Milk Metabolomics Reveals Potential Biomarkers for Early Prediction of Pregnancy in Buffaloes Having Undergone Artificial Insemination
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Farm and Animals
2.2. Synchronization Treatment and AI
2.3. Milk Sampling and Ultrasound Examination
2.4. Metabolomic Analysis
2.5. Metabolite Identification and Quantification
2.6. Statistical Analyses
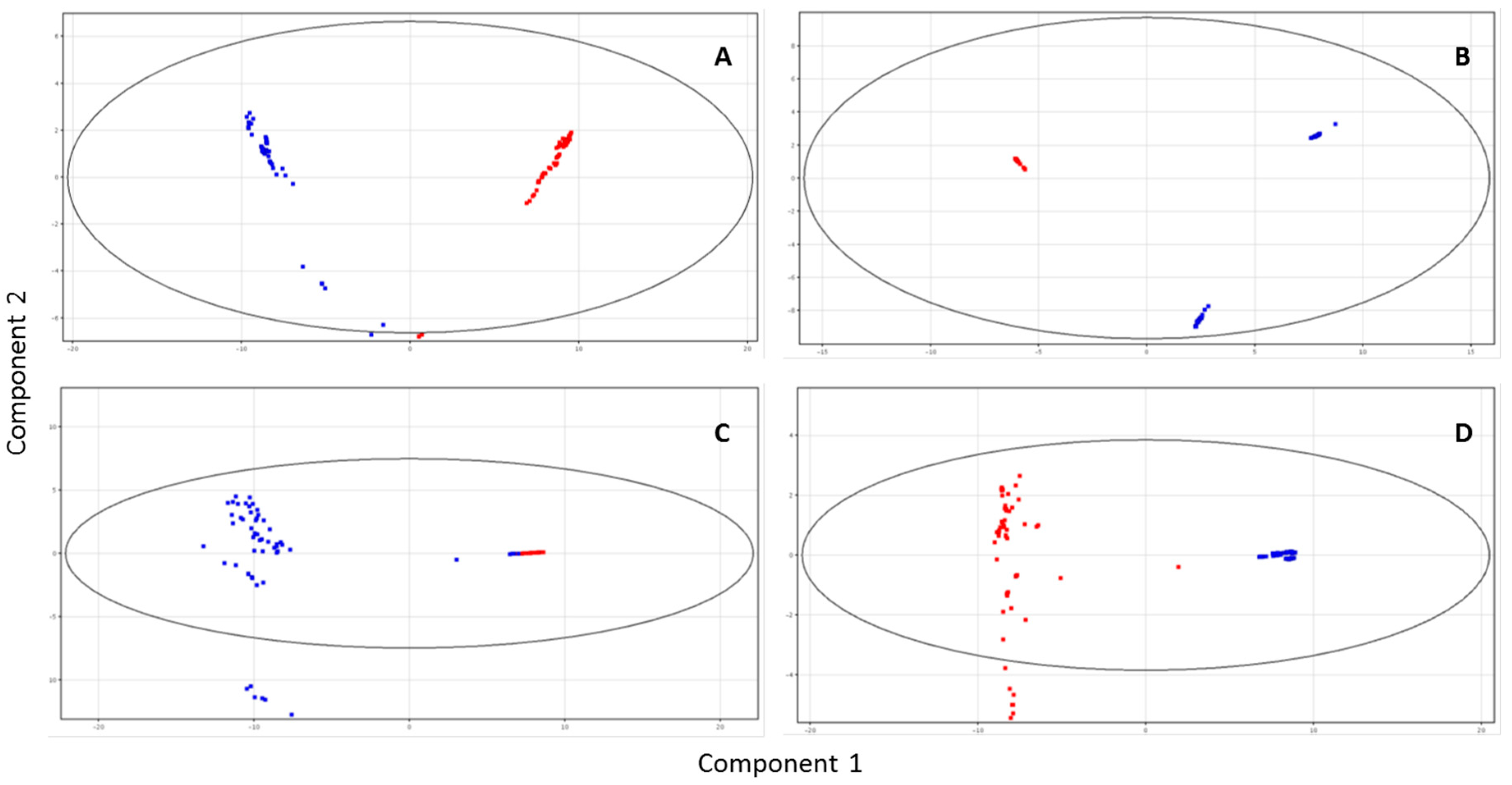
3. Results
3.1. Reproductive Activity
3.2. Milk Metabolic Profile
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Campanile, G.; Neglia, G.; Vecchio, D.; Zicarelli, L. Protein nutrition and nitrogen balance in buffalo cows. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2010, 5, 1–8. [Google Scholar] [CrossRef]
- Thibier, M. The zootechnical applications of biotechnology in animal reproduction: Current methods and perspectives. Reprod. Nutr. Dev. 2005, 45, 235–242. [Google Scholar] [CrossRef][Green Version]
- Baruselli, P.S.; Bernardes, O.; Braga, D.P.A.F.; De Araujo berber, C.; Tonhati, H. Calving distribution throughout the year in buffalo raised all over Brazil. In Proceedings of the 6th World Buffalo Congress, Maracaibo, Venezuela, 20–23 May 2001; pp. 234–239. [Google Scholar]
- Campanile, G.; Gasparrini, B.; Vecchio, D.; Neglia, G.; Senatore, E.M.; Bella, A.; Presicce, G.A.; Zicarelli, L. Pregnancy rates following AI with sexed semen in Mediterranean Italian buffalo heifers (Bubalus bubalis). Theriogenology 2011, 76, 500–506. [Google Scholar] [CrossRef]
- Neglia, G.; Gasparrini, B.; Salzano, A.; Vecchio, D.; De Carlo, E.; Cimmino, R.; Balestrieri, A.; D’Occhio, M.J.; Campanile, G. Relationship between the ovarian follicular response at the start of an Ovsynch-TAI program and pregnancy outcome in the Mediterranean river buffalo. Theriogenology 2016, 86, 2328–2333. [Google Scholar] [CrossRef] [PubMed]
- Neglia, G.; De Nicola, D.; Esposito, L.; Salzano, A.; D’Occhio, M.J.; Fatone, G. Reproductive management in buffalo by artificial insemination. Theriogenology 2020, 86, 2328–2333. [Google Scholar] [CrossRef] [PubMed]
- Campanile, G.; Baruselli, P.S.; Neglia, G.; Vecchio, D.; Gasparrini, B.; Gimenes, L.U.; Zicarelli, L.; D’Occhio, M.J. Ovarian function in the buffalo and implications for embryo development and assisted reproduction. Anim. Reprod. Sci. 2010, 121, 1e11. [Google Scholar] [CrossRef] [PubMed]
- Campanile, G.; Neglia, G. Embryonic mortality in buffalo cows. Ital. J. Anim. Sci. 2007, 6, 119e29. [Google Scholar] [CrossRef]
- Campanile, G.; Neglia, G.; D’Occhio, M.J. Embryonic and foetal mortality in River buffalo (Bubalus bubalis). Theriogenology 2016, 86, 207e13. [Google Scholar] [CrossRef]
- Oropeza, A.J.; Rojas, A.F.; Velazquez, M.A.; Muro, J.D.; Marquez, Y.C.; Vilanova, L.T. Efficiency of two-timed artificial insemination protocols in Murrah buffaloes managed under a semi-intensive system in the tropics. Trop. Anim. Health Prod. 2010, 42, 149e54. [Google Scholar] [CrossRef]
- Ohashi, O.M. Estrous detection in buffalo cow. Buffalo J. 1994, 2, 61–64. [Google Scholar]
- Rossi, P.; Vecchio, D.; Neglia, G.; Di Palo, R.; Gasparrini, B.; D’Occhio, M.J.; Campanile, G. Seasonal fluctuations in the response of Italian Mediterranean buffaloes to synchronization of ovulation and timed artificial insemination. Theriogenology 2014, 82, 132e7. [Google Scholar] [CrossRef] [PubMed]
- Neglia, G.; Capuano, M.; Balestrieri, A.; Cimmino, R.; Iannaccone, F.; Palumbo, F.; Presicce, G.A.; Campanile, G. Effect of consecutive re-synchronization protocols for ovulation on pregnancy rate in buffalo (Bubalus bubalis) heifers out of the breeding season. Theriogenology 2018, 113, 120e6. [Google Scholar] [CrossRef] [PubMed]
- Jaśkowski, J.M.; Gehrke, M.; Herudzińska, M.; Jaśkowski, B.M.; Brüssow, K.P. Resynchronisation as an element of improving cattle reproduction efficiency. J. Vet. Res. 2019, 63, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Johnston, D.; Malo Estepa, I.; Ebhardt, H.A.; Crowe, M.A.; Diskin, M.G. Differences in the bovine milk whey proteome between early pregnancy and the estrous cycle. Theriogenology 2018, 114, 301–307. [Google Scholar] [CrossRef]
- Humblot, P.; Camous, S.; Martal, J.; Charlery, J.; Jeanguyot, N.; Thibier, M.; Sasser, R.G. Diagnosis of pregnancy by radioimmunoassay of a pregnancy-specific proteinin the plasma of dairy cows. Theriogenology 1988, 30, 257–267. [Google Scholar] [CrossRef]
- Cordoba, M.C.; Sartori, R.; Fricke, P.M. Assessment of a commercially available early conception factor (ECF) test for determining pregnancy status of dairy cattle. J. Dairy Sci. 2001, 84, 1884–1889. [Google Scholar] [CrossRef]
- Shemesh, M.; Ayalon, N.; Lindner, H.R. Early effect of conceptus on plasma progesterone level in the cow. J. Reprod. Ferfil. 1968, 15, 161. [Google Scholar] [CrossRef]
- Perera, B.M.; Pathiraja, N.; Abeywardena, S.A.; Motha, M.X.; Abeygunawardena, H. Early pregnancy diagnosis in buffaloes from plasma progesterone concentration. Vet. Rec. 1980, 106, 104–106. [Google Scholar] [CrossRef]
- Boscos, C.M.; Samartzi, F.C.; Lymberopoulos, A.G.; Stefanakis, A.; Belibasaki, S. Assessment of Progesterone Concentration Using Enzymeimmunoassay, for Early Pregnancy Diagnosis in Sheep and Goats. Reprod. Domest. Anim. 2003, 38, 170–174. [Google Scholar] [CrossRef]
- Ambrose, D.J.; Radke, B.; Pitney, P.A.; Goonewardene, L.A. Evaluation of early conception factor lateral flow test to determine nonpregnancy in dairy cattle. Can. Vet. J. 2007, 48, 831–835. [Google Scholar]
- Wishart, D.S.; Lewis, M.J.; Morrissey, J.A.; Flegel, M.D.; Jeroncic, K.; Xiong, Y.; Cheng, D.; Eisner, R.; Gautam, B.; Tzur, D.; et al. The human cerebrospinal fluid metabolome. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 871, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Putri, S.P.; Yamamoto, S.; Tsugawa, H.; Fukusaki, E. Current metabolomics: Technological advances. J. Biosci. Bioeng. 2013, 116, 9–16. [Google Scholar] [CrossRef]
- Maher, A.D.; Hayes, B.; Cocks, B.; Marett, L.; Wales, W.J.; Rochfort, S.J. Latent biochemical relationships in the blood-milk metabolic axis of dairy cows revealed by statistical integration of 1H NMR spectroscopic data. J. Proteome Res. 2013, 12, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
- Schanzenbach, C.I.; Kirchner, B.; Ulbrich, S.E.; Pfaffl, M.W. MicroRNA of whole milk samples are not suitable for pregnancy detection in cattle. Gene 2019, 692, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Ciborowski, M.; Lipska, A.; Godzien, J.; Ferrarini, A.; Korsak, J.; Radziwon, P.; Tomasiak, M.; Barbas, C. Combination of LC-MS- and GC-MS-based metabolomics to study the effect of ozonated autohemotherapy on human blood. J. Proteome Res. 2012, 11, 6231–6241. [Google Scholar] [CrossRef]
- Goodacre, R.; Vaidyanathan, S.; Dunn, W.B.; Harrigan, G.G.; Kell, D.B. Metabolomics bynumbers: Acquiring and understanding global metabolite data. Trends Biotechnol. 2004, 22, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Puchades-Carrasco, L.; Pineda-Lucena, A. Metabolomics applications in precision medicine: An oncological perspective. Curr. Top. Med. Chem. 2017, 17, 2740–2751. [Google Scholar] [CrossRef]
- Nanda, T.; Das, M.; Tripathy, K.; Ravi, T.Y. Metabolomics: The future of systems biology. J. Comput. Sci. Syst. Biol. 2011, 1, 2–6. [Google Scholar] [CrossRef]
- D’Occhio, M.J.; Baruselli, P.S.; Campanile, G. Influence of nutrition, body condition, and metabolic status on reproduction in female beef cattle: A review. Theriogenology 2019, 125, 277–284. [Google Scholar] [CrossRef]
- Neglia, G.; Restucci, B.; Russo, M.; Vecchio, D.; Gasparrini, B.; Prandi, A.; Di Palo, R.; D’Occhio, M.J.; Campanile, G. Early development and function of the corpus luteum and relationship to pregnancy in the buffalo. Theriogenology 2015, 83, 959e67. [Google Scholar] [CrossRef]
- Campanile, G.; Neglia, G.; Vecchio, D.; Russo, M.; Zicarelli, L. Pregnancy in buffalo cows. In Pregnancy Protein Research; O’Leary, M., Arnett, J., Eds.; Nova Science Publishers Inc.: New York, NY, USA, 2009; pp. 31–91. [Google Scholar]
- Balestrieri, M.L.; Gasparrini, B.; Neglia, G.; Vecchio, D.; Strazzullo, M.; Giovane, A.; Servillo, L.; Zicarelli, L.; D’Occhio, M.J.; Campanile, G. Proteomic profiles of the embryonic chorioamnion and uterine caruncles in buffaloes (Bubalus bubalis) with normal and retarded embryonic development. Biol. Reprod. 2013, 88, 119. [Google Scholar] [CrossRef] [PubMed]
- Campanile, G.; Neglia, G.; Gasparrini, B.; Galiero, G.; Prandi, A.; Di Palo, R.; D’Occhio, M.J.; Zicarelli, L. Embryonic mortality in buffaloes synchronized and mated by AI during the seasonal decline in reproductive function. Theriogenology 2005, 63, 2334–2340. [Google Scholar] [CrossRef] [PubMed]
- Baruselli, P.S.; Mucciolo, R.G.; Visintin, J.A.; Viana, W.G.; Arruda, R.P.; Madureira, E.H.; Oliveira, C.A.; Molero-Filho, J.R. Ovarian follicular dynamics during the estrous cycle in buffalo (Bubalus bubalis). Theriogenology 1997, 47, 1531e47. [Google Scholar] [CrossRef]
- Vecchio, D.; Neglia, G.; Gasparrini, B.; Russo, M.; Pacelli, C.; Prandi, A.; D’Occhio, M.J.; Campanile, G. Corpus luteum development and function and relationship to pregnancy during the breeding season in the Mediterranean buffalo. Theriogenology 2012, 77, 1811e5. [Google Scholar] [CrossRef] [PubMed]
- Wiltbank, M.C.; Sartori, R.; Herlihy, M.M.; Vasconcelos, J.L.M.; Nascimento, A.B.; Souza, A.H.; Ayres, H.; Cunha, A.P.; Keskin, A.; Guenther, J.N.; et al. Managing the dominant follicle in lactating dairy cows. Theriogenology 2011, 76, 1568–1582. [Google Scholar] [CrossRef] [PubMed]
- Bello, N.M.; Steibel, J.P.; Pursley, J.R. Optimizing ovulation to first GnRH improved outcomes to each hormonal injection of Ovsynch in lactating dairy cows. J. Dairy Sci. 2006, 89, 3413–3424. [Google Scholar] [CrossRef]
- Wiltbank, M.C.; Pursley, J.R. The cow as an induced ovulator: Timed AI after synchronization of ovulation. Theriogenology 2014, 81, 170–185. [Google Scholar] [CrossRef]
- Sun, H.-Z.; Wang, D.-M.; Wang, B.; Wang, J.-K.; Liu, H.-Y.; Guan, L.L.; Liu, J.-X. Metabolomics of four biofluids from dairy cows: Potential biomarkers for milk production and quality. J. Proteome Res. 2015, 14, 1287–1298. [Google Scholar] [CrossRef]
- Salzano, A.; Manganiello, G.; Neglia, G.; Vinale, F.; De Nicola, D.; D’Occhio, M.J.; Campanile, G. A preliminary study on metabolome profiles of buffalo milk and corresponding mozzarella cheese: Safeguarding the authenticity and traceability of protected status buffalo dairy products. Molecules 2020, 25, 304. [Google Scholar] [CrossRef]
- Zhang, H.; Tong, J.; Zhang, Y.; Xiong, B.; Jiang, L. Metabolomics reveals potential biomarkers in the rumen fluid of dairy cows with different levels of milk production. Asian-Australas J. Anim. Sci. 2020, 33, 79–90. [Google Scholar] [CrossRef]
- Zoli, A.P.; Guilbault, L.A.; Delahaut, P.; Ortiz, W.B.; Beckers, J.F. Radioimmunoassay of a bovine pregnancy-associated glycoprotein in serum: Its application for pregnancy diagnosis. Biol. Reprod. 1992, 46, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.T.; Pereira, M.H.C.; Edwards, J.L.; Vasconcelos, J.L.M.; Pohler, K.G. Pregnancy diagnosis in cattle using pregnancy associated glycoprotein concentration in circulation at day 24 of gestation. Theriogenology 2018, 106, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Barbato, O.; Menchetti, L.; Sousa, N.M.; Malfatti, A.; Brecchia, G.; Canali, C.; Beckers, J.F.; Barile, V.L. Pregnancy-associated glycoproteins (PAGs) concentrations in water buffaloes (Bubalus bubalis) during gestation and the postpartum period. Theriogenology 2017, 97, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Vlaeminck, B.; Fievez, V.; Cabrita, A.R.J.; Fonseca, A.J.M.; Dewhurst, R.J. Factors affecting odd- and branched-chain fatty acids in milk: A review. Anim. Feed Sci. Technol. 2006, 131, 389–417. [Google Scholar] [CrossRef]
- Stefanov, I.; Baeten, V.; Abbas, O.; Vlaeminck, B.; De Baets, B.; Fievez, V. Evaluation of FT-NIR and ATR-FTIR spectroscopy techniques for determination of minor odd- and branched-chain saturated and trans unsaturated milk fatty acids. J. Agric. Food Chem. 2013, 61, 3403–3413. [Google Scholar] [CrossRef]
- Jenkins, B.; West, J.A.; Koulman, A. A review of odd-chain fatty acid metabolism and the role of pentadecanoic acid (C15:0) and heptadecanoic acid (C17:0) in health and disease. Molecules 2015, 20, 2425–2444. [Google Scholar] [CrossRef]
- Nicassio, L.; Fracasso, F.; Sirago, G.; Musicco, C.; Picca, A.; Marzetti, E.; Calvani, R.; Cantatore, P.; Gadaleta, M.N.; Pesce, V. Dietary supplementation with acetyl-l-carnitine counteracts agerelated alterations of mitochondrial biogenesis, dynamics and antioxidant defenses in brain of old rats. Exp. Gerontol. 2017, 98, 99–109. [Google Scholar] [CrossRef]
- Sergi, G.; Pizzato, S.; Piovesan, F.; Trevisan, C.; Veronese, N.; Manzato, E. Effects of acetyl-L-carnitine in diabetic neuropathy and other geriatric disorders. Aging Clin. Exp. Res. 2018, 30, 133–138. [Google Scholar] [CrossRef]
- Maldonado, C.; Vázquez, M.; Fagiolino, P. Potential therapeutic role of carnitine and acetylcarnitine in neurological disorders. Curr. Pharm. Des. 2020. Online ahead of print. [Google Scholar] [CrossRef]
- Cacciari, B.; Pastorin, G.; Bolcato, C.; Spalluto, G.; Bacilieri, M.; Moro, S. A2B adenosine receptor antagonists: Recent developments. Mini. Rev. Med. Chem. 2005, 5, 1053–1060. [Google Scholar] [CrossRef]
- Polosa, R.; Holgate, S.T. Adenosine receptors as promising therapeutic targets for drug development in chronic airway inflammation. Curr. Drug Targets 2006, 7, 699–706. [Google Scholar] [CrossRef]
- Boison, D.; Singer, P.; Shen, H.; Feldon, J.; Yee, B.K. Adenosine hypothesis of schizophrenia – Opportunities for pharmacotherapy. Neuropharmacology 2012, 62, 1527–1543. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, S.; Gupta, M. Adenosine and its receptors as therapeutic targets: An overview. Saudi Pharm. J. 2013, 21, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Kazemzadeh-Narbat, M.; Annabi, N.; Tamayol, A.; Oklu, R.; Ghanem, A.; Khademhosseini, A. Adenosine-associated delivery systems. J. Drug Target 2015, 23, 580–596. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Toti, K.S.; Chakraborty, S.; Kumar, T.S.; Cronin, C.; Liang, B.T.; Jacobson, K.A. Prevention and rescue of cardiac dysfunction by methanocarba adenosine monophosphonate derivatives. Purinergic Signal. 2020. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Rosas, M.G.; Chávez, E.; Velasco-Loyden, G.; Domínguez-López, M.; Martínez-Pérez, L.; De Sánchez, V.G. Diminished S-adenosylmethionine biosynthesis and its metabolism in a model of hepatocellular carcinoma is recuperated by an adenosine derivative. Cancer Biol. Ther. 2020, 21, 81–94. [Google Scholar] [CrossRef]
- Lian, B.; Ren, Y.; Zhang, H.; Lin, T.; Wang, Y. An adenosine derivative (IFC-305) reduced the risk of radiation-induced intestinal toxicity in the treatment of colon cancer by suppressing the methylation of PPAR-r promoter. Biomed. Pharmacother. 2019, 118, 109202. [Google Scholar] [CrossRef]
- Zuchi, C.; Ambrosio, G.; Lüscher, T.F.; Landmesser, U. Nutraceuticals in cardiovascular prevention: Lessons from studies on endothelial function. Cardiovasc. Ther. 2010, 28, 187–201. [Google Scholar] [CrossRef]
- Popovic, P.J.; Zeh, H.J.; Ochoa, J.B. Arginine and immunity. J. Nutr. 2007, 137, 1681S–1686S. [Google Scholar] [CrossRef]
- López-Jaramillo, P.; Arenas, W.D.; García, R.G.; Rincon, M.Y.; López, M. The role of the L-arginine-nitric oxide pathway in preeclampsia. Ther. Adv. Cardiovasc. Dis. 2008, 2, 261–275. [Google Scholar] [CrossRef]
- Menge, B.A.; Schrader, H.; Ritter, P.R.; Ellrichmann, M.; Uhl, W.; Schmidt, W.E.; Meier, J.J. Selective amino acid deficiency in patients with impaired glucose tolerance and type 2 diabetes. Regul. Pept. 2010, 160, 75–80. [Google Scholar] [CrossRef]
- Pahlavani, N.; Jafari, M.; Sadeghi, O.; Rezaei, M.; Rasad, H.; Rahdar, H.A.; Entezari, M.H. L-arginine supplementation and risk factors of cardiovascular diseases in healthy men: A double-blind randomized clinical trial. F1000Res 2014, 3, 306. [Google Scholar] [CrossRef] [PubMed]
- Kurpad, A.V.; Young, V.R. What is apparent is not always real: Lessons from lysine requirement studies in adult humans. J. Nutr. 2003, 133, 1227–1230. [Google Scholar] [CrossRef] [PubMed]
- Pena, I.A.; Marques, L.A.; Laranjeira, A.B.; Yunes, J.A.; Eberlin, M.N.; MacKenzie, A.; Arruda, P. Mouse lysine catabolism to aminoadipate occurs primarily through the saccharopine pathway; implications for pyridoxine dependent epilepsy (PDE). Biochim. Et Biophys. Acta (Bba)-Mol. Basis Dis. 2017, 1863, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Hayamizu, K.; Oshima, I.; Fukuda, Z.; Kuraamochi, Y.; Nagai, Y.; Izumo, N.; Nakano, M. Safety assessment of L-lysine oral intake: A systematic review. Amino Acids 2019, 51, 647–659. [Google Scholar] [CrossRef]

| Groups | Time (Days) | |||
|---|---|---|---|---|
| −10 | 0 | +7 | +18 | |
| P | 8.78 ± 0.9 | 8.15 ± 0.9 | 9.02 ± 1.0 | 8.25 ± 0.8 |
| NP | 7.48 ± 0.8 | 6.88 ± 0.9 | 7.43 ± 0.8 | 7.49 ± 0.8 |
| Metabolites | Sampling Times | |||
|---|---|---|---|---|
| D −10 | D0 | D7 | D18 | |
| Acetyl carnitine (3-Acetoxy-4-(trimethylammonio)butanoate) | ↓ | ↓ | ↓ | ↑ |
| Arginine-succinic acid hydrate | ↑ | ↑ | ↔ | ↔ |
| 5′-O-{[3-({4-[(3 Aminopropyl)amino]butyl}amino)propyl]carbamoyl}-2′-deoxyadenosine | ↔ | ↓ | ↓ | ↔ |
| N-(1-Hydroxy-2-hexadecanyl) pentadecanamide | ↑ | ↑ | ↔ | ↔ |
| N-[2,3-Bis(dodecyloxy)propyl]-L-lysinamide | ↔ | ↔ | ↔ | ↓ |
| Group | N-acetyl Carnitine (g/mL) | |||
|---|---|---|---|---|
| Sampling Times | ||||
| D −10 | D0 | D7 | D18 | |
| P | N.D. | N.D. | N.D. | 0.3 ± 0.5 |
| NP | 2.1 ± 1.1 | 3.3 ± 3.2 | 0.8 ± 0.5 | 0.2 ± 0.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Nicola, D.; Vinale, F.; Salzano, A.; d’Errico, G.; Vassetti, A.; D’Onofrio, N.; Balestrieri, M.L.; Neglia, G. Milk Metabolomics Reveals Potential Biomarkers for Early Prediction of Pregnancy in Buffaloes Having Undergone Artificial Insemination. Animals 2020, 10, 758. https://doi.org/10.3390/ani10050758
de Nicola D, Vinale F, Salzano A, d’Errico G, Vassetti A, D’Onofrio N, Balestrieri ML, Neglia G. Milk Metabolomics Reveals Potential Biomarkers for Early Prediction of Pregnancy in Buffaloes Having Undergone Artificial Insemination. Animals. 2020; 10(5):758. https://doi.org/10.3390/ani10050758
Chicago/Turabian Stylede Nicola, Donato, Francesco Vinale, Angela Salzano, Giada d’Errico, Anastasia Vassetti, Nunzia D’Onofrio, Maria Luisa Balestrieri, and Gianluca Neglia. 2020. "Milk Metabolomics Reveals Potential Biomarkers for Early Prediction of Pregnancy in Buffaloes Having Undergone Artificial Insemination" Animals 10, no. 5: 758. https://doi.org/10.3390/ani10050758
APA Stylede Nicola, D., Vinale, F., Salzano, A., d’Errico, G., Vassetti, A., D’Onofrio, N., Balestrieri, M. L., & Neglia, G. (2020). Milk Metabolomics Reveals Potential Biomarkers for Early Prediction of Pregnancy in Buffaloes Having Undergone Artificial Insemination. Animals, 10(5), 758. https://doi.org/10.3390/ani10050758








