HPLC MS-MS Analysis Shows Measurement of Corticosterone in Egg Albumen Is Not a Valid Indicator of Chicken Welfare
Simple Summary
Abstract
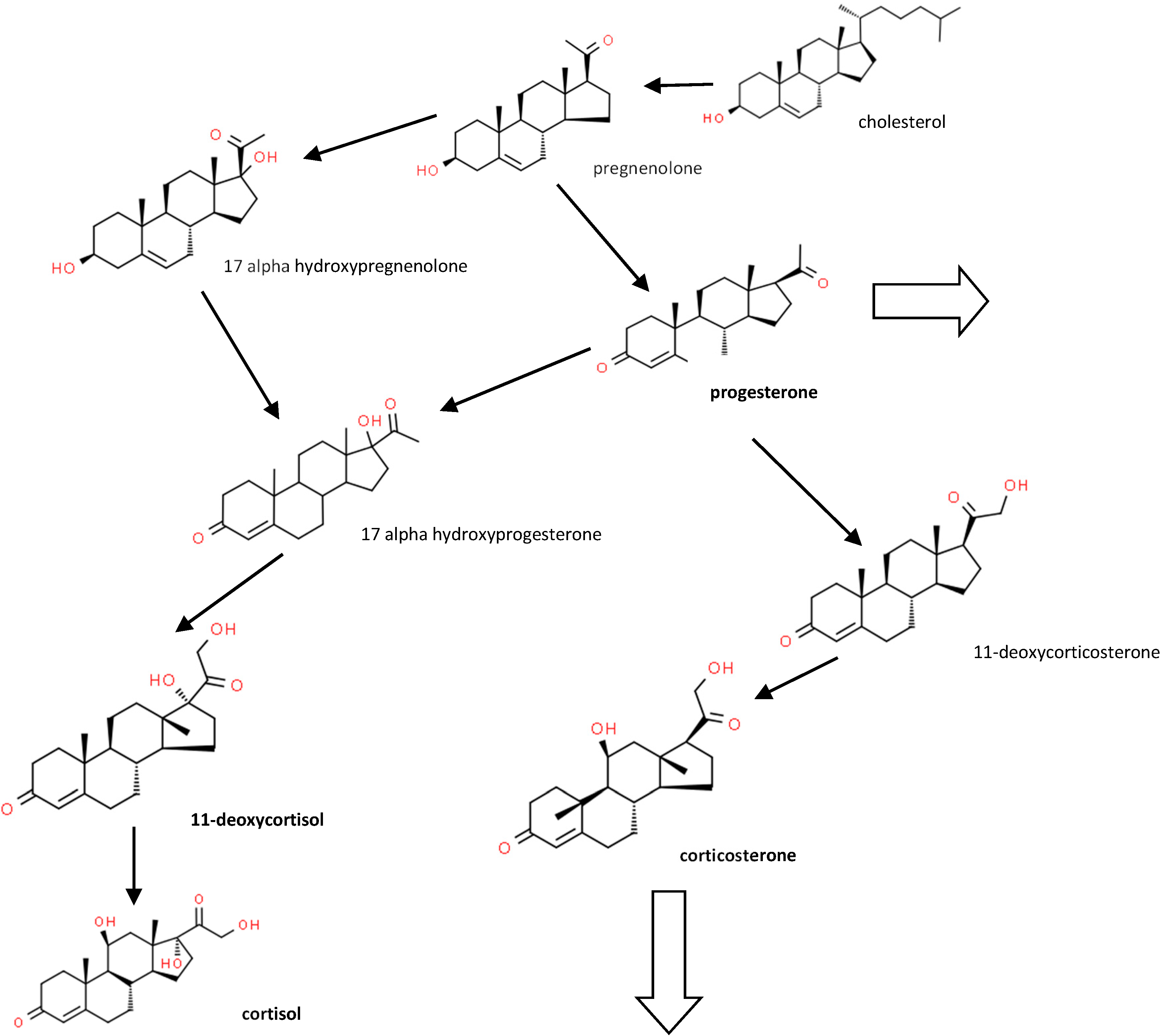
1. Introduction
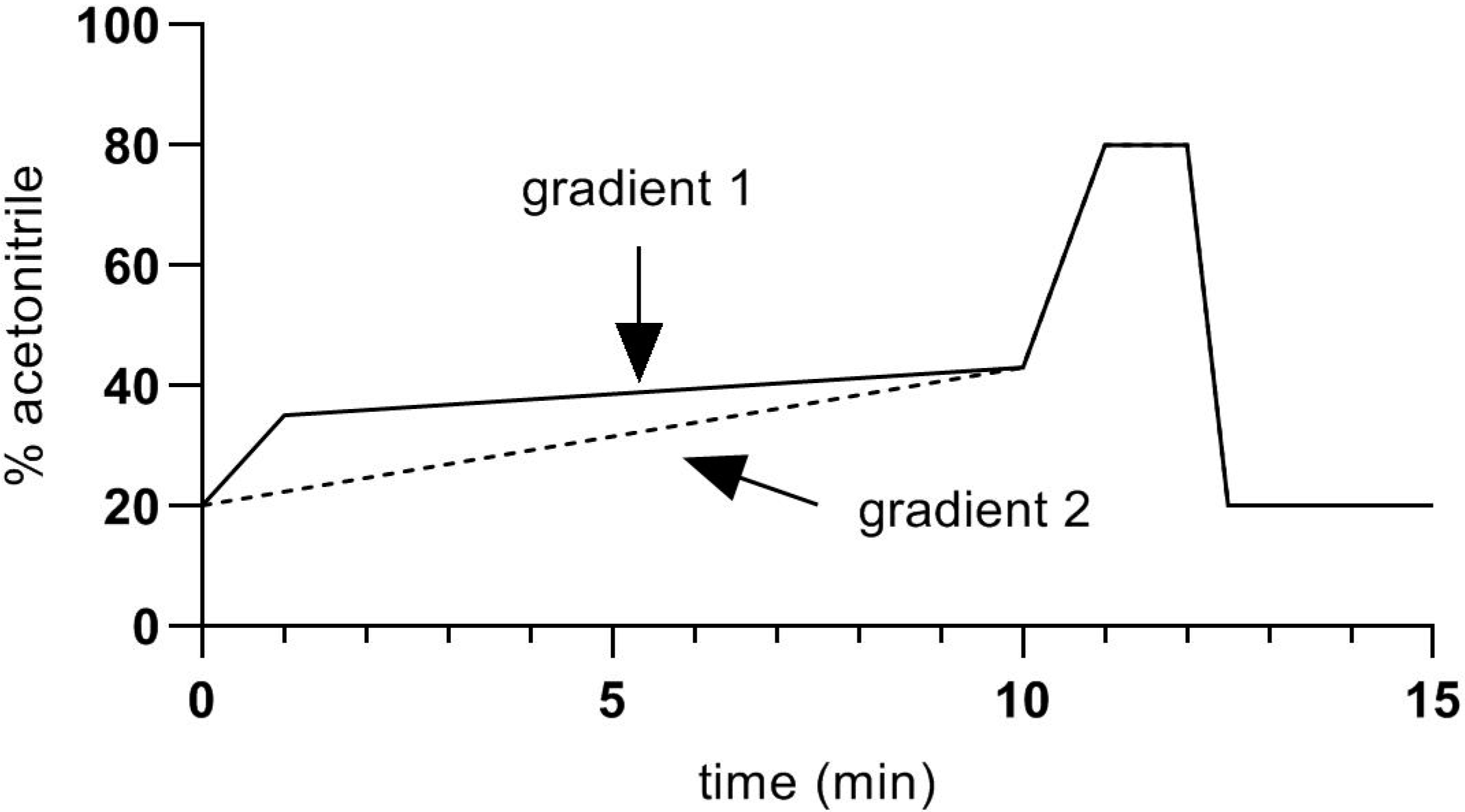
2. Materials and Methods
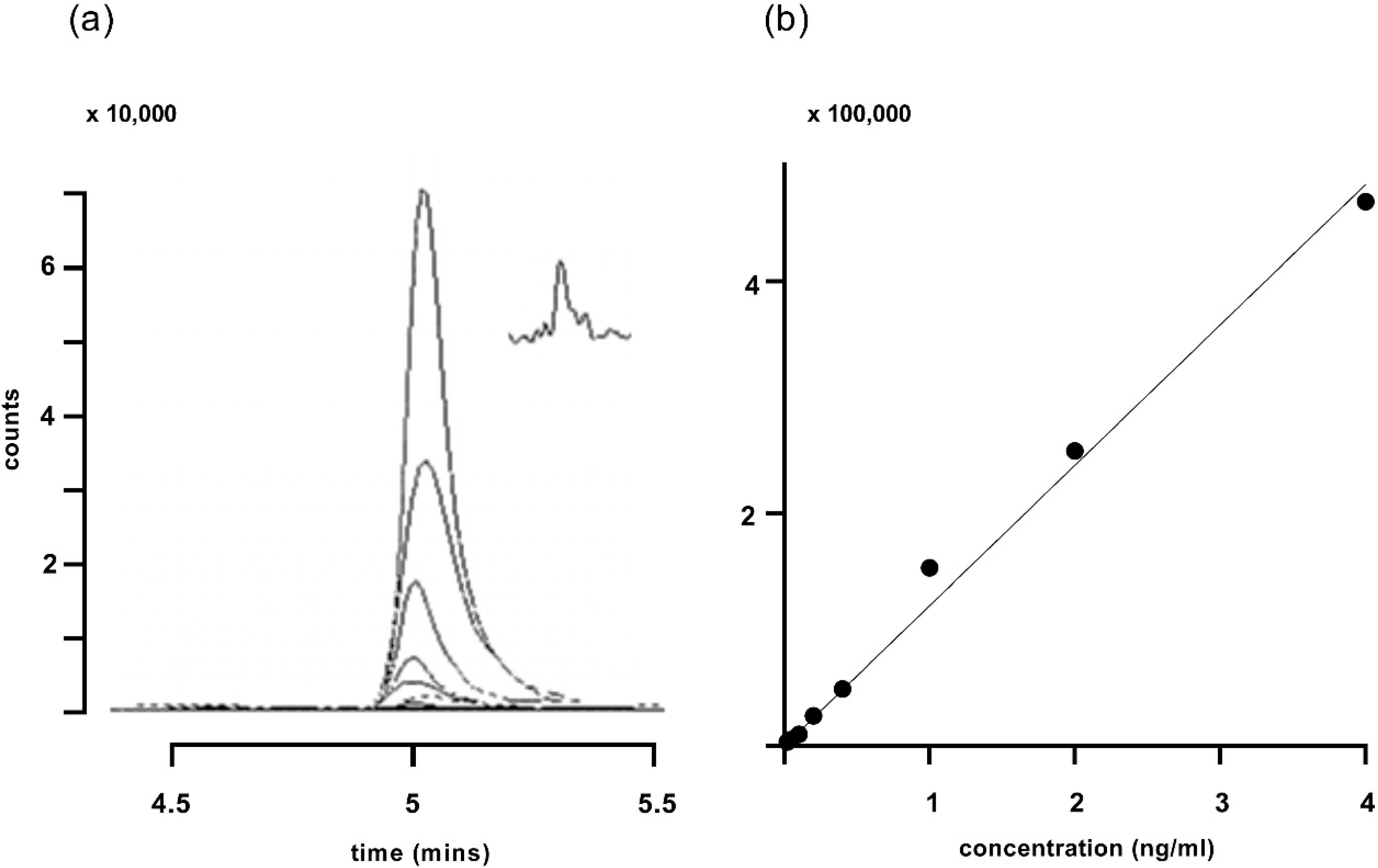
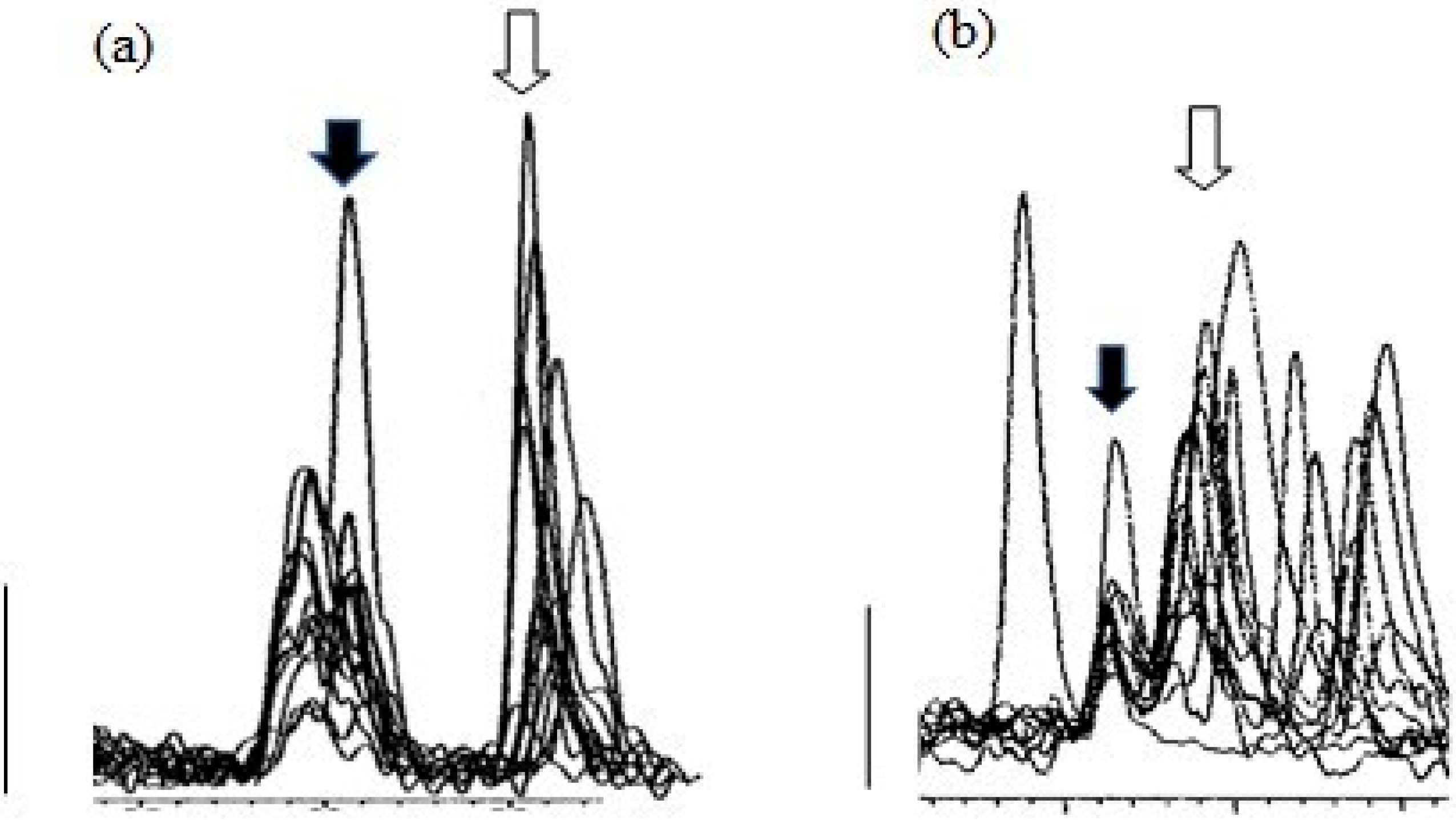
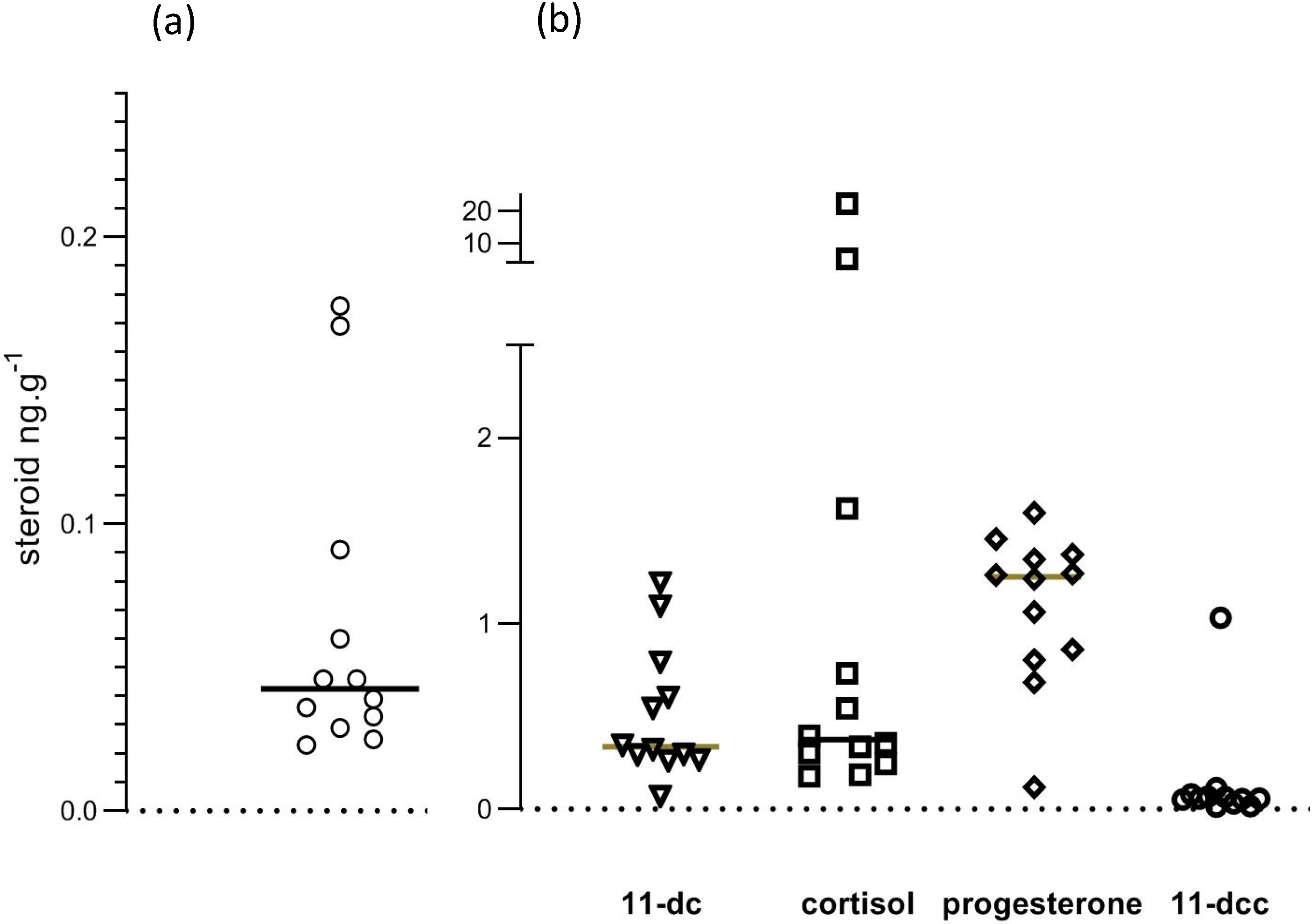
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Nicol, C.J.; Bouwsema, J.; Caplen, G.; Davies, A.C.; Hockenhull, J.; Lambton, S.L.; Lines, J.A.; Mullan, S.; Weeks, C.A. Farmed Bird Welfare Science Review. Available online: http://agriculture.vic.gov.au/__data/assets/pdf_file/0019/370126/Farmed-Bird-Welfare-Science-Review-Oct-2017.pdf (accessed on 14 February 2020).
- Hemsworth, P.H.; Mellor, D.J.; Cronin, G.M.; Tilbrook, A.J. Scientific assessment of animal welfare. N. Z. Vet. J. 2015, 63, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Spencer, R.L.; Deak, T. A users guide to HPA axis research. Physiol. Behav. 2017, 178, 43–65. [Google Scholar] [CrossRef] [PubMed]
- Carsia, R.V. Adrenals. In Sturkie’s Avian Physiology, 6th ed.; Scanes, C.G., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 577–611. [Google Scholar]
- Rushen, J.A. Problems associated with the interpretation of physiological data in the assessment of animal welfare. Appl. Anim. Behav. Sci. 1991, 28, 381–386. [Google Scholar] [CrossRef]
- Scanes, C.G. Biology of stress in poultry with emphasis on glucocorticoids and the heterophil to lymphocyte ratio. Poult. Sci. 2016, 95, 2208–2215. [Google Scholar] [CrossRef] [PubMed]
- Palme, R. Non-invasive measurement of glucocorticoids: Advances and problems. Physiol. Behav. 2019, 199, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Downing, J.A.; Bryden, W.L. Determination of corticosterone concentrations in egg albumen: A non-invasive indicator of stress in laying hens. Physiol. Behav. 2008, 95, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Downing, J. Non-Invasive Assessment of Stress in Commercial Housing Systems. A Report for the Australian Egg Corporation Limited. 2012. Available online: https://www.australianeggs.org.au/dmsdocument/529-non-invasive-assessment-of-stress-in-commercial-housing-systems (accessed on 14 February 2020).
- University of Sydney News. Cage or Free Range Don’t Put Too Much Stress on Housing Systems. Available online: https://www.sydney.edu.au/news/84.html?newsstoryid=8876 (accessed on 25 March 2020).
- Campbell, D.L.M.; Hinch, G.N.; Downing, J.A.; Lee, C. Early enrichment in free-range laying hens: Effects on ranging behavior, welfare and response to stressors. Animal 2017, 12, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.L.; Tauson, R.; Downing, J.A.; Janardhana, V.; Lowenthal, J.W.; Butler, K.L.; Cronin, G.M. The effects of a perch, dust bath, and nest box, either alone or in combination as used in furnished cages, on the welfare of laying hens. Poult. Sci. 2009, 88, 456–470. [Google Scholar] [CrossRef] [PubMed]
- Engel, J.M. The Effects of Floor Space Allowance and Nest Box Access on the Welfare of Caged Laying Hens (Gallus gallus domesticus). Ph.D. Thesis, University of Melbourne, Melbourne, Australia, 2016. [Google Scholar]
- Engel, J.M.; Widowski, T.M.; Tilbrook, A.J.; Butler, K.L.; Hemsworth, P.H. The effects of floor space and nest box access on the physiology and behavior of caged laying hens. Poult. Sci. 2019, 98, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Handelsman, D.J. Mass spectrometry, immunoassay and valid steroid measurements in reproductive medicine and science. Hum. Reprod. 2017, 32, 1147–1150. [Google Scholar] [CrossRef] [PubMed]
- Quillfeldt, P.; Poisbleau, M.; Parenteau, C.; Trouve, C.; Demongin, L.; Van Noordwijk, H.; Mostl, E. Measuring corticosterone in seabird egg yolk and the presence of high yolk gestagen concentrations. Gen. Comp. Endocr. 2011, 173, 11–14. [Google Scholar] [CrossRef] [PubMed]
- De Baere, S.; Rosendahl Larsen, T.; Devreese, M.; De Backer, P.; De Neve, L.; Fairhurst, G.; Lens, L.; Croubels, S. Use of LC-MS-MS as an alternative to currently available immunoassay methods to quantitate corticosterone in egg yolk and albumen. Anal. Bioanal. Chem. 2015, 407, 4351–4362. [Google Scholar] [CrossRef] [PubMed]
- Makin, H.L.J.; Honour, J.W.; Shackleton, C.H.L.; Griffiths, W.J. General methods for the extraction, purification, and measurement of steroids by chromatography and mass spectrometry. In Steroid Analysis; Makin, H.L.J., Gower, D.B., Eds.; Springer Science+Business Media: London, UK, 2010. [Google Scholar] [CrossRef]
- Karvaly, G.; Kovacs, K.; Meszaros, K.; Kocsis, I.; Patocs, A.; Vasarhelyi, B. The comprehensive characterization of adrenocortical steroidogenesis using two-dimensional ultra-performance liquid chromatography—electrospray ionization tandem mass spectrometry. J. Pharm. Biomed. 2018, 153, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Sheng, L.-P.; Wang, B.; Yang, Z.-L.; Liu, S.-Y. An optimized method for corticosterone analysis in mouse plasma by ultra-performance liquid chromatography-full-scan high-resolution accurate mass spectrometry. J. Chromatogr. Sci. 2015, 53, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.A. Animal Welfare: Limping Towards Eden; Universities Federation for Animal Welfare: Wallingford, UK, 2005; p. 30. [Google Scholar]
- Aerts, J. Quantification of a glucocorticoid profile in non-pooled samples is pivotal in stress research across vertebrates. Front. Endocrinol. 2018, 9, 635. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.L. Reproduction in the female. In Sturkie’s Avian Physiology, 6th ed.; Scanes, C.G., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 635–665. [Google Scholar]
- Fallahsharoudi, A.; De Kock, N.; Johnsson, M.; Ubhayasekera, S.J.K.A.; Bergquist, J.; Wright, D.; Jensen, P. Domestication effects on stress induced steroid secretion and adrenal gene expression in chickens. Sci. Rep. 2015, 5, 15345. [Google Scholar] [CrossRef] [PubMed]
- Sarabdjitsingh, R.A.; Joels, M.; De Kloet, E.R. Glucocorticoid pulsatility and rapid corticosteroid actions in the central stress response. Physiol. Behav. 2012, 106, 73–80. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, J.M.; Curtis, S.E.; Shanks, R.D.; Carmer, G.G. Multiple concurrent stressors in chick. 1. Effects on weight gain, feed intake and behavior. Poult. Sci. 1989, 68, 501–509. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, J.M.; Curtis, S.E.; Simon, J.; Izquierdo, O.A. Multiple concurrent stressors in chicks. 2. Effects on haematologic, body composition and pathologic traits. Poult. Sci. 1989, 68, 510–521. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, J.M.; Curtis, S.E. Multiple concurrent stressors in chicks. 3. Effects on plasma corticosterone and the heterophil/lymphocyte ratio. Poult. Sci. 1989, 68, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Nicol, C.J. The Behavioural Biology of Chickens; CABI: Wallingford, UK, 2015. [Google Scholar]
- Animal Health Australia. Proposed Australian Animal Welfare Standards and Guidelines. Poultry. Consultation Regulatory Impact Statement. 2017. Available online: http://www.animalwelfarestandards.net.au/files/2015/07/Public-Cons-Version-Poultry-RIS-Nov-2017.pdf (accessed on 14 February 2020).
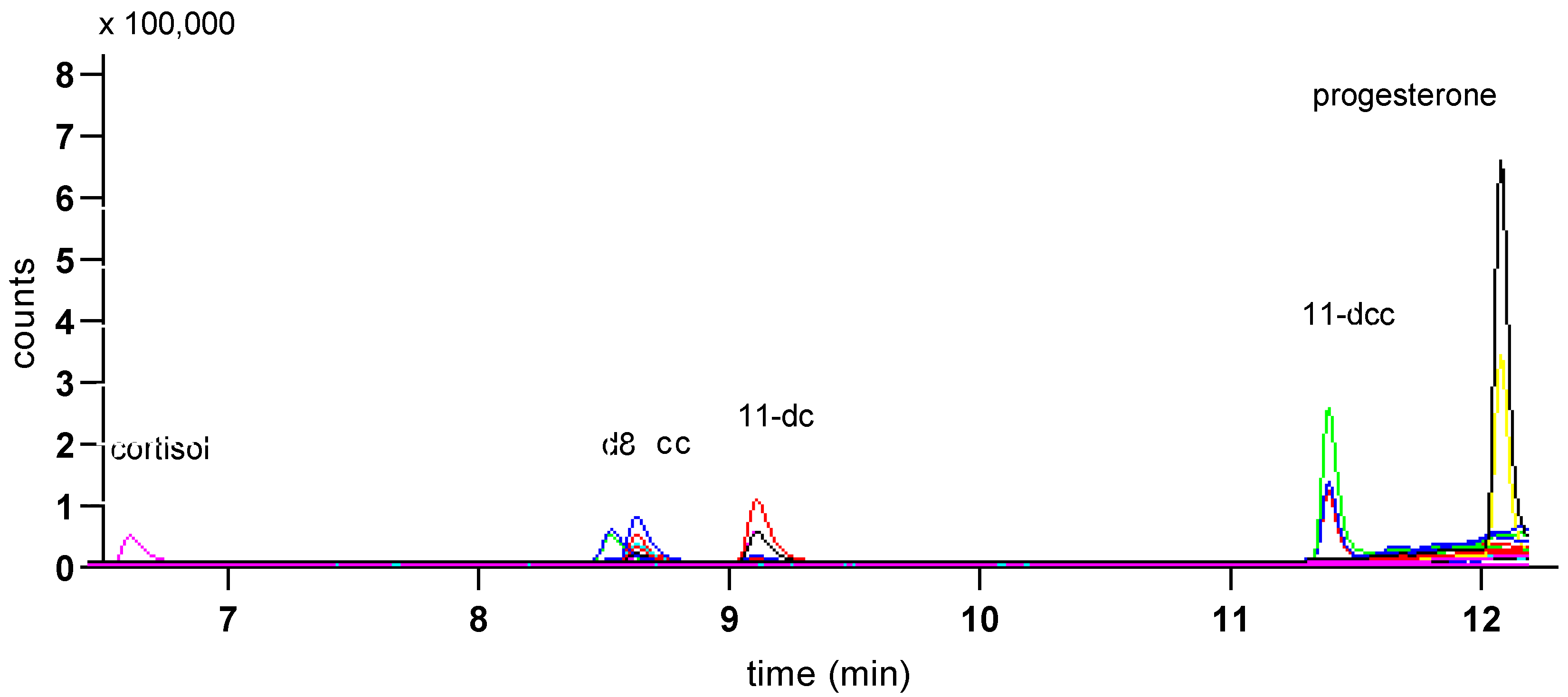
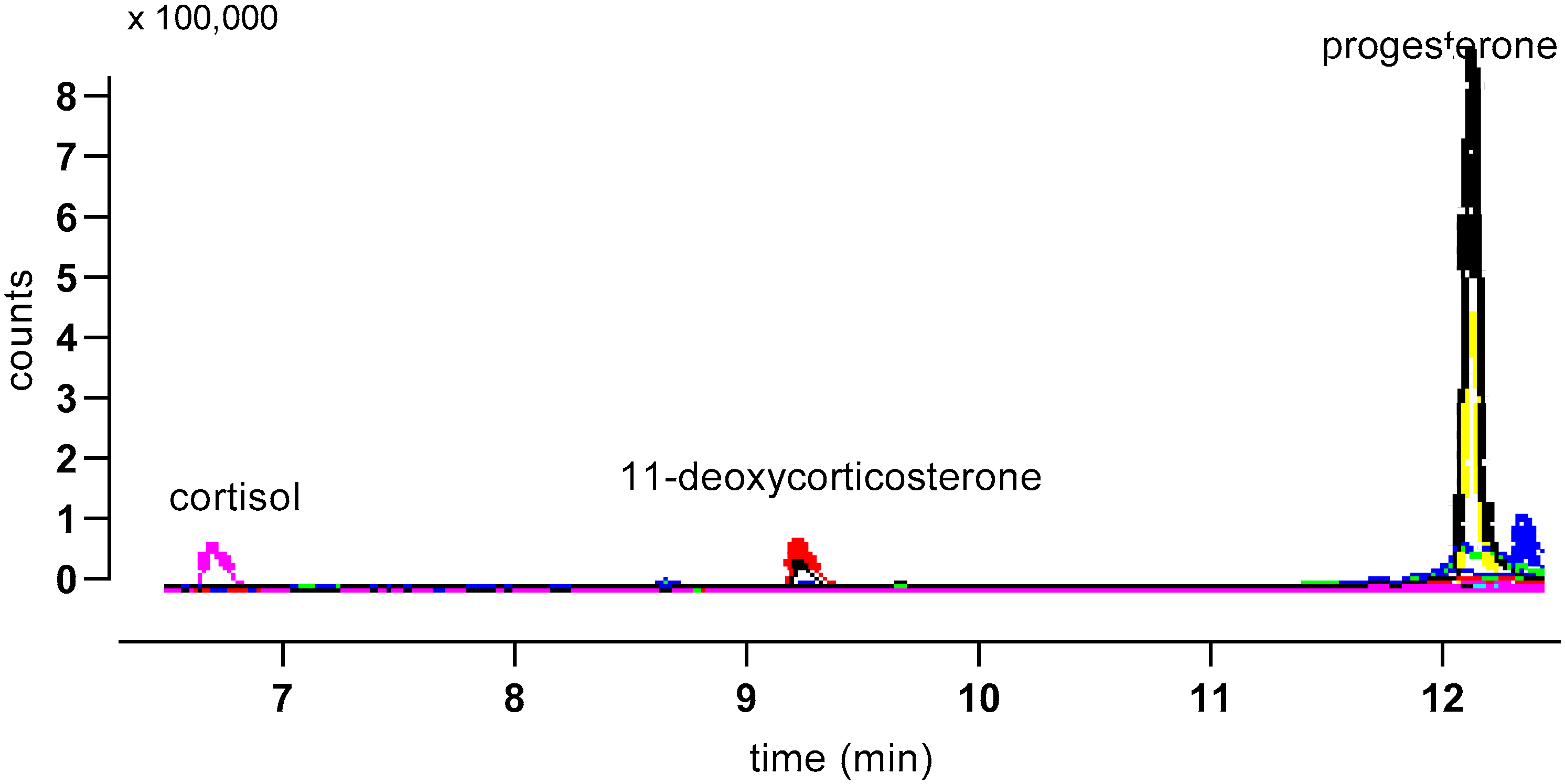
| Steroid | Precursor Ion | Quantifier Ion | Qualifier Ion | Retention Time (min) |
|---|---|---|---|---|
| corticosterone | 347.2 | 121.1 | 329.3 | 8.60 5.01 1 |
| d8 corticosterone | 355.2 | 125.1 | 337.3 | 8.49 |
| cortisol | 363.2 | 121.1 | 91.1 | 6.54 |
| 11-deoxy cc | 331.1 | 97.1 | 331.1 | 11.40 |
| 11-deoxycortisol | 347.1 | 97.1 | 109.1 | 9.08 |
| progesterone | 315.1 | 97.1 | 109.1 | 12.10 |
| Steroid 1 | Concentration (ng/g) |
|---|---|
| corticosterone | 0.043 (0.029–0.091) |
| 11-deoxycortisol | 0.335 (0.27–0.79) |
| cortisol | 0.374 (0.24–1.60) |
| 11-deoxycorticosterone | 0.057 (0.033–0.08) |
| progesterone | 1.251 (0.81–0.84) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caulfield, M.P.; Padula, M.P. HPLC MS-MS Analysis Shows Measurement of Corticosterone in Egg Albumen Is Not a Valid Indicator of Chicken Welfare. Animals 2020, 10, 821. https://doi.org/10.3390/ani10050821
Caulfield MP, Padula MP. HPLC MS-MS Analysis Shows Measurement of Corticosterone in Egg Albumen Is Not a Valid Indicator of Chicken Welfare. Animals. 2020; 10(5):821. https://doi.org/10.3390/ani10050821
Chicago/Turabian StyleCaulfield, Malcolm P., and Matthew P. Padula. 2020. "HPLC MS-MS Analysis Shows Measurement of Corticosterone in Egg Albumen Is Not a Valid Indicator of Chicken Welfare" Animals 10, no. 5: 821. https://doi.org/10.3390/ani10050821
APA StyleCaulfield, M. P., & Padula, M. P. (2020). HPLC MS-MS Analysis Shows Measurement of Corticosterone in Egg Albumen Is Not a Valid Indicator of Chicken Welfare. Animals, 10(5), 821. https://doi.org/10.3390/ani10050821






