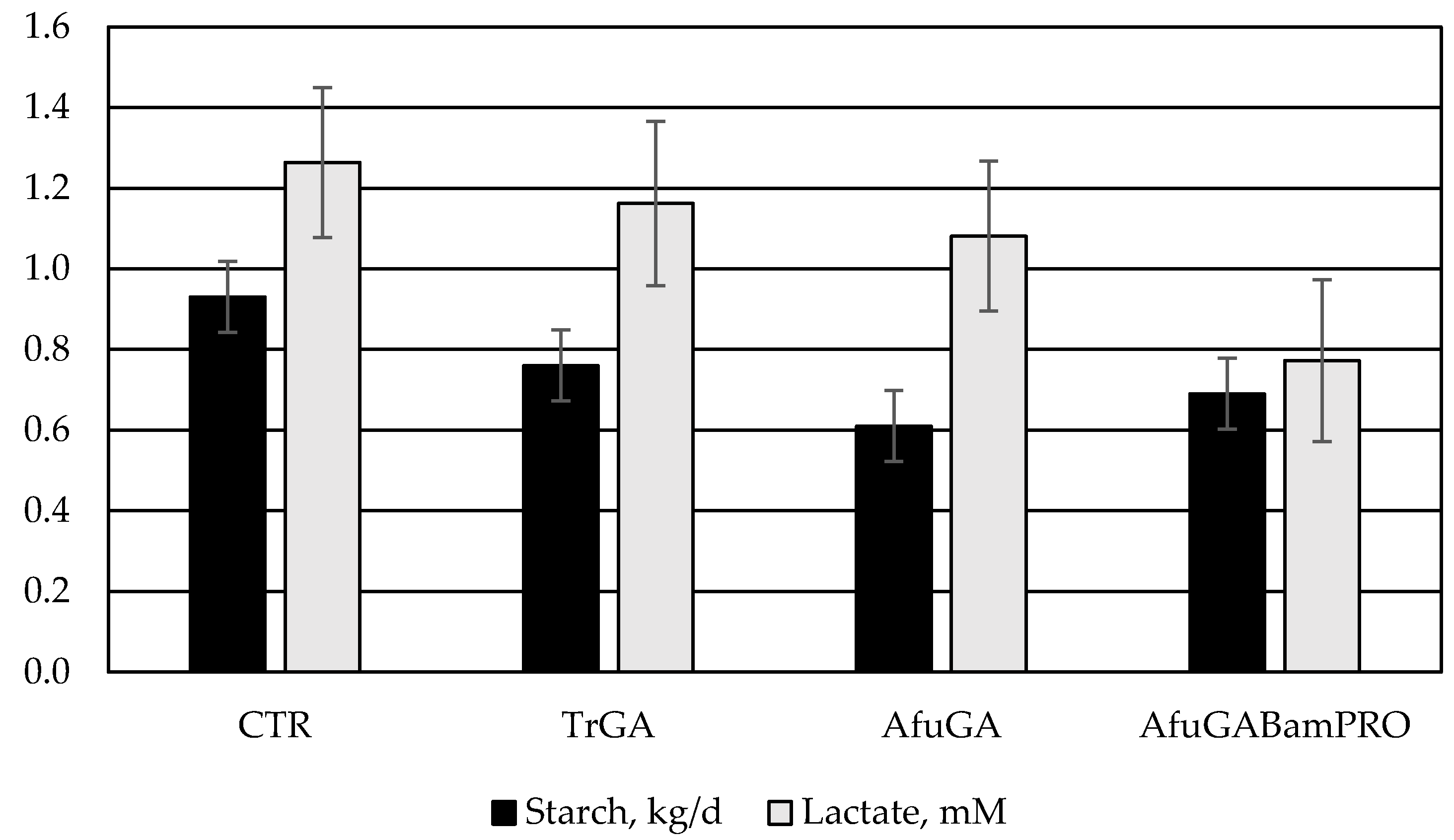
| Starch | 0.93 | 0.76 | 0.61 | 0.69 | 0.088 | 0.07 |
| CP | 0.40 | 0.37 | 0.35 | 0.35 | 0.027 | 0.45 |
| Ether extract | 0.18 | 0.18 | 0.17 | 0.17 | 0.013 | 0.86 |
| NDF | 0.79 | 0.76 | 0.72 | 0.71 | 0.050 | 0.61 |
| Apparent total tract digestibility, % | ||||||
| DM | 66.7 b | 71.1 a | 74.7 a | 72.8 a | 2.01 | 0.05 |
| OM | 66.8 b | 71.3 a | 74.9 a | 72.9 a | 2.03 | 0.05 |
| Starch | 74.7 b | 80.2 a | 84.1 a | 81.3 a | 2.25 | 0.04 |
| CP | 67.7 | 70.8 | 74.4 | 72.4 | 2.14 | 0.17 |
| Ether extract | 61.2 | 62.5 | 64.7 | 62.7 | 2.87 | 0.84 |
| NDF | 50.0 | 53.9 | 58.4 | 58.1 | 2.91 | 0.15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devant, M.; Yu, S.; Genís, S.; Larsen, T.; Wenting, L. Effects of Exogenous Glucoamylase Enzymes Alone or in Combination with a Neutral Protease on Apparent Total Tract Digestibility and Feces D-Lactate in Crossbred Angus Bulls Fed a Ration Rich in Rolled Corn. Animals 2020, 10, 1077. https://doi.org/10.3390/ani10061077
Devant M, Yu S, Genís S, Larsen T, Wenting L. Effects of Exogenous Glucoamylase Enzymes Alone or in Combination with a Neutral Protease on Apparent Total Tract Digestibility and Feces D-Lactate in Crossbred Angus Bulls Fed a Ration Rich in Rolled Corn. Animals. 2020; 10(6):1077. https://doi.org/10.3390/ani10061077
Chicago/Turabian StyleDevant, Maria, Shukun Yu, Sandra Genís, Torben Larsen, and Li Wenting. 2020. "Effects of Exogenous Glucoamylase Enzymes Alone or in Combination with a Neutral Protease on Apparent Total Tract Digestibility and Feces D-Lactate in Crossbred Angus Bulls Fed a Ration Rich in Rolled Corn" Animals 10, no. 6: 1077. https://doi.org/10.3390/ani10061077
APA StyleDevant, M., Yu, S., Genís, S., Larsen, T., & Wenting, L. (2020). Effects of Exogenous Glucoamylase Enzymes Alone or in Combination with a Neutral Protease on Apparent Total Tract Digestibility and Feces D-Lactate in Crossbred Angus Bulls Fed a Ration Rich in Rolled Corn. Animals, 10(6), 1077. https://doi.org/10.3390/ani10061077




