Seaweed and Seaweed Bioactives for Mitigation of Enteric Methane: Challenges and Opportunities
Abstract
Simple Summary
Abstract
1. Introduction
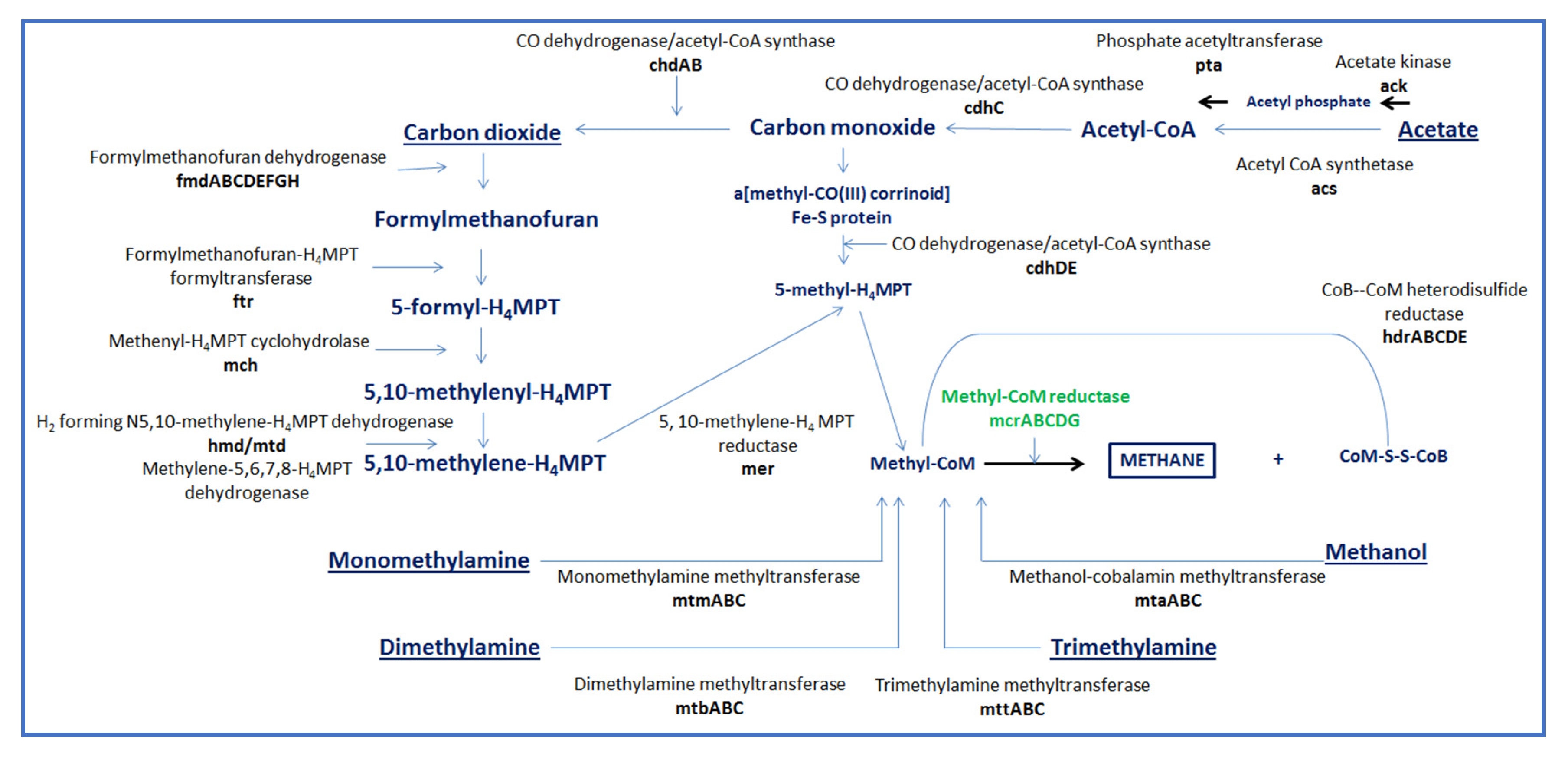
2. Influence of the Rumen Microbiome on CH4 Emissions
3. Overview of In Vitro Studies Which Used Seaweeds for Feed and Impact on CH4 Emissions
3.1. Brown Seaweeds
3.2. Green Seaweeds
3.3. Red Seaweeds
3.4. Seaweeds and Known Impacts on the Rumen Microbiome
4. Seaweed Bioactives and CH4 Emissions Reductions
4.1. Biogenic Halocarbons and Bromoform
4.2. Peptides and Bacteriocins
4.3. Phlorotannins
4.4. Lipids
4.5. Carbohydrates
4.6. Alkaloids and Saponins
5. Effect of Seaweeds/Seaweed-Derived Bioactives on the Microbiome of Ruminant Livestock
6. Potential for Scale-Up Trials and Economic Feasibility
Cultivation of Seaweed
7. Gaps in Current Knowledge
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gerber, P.J.H.; Steinfeld, B.; Henderson, A.; Mottet, C.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock: A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013. [Google Scholar]
- FAO (Food and Agriculture Organization of the United Nations). World Livestock 2011: Livestock in Food Security; FAO: Rome, Italy, 2011. [Google Scholar]
- IPCC. 2007: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K.B., Tignor, M., Miller, H.L., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- McAllister, T.A.; Beauchemin, K.A.; McGinn, S.M.; Hao, X. Greenhouse gases in animal agriculture—Finding a balance between Food Production and emissions. Anim. Feed Sci. Technol. 2011, 166–167, 1–6. [Google Scholar] [CrossRef]
- Carberry, C.A.; Kenny, D.A.; Han, S.; McCabe, M.S.; Waters, S.A. Effect of phenotypic residual feed intake and dietary forage content on the rumen microbial community of beef cattle. Appl. Environ. Microbiol. 2012, 78, 4949–4958. [Google Scholar] [CrossRef] [PubMed]
- Huws, S.A.; Creevey, C.J.; Oyama, L.B.; Mizrahi, I.; Denman, S.E.; Popova, M.; Muñoz-Tamayo, R.; Forano, E.; Waters, S.M.; Hess, M.; et al. Addressing global ruminant agricultural challenges through understanding the rumen microbiome: Past, Present and Future. Front. Microbiol. 2018, 9, 2161. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Johnson, D.E. Methane emissions from cattle. J. Anim. Sci. 1995, 73, 2483–2492. [Google Scholar] [CrossRef]
- van Gastelen, S.; Dijkstra, J.; Bannink, A. Are dietary strategies to mitigate enteric methane emissions equally effective across dairy cattle, beef cattle, and sheep? J. Dairy Sci. 2019, 102, 6109–6130. [Google Scholar] [CrossRef]
- Ramin, M.; Franco, M.; Roleda, M.; Aasen, I.M.; Hetta, M.; Steinshamn, H. In vitro evaluation of utilisable crude protein and methane production for a diet in which grass silage was replaced by different levels and fractions of extracted seaweed proteins. Anim. Feed Sci. Technol. 2018, 255, 114225. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Z.; Bach, S.J.; McAllister, T.A. Effects of phlorotannins from Ascophyllum nodosum (brown seaweed) on in vitro ruminal digestion of mixed forage or barley grain. Anim. Feed Sci. Technol. 2008, 145, 375–395. [Google Scholar] [CrossRef]
- Kinley, R.D.; de Nys, R.; Vucko, M.J.; Machado, L.; Tomkins, N.W. The red macroalgae Asparagopsis taxiformis is a potent natural anti-methanogenic that reduces methane production during in vitro fermentation with rumen fluid. Anim. Prod. 2016, 56, 282–289. [Google Scholar] [CrossRef]
- Machado, L.; Kinley, R.D.; Magnusson, M.; de Nys, R.; Tomkins, N.W. The potential of macroalgae for beef production systems in Northern Australia. J. Appl. Phycol. 2015, 27, 2001–2005. [Google Scholar] [CrossRef]
- Bang, C.; Schilhabel, A.; Weidenbach, K.; Kopp, A.; Goldmann, T.; Gutsmann, T.; Schmitz, R.A. Effect of Antimicrobial peptides on methanogenic Archaea. Antimicrob. Agents Chemother. 2012, 56, 4123–4130. [Google Scholar] [CrossRef]
- Roque, B.M.; Brooke, C.G.; Ladau, J.; Polley, T.; Marsh, L.J.; Najafi, N.; Pandey, P.; Singhi, H.; Kinley, R.; Salwen, J.R.; et al. Effect of the macroalgae Asparagopsis taxiformis on methane production and rumen microbiome assemblage. Anim. Microbiome 2019, 1, 3. [Google Scholar] [CrossRef]
- Kinley, R.D.; Martinez-Fernandez, G.; Matthews, M.K.; de Nys, R.; Magnusson, M.; Tomkins, N.W. Mitigating the carbon footprint and improving productivity of ruminant livestock agriculture using a red seaweed. J. Clean. Prod. 2020, 259, 120836–120842. [Google Scholar] [CrossRef]
- Henderson, G.; Cox, F.; Ganesh, S.; Jonker, A.; Young, W.; Global Rumen Census collaboration; Janssen, P.H. Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Rep. 2015, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Sasson, G.; Kruger Ben-Shabat, S.; Seroussi, E.; Doron-Faigenboim, A.; Shterzer, N.; Yaacoby, S.; Berg Miller, M.E.; White, B.A.; Halperin, E.; Mizrahi, I. Heritable bovine rumen bacteria are phylogenetically related and correlated with the cow’s capacity to harvest energy from its feed. mBio 2017, 8, e00703-17. [Google Scholar] [CrossRef] [PubMed]
- Tapio, I.; Snelling, T.J.; Strozzi, F.; Wallace, R.J. The ruminal microbiome associated with methane emissions from ruminant livestock. J. Anim. Sci. Biotechnol. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report; Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; pp. 151–172. [Google Scholar]
- Patra, A.; Park, T.; Kim, M.; Yu, Z. Rumen methanogens and mitigation of methane emission by anti-methanogenic compounds and substances. J. Anim. Sci. Biotechnol. 2017, 8, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, R.; Dicksved, J.; Sun, L.; Gonda, H.; Muller, B.; Schnurer, A.; Bertilsson, J. Methane production in dairy cows correlates with rumen methanogenic and bacterial community structure. Front. Microbiol. 2017, 8, 266. [Google Scholar] [CrossRef]
- Kittelmann, S.; Pinares-Patino, C.S.; Seedorf, H.; Kirk, M.R.; Ganesh, S. McEwan, J.C.; Janssen, P.H. Two different bacterial community types are linked with the low methane emission trait in sheep. PLoS ONE 2014, 9, e103717. [Google Scholar] [CrossRef]
- Shi, W.; Moon, C.D.; Leahy, S.C.; Kang, D.; Foula, J.; Kittelmann, S.; Fan, C.; Deutsch, S.; Gagic, D.; Seedorf, H.; et al. Methane yield phenotypes linked to differential gene expression in the sheep rumen microbiome. Genome Res. 2014, 24, 1517–1525. [Google Scholar] [CrossRef]
- Leahy, S.C.; Kelly, W.J.; Altermann, E.; Ronimns, R.S.; Yeoman, C.J.; Pacheco, D.M.; Li, D.; Kong, Z.; McTowish, S.; Sang, C.; et al. The genome sequence of the rumen methanogen Methanobrevibacter ruminantium reveals new possibilities for controlling ruminant methane emissions. PLoS ONE 2010, 5, e8926. [Google Scholar] [CrossRef]
- Reeve, J.N.; Nolling, J.; Morgan, R.M.; Smith, D.R. Methanogenesis: Genes, genomes, and who’s on first? J. Bacteriol. 1997, 179, 5945–5986. [Google Scholar] [CrossRef]
- Kamke, J.; Kittelmann, S.; Soni, P. Rumen metagenome and metatranscriptome analyses of low methane yield sheep reveals a Sharpea-enriched microbiome characterised by lactic acid formation and utilisation. Microbiome 2016, 4, 56. [Google Scholar] [CrossRef]
- Janssen, P.H. Influence of hydrogen on rumen methane formulation and fermentation balances through microbial growth kinetics and fermentation thermodynamics. Anim. Feed Sci. Technol. 2010, 160, 1–22. [Google Scholar] [CrossRef]
- Wolin, M.J.; Miller, T.L.; Stewart, C.S. Microbe-microbe interactions. In The Rumen Microbial Ecosystem; Springer: Dordrecht, The Netherlands, 1997; pp. 467–491. [Google Scholar]
- Wallace, R.J.; Rooke, J.A.; McKain, N.; Duthie, C.-A.; Hyslop, J.J.; Ross, D.W.; Waterhouse, A.; Watson, M.; Roehe, R. The rumen microbial metagenome associated with high methane production in cattle. BMC Genom. 2015, 16, 839. [Google Scholar] [CrossRef]
- Edwards, J.E.; Forster, R.J.; Callaghan, T.M.; Dollhofer, V.; Dagar, S.S.; Cheng, Y.; Chang, J.; Kittelmann, S.; Fliegerova, K.; Puniya, A.K.; et al. PCR and Omics Based Techniques to Study the Diversity, Ecology and Biology of Anaerobic Fungi: Insights, Challenges and Opportunities. Front. Microbiol. 2017, 8, 1657. [Google Scholar] [CrossRef]
- Cunha, C.S.; Veloso, C.M.; Marcondes, M.I.; Mantouvani, H.C.; Tomich, T.R.; Pereira, L.G.R.; Ferreira, M.F.L.; Dill-McFarland, K.A.; Suen, G. Assessing the impact of rumen microbial communities on methane emissions and production traits in Holstein cows in a tropical climate. Syst. Appl. Microbiol. 2017, 40, 492–499. [Google Scholar] [CrossRef]
- Aydin, S.; Yildirim, E.; Ince, O.; Ince, B. Rumen anaerobic Fungi create new opportunities for enhanced methane production from microalgae biomass. Algal Res. 2017, 23, 150–160. [Google Scholar] [CrossRef]
- Newbold, C.J.; de la Fuente, G.; Belanche, A.; Ramos-Morales, E.; McEwan, N.R. The role of ciliate protozoa in the rumen. Front. Microbiol. 2015, 6, 1313. [Google Scholar] [CrossRef]
- Weimer, P.J. Redundancy, resilience, and host specificity of the ruminal microbiota: Implications for engineering improved ruminal fermentations. Front. Microbiol. 2015, 6, 296. [Google Scholar] [CrossRef]
- Difford, G.F.; Plichta, D.R.; Løvendahl, P.; Lassen, J.; Noel, S.J.; Højberg, O.; Wright, A.-D.G.; Zhu, Z.; Kristensen, L.; Nielsen, H.B.; et al. Host genetics and the rumen microbiome jointly associate with methane emissions in dairy cows. PLoS Genet. 2018, 14, e1007580. [Google Scholar] [CrossRef]
- Roehe, R.; Dewhurst, R.J.; Duthie, C.-A.; Rooke, J.A.; McKain, N.; Ross, D.W.; Hyslop, J.J.; Waterhouse, A.; Freeman, T.C.; Watson, M.; et al. Bovine Host Genetic Variation Influences Rumen Microbial Methane Production with Best Selection Criterion for Low Methane Emitting and Efficiently Feed Converting Hosts Based on Metagenomic Gene Abundance. PLoS Genet. 2016, 12, e1005846. [Google Scholar] [CrossRef]
- Zhang, Q.; Difford, G.; Sahana, G.; Løvendahl, P.; Lassen, J.; Lund, M.S.; Guldbrandtsen, B.; Janss, L. Bayesian modeling reveals host genetics associated with rumen microbiota jointly influence methane emission in dairy cows. ISME J. 2020, 14, 2019–2033. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; McGinn, S.M.; Petit, H.V. Methane abatement strategies for cattle: Lipid supplementation of diets. Can. J. Anim. Sci. 2007, 87, 431–440. [Google Scholar] [CrossRef]
- Patra, A.K. The effect of dietary fat on methane emissions, and its other effects on digestibility, rumen fermentation and lactation performance in cattle: A meta-analysis. Livest. Sci. 2013, 155, 244–254. [Google Scholar] [CrossRef]
- Jayanegara, A.; Goel, G.; Makkar, H.P.S.; Becker, K. Divergence between purified hydrolysable and condensed tannin effect on methane emission, rumen fermentation and microbial populations in vitro. Anim. Feed Sci. Technol. 2015, 209, 60–68. [Google Scholar] [CrossRef]
- Cobellis, G.; Petrozzi, A.; Forte, C.; Acuti, G.; Orrui, M.; Marcotullio, M.C.; Aquino, a.; Nicolini, A.; Mazza, U. and Trabalza-Marinucci, M. Evaluation of the effects of methane and ammonia production by using Origanum vulgare L. and Rosmarinus officinalis L. essential oils on in vitro rumen fermentation systems. Sustainability 2015, 7, 12856–12869. [Google Scholar] [CrossRef]
- Bay-Larsen, I.; Risvoll, C.; Bjorkhang, H. Local protein sources in animal feed—Perceptions among arctic sheep farmers. J. Rural Stud. 2018, 59, 98–110. [Google Scholar] [CrossRef]
- Molina-Alcaide, E.; Carro, M.D.; Roleda, M.Y.; Weisbjerg, M.R.; Lind, V.; Novoa-Garrido, M. In vitro ruminal fermentation and methane production of different seaweed species. Anim. Feed Sci. Technol. 2017, 228, 1–12. [Google Scholar] [CrossRef]
- Lind, V.; Weisbjerg, M.R.; Jørgensen, G.M.; Fernandez-Yepes, J.E.; Arbesú, L.; Molina-Alcaide, E. Ruminal Fermentation, Growth Rate and Methane Production in Sheep Fed Diets Including White Clover, Soybean Meal or Porphyra sp. Animals 2020, 10, 79. [Google Scholar] [CrossRef]
- Chagas, J.C.; Ramin, M.; Krizsan, S.J. In vitro evaluation of different dietary methane mitigation strategies. Animals 2019, 9, 1120. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; Nys, R.; Tomkins, N. Identification of bioactives from the red seaweed Asparagopsis taxiformis that promote antimethanogenic activity in vitro. J. Appl. Phycol. 2016, 28, 3117–3126. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; de Nys, R.; Tomkins, N. Dose-response effects of Asparagopsis taxiformis and Oedogonium sp. on in vitro fermentation and methane production. J. Appl. Phycol. 2016, 28, 1443–1452. [Google Scholar] [CrossRef]
- Brooke, C.G.; Roque, B.M.; Najafi, N.; Hess, M. Methane reduction potential of two pacific coast macroalgae during in vitro ruminant fermentation. Front. Mar. Sci. 2020, 7, 7. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; de Nys, R.; Tomkins, N. Effects of Marine and Freshwater Macroalgae on In-Vitro Total Gas and Methane Production. PLoS ONE 2014, 9, e85289. [Google Scholar] [CrossRef]
- Dubois, B.; Tomkins, N.W.; Kinley, R.D.; Bai, M.; Scott, S.; Paul, N.A.; de Nys, R. Effect of topical algae as additives on rumen in vitro gas production and fermentation characteristics. Am. J. Plant Sci. 2013, 4, 34–43. [Google Scholar] [CrossRef]
- Maia, M.R.G.; Fonseca, A.J.M.; Oliveria, H.M.; Medonca, C.; Cabrita, A.R.J. The potential role of seaweeds in the natural manipulation of rumen fermentation and methane production. Sci. Rep. 2016, 6, 32321. [Google Scholar] [CrossRef]
- Brooke, C.G.; Roque, B.M.; Najafi, N.; Gonzalez, M.; Pfefferien, A.; DeAnde, V.; Ginsberg, D.W.; Harden, M.C.; Nuzhdin, S.V.; Salwen, J.K.; et al. Evaluation of the potential of two common Pacific coast macroalgae for mitigating methane emissions from ruminants. bioRxiv 2018, 16, 434480. [Google Scholar] [CrossRef]
- Machado, L.; Tomkins, N.; Magnusson, M.; Midgley, D.J.; De Nys, R.; Rosewarne, C.P. In Vitro Response of Rumen Microbiota to the Antimethanogenic Red Macroalga Asparagopsis taxiformis. Microb. Ecol. 2018, 75, 811–818. [Google Scholar] [CrossRef]
- Kinley, R.D.; Fredeen, A.H. In vitro evaluation of feeding North Atlantic stormtoss seaweeds on ruminal digestion. J. Appl. Phycol. 2015, 27, 2387–2393. [Google Scholar] [CrossRef]
- Prayitno, C.H.; Utami, F.K.; Nugroho, A.; Widyastuti, T. The effect of seaweed (Gracilaria sp.) supplementation in sheep feed on methanogenesis inhibition in vitro. In Proceedings of the IOP Conference Series Earth and Environmental Science, Makassar, Indonesia, 6–7 November 2018; Volume 247, p. 012069. [Google Scholar]
- Gemeda, B.S.; Hassen, A. Effect of tannin and species variation on in vitro digestibility, gas, and methane production of tropical browse plants. Asian-Australas. J. Anim. Sci. 2015, 28, 188–199. [Google Scholar] [CrossRef]
- Burtin, P. Nutritional value of seaweed. Electron. J. Environ. Agric. Food Chem. 2003, 2, 498–503. [Google Scholar]
- Belanche, A.; Jones, E.; Parveen, I.; Newbold, C.J. A metagenomic approach to evaluate the impact of dietary supplementation with Ascophyllum nodosum or Laminaria digitata on rumen function in Rusitec fermenters. Front. Microbiol. 2016, 7, 299. [Google Scholar] [CrossRef] [PubMed]
- Hymes-Fecht, U.C.; Broderick, G.A.; Muck, R.E.; Grabber, J.H. Replacing alfalfa or red clover silage with birdsfoot trefoil silage in total mixed rations increases production of lactating dairy cows. J. Dairy Sci. 2013, 96, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Cabrita, A.R.J.; Maia, M.R.G.; Oliveira, H.M.; Pinto, I.S.; Almeida, A.; Pinto, E.; Fonseca, A.J.M. Tracing seaweeds as mineral sources for farm-animals. J. Appl. Phycol. 2016, 28, 3135–3150. [Google Scholar] [CrossRef]
- Gulzari, S.O.; Lind, V.; Aasen, I.M.; Steinshamn, H. Effect of supplementing sheep diets with macroalgae species on in vivo nutrient digestibility, rumen fermentation and blood amino acid profile. Animal 2019, 13, 2792–2801. [Google Scholar] [CrossRef]
- Hansen, H.R.; Hector, B.L.; Feldmann, J. A qualitative and quantitative evaluation of the seaweed diet of North Ronaldsay sheep. Anim. Feed Sci. Technol. 2003, 105, 21–28. [Google Scholar] [CrossRef]
- Biancarosa, I.; Espe, M.; Bruckner, C.G.; Heesch, S.; Liland, N.; Waagbo, R.; Torstensen, B.; Lock, E.J. Amino acid composition, protein content, and nitrogen to protein conversion factors of 21 seaweed species. J. Appl. Phycol. 2017, 29, 1001–1009. [Google Scholar] [CrossRef]
- Burt, A.W.A.; Bartlett, S.; Rowland, S.J. The use of seaweed meals in concentrate mixtures for dairy cows. J. Dairy Res. 1954, 21, 299–304. [Google Scholar] [CrossRef]
- Tayyab, U.; Novoa-Garrido, M.; Roleda, M.Y.; Lind, V.; Weisbjerg, M.R. Ruminal and intestinal protein degradability of various seaweed species measured in situ in dairy cows. Anim. Feed Sci. Technol. 2016, 213, 44–54. [Google Scholar] [CrossRef]
- Haq, S.H.; Al-Ruwaished, G.; Al-Mutlaq, M.A.; Naki, S.A.; Al-Mogren, M.; Al-Rashed, S.; Ain, Q.T.; Al-Amro, A.A.; Al-Mussallam, A. Antioxidant, anticancer activity and phytochemical analysis of green algae, Chaetomorpha collected from the Arabian Gulf. Sci. Rep. 2019, 9, 18906. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, C.; Aluko, R.E.; Hossain, M.; Rai, D.K.; Hayes, M. Potential of a renin inhibitory peptide from the red seaweed Palmaria palmata as a functional food ingredient following confirmation and characterization of a hypotensive effect in spontaneously hypertensive rats. J. Agric. Food Chem. 2014, 62, 8352–8356. [Google Scholar] [CrossRef] [PubMed]
- Sharifuddin, Y.; Chin, Y.-X.; Lim, P.-E.; Phang, S.-M. Potential bioactive compounds from seaweeds for diabetes management. Mar. Drugs 2015, 13, 5447–5491. [Google Scholar] [CrossRef]
- Young, E.G.; Smith, D.G. Amino acids, peptides and proteins of Irish Moss, Chondrus crispus. J. Biol. Chem. 1958, 233, 406–410. [Google Scholar]
- Orpin, C.G.; Greenwood, Y.; Hall, F.J.; Paterson, I.W. The rumen microbiology of seaweed digestion in Orkney sheep. J. Appl. Bacteriol. 1985, 58, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Hehemann, J.H.; Correc, G.; Barbeyron, T.; Helbert, W.; Czjzek, M.; Michel, G. Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature 2010, 464, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Bach, S.J.; Wang, Y.; McAllister, T.A. Effect of feeding sun-dried seaweed (Ascophyllum nodosum) on fecal shedding of Escherichia coli O157:H7 by feedlot cattle and on growth performance of lambs. Anim. Feed Sci. Technol. 2008, 142, 17–32. [Google Scholar] [CrossRef]
- Wang, Y.; Alexander, T.W.; McAllister, T.A. In vitro effects of phlorotannins from Ascophyllum nodosum on rumen bacterial populations and fermentation. J. Sci. Food Agric. 2009, 89, 2252–2260. [Google Scholar]
- Lee, S.; Jeong, J.S.; Shin, W.H.; Lee, S.K.; Kim, H.S.; Eom, J.S.; Lee, S.S. Impact of Ecklonia stolonifera extract on in vitro ruminal fermentation characteristics, methanogenesis and microbial populations. Asian-Australas. J. Anim. Sci. 2019, 32, 1864–1872. [Google Scholar] [CrossRef]
- Zhou, M.; Hünerberg, M.; Chen, Y.; Reuter, T.; McAllister, T.A.; Evans, F.; Critchley, A.T.; Guan, L.L. Air-dried brown seaweed, Ascophyllum nodosum, alters the rumen microbiome in a manner that changes rumen fermentation profiles and lowers the prevalence of foodborne pathogens. mSphere 2018, 3, e00017-18. [Google Scholar] [CrossRef]
- Newbold, C.J.; Lassalas, B.; Jouany, J.P. The importance of methanogens associated with ciliate protozoa in ruminal methane production in vitro. Lett. Appl. Microbiol. 1995, 21, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Newbold, C.J. Assessing antiprotozoal agents. In In Vitro Screening of Plant Resources for Extra-Nutritional Attributes in Ruminants; Vercoe, P.E., Makkar, H.P.S., Schilink, C., Eds.; Springer: Dordrecht, The Netherland, 2010; pp. 47–53. [Google Scholar]
- Makkar, H.P.S.; Blummel, M.; Becker, K. In vitro effects of and interactions between tannins and saponins and fate of tannins in the rumen. J. Sci. Food Agric. 1995, 69, 481–495. [Google Scholar] [CrossRef]
- Khelaifia, S.; Drancourt, M. Susceptibility of Archaea to antimicrobial agents: Applications to clinical microbiology. Clin. Microbiol. Infect. 2012, 18, 841–848. [Google Scholar] [CrossRef]
- Varnava, K.G.; Ronimus, R.S.; Sarojini, V. A review on comparative mechanistic studies of antimicrobial peptides against Archaea. Biotechnol. Bioeng. 2017, 114, 2457–2473. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, L.J.; Liss, P.S. On temperate sources of bromoform and other reactive organic bromine gases. J. Geophys. Res. 2000, 105, 20539–20547. [Google Scholar] [CrossRef]
- Paul, N.A.; de Nys, R.; Steinberg, P.D. Chemical Defence against Bacteria in the Red Alga Asparagopsis armata: Linking Structure with Function. Mar. Ecol. Prog. Ser. 2006, 306, 87–101. [Google Scholar] [CrossRef]
- Maschek, J.A.; Baker, B.J. The chemistry of algal secondary metabolism. In Algal Chemical Ecology; Amsler, C.D., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 1–24. [Google Scholar]
- Leedham, E.C.; Hughes, C.; Keng, F.S.L.; Phang, S.-M.; Malin, G.; Sturges, W.T. Emission of atmospherically significant halocarbons by naturally occurring and farmed tropical macroalgae. BiogeoSciences 2013, 10, 3615–3633. [Google Scholar] [CrossRef]
- Tomkins, N.; Colegate, S.; Hunter, R. A bromochloromethane formulation reduces enteric methanogenesis in cattle fed grain-based diets. Anim. Prod. Sci. 2009, 49, 1053–1058. [Google Scholar] [CrossRef]
- Kasanah, N.; Seto, T.D.S.; Ameila, W.; Isnansetyo, A. Antibacterial compounds from red seaweeds (Rhodophyta). Indones. J. Chem. 2015, 15, 201–209. [Google Scholar] [CrossRef]
- Genovese, G.; Tedone, L.; Hamann, M.T.; Morabito, M. The Mediterranean Red Alga Asparagopsis: A source of compounds against Leishmania. Mar. Drugs 2009, 7, 361–399. [Google Scholar] [CrossRef]
- Mata, L.; Gasper, H.; Santos, R. Carbon/Nutrient balance in relation to biomass production and halogenated compound content in the red alga Asparagopsis taxiformis (Bonnemaisoniaceae). J. Phycol. 2012, 48, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, B.; Vadalá, M.; Laurino, C. Clinical effect of overwintered, stressed Chondrus crispus and non-overwintered-stressed Chondrus crispus on dietary supplementation. Asian J. Med. Sci. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Ismail, A.; Ktari, L.; Ahmed, M.; Bolhuis, H.; Boudabbous, A.; Stal, L.J.; Cretoiu, M.S.; El Bour, M. Antimicrobial Activities of Bacteria Associated with the Brown Alga Padina pavonica. Front. Microbiol. 2016, 7, 1072. [Google Scholar] [CrossRef] [PubMed]
- Egan, S.; Harder, T.; Burke, C.; Steinberg, P.; Kjelleberg, S.; Thomas, T. The seaweed holobiont: Understanding seaweed–bacteria interactions. FEMS Microbiol. Rev. 2013, 37, 462–476. [Google Scholar] [CrossRef]
- Goecke, F.; Wiese, L.A.; Imhoff, J. Chemical interaction between marine macroalgae and bacteria. Mar. Ecol. Prog. 2010, 409, 267–300. [Google Scholar] [CrossRef]
- Chellaram, C.; Raja, P.; Alex John, A.; Krithika, S. Antagonistic effect of epiphytic bacteria from marine algae, southeastern India. Pak. J. Biol. Sci. 2013, 16, 431–434. [Google Scholar] [CrossRef]
- Horta, A.; Pinteus, S.; Alves, C.; Fino, N.; Silva, J.; Fernandez, S.; Rodrigues, A.; Pedrosa, R. Antioxidant and antimicrobial potential of the Bifurcaria bifurcata epiphytic bacteria. Mar. Drugs 2012, 12, 1676–1689. [Google Scholar] [CrossRef]
- Martin, M.; Portetelle, D.; Michel, G.; Vandenbol, M. Microorganisms living on macroalgae, Diversity, interactions, and biotechnological applications. Appl. Microbiol. Biotechnol. 2014, 98, 2917–2935. [Google Scholar] [CrossRef]
- Sawabe, T.; Makino, H.; Tatsumi, M.; Nakano, K.; Tajima, K.; Iqbal, M.M.; Yumoto, I.; Ezura, Y.; Christen, R. Pseudoalteromonas bacteriolytica sp. nov.; a marine bacterium that is the causative agent of red spot disease of Laminaria japonica. Int. J. Syst. Bacteriol. 1998, 48, 769–774. [Google Scholar] [CrossRef]
- Johansen, J.E.; Nielsen, P.; Sjoholm, C. Description of Cellulophaga baltica gen. nov.; sp. nov. and Cellulophaga fucicola gen. nov.; sp. nov. and reclassification of Cytophaga lytica to Cellulophaga lytica gen. nov.; comb. nov. Int. J. Syst. Evol. Microbiol. 1999, 49, 1231–1240. [Google Scholar] [CrossRef]
- Ivanova, E.P.; Christen, R.; Alexeeva, Y.V.; Zhukova, N.V.; Gorshkova, N.M.; Lysenko, A.M.; Mikhailov, V.V.; Nicolau, D.V. Brevibacterium celere sp. nov.; isolated from degraded thallus of a brown alga. Int. J. Syst. Evol. Microbiol. 2004, 54, 2107–2111. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Jung, H.J.; Lee, H.K. Marine bacteria associated with the Korean brown alga, Undaria pinnatifida. Microbiol. J. 2004, 44, 694–698. [Google Scholar]
- Suresh, M.; Lyappuraj, P.; Anantharaman, P. Optimisation, characterisation and partial purification of bacteriocin produced by Staphylococcus haemolyticus MSM an isolate from seaweed. Biocatal. Agric. Biotechnol. 2014, 3, 161–166. [Google Scholar] [CrossRef]
- Simmons, T.L.; Engene, N.; Ureña, L.D.; Romero, L.I.; Ortega-Barría, E.; Gerwick, L.; Gerwick, W.H. Viridamides A and B, lipodepsipeptides with antiprotozoal activity from the marine cyanobacterium Oscillatoria nigro-viridis. J. Nat. Prod. 2008, 71, 1544–1550. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fairhead, V.A.; Amsler, C.D.; McClintock, J.B.; Baker, B.J. Variation in phlorotannin content with two species of brown macroalgae (Desmarestia anceps and D. menziesii) from the Western Antartic Peninsula. Polar Biol. 2005, 28, 680–699. [Google Scholar] [CrossRef]
- Hay, M.E.; Fenical, W. Galactolipids rather than phlorotannins as herbivore deterrents in the brown seaweed Fucus vesiculosus. Oecologia 2003, 136, 107–114. [Google Scholar]
- Mueller-Harvey, I. Unravelling the conundrum of tannins in animal nutrition and health. J. Sci. Food Agric. 2006, 86, 2010–2037. [Google Scholar]
- Rochfort, S.; Parker, A.J.; Dunshea, F.R. Plant bioactives for ruminant health and productivity. Phytochemistry 2008, 69, 299–322. [Google Scholar] [CrossRef]
- Wang, Y.; Majak, W.; McAllister, T.A. Frothy bloat in ruminants: Causes, occurrence and mitigation strategies. Anim. Feed Sci. Technol. 2012, 172, 103–114. [Google Scholar] [CrossRef]
- Jormalainen, V.; Honkanen, T.; Koivikko, R.; Eranen, J. Induction of phlorotannin production in a brown alga: Defense or resource dynamics? OIKOS 2003, 103, 140–150. [Google Scholar] [CrossRef]
- Ank, G.; da Gama, B.A.P.; Pereira, R.C. Latitudinal variation in phlorotannin contents from Southwestern Atlantic brown seaweeds. PeerJ Life Environ. 2019, 7, 7379–7398. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Silva, L.R.; Pinto, E.; Andrade, P.B.; Bernardo, J.; Mouga, T.; Valentão, P. Can Phlorotannins Purified Extracts Constitute a Novel Pharmacological Alternative for Microbial Infections with Associated Inflammatory Conditions? PLoS ONE 2012, 7, e31145. [Google Scholar] [CrossRef]
- Visser, A.M.; Brok, A.E.; Westphal, A.H.; Hendriks, W.H.; Gruppen, H.; Vincken, J.-P. Resolubilisation of protein from water-insoluble phlorotannin-protein complexes upon classification. J. Agric. Food Chem. 2017, 65, 9595–9602. [Google Scholar] [CrossRef]
- de la Moneda, A.; Carro, M.D.; Weisbjerg, M.R.; Roleda, M.Y.; Lind, V.; Novoa-Garrido, M.; Molina-Alcaide, E. Variability and potential of seaweeds as ingredients of ruminant diets: An in vitro study. Animals 2019, 9, 851. [Google Scholar] [CrossRef] [PubMed]
- Pavia, H.; Toth, G.B. Inducible chemical resistance to herbivory in the brown seaweed Ascophyllum nodosum. Ecology 2000, 81, 3212–3225. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; Kreuzer, M.; O’Mara, F.; McAllister, T.A. Nutritional management for enteric methane abatement: A review. Aust. J. Exp. Agric. 2008, 48, 21–27. [Google Scholar] [CrossRef]
- Bikker, P.; Stokvis, L.; van Krimpen, M.M.; van Wikselaar, P.G.; Cone, K.N. Evaluation of seaweeds from marine waters in Northwestern Europe for application in animal nutrition. Anim. Feed Sci. Technol. 2020, 263, 114460. [Google Scholar] [CrossRef]
- Poulsen, M.; Schwab, C.; Jensen, B.B.; Engberg, R.M.; Spang, A.; Canibe, N.; Højberg, O.; Milinovich, G.; Fragner, L.; Schleper, C.; et al. Methylotrophic methanogenic Thermoplasmata implicated in reduced methane emissions from bovine rumen. Nat. Commun. 2013, 4, 1428. [Google Scholar] [CrossRef] [PubMed]
- Stiger-Pouvreau, V.; Bourgougnon, N.; Deslandes, E. Chapter 8—Carbohydrates From Seaweeds. In Seaweed in Health and Disease Prevention; Fleurence, J., Levine, I., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 223–274. [Google Scholar]
- Becker, S.; Tebben, J.; Coffinet, S.; Wiltshire, K.; Iversen, M.H.; Harder, T.; Hinrichs, K.-U.; Hehemann, J.-H. Laminarin is a major molecule in the marine carbon cycle. Proc. Natl. Acad. Sci. USA 2020, 117, 6599–6607. [Google Scholar] [CrossRef]
- Yu, S.; Blennow, A.; Bojko, M.; Madsen, F.; Olsen, C.E.; Engelsen, S.B. Physico-chemical Characterization of Floridean Starch of Red Algae. Starch-Stärke 2002, 54, 66–74. [Google Scholar] [CrossRef]
- Usov, A.I. Chapter 4—Polysaccharides of the red algae. In Advances in Carbohydrate Chemistry and Biochemistry; Horton, D., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 65, pp. 115–217. [Google Scholar]
- Kidgell, J.T.; Magnusson, M.; de Nys, R.; Glasson, C.R.K. Ulvan: A systematic review of extraction, composition and function. Algal Res. 2019, 39, 101422. [Google Scholar] [CrossRef]
- Morais, T.; Inácio, A.; Coutinho, T.; Ministro, M.; Cotas, J.; Pereira, L.; Bahcvandziev, K. Seaweed potential in the animal feed: A review. J. Mar. Sci. Eng. 2020, 8, 559. [Google Scholar] [CrossRef]
- Wink, M. Medicinal plants: A source of anti-parasitic secondary metabolites. Molecules 2012, 17, 12771–12791. [Google Scholar] [CrossRef] [PubMed]
- Desgagné-Penix, I. Biosynthesis of alkaloids in Amaryllidaceae plants: A review. Phytochem. Rev. 2020, 5, 239–270. [Google Scholar] [CrossRef]
- Guven, K.C.; Percot, A.; Sezik, E. Alkaloids in marine algae. Mar. Drugs 2010, 8, 269–284. [Google Scholar] [CrossRef]
- Liu, D.-Q.; Mao, S.-C.; Zhang, H.-Y.; Yu, X.-Q.; Feng, M.-T.; Wang, B.; Feng, L.-H.; Guo, Y.-W. Racemosins A and B, two novel bisindole alkaloids from the green alga Caulerpa racemosa. Fitoterapia 2013, 91, 15–20. [Google Scholar] [CrossRef]
- Yang, H.; Liu, D.-Q.; Liang, T.-J.; Li, J.; Liu, A.-H.; Yang, P.; Lin, K.; Yu, X.-Q.; Guo, Y.-W.; Mao, S.-C.; et al. Racemosin C, a novel minor bisindole alkaloid with protein tyrosine phosphatase-1B inhibitory activity from the green alga Caulerpa racemosa. J. Asian Nat. Prod. Res. 2014, 16, 1158–1165. [Google Scholar] [CrossRef]
- Wahlström, N.; Stensland, B.; Bergman, J. Synthesis of the marine alkaloid caulersin. Tetrahedron 2004, 60, 2147–2153. [Google Scholar] [CrossRef]
- Chen, Y.F.; Yang, C.H.; Chang, M.S.; Ciou, Y.P.; Huang, Y.C. Foam properties and detergent abilities of the saponins from Camellia oleifera. Int. J. Mol. Sci. 2010, 11, 4417–4425. [Google Scholar] [CrossRef]
- Holtshausen, L.; Chaves, A.V.; Beauchemin, K.A.; McGinn, S.M.; McAllister, T.A.; Odongo, N.F.; Cheeke, P.R.; Benchaar, C. Feeding saponin-containing Yucca schidigera and Quillaja saponaria to decrease enteric methane production in dairy cows. J. Dairy Sci. 2009, 92, 2809–2821. [Google Scholar] [CrossRef]
- Cieslak, A.; Zmora, P.; Stochmal, A.; Pecio, L.; Oleszek, W.; Pers-Kamczyc, E.; Szczechowiak, J.; Nowak, A.; Szumacher-Strabel, M. Rumen anti-methanogenic effect of Saponaria officinalis L. phytochemicals in vitro. J. Agric. Sci. 2014, 152, 981–993. [Google Scholar] [CrossRef]
- Wang, Y.X.; McAllister, T.A.; Newbold, C.J.; Cheeke, P.R.; Cheng, K.J. Effect of Yucca schidigera extract on fermentation and degradation of steroidal saponins in the rumen simulation technique (RUSITEC). Anim. Feed Sci. Technol. 1998, 74, 143–153. [Google Scholar] [CrossRef]
- Patra, A.K.; Saxena, J. Dietary phytochemicals as rumen modifiers: A review of the effects on microbial populations. Antonie van Leeuwenhoek 2009, 96, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Feroz, B. Saponins from marine macroalgae: A review. J. Mar. Sci. Res. Dev. 2018, 8, 4. [Google Scholar] [CrossRef]
- Leelavathi, M.S.; Prasad, M.P. Comparative analysis of phytochemical compounds of marine algae isolated from Gulf of Mannar. World J. Pharm. Pharmalogical Sci. 2015, 4, 640–654. [Google Scholar]
- Venkatesh, R.; Shanthi, S.; Rajapandian, K.; Elamathi, S.; Thenmozhi, S.; Radha, N. Preliminary study on anti-xanthomonas activity, phytochemical analysis and characterization of antimicrobial compounds from Kappaphycus alvarezii. Asian J. Pharm. Clin. Res. 2011, 4, 46–51. [Google Scholar]
- Novoa-Garrido, M.; Rebours, C.; Aaensen, L.; Torp, T.; Lind, V.; Steinshamn, H. Effect of seaweed on gastrointestinal microbiota isolated from Norwegian White sheep. Acta Agric. Scand. Sect. A Anim. Sci. 2017, 66, 152–160. [Google Scholar] [CrossRef]
- Jaspars, M.; Folmer, F. Sea Vegetables for Health; Food and Health innovation Service: Aberdeen, UK, 2013. [Google Scholar]
- Novoa-Garrido, M.; Aanensen, L.; Lind, V.; Larsen, H.J.S.; Jensen, S.K.; Govasmark, E.; Steinshamn, H. Immunological effects of feeding macroalgae and various vitamin E supplements in Norwegian White sheep-ewes and their offspring. Livest. Sci. 2014, 167, 126–136. [Google Scholar] [CrossRef]
- Gaillard, C.; Bhatti, H.S.; Novoa-Garrido, M.; Lind, V.; Roleda, M.Y.; Weisbjerg, M.R. Amino acid profiles of nine seaweed species and their in situ degradability in dairy cows. Anim. Feed Sci. Technol. 2018, 241, 210–222. [Google Scholar] [CrossRef]
- Li, X.; Norman, H.C.; Kinley, R.D.; Laurence, M.; Wilmot, M.; Bender, H.; de Nys, R.; Tomkins, N. Asparagopsis taxiformis decreases enteric methane production from sheep. Anim. Prod. Sci. 2018, 58, 681–688. [Google Scholar] [CrossRef]
- McDonnell, R.P.; O’Doherty, J.V.; Earley, B.; Clarke, A.M.; Kenny, D.A. Effect of supplementation with n-3 polyunsaturated fatty acids and/or β-glucans on performance, feeding behaviour and immune status of Holstein Friesian bull calves during the pre- and post-weaning periods. J. Anim. Sci. Biotechnol. 2019, 10, 1–17. [Google Scholar]
- Baumont, R. Palatability and feeding behaviour in ruminants. A review. Annales de Zootechnie INRA/EDP Sci. 1996, 45, 385–400. [Google Scholar] [CrossRef]
- Harper, M.T.; Oh, J.; Giallongo, F.; Lopes, J.C.; Weeks, H.L.; Faugeron, J.; Hristov, A.N. Short communication: Preference for flavoured concentrated premixes by dairy cows. J. Dairy Sci. 2016, 99, 6585–6589. [Google Scholar] [CrossRef] [PubMed]
- Carlotto, S.B. Performance and behavior of dairy calves fed diets containing milk and citric flavor agents. Ciência e Agrotecnologia 2007, 31, 889–895. [Google Scholar] [CrossRef]
- Mora, L.M.; Oddo, J.M. Flavours in Ruminants. Technical Bulletin No. 27. Norel Animal Nutrition. 1-3. Available online: https://norel.net/en/system/files/TB_27_Use%20of%20flavors%20in%20ruminants_ENG.pdf (accessed on 14 October 2020).
- Robertson, E.; Gordan, I.J.; Pérez-Barbería, J.F. Preference of sheep and goats for straw pellets treated with different food-flavouring agents. Small Rumin. Res. 2006, 63, 50–75. [Google Scholar] [CrossRef]
- Antaya, N.T.; Soder, K.J.; Kraft, J.; Whitehouse, N.L.; Guidon, N.E.; Erickson, P.S.; Conroy, A.B.; Brito, A.F. Incremental amounts of Ascophyllum nodosum meal do not improve animal performance but do increase milk iodine output in early lactation dairy cows fed high-forage diets. J. Dairy Sci. 2015, 98, 1991–2004. [Google Scholar] [CrossRef]
| Seaweed | Seaweed Dose (% of Dry Matter (DM) or OM Incubated) | CH4 Decrease vs. Control | Effects on TGP, TVFA and Digestibility | Reference |
|---|---|---|---|---|
| Alaria esculenta extract (B) | 13, 23, 31 | Linear ↓ with increasing dose | TGP, n.e.; ↓DOM with ↑ dose | [9] |
| Ascophyllum nodosum (B) | 11.1 | ↓15% at 24 h | ↓TGP, ↓TVFA | [10] |
| Asparagopsis taxiformis (R) | 5 | n.e. at 24 h, ↓74% at 48 h | Not measured | [52] |
| 1, 2 | ↓>99% | n.e. on TGP or DOM | [45] | |
| 0.006, 0.013, 0.025, 0.05, and 0.1 | ↓100 for ≥0.05 | ↓TGP (24, 48 h) for ≥0.05 | [45] | |
| 0.5, 1, 2, 5, 10 | ↓100% for ≥1% | ↓TGP for ≥2%; ↓DOM for 10%; ↓VFA for ≥1% | [11] | |
| 2% | ↓100% | ↓TGP; ↓TVFA; DOM-72 h, n.e. | [11] | |
| 16.6 | ↓100% | ↓TGP | [49] | |
| 0.07, 0.125, 0.25, 0.5, 1, 2, 5, 10, and 16.8 | n.e. for ≤0.5%, ↓85% for 1%, ↓100% for ≥2% | ↓TGP for ≥1%; ↓DOM for ≥10%; ↓TVFA for ≥0.5% | [46,47] | |
| 2 | ↓100% | ↓TGP | [53] | |
| Caulerpa taxifolia (G) | 16.6 | ↓33% | TGP, n.e. | [49] |
| Chaetomorpha linum (G) | 16.6 | ↓40% | ↓TGP | [49] |
| Chondrus crispus (Irish moss) (R) | 0.5 | ↓12% | n.e. on TVFA or DOM | [54] |
| Cladophora patentiramea (G) | 16.6 | ↓66% | ↓TGP | [49] |
| Colpomenia sinuosa (B) | 16.6 | ↓49% | ↓TGP | [49] |
| Cystoseira trinodis (B) | 2, 3.8, 7.4, 13.8 | ↓73% for ≥3.8% only | ↓TGP | [50] |
| 16.6 | ↓45% | ↓TGP | [49] | |
| Dictyota bartayresii (B) | 16.6 | ↓92% | ↓TGP | [49] |
| Furcellaria spp. (R) | 0.5 | ↓10% | n.e. on TVFA or DOM | [54] |
| Gigartina spp. (R) | 25 | ↓56% | TGP, n.e. | [51] |
| Gracilaria spp. (R) | 2, 4, 5, 7 | ↓49% for 2%, small ↓ for ≥4% | ↓TGP for 2%; other doses, n.e. | [55] |
| Gracilaria vermiculophylla (R) | 25 | ↓41% | TGP, n.e. | [51] |
| 25 | ↓37% | ↓TGP | [51] | |
| Hormophysa triquetra (B) | 16.6 | ↓44% | TGP, n.e. | [49] |
| Hypnea pannosa (R) | 16.6 | ↓43% | TGP, n.e. | [49] |
| Laurencia filiformis (R) | 16.6 | ↓40% | ↓TGP | [49] |
| Oedogonium spp. (FW) | 16.6 | ↓30% | ↓TGP | [49] |
| 10, 16.7, 25, 50, 75, and 100 | n.e. at ≤25%, ↓17% for 50%, ↓55% for 75%, ↓72.5% for 100% | ↓TGP for ≥10%; ↓DOM and ↓TVFA for ≥16.7% | [47] | |
| Padina australis (B) | 16.6 | ↓51% | ↓TGP | [49] |
| Sargassum flavicans (B) | 16.6 | ↓34% | TGP, n.e. | [49] |
| Ulva ohnoi (G) | 16.6 | ↓45% | ↓TGP | [49] |
| Ulva spp. (G) | 16.6 | ↓50% | ↓TGP | [49] |
| 25 | ↓45% | TGP, n.e. | [51] | |
| Zonaria farlowii (B) | 5 | ↓11% at 24 h only | Not measured | [52] |
| Chemical Composition | Macrominerals (g/kg DM) | Trace Elements (mg/kg DM) | Potentially Toxic Trace Elements (mg/kg DM) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ash (% DM) | CP (% DM) | NDF (% DM) | EE (% DM) | DMD (%) | Ca | P | Mg | Fe | I | Zn | Br | As | Pb | Hg | |
| Saccharina latissimi | 40 * | 12–(17.5 *) | 40 | 60 | 10 | 3 | 5 | 30 | 960–120 | 40 | 550 | 26–70 | 0 | 0.1 | |
| Fucus serratus/spp. | 30 | 6 | 26 | 15–50 | 9–13 | 2 | 7–9 | 40–310 | 300 | 40–50 | 420 | 25–40 | 1 | 0.1 | |
| Laminiaria digitata/spp. | 25–38 | 9–12 | 17–22 | 0.30 | 70–75 | 10–13 | 3 | 6 | 30–179 | 880 | 18–25 | 280 | 50–70 | 0 | 0 |
| Pelvetia canaliculata | 21 | 7 | 2.80 | 9 | 1 | 8 | 200 | 250 | 70 | 520 | 50 | 0 | 0.1 | ||
| Ascophyllum nodosum | 220 | 4.5–6 | 32 | 10–30 | 1–2 | 5–10 | 35–100 | <1000 | 35–100 | <3–22 | |||||
| Seaweed Species | Bromoform (CHBr3) ng g−1 h Fresh Weight |
|---|---|
| Brown | |
| Fucus vesiculosus | 4.9 |
| Fucus serratus | 2.1 |
| Ascophyllum nodosum | 2.7 |
| Laminaria digitata | 49.7 |
| Laminaria saccharina | 32 |
| Macrocystis pyrifera | 125 |
| Red | |
| Meristiella gelidium | 25 |
| Rhodymenia californica | 47 |
| Pterocladia capillacea | 500 |
| Cordllina officinalis | 1.4–20 |
| Gigartina stellata | 4.1–26 |
| Asparagopsis spp. | 43–1256 |
| Chondrus crispus | 0–1.3 |
| Polysiphonia lanosa | 2.1 |
| Green | |
| Ulva intestinalis | 87–192 |
| Ulva linza | 11 |
| Ulva spp. | 150 |
| Ulva spp. (formerly lactuca) | 13.0–150 |
| Cladophoria albida | 0 |
| Brown Seaweed | Phlorotannin Content (mg/Kg) |
|---|---|
| Ascophyllum nodosum | 34.9 |
| Fucus vesiculosus | 42.3 |
| Cladostephus spongiosus | 81.64 |
| Cytoseira nodicaulis | 516.24 |
| Cytoseira tamariscifolia | 815.82 |
| Cytoseira usnevides | 288.2 |
| Fucus spiralis | 968.57 |
| Halopteris filicina | 101.97 |
| Saccorhiza polyschides | 36.68 |
| Sargassum vulgare | 74.96 |
| Seaweed Species | Saponin Content (% of Dry Weight of Alga) |
|---|---|
| Gracilaria crassa | 15 |
| Gracilaria edulis | 17 |
| Cymodoceae rotudata | 13 |
| Cymodoceae serrulata | 14 |
| Ulva lactuca | 14 |
| Ulva reticulate | 16 |
| Gracilaria foliifera | 14 |
| Kappaphycus alvarezii | 14 |
| Galidiella accrosa | 13 |
| Seaweed | Seaweed Dose (% of Dry Matter (DM)) and Animal Trial Used | CH4 Decrease vs. Control | Effects of TGP, TVFA and Digestibility | Reference |
|---|---|---|---|---|
| Asparagopsis taxiformis (R) | <2% OM | Asparagopsis inclusion resulted in a consistent and dose-dependent reduction in enteric CH4 production in sheep over time, with up to 80% CH4 mitigation at the 3% offered rate compared with the group fed no Asparagopsis spp. (p < 0.05). | Sheep fed Asparagopsis had a significantly lower concentration of total volatile fatty acids and acetate, but higher propionate concentration. No changes in live weight gain were identified. Supplementing Asparagopsis in a high-fibre diet (<2% OM) results in significant and persistent decreases in enteric methanogenesis over a 72 d period in sheep. | [141] |
| 0.05–0.20% feed | Asparagopsis spp. was included in the feed of Brahman-Angus cross steers at 0.00%, 0.05%, 0.10%, and 0.20% of feed organic matter. Emissions were monitored in respiration chambers fortnightly over 90 d of treatment. Asparagopsis demonstrated decreased CH4 up to 40% and 98%. | Asparagopsis resulted in weight gain improvements of 53% and 42%, respectively. | [15] | |
| 5% inclusion rate in OM | Feeding cows a 1–2% dry mass supplement of Asparagopsis per day reduced CH4 release by up to 95% without altering fermentation processes. | No negative impact on milk taste. No obvious negative impacts on volatile fatty acid production. | [14] | |
| Ascophyllum nodosum (B) | 2% of diet dry matter (DM) | Limited effect on rumen microbiome. | Feeding 8 Canadian rams with sun-dried seaweed extract (Tasco; Acadian Seaplants Ltd., Dartmouth, NS, Canada) containing a mixture of polysaccharides and oligosaccharides and derived from Ascophyllum nodosum resulted in decreased faecal shedding of E. coli. | [76] |
| Seaweed-derived beta-glucans and marine omega-3 oils (fish oil) | 5 L (120 g/L) per day of milk replacer (MR) and one of the following: (1) 40 g n-3 PUFA per day; (2) 1 g β-glucans per day (GL) and (3) 40 g n-3 PUFA per day and 1 g/d β-glucans in a 2 × 2 factorial design. | Improved immunity. | 44 Holstein Friesian bull calves pre- and post-weaning. | [142] |
| Company | Location | Activities |
|---|---|---|
| Symbrosia | USA | Production techniques for Asparagopsis spp. seaweeds |
| Volta Greentech | Sweden | Production of A.s taxiformis species |
| Taighde Mara Teo | Ireland | Production of A. aramata |
| DúlaBio | Ireland | Production of seaweed “blends” for CH4 reduction |
| BMRS | Ireland | Production of Asparagopsis spp. |
| CH4 Global | Australia | Global supply of Asparagopsis spp. |
| SeaExpert—Consultoria na Área das Pescas, Lda. | Portugal | Sustainable harvest and supply of Asparagopsis spp. |
| Acadian SeaPlants Ltd. | Canada; Ireland and UK | Sustainable harvest and supply of a range of different seaweeds |
| SeaLac Ltd. | Ireland | Supply of sustainable seaweeds |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbott, D.W.; Aasen, I.M.; Beauchemin, K.A.; Grondahl, F.; Gruninger, R.; Hayes, M.; Huws, S.; Kenny, D.A.; Krizsan, S.J.; Kirwan, S.F.; et al. Seaweed and Seaweed Bioactives for Mitigation of Enteric Methane: Challenges and Opportunities. Animals 2020, 10, 2432. https://doi.org/10.3390/ani10122432
Abbott DW, Aasen IM, Beauchemin KA, Grondahl F, Gruninger R, Hayes M, Huws S, Kenny DA, Krizsan SJ, Kirwan SF, et al. Seaweed and Seaweed Bioactives for Mitigation of Enteric Methane: Challenges and Opportunities. Animals. 2020; 10(12):2432. https://doi.org/10.3390/ani10122432
Chicago/Turabian StyleAbbott, D. Wade, Inga Marie Aasen, Karen A. Beauchemin, Fredrik Grondahl, Robert Gruninger, Maria Hayes, Sharon Huws, David A. Kenny, Sophie J. Krizsan, Stuart F. Kirwan, and et al. 2020. "Seaweed and Seaweed Bioactives for Mitigation of Enteric Methane: Challenges and Opportunities" Animals 10, no. 12: 2432. https://doi.org/10.3390/ani10122432
APA StyleAbbott, D. W., Aasen, I. M., Beauchemin, K. A., Grondahl, F., Gruninger, R., Hayes, M., Huws, S., Kenny, D. A., Krizsan, S. J., Kirwan, S. F., Lind, V., Meyer, U., Ramin, M., Theodoridou, K., von Soosten, D., Walsh, P. J., Waters, S., & Xing, X. (2020). Seaweed and Seaweed Bioactives for Mitigation of Enteric Methane: Challenges and Opportunities. Animals, 10(12), 2432. https://doi.org/10.3390/ani10122432






